Identifying Active Compounds and Targets of Fritillariae thunbergii against Influenza-Associated Inflammation by Network Pharmacology Analysis and Molecular Docking
Abstract
1. Introduction
2. Results
2.1. Active Compounds Selection Using Physicochemical Characteristics
2.2. Compounds Associated with Biological Targets
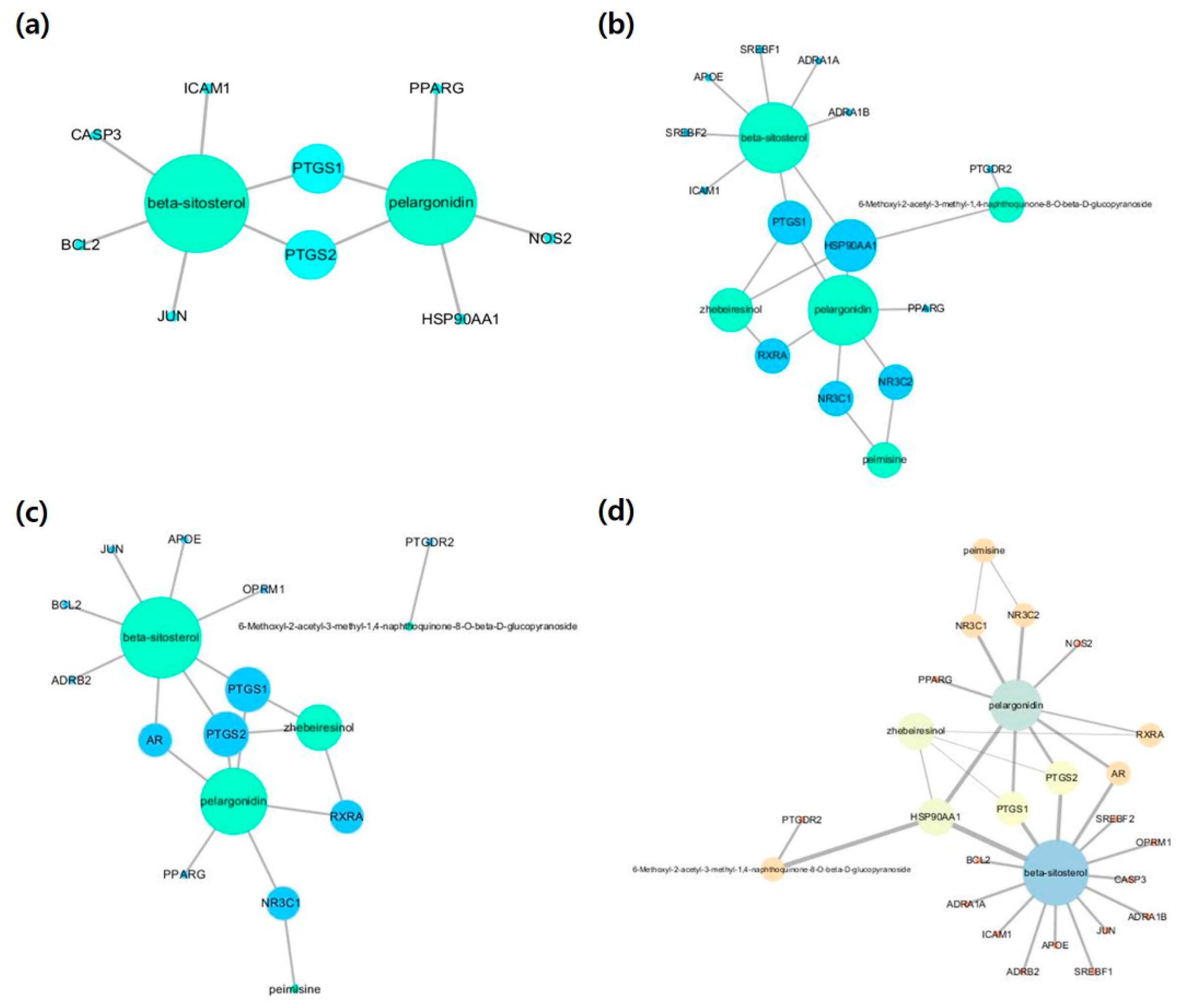
2.3. Compound-Disease-Target (C-D-T) Networks
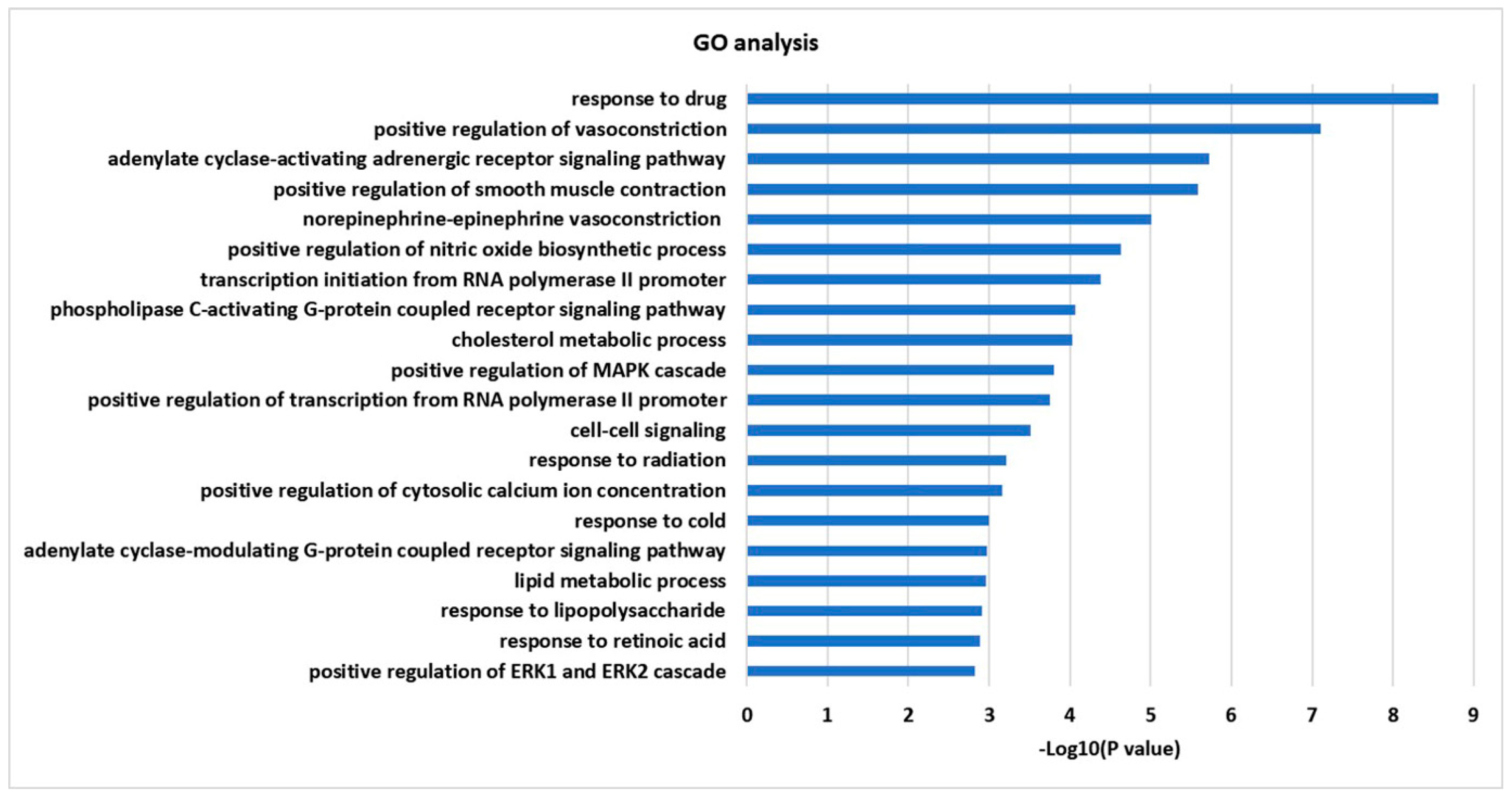
2.4. GO Enrichment and KEGG Pathway Analysis
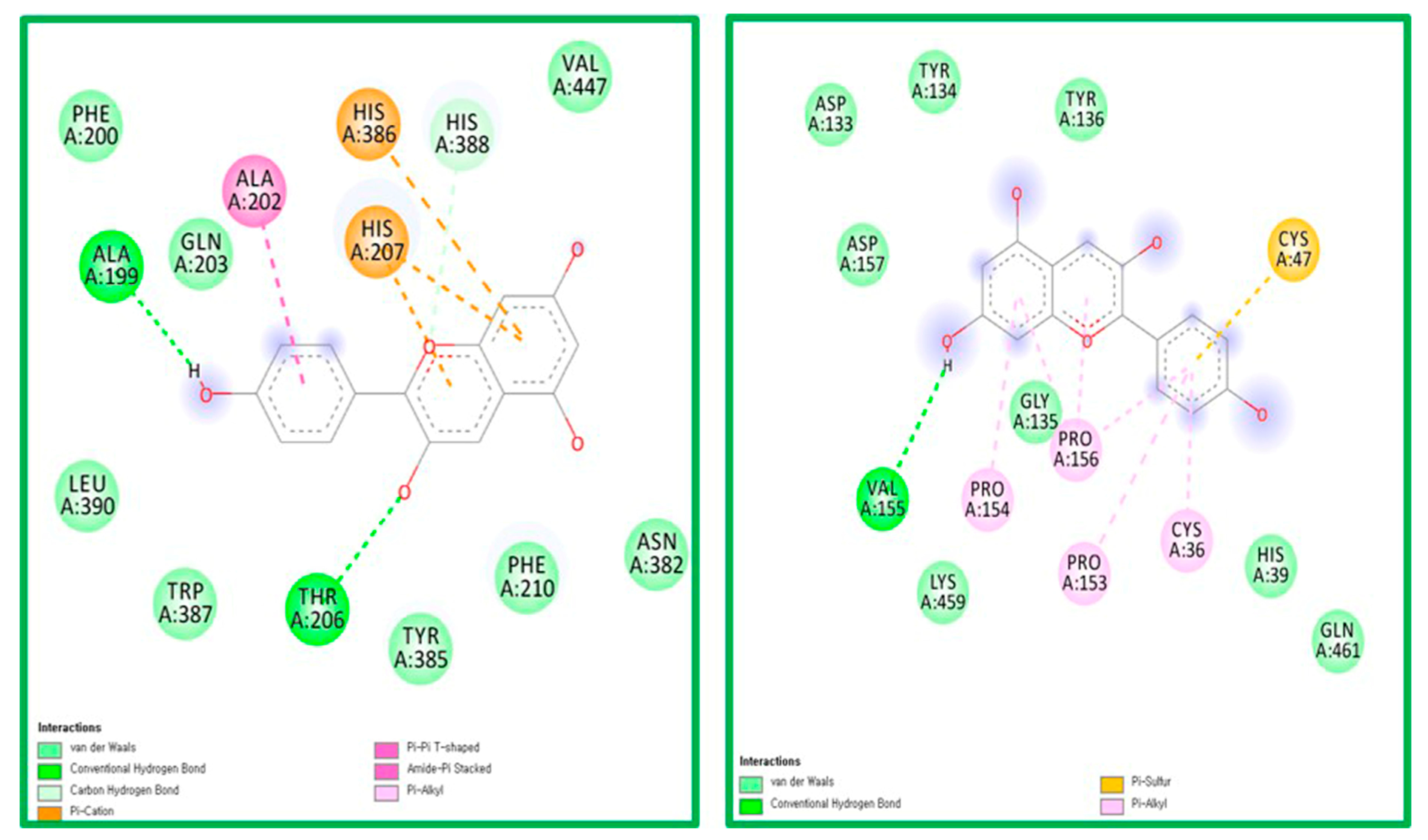
2.5. Molecular Docking Analysis
3. Discussion
3.1. Prediction of Potential Active Compounds of FT
3.2. Potential Therapeutic Targets Analysis
3.3. Pathway Analysis and Network Construction
3.4. Molecular Docking Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Herold, S.; Becker, C.; Ridge, K.M.; Budinger, G.R.S. Influenza virus-induced lung injury: Pathogenesis and implications for treatment. Eur. Respir. J. 2015, 45, 1463–1478. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Liu, D.-Y.; Yang, Z.-Q. Characteristics of human infection with avian influenza viruses and development of new antiviral agents. Acta Pharmacol. Sin. 2013, 34, 1257–1269. [Google Scholar] [CrossRef] [PubMed]
- Tisoncik, J.R.; Korth, M.J.; Simmons, C.P.; Farrar, J.; Martin, T.R.; Katze, M.G. Into the Eye of the Cytokine Storm. Microbiol. Mol. Biol. Rev. 2012, 76, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Peiris, J.S.M.; Hui, K.P.Y.; Yen, H.-L. Host response to influenza virus: Protection versus immunopathology. Curr. Opin. Immunol. 2010, 22, 475–481. [Google Scholar] [CrossRef]
- Turner, D.L.; Bickham, K.L.; Farber, D.L.; Lefrançois, L. Splenic Priming of Virus-Specific CD8 T Cells following Influenza Virus Infection. J. Virol. 2013, 87, 4496–4506. [Google Scholar] [CrossRef]
