Hyaluronic Acid as a Component of Natural Polymer Blends for Biomedical Applications: A Review
Abstract
1. Introduction
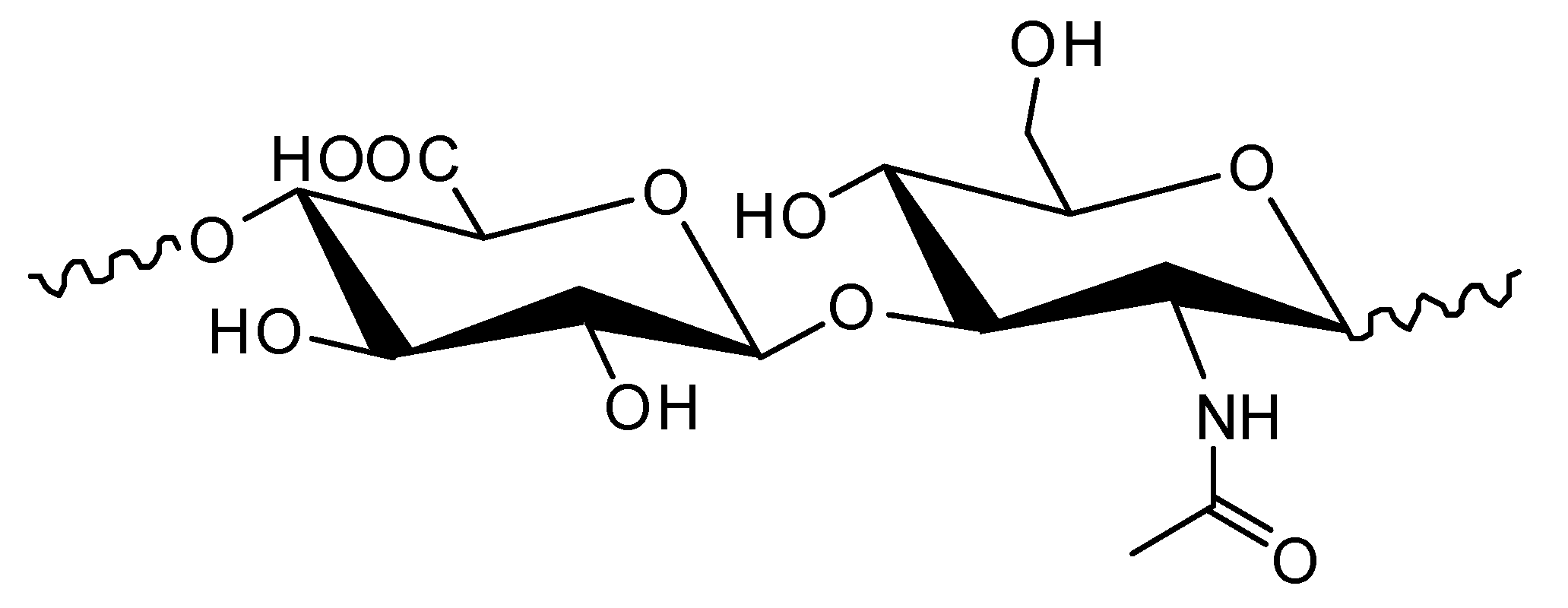
2. Blends of Hyaluronic Acid and Collagen
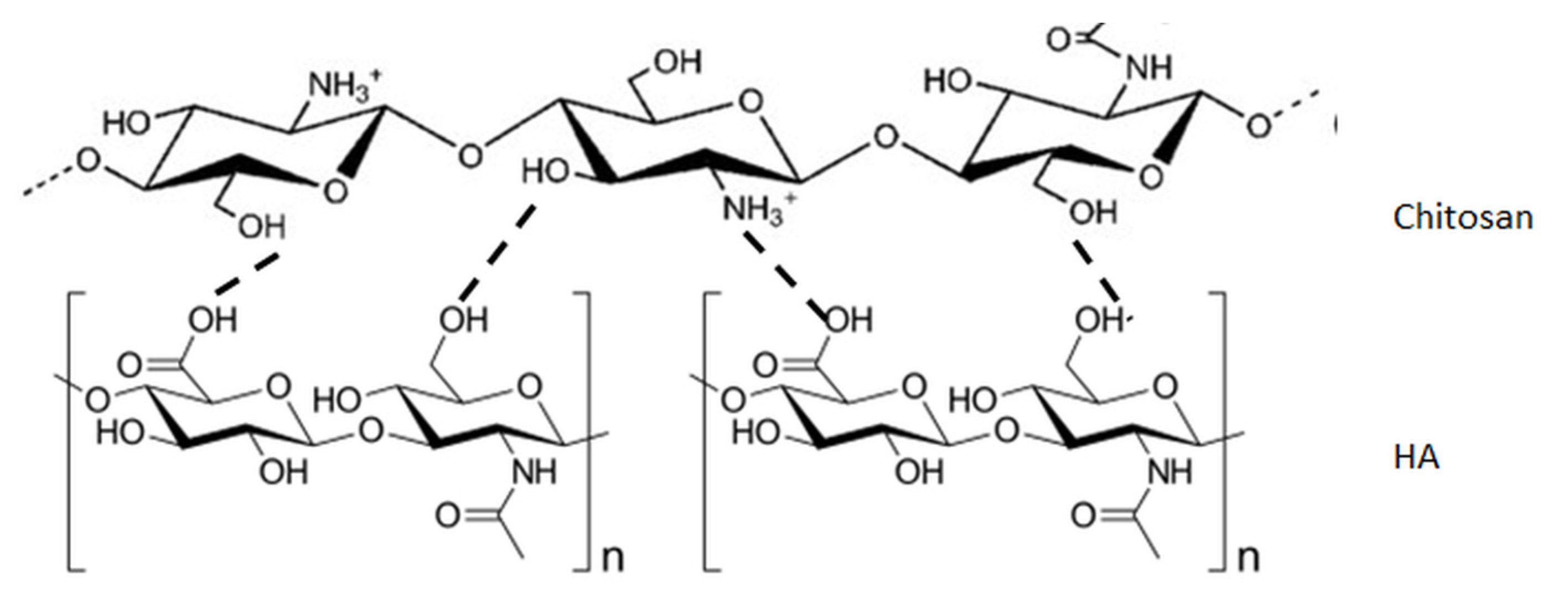
3. Blends of Hyaluronic Acid and Chitosan
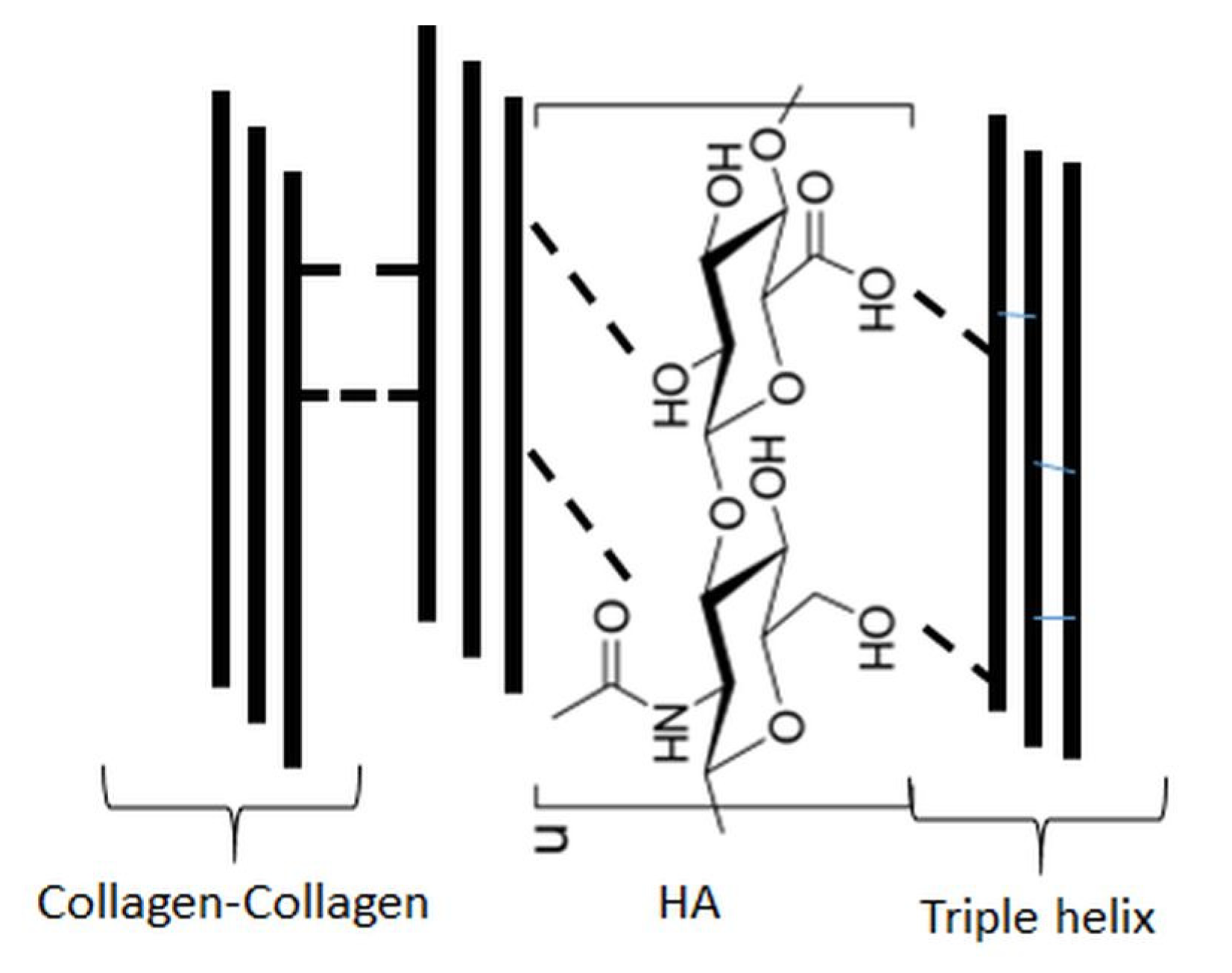
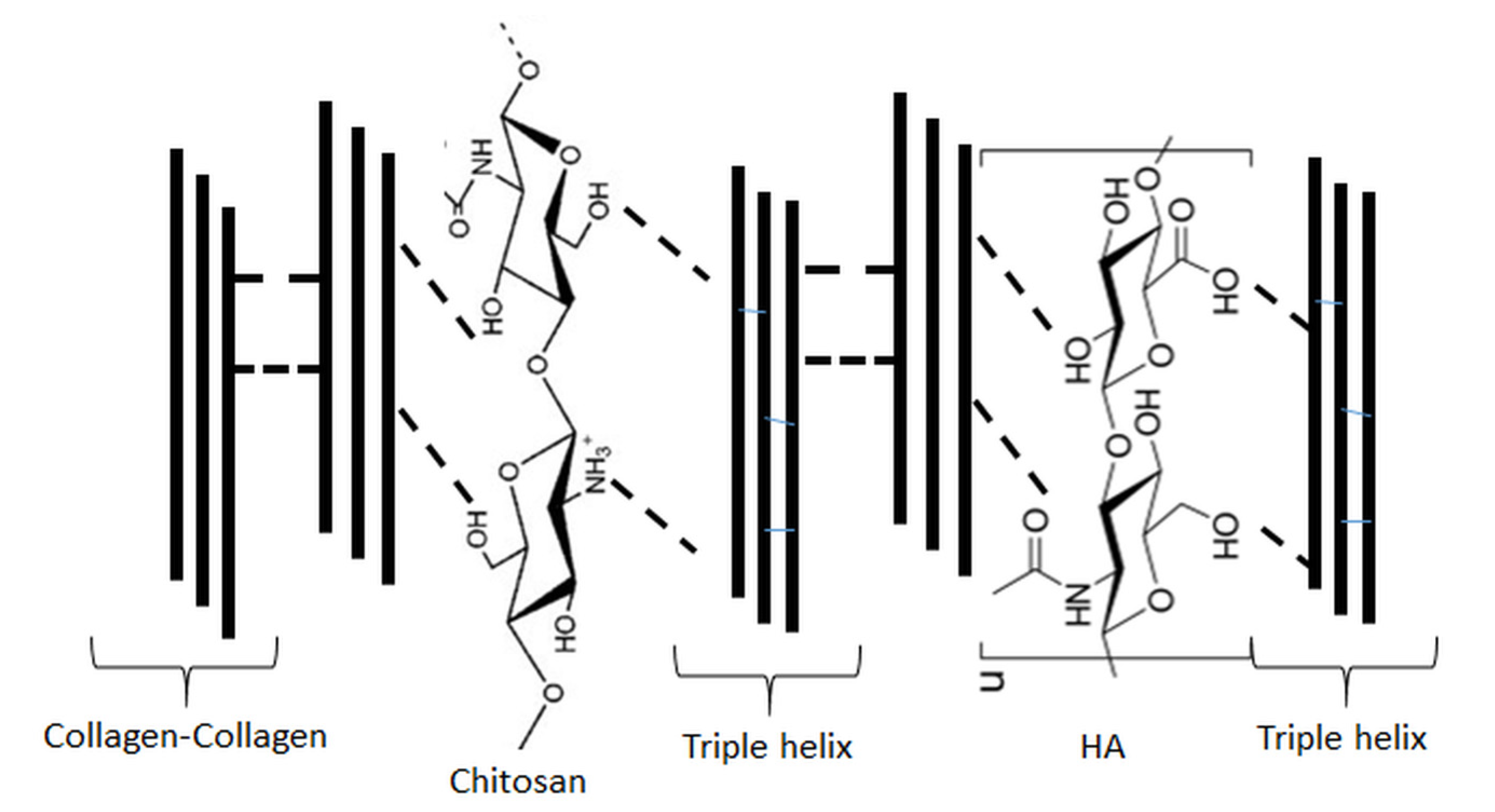
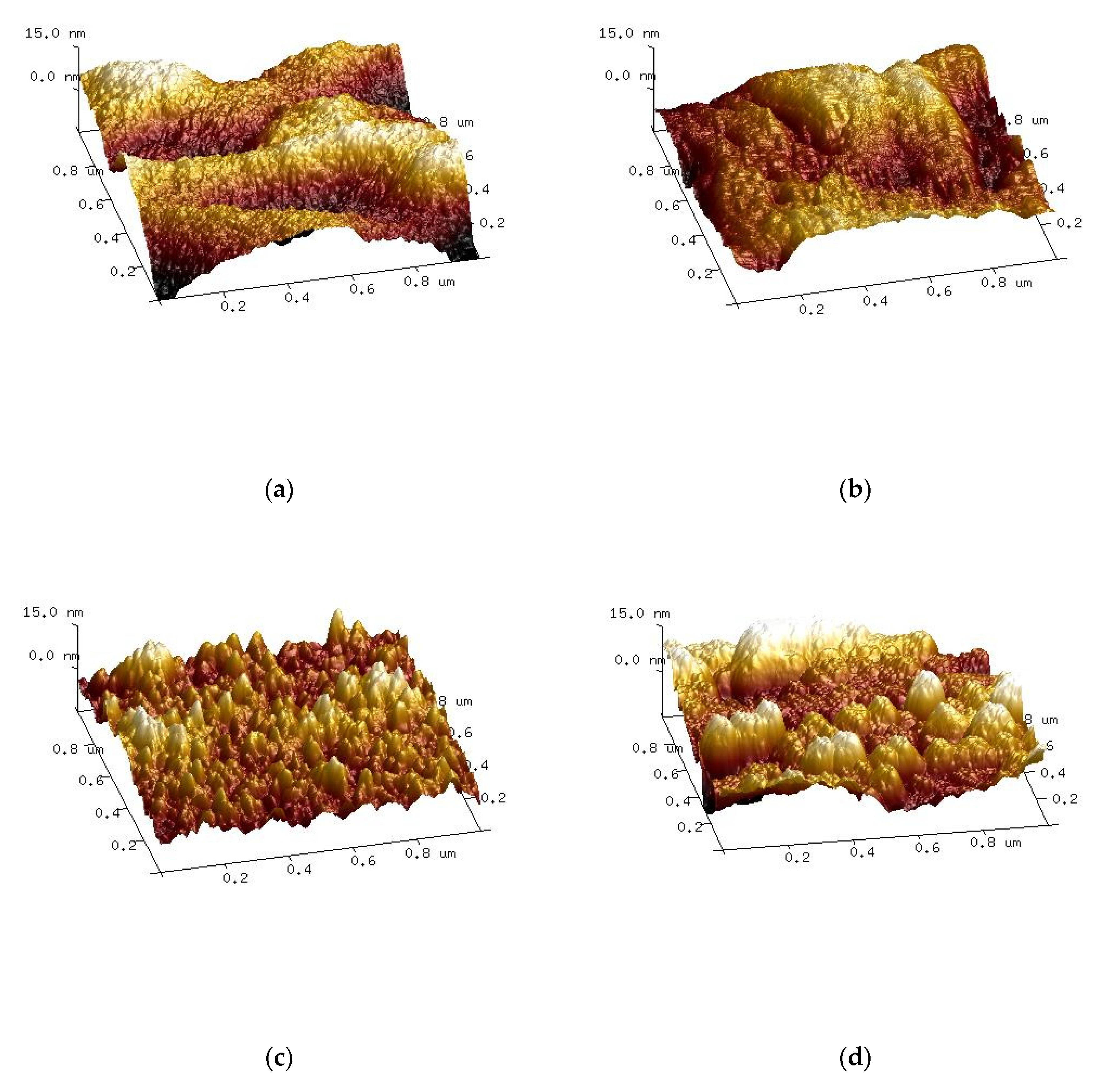
4. Ternary Blends of Hyaluronic Acid, Collagen, and Chitosan
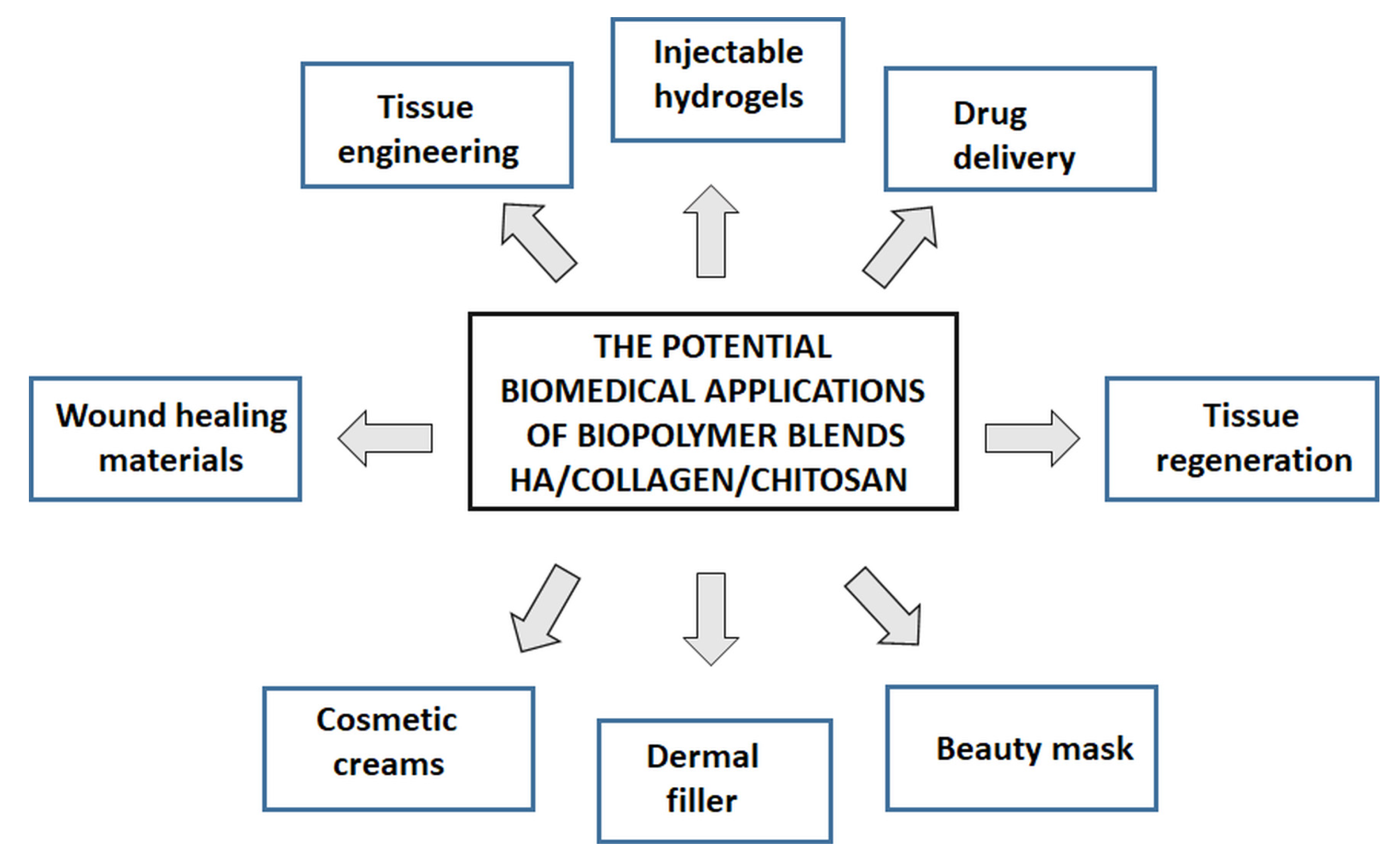
5. Possible Application of New Materials Based on the Blends of Hyaluronic Acid with Other Natural Polymers
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Schante, C.; Zuber, G.; Herlin, C.; Vandamme, T. Chemical modifications of hyaluronic acid for the synthesis of derivatives for a broad range of biomedical applications. Carbohydr. Polym. 2011, 85, 469–489. [Google Scholar] [CrossRef]
- Vasvani, S.; Kulkarni, P.; Rawtani, D. Hyaluronic acid: A reviewon its biology, aspects of drug delivery, route ofadministrations and a special emphasis on its approved marketedproducts and recent clinical studies. Int. J. Biol. Macromol. 2020, 151, 1012–1029. [Google Scholar] [CrossRef]
- Al-Sibani, M.; Al-Harrasi, A.; Neubert, R.H.H. Effect of hyaluronic acid initial concentration on cross-linking efficiency of hyaluronic acid-based hydrogels used in biomedical and cosmetic applications. Pharmazie 2017, 72, 81–86. [Google Scholar]
- Luo, Y.; Kirker, K.R.; Prestwich, G.D. Cross-linked hyaluronic acid hydrogel films: New biomaterials for drug delivery. J. Control. Release 2008, 69, 169–184. [Google Scholar] [CrossRef]
- Berko, S.; Maroda, M.; Bondar, M.; Eros, G.; Hartmann, P.; Szentner, K.; Szabo-Revesz, P.; Kemeny, L.; Borbely, J.; Csanyi, E. Advantages of cross-linked versus linear hyaluronic acid for semisolid skin delivery systems. Eur. Polym. J. 2013, 49, 2511–2517. [Google Scholar] [CrossRef]
- Masters, K.S.; Shah, D.N.; Leinwand, L.A.; Anseth, K.S. Crosslinked hyaluronan scaffolds as a biologically active carrier for valvular interstitial cells. Biomaterials 2005, 26, 2517–2525. [Google Scholar] [CrossRef]
- Kim, I.L.; Mauck, R.L.; Burdick, J.A. Hydrogel design for cartilage tissue engineering: A case study with hyaluronic acid. Biomaterials 2011, 32, 8771–8782. [Google Scholar] [CrossRef] [PubMed]
- Bukhari, S.N.; Roswandi, N.L.; Waqas, M.; Habib, H.; Hussain, F.; Khan, S.; Sohail, M.; Ramli, N.A.; Thu, H.E.; Hussain, Z. Hyaluronic acid, a promising skin rejuvenating biomedicine: A review of recent updates and pre-clinical and clinical investigations on cosmetic and nutricosmetic effects. Int. J. Biol. Macromol. 2018, 120, 1682–1695. [Google Scholar] [CrossRef] [PubMed]
- Ievdokimova, N.I. Hyaluronic acid, receptor CD44, and their role in diabetic complications. Ukr. Kyi Biokhimichnyi Zhurnal 2008, 80, 5–44. [Google Scholar]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinol 2012, 4, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Narurkar, V.A.; Fabi, S.G.; Bucay, V.W.; Tedaldi, R.; Downie, J.B.; Zeichner, J.A.; Butterwick, K.; Taub, A.; Kadoya, K.; Makino, E.T.; et al. Rejuvenating hydrator: Restoring epidermal hyaluronic acid homeostasis with instant benefits. J. Drugs Dermatol. 2016, 15, 24–37. [Google Scholar]
- Pavicic, T.; Gauglitz, G.G.; Lersch, P.; Schwach-Abdellaoui, K.; Malle, B.; Korting, H.C.; Farwick, M. Efficacy of cream-based novel formulations of hyaluronic acid of different molecular weights in anti-wrinkle treatment. J. Drugs Dermatol. 2011, 10, 990–1000. [Google Scholar] [PubMed]
- Brandt, F.; Bassichis, B.; Bassichis, M.; O’Connell, C.; Lin, X. Safety and effectiveness of small and large gel-particle hyaluronic acid in the correction of perioral wrinkles. J. Drugs Dermatol. 2011, 10, 982–987. [Google Scholar] [PubMed]
- Rzany, B.; Cartier, H.; Kestemont, P.; Trevidic, P.; Sattler, G.; Kerrouche, N.; Dhuin, J.C.; Ma, M.Y. Full-face rejuvenation using a range of hyaluronic acid fillers: Efficacy, safety, and patient satisfaction over 6 months. Dermatol. Surg. 2012, 38, 1153–1161. [Google Scholar] [CrossRef]
- Streit, M.; Brand, C.U.; Braathen, L.R. Soft tissue augmentation for treatment of wrinkles and scars of the face. Ther. Umsch. 1999, 56, 212–218. [Google Scholar] [CrossRef]
- Sionkowska, A. The potential of polymers from naturals sources as components of the blends for biomedical and cosmetic applications. Pure Appl. Chem. 2015, 87, 1075–1084. [Google Scholar] [CrossRef]
- Xie, H.; Chen, X.; Shen, X.; He, Y.; Chen, W.; Luo, Q.; Ge, W.; Yuan, W.; Tang, X.; Hou, D.; et al. Preparation of chitosan-collagen-alginate composite dressing and its promoting effects on wound healing. Int. J. Biol. Macromol. 2018, 107, 93–104. [Google Scholar] [CrossRef]
- Vieira, S.; Strymecka, P.; Stanaszek, L.; Silva-Correia, J.; Drela, K.; Fiedorowicz, M.; Malysz-Cymborska, I.; Rogujski, P.; Janowski, M.; Reis, R.L.; et al. Methacrylated gellan gum and hyaluronic acid hydrogel blends for image-guided neurointerventions. J. Mat. Chem. B 2020, 8, 5928–5937. [Google Scholar] [CrossRef]
- Ronca, A.; D’Amora, U.; Raucci, M.G.; Lin, H.; Fan, Y.J.; Zhang, X.D.; Ambrosio, L. A Combined Approach of Double Network Hydrogel and Nanocomposites Based on Hyaluronic Acid and Poly (ethylene glycol) Diacrylate Blend. Materials 2018, 11, 2454. [Google Scholar] [CrossRef]
- Pokorny, M.; Rassushin, V.; Wolfova, L.; Velebny, V. Increased Production of Nanofibrous Materials by Electroblowing from Blends of Hyaluronic Acid and Polyethylene Oxide. Polym. Eng. Sci. 2016, 56, 932–938. [Google Scholar] [CrossRef]
- Sionkowska, A.; Skrzyński, S.; Śmiechowski, K.; Kołodziejczak, A. The review of versatile application of collagen. Polym. Adv. Technol. 2017, 28, 4–9. [Google Scholar] [CrossRef]
- Taguchi, T.; Tanaka, J. Swelling behavior of hyaluronic acid and type II collagen hydrogels prepared by using conventional crosslinking and subsequent additional polymer interactions. J. Biomater. Sci. Polym. Ed. 2002, 13, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Chung, E.J.; Jakus, A.E.; Shah, R.N. In situ forming collagen-hyaluronic acid membrane structures: Mechanism of self-assembly and applications in regenerative medicine. Acta Biomater. 2013, 9, 5153–5161. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, T.; Ikoma, T.; Tanaka, J. An improved method to prepare hyaluronic acid and type II collagen composite matrices. J. Biomed. Mater. Res. 2002, 61, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Park, S.N.; Lee, H.J.; Lee, K.H.; Suh, H. Biological characterization of EDC-crosslinked collagen-hyaluronic acid matrix in dermal tissue restoration. Biomaterials 2003, 24, 1631–1641. [Google Scholar] [CrossRef]
- Lin, Y.K.; Liu, D.C. Studies of novel hyaluronic acid-collagen sponge materials composed of two different species of type I collagen. J. Biomater. Appl. 2007, 21, 265–281. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, S.Y.; Lee, Y.M. Preparation of porous collagen/hyaluronic acid hybrid scaffolds for biomimetic functionalization through biochemical binding affinity. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82B, 506–518. [Google Scholar] [CrossRef]
- Fujie, T.; Furutate, S.; Niwa, D.; Takeoka, S. A nano-fibrous assembly of collagen-hyaluronic acid for controlling cell-adhesive properties. Soft Matter 2010, 6, 4672–4676. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, K.K.; Park, I.K.; Choi, B.S.; Kim, J.H.; Kim, M.S. Hybrid scaffolds composed of hyaluronic acid and collagen for cartilage regeneration. Tissue Eng. Regen. Med. 2012, 9, 57–62. [Google Scholar]
- Liu, Y.; Fan, D. Novel hyaluronic acid-tyrosine/collagen-based injectable hydrogels as soft filler for tissue engineering. Int. J. Biol. Macromol. 2019, 141, 700–712. [Google Scholar] [CrossRef]
- Ying, H.Y.; Zhou, J.; Wang, M.Y.; Su, D.D.; Ma, Q.Q.; Lv, G.Z.; Chen, J.H. In situ formed collagen-hyaluronic acid hydrogel as biomimetic dressing for promoting spontaneous wound healing. Mater. Sci. Eng. C-Mater. Biol. Appl. 2019, 101, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Ma, X.X.; Fan, D.D.; Zhu, C.H.; Deng, J.J.; Hui, J.F.; Ma, P. Synthesis and characterization of hyaluronic acid/human-like collagen hydrogels. Mater. Sci. Eng. C-Mater. Biol. Appl. 2014, 43, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Segura, T.; Chung, P.H.; Shea, L.D. DNA delivery from hyaluronic acid-collagen hydrogels via a substrate-mediated approach. Biomaterials 2005, 26, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Mineo, A.; Suzuki, R.; Kuroyanagi, Y. Development of an artificial dermis composed of hyaluronic acid and collagen. J. Biomater. Sci. Polym. Ed. 2013, 24, 726–740. [Google Scholar] [CrossRef]
- Kondo, S.; Niiyama, H.; Yu, A.; Kuroyanagi, Y. Evaluation of a wound dressing composed of hyaluronic acid and collagen sponge containing epidermal growth factor in diabetic mice. J. Biomater. Sci. Polym. Ed. 2012, 23, 1729–1740. [Google Scholar] [CrossRef]
- Kondo, S.; Kuroyanagi, Y. Development of a wound dressing composed of hyaluronic acid and collagen sponge with epidermal growth factor. J. Biomater. Sci. Polym. Ed. 2012, 23, 629–643. [Google Scholar] [CrossRef]
- Yu, A.; Niiyama, H.; Kondo, S.; Yamamoto, A.; Suzuki, R.; Kuroyanagi, Y. Wound dressing composed of hyaluronic acid and collagen containing EGF or bFGF: Comparative culture study. J. Biomater. Sci. Polym. Ed. 2013, 24, 1015–1026. [Google Scholar] [CrossRef]
- Kim, Z.H.; Lee, Y.; Kim, S.M.; Kim, H.; Yun, C.K.; Choi, Y.S. A composite dermal filler comprising cross-linked hyaluronic acid and human collagen for tissue reconstruction. J. Microbiol. Biotechnol. 2015, 25, 399–406. [Google Scholar] [CrossRef]
- Kim, T.G.; Chung, H.J.; Park, T.G. Macroporous and nanofibrous hyaluronic acid/collagen hybrid scaffold fabricated by concurrent electrospinning and deposition/leaching of salt particles. Acta Biomater. 2008, 4, 1611–1619. [Google Scholar] [CrossRef]
- Fischer, R.L.; McCoy, M.G.; Grant, S.A. Electrospinning collagen and hyaluronic acid nanofiber meshes. J. Mater. Sci. Mater. Med. 2012, 23, 1645–1654. [Google Scholar] [CrossRef]
- Nazir, R. Collagen—hyaluronic acid based interpenetrating polymer networks as tissue engineered heart valve. Mater. Sci. Technol. 2016, 32, 871–882. [Google Scholar] [CrossRef]
- Crini, G. Historical review on chitin and chitosan biopolymers. Environ. Chem. Lett. 2019, 17, 1623–1643. [Google Scholar] [CrossRef]
- Lewandowska, K.; Sionkowska, A.; Grabska, S. Chitosan blends containing hyaluronic acid and collagen. Compatibility behaviour. J. Mol. Liq. 2015, 212, 879–884. [Google Scholar] [CrossRef]
- Lewandowska, K.; Sionkowska, A.; Grabska, S. The influence of the type solvent on the structure of chitosan blends with hyaluronic acid. PCACD 2016, 21, 147–153. [Google Scholar] [CrossRef]
- Lalevee, G.; Sudre, G.; Montembault, A.; Meadows, J.; Malaise, S.; Crepet, A.; David, L.; Delair, T. Polyelectrolyte complexes via desalting mixtures of hyaluronic acid and chitosan-Physicochemical study and structural analysis. Carbohydr. Polym. 2016, 154, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Kayitmazer, A.B.; Koksal, A.F.; Iyilik, E.K. Complex coacervation of hyaluronic acid and chitosan: Effects of pH, ionic strength, charge density, chain length and the charge ratio. Soft Matter. 2015, 11, 8605–8612. [Google Scholar] [CrossRef]
- Monteiro do Nascimento, M.H.; Lombello, C.B. Hyaluronic acid and chitosan based hydrogels for cartilage tissue engineering. Polim. Cienc. Tecnol. 2016, 26, 360–370. [Google Scholar]
- Park, H.; Choi, B.; Hu, J.L.; Lee, M. Injectable chitosan hyaluronic acid hydrogels for cartilage tissue engineering. Acta Biomater. 2013, 9, 4779–4786. [Google Scholar] [CrossRef]
- Neethu, M.; Mohanan, P.V.; Sabareeswaran, A.; Prabha, N. Chitosan-hyaluronic acid hydrogel for cartilage repair. Int. J. Biol. Macromol. 2017, 104, 1936–1945. [Google Scholar]
- Acar, O.K.; Kayitmazer, A.B.; Kose, G.T. Hyaluronic acid/chitosan coacervate-based scaffolds. Biomacromolecules 2018, 19, 1198–1211. [Google Scholar] [CrossRef]
- Zhang, W.; Jin, X.; Li, H.; Zhang, R.R.; Wu, C.W. Injectable and body temperature sensitive hydrogels based on chitosan and hyaluronic acid for pH sensitive drug release. Carbohydr. Polym. 2018, 186, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Valverde, A.; Perez-Alvarez, L.; Ruiz-Rubio, L.; Olivenza, M.A.P.; Blanco, M.B.G.; Diaz-Fuentes, M.; Vilas-Vilela, J.L. Antibacterial hyaluronic acid/chitosan multilayers onto smooth and micropatterned titanium surfaces. Carbohydr. Polym. 2019, 207, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Vigani, B.; Rossi, S.; Sandri, G.; Bonferoni, M.C.; Caramella, C.M.; Ferrari, F. Hyaluronic acid and chitosan-based nanosystems: A new dressing generation for wound care. Expert Opin. Drug Deliv. 2019, 16, 715–740. [Google Scholar] [CrossRef] [PubMed]
