Biosurfactant Production and Growth Kinetics Studies of the Waste Canola Oil-Degrading Bacterium Rhodococcus erythropolis AQ5-07 from Antarctica
Abstract
1. Introduction
2. Results and Discussion
2.1. Growth and Biosurfactant Production
2.1.1. Haemolytic Activity
2.1.2. Drop Collapse Test
2.1.3. Oil-Spreading Test
2.1.4. Surface Tension Measurement
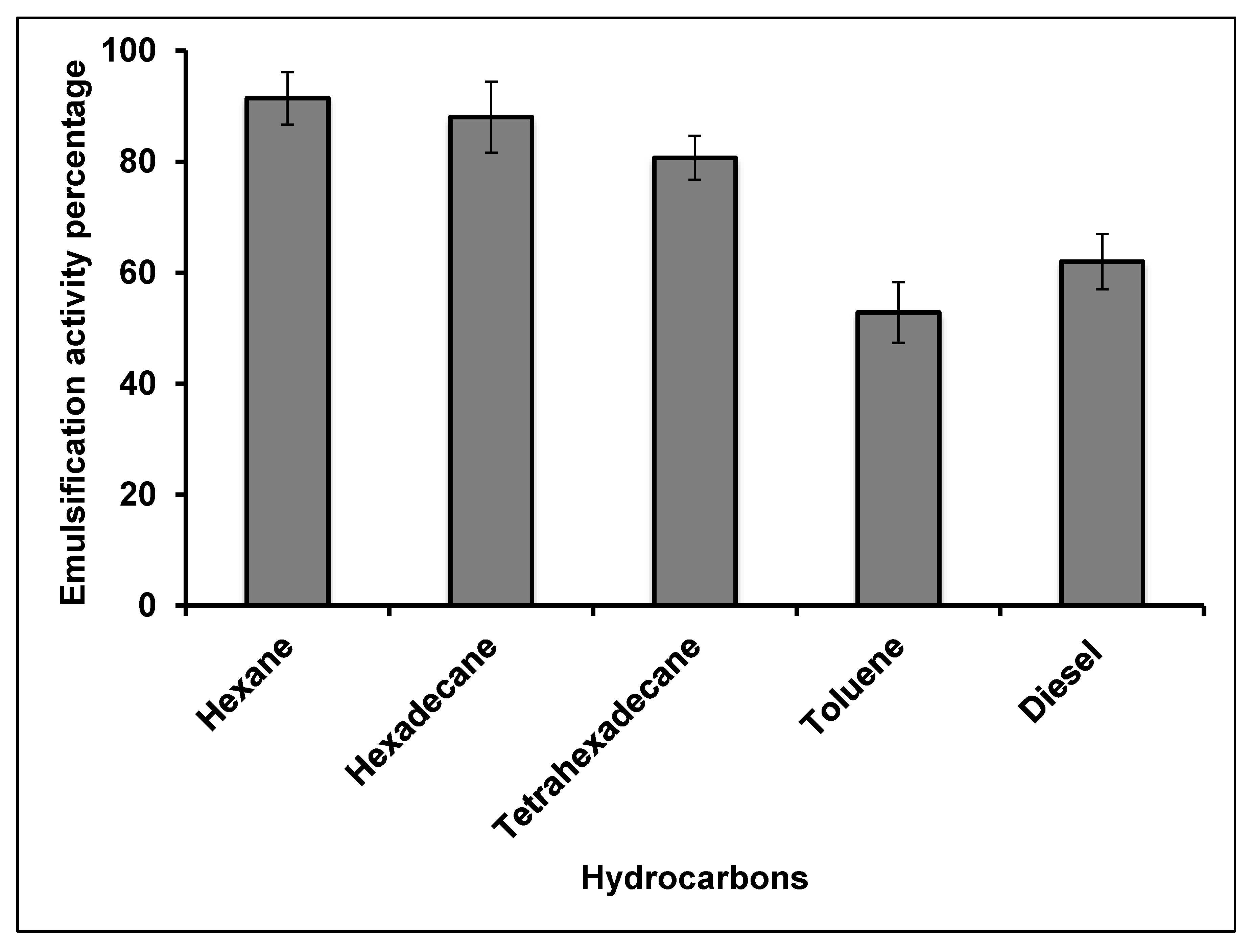
2.1.5. Emulsification Activity
2.1.6. Microbial Adhesion to Hydrocarbons (MATH) Assay
2.1.7. Stability of Biosurfactant
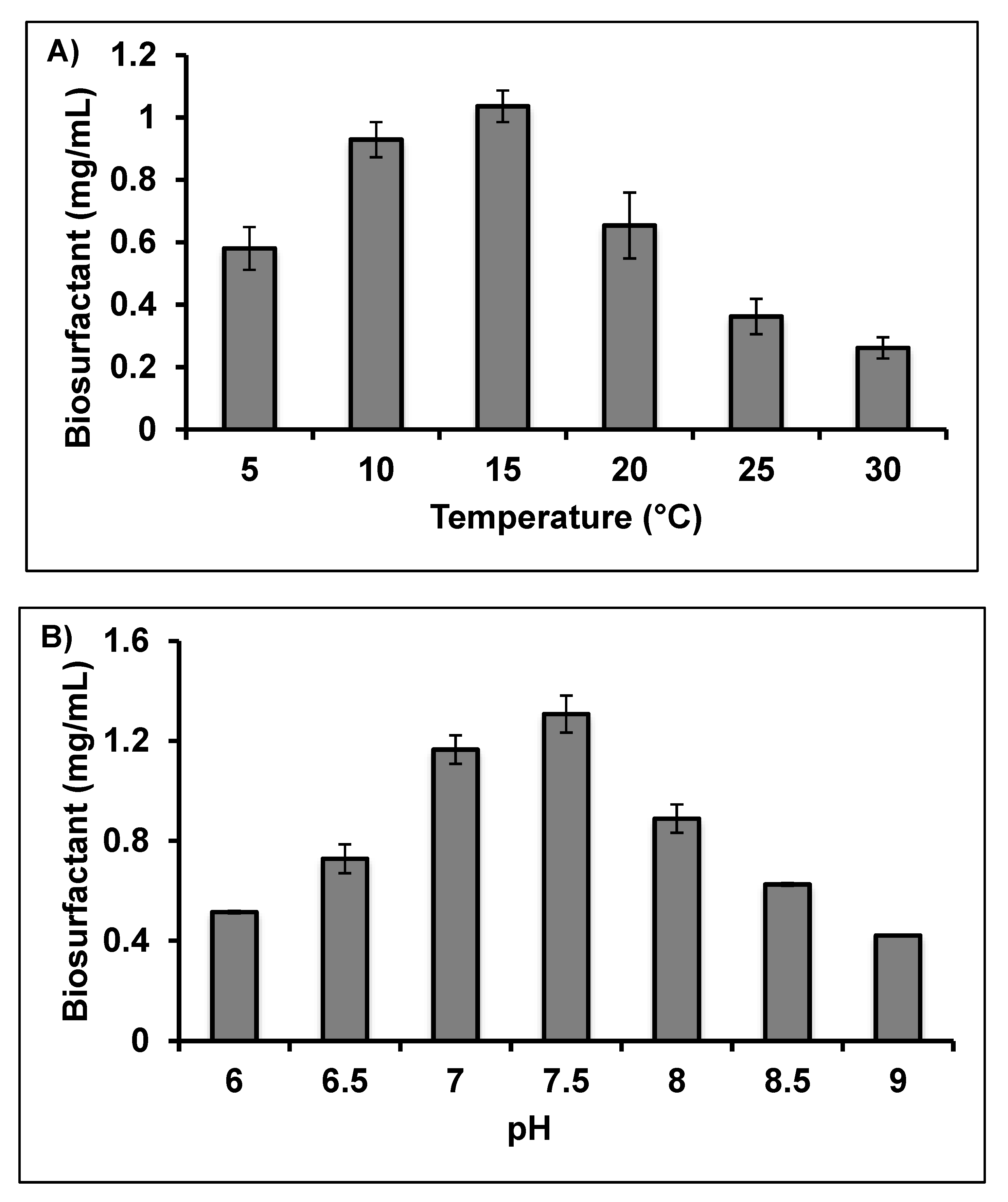
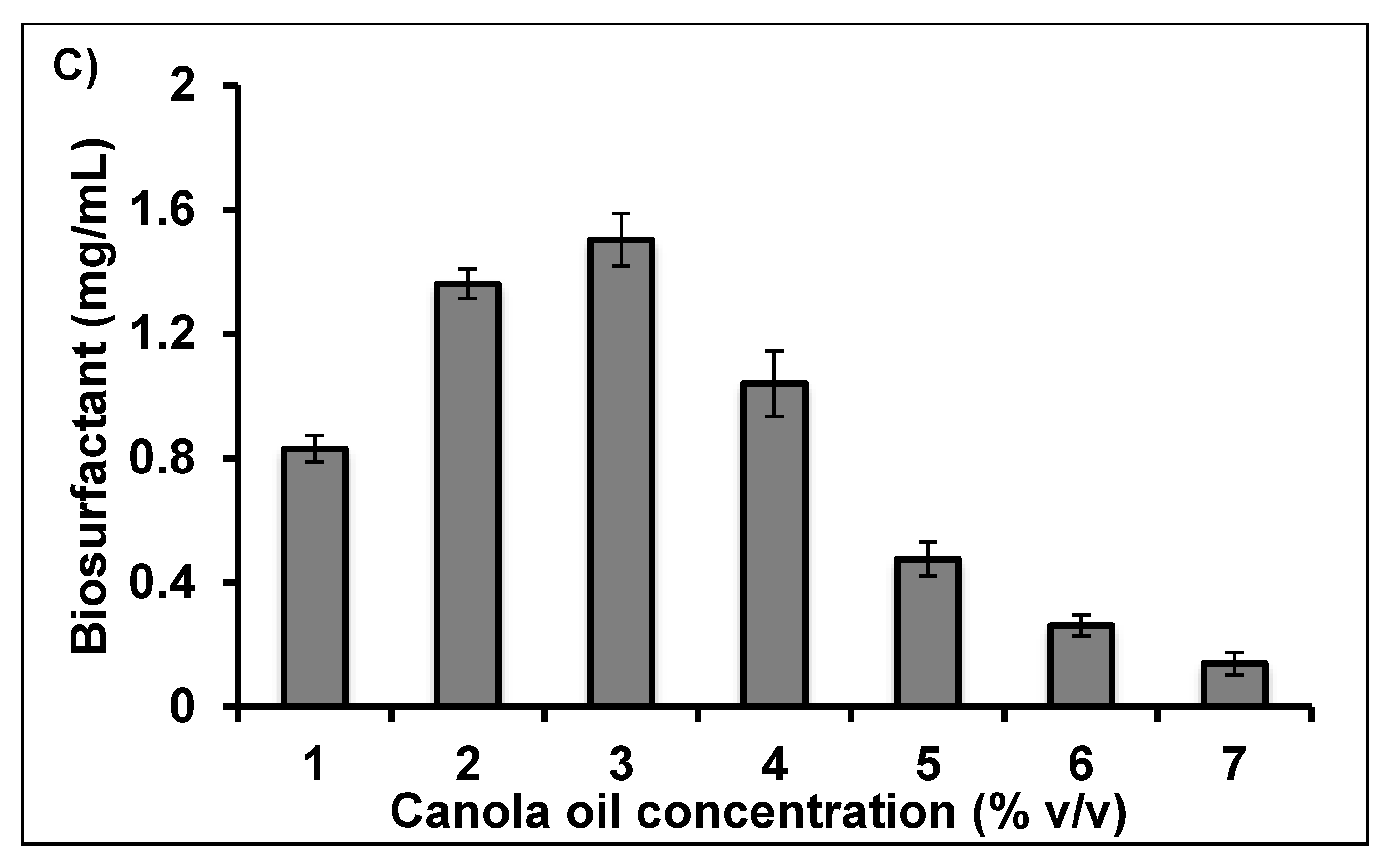
2.2. Optimisation of Crude Biosurfactant Production
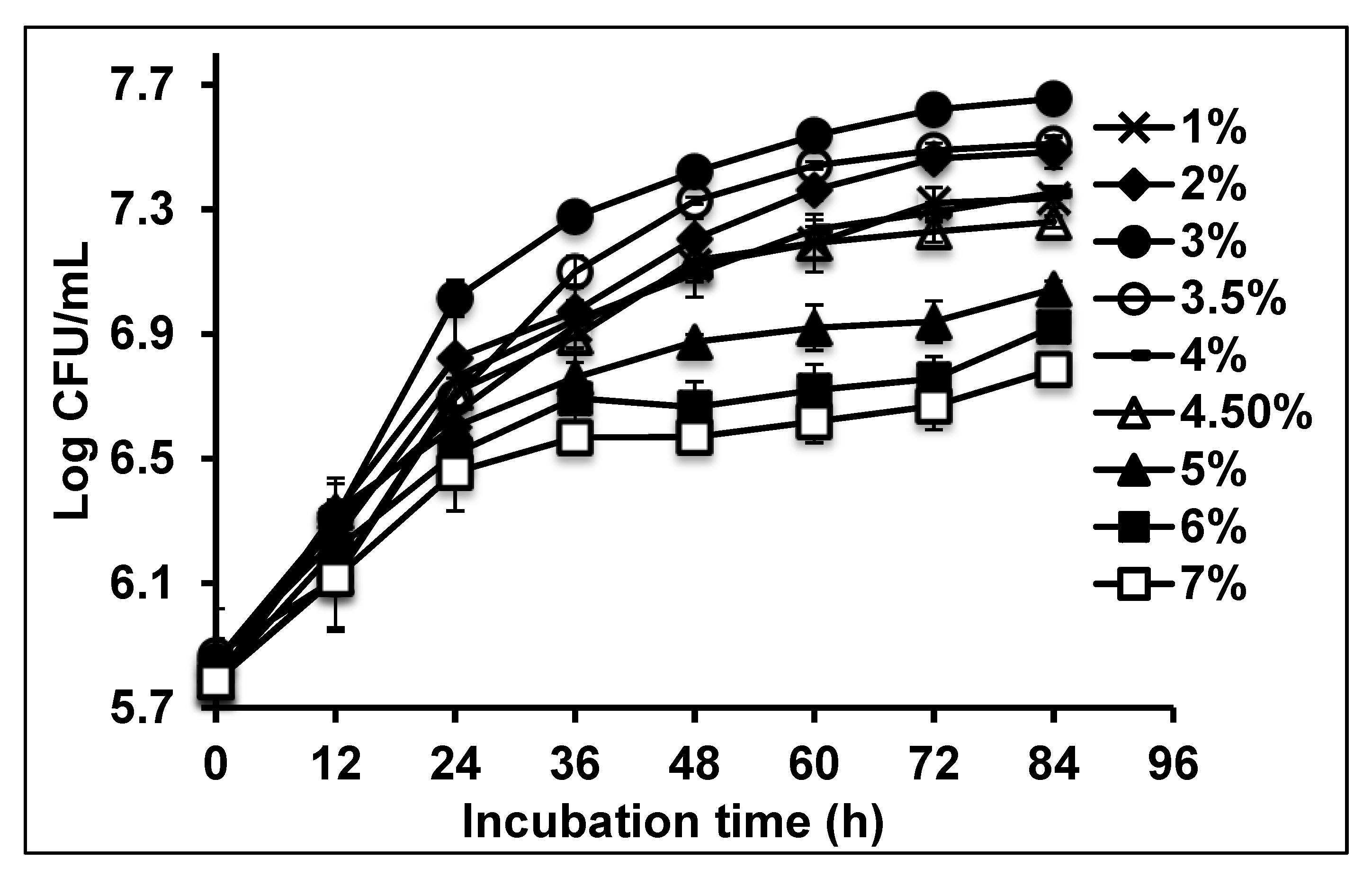
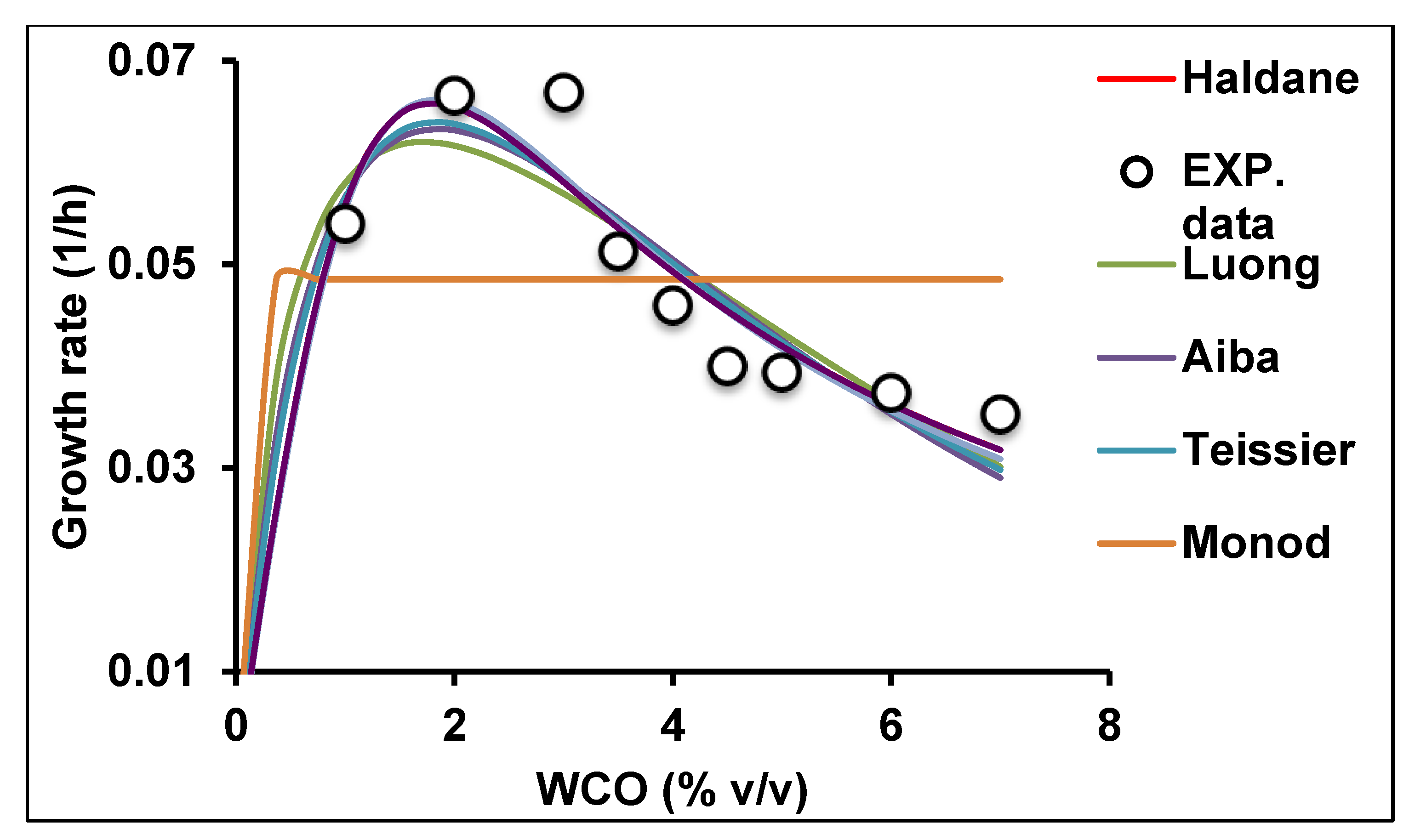
2.3. Growth on Canola Oil Modelled Using Secondary Models
3. Materials and Methods
3.1. Microorganism and Shake Flask Culture Condition
3.2. Biosurfactant Analysis
3.2.1. Haemolytic Activity
3.2.2. Drop Collapse Test
3.2.3. Oil-Spreading Test
3.2.4. Emulsification Index Test
3.2.5. Surface Tension
3.2.6. Microbial Adhesion to Hydrocarbon (MATH) Assay
3.2.7. Biosurfactant Stability Test
3.2.8. Optimisation of Biosurfactant Production
3.2.9. Biosurfactant Extraction
3.3. Bacterial Growth Kinetics Modelling
| Model | Equation | No. of Parameters | References |
|---|---|---|---|
| Monod | 2 | [54] | |
| Haldane | 3 | [55] | |
| Teissier | 3 | [56] | |
| Luong | 4 | [57] | |
| Aiba | 4 | [58] | |
| Yano and Koga | 4 | [59] | |
| Webb | 4 | [60] |
Verification of the Models
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Subramaniam, K.; Mazuki, T.A.T.; Shukor, M.Y.; Ahmad, S.A. Isolation and optimisation of phenol degradation by Antarctic isolate using ine factor at time. Malays. J. Biochem. Mol. Biol. 2019, 1, 79–86. [Google Scholar]
- Tengku-Mazuki, T.A.; Subramanian, K.; Zakaria, N.N.; Convey, P.; Khalil, K.A.; Lee, G.L.Y.; Zulkharnain, A.; Shaharuddin, N.A.; Ahmad, S.A. Optimization of phenol degradation by Antarctic bacterium Rhodococcus sp. Antarct. Sci. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Roslee, A.F.A.; Zakaria, N.N.; Convey, P.; Zulkharnain, A.; Lee, G.L.Y.; Gomez-Fuentes, C.; Ahmad, S.A. Statistical optimisation of growth conditions and diesel degradation by the Antarctic bacterium, Rhodococcus sp. strain AQ5‒07. Extremophiles 2020, 24, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Abdulrasheed, M.; Zakaria, N.N.; Ahmad Roslee, A.F.; Shukor, M.Y.; Zulkharnain, A.; Napis, S.; Convey, P.; Alias, S.A.; Gonzalez-Rocha, G.; Ahmad, S.A. Biodegradation of diesel oil by cold-adapted bacterial strains of Arthrobacter spp. from Antarctica. Antarct. Sci. 2020, 13, 1–13. [Google Scholar] [CrossRef]
- Zakaria, N.N.; Roslee, A.F.A.; Gomez-Fuentes, C.; Zulkharnain, A.; Abdulrasheed, M.; Sabri, S.; Ramírez-Moreno, N.; Calisto-Ulloa, N.; Ahmad, S.A. Kinetic studies of marine psychrotolerant microorganisms capable of degrading diesel in the presence of heavy metals. Rev. Mex. Ing. Química 2020, 19, 1375–1388. [Google Scholar] [CrossRef]
- Kaczorek, E.; Chrzanowski, L.; Pijanowska, A.; Olszanoswski, A. Yeast and bacteria cell hydrophobicity and hydrocarbon biodegradation in the presence of natural surfactants: Rhamnolipids and saponins. Bioresour. Technol. 2008, 99, 4285–4291. [Google Scholar] [CrossRef]
- Sáenz-Marta, C.I.; de Lourdes Ballinas-Casarrubias, M.; Rivera-Chavira, B.E.; Nevárez-Moorillón, G.V. Biosurfactants as useful tools in bioremediation. IntechOpen 2015, 2, 1–19. [Google Scholar] [CrossRef]
- Zakaria, N.N.; Man, Z.; Zulkharnain, A.; Ahmad, S.A. Psychrotolerant biosurfactant-producing bacteria for hydrocarbon degradation: A mini review. Malays. J. Biochem. Mol. Biol. 2019, 22, 52–59. [Google Scholar]
- Ilory, M.; Adebusoye, E.; Ojo, A. Isolation and characterization of hydrocarbon-degrading and biosurfactant-producing yeast strains obtained from a polluted lagoon water. World J. Microbiol. Biotechnol. 2008, 24, 2539–2545. [Google Scholar] [CrossRef]
- Abouseoud, M.; Maachi, R.; Amrane, A.; Boudergua, S.; Nabi, A. Evaluation of different carbon and nitrogen sources in production of biosurfactant by Pseudomonas fluorescens. Desalination 2008, 223, 143–151. [Google Scholar] [CrossRef]
- Kügler, J.H.; Le Roes-Hill, M.; Syldatk, C.; Hausmann, R. Surfactants tailored by the class Actinobacteria. Front. Microbiol. 2015, 6, 5–23. [Google Scholar] [CrossRef]