- Wang, D.; Zhu, J.; Wang, S.; Wang, X.; Ou, Y.; Wei, D.; Li, X. Antitussive, expectorant and anti-inflammatory alkaloids from Bulbus Fritillariae Cirrhosae. Fitoterapia 2011, 82, 1290–1294. [Google Scholar] [CrossRef]
- Kim, M.; Nguyen, D.-V.; Heo, Y.; Park, K.H.; Paik, H.-D.; Kim, Y.B. Antiviral Activity of Fritillaria thunbergii extract against Human Influenza Virus H1N1 (PR8) in vitro, in ovo and in vivo. J. Microbiol. Biotechnol. 2020, 30, 172–177. [Google Scholar] [CrossRef]
- Hopkins, A.L. Network pharmacology: The next paradigm in drug discovery. Nat. Methods 2008, 4, 682–690. [Google Scholar] [CrossRef]
- Wang, X.; Xu, X.; Li, Y.; Li, X.; Tao, W.; Li, B.; Wang, Y.; Yang, L. Systems pharmacology uncovers Janus functions of botanical drugs: Activation of host defense system and inhibition of influenza virus replication. Integr. Biol. 2012, 5, 351–371. [Google Scholar] [CrossRef]
- Shi, X.-Q.; Yue, S.-J.; Tang, Y.-P.; Chen, Y.-Y.; Zhou, G.-S.; Zhang, J.; Zhu, Z.-H.; Liu, P.; Duan, J.-A. A network pharmacology approach to investigate the blood enriching mechanism of Danggui buxue Decoction. J. Ethnopharmacol. 2019, 235, 227–242. [Google Scholar] [CrossRef]
- Wang, N.; Zhu, F.; Shen, M.; Qiu, L.-P.; Tang, M.; Xia, H.; Chen, L.; Yuan, Y.; Ma, S.; Chen, K. Network pharmacology-based analysis on bioactive anti-diabetic compounds in Potentilla discolor bunge. J. Ethnopharmacol. 2019, 241, 111905. [Google Scholar] [CrossRef]
- Yu, H.; Chen, J.; Xu, X.; Li, Y.; Zhao, H.; Fang, Y.; Li, X.; Zhou, W.; Wang, W.; Wang, Y. A Systematic Prediction of Multiple Drug-Target Interactions from Chemical, Genomic, and Pharmacological Data. PLoS ONE 2012, 7, e37608. [Google Scholar] [CrossRef] [PubMed]
- Shawky, E. Prediction of potential cancer-related molecular targets of North African plants constituents using network pharmacology-based analysis. J. Ethnopharmacol. 2019, 238, 111826. [Google Scholar] [CrossRef] [PubMed]
- Ru, J.; Li, P.; Wang, J.; Zhou, W.; Li, B.; Huang, C.; Li, P.; Guo, Z.; Tao, W.; Yang, Y.; et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J. Chemin. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Herold, S.; Mayer, K.; Lohmeyer, J. Acute Lung Injury: How Macrophages Orchestrate Resolution of Inflammation and Tissue Repair. Front. Immunol. 2011, 2, 65. [Google Scholar] [CrossRef]
- Yoo, J.-K.; Galligan, C.L.; Virtanen, C.; Fish, E.N. Identification of a novel antigen-presenting cell population modulating antiinfluenza type 2 immunity. J. Exp. Med. 2010, 207, 1435–1451. [Google Scholar] [CrossRef]
- Kim, E.J.; Yoon, Y.P.; Woo, K.W.; Kim, J.-H.; Min, S.Y.; Lee, H.J.; Lee, S.K.; Hong, J.H.; Lee, K.R.; Lee, C.J. Verticine, ebeiedine and suchengbeisine isolated from the bulbs of Fritillaria thunbergii Miq. inhibited the gene expression and production of MUC5AC mucin from human airway epithelial cells. Phytomedicine 2016, 23, 95–104. [Google Scholar] [CrossRef]
- Li, H.; Hung, A.; Li, M.; Yang, A.W.H. Fritillariae thunbergii Bulbus: Traditional Uses, Phytochemistry, Pharmacodynamics, Pharmacokinetics and Toxicity. Int. J. Mol. Sci. 2019, 20, 1667. [Google Scholar] [CrossRef]
- Zhou, M.; Ma, X.; Ding, G.; Wang, Z.; Liu, D.; Tong, Y.; Zhou, H.; Gao, J.; Hou, Y.; Jiang, M.; et al. Comparison and evaluation of antimuscarinic and anti-inflammatory effects of five Bulbus fritillariae species based on UPLC-Q/TOF integrated dual-luciferase reporter assay, PCA and ANN analysis. J. Chromatogr. B 2017, 1041, 60–69. [Google Scholar] [CrossRef]
- Xu, J.; Zhao, W.; Pan, L.; Zhang, A.; Chen, Q.; Xu, K.; Lu, H.; Chen, Y. Peimine, a main active ingredient of Fritillaria, exhibits anti-inflammatory and pain suppression properties at the cellular level. Fitoterapia 2016, 111, 1–6. [Google Scholar] [CrossRef]
- Sorenson, W.R.; Sullivan, D.; Baugh, S.; Collison, M.; Das, R.; Erickson, A.; Harmon, T.; Heathman, S.; Ji, D.; Khandelwal, B.; et al. Determination of Campesterol, Stigmasterol, and Beta-Sitosterol in Saw Palmetto Raw Materials and Dietary Supplements by Gas Chromatography: Collaborative Study. J. AOAC Int. 2007, 90, 670–678. [Google Scholar] [CrossRef]
- Lampronti, I.; Dechecchi, M.C.; Rimessi, A.; Bezzerri, V.; Nicolis, E.; Guerrini, A.; Tacchini, M.; Tamanini, A.; Munari, S.; D’Aversa, E.; et al. Beta-Sitosterol Reduces the Expression of Chemotactic Cytokine Genes in Cystic Fibrosis Bronchial Epithelial Cells. Front. Pharmacol. 2017, 8, 236. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Korm, S.; Chung, H.J.; Choi, S.J.; Jang, J.; Cho, S.; Lim, Y.T.; Kim, H.; Lee, J.Y. RAP80 regulates epithelial-mesenchymal transition related with metastasis and malignancy of cancer. Cancer Sci. 2016, 107, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Hao, D.; Xu, W.; Li, J.; Li, X.; Shen, D.; Sheng, K.; Zhao, L.; Gao, Z.; Zhao, X.; et al. Beta-Sitosterol modulates macrophage polarization and attenuates rheumatoid inflammation in mice. Pharm. Biol. 2019, 57, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Yu, D.; Zhang, Y. beta-Sitosterol Attenuates the Intracranial Aneurysm Growth by Suppressing TNF-alpha-Mediated Mechanism. Pharmacology 2019, 104, 303–311. [Google Scholar] [CrossRef]
- Dangles, O.; Fenger, J.-A. The Chemical Reactivity of Anthocyanins and Its Consequences in Food Science and Nutrition. Molecules 2018, 23, 1970. [Google Scholar] [CrossRef]
- Lee, B.-S.; Lee, C.; Yang, S.; Park, E.K.; Ku, S.-K.; Bae, J.-S. Suppressive effects of pelargonidin on lipopolysaccharide-induced inflammatory responses. Chem. Interact. 2019, 302, 67–73. [Google Scholar] [CrossRef]
- Ramos, I.; Fernandez-Sesma, A. Modulating the Innate Immune Response to Influenza A Virus: Potential Therapeutic Use of Anti-Inflammatory Drugs. Front. Immunol. 2015, 6, 361. [Google Scholar] [CrossRef]
- Shi, X.; Zhou, W.; Huang, H.; Zhu, H.-G.; Zhou, P.; Zhu, H.; Ju, D. Inhibition of the inflammatory cytokine tumor necrosis factor-alpha with etanercept provides protection against lethal H1N1 influenza infection in mice. Crit. Care 2013, 17, R301. [Google Scholar] [CrossRef]
- De Flora, S.; Grassi, C.; Carati, L. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur. Respir. J. 1997, 10, 1535–1541. [Google Scholar] [CrossRef]
- Kikuchi, T.; Hagiwara, K.; Honda, Y.; Gomi, K.; Kobayashi, T.; Takahashi, H.; Tokue, Y.; Watanabe, A.; Nukiwa, T. Clarithromycin suppresses lipopolysaccharide-induced interleukin-8 production by human monocytes through AP-1 and NF-kappaB transcription factors. J. Antimicrob. Chemother. 2002, 49, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Burggraaf, S.; Bingham, J.; Payne, J.; Kimpton, W.G.; Lowenthal, J.W.; Bean, A.G.D. Increased Inducible Nitric Oxide Synthase Expression in Organs Is Associated with a Higher Severity of H5N1 Influenza Virus Infection. PLoS ONE 2011, 6, e14561. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |


| Active Compounds | |||||||
|---|---|---|---|---|---|---|---|
| PG | BS | PM | ZBR | ZBM | 6GP | CS | |
| MW | 271.26 | 414.79 | 427.69 | 280.3 | 413.71 | 422.42 | 450.66 |
| OB | 37.99 | 36.91 | 57.4 | 58.72 | 64.25 | 33.31 | 65.63 |
| DL | 0.21 | 0.75 | 0.81 | 0.19 | 0.7 | 0.57 | 0.66 |
| Caco-2 | 0.31 | 1.32 | 0.18 | 0.53 | 0.81 | −1.21 | −0.01 |
| HL | 0.48 | 5.37 | 14.39 | 3.32 | 7.81 | 31.01 | 0.58 |
| Compound | Target |
|---|---|
| BS | ABCB11, ABCG5, ABCG8, ADRA1A, ADRA1B, ADRB2, APOE, BCL2, CASP3, CHRM1, CHRM2, CHRM3, CHRM4, CHRNA2, CYP7A1, DRD1, GABRA1, HSP90AA1, HTR2A, ICAM1, JUN, KCNH2, MAP2, NCOA2, OPRM1, PDE3A, PGR, PON1, PTGS1, PTGS2, SCN5A, SLC6A4, SREBF1, SREBF2 |
| PG | ACHE, AR, CA2, HSP90AA1, NCOA1, NCOA2, NOS2, NR3C1, NR3C2, PGR, PPARG, PTGS1, PTGS2, RXRA |
| ZBR | ADRB2, GABRA1, HSP90AA1, PDE3A, PTGS1, PTGS2, RXRA, SCN5A |
| 6GP | CA2, ESR1, F7, HSP90AA1, NCOA2, PTGDR2, TOP2B |
| PM | NR3C1, NR3C2 |
| Ligand-Target | Binding Affinity (kcal/mol) |
|---|---|
| PG-COX-1 | −8.6 |
| PG-COX-2 | −7.9 |
| BS-COX-1 | −6.9 |
| BS-COX-2 | −7.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.; Park, K.H.; Kim, Y.B. Identifying Active Compounds and Targets of Fritillariae thunbergii against Influenza-Associated Inflammation by Network Pharmacology Analysis and Molecular Docking. Molecules 2020, 25, 3853. https://doi.org/10.3390/molecules25173853
Kim M, Park KH, Kim YB. Identifying Active Compounds and Targets of Fritillariae thunbergii against Influenza-Associated Inflammation by Network Pharmacology Analysis and Molecular Docking. Molecules. 2020; 25(17):3853. https://doi.org/10.3390/molecules25173853
Chicago/Turabian StyleKim, Minjee, Ki Hoon Park, and Young Bong Kim. 2020. "Identifying Active Compounds and Targets of Fritillariae thunbergii against Influenza-Associated Inflammation by Network Pharmacology Analysis and Molecular Docking" Molecules 25, no. 17: 3853. https://doi.org/10.3390/molecules25173853
APA StyleKim, M., Park, K. H., & Kim, Y. B. (2020). Identifying Active Compounds and Targets of Fritillariae thunbergii against Influenza-Associated Inflammation by Network Pharmacology Analysis and Molecular Docking. Molecules, 25(17), 3853. https://doi.org/10.3390/molecules25173853




