The Role of β-Cyclodextrin in the Textile Industry—Review
Abstract
1. Introduction
2. General Characteristics of Cyclodextrins
Host-Guest Complex Formation
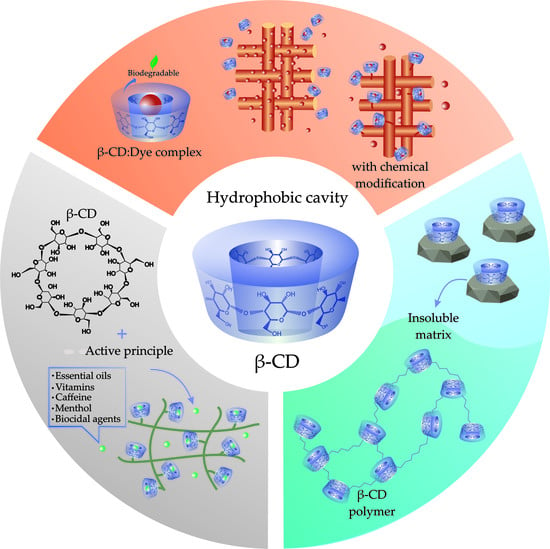
3. Application of Cyclodextrins in the Textile Area
3.1. Dyeing Process
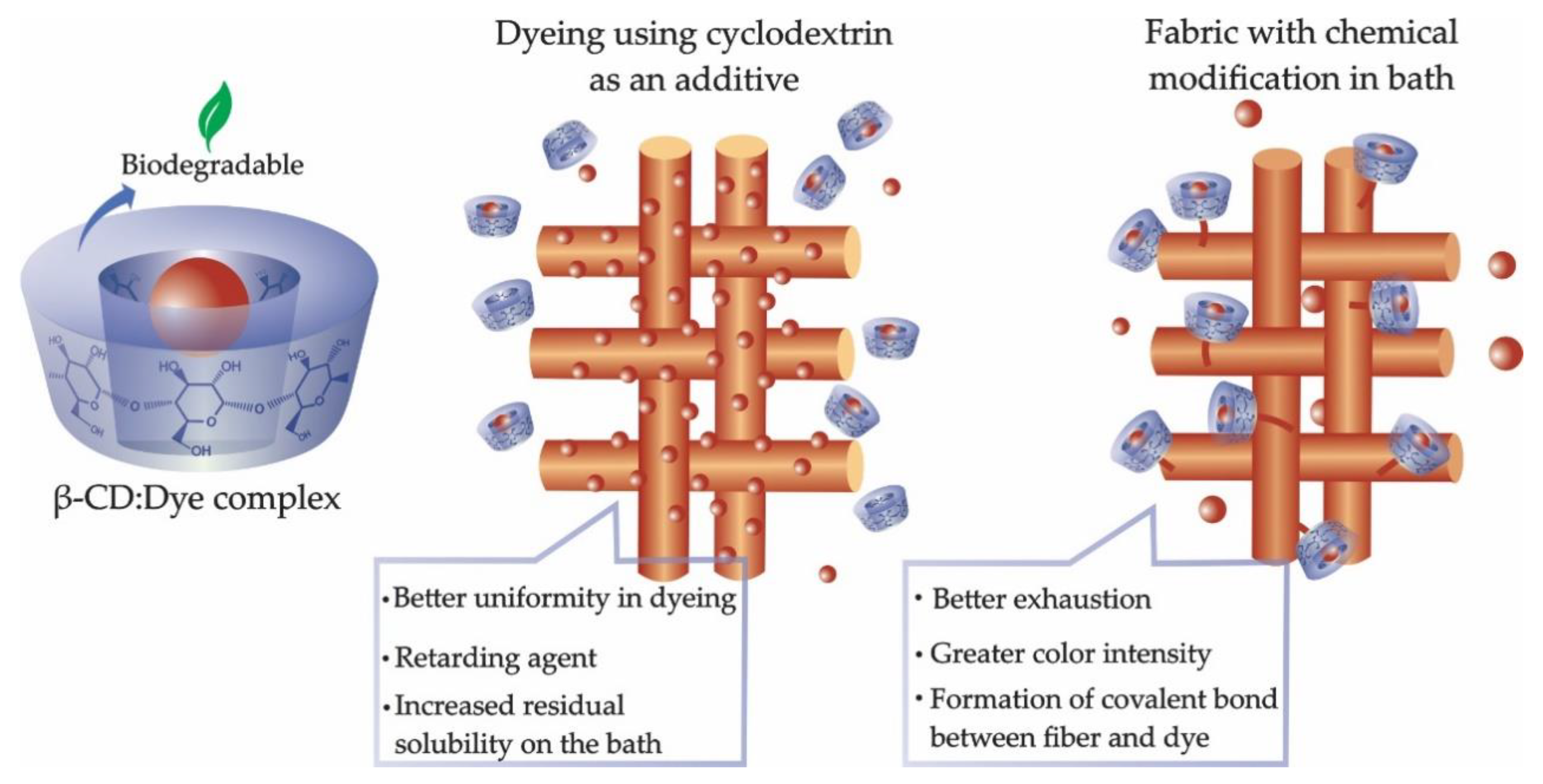
3.1.1. Cyclodextrin as an Auxiliary Agent in Dyeing
3.1.2. Dyeing Chemical Modification
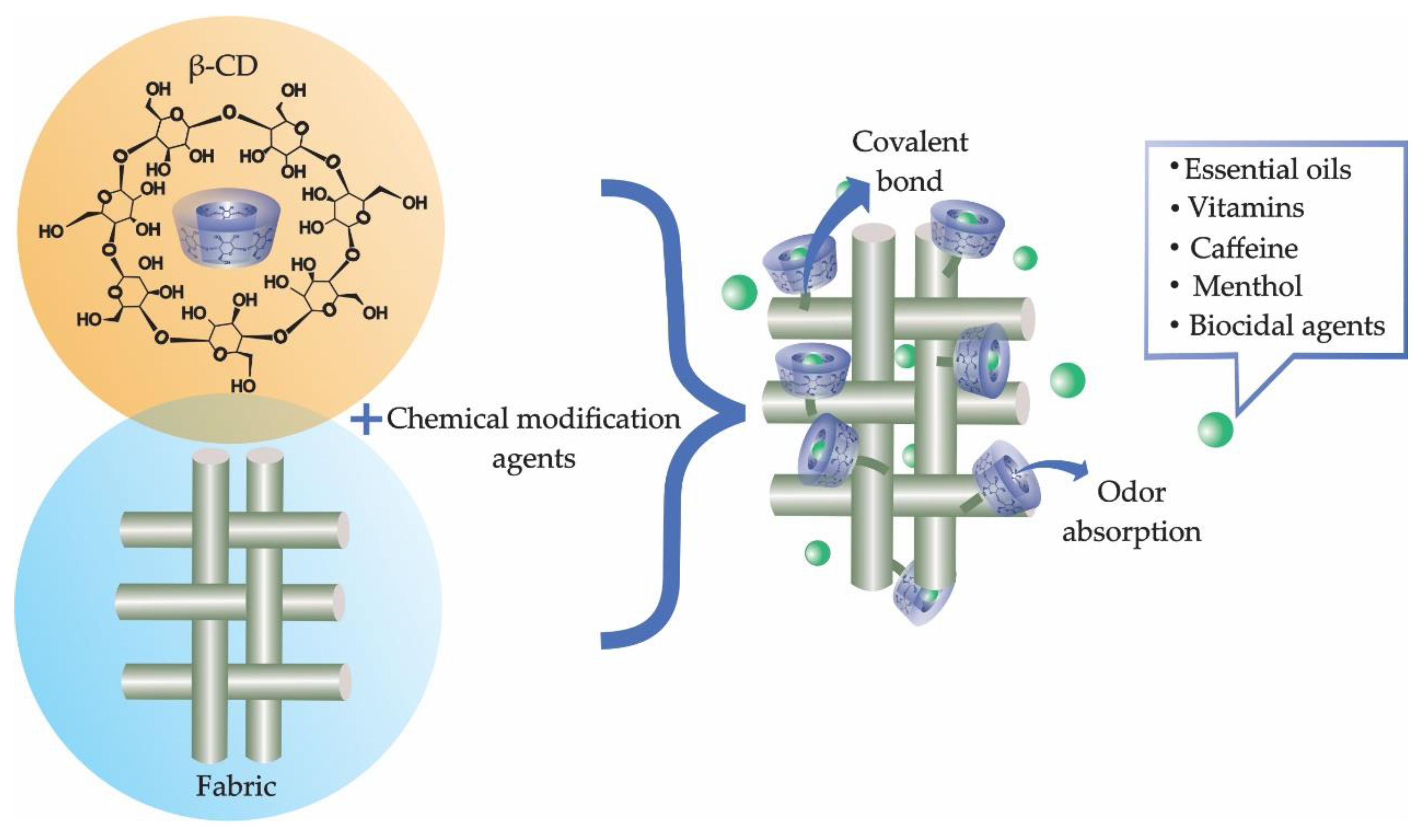
3.2. Textile Finishing
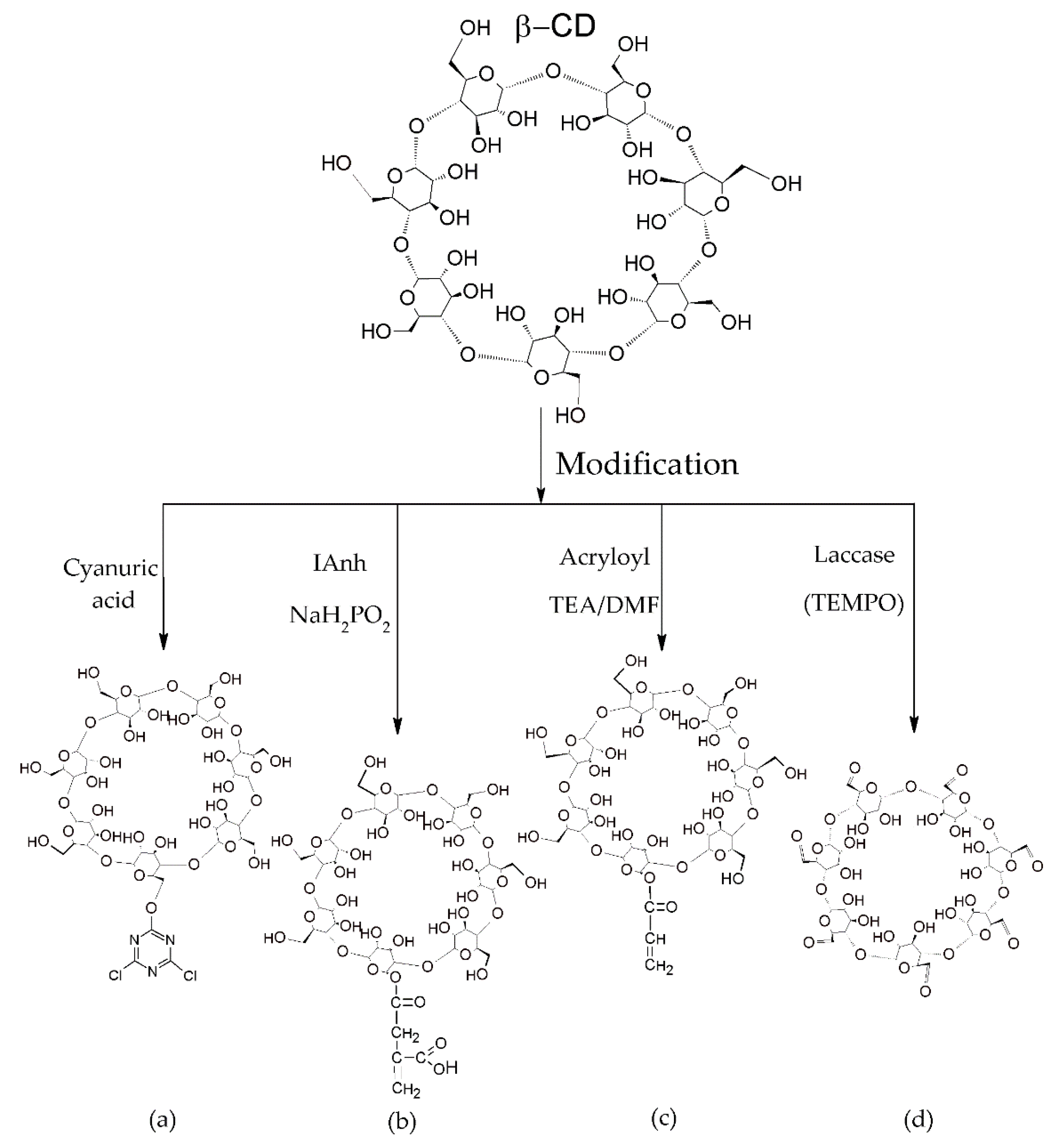
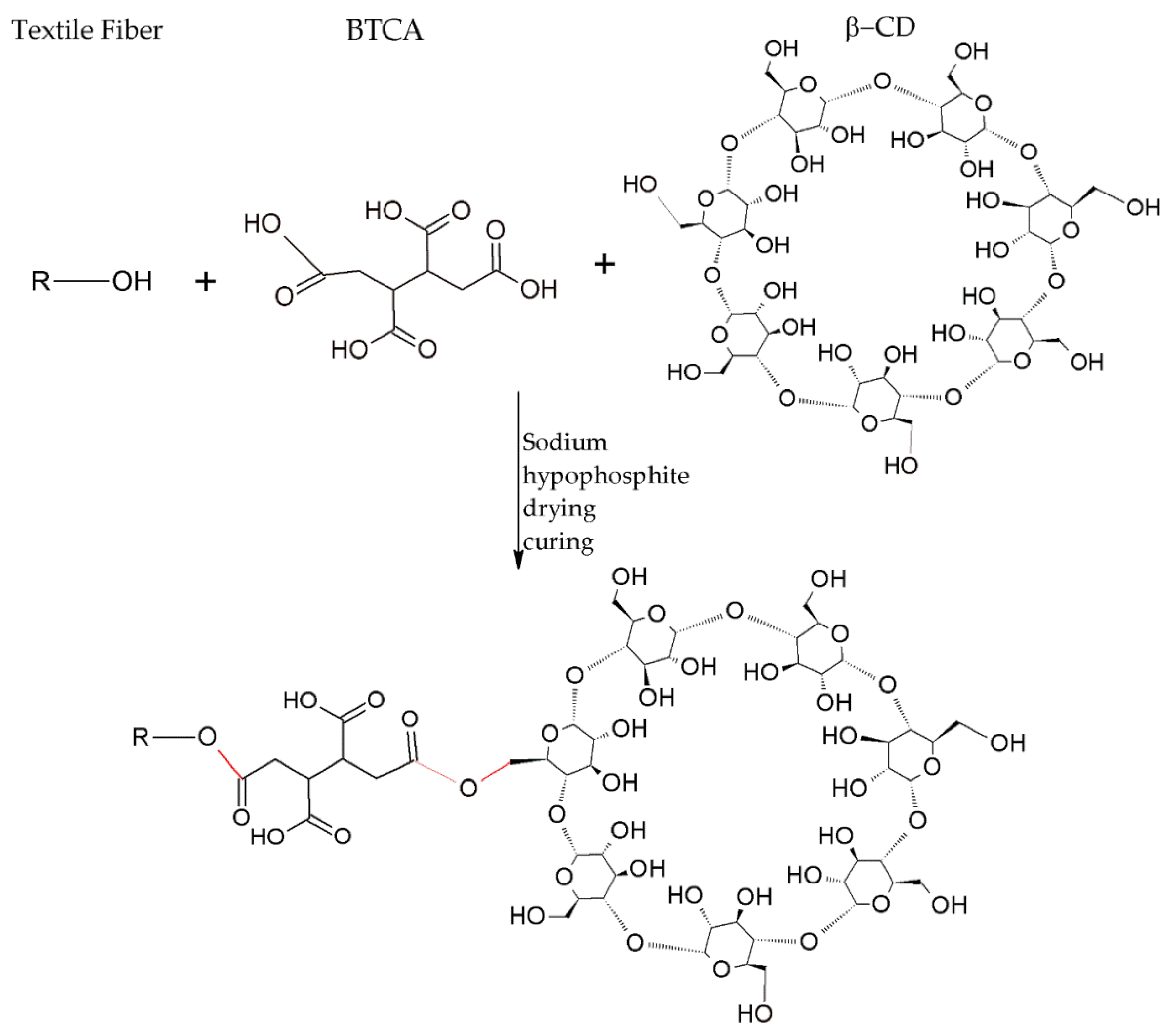
3.2.1. Preparation of Cyclodextrins
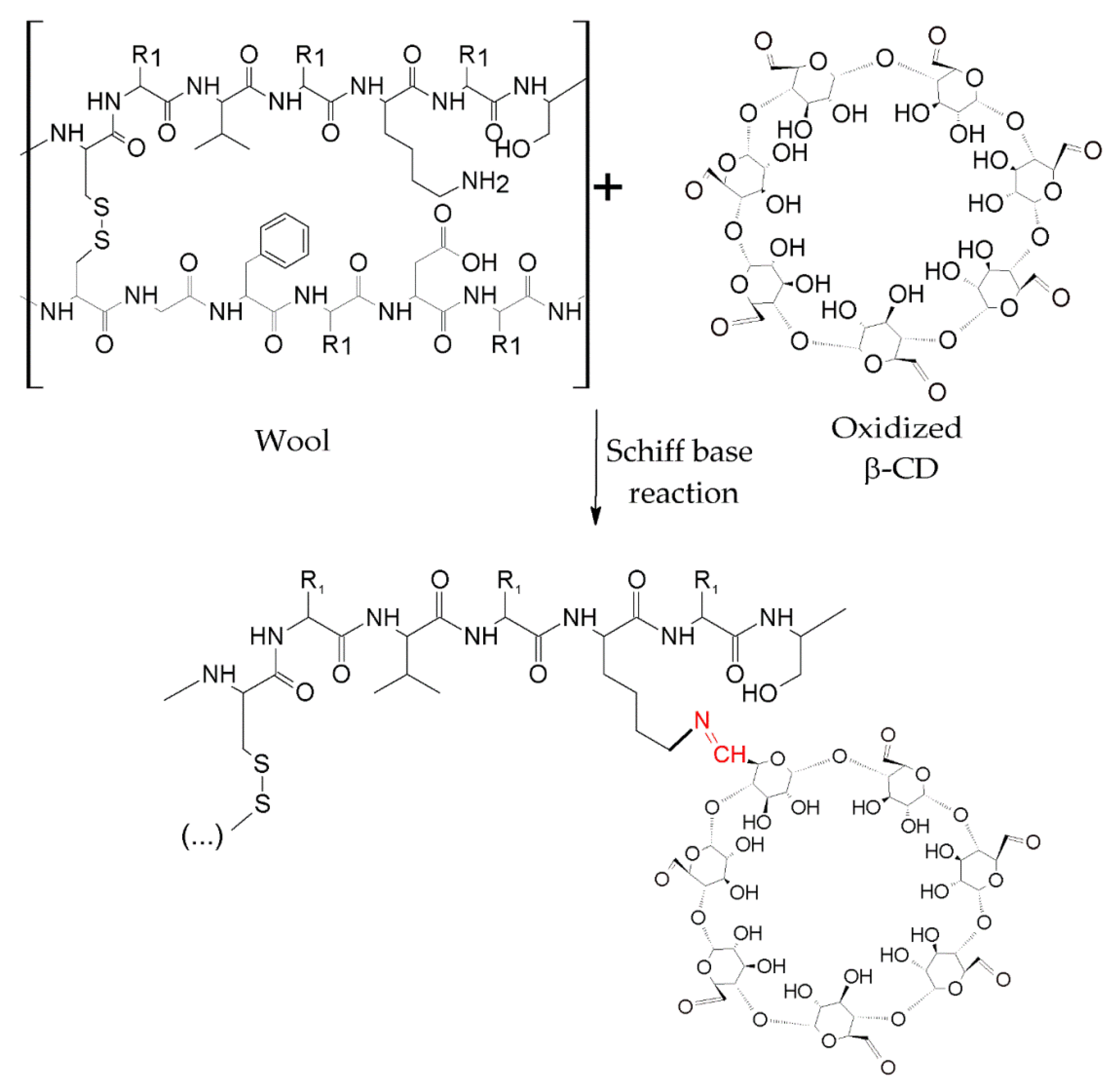
3.2.2. Grafting of Cyclodextrins onto Textile Substrates
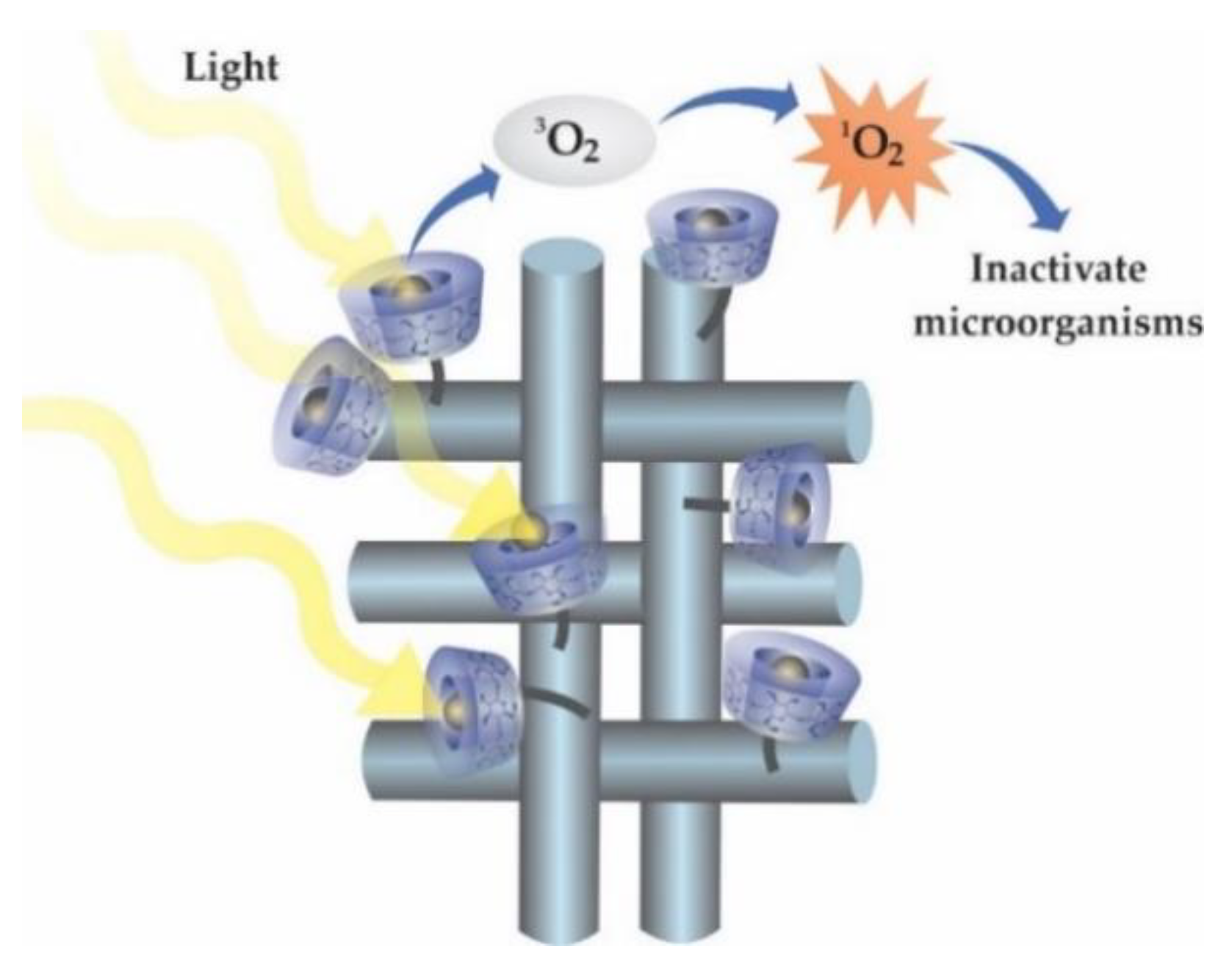
3.3. New Trends in Textile Finishes Using Cyclodextrins
3.4. Cyclodextrins in Textile Effluent Treatment
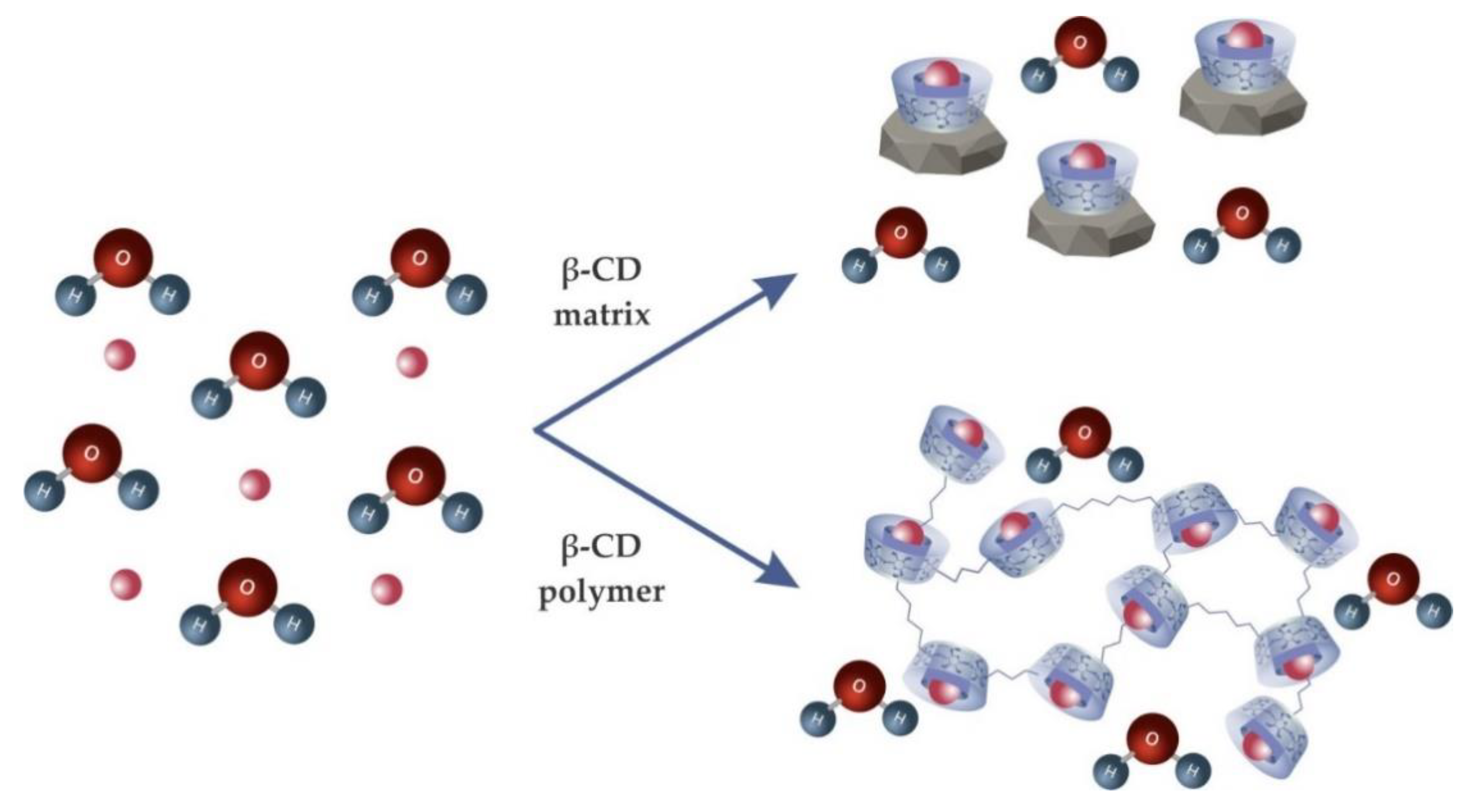
3.4.1. Cyclodextrin Matrix
3.4.2. Cyclodextrin Polymers
4. Final Considerations and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Villiers, A. Sur la fermentation de la fécule par l’action du ferment butyrique. C. R. Acad. Sci. 1891, 112, 536–537. [Google Scholar]
- Szejtli, J. Introduction and General Overview of Cyclodextrin Chemistry. Chem. Rev. 1998, 98, 1743–1754. [Google Scholar] [CrossRef] [PubMed]
- Alonso, C.; Martí, M.; Barba, C.; Lis, M.; Rubio, L.; Coderch, L. Skin penetration and antioxidant effect of cosmeto-textiles with gallic acid. J. Photochem. Photobiol. B Boil. 2016, 156, 50–55. [Google Scholar] [CrossRef]
- Rasheed, A. Cyclodextrins as Drug Carrier Molecule: A Review. Sci. Pharm. 2008, 76, 567–598. [Google Scholar] [CrossRef]
- Radu, C.-D.; Parteni, O.; Ochiuz, L. Applications of cyclodextrins in medical textiles—Review. J. Control. Release 2016, 224, 146–157. [Google Scholar] [CrossRef]
- Nardello-Rataj, V.; Leclercq, L. Encapsulation of biocides by cyclodextrins: Toward synergistic effects against pathogens. Beilstein J. Org. Chem. 2014, 10, 2603–2622. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, L. 17 - Smart medical textiles based on cyclodextrins for curative or preventive patient care. In Active Coatings for Smart Textiles; Hu, J., Ed.; Woodhead Publishing Series in Textiles; Woodhead Publishing: Cambridge, UK, 2016; pp. 391–427. ISBN 978-0-08-100263-6. [Google Scholar]
- Crini, G. Review: A History of Cyclodextrins. Chem. Rev. 2014, 114, 10940–10975. [Google Scholar] [CrossRef]
- Buschmann, H.-J.; Schollmeyer, E. Applications of cyclodextrins in cosmetic products: A review. J. Cosmet. Sci. 2002, 53, 185–191. [Google Scholar]
- Pinho, E.; Grootveld, M.; Soares, G.; Henriques, M. Cyclodextrins as encapsulation agents for plant bioactive compounds. Carbohydr. Polym. 2014, 101, 121–135. [Google Scholar] [CrossRef]
- French, D. The Schardinger Dextrins. In Advances in Carbohydrate Chemistry; Wolfrom, M.L., Tipson, R.S., Eds.; Academic Press: New York, NY, USA, 1957; Volume 12, pp. 189–260. [Google Scholar]
- Fernández, M.A.; Silva, O.F.; Vico, R.V.; de Rossi, R.H. Complex systems that incorporate cyclodextrins to get materials for some specific applications. Carbohydr. Res. 2019, 480, 12–34. [Google Scholar] [CrossRef]
- Loftsson, T.; Duchêne, D. Cyclodextrins and their pharmaceutical applications. Int. J. Pharm. 2007, 329, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Matioli, G. CICLODEXTRINAS E SUAS APLICAÇÕES EM: Alimentos, Fármacos, Cosméticos, Agricultura, Biotecnologia, Química Analítica e Produtos Gerais|Eduem—Editora da UEM, 1st ed.; Eduem: Maringá, Brazil, 2000; ISBN 85-85545-46-1. [Google Scholar]
