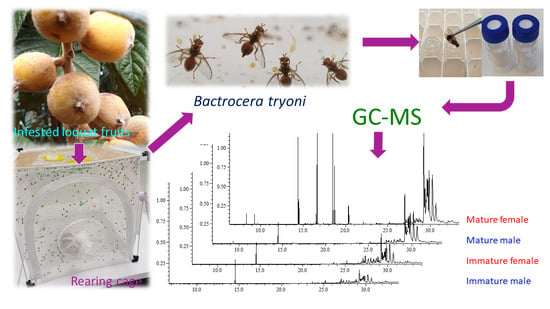
Cuticular Chemistry of the Queensland Fruit Fly Bactrocera tryoni (Froggatt)
Abstract
1. Introduction
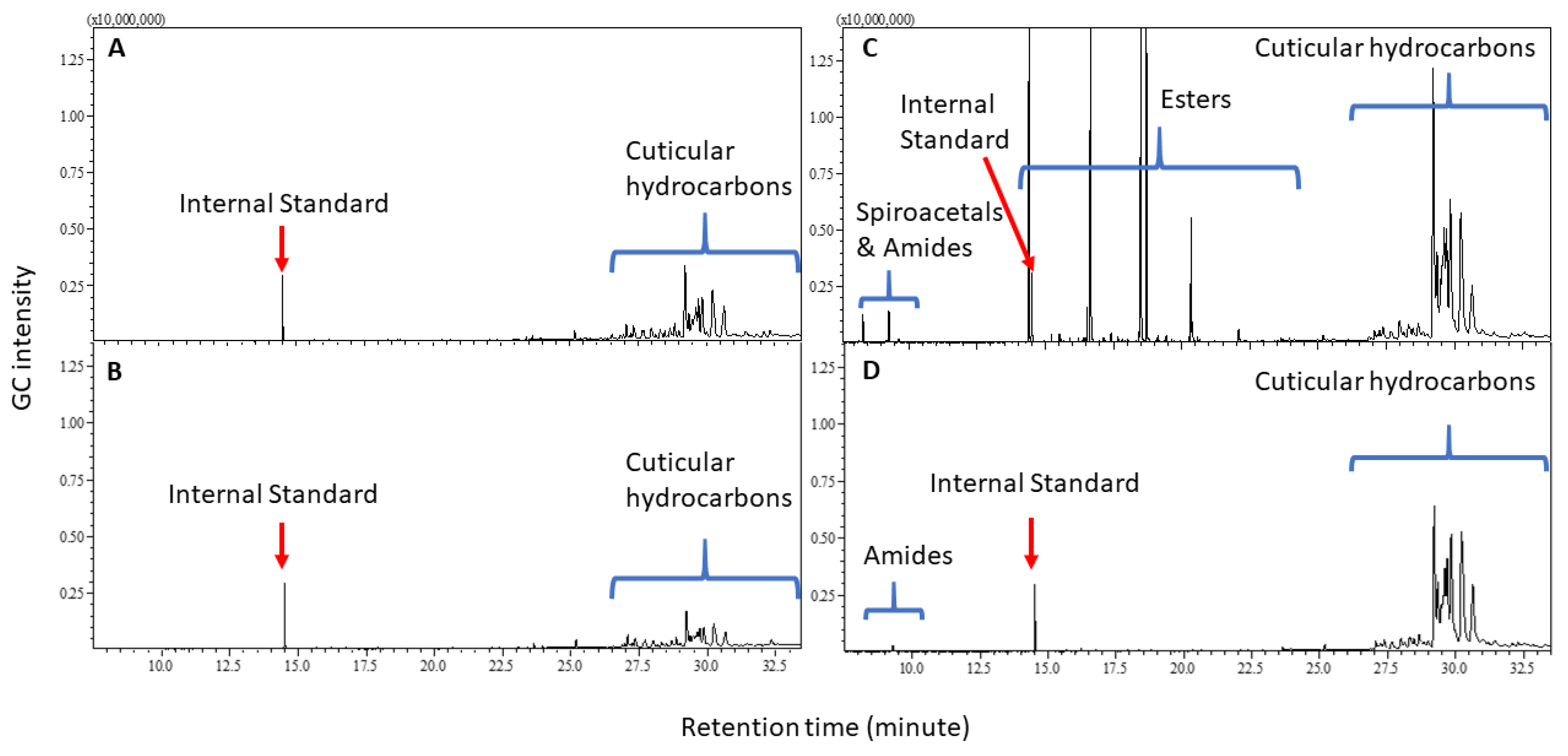
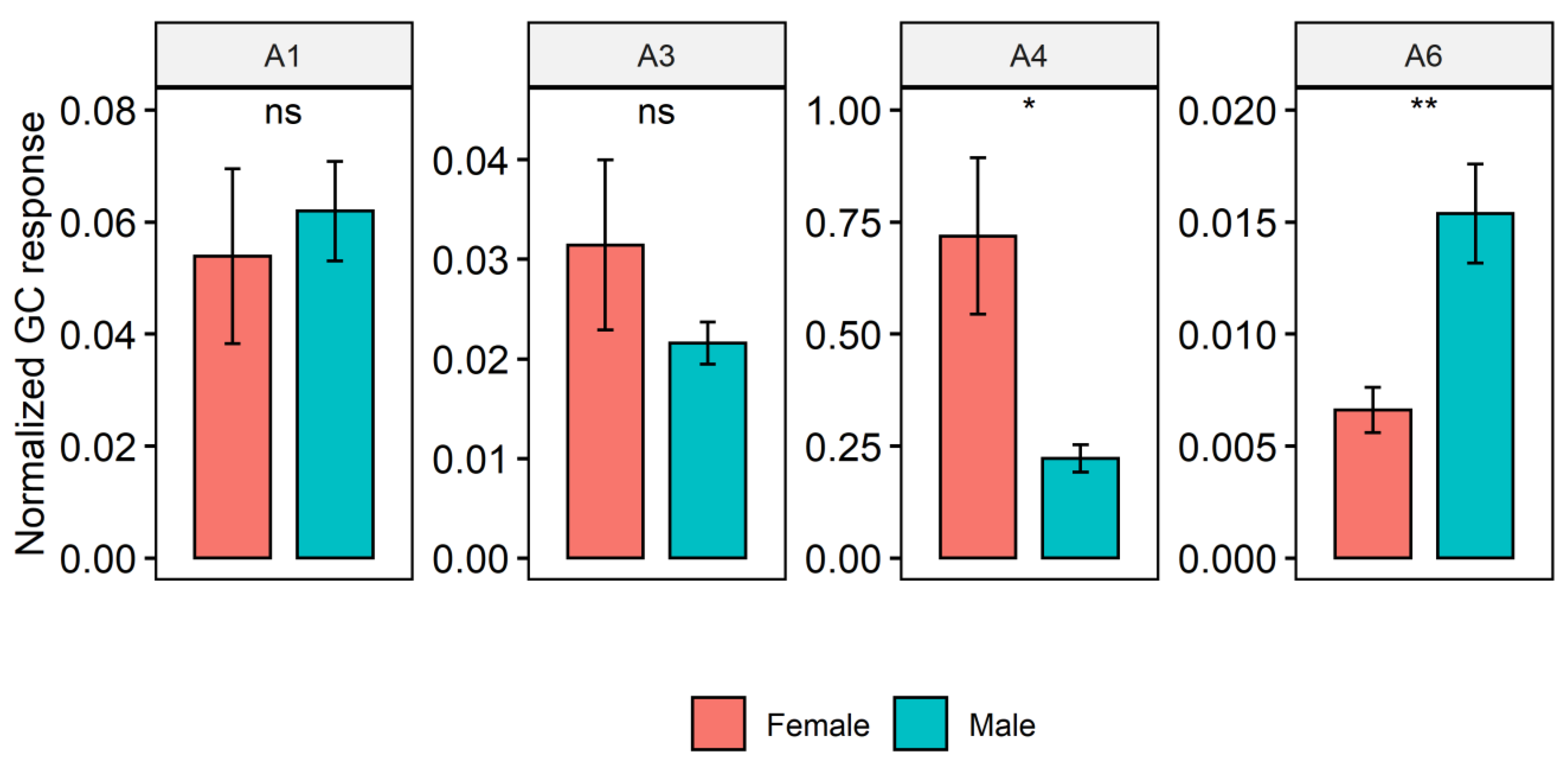
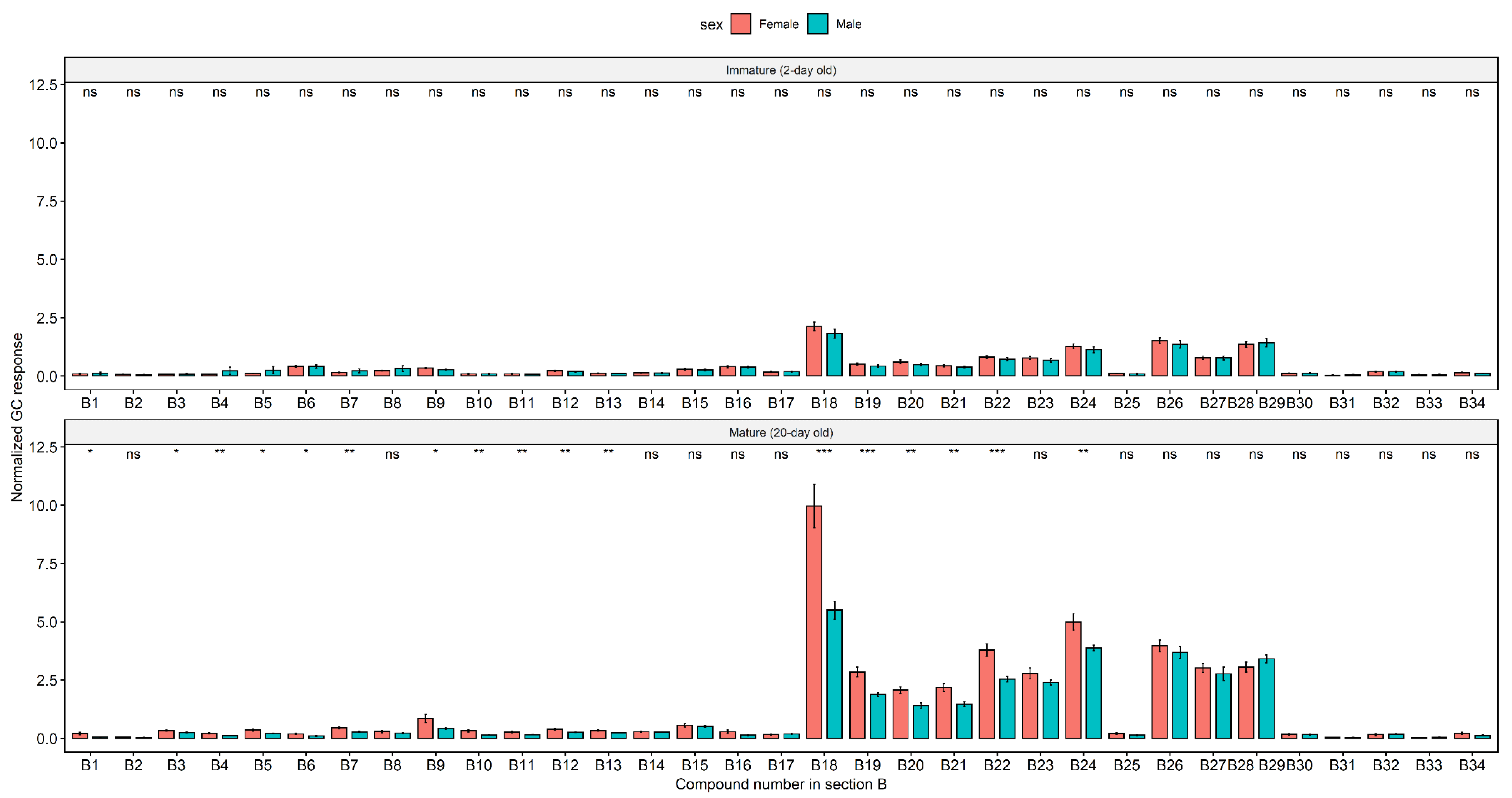
2. Results
Cuticular Chemistry and Statistical Analysis
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Origin of Flies
4.3. Extraction of Cuticular Compounds
4.4. Gas Chromatography Mass Spectrometry (GC-MS) Analysis
4.5. Comparison between Groups
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gibbs, A.; Crockett, E.L. The Biology of Lipids: Integrative and Comparative Perspectives. Am. Zool. 1998, 38, 265–267. [Google Scholar] [CrossRef]
- Howard, R.W.; Blomquist, G.J. Chemical Ecology and Biochemistry of Insect Hydrocarbons. Annu. Rev. Èntomol. 1982, 27, 149–172. [Google Scholar] [CrossRef]
- Lockey, K.H. Lipids of the insect cuticle: Origin, composition and function. Comp. Biochem. Physiol. Part B Comp. Biochem. 1988, 89, 595–645. [Google Scholar] [CrossRef]
- Blomquist, G.J.; Jackson, L.L. Chemistry and biochemistry of insect waxes. Prog. Lipid Res. 1979, 17, 319–345. [Google Scholar] [CrossRef]
- Blomquist, G.J.; Nelson, D.R.; De Renobales, M. Chemistry, biochemistry, and physiology of insect cuticular lipids. Arch. Insect Biochem. Physiol. 1987, 6, 227–265. [Google Scholar] [CrossRef]
- Ferveur, J.-F. Cuticular Hydrocarbons: Their Evolution and Roles in Drosophila Pheromonal Communication. Behav. Genet. 2005, 35, 279–295. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Boguś, M.I.; Paszkiewicz, M.; Stepnowski, P. Cuticular lipids of insects as potential biofungicides: Methods of lipid composition analysis. Anal. Bioanal. Chem. 2010, 399, 3177–3191. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Urbanek, A.; Pietrzak, A.; Naczk, A.M.; Bojke, A.; Tkaczuk, C.; Stepnowski, P. Effects of the entomopathogenic fungus Metarhizium flavoviride on the fat body lipid composition of Zophobas morio larvae (Coleoptera: Tenebrionidae). Naturwissenschaften 2020, 107, 1–11. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Cerkowniak, M.; Urbanek, A.; Dawgul, M.; Kamysz, W.; Boguś, M.I.; Stepnowski, P. Identification and antifungal activity of novel organic compounds found in cuticular and internal lipids of medically important flies. Microbiol. Res. 2015, 170, 213–222. [Google Scholar] [CrossRef]
- Boguś, M.I.; Czygier, M.; Gołębiowski, M.; Kędra, E.; Kucińska, J.; Mazgajska, J.; Samborski, J.; Wieloch, W.; Włóka, E. Effects of insect cuticular fatty acids on in vitro growth and pathogenicity of the entomopathogenic fungus Conidiobolus coronatus. Exp. Parasitol. 2010, 125, 400–408. [Google Scholar] [CrossRef]
- Urbanek, A.; Szadziewski, R.; Stepnowski, P.; Boros-Majewska, J.; Gabriel, I.; Dawgul, M.; Kamysz, W.; Sosnowska, D.; Gołębiowski, M. Composition and antimicrobial activity of fatty acids detected in the hygroscopic secretion collected from the secretory setae of larvae of the biting midge Forcipomyia nigra (Diptera: Ceratopogonidae). J. Insect Physiol. 2012, 58, 1265–1276. [Google Scholar] [CrossRef] [PubMed]
- Gołębiowski, M.; Cerkowniak, M.; Dawgul, M.; Kamysz, W.; Boguś, M.I.; Stepnowski, P. The antifungal activity of the cuticular and internal fatty acid methyl esters and alcohols in Calliphora vomitoria. Parasitology 2013, 140, 972–985. [Google Scholar] [CrossRef]
- Howard, R.W.; Blomquist, G.J. Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu. Rev. Èntomol. 2005, 50, 371–393. [Google Scholar] [CrossRef] [PubMed]
- Carlson, D.A.; Nelson, D.R.; Langley, P.A.; Coates, T.W.; Davis, T.L.; Der Linden, M.E.L.-V. Contact sex pheromone in the tsetse flyGlossina pallidipes (Austen) Identification and Synthesis. J. Chem. Ecol. 1984, 10, 429–450. [Google Scholar] [CrossRef]
- Ginzel, M.D.; Blomquist, G.J.; Millar, J.G.; Hanks, L.M. Role of contact pheromones in mate recognition in Xylotrechus colonus. J. Chem. Ecol. 2003, 29, 533–545. [Google Scholar] [CrossRef]
- Wilson, E.O. Chemical Communication within Animal Species; Academic Press, Inc.: New York, NY, USA, 1970; pp. 133–155. [Google Scholar]
- Xue, H.-J.; Segraves, K.A.; Wei, J.; Zhang, B.; Nie, R.-E.; Li, W.-Z.; Yang, X.-K. Chemically mediated sexual signals restrict hybrid speciation in a flea beetle. Behav. Ecol. 2018, 29, 1462–1471. [Google Scholar] [CrossRef]
- Dettner, K.; Liepert, C. Chemical Mimicry and Camouflage. Annu. Rev. Entomol. 1994, 39, 129–154. [Google Scholar] [CrossRef]
- Heifetz, Y.; Voet, H.; Applebaum, S.W. Factors affecting behavioral phase transition in the desert locust, Schistocerca gregaria (Forskål) (Orthoptera: Acrididae). J. Chem. Ecol. 1996, 22, 1717–1734. [Google Scholar] [CrossRef]
- Carlson, D.A.; Mayer, M.S.; Silhacek, D.L.; James, J.D.; Beroza, M.; Bierl, B.A. Sex Attractant Pheromone of the House Fly: Isolation, Identification and Synthesis. Science 1971, 174, 76–78. [Google Scholar] [CrossRef]
- Jennings, J.H.; Etges, W.J.; Schmitt, T.; Hoikkala, A. Cuticular hydrocarbons of Drosophila montana: Geographic variation, sexual dimorphism and potential roles as pheromones. J. Insect Physiol. 2014, 61, 16–24. [Google Scholar] [CrossRef]
- Roelofs, W.L.; Liu, W.; Hao, G.; Jiao, H.; Rooney, A.P.; Linn, J.C.E. Evolution of moth sex pheromones via ancestral genes. Proc. Natl. Acad. Sci. USA 2002, 99, 13621–13626. [Google Scholar] [CrossRef] [PubMed]
- Mant, J.; Brändli, C.; Vereecken, N.J.; Schulz, C.M.; Francke, W.; Schiestl, F.P. Cuticular Hydrocarbons as Sex Pheromone of the Bee Colletes cunicularius and the Key to its Mimicry by the Sexually Deceptive Orchid, Ophrys exaltata. J. Chem. Ecol. 2005, 31, 1765–1787. [Google Scholar] [CrossRef] [PubMed]
- Nojima, S.; Shimomura, K.; Honda, H.; Yamamoto, I.; Ohsawa, K. Contact Sex Pheromone Components of the Cowpea Weevil, Callosobruchus maculatus. J. Chem. Ecol. 2007, 33, 923–933. [Google Scholar] [CrossRef]
- Keesey, I.W.; Koerte, S.; Retzke, T.; Haverkamp, A.; Hansson, B.S.; Knaden, M. Adult Frass Provides a Pheromone Signature for Drosophila Feeding and Aggregation. J. Chem. Ecol. 2016, 42, 739–747. [Google Scholar] [CrossRef]
- Mitaka, Y.; Matsuyama, S.; Mizumoto, N.; Matsuura, K.; Akino, T. Chemical identification of an aggregation pheromone in the termite Reticulitermes speratus. Sci. Rep. 2020, 10, 7424. [Google Scholar] [CrossRef]
- Hamilton, J.A.; Wada-Katsumata, A.; Schal, C. Role of Cuticular Hydrocarbons in German Cockroach (Blattodea: Ectobiidae) Aggregation Behavior. Environ. Èntomol. 2019, 48, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Cooper, W.E.; Andersson, M. Sexual Selection. Copeia 1995, 756. [Google Scholar] [CrossRef]
- Blomquist, G.J.; Bagnères, A.-G. Introduction: History and overview of insect hydrocarbons. In Insect Hydrocarbons: Biology, Biochemistry, and Chemical Ecologyi; Bagnères, A.-G., Blomquist, G.J., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 3–18. [Google Scholar]
- Luo, Y.; Zhang, Y.; Farine, J.; Ferveur, J.; Ramírez, S.; Kopp, A. Evolution of sexually dimorphic pheromone profiles coincides with increased number of male-specific chemosensory organs in Drosophila prolongata. Ecol. Evol. 2019, 9, 13608–13618. [Google Scholar] [CrossRef]
- Ngumbi, E.N.; Hanks, L.M.; Suarez, A.V.; Millar, J.G.; Berenbaum, M.R. Author Correction: Factors Associated with Variation in Cuticular Hydrocarbon Profiles in the Navel Orangeworm, Amyelois transitella (Lepidoptera: Pyralidae). J. Chem. Ecol. 2020, 46, 232. [Google Scholar] [CrossRef]
- Farine, J.-P.; Ferveur, J.-F.; Everaerts, C. Volatile Drosophila Cuticular Pheromones Are Affected by Social but Not Sexual Experience. PLoS ONE 2012, 7, e40396. [Google Scholar] [CrossRef]
- Dias, N.P.; Zotti, M.J.; Montoya, P.; Carvalho, I.R.; Nava, D.E. Fruit fly management research: A systematic review of monitoring and control tactics in the world. Crop. Prot. 2018, 112, 187–200. [Google Scholar] [CrossRef]
- Robledo, N.; Arzuffi, R.; Robledo-Quintos, N. Influence of Host Fruit and Conspecifics on the Release of the Sex Pheromone By Toxotrypana curvicauda Males (Diptera: Tephritidae). Environ. Èntomol. 2012, 41, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.K.; Nishida, R. Incorporation of Raspberry Ketone in the Rectal Glands of Males of the Queensland Fruit Fly, Bactrocera tryoni FROGGATT (Diptera: Tephritidae). Appl. Èntomol. Zool. 1995, 30, 494–497. [Google Scholar] [CrossRef][Green Version]
- Jang, E.B.; Carvalho, L.A.F.N.; Chen, C.-C.; Siderhurst, M.S. Cucumber Lure Trapping of Zeugodacus cucurbitae (Diptera:Tephritidae) in Hawaii and Taiwan: Longevity and Nontargets Captures. J. Econ. Èntomol. 2016, 110, 201–207. [Google Scholar] [CrossRef][Green Version]
- Royer, J.E.; De Faveri, S.G.; Lowe, G.E.; Wright, C.L. Cucumber volatile blend, a promising female-biased lure for B actrocera cucumis (French 1907) (Diptera: Tephritidae: Dacinae), a pest fruit fly that does not respond to male attractants. Austral Èntomol. 2014, 53, 347–352. [Google Scholar] [CrossRef]
- Siderhurst, M.S.; Jang, E.B. Female-Biased Attraction of Oriental Fruit Fly, Bactrocera dorsalis (Hendel), to a Blend of Host Fruit Volatiles From Terminalia catappa L. J. Chem. Ecol. 2006, 32, 2513–2524. [Google Scholar] [CrossRef]
- Siderhurst, M.S.; Jang, E.B. Attraction of female Oriental fruit fly, Bactrocera dorsalis, to Terminalia catappa fruit in wind tunnel and olfactometer tests. Formos. Entomol. 2006, 26, 45–55. [Google Scholar]
- Siderhurst, M.S.; Jang, E.B. Cucumber Volatile Blend Attractive to Female Melon Fly, Bactrocera cucurbitae (Coquillett). J. Chem. Ecol. 2010, 36, 699–708. [Google Scholar] [CrossRef]
- Tan, K.H. Fruit fly pests as pollinators of wild orchids. In Proceedings of the 7th International Symposium on Fruit Flies of Economic Importance, Salvador, Brazil, 10–15 September 2006; pp. 195–206. [Google Scholar]
- Tan, K.H.; Nishida, R. Zingerone in the floral synomone of Bulbophyllum baileyi (Orchidaceae) attracts Bactrocera fruit flies during pollination. Biochem. Syst. Ecol. 2007, 35, 334–341. [Google Scholar] [CrossRef]
- Tan, K.H.; Tan, L.T.; Nishida, R. Floral Phenylpropanoid Cocktail and Architecture of Bulbophyllum vinaceum Orchid in Attracting Fruit Flies for Pollination. J. Chem. Ecol. 2006, 32, 2429–2441. [Google Scholar] [CrossRef]
- Tan, K.-H.; Nishida, R. Synomone or kairomone?—Bulbophyllum apertum flower releases raspberry ketone to attract Bactrocera fruit flies. J. Chem. Ecol. 2005, 31, 497–507. [Google Scholar]
- Tan, K.-H.; Nishida, R.; Toong, Y.-C. Floral synomone of a wild orchid, Bulbophyllum cheiri, lures Bactrocera fruit flies for pollination. J. Chem. Ecol. 2002, 28, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Jang, E.B.; Carvalho, L.A.; Stark, J.D. Attraction of Female Oriental Fruit Fly, Bactrocera dorsalis, to Volatile Semiochemicals from Leaves and Extracts of a Nonhost Plant, Panax (Polyscias guilfoylei) in Laboratory and Olfactometer Assays. J. Chem. Ecol. 1997, 23, 1389–1401. [Google Scholar] [CrossRef]
- Siderhurst, M.S.; Park, S.J.; Buller, C.N.; Jamie, I.M.; Manoukis, N.C.; Jang, E.B.; Taylor, P.W. Raspberry Ketone Trifluoroacetate, a New Attractant for the Queensland Fruit Fly, Bactrocera Tryoni (Froggatt). J. Chem. Ecol. 2016, 42, 156–162. [Google Scholar] [CrossRef]
- Kather, R.; Martin, S.J. Cuticular hydrocarbon profiles as a taxonomic tool: Advantages, limitations and technical aspects. Physiol. Èntomol. 2012, 37, 25–32. [Google Scholar] [CrossRef]
- Vanickova, L.; Hernández-Ortiz, V.; Bravo, I.S.J.; Dias, V.S.; Roriz, A.K.P.; Laumann, R.A.; Mendonça, A.D.L.; Paranhos, B.A.J.; Nascimento, R.R.D. Current knowledge of the species complex Anastrepha fraterculus (Diptera, Tephritidae) in Brazil. ZooKeys 2015, 540, 211–237. [Google Scholar] [CrossRef]
- Carlson, D.A.; Yocom, S.R. Cuticular hydrocarbons from six species of tephritid fruit flies. Arch. Insect Biochem. Physiol. 1986, 3, 397–412. [Google Scholar] [CrossRef]
- Sutton, B.D.; Carlson, D.A. Interspecific variation in tephritid fruit fly larvae surface hydrocarbons. Arch. Insect Biochem. Physiol. 1993, 23, 53–65. [Google Scholar] [CrossRef]
- Sutton, B.D.; Steck, G.J. Discrimination of Caribbean and Mediterranean Fruit Fly Larvae (Diptera: Tephritidae) by Cuticular Hydrocarbon Analysis. Fla. Èntomol. 1994, 77, 231. [Google Scholar] [CrossRef]
- Vaníčková, L.; Virgilio, M.; Tomčala, A.; Břízová, R.; Ekesi, S.; Hoskovec, M.; Kalinová, B.; Do Nascimento, R.R.; De Meyer, M. Resolution of three cryptic agricultural pests (Ceratitis fasciventris, C. anonae, C. rosa, Diptera: Tephritidae) using cuticular hydrocarbon profiling. Bull. Entomol. Res. 2014, 104, 631–638. [Google Scholar]
- Vanickova, L.; Břízová, R.; Mendonça, A.L.; Pompeiano, A.; Nascimento, R.R.D. Intraspecific variation of cuticular hydrocarbon profiles in theAnastrepha fraterculus(Diptera:Tephritidae) species complex. J. Appl. Èntomol. 2015, 139, 679–689. [Google Scholar] [CrossRef]
- Vanickova, L.; Břízová, R.; Pompeiano, A.; Ekesi, S.; De Meyer, M. Cuticular hydrocarbons corroborate the distinction between lowland and highland Natal fruit fly (Tephritidae, Ceratitis rosa) populations. ZooKeys 2015, 540, 507–524. [Google Scholar] [CrossRef] [PubMed]
- Vanickova, L.; Břízová, R.; Pompeiano, A.; Ferreira, L.L.; De Aquino, N.C.; Tavares, R.D.F.; Rodriguez, L.D.; Mendonça, A.D.L.; Canal, N.A.; Nascimento, R.R.D. Characterisation of the chemical profiles of Brazilian and Andean morphotypes belonging to the Anastrepha fraterculus complex (Diptera, Tephritidae). ZooKeys 2015, 540, 193–209. [Google Scholar] [CrossRef] [PubMed]
- Goh, S.; Ooi, K.; Chuah, C.; Yong, H.; Khoo, S.; Ong, S. Cuticular hydrocarbons from two species of Malaysian Bactrocera fruit flies. Biochem. Syst. Ecol. 1993, 21, 215–226. [Google Scholar] [CrossRef]
- Vanickova, L.; Nagy, R.; Pompeiano, A.; Kalinová, B. Epicuticular chemistry reinforces the new taxonomic classification of the Bactrocera dorsalis species complex (Diptera: Tephritidae, Dacinae). PLoS ONE 2017, 12, e0184102. [Google Scholar] [CrossRef]
- Shen, J.; Hu, L.; Zhou, X.; Dai, J.; Chen, B.; Li, S. Allyl-2,6-dimethoxyphenol, a female-biased compound, is robustly attractive to conspecific males of Bactrocera dorsalis at close range. Èntomol. Exp. Appl. 2019, 167, 811–819. [Google Scholar] [CrossRef]
- Clarke, A.R.; Powell, K.S.; Weldon, C.W.; Taylor, P.W. The ecology of Bactrocera tryoni (Diptera: Tephritidae): What do we know to assist pest management? Ann. Appl. Biol. 2011, 158, 26–54. [Google Scholar] [CrossRef]
- Gilchrist, A.S.; Dominiak, B.; Gillespie, P.S.; Sved, J.A. Variation in population structure across the ecological range of the Queensland fruit fly. Bactrocera tryoni. Aust. J. Zool. 2006, 54, 87. [Google Scholar] [CrossRef]
- Sutherst, R.W.; Collyer, B.S.; Yonow, T. The vulnerability of Australian horticulture to the Queensland fruit fly, Bactrocera (Dacus) tryoni, under climate change. Aust. J. Agric. Res. 2000, 51, 467. [Google Scholar] [CrossRef]
- Dominiak, B.; Ekman, J.H.; Broughton, S. Mass trapping and other management options for Mediterranean fruit fly and Queensland fruit fly in Australia. Gen. App. Entomol. 2016, 44, 1–8. [Google Scholar]
- Florec, V.; Sadler, R.J.; White, B.; Dominiak, B.C.; White, B. Choosing the battles: The economics of area wide pest management for Queensland fruit fly. Food Policy 2013, 38, 203–213. [Google Scholar] [CrossRef]
- Bellas, T.E.; Fletcher, B.S. Identification of the major components in the secretion from the rectal pheromone glands of the queensland fruit fliesDacus tryoni andDacus neohumeralis (Diptera: Tephritidae). J. Chem. Ecol. 1979, 5, 795–803. [Google Scholar] [CrossRef]
- Fletcher, B.S. Storage and Release of a Sex Pheromone by the Queensland Fruit Fly, Dacus tryoni (Diptera: Trypetidae). Nature 1968, 219, 631–632. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, A.M.; Venkatesham, U.; Unelius, C.R.; Sporle, A.; Pérez, J.; Taylor, P.W.; Suckling, D.M. Chemical Composition of the Rectal Gland and Volatiles Released by Female Queensland Fruit Fly, Bactrocera tryoni (Diptera: Tephritidae). Environ. Èntomol. 2019, 48, 807–814. [Google Scholar] [CrossRef]
- Pérez, J.; Park, S.J.; Taylor, P.W. Domestication modifies the volatile emissions produced by male Queensland fruit flies during sexual advertisement. Sci. Rep. 2018, 8, 16503. [Google Scholar] [CrossRef] [PubMed]
- Noushini, S.; Park, S.J.; Jamie, I.M.; Jamie, J.F.; Taylor, P.W. Sampling technique biases in the analysis of fruit fly pheromones: A case study of Queensland fruit fly. Sci. Rep. 2020, in press. [Google Scholar]
- Booth, Y.K.; Schwartz, B.D.; Fletcher, M.; Lambert, L.K.; Kitching, W.; De Voss, J.J. A diverse suite of spiroacetals, including a novel branched representative, is released by female Bactrocera tryoni (Queensland fruit fly). Chem. Commun. 2006, 42, 3975–3977. [Google Scholar] [CrossRef]
- Lin, Y.; Jin, T.; Zeng, L.; Lu, Y. Cuticular penetration of β-cypermethrin in insecticide-susceptible and resistant strains of Bactrocera dorsalis. Pestic. Biochem. Physiol. 2012, 103, 189–193. [Google Scholar] [CrossRef]
- Sultana, S.; Baumgartner, J.B.; Dominiak, B.C.; Royer, J.E.; Beaumont, L.J. Potential impacts of climate change on habitat suitability for the Queensland fruit fly. Sci. Rep. 2017, 7, 13025. [Google Scholar] [CrossRef]
- Weldon, C.W.; Taylor, P.W. Desiccation resistance of adult Queensland fruit flies Bactrocera tryoni decreases with age. Physiol. Èntomol. 2010, 35, 385–390. [Google Scholar] [CrossRef]
- Weldon, C.W.; Yap, S.; Taylor, P. Desiccation resistance of wild and mass-reared Bactrocera tryoni (Diptera: Tephritidae). Bull. Èntomol. Res. 2013, 103, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Dominiak, B.C.; Westcott, A.E.; Barchia, I.M. Release of sterile Queensland fruit fly, Bactrocera > tryoni (Froggatt) (Diptera: Tephritidae), at Sydney, Australia. Aust. J. Exp. Agric. 2003, 43, 519–528. [Google Scholar] [CrossRef]
- Meats, A.; Duthie, R.; Clift, A.D.; Dominiak, B.C. Trials on variants of the Sterile Insect Technique (SIT) for suppression of populations of the Queensland fruit fly in small towns neighbouring a quarantine (exclusion) zone. Aust. J. Exp. Agric. 2003, 43, 389–395. [Google Scholar] [CrossRef]
- Reynolds, O.L.; Smallridge, C.J.; Cockington, V.G.; Penrose, L.D. The effect of release method and trial site on recapture rates of adult sterile Queensland fruit fly, Bactrocera tryoni (Froggatt) (Diptera: Tephritidae). Aust. J. Èntomol. 2011, 51, 116–126. [Google Scholar] [CrossRef]
- Noushini, S.; Pérez, J.; Park, S.J.; Holgate, D.; Alvarez, V.M.; Jamie, I.M.; Jamie, J.F.; Taylor, P.W. Attraction and Electrophysiological Response to Identified Rectal Gland Volatiles in Bactrocera frauenfeldi (Schiner). Molecules 2020, 25, 1275. [Google Scholar] [CrossRef]
- El-Sayed, A.M.; Heppelthwaite, V.J.; Manning, L.-A.; Gibb, A.R.; Suckling, D.M.; El-Sayed, A.M. Volatile Constituents of Fermented Sugar Baits and Their Attraction to Lepidopteran Species. J. Agric. Food Chem. 2005, 53, 953–958. [Google Scholar] [CrossRef]
- Pino, J.A.; Mesa, J.; Muñoz, Y.; Martí, M.P.; Marbot, R. Volatile Components from Mango (Mangifera indica L.) Cultivars. J. Agric. Food Chem. 2005, 53, 2213–2223. [Google Scholar] [CrossRef]
- Peppard, T.L. Volatile flavor constituents of Monstera deliciosa. J. Agric. Food Chem. 1992, 40, 257–262. [Google Scholar] [CrossRef]
- Silva, D.B.; Pott, A.; Oliveira, D.C.R. Analyses of the Headspace Volatile Constituents of Aerial Parts (leaves and stems), Flowers and Fruits of Bidens gardneri Bak. and Bidens sulphurea (Cav.) Sch.Bip. Using Solid-Phase Microextraction. J. Essent. Oil Res. 2010, 22, 560–563. [Google Scholar] [CrossRef]
- Isidorov, V.; Krajewska, U.; Dubis, E.N.; Jdanova, M. Partition coefficients of alkyl aromatic hydrocarbons and esters in a hexane-acetonitrile system. J. Chromatogr. A 2001, 923, 127–136. [Google Scholar] [CrossRef]
- Robinson, A.L.; Adams, D.O.; Boss, P.K.; Heymann, H.; Solomon, P.S.; Trengove, R. Influence of Geographic Origin on the Sensory Characteristics and Wine Composition of Vitis vinifera cv. Cabernet Sauvignon Wines from Australia. Am. J. Enol. Vitic. 2012, 63, 467–476. [Google Scholar] [CrossRef]
- Palmeira, S.F.; Moura, F.D.S.; Alves, V.D.L.; De Oliveira, F.M.; Bento, E.S.; Conserva, L.M.; Andrade, E.D.A. Neutral components from hexane extracts ofCroton sellowii. Flavour Fragr. J. 2004, 19, 69–71. [Google Scholar] [CrossRef]
- Pino, J.A.; Almora, K.; Marbot, R. Volatile components of papaya (Carica papaya L., Maradol variety) fruit. Flavour Fragr. J. 2003, 18, 492–496. [Google Scholar] [CrossRef]
- Palmeira, S.F.; Conserva, L.M.; Andrade, E.H.D.A.; Guilhon, G.M.S.P. Analysis by GC-MS of the hexane extract of the aerial parts of Aristolochia acutifolia Duchtr. Flavour Fragr. J. 2001, 16, 85–88. [Google Scholar] [CrossRef]
- Demyttenaere, J.C.; Martínez, J.I.S.; Verhé, R.; Sandra, P.; De Kimpe, N.; Sanchezmartinez, J. Analysis of volatiles of malt whisky by solid-phase microextraction and stir bar sorptive extraction. J. Chromatogr. A 2003, 985, 221–232. [Google Scholar] [CrossRef]
- Andriamaharavo, N.R. Retention Data: NIST Mass Spectrometry Data Center. 2014. Available online: https://webbook.nist.gov/cgi/cbook.cgi?Source=2014AND%2319410M&Units=SI&Mask=2000 (accessed on 10 September 2020).
- Tuberoso, C.; Barra, A.; Angioni, A.; Sarritzu, E.; Pirisi, F.M. Chemical Composition of Volatiles in Sardinian Myrtle (Myrtus communis L.) Alcoholic Extracts and Essential Oils. J. Agric. Food Chem. 2006, 54, 1420–1426. [Google Scholar] [CrossRef]
- Kenig, F.; Damsté, J.S.S.; Dalen, A.K.-V.; Rijpstra, W.C.; Huc, A.Y.; De Leeuw, J.W. Occurrence and origin of mono-, di-, and trimethylalkanes in modern and Holocene cyanobacterial mats from Abu Dhabi, United Arab Emirates. Geochim. Cosmochim. Acta 1995, 59, 2999–3015. [Google Scholar] [CrossRef]
- Nelson, D.R.; Charlet, L.D. Cuticular hydrocarbons of the sunflower beetle, Zygogramma exclamationis. Comp. Biochem. Physiol. Part B Biochem. Mol. Boil. 2003, 135, 273–284. [Google Scholar] [CrossRef]
- Carlson, D.; Geden, C.; Bernier, U. Identification of Pupal Exuviae of Nasonia vitripennis and Muscidifurax raptorellus Parasitoids Using Cuticular Hydrocarbons. Boil. Control. 1999, 15, 97–106. [Google Scholar] [CrossRef]
- Lockey, K.H. Cuticular hydrocarbons of adult Cylindrinotus laevioctostriatus (Goeze) and Phylan gibbus (Fabricius) (Coleoptera: Tenebrionidae). Insect Biochem. 1981, 11, 549–561. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Chen, K.; Maran, U.; Carlson, D.A. QSPR correlation and predictions of GC retention indexes for methyl-branched hydrocarbons produced by insects. Anal. Chem. 2000, 72, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Lockey, K.H. The adult cuticular hydrocarbons of Tenebrio molitor L. and Tenebrio obscurus F. (Coleoptera: Tenebrionidae). Insect Biochem. 1978, 8, 237–250. [Google Scholar] [CrossRef]
- Bernier, U.R.; Carlson, D.A.; Geden, C.J. Gas chromatography/mass spectrometry analysis of the cuticular hydrocarbons from parasitic wasps of the genus Muscidifurax. J. Am. Soc. Mass Spectrom. 1998, 9, 320–332. [Google Scholar] [CrossRef]
- Ekanayake, D. [Leave for Adam-Compressed File] The Mating System and Courtship Behaviour of the Queensland Fruit Fly, Bactrocera tryoni (Froggatt) (Diptera: Tephritidae); Queensland University of Technology: Brisbane, Australia, 2017; p. 271. [Google Scholar]
- Blomquist, G.J.; Tittiger, C.; Jurenka, R. Cuticular Hydrocarbons and Pheromones of Arthropods. In Hydrocarbons, Oils and Lipids: Diversity, Origin, Chemistry and Fate. Available online: https://doi.org/10.1007/978-3-319-54529-5_11-1 (accessed on 10 September 2020).
- Vanickova, L.; Svatoš, A.; Kroiss, J.; Kaltenpoth, M.; Nascimento, R.R.D.; Hoskovec, M.; Břízová, R.; Kalinová, B. Cuticular Hydrocarbons of the South American Fruit Fly Anastrepha fraterculus: Variability with Sex and Age. J. Chem. Ecol. 2012, 38, 1133–1142. [Google Scholar] [CrossRef]
- Lucas, C.; Pho, D.; Fresneau, D.; Jallon, J. Hydrocarbon circulation and colonial signature in Pachycondyla villosa. J. Insect Physiol. 2004, 50, 595–607. [Google Scholar] [CrossRef]
- Soroker, V.; Vienne, C.; Hefetz, A. Hydrocarbon dynamics within and between nestmates in Cataglyphis niger (Hymenoptera: Formicidae). J. Chem. Ecol. 1995, 21, 365–378. [Google Scholar] [CrossRef]
- Bagnères, A.-G.; Trabalon, M.; Blomquist, G.J.; Schulz, S. Waxes of the social spider Anelosimus eximius (Araneae, Theridiidae): Abundance of noveln-propyl esters of long-chain methyl-branched fatty acids. Arch. Insect Biochem. Physiol. 1997, 36, 295–314. [Google Scholar] [CrossRef]
- Chinta, S.P.; Goller, S.; Uhl, G.; Schulz, S. Identification and Synthesis of Branched Wax-type Esters, Novel Surface Lipids from the Spider Argyrodes elevates (Araneae: Theridiidae). Chem. Biodivers. 2016, 13, 1202–1220. [Google Scholar] [CrossRef]
- Gu, P.; Welch, W.H.; Blomquist, G.J. Methyl-branched fatty acid biosynthesis in the German cockroach, Blatella germanica: Kinetic studies comparing a microsomal and soluble fatty acid synthetase. Insect Biochem. Mol. Boil. 1993, 23, 263–271. [Google Scholar] [CrossRef]
- Wang, H.-L.; Brattström, O.; Brakefield, P.M.; Francke, W.; Löfstedt, C. Identification and Biosynthesis of Novel Male Specific Esters in the Wings of the Tropical Butterfly, Bicyclus martius sanaos. J. Chem. Ecol. 2014, 40, 549–559. [Google Scholar] [CrossRef]
- Morgan, E. Biosynthesis in Insects; Royal Society of Chemistry (RSC): Cambridge, UK, 2006. [Google Scholar]
- Gibbs, A.; Pomonis, J. Physical properties of insect cuticular hydrocarbons: The effects of chain length, methyl-branching and unsaturation. Comp. Biochem. Physiol. Part B Biochem. Mol. Boil. 1995, 112, 243–249. [Google Scholar] [CrossRef]
- Foley, B.R.; Telonis-Scott, M. Quantitative genetic analysis suggests causal association between cuticular hydrocarbon composition and desiccation survival in Drosophila melanogaster. Heredity 2010, 106, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Gefen, E.; Talal, S.; Brendzel, O.; Dror, A.; Fishman, A. Variation in quantity and composition of cuticular hydrocarbons in the scorpion Buthus occitanus (Buthidae) in response to acute exposure to desiccation stress. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2015, 182, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Ferveur, J.-F.; Cortot, J.; Rihani, K.; Cobb, M.; Everaerts, C. Desiccation resistance: Effect of cuticular hydrocarbons and water content in Drosophila melanogaster adults. PeerJ 2018, 6, 4318. [Google Scholar] [CrossRef] [PubMed]
- Pike, N.; Wang, W.Y.; Meats, A. The likely fate of hybrids of Bactrocera tryoni and Bactrocera neohumeralis. Heredity 2003, 90, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Popa-Báez, Á.-D.; Catullo, R.; Lee, S.F.; Yeap, H.L.; Mourant, R.G.; Frommer, M.; Sved, J.A.; Cameron, E.C.; Edwards, O.R.; Taylor, P.W.; et al. Genome-wide patterns of differentiation over space and time in the Queensland fruit fly. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Fedina, T.Y.; Kuo, T.-H.; Dreisewerd, K.; Dierick, H.A.; Yew, J.Y.; Pletcher, S.D. Dietary Effects on Cuticular Hydrocarbons and Sexual Attractiveness in Drosophila. PLoS ONE 2012, 7, e49799. [Google Scholar] [CrossRef]
- Otte, T.; Hilker, M.; Geiselhardt, S. The effect of eietary fatty acids on the cuticular hydrocarbon phenotype of an herbivorous insect and consequences for mate recognition. J. Chem. Ecol. 2015, 41, 32–43. [Google Scholar] [CrossRef]
- Otte, T.; Hilker, M.; Geiselhardt, S. Phenotypic Plasticity of Cuticular Hydrocarbon Profiles in Insects. J. Chem. Ecol. 2018, 44, 235–247. [Google Scholar] [CrossRef]
- Drew, R.A.I. The tropical fruit flies (Diptera: Tephritidae: Dacinae) of the Australian and Oceanian regions. Mem Queensl. Mus. 1989, 26, 521. [Google Scholar]
- Pérez-Staples, D.; Prabhu, V.; Taylor, P.W. Post-teneral protein feeding enhances sexual performance of Queensland fruit flies. Physiol. Èntomol. 2007, 32, 225–232. [Google Scholar] [CrossRef]
- Kováts, E. Gas-chromatographische Charakterisierung organischer Verbindungen. Teil 1: Retentionsindices aliphatischer Halogenide, Alkohole, Aldehyde und Ketone. Helvetica Chim. Acta 1958, 41, 1915–1932. [Google Scholar] [CrossRef]
- McCarthy, E.D.; Han, J.; Calvin, M. Hydrogen atom transfer in mass spectrometric fragmentation patterns of saturated aliphatic hydrocarbons. Anal. Chem. 1968, 40, 1475–1480. [Google Scholar] [CrossRef]
- Nelson, D.R.; Sukkestad, D.R. Normal and branched aliphatic hydrocarbons from the eggs of the tobacco hornworm. Biochem. 1970, 9, 4601–4611. [Google Scholar] [CrossRef]
- Nelson, D.R.; Sukkestad, D.R.; Zaylskie, R.G. Mass spectra of methyl-branched hydrocarbons from eggs of the tobacco hornworm. J. Lipid Res. 1972, 13, 413–421. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
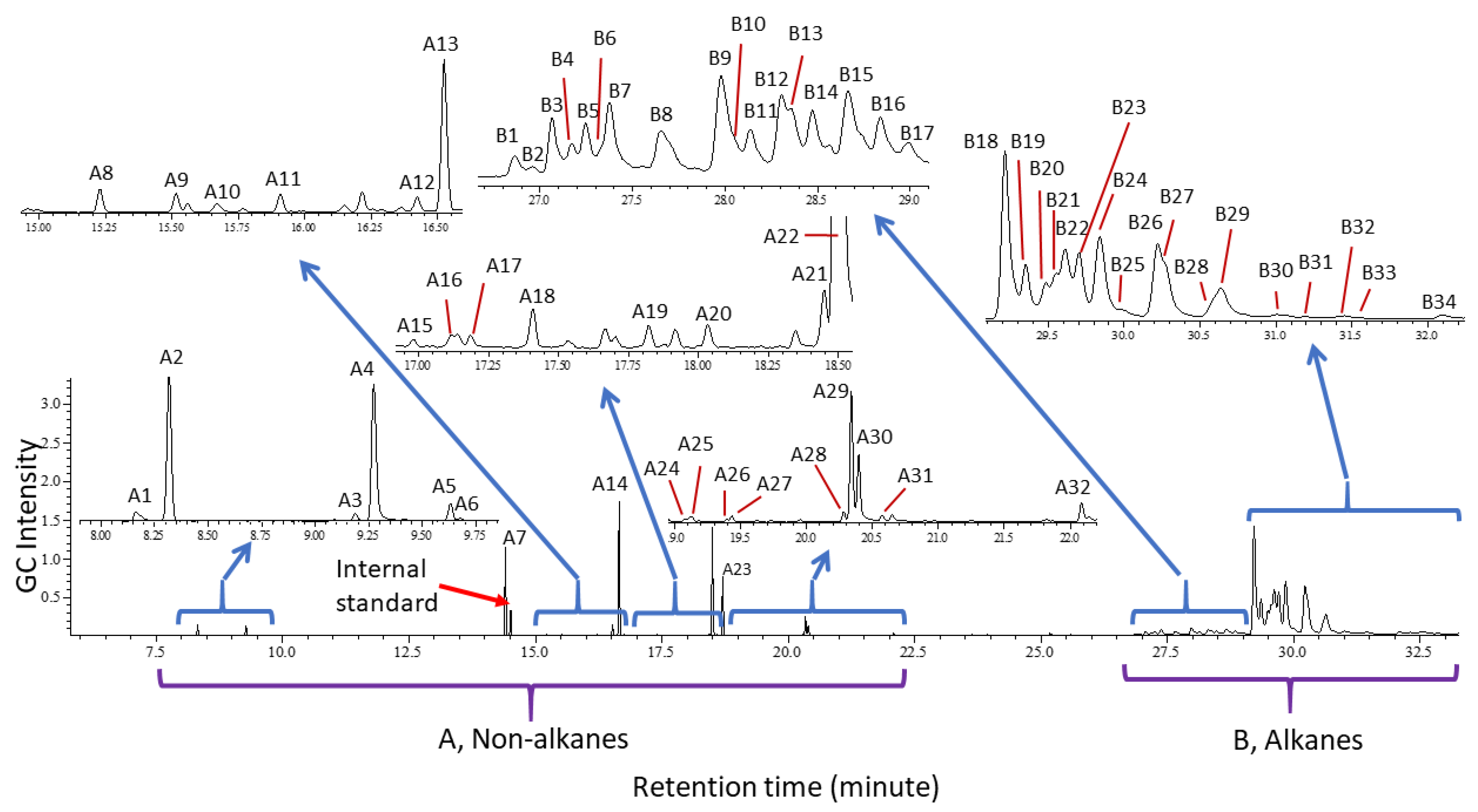
| No | Identity | MM | KI | Ref.KI (Ref) | Characteristic/Diagnostic EI Ions |
|---|---|---|---|---|---|
| A1 * | N-(3-Methylbutyl)acetamide | 129.12 | 1131 | 1137 [78] | 129 (M+), 114, 86, 73, 60 (CH3COHNH2+) |
| A2 | 2,8-Dimethyl-1,7-dioxaspiro[5,5]undecane | 184.15 | 1140 | 1147 [78] | 184 (M+), 140, 115/112 (M-C5H8/C5H8O), 97, 69 |
| A3 * | N-(2-Methylbutyl)propanamide | 143.13 | 1198 | 143 (M+), 114, 86, 74, 57 | |
| A4 * | N-(3-Methylbutyl)propanamide | 143.13 | 1204 | 143 (M+), 128 (M-CH3), 114 (M-C2H5), 100, 87, 74, 57 | |
| A5 # | 2-Ethyl-8-methyl-1,7-dioxaspiro[5,5]undecane | 198.16 | 1230 | 1237 [78] | 198 (M+), 169, 129/126 (M-C5H8/C5H8O), 115/112 (C6H10/C6H10O), 97, 83, 69, 55 |
| A6 * | N-(3-Methylbutyl)isobutyramide | 157.15 | 1233 | 157 (M+), 142, 101, 71, 57 | |
| A7 | Ethyl dodecanoate (ethyl laurate) | 228.38 | 1591 | 1593 [79] | 228 (M+), 183, 157, 115, 101, 88, 73, 70, 60 (CH3CO=OH+) |
| A8 # | Ethyl 6-methyldodecanoate | 242.22 | 1662 | 242 (M+), 213, 199, 185, 157, 143, 101, 88, 83, 70, 55 | |
| A9 | Propyl dodecanoate | 242.22 | 1680 | 1685 [80] | 242 (M+), 201, 183, 157, 143, 115, 102, 61 (C3H7OH2+, base peak) |
| A10 | Ethyl tridecanoate | 242.22 | 1691 | 1695 [81] | 242 (M+), 199, 197, 157, 101, 88 |
| A11 | Methyl tetradecanoate | 242.22 | 1722 | 1724 [82] | 242 (M+), 157, 143, 101, 87, 74 |
| A12 | Ethyl (E)-9-tetradecenoate (ethyl myristolaidate) | 254.22 | 1769 | 254 (M+), 208/209 (loss of EtOH/EtO), 166, 124, 88, 55 | |
| A13 | Ethyl (Z)-9-tetradecenoate (ethyl myristoleate) | 254.22 | 1778 | 254 (M+), 208/209 (loss of EtOH/EtO), 166, 124, 88, 55 | |
| A14 | Ethyl tetradecanoate (ethyl myristate) | 256.43 | 1790 | 1793 [80] | 256 (M+), 213, 157, 101, 88 |
| A15 # | Ethyl 4-methyltetradecanoate | 270.26 | 1836 | 270 (M+), 213, 101 (M-C12H25, base peak), 88 | |
| A16 # | Ethyl 12-methyltetradecanoate | 270.26 | 1862 | 270 (M+), 227, 213, 157, 101, 88 | |
| A17 # | Propyl tetradecanoate | 270.26 | 1887 | 1893 [83] | 270 (M+), 229, 211, 172, 129, 102, 61 (C3H7OH2+, base peak) |
| A18 | Ethyl pentadecanoate | 270.26 | 1890 | 1897 [84] | 270 (M+), 227, 199, 157, 101, 88 |
| A19 | Methyl (Z)-9-hexadecenoate | 268.44 | 1902 | 1909 [78] | 268 (M+), 236/237 (loss of MeOH/MeO), 194, 152, 96, 74, 55 |
| A20 | Methyl hexadecanoate | 270.26 | 1923 | 1927 [85] | 270 (M+), 227, 143, 87, 74 |
| A21 | Ethyl (E)-9-hexadecenoate (ethyl palmitelaidate) | 282.26 | 1965 | 282 (M+), 236/237 (loss of EtOH/EtO), 194, 152, 96, 88, 69, 55 | |
| A22 | Ethyl (Z)-9-hexadecenoate (ethyl plamitoleate) | 282.26 | 1970 | 1975 [86] | 282 (M+), 236/237 (loss of EtOH/EtO), 194, 152, 96, 88, 69, 55 |
| A23 | Ethyl hexadecanoate (ethyl palmitate) | 284.27 | 1990 | 1993 [80] | 284 (M+), 241, 157, 101, 88 |
| A24 # | Ethyl 15-methylhexadecanoate | 298.29 | 2029 | 298 (M+), 255, 157 (M-C10H21), 101, 88 | |
| A25 # | Ethyl 4-methylhexadecanoate | 298.29 | 2035 | 298 (M+), 241 (M-C4H9), 101 (base peak), 88 | |
| A26 # | Ethyl 14-methylhexadecanoate | 298.29 | 2062 | 298 (M+), 269, 255, 241, 199, 157, 101, 88 | |
| A27 # | Propyl 9-hexadecenoate | 296.27 | 2067 | 296 (M+) 281, 237, 194 (M-C3H7COOCH2) | |
| A28 | Ethyl (Z,Z)-octadeca-9,12-dienoate (ethyl linoleate) | 308.27 | 2158 | 2155 [87] | 308 (M+), 262/263 (loss of EtOH/EtO), 178, 135, 95, 81 |
| A29 | Ethyl (Z)-9-octadecenoate (ethyl oleate) | 310.29 | 2168 | 2168 [88] | 310 (M+), 264/265 (loss of EtOH/EtO), 222, 180, 97, 55 |
| A30 | Ethyl (E)-9-octadecenoate (ethyl elaidate) | 310.29 | 2171 | 2174 [89] | 310 (M+), 264/265 (loss of EtOH/EtO), 222, 180, 97, 55 |
| A31 | Ethyl octadecanoate | 312.54 | 2190 | 2191 [90] | 312 (M+), 269, 157, 101, 88 |
| A32 # | Ethyl 11-eicosenoate | 338.57 | 2366 | 338 (M+), 292/293 (M-EtOH/EtO), 250, 208, 97, 55 |
| No | Identity | MM | KI | Ref.KI (Ref) | Characteristic/Diagnostic EI Ions |
|---|---|---|---|---|---|
| B1 | 11-; 13-; 15-MeC29 | 422.82 | 2929 | 2932 [91] | 280/281,168/169; 252/253, 196/197; 224/225(s) |
| B2 | 7-MeC29 | 422.82 | 2946 | 2940 [51] | 336/337, 112/113 |
| B3 | 5-MeC29 | 422.82 | 2952 | 2949 [91] | 364/365, 84/85 |
| B4 | 9,13-DiMeC29 | 436.50 | 2966 | 2963 [91] | 322/323, 140/141, 252/253, 210/211 |
| B5 | 7,11-DiMeC29 | 436.50 | 2970 | 350/351, 112/113, 280/281, 182/183 | |
| B6 | 3-MeC29 | 422.82 | 2976 | 2973 [91] | 392/393, 56/57 |
| B7 | 5,11-DiMeC29; 5,13-DiMeC29 | 436.50 | 2986 | 2983 [91] | 378/379, 84/85,280/281, 182/183; 378/379, 84/85, 280/281, 210/211 |
| B8 | 4, x, 22-TriMeC29 (x = 14 or 16) | 450.52 | 3009 | 392/393, 84/85, 252/253, 224/225, 336/337, 126/127 | |
| B9 | 12-Me; 14-MeC30 | 436.50 | 3025 | 3031 [92] | 280/281,182/183; 252/253, 210/211 |
| B10 | 8-MeC30 | 436.50 | 3034 | 3040 [93] | 336/337, 126/127 |
| B11 | 6-MeC30 | 436.50 | 3041 | 3045 [93] | 364/365, 98/99 |
| B12 | 4-MeC30 | 436.50 | 3055 | 3065 [93] | 392/393, 70/71 |
| B13 | 8,12-DiMeC30; 8,14-DiMeC30 | 450.52 | 3061 | 3064 [94] | 350/351, 126/127, 280/281, 196/197; 350/351, 126/127, 253/252, 225.224 |
| B14 | 6,14-DiMeC30; 6,12-DiMeC30 | 450.50 | 3071 | 378/379, 98/99, 252/253, 224/225; 378/379, 98/99, 280/281, 196/197 | |
| B15 | 4,12-DiMeC30; 4,14-DiMeC30; 4,20-DiMeC30 | 450.52 | 3088 | 3098 [94] | 406/407, 70/71, 280/281, 196/197; 406/407, 70/71, 225/224, 253/252; 406/407, 70/71, 309/308, 169/168 |
| B16 | n-C31 | 436.50 | 3100 | 436 | |
| B17 | 4,8,12-TriMeC30; 4,8,14-TriMeC30; 4,8,20-TriMeC30 | 464.53 | 3115 | 70/71, 420/421, 350/351, 140/141, 280/291, 210/211; 70/71, 420/421, 350/351, 140/141, 252/253, 238/239; 70/71, 420/421, 350/351, 140/141, 322/323, 168/169 | |
| B18 | 11-; 13-; 15-MeC31 | 450.52 | 3129 | 3130 [92] | 308/309, 168/169; 280/281, 196/197; 250/251, 224/225 |
| B19 | 7-MeC31; 9-MeC31 | 450.52 | 3137 | 3140 [93] | 364/365, 112/113; 336/337, 141/140 |
| B20 | 5-MeC31 | 450.52 | 3147 | 3150 [93] | 392/393, 84/85 |
| B21 | 11,15-DiMeC31 | 464.53 | 3153 | 3155 [93] | 322/323, 168/169, 252/253, 238/239 |
| B22 | 9,13-DiMeC31; 9,15-DiMeC31; 11,13-DiMeC31; 13,15-DiMeC31 | 464.53 | 3157 | 3159 [92] | 350/351, 140/141, 280/281, 210/211; 350/351, 140/141, 252/253, 238/239; 322/323, 168/169, 280/281, 210/211; 294/295, 196/197, 252/253, 238/239 |
| B23 | 7,13-DiMeC31;7,15-DiMeC31 | 464.53 | 3164 | 378/379, 112/113, 280/281, 210/211; 378/379, 112/113, 252/253, 238/239 | |
| B24 | 3-MeC31 | 450.52 | 3170 | 3172 [91] | 420/421, 56/57 |
| B25 | 5,11-DiMeC31; 5,13-DiMeC31 | 464.53 | 3178 | 3180 [95] | 406/407, 84/85, 182/183, 308/309; 406/407, 84/85, 280/281, 210/211 |
| B26 | 12-; 14-; 16-MeC32 | 464.53 | 3223 | 3225 [57] | 308/309, 182/183; 280/281, 210/211; 252/253, 238/239 |
| B27 | 8-; 10-MeC32 | 464.53 | 3225 | 3225 [57] | 364/365, 126/127; 336/337, 154/155 |
| B28 | 10,14-DiMeC32 | 478.55 | 3233 | 3254 [94] | 350/351, 154/155, 280/281, 224/225 |
| B29 | 8,12-DiMeC32; 8,14-DiMeC32; 8,16-DiMeC32 | 478.55 | 3257 | 3263 [96] | 378/379, 126/127, 308/309, 196/197; 378/379, 126/127, 280/281, 224/225; 378/379, 126/127, 252/253 (s) |
| B30 | 4-MeC32 | 464.53 | 3267 | 3265 [97] | 420/421, 70/71 |
| B31 | 6,16-DiMeC32 | 478.55 | 3285 | 406/407, 98/99, 252/253 | |
| B32 | 9-; 11-MeC33 | 478.55 | 3327 | 3335 [93] | 364/365, 140/141; 336/337, 168/169 |
| B33 | 13-; 15-; 17-MeC33 | 478.55 | 3330 | 3335 [93] | 308/309, 196/197; 280/281, 224/225; 252/253 (s) |
| B34 | 9,23-DiMeC33 | 492.56 | 3359 | 378/379, 140/141, 350/351, 168/169 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.J.; Pandey, G.; Castro-Vargas, C.; Oakeshott, J.G.; Taylor, P.W.; Mendez, V. Cuticular Chemistry of the Queensland Fruit Fly Bactrocera tryoni (Froggatt). Molecules 2020, 25, 4185. https://doi.org/10.3390/molecules25184185
Park SJ, Pandey G, Castro-Vargas C, Oakeshott JG, Taylor PW, Mendez V. Cuticular Chemistry of the Queensland Fruit Fly Bactrocera tryoni (Froggatt). Molecules. 2020; 25(18):4185. https://doi.org/10.3390/molecules25184185
Chicago/Turabian StylePark, Soo J., Gunjan Pandey, Cynthia Castro-Vargas, John G. Oakeshott, Phillip W. Taylor, and Vivian Mendez. 2020. "Cuticular Chemistry of the Queensland Fruit Fly Bactrocera tryoni (Froggatt)" Molecules 25, no. 18: 4185. https://doi.org/10.3390/molecules25184185
APA StylePark, S. J., Pandey, G., Castro-Vargas, C., Oakeshott, J. G., Taylor, P. W., & Mendez, V. (2020). Cuticular Chemistry of the Queensland Fruit Fly Bactrocera tryoni (Froggatt). Molecules, 25(18), 4185. https://doi.org/10.3390/molecules25184185