- Perfumo, A.; Banat, I.M.; Marchant, R. Going green and cold: Biosurfactants from low-temperature environments to biotechnology applications. Trends Biotechnol. 2018, 36, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Zahri, N.K.M.; Zulkharnain, A.; Ibrahim, S.; Gomez-fuentes, C.; Sabri, S.; Nancy, C.-U.; Ahmad, S.A. Kinetic analysis on the effects of lead (Pb) and silver (Ag) on waste canola oil (WCO) biodegradation by selected Antarctic microbial consortium. Malays. J. Biochem. Mol. Biol. 2020, 1, 20–23. [Google Scholar]
- Zakaria, N.N.; Roslee, A.F.A.; Zulkharnain, A.; Gomez-Fuentes, C.; Abdulrasheed, M.; Sabri, S.; Calisto-Ulloa, N.; Ahmad, S.A. Bacterial growth and diesel biodegradation in the presence of As, Cu and Pb by Antarctic marine bacteria. Malays. J. Biochem. Mol. Biol. 2019, 22, 1–7. [Google Scholar]
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; Van’t Riet, K. Modeling of the bacterial growth curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Mercurio, P.; Burns, K.A.; Negri, A. Testing the ecotoxicology of vegetable versus mineral based lubricating oils: 1. Degradation rates using tropical marine microbes. Environ. Pollut. 2004, 129, 165–173. [Google Scholar] [CrossRef]
- Campo, P.; Zhao, Y.; Suidan, M.T.; Venosa, A.D.; Sorial, G.A. Biodegradation kinetics and toxicity of vegetable oil triacylglycerols under aerobic conditions. Chemosphere 2007, 68, 2054–2062. [Google Scholar] [CrossRef]
- Lee, G.L.Y.; Ahmad, S.A.; Yasid, N.A.; Zulkharnain, A.; Convey, P.; Wan Johari, W.L.; Alias, S.A.; Gonzalez-Rocha, G.; Shukor, M.Y. Biodegradation of phenol by cold-adapted bacteria from Antarctic soils. Polar Biol. 2018, 41, 553–562. [Google Scholar] [CrossRef]
- Ibrahim, S.; Zulkharnain, A.; Zahri, K.N.M.; Lee, G.L.Y.; Convey, P.; Gomez-Fuentes, C.; Sabri, S.; Khalil, K.A.; Gonazalez-Rocha, G.; Ahmad, S.A. Effect of heavy metals and other xenobiotics on biodegradation of waste canola oil by cold-adapted Rhodococcus sp. AQ5-07. Rev. Mex. Ing. Química 2020, 19, 1041–1052. [Google Scholar] [CrossRef]
- Mulligan, C.N. Environmental applications for biosurfactants. Environ. Pollut. 2005, 133, 183–198. [Google Scholar] [CrossRef]
- Yonebayashi, H.; Yoshida, S.; Ono, K.; Enomoto, H. Screening of microorganisms for microbial enhanced oil recovery processes. J. Japan Pet. Inst. 2000, 43, 59–69. [Google Scholar] [CrossRef]
- Ibrahim, M.L.; Ijah, U.J.J.; Manga, S.B.; Bilbis, L.S.; Umar, S. Production and partial characterization of biosurfactant produced by crude oil degrading bacteria. Int. Biodeterior. Biodegrad. 2013, 81, 28–34. [Google Scholar] [CrossRef]
- Chandran, P.; Das, N. Biosurfactant production and diesel oil degradation by yeast species Trichosporon asahii isolated from petroleum hydrocarbon contaminated soil. Int. J. Eng. Sci. Technol. 2010, 2, 6942–6953. [Google Scholar]
- Thavasi, R.; Sharma, S.; Jayalakshmi, S. Evaluation of screening methods for the isolation of biosurfactant producing marine bacteria. J. Pet. Environ. Biotechnol. 2011, 4, 1–6. [Google Scholar] [CrossRef]
- Youssef, N.H.; Duncan, K.E.; Nagle, D.P.; Savage, K.N.; Knapp, R.M.; McInerney, M.J. Comparison of methods to detect biosurfactant production by diverse microorganisms. J. Microbiol. Methods 2004, 56, 339–347. [Google Scholar] [CrossRef]
- Morikawa, M.; Hirata, Y.; Imanaka, T. A study on the structure-function relationship of lipopeptide biosurfactants. Biochim. Biophys. Acta 2000, 1488, 211–218. [Google Scholar] [CrossRef]
- Bodour, A.A.; Miller-Maier, R.M. Application of a modified drop-collapse technique for surfactant quantitation and screening of biosurfactant-producing microorganisms. J. Microbiol. Methods 1998, 32, 273–280. [Google Scholar] [CrossRef]
- Morais, I.M.C.; Cordeiro, A.L.; Teixeira, G.S.; Domingues, V.S.; Nardi, R.M.D.; Monteiro, A.S.; Alves, R.J.; Siqueira, E.P.; Santos, V.L. Biological and physicochemical properties of biosurfactants produced by Lactobacillus jensenii P6A and Lactobacillus gasseri P65. Microb. Cell Fact. 2017, 16, 1–15. [Google Scholar] [CrossRef]
- Banat, I.M.; Makkar, R.S.; Cameotra, S.S. Potential commercial applications of microbial surfactants. Appl. Microbiol. Biotechnol. 2000, 53, 495–508. [Google Scholar] [CrossRef]
- Kumari, B.; Singh, S.N.; Singh, D.P. Characterization of two biosurfactant producing strains in crude oil degradation. Process Biochem. 2012, 47, 2463–2471. [Google Scholar] [CrossRef]
- Rosenberg, M. Bacterial adherence to hydrocarbons: A useful technique for studying cell surface hydrophobicity. FEMS Microbiol. Lett. 1984, 22, 289–295. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, D.; Li, M.; Xiang, W.N.; Achal, V. Isolation and characterization of diesel degrading bacteria, Sphingomonas sp. and Acinetobacter junii from petroleum contaminated soil. Front. Earth Sci. 2014, 8, 58–63. [Google Scholar] [CrossRef]
- Volchenko, N.N.; Karasev, S.G.; Nimchenko, D.V.; Karaseva, E.V. Cell hydrophobicity as a criterion of selection of bacterial producers of biosurfactants. Microbiology 2007, 76, 112–114. [Google Scholar] [CrossRef]
- Obayori, O.S.; Ilori, M.O.; Adebussoye, S.A.; Oyetibo, G.O.; Omoayo, A.E.; Amund, O.O. Degradation of hydrocarbons an biosurfactant production by Pseudomonas sp. strain LP1. World J. Microbiol. Biotechnol. 2009, 25, 1615–1623. [Google Scholar] [CrossRef]
- Helmke, E.; Weyland, H. Psychrophilic versus psychrotolerant bacteria-occurrence and significance in polar and temperate marine habitats. Cell. Mol. Biol. 2004, 50, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Peck, L.S.; Convey, P.; Barnes, D.K.A. Environmental constraints on life histories in Antarctic ecosystems: Tempos, timings and predictability. Biol. Rev. 2006, 81, 75–109. [Google Scholar] [CrossRef]
- Convey, P.; Coulson, S.J.; Worland, M.R.; Sjöblom, A. The importance of understanding annual and shorter-term temperature patterns and variation in the surface levels of polar soils for terrestrial biota. Polar Biol. 2018, 41, 1587–1605. [Google Scholar] [CrossRef]
- Hamzah, A.; Sabturani, N.; Radiman, S. Screening and optimization of biosurfactant production by the hydrocarbon-degrading bacteria. Sains Malays. 2013, 42, 615–623. [Google Scholar] [CrossRef]
- Chen, S.Y.; Lu, W.B.; Wei, Y.H.; Chen, W.M.; Chang, J.S. Improved production of biosurfactant with newly isolated Pseudomonas aeruginosa S2. Biotechnol. Prog. 2007, 23, 661–666. [Google Scholar] [CrossRef]
- Ahmad, S.A.; Ibrahim, S.; Shukor, M.Y.; Johari, W.L.W.J.; Rahman, N.A.; Syed, M.A.S. Biodegradation kinetics of caffeine by Leifsonia sp. strain SIU. J. Chem. Pharm. Sci. 2015, 8, 312–316. [Google Scholar]
- Ibrahim, S.; Shukor, M.Y.; Syed, M.A.; Wan Johari, W.L.; Ahmad, S.A. Characterisation and growth kinetics studies of caffeine-degrading bacterium Leifsonia sp. strain SIU. Ann. Microbiol. 2016, 66, 289–298. [Google Scholar] [CrossRef]
- Agarwal, R.; Mahanty, B.; Dasu, V.V. Modeling growth of Cellulomonas cellulans NRRL B 4567 under substrate inhibition during Cellulase production. Chem. Biochem. Eng. Q. 2009, 23, 213–218. [Google Scholar]
- Agarry, S.E.; Solomon, B.O. Kinetics of batch microbial degradation of phenols by indigenous Pseudomonas fluorescence. Int. J. Environ. Sci. Technol. 2008, 5, 223–232. [Google Scholar] [CrossRef]
- Othman, A.R.; Bakar, N.A.; Halmi, M.I.E.; Johari, W.L.W.; Ahmad, S.A.; Jirangon, H.; Syed, M.A.; Shukor, M.Y. Kinetics of molybdenum reduction to molybdenum blue by Bacillus sp. strain A.rzi. BioMed Res. Int. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wayman, M.; Tseng, M.C. Inhibition threshold substrate concentrations. Biotechnol. Bioeng. 1976, 18, 383–387. [Google Scholar] [CrossRef]
- Mulchandani, A.; Luong, J.H.T.; Groom, C. Substrate inhibition kinetics for microbial growth and synthesis of poly-β-hydroxybutyric acid by Alcaligenes eutrophus ATCC 17697. Appl. Microbiol. Biotechnol. 1989, 30, 11–17. [Google Scholar] [CrossRef]
- Ahmad, S.A.; Ku Ahamad, K.N.E.; WanJohari, W.L.; Halmi, M.I.E.; Shukor, M.Y.; Yusof, M.T. Kinetics of diesel degradation by an acrylamide-degrading bacterium. Rend. Lincei 2014, 25, 505–512. [Google Scholar] [CrossRef]
- Rajan, A.; Kumar, S.; Nair, A.J. Isolation of novel alkaline lipase producing fungus Aspergillus fumigatus MTCC 9657 from aged and crude rice bran oil and quantification by HPTLC. Int. J. Biol. Chem. 2011, 5, 116–126. [Google Scholar] [CrossRef]
- Shoeb, E.; Ahmed, N.; Akhter, J.; Badar, U.; Siddiqui, K.; Ansari, F.A.; Waqar, M.; Imtiaz, S.; Akhtar, N.; Shaikh, Q.U.A.; et al. Screening and characterization of biosurfactant-producing bacteria isolated from the Arabian Sea coast of Karachi. Turk. J. Biol. 2015, 39, 210–216. [Google Scholar] [CrossRef]
- Rodrigues, L.; Banat, I.M.; Teixeira, J.; Oliveira, R. Biosurfactants: Potential applications in medicine. J. Antimicrob. Chemother. 2006, 57, 609–618. [Google Scholar] [CrossRef]
- Zoueki, C.W.; Tufenkji, N.; Ghoshal, S. A modified microbial adhesion to hydrocarbons assay to account for the presence of hydrocarbon droplets. J. Colloid Interface Sci. 2010, 344, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Patowary, K.; Patowary, R.; Kalita, M.C.; Deka, S. Characterization of biosurfactant produced during degradation of hydrocarbons using crude oil as sole sole of carbon. Front. Microbiol. 2017, 8, 2–14. [Google Scholar] [CrossRef] [PubMed]
- Karamba, K.I.; Aqlima, S.A.; Zulkharnain, A.; Yasid, N.A.; Ibrahim, S.; Shukor, M.Y. Batch growth kinetic studies of locally isolated cyanide-degrading Serratia marcescens strain AQ07. 3 Biotech 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Monod, J. The growth of bacterial cultures. Annu. Rev. Microbiol. 1949, 3, 371–394. [Google Scholar] [CrossRef]
- Haldane, J.B.S. Enzymes; Longmans, Green & Co: London, UK, 1930. [Google Scholar]
- Teissier, G. Croissance des populations bacte’riennes et quantite’d’aliment disponible (Growth of bacterial populations and the available substrate concentration). Revis. Sci. 1942, 80, 209. [Google Scholar]
- Luong, J.H.T. Generalization of Monod kinetics for analysis of growth data with substrate inhibition. Biotechnol. Bioeng. 1987, 29, 242–248. [Google Scholar] [CrossRef]
- Aiba, S.; Shoda, M.; Nagalani, M. Kinetics of product inhibition in alcohol fermentation. Biotechnol. Bioeng. 1968, 10, 845–864. [Google Scholar] [CrossRef]
- Yano, T.; Koga, S. Dynamic behavior of the chemostat subject to substrate inhibition. Biotechnol. Bioeng. 1969, 11, 139–153. [Google Scholar] [CrossRef]
- Webb, J.L. Enzymes and Metabolic Inhibitors; Academic Press: Boston, MA, USA, 1963. [Google Scholar]
- López, S.; Prieto, M.; Dijkstra, J.; Dhanoa, M.S.; France, J. Statistical evaluation of mathematical models for microbial growth. Int. J. Food Microbiol. 2004, 96, 289–300. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


| Model | Parameter | R2 | ADJ R2 | AICc | RMSE | SSE | BF | AF |
|---|---|---|---|---|---|---|---|---|
| Haldane | 3 | 0.9547 | 0.9364 | −105.46 | 0.004733 | 0.000162 | 1.00 | 1.07 |
| Teissier | 3 | 0.8971 | 0.8677 | −92.29 | 0.006949 | 0.000338 | 1.00 | 1.04 |
| Monod | 2 | 0.7181 | 0.8272 | −90.38 | 0.01138 | 0.001166 | 0.98 | 1.12 |
| Yano | 4 | 0.9464 | 0.9196 | −102.10 | 0.005418 | 0.000176 | 1.00 | 1.07 |
| Luong | 4 | 0.9064 | 0.8797 | −94.68 | 0.006626 | 0.000301 | 1.00 | 1.08 |
| Aiba | 3 | 0.9333 | 0.9661 | −101.57 | 0.005592 | 0.000219 | 1.00 | 1.09 |
| Webb | 4 | 0.9646 | 0.8520 | −103.61 | 0.007349 | 0.000324 | 1.01 | 1.08 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibrahim, S.; Abdul Khalil, K.; Zahri, K.N.M.; Gomez-Fuentes, C.; Convey, P.; Zulkharnain, A.; Sabri, S.; Alias, S.A.; González-Rocha, G.; Ahmad, S.A. Biosurfactant Production and Growth Kinetics Studies of the Waste Canola Oil-Degrading Bacterium Rhodococcus erythropolis AQ5-07 from Antarctica. Molecules 2020, 25, 3878. https://doi.org/10.3390/molecules25173878
Ibrahim S, Abdul Khalil K, Zahri KNM, Gomez-Fuentes C, Convey P, Zulkharnain A, Sabri S, Alias SA, González-Rocha G, Ahmad SA. Biosurfactant Production and Growth Kinetics Studies of the Waste Canola Oil-Degrading Bacterium Rhodococcus erythropolis AQ5-07 from Antarctica. Molecules. 2020; 25(17):3878. https://doi.org/10.3390/molecules25173878
Chicago/Turabian StyleIbrahim, Salihu, Khalilah Abdul Khalil, Khadijah Nabilah Mohd Zahri, Claudio Gomez-Fuentes, Peter Convey, Azham Zulkharnain, Suriana Sabri, Siti Aisyah Alias, Gerardo González-Rocha, and Siti Aqlima Ahmad. 2020. "Biosurfactant Production and Growth Kinetics Studies of the Waste Canola Oil-Degrading Bacterium Rhodococcus erythropolis AQ5-07 from Antarctica" Molecules 25, no. 17: 3878. https://doi.org/10.3390/molecules25173878
APA StyleIbrahim, S., Abdul Khalil, K., Zahri, K. N. M., Gomez-Fuentes, C., Convey, P., Zulkharnain, A., Sabri, S., Alias, S. A., González-Rocha, G., & Ahmad, S. A. (2020). Biosurfactant Production and Growth Kinetics Studies of the Waste Canola Oil-Degrading Bacterium Rhodococcus erythropolis AQ5-07 from Antarctica. Molecules, 25(17), 3878. https://doi.org/10.3390/molecules25173878