- Challa, R.; Ahuja, A.; Ali, J.; Khar, R.K. Cyclodextrins in drug delivery: An updated review. AAPS PharmSciTech 2005, 6, E329–E357. [Google Scholar] [CrossRef] [PubMed]
- Schöffer, J.D.N.; Klein, M.P.; Rodrigues, R.C.; Hertz, P.F. Continuous production of β-cyclodextrin from starch by highly stable cyclodextrin glycosyltransferase immobilized on chitosan. Carbohydr. Polym. 2013, 98, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Andreaus, J.; Dalmolin, M.C.; Junior, I.B.D.O.; Barcellos, I.O. Application of cyclodextrins in textile processes. Química Nova 2010, 33, 929–937. [Google Scholar] [CrossRef]
- Amiri, S.; Amiri, S. Cyclodextrins: Properties and Industrial Applications; John Wiley&Sons: Hoboken, NJ, USA, 2017; ISBN 978-1-119-24760-9. [Google Scholar]
- Harata, K. Structural Aspects of Stereodifferentiation in the Solid State. Chem. Rev. 1998, 98, 1803–1828. [Google Scholar] [CrossRef]
- Faisal, Z.; Kunsági-Máté, S.; Lemli, B.; Szente, L.; Bergmann, D.; Humpf, H.-U.; Poór, M. Interaction of Dihydrocitrinone with Native and Chemically Modified Cyclodextrins. Molecules 2019, 24, 1328. [Google Scholar] [CrossRef]
- Del Valle, E.M.M. Cyclodextrins and their uses: A review. Process. Biochem. 2004, 39, 1033–1046. [Google Scholar] [CrossRef]
- Singh, N.; Yadav, M.; Khanna, S.; Sahu, O. Sustainable fragrance cum antimicrobial finishing on cotton: Indigenous essential oil. Sustain. Chem. Pharm. 2017, 5, 22–29. [Google Scholar] [CrossRef]
- Shabbir, M.; Ahmed, S.; Sheikh, J.N. Frontiers of Textile Materials: Polymers, Nanomaterials, Enzymes, and Advanced Modification Techniques; Scrivener Publishing: Beverly, MA, USA, 2020; ISBN 978-1-119-62036-5. [Google Scholar]
- Semeraro, P.; Rizzi, V.; Fini, P.; Matera, S.; Cosma, P.; Franco, E.; García, R.; Ferrándiz, M.; Núñez, E.; Gabaldón, J.A.; et al. Interaction between industrial textile dyes and cyclodextrins. Dye. Pigment. 2015, 119, 84–94. [Google Scholar] [CrossRef]
- Gao, S.; Liu, Y.; Jiang, J.; Ji, Q.; Fu, Y.; Zhao, L.; Li, C.; Ye, F. Physicochemical properties and fungicidal activity of inclusion complexes of fungicide chlorothalonil with β-cyclodextrin and hydroxypropyl-β-cyclodextrin. J. Mol. Liq. 2019, 293, 111513. [Google Scholar] [CrossRef]
- Saenger, W. Cyclodextrin Inclusion Compounds in Research and Industry. Angew. Chem. Int. Ed. 1980, 19, 344–362. [Google Scholar] [CrossRef]
- Stella, V.J.; He, Q. Cyclodextrins. Toxicol. Pathol. 2008, 36, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Irie, T.; Uekama, K. Pharmaceutical Applications of Cyclodextrins. III. Toxicological Issues and Safety Evaluation. J. Pharm. Sci. 1997, 86, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Cunha-Filho, M.S.S.D.; SÁ-BARRETO, L.C.L. Utilização de ciclodextrinas na formação de complexos de inclusão de interesse farmacêutico. Revista de Ciências Farmacêuticas Básica e Aplicada 2007, 28, 1–9. [Google Scholar]
- Bilensoy, E. Cyclodextrins in Pharmaceutics, Cosmetics, and Biomedicine: Current and Future Industrial Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011. [Google Scholar]
- Bhaskara-Amrit, U.R.; Agrawal, P.B.; Warmoeskerken, M.M.C.G. Applications of β-cyclodextrins in textiles. Autex Res. J. 2011, 11, 94–101. [Google Scholar]
- Savarino, P.; Viscardi, G.; Quagliotto, P.; Montoneri, E.; Barni, E. Reactivity and effects of cyclodextrins in textile dyeing. Dye. Pigment. 1999, 42, 143–147. [Google Scholar] [CrossRef]
- Bezerra, F.M.; Moraes, F.F.D.; Santos, W.L.F.; Santos, J.C. Emprego de β-ciclodextrina como auxiliar no tingimento de fibras têxteis. Química Têxt. 2013, 27, 12–21. [Google Scholar]
- Grigoriu, A.; Luca, C.; Grigoriu, A. Cyclodextrins Applications in the Textile Industry. Cellul. Chem. Technol. 2007, 1, 103–112. [Google Scholar]
- Crupi, V.; Ficarra, R.; Guardo, M.; Majolino, D.; Stancanelli, R.; Venuti, V. UV–vis and FTIR–ATR spectroscopic techniques to study the inclusion complexes of genistein with β-cyclodextrins. J. Pharm. Biomed. Anal. 2007, 44, 110–117. [Google Scholar] [CrossRef]
- Abdel-Halim, E.S.; Abdel-Mohdy, F.A.; Fouda, M.M.G.; El-Sawy, S.M.; Hamdy, I.A.; Al-Deyab, S.S. Antimicrobial activity of monochlorotriazinyl-β-cyclodextrin/chlorohexidin diacetate finished cotton fabrics. Carbohydr. Polym. 2011, 86, 1389–1394. [Google Scholar] [CrossRef]
- Marques, H.M.C. A review on cyclodextrin encapsulation of essential oils and volatiles. Flavour Fragr. J. 2010, 25, 313–326. [Google Scholar] [CrossRef]
- Lis, M.J.; Carmona, O.G.; Carmona, C.G.; Bezerra, F.M. Inclusion Complexes of Citronella Oil with β-Cyclodextrin for Controlled Release in Biofunctional Textiles. Polymers 2018, 10, 1324. [Google Scholar] [CrossRef] [PubMed]
- Popescu, V.; Vasluianu, E.; Popescu, G. Quantitative analysis of the multifunctional finishing of cotton fabric with non-formaldehyde agents. Carbohydr. Polym. 2014, 111, 870–882. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Hu, Y.; Xu, G.; Li, M.; Zhu, Y.; Jiang, L.; Tu, Y.; Zhu, X.; Xie, X.; Li, A. Green synthesis of a magnetic β-cyclodextrin polymer for rapid removal of organic micro-pollutants and heavy metals from dyeing wastewater. Environ. Res. 2020, 180, 108796. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.-Y.; Jiang, H.-L.; Xie, Z.-W.; Li, Z.-T.; Xu, D.; He, F.-A. Highly efficient selective adsorption of anionic dyes by modified β-cyclodextrin polymers. J. Taiwan Inst. Chem. Eng. 2020, 108, 114–128. [Google Scholar] [CrossRef]
- Zhou, Y.; Hu, Y.; Huang, W.; Cheng, G.; Cui, C.; Lu, J. A novel amphoteric β-cyclodextrin-based adsorbent for simultaneous removal of cationic/anionic dyes and bisphenol A. Chem. Eng. J. 2018, 341, 47–57. [Google Scholar] [CrossRef]
- Teng, M.; Li, F.; Zhang, B.; Taha, A.A. Electrospun cyclodextrin-functionalized mesoporous polyvinyl alcohol/SiO2 nanofiber membranes as a highly efficient adsorbent for indigo carmine dye. Colloids Surf. A: Physicochem Eng. Asp. 2011, 385, 229–234. [Google Scholar] [CrossRef]
- Nostro, P.L.; Fratoni, L.; Ridi, F.; Baglioni, P. Surface treatments on Tencel fabric: Grafting with β-cyclodextrin. J. Appl. Polym. Sci. 2003, 88, 706–715. [Google Scholar] [CrossRef]
- Perchyonok, V.T.; Oberholzer, T. Cyclodextrins as Oral Drug Carrier Molecular Devices: Origins, Reasons and In-vitro Model Applications. Curr. Org. Chem. 2012, 16, 2365–2378. [Google Scholar] [CrossRef]
- Acartürk, F.; Çelebi, N. Cyclodextrins as Bioavailability Enhancers. In Cyclodextrins in Pharmaceutics, Cosmetics, and Biomedicine; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2011; pp. 45–64. [Google Scholar]
- Andersen, F.M.; Bundgaard, H. Inclusion complexation of metronidazole benzoate with β-cyclodextrin and its depression of anhydrate-hydrate transition in aqueous suspensions. Int. J. Pharm. 1984, 19, 189–197. [Google Scholar] [CrossRef]
- Venturini, C.D.G.; Nicolini, J.; Machado, C.; Machado, V.G. Propriedades e aplicações recentes das ciclodextrinas. Química Nova 2008, 31, 360–368. [Google Scholar] [CrossRef]
- Rekharsky, M.V.; Inoue, Y. Complexation Thermodynamics of Cyclodextrins. Chem. Rev. 1998, 98, 1875–1918. [Google Scholar] [CrossRef] [PubMed]
- Ross, P.D.; Rekharsky, M.V. Thermodynamics of hydrogen bond and hydrophobic interactions in cyclodextrin complexes. Biophys. J. 1996, 71, 2144–2154. [Google Scholar] [CrossRef]
- Loftsson, T.; Björnsdóttir, S.; Pálsdóttir, G.; Bodor, N. The effects of 2-hydroxypropyl-β-cyclodextrin on the solubility and stability of chlorambucil and melphalan in aqueous solution. Int. J. Pharm. 1989, 57, 63–72. [Google Scholar] [CrossRef]
- Pitha, J.; Milecki, J.; Fales, H.; Pannell, L.; Uekama, K. Hydroxypropyl-β-cyclodextrin: Preparation and characterization; effects on solubility of drugs. Int. J. Pharm. 1986, 29, 73–82. [Google Scholar] [CrossRef]
- Loftsson, T. Cyclodextrins and the Biopharmaceutics Classification System of Drugs. J. Incl. Phenom. 2002, 44, 63–67. [Google Scholar] [CrossRef]
- Chow, D.D.; Karara, A.H. Characterization, dissolution and bioavailability in rats of ibuprofen-β-cyclodextrin complex system. Int. J. Pharm. 1986, 28, 95–101. [Google Scholar] [CrossRef]
- Vila-Jato, J.L.; Blanco, J.; Torres, J.J. Biopharmaceutical aspects of the tolbutamide-beta-cyclodextrin inclusion compound. Farm. Prat. 1988, 43, 37–45. [Google Scholar]
- Wang, Z.; Landy, D.; Sizun, C.; Cézard, C.; Solgadi, A.; Przybylski, C.; de Chaisemartin, L.; Herfindal, L.; Barratt, G.; Legrand, F.-X. Cyclodextrin complexation studies as the first step for repurposing of chlorpromazine. Int. J. Pharm. 2020, 584, 119391. [Google Scholar] [CrossRef]
- Kfoury, M.; Landy, D.; Fourmentin, S. Characterization of Cyclodextrin/Volatile Inclusion Complexes: A Review. Molecules 2018, 23, 1204. [Google Scholar] [CrossRef]
- Takahashi, K. Organic Reactions Mediated by Cyclodextrins. Chem. Rev. 1998, 98, 2013–2034. [Google Scholar] [CrossRef] [PubMed]
- Rama, A.C.R.; Veiga, F.; Figueiredo, I.V.; Sousa, A.; Caramona, M. Aspectos biofarmacêuticos da formulação de medicamentos para neonatos: Fundamentos da complexação de indometacina com hidroxipropil-beta-ciclodextrina para tratamento oral do fechamento do canal arterial. Revista Brasileira de Ciências Farmacêuticas 2005, 41, 281–299. [Google Scholar] [CrossRef]
- Charumanee, S.; Titwan, A.; Sirithunyalug, J.; Weiss-Greiler, P.; Wolschann, P.; Viernstein, H.; Okonogi, S. Thermodynamics of the encapsulation by cyclodextrins. J. Chem. Technol. Biotechnol. 2006, 81, 523–529. [Google Scholar] [CrossRef]
- Ameen, H.M.; Kunsági-Máté, S.; Bognár, B.; Szente, L.; Poór, M.; Lemli, B. Thermodynamic Characterization of the Interaction between the Antimicrobial Drug Sulfamethazine and Two Selected Cyclodextrins. Molecules 2019, 24, 4565. [Google Scholar] [CrossRef] [PubMed]
- Vončina, B.; Vivod, V.; Jaušovec, D. β-Cyclodextrin as retarding reagent in polyacrylonitrile dyeing. Dye. Pigment. 2007, 74, 642–646. [Google Scholar] [CrossRef]
- Hedges, A.R. Industrial Applications of Cyclodextrins. Chem. Rev. 1998, 98, 2035–2044. [Google Scholar] [CrossRef]
- Cirri, M.; Maestrelli, F.; Orlandini, S.; Furlanetto, S.; Pinzauti, S.; Mura, P. Determination of stability constant values of flurbiprofen-cyclodextrin complexes using different techniques. J. Pharm. Biomed. Anal. 2005, 37, 995–1002. [Google Scholar] [CrossRef]
- Vozone, C.M.; Marques, H.M.C. Complexation of Budesonide in Cyclodextrins and Particle Aerodynamic Characterization of the Complex Solid Form for Dry Powder Inhalation. J. Incl. Phenom. 2002, 44, 111–116. [Google Scholar] [CrossRef]
- Cao, F.; Guo, J.; Ping, Q. The physicochemical characteristics of freeze-dried scutellarin-cyclodextrin tetracomponent complexes. Drug Dev. Ind. Pharm. 2005, 31, 747–756. [Google Scholar] [CrossRef]
- Miro, A.; Ungaro, F.; Quaglia, F. Cyclodextrins as Smart Excipients in Polymeric Drug Delivery Systems. In Cyclodextrins in Pharmaceutics, Cosmetics, and Biomedicine; John Wiley & Sons, Ltd.: New Jersey, NY, USA, 2011; pp. 65–89. [Google Scholar]
- Buschmann, H.-J.; Denter, U.; Knittel, D.; Schollmeyer, E. The Use of Cyclodextrins in Textile Processes—An Overview. J. Text. Inst. 1998, 89, 554–561. [Google Scholar] [CrossRef]
- Loftsson, T.; Brewster, M.E. Pharmaceutical Applications of Cyclodextrins. 1. Drug Solubilization and Stabilization. J. Pharm. Sci. 1996, 85, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Alzate-Sánchez, D.M.; Smith, B.J.; Alsbaiee, A.; Hinestroza, J.P.; Dichtel, W.R. Cotton Fabric Functionalized with a β-Cyclodextrin Polymer Captures Organic Pollutants from Contaminated Air and Water. Chem. Mater. 2016, 28, 8340–8346. [Google Scholar] [CrossRef]
- Tonelli, A.E. Cyclodextrins as a means to nanostructure and functionalize polymers. J. Incl. Phenom. Macrocycl. Chem. 2008, 60, 197–202. [Google Scholar] [CrossRef]
- Hodul, P.; Duris, M.; Králik, M. Inclusion complexes of B-cyclodextrin with non-ionic surfactants in textile preparation process. Vlakna a Text. 1996, 3, 16–19. [Google Scholar]
- Carpignano, R.; Parlati, S.; Piccinini, P.; Savarino, P.; Giorgi, M.R.D.; Fochi, R. Use of β-cyclodextrin in the dyeing of polyester with low environmental impact. Color. Technol. 2010, 126, 201–208. [Google Scholar] [CrossRef]
- Sricharussin, W.; Sopajaree, C.; Maneerung, T.; Sangsuriya, N. Modification of cotton fabrics with β-cyclodextrin derivative for aroma finishing. J. Text. Inst. 2009, 100, 682–687. [Google Scholar] [CrossRef]
- Raslan, W.M.; El-Aref, A.T.; Bendak, A. Modification of cellulose acetate fabric with cyclodextrin to improve its dyeability. J. Appl. Polym. Sci. 2009, 112, 3192–3198. [Google Scholar] [CrossRef]
- Scacchetti, F.A.P.; Pinto, E.; Soares, G.M.B. Functionalization and characterization of cotton with phase change materials and thyme oil encapsulated in beta-cyclodextrins. Prog. Org. Coatings 2017, 107, 64–74. [Google Scholar] [CrossRef]
- Mihailiasa, M.; Caldera, F.; Li, J.; Peila, R.; Ferri, A.; Trotta, F. Preparation of functionalized cotton fabrics by means of melatonin loaded β-cyclodextrin nanosponges. Carbohydr. Polym. 2016, 142, 24–30. [Google Scholar] [CrossRef]
- Bezerra, F.M.; Carmona, O.G.; Carmona, C.G.; Plath, A.S.; Lis, M. Biofunctional wool using β-cyclodextrins as vehiculizer of citronella oil. Process. Biochem. 2019, 77, 151–158. [Google Scholar] [CrossRef]
- Chang, Y.; Dou, N.; Liu, M.; Jiang, M.; Men, J.; Cui, Y.; Li, R.; Zhu, Y. Efficient removal of anionic dyes from aqueous solution using CTAB and β-cyclodextrin-induced dye aggregation. J. Mol. Liq. 2020, 309, 113021. [Google Scholar] [CrossRef]
- Keskin, N.O.S.; Celebioglu, A.; Sarioglu, O.F.; Uyar, T.; Tekinay, T. Encapsulation of living bacteria in electrospun cyclodextrin ultrathin fibers for bioremediation of heavy metals and reactive dye from wastewater. Colloids Surf. B Biointerfaces 2018, 161, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Crini, G. Studies on adsorption of dyes on beta-cyclodextrin polymer. Bioresour. Technol. 2003, 90, 193–198. [Google Scholar] [CrossRef]
- Cireli, A.; Yurdakul, B. Application of cyclodextrin to the textile dyeing and washing processes. J. Appl. Polym. Sci. 2006, 100, 208–218. [Google Scholar] [CrossRef]
- Park, J.S.; Kim, I.-S. Use of β-cyclodextrin in an antimigration coating for polyester fabric. Color. Technol. 2013, 129, 347–351. [Google Scholar] [CrossRef]
- Kacem, I.; Laurent, T.; Blanchemain, N.; Neut, C.; Chai, F.; Haulon, S.; Hildebrand, H.F.; Martel, B. Dyeing and antibacterial activation with methylene blue of a cyclodextrin modified polyester vascular graft. J. Biomed. Mater. Res. Part A 2014, 102, 2942–2951. [Google Scholar] [CrossRef]
- Dardeer, H.M.; El-sisi, A.A.; Emam, A.A.; Hilal, N.M. Synthesis, Application of a Novel Azo Dye and Its Inclusion Complex with Beta-cyclodextrin onto Polyester Fabric. Int. J. Text. Sci. 2017, 6, 79–87. [Google Scholar]
- Dutra, F.V.; Santos, K.R.M.D.; Bezerra, F.M. Complexação de corantes dispersos utilizando ß-ciclodextrina em tricromia para poliéster. Química Têxt. 2019, 43, 40–49. [Google Scholar]
- Ferreira, B.T.M.; Espinoza-Quiñones, F.R.; Borba, C.E.; Módenes, A.N.; Santos, W.L.F.; Bezerra, F.M. Use of the β-Cyclodextrin Additive as a Good Alternative for the Substitution of Environmentally Harmful Additives in Industrial Dyeing Processes. Fibers Polym. 2020, 21, 1266–1274. [Google Scholar] [CrossRef]
- Savarino, P.; Piccinini, P.; Montoneri, E.; Viscardi, G.; Quagliotto, P.; Barni, E. Effects of additives on the dyeing of nylon-6 with dyes containing hydrophobic and hydrophilic moieties. Dye. Pigment. 2000, 47, 177–188. [Google Scholar] [CrossRef]
- Savarino, P.; Parlati, S.; Buscaino, R.; Piccinini, P.; Degani, I.; Barni, E. Effects of additives on the dyeing of polyamide fibres. Part I: β-cyclodextrin. Dye. Pigment. 2004, 60, 223–232. [Google Scholar] [CrossRef]
- Savarino, P.; Parlati, S.; Buscaino, R.; Piccinini, P.; Barolo, C.; Montoneri, E. Effects of additives on the dyeing of polyamide fibres. Part II: Methyl-β-cyclodextrin. Dye. Pigment. 2006, 69, 7–12. [Google Scholar] [CrossRef]
- Parlati, S.; Gobetto, R.; Barolo, C.; Arrais, A.; Buscaino, R.; Medana, C.; Savarino, P. Preparation and application of a β-cyclodextrin-disperse/reactive dye complex. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 463–470. [Google Scholar] [CrossRef]
- Shibusawa, T.; Okamoto, J.; Abe, K.; Sakata, K.; Ito, Y. Inclusion of azo disperse dyes by cyclodextrins at dyeing temperature. Dye. Pigment. 1998, 36, 79–91. [Google Scholar] [CrossRef]
- Dutra, F.V.; Caruzi, B.B.; Kawasaki, I.; Silva, T.L.; Bezerra, F.M. Utilização de β-ciclodextrina no tingimento de lã com Extratode Urucum (BiXa orellana). Química Têxt. 2014, 37, 58–64. [Google Scholar]
- Chen, L.; Wang, C.; Tian, A.; Wu, M. An attempt of improving polyester inkjet printing performance by surface modification using β-cyclodextrin. Surf. Interface Anal. 2012, 44, 1324–1330. [Google Scholar] [CrossRef]
- Lu, M.; Liu, Y.P. Dyeing Kinetics of Vinylon Modified with β-Cyclodextrin. Fibres Text. East. Eur. 2011, 5, 88. [Google Scholar]
- Rehan, M.; Mahmoud, S.A.; Mashaly, H.M.; Youssef, B.M. β-Cyclodextrin assisted simultaneous preparation and dyeing acid dyes onto cotton fabric. React. Funct. Polym. 2020, 151, 104573. [Google Scholar] [CrossRef]
- Zhang, W.; Ji, X.; Wang, C.; Yin, Y. One-bath one-step low-temperature dyeing of polyester/cotton blended fabric with cationic dyes via β-cyclodextrin modification. Text. Res. J. 2019, 89, 1699–1711. [Google Scholar] [CrossRef]
- Ibrahim, N.A.; El-Zairy, E.M.R. Union disperse printing and UV-protecting of wool/polyester blend using a reactive β-cyclodextrin. Carbohydr. Polym. 2009, 76, 244–249. [Google Scholar] [CrossRef]
- Ghoul, Y.E.; Martel, B.; Achari, A.E.; Campagne, C.; Razafimahefa, L.; Vroman, I. Improved dyeability of polypropylene fabrics finished with β-cyclodextrin–citric acid polymer. Polym. J. 2010, 42, 804–811. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Liu, K.; Salihu, G. The wash-off of dyeings using interstitial water Part 5: Residual dyebath and wash-off liquor generated during the application of disperse dyes and reactive dyes to polyester/cotton fabric. Dye. Pigment. 2019, 171, 106367. [Google Scholar] [CrossRef]
- Hou, A.; Chen, B.; Dai, J.; Zhang, K. Using supercritical carbon dioxide as solvent to replace water in polyethylene terephthalate (PET) fabric dyeing procedures. J. Clean. Prod. 2010, 18, 1009–1014. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Lagonika, K. Sulphur dyes on nylon 6,6. Part 3. Preliminary studies of the nature of dye–fibre interaction. Dye. Pigment. 2006, 69, 185–191. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Son, Y.-A. A comparison of the colour strength and fastness to repeated washing of acid dyes on standard and deep dyeable nylon 6,6. Dye. Pigment. 2006, 70, 156–163. [Google Scholar] [CrossRef]
- Xie, K.; Liu, H.; Wang, X. Surface modification of cellulose with triazine derivative to improve printability with reactive dyes. Carbohydr. Polym. 2009, 78, 538–542. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Mignanelli, M.; Froehling, P.E.; Bide, M.J. The use of dendrimers to modify the dyeing behaviour of reactive dyes on cotton. Dye. Pigment. 2000, 47, 259–267. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Salihu, G. The role of auxiliaries in the immersion dyeing of textile fibres part 2: Analysis of conventional models that describe the manner by which inorganic electrolytes promote direct dye uptake on cellulosic fibres. Dye. Pigment. 2019, 161, 531–545. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Salihu, G. The role of auxiliaries in the immersion dyeing of textile fibres: Part 4 theoretical model to describe the role of liquor ratio in dyeing cellulosic fibres with direct dyes in the absence and presence of inorganic electrolyte. Dye. Pigment. 2019, 161, 565–580. [Google Scholar] [CrossRef]
- Hunger, K. Industrial Dyes: Chemistry, Properties, Applications; Wiley-VCH: Weinheim, Germany, 2003; ISBN 978-3-527-30426-4. [Google Scholar]
- Wang, H.; Lewis, D.M. Chemical modification of cotton to improve fibre dyeability. Color. Technol. 2002, 118, 159–168. [Google Scholar] [CrossRef]
- Udrescu, C.; Ferrero, F.; Periolatto, M. Ultrasound-assisted dyeing of cellulose acetate. Ultrason. Sonochem. 2014, 21, 1477–1481. [Google Scholar] [CrossRef] [PubMed]
- Martel, B.; Ruffin, D.; Weltrowski, M.; Lekchiri, Y.; Morcellet, M. Water-soluble polymers and gels from the polycondensation between cyclodextrins and poly(carboxylic acid)s: A study of the preparation parameters. J. Appl. Polym. Sci. 2005, 97, 433–442. [Google Scholar] [CrossRef]
- Arias, M.J.L.; Coderch, L.; Martí, M.; Alonso, C.; Carmona, O.G.; Carmona, C.G.; Maesta, F. Vehiculation of Active Principles as a Way to Create Smart and Biofunctional Textiles. Materials 2018, 11, 2152. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, A.; Mallard, I.; Landy, D.; Brabie, G.; Nistor, D.; Fourmentin, S. Retention of aroma compounds from Mentha piperita essential oil by cyclodextrins and crosslinked cyclodextrin polymers. Food Chem. 2013, 138, 291–297. [Google Scholar] [CrossRef]
- Peila, R.; Migliavacca, G.; Aimone, F.; Ferri, A.; Sicardi, S. A comparison of analytical methods for the quantification of a reactive β-cyclodextrin fixed onto cotton yarns. Cellulose 2012, 19, 1097–1105. [Google Scholar] [CrossRef]
- Hedayati, N.; Montazer, M.; Mahmoudirad, M.; Toliyat, T. Ketoconazole and Ketoconazole/β-cyclodextrin performance on cotton wound dressing as fungal skin treatment. Carbohydr. Polym. 2020, 240, 116267. [Google Scholar] [CrossRef]
- El-Ghoul, Y. Biological and microbiological performance of new polymer-based chitosan and synthesized amino-cyclodextrin finished polypropylene abdominal wall prosthesis biomaterial. Text. Res. J. 2020, 0040517520926624. [Google Scholar] [CrossRef]
- McQueen, R.H.; Vaezafshar, S. Odor in textiles: A review of evaluation methods, fabric characteristics, and odor control technologies. Text. Res. J. 2019, 90, 1–17. [Google Scholar] [CrossRef]
- Kadam, V.; Kyratzis, I.L.; Truong, Y.B.; Wang, L.; Padhye, R. Air filter media functionalized with β-Cyclodextrin for efficient adsorption of volatile organic compounds. J. Appl. Polym. Sci. 2020, 137, 49228. [Google Scholar] [CrossRef]
- Azizi, N.; Ben Abdelkader, M.; Chevalier, Y.; Majdoub, M. New β-Cyclodextrin-Based Microcapsules for Textiles Uses. Fibers Polym. 2019, 20, 683–689. [Google Scholar] [CrossRef]
- Wang, C.X.; Chen, S.L. Aromachology and its application in the textile field. Fibres Text. East. Eur. 2005, 13, 41–44. [Google Scholar]
- Agrawal, P.B.; Warmoeskerken, M.M.C.G. Permanent fixation of β-cyclodextrin on cotton surface—An assessment between innovative and established approaches. J. Appl. Polym. Sci. 2012, 124, 4090–4097. [Google Scholar] [CrossRef]
- Nazi, M.; Malek, R.M.A.; Kotek, R. Modification of β-cyclodextrin with itaconic acid and application of the new derivative to cotton fabrics. Carbohydr. Polym. 2012, 88, 950–958. [Google Scholar] [CrossRef]
- Zhang, F.; Islam, M.S.; Berry, R.M.; Tam, K.C. β-Cyclodextrin-Functionalized Cellulose Nanocrystals and Their Interactions with Surfactants. ACS Omega 2019, 4, 2102–2110. [Google Scholar] [CrossRef] [PubMed]
- Reuscher, H.; Hirsenkorn, R. BETA W7 MCT—New ways in surface modification. J. Incl. Phenom. Macrocycl. Chem. 1996, 25, 191–196. [Google Scholar] [CrossRef]
- Shown, I.; Murthy, C.N. Grafting of cotton fiber by water-soluble cyclodextrin-based polymer. J. Appl. Polym. Sci. 2009, 111, 2056–2061. [Google Scholar] [CrossRef]
- Nichifor, M.; Constantin, M.; Mocanu, G.; Fundueanu, G.; Branisteanu, D.; Costuleanu, M.; Radu, C.D. New multifunctional textile biomaterials for the treatment of leg venous insufficiency. J. Mater. Sci. Mater. Med. 2009, 20, 975–982. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, Q.; Yuan, J.; Fan, X.; Wang, P. A novel approach for grafting of β-cyclodextrin onto wool via laccase/TEMPO oxidation. Carbohydr. Polym. 2016, 153, 463–470. [Google Scholar] [CrossRef]
- Haji, A.; Mehrizi, M.K.; Akbarpour, R. Optimization of β-cyclodextrin grafting on wool fibers improved by plasma treatment and assessment of antibacterial activity of berberine finished fabric. J. Incl. Phenom. Macrocycl. Chem. 2015, 81, 121–133. [Google Scholar] [CrossRef]
- Montazer, M.; Jolaei, M.M. β-Cyclodextrin stabilized on three-dimensional polyester fabric with different crosslinking agents. J. Appl. Polym. Sci. 2010, 116, 210–217. [Google Scholar] [CrossRef]
- Abdel-Halim, E.S.; Abdel-Mohdy, F.A.; Al-Deyab, S.S.; El-Newehy, M.H. Chitosan and monochlorotriