1. Introduction
The infrared (IR) spectrum of type I collagen has been studied for decades [
1,
2,
3,
4,
5,
6,
7]. The importance of collagen in the health and maintenance of the human body is extensive; the hierarchical structure and radial symmetry of collagen render it a suitable target for study with polarized IR light, for example: type I collagen [
1,
2,
6,
7,
8] and type II collagen [
9]. The dichroic behavior of ordered collagen fibrils was first demonstrated with IR spectro-microscopy nearly 70 years ago [
1]. We have studied collagen in scar tissue [
10,
11] and in mechanically damaged tendon [
7], where we exploited IR polarization imaging to probe the degree of disorder in the damaged tissue.
A limitation to the IR spectroscopic analyses of collagen has been the lack of definition of the spectral profile that constitutes a molecular level of order in the normal control tendon and fibrils. Typically, the IR polarization contrast is defined chiefly by changes in the relative intensity of the Amide I (primarily carbonyl stretch) and Amide II (primarily C-N backbone stretch with some C-N-H angle bend) vibrational bands. In fact, numerous bands in the complicated spectrum of highly ordered collagen type I respond differently under polarized infrared light [
1,
2,
7,
9,
12]. Our previous work was primarily based on far-field Fourier transform IR (FF-FTIR) imaging with focal plane array (FPA) detection, which is subject to the usual Rayleigh criterion resolution limits for λ = ~7 μm wavelength, corresponding to the main protein IR absorbance bands, Amide I and II [
7]. We then began using nanoscale FTIR (nano-FTIR) spectroscopy employing the scattering-type scanning near-field optical microscopy (s-SNOM) method [
13,
14,
15,
16,
17], wherein the diffraction limit is circumvented and IR spectra may be obtained with a spatial resolution of ~20 nm, far below the wavelength of the IR radiation (typically 5–10 μm). Another complementary method is optical photothermal IR (O-PTIR) imaging and spectroscopy [
18,
19,
20], a relatively new far-field technique that opens a new window onto the study of collagen, both as intact tissue and as fibrils, with IR wavelength-independent resolution of better than 500 nm.
In this paper, we seek a better understanding of the absolute variation in spectra of ordered collagen, as this can improve the evaluation of disorder in damaged collagen in the future. To this end, we employed multiple techniques that provide a variety of polarized IR sample illumination and spatial resolutions to investigate the dependence of collagen IR spectra on these parameters. Polarized IR spectra and images of intact, mechanically aligned tendon were obtained with FF-FTIR equipped with focal plane array (FPA) detection and with O-PTIR. To polarize the IR radiation in the FF-FTIR measurements, we inserted a polarizer in the path of the IR beam, after passage through the sample; the lasers employed in O-PTIR are inherently linearly polarized. It was also possible to obtain spectra of control, unstretched fibrils with nano-FTIR spectroscopy, and of similar fibrils with a quantum cascade laser (QCL) oriented with (parallel) and across (perpendicular) the fibril direction by O-PTIR. The nano-FTIR spectra are necessarily polarized with respect to the probe tip axis, and thus yield spectra perpendicular to the fibril direction only. The results from the intact tendon (FF-FTIR and O-PTIR) are contrasted with those from sub-micron fibrils (O-PTIR and nano-FTIR), in an effort to obtain a clearer understanding of the variations observed and their relation to internal structural order.
3. Discussion
Collagen molecules in a positional tendon are aligned in an inherently organized, hierarchical manner, thus tendon is eminently suitable for analysis with polarized IR light [
1]. While polarization studies with other methods are common, few have used polarized IR spectroscopy [
1,
2,
7,
9,
12]. IR wavelength-independent spatial resolution FF O-PTIR and nano-FTIR technologies are rapidly advancing our ability to collect IR spectra at spatial resolutions below the IR diffraction limit. Our motivation in this work has been to achieve a deeper understanding of what the different experimental approaches can reveal for type I collagen, in order that we can better evaluate mechanically damaged tissue and ordered/disordered scar tissue, where the organization is significantly altered.
Early IR polarization studies of collagen quickly addressed the complexity of dichroism in collagen [
2,
3], with recognition that the direction of the change of the molecular dipole moment in the Amide modes would not be precisely parallel to the orientation of the bonds. Some band assignments have been achieved through analysis of synthetic biopolymer mimics [
21] and temperature-dependent studies [
4]. The Amide I mode is generally referred to as a C=O stretch, but vibrational energy and alignment of the dipole moment change depending on the amide orientation. The Amide II is mostly due to C-N stretch with some C-N-H angle bend, but the fundamental mode could well be nearly perpendicular to the direction of the N-H bond. The presence of other amino acid residues, and unknown quantities of water of hydration, lead to further unresolvable spectral contributions even in pure collagen.
The hierarchical organizational structures in tendons are loosely classified by their diameter as fiber (2000–500 nm), fibril (500–100 nm) and sub-fibril (<100 nm) diameter, while the diameter of an individual collagen triple helical molecule is about 1.5 nm. Given the mode of self-assembly, it is not realistic to define these more precisely [
22,
23]. As we showed previously [
7], actual rupture results in a mixture of orientations on a micron spatial scale, producing spectra that are intermediate between the primarily parallel and perpendicular extremes. Some variation in orientation must exist simply because of tissue depth, as those tendons were cryosectioned at 5 and 8 μm thickness. Intact tendons contain additional non-collagenous components that help to maintain the gliding functions, as well as cells, such as fibroblasts and synovial cells [
22]. The absence of a lipid carbonyl band at 1735–1740 cm
−1 and of the symmetric CH
2 stretch band (data not shown) shows that we are not detecting any significant amounts of cellular material. These bands were detected in some spectra of intact tendon, and for this reason were not selected for the data presented here.
The spectral information shown in
Figure 1 has spatial resolution that is IR wavelength dependent. The volume probed with the FF-FTIR FPA microscope and high magnification option is at best ~1.1 μm × 1.1 μm × 5 μm [
24] when applied to bands at short wavelengths, such as 3 μm for NH and CH stretch modes. For the Amide bands considered here, the Rayleigh criterion spatial resolution is ~4 μm, with 1.1 μm pixel size oversampling [
24,
25,
26], and the probed volumes are estimated to be on the order of 3 μm × 3 μm × 5 μm. In comparison with the fibril spectra below, the Amide bands are relatively broad, as can be expected for the heterogeneity present, even though the tendons had been stretched sufficiently to eliminate normal crimping. The spectra show the expected profiles under parallel and perpendicular polarization, wherein the distinct polarization contrast permits assessment of relative tissue organization at the micron length scale.
The application of O-PTIR for analysis of oriented materials was recently demonstrated for a cross-section of a commercial plastic bottle [
19]. The plastic wall was 350 μm thick, thus it was possible to obtain polarized spectra with an estimated pixel resolution of 500 nm across the wall thickness. Linear dichroism was observed in the spectra for polyethylene terephthalate PET backbone methylene wagging modes, in contrast to C=O stretch modes, which were anticipated to be mainly perpendicular to the chain direction. Here, we present the first example of O-PTIR polarized spectroscopy of a biopolymer, collagen.
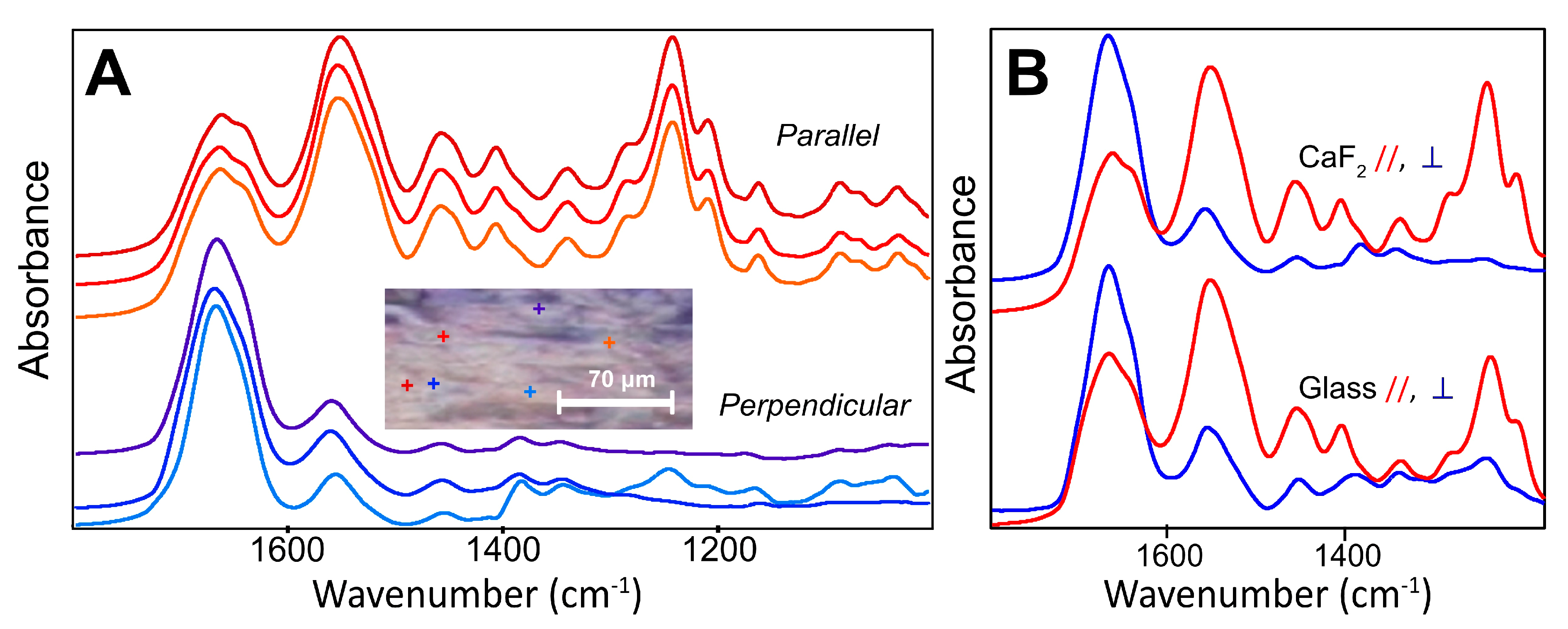
From the FF O-PTIR spectra of intact tendon (
Figure 2A), we can conclude that the polarization of the lasers in the mIRage microscope give results that are essentially equivalent to those obtained with a thermal source FF-FTIR FPA and IR polarizer. The SNR is excellent, particularly on noting that, for single points at least, the data collection time was 30 s under our operating conditions. The band profiles reveal further differences in the Amide bands. The low energy shoulder in the Amide I under parallel polarized light is even more clearly defined with O-PTIR. It is still apparent, though greatly diminished, in the perpendicular orientation. The depth penetration of the O-PTIR spectra is not precisely definable, though presumably a few microns, and possibly the entire depth of these dried sections as it depends on the material probed; however; the spot size is ~500 nm and the spectrum must be more localized than that of FF-FTIR FPA.
Interestingly, relative intensities of the Amide I and II bands in O-PTIR differed slightly more than was observed with the FPA, as did the contrasting profiles within the Amide I. This is possibly due to a higher IR polarization contrast with the QCL, though this seems unlikely, as the IR polarizer is very efficient. Instead, the higher spatial resolution afforded by O-PTIR may probe a more purely oriented local section of the tendon fibrils. The FF-FTIR gives an average of the chemistry from within a few to several micron diameter voxels, while with better spatial resolution, the submicron heterogeneity may be clearer.
The O-PTIR signal intensity was not dependent on the substrate, as numerically comparable results were obtained from the section on glass compared to that on CaF
2, under the same measurement conditions and sample orientation (
Figure 2B). Point to point variation was no greater than that typically observed under FF-FTIR and is ascribed to slight variations in tissue thickness or condition. The ability to measure samples on standard glass slides, without the loss of signal below 1600 cm
−1, as occurs with transmission FTIR on salt windows, is another key advantage of using O-PTIR. The sample may be stained after spectra have been recorded; the glass substrate is inexpensive and is a normal part of histology workflow.
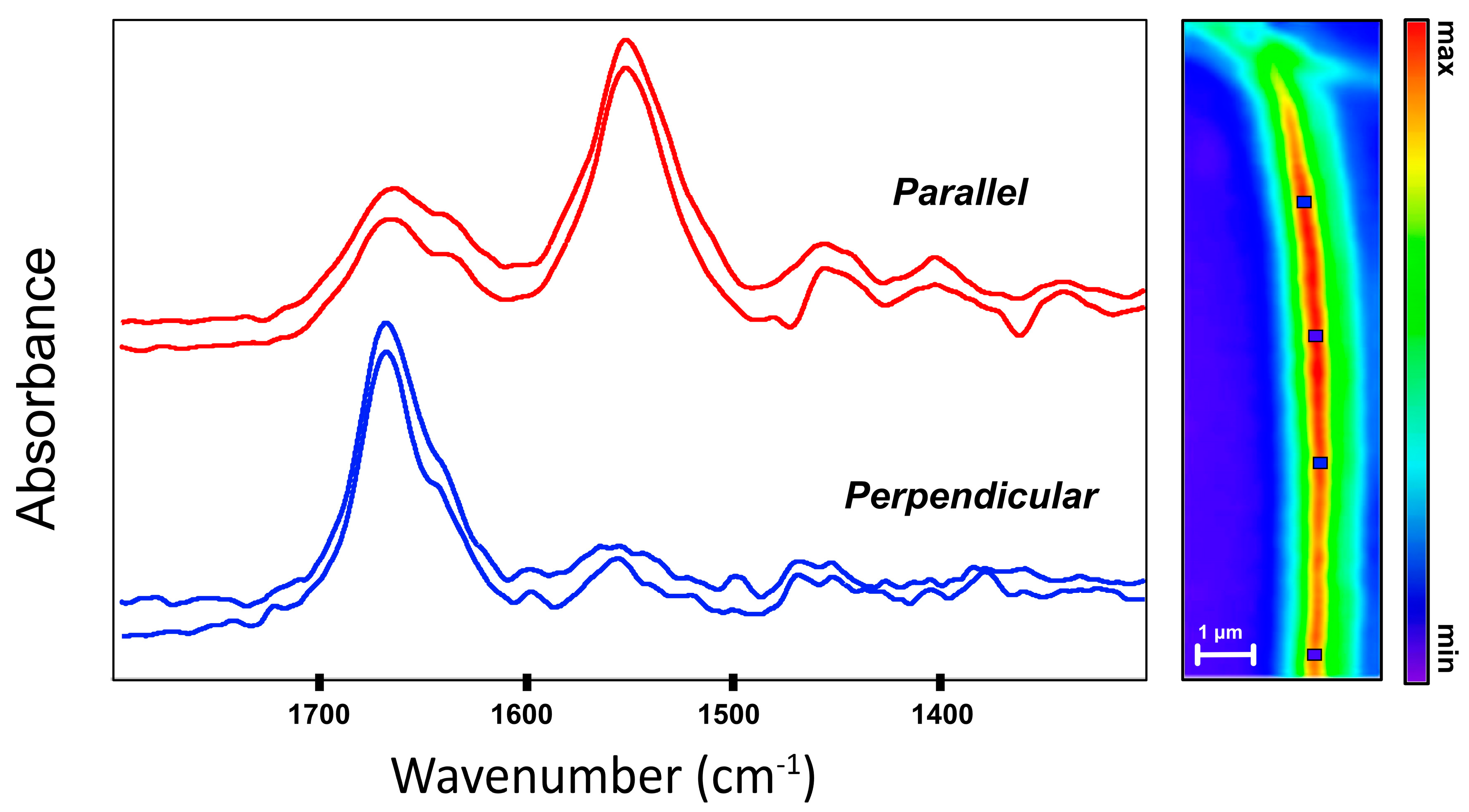
The profiles for the Amide bands of fibrils obtained with O-PTIR (
Figure 3) are similar to those of intact tendon obtained with O-PTIR (
Figure 2) but with cleaner and more pronounced differences. The spectra parallel to the fibril length exhibit a strong Amide I shoulder at lower energy that is nearly absent in the perpendicular spectrum. The increased intensity of the main Amide I absorbance, centered around 1655–1660 cm
−1 could be masking the shoulder but relative intensities of individual bands under different polarizations cannot be ascertained.
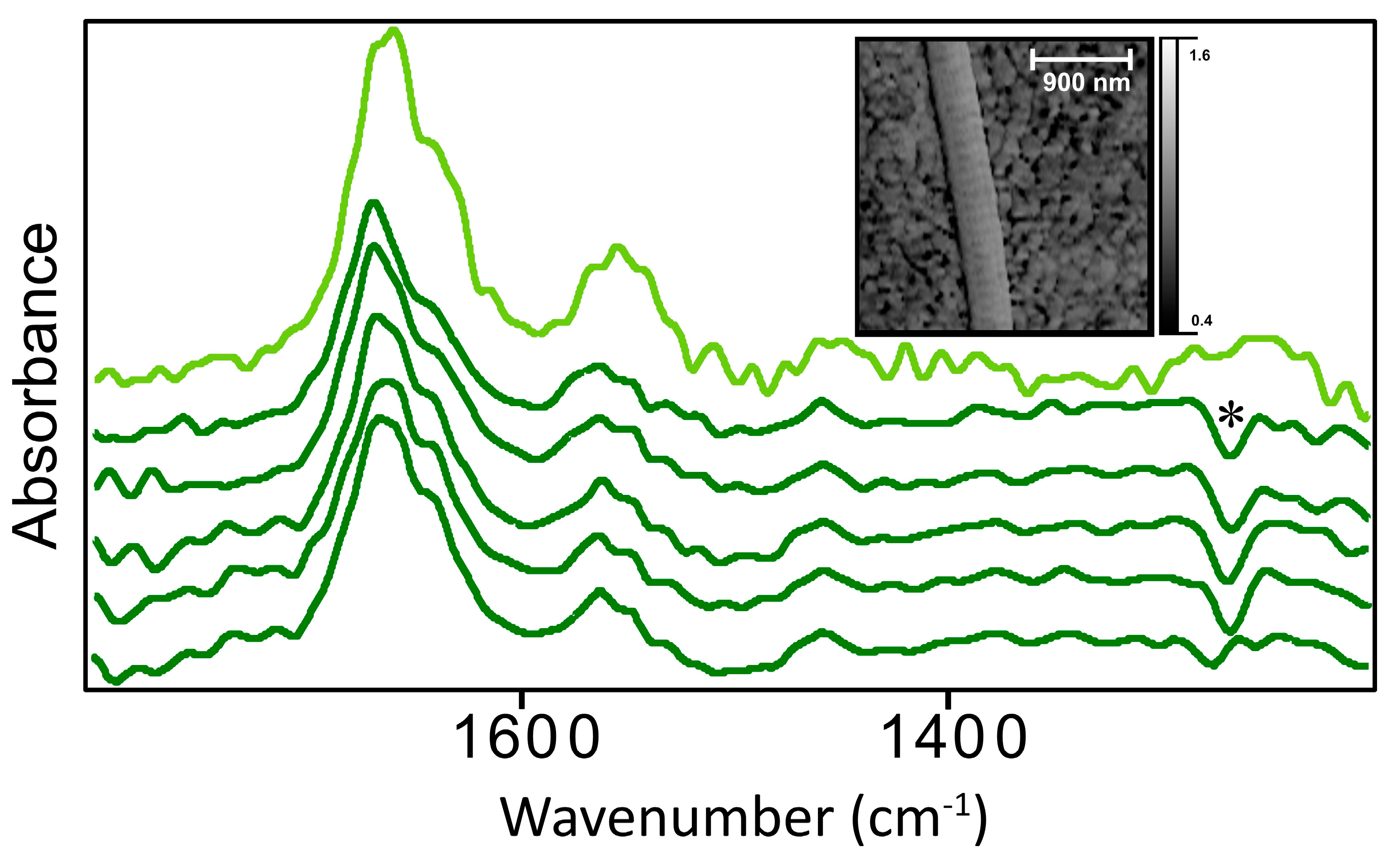
Owing to the radial symmetry of the collagen fibril, and the high polarization in the z-direction (parallel to AFM tip), the nano-FTIR spectra of collagen fibrils are expected to be equivalent to spectra acquired perpendicular to the fibril direction, in the sample x-y plane. Spectra in
Figure 4 are very similar to those observed in our previous work [
7], whether acquired with synchrotron IR light, or the nano-FTIR laser system.
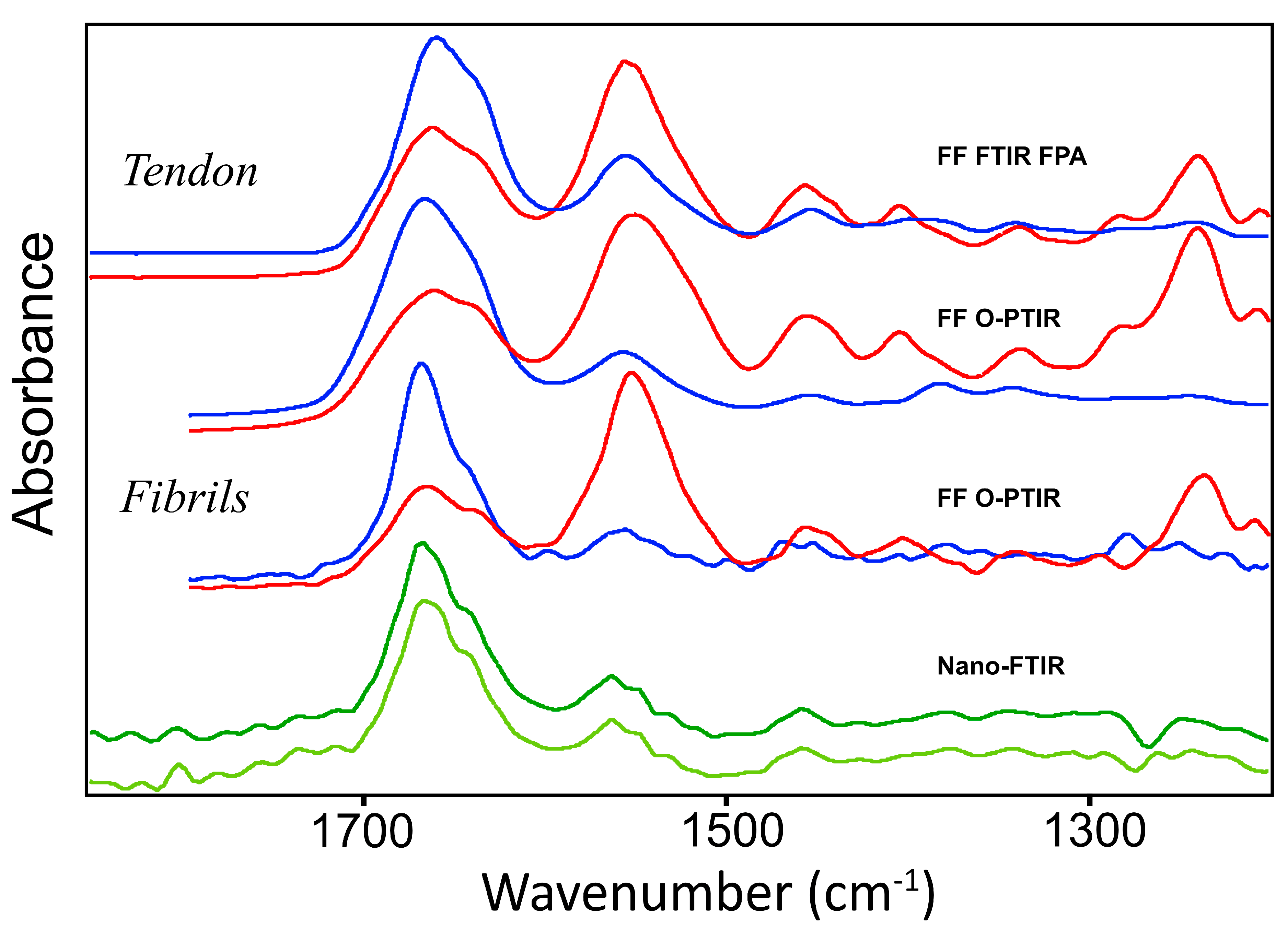
Direct comparison of the averaged spectra from intact tendon and from fibrils, from all experimental techniques (
Figure 5), shows clearly that the Amide bands from intact tendon spectra are considerably broader than those obtained from fibrils, regardless of whether intact tissue spectra were obtained by FF-FTIR FPA or FF O-PTIR. The relative intensity of Amide II to Amide I band drops from about 1.3 (parallel) to 0.3 (perpendicular).
Owing to the sample dimension and sub-fibril heterogeneity [
22], it is not possible to rationally resolve the multiple contributions within either band. It is important to note that, historically, procedures such as curve-fitting, second derivatization (2ndD), and Fourier self-deconvolution (FSD), have sometimes been applied to spectra in an effort to resolve overlapping bands [
4]. We applied FSD and 2ndD to the spectra of intact tissue obtained with FF-FTIR FPA and FF O-PTIR. FSD parameters are typically left to the operator choice; we used a bandwidth of 16 cm
−1 and enhancement factor of 2. The pseudo-deconvolved spectra bore little resemblance to the fibril spectra acquired with sub-micron techniques: relative intensities did not match, multiplets appeared at wavelengths where no bands appeared in the fibril spectra (data not shown). It might be argued that these bands could reflect multiple oblique fibril orientations within the tissue thickness, but these are not unique solutions and cannot provide the information that we sought. There can be no substitute for data acquired at genuinely wavelength-independent submicron spatial resolution.
Comparisons among all spectra show that Amide bands taken with IR light parallel or perpendicular to the control fibril direction are much narrower than those from intact tendon. This indicates that the fibril target is more uniform, whether assessed at ~500 nm (O-PTIR) or 200 nm (nano-FTIR spectroscopy). These dimensions represent the typical range of the “fibril” width; thus, it is very satisfying to see the similarity.
The Amide I band is sharply narrowed in the perpendicular polarized spectra from either method. The principal contribution is from the higher energy band centered at ~1660 cm
−1. The ratio of the Amide II to Amide I integrated band areas is still about 0.3; however, this now reflects the dominant higher energy component. The lower energy shoulder at 1635 cm
−1 is generally thought to arise from the glycine C=O, which points outward from the triple helix, and is mostly available for H-bonding with water [
3,
4]. Such H-bonding would weaken the C=O bond and lower the vibrational energy. If so, then the spectra can be interpreted to mean that this carbonyl is generally aligned more with the fibril direction. Only the O-PTIR technique allows for spectra with polarization parallel to the direction of an individual fibril. The intensity of the 1660 cm
−1 band drops significantly, while that of the lower energy band at 1635 cm
−1 remains distinct. This observation is in keeping with the perpendicular spectra, in that a band more aligned with the fibril direction (1635 cm
−1) should remain strong when the polarization is rotated to the parallel. The triple helix axis is tilted by up to 15 degrees with respect to the microfibril axis (bundle of five collagen triple helices where the molecules are axially quarter-staggered), based on X-ray scattering data on tendons [
22]. Given the dimensions of the fibrils, we must assume this amount of variation is present. The absence of a lipid carbonyl band at the 1735–1740 cm
−1 region confirms that there is little, if any, cellular debris remaining, and that these are spectra of clean, normal D-banded, undamaged collagen fibrils. The O-PTIR and nano-FTIR spectra of fibrils give confirmatory results for spectra with polarization perpendicular to the fibril axis, while the O-PTIR also enables the complementary result for polarization parallel to the fibril axis. We conclude that these spectra represent reliable profiles for control collagen type I fibrils of this dimension, under polarized IR light, and will serve as benchmark references for future studies.
4. Materials and Methods
Tendon samples came from a series described previously [
7]; preparation is summarized briefly here. Tendons were dissected from the tails of young adult steers killed for food, and immediately stored at 4 °C in phosphate buffered saline (PBS) containing 1% antibiotic/antimycotic solution (product A5955, Sigma-Aldrich, Oakville, ON, Canada). Prior to the mechanical damage procedure, a 15-mm-long sample was removed from each tendon to serve as an unloaded control. Using a servo-hydraulic material testing system, the remainder of each tendon was subjected to 5 cycles of sub-rupture overload at a strain rate of 1%/s, as described previously [
7,
27]. Control and stretched tendon segments, approximately 10 mm long by 4 mm diameter, were stored at room temperature for 24 h in PBS containing 1% antibiotic/antimycotic solution and 1% protease inhibitor (product S8820, Sigma-Aldrich, Oakville, ON, Canada); shipped overnight to the University of Manitoba, where they were immediately removed from the PBS medium, covered with OCT (Sakura Finetek Inc., Torrance, CA, USA), frozen in isopentane-cooled in liquid N
2, and stored at −80 °C. Cryosections were cut to 5 or 8 μm thickness at −21 °C, and mounted on CaF
2 or BaF
2 salt windows, as well as on glass microscope slides (for O-PTIR).
For fibril analyses, a thawed control tendon segment was washed as follows: the thawed segment was rinsed in ultra-pure water to remove the majority of the OCT; it was then transferred to a 1.5 mL Eppendorf with ultra-pure water, soaked for 20 min, rinsed again and transferred to a fresh Eppendorf.; this was repeated 6 times. The rinsed segment was dissected further using tweezers and a fine-tipped glass rod in 1 mL of ultra-pure water until the mixture appeared cloudy, releasing many suitable fibrils [
28]. Droplets of the suspensions were deposited on CaF
2 salt windows or on 1 cm
2 gold-coated silicon wafers for O-PTIR and nano-FTIR analysis, respectively.
For FF-FTIR, tissue sections of stretched, sub-rupture tendon were imaged with an Agilent Cary 670 interferometer and 620 IR microscope, equipped with a 64 × 64 Focal Plane Array Mercury Cadmium Telluride (MCT) detector. High magnification optics (1.1 μm × 1.1 μm pixels, 15×, 0.62NA objective) [
25] were used to explore collagen orientation within intact tissue. All spectra were acquired as averages of 128 scans, 4 cm
−1 spectral resolution, ratioed against a background of 512 scans acquired on a clean region of the salt window. Spectral acquisition and data processing were performed with the ResolutionsPro
TM FTIR spectroscopy software (Ver. 5.3.01964, Agilent Technologies Inc., Santa Clara, CA, USA).
O-PTIR spectra and images of tissue and of fibrils were collected on a mIRage+R™ Infrared and Raman microscope (Photothermal Spectroscopy Corp, Santa Barbara, CA, USA). Samples were presented on CaF2 windows and glass microscope slides and measured in reflectance mode. The system was equipped with a pulsed, broadly tunable high-power QCL covering 1800–800 cm−1 as the pump beam (IR source) and a 532 nm continuous wave (CW) probe beam laser. Upon IR frequency-dependent absorption, the photothermal expansion and refractive index changes are detected via lock-in amplifier detection of variations in the reflected probe beam (532 nm) intensity as a function of IR wavelength sweeping. Consequently, the spatial resolution of the IR spectra is now diffraction-limited by the wavelength of the probe beam, to about 350 nm.
The system was purged with dry nitrogen gas to remove water vapor interferences. Data were collected by focusing the sample using the visible image, which was collected with both the 10× visible objective (for sample overview) and the 40×, 0.78 NA reflective Cassegrain-style objective for both sample visualization and O-PTIR data collection. Background spectra were collected on aluminized polypropylene mounted on epoxy to provide an accurate power spectrum of the QCL. This was carried out once at the beginning of the day and used throughout; the QCL was set to 100% power, 100 kHz repetition rate, with a pulse width of 300 ns (3% duty cycle). The probe beam was set to 25% power. Twenty scans were co-added at 6 cm−1 spectral resolution, with a QCL scan rate of 100 cm−1/s. Sample spectra were collected at 79% QCL power, 100 kHz repetition rate, 300 nm pulse width (3% duty cycle). The probe beam was also set at 79% power. Three scans were co-added at 6 cm−1 spectral resolution with a QCL scan rate of 100 cm−1/s. All sample spectra were automatically normalized to the collected background spectrum. IR images were collected at discrete frequencies as specified in the results section. Images were collected with 100 nm pixel sizes at a line scan rate of 1 Hz, with 10 Hz retrace.
Nano-FTIR data were acquired with a neaSNOM (neaspec GmbH, Munich-Haar, Germany) microscope at beamline 2.4 and with the synchrotron infrared nanospectroscopy (SINS) system [
16] at beamline 5.4, ALS, according to protocols detailed previously [
7]. With SINS, spectra were recorded with a modified commercial rapid-scan FTIR spectrometer (Nicolet 6700, Thermo-Scientific, Madison, WI, USA), with an asymmetric Michaelson interferometer. The second order amplitude (s(ω)) and phase (φ(ω)) spectra of the backscattered light were recorded at locations on (sample) and off (reference) the fibrils as a function of frequency, ω. For each sample and reference spectrum, 512 scans were co-added, with a spectral data spacing of 8 cm
−1. Spectra were processed with a fast Fourier-transform analysis suite (Nanospectroscopy Data Analysis (V.1, Advanced Light Source, Berkeley, CA, USA). Data collection with the commercial neaSNOM system at beamline 2.4 was conducted similarly, within the standard operating protocols of the system, synchrotron IR illumination and MCT detector. Finally, spectra were acquired with a neaSNOM microscope and 4.5–15 μm nano-FTIR laser illumination. Spectra were analyzed with the neaPLOT
TM software package (Ver. 1.9.702, neaspec GmbH, Munich-Haar, Germany). AFM and s-SNOM image data were processed in Gwyddion (Ver. 2.51, Brno, Czechia)) [
29].
For all nano-FTIR spectra, whether acquired with synchrotron source IR or nano-FTIR laser illumination, the sample signal was normalized against a signal obtained from a clean location on the AuSi substrate. The gold- or platinum-coated AFM tips used in these experiments are often provided on a polydimethylsiloxane (PDMS) gel substrate that can lead to slight contamination of the tip. Even when transported in gel-free packaging, some contamination may occur in the manufacturing process. As a consequence, PDMS appears in the reference spectrum, observed as negative peaks in the sample spectra.
For post-processing of near-field data, the second order amplitude, s
2(ω), and phase φ
2(ω) spectra of the demodulated complex-valued scattering coefficient σ were recorded, and the imaginary part, which corresponds to the near-field IR absorption, was calculated [
14,
17] according to the relationship:
The near-field nano-FTIR absorbance spectrum was obtained by normalizing the signal on a fibril against a signal from on a flat, non-absorbing surface location on the gold-coated wafer:
Spectra from all sources were exported in spc file format for common display purposes.
5. Conclusions
We have used three different techniques to obtain polarized IR spectra of collagen in intact tendon and as isolated fibrils. The highly linearly polarized IR laser source in the FF O-PTIR yielded spectra of intact tendon that closely matched those obtained from IR polarized FF-FTIR FPA, and the expected dichroic behavior of spectra with respect to fiber orientation was easily displayed. FF-FTIR with FPA allows for the rapid survey of large areas of intact tissue, with or without polarization, though spatial resolution is wavelength-dependent across the 5–10 μm spectral range. With O-PTIR, individual spectra could be acquired rapidly (30s), with equally good SNR, from tendons mounted on salt windows or glass microscope slides. An obvious advantage of obtaining spectra from samples on glass is that subsequent histochemical or immunohistochemical analysis could be performed.
Acquisition of FF-FTIR FPA data from individual fibrils was not feasible; however, in this first demonstration of FF O-PTIR spectra and images of a biopolymer, good data were obtained from fibrils of ~500 nm diameter. Nano-FTIR spectroscopy gave access to the smallest feasible voxel, and from fibrils of 100 to 300 nm diameter. The O-PTIR spectra with perpendicular orientation were an excellent match for nano-FTIR spectra from the smaller fibrils, obtained under synchrotron infrared light and with nano-FTIR lasers.
Relative intensities and band profiles changed between parallel and perpendicular orientations of the fibers and fibrils to polarized IR light, as expected, but distinct differences were noted among the different samples and techniques. For the intact tendon, with IR light polarized parallel to fiber direction, the Amide II band intensity increased relative to the Amide I, again as expected, since the Amide II backbone mode is known to be well aligned with the fiber direction. Interestingly, the 1635 cm−1 shoulder in the Amide I band was more clearly defined in the O-PTIR spectra of intact tendon, in comparison to the FF-FTIR FPA results. This shoulder is often described as being mainly due to the C=O modes of the glycine residue. We conclude that the dipole moment change for this mode is more closely aligned with the molecular backbone. The increased definition of this band in the O-PTIR compared to FF-FTIR FPA spectra is thought to be due to the greater spatial resolution, with O-PTIR providing for a more pure local spectrum. The larger voxel probed with FF-FTIR FPA is averaged across more variations in fiber orientation, and could include additional, non-collagen materials. The Amide I and II bands were significantly narrower in fibril spectra compared to those from the intact tendon. The low energy shoulder at 1635 cm−1 was barely discernible against the high energy band at 1660 cm−1 whether from perpendicular O-PTIR or in the smallest fibrils with nano-FTIR. The collagen fibrils have radial symmetry at this length scale, and the electrical field is parallel to the AFM tip in s-SNOM nano-FTIR; these spectra were also effectively perpendicular. Only the FF O-PTIR could deliver spectra of fibrils with polarization parallel to the fibril orientation. The Amide II band was seen to be narrower than in the intact tendon, probably owing to greater orientation purity, while the 1660 and 1635 cm−1 maxima were again more clearly resolved. This is the first demonstration that O-PTIR and nano-FTIR spectra give confirmatory and complementary results on collagen fibrils with diameters from 100 to 500 nm. We conclude that these spectra represent reliable profiles for control collagen type I fibrils; taken together, these results provide a broad basis for further studies of collagen in biological samples.