- Bazmandeh, A.Z.; Mirzaei, E.; Ghasemi, Y.; Kouhbanani, M.A.J. Hyaluronic acid coated electrospun chitosan-based nanofibers prepared by simultaneous stabilizing and coating. Int. J. Biol. Macromol. 2019, 138, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.F.; Perry, S.L.; Schiffman, J.D. Electrospinning Nanofibers from Chitosan/Hyaluronic Acid Complex Coacervates. Biomacromolecules 2019, 20, 4191–4198. [Google Scholar] [CrossRef]
- Lee, E.J.; Kang, E.; Kang, S.W.; Huh, K.M. Thermo-irreversible glycol chitosan/hyaluronic acid blend hydrogel for injectable tissue engineering. Carbohydr. Polym. 2020, 244, 116432. [Google Scholar] [CrossRef]
- Engkagul, V.; Sereemaspun, A.; Chirachanchai, S. One pot preparation of chitosan/hyaluronic acid-based triple network hydrogel via in situ click reaction, metal coordination and polyion complexation in water. Carbohydr. Polym. 2018, 200, 616–623. [Google Scholar] [CrossRef]
- Erickson, A.E.; Levengood, S.K.L.; Sun, J.L.; Chang, F.C.; Zhang, M.Q. Fabrication and Characterization of Chitosan-Hyaluronic Acid Scaffolds with Varying Stiffness for Glioblastoma Cell Culture. Adv. Healthc. Mater. 2018, 7, 1800295. [Google Scholar] [CrossRef]
- Xu, H.T.; Ma, L.; Shi, H.F.; Gao, C.Y.; Han, C.M. Chitosan-hyaluronic acid hybrid film as a novel wound dressing: In vitro and in vivo studies. Polym. Adv. Technol. 2007, 18, 869–875. [Google Scholar] [CrossRef]
- Iacob, A.T.; Dragan, M.; Ghetu, N.; Pieptu, D.; Vasile, C.; Buron, F.; Routier, S.; Giusca, S.E.; Caruntu, I.D.; Profire, L. Preparation, Characterization and Wound Healing Effects of New Membranes Based on Chitosan, Hyaluronic Acid and Arginine Derivatives. Polymers 2018, 10, 607. [Google Scholar] [CrossRef]
- Mitura, S.; Sionkowska, A.; Jaiswal, A. Biopolymers for hydrogels in cosmetics: Review. J. Mater. Sci. Mater. Med. 2020, 31, 50. [Google Scholar] [CrossRef] [PubMed]
- Lewandowska, K.; Sionkowska, A.; Grabska, S.; Kaczmarek, B.; Michalska, M. The miscibility of collagen/hyaluronic acid/chitosan blends investigated in dilute solutions and solids. J. Mol. Liq. 2016, 220, 726–730. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Markiewicz, E. L-ascorbic acid release from polymeric matrixes based on blends of chitosan, collagen and hyaluronic acid. Mol. Cryst. Liq. Cryst. 2016, 640, 46–53. [Google Scholar]
- Sionkowska, A.; Lewandowska, K.; Grabska, S.; Kaczmarek, B.; Michalska, M. Physico-chemical properties of three-component mixtures based on chitosan, hyaluronic acid and collagen. Mol. Cryst. Liq. Cryst. 2016, 640, 21–29. [Google Scholar] [CrossRef]
- Lewandowska, K.; Sionkowska, A.; Grabska, S.; Kaczmarek, B. Surface and thermal properties of collagen/hyaluronic acid blends containing chitosan. Int. J. Biol. Macromol. 2016, 92, 371–376. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Michalska, M.; Lewandowska, K.; Grabska, S. Preparation and characterization of collagen/chitosan/hyaluronic acid thin films for application in hair care cosmetics. Pure Appl. Chem. 2017, 89, 1829–1839. [Google Scholar] [CrossRef]
- Sionkowska, A.; Michalska-Sionkowska, M.; Walczak, M. Preparation and characterization of collagen/hyaluronic acid/chitosan film crosslinked with dialdehyde starch. Int. J. Biol. Macromol. 2020, 149, 290–295. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Lewandowska, K.; Grabska, S.; Pokrywcznska, M.; Kloskowski, T.; Drewa, T. 3D composites based on the blends of chitosan and collagen with the addition of hyaluronic acid. Int. J. Biol. Macromol. 2016, 89, 442–448. [Google Scholar] [CrossRef]
- Kaczmarek, B.; Sionkowska, A.; Kozlowska, J.; Osyczka, A.M. New composite materials prepared by calcium phosphate precipitation in chitosan/collagen/hyaluronic acid sponge cross-linked by EDC/NHS. Int. J. Biol. Maacromol. 2018, 107, 247–253. [Google Scholar] [CrossRef]
- Kaczmarek, B.; Sionkowska, A.; Osyczka, A.M. The application of chitosan/collagen/hyaluronic acid sponge cross-linked by dialdehyde starch addition as a matrix for calcium phosphate in situ precipitation. Int. J. Biol. Maacromol. 2018, 107, 470–477. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B. Preparation and characterization of composites based on the blends of collagen, chitosan and hyaluronic acid with nano-hydroxyapatite. Int. J. Biol. Macromol. 2017, 102, 658–666. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, B.; Sionkowska, A.; Osyczka, A.M. The comparison of physic-chemical properties of chitosan/collagen/hyaluronic acid composites with nano-hydroxyapatite cross-linked by dialdehyde starch and tannic acid. Polym. Test. 2017, 62, 171–176. [Google Scholar] [CrossRef]
- Lewandowska-Lancucka, J.; Gilarska, A.; Bula, A.; Horak, W.; Latkiewicz, A.; Nowakowska, M. Genipin crosslinked bioactive collagen/chitosan/hyaluronic acid injectable hydrogels structurally amended via covalent attachment of surface-modified silica particles. Int. J. Biol. Maacromol. 2019, 136, 1196–1208. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, B.; Sionkowska, A.; Golynska, M.; Polkowska, I.; Szponder, T.; Nehrbass, D.; Osyczka, A.M. In vivo study on scaffolds based on chitosan, collagen, and hyaluronic acid with hydroxyapatite. Int. J. Biol. Macromol. 2018, 118, 938–944. [Google Scholar] [CrossRef] [PubMed]
- Michalska-Sionkowska, M.; Kaczmarek, B.; Walczak, M.; Sionkowska, A. Antimicrobial activity of new materials based on the blends of collagen/chitosan/hyaluronic acid with gentamicin sulfate addition. Mater Sci. Eng. C Mater Biol. Appl. 2018, 86, 103–108. [Google Scholar] [CrossRef]
- Hu, Y.; Wu, Y.; Cai, J.; Ma, S.; Wang, X. The procoagulant properties of hyaluronic acid-collagen (i)/chitosan complex film. J. Biomater. Sci. Polym. Ed. 2009, 20, 1111–1118. [Google Scholar] [CrossRef]
- Wu, Y.; Hu, Y.; Cai, J.; Ma, S.; Wang, X. Coagulation property of hyaluronic acid –collagen/chitosan complex film. J. Mater. Sci. Mater. Med. 2008, 19, 3621–3629. [Google Scholar] [CrossRef]
- Sionkowska, A.; Grabska, S. Incorporation of magnetite particles in 3D matrices made from the blends of collagen, chitosan, and hyaluronic acid. Adv. Polym. Technol. 2018, 37, 2905–2914. [Google Scholar] [CrossRef]
- Highley, C.B.; Prestwich, G.D.; Burdick, J.A. Recent advances in hyaluronic acid hydrogels for biomedical applications. Curr. Opin. Biotechnol. 2016, 40, 35–40. [Google Scholar] [CrossRef]
- Shaharudin, A.; Aziz, Z. Effectiveness of hyaluronic acid and its derivatives on chronic wounds: A systematic review. J. Wound Care 2016, 25, 585–592. [Google Scholar] [CrossRef]
- Genasetti, A.; Vigetti, D.; Viola, M.; Karousou, E.; Moretto, P.; Rizzi, M.; Bartolini, B.; Clerici, M.; Pallotti, F.; De Luca, G.; et al. Hyaluronan and human endothelial cell behaviour. Connect. Tissue Res. 2008, 49, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Chennazhi, K.P.; Srinivasan, S.; Nair, S.V.; Furuike, T.; Tamura, H. Chitin scaffolds in tissue engineering. Int. J. Mol. Sci. 2011, 12, 1876–1887. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Buri, P.A. Surface, interfacial and molecular aspects of polymer bioadhesion on soft tissues. J. Control. Release 1985, 2, 257–275. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kang, M.S.; Jeong, W.Y.; Dong-Wook, H.; Kim, K.S. Hyaluronic Acid-Based Theranostic Nanomedicines for Targeted Cancer Therapy. Cancers 2020, 1, 940. [Google Scholar] [CrossRef] [PubMed]
- Abou-Okeil, A.H.; Fahmy, M.; El-Bisi, M.K.; Ahmed-Farid, O.A. Hyaluronic acid/Naalginate films as topical bioactive wound dressings. Eur. Polym. J. 2018, 109, 101–109. [Google Scholar] [CrossRef]
- Jegasothy, S.M.; Zabolotniaia, V.; Bielfeldt, S. Efficacy of a new topical nanohyaluronic acid in humans. J. Clin. Aest. Dermatol. 2014, 7, 27–29. [Google Scholar]
- Amirian, J.; Makkar, P.; Lee, G.H.; Paul, K.; Lee, B.T. Incorporation of alginate-hyaluronic acid microbeads in injectable calcium phosphate cement for improved bone regeneration. Mater. Lett. 2020, 272, 127830. [Google Scholar] [CrossRef]
- Yang, S.; Zhu, B.; Yin, P.; Zhao, L.; Wang, Y.; Fu, Z.; Dang, R.; Xu, J.; Zhang, J.; Wen, N. Integration of Human Umbilical Cord Mesenchymal Stem Cells-Derived Exosomes with Hydroxyapatite-Embedded Hyaluronic Acid-Alginate Hydrogel for Bone Regeneration. ACS Biomater. Sci. Eng. 2020, 6, 1590–1602. [Google Scholar] [CrossRef]
- Makvandi, P.; Ali, G.W.; Della Sala, F.; Abdel-Fattah, W.I.; Borzacchiello, A. Hyaluronic acid/corn silk extract based injectable nanocomposite: A biomimetic antibacterial scaffold for bone tissue regeneration. Mater. Sci. Eng. C Mater. 2020, 107, 110195. [Google Scholar] [CrossRef]
- Chomoucka, J.; Drbohlavova, J.; Huska, D.; Adam, V.; Kizek, R.; Hubalek, J. Magnetic nanoparticles and targeted drug delivering. Pharmacol. Res. 2010, 62, 144–149. [Google Scholar] [CrossRef]
- Ito, A.; Shinkai, M.; Honda, H.; Kobayashi, T. Medican application of funcionalized magnetic nanoparticles. J. Biosci. Bioeng. 2005, 100, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dias, A.M.; Hussain, A.; Marcos, A.S.; Roque, A.C. A biotechnological perspective on the application of iron oxide magnetic colloids modified with polysaccharides. Biotechnol. Adv. 2011, 29, 142–155. [Google Scholar] [CrossRef] [PubMed]
- Chełminiak, D.; Ziegler-Borowska, M.; Kaczmarek, H. Polymer coated magnetite nanoparticles for biomedical applications. Part I. Preparation of nanoparticles Fe3O4 coated by polysaccharides. Polimery 2015, 60, 12–17. [Google Scholar] [CrossRef]
- Kim, S.; Cho, D.H.; Kweon, D.K.; Jang, E.H.; Hong, J.Y.; Lim, S.T. Improvement of mechanical properties of orodispersible hyaluronic acid film by carboxymethyl cellulose addition. Food Sci. Biotechnol. 2020, 29, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Ducker, M.; Sun, B.; Szele, F.G.; Czernuszka, J.T. Interpenetrating polymer networks of collagen, hyaluronic acid, and chondroitin sulfate as scaffolds for brain tissue engineering. Acta Biomater. 2020, 112, 122–135. [Google Scholar] [CrossRef]
- Ghorbani, F.; Zamanian, A.; Behnamghader, A.; Behnamghader, A.; Joupari, M.D. Bioactive and biostable hyaluronic acid-pullulan dermal hydrogels incorporated with biomimetic hydroxyapatite spheres. Mater. Sci. Eng. C Mater. 2020, 112, 110906. [Google Scholar] [CrossRef]
- Guan, Y.; You, H.; Cai, J.; Zhang, Q.; Yan, S.Q.; You, R.C. Physically crosslinked silk fibroin/hyaluronic acid scaffolds. Carbohyd. Polym. 2020, 239, 116232. [Google Scholar] [CrossRef]
- Unal, S.; Arslan, S.; Yilmaz, B.K.; Oktar, F.N.; Ficai, D.; Ficai, A.; Gunduz, O. Polycaprolactone/Gelatin/Hyaluronic Acid Electrospun Scaffolds to Mimic Glioblastoma Extracellular Matrix. Materials 2020, 13, 2661. [Google Scholar] [CrossRef]
- Najberg, M.; Mansor, M.H.; Taille, T.; Boure, C.; Molina-Pena, R.; Boury, F.; Cenis, J.L.; Garcion, E.; Alvarez-Lorenzo, C. Aerogel sponges of silk fibroin, hyaluronic acid and heparin for soft tissue engineering: Composition-properties relationship. Carbohydr. Polym. 2020, 237, 116107. [Google Scholar] [CrossRef]
- Zineh, B.R.; Shabgard, M.R.; Roshangar, L.; Jahani, K. Experimental and numerical study on the performance of printed alginate/hyaluronic acid/halloysite nanotube/polyvinylidene fluoride bio-scaffolds. J. Biomech. 2020, 104, 109764. [Google Scholar] [CrossRef]
- Antich, C.; de Vicente, J.; Jimenez, G.; Chocarro, C.; Carrillo, E.; Montanez, E.; Galvez-Martin, P.; Marchal, J.A. Bio-inspired hydrogel composed of hyaluronic acid and alginate as a potential bioink for 3D bioprinting of articular cartilage engineering constructs. ACTA Biomater. 2020, 106, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Phuong, L.T.; Son, J.Y.; Lee, Y.; Ryu, S.B.; Park, K.M.; Park, K.D. Enzymatically Crosslinkable Hyaluronic Acid-Gelatin Hybrid Hydrogels as Potential Bioinks for Tissue Regeneration. Macromol. Res. 2020, 28, 400–406. [Google Scholar]
- Andrysiak, T.; Bełdowski, P.; Siódmiak, J.; Weber, P.; Ledzinski, D. Hyaluronan-chondroitin sulfate anomalous crosslinking due to temperature changes. Polymers 2018, 10, 560. [Google Scholar] [CrossRef] [PubMed]
- Dėdinaitė, A.; Wieland, D.C.F.; Beldowski, P.; Claesson, P.M. Biolubrication synergy: Hyaluronan–Phospholipid interactions at interfaces. Adv. Coll. Interf. Sci. 2019, 274, 102050. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sionkowska, A.; Gadomska, M.; Musiał, K.; Piątek, J. Hyaluronic Acid as a Component of Natural Polymer Blends for Biomedical Applications: A Review. Molecules 2020, 25, 4035. https://doi.org/10.3390/molecules25184035
Sionkowska A, Gadomska M, Musiał K, Piątek J. Hyaluronic Acid as a Component of Natural Polymer Blends for Biomedical Applications: A Review. Molecules. 2020; 25(18):4035. https://doi.org/10.3390/molecules25184035
Chicago/Turabian StyleSionkowska, Alina, Magdalena Gadomska, Katarzyna Musiał, and Jacek Piątek. 2020. "Hyaluronic Acid as a Component of Natural Polymer Blends for Biomedical Applications: A Review" Molecules 25, no. 18: 4035. https://doi.org/10.3390/molecules25184035
APA StyleSionkowska, A., Gadomska, M., Musiał, K., & Piątek, J. (2020). Hyaluronic Acid as a Component of Natural Polymer Blends for Biomedical Applications: A Review. Molecules, 25(18), 4035. https://doi.org/10.3390/molecules25184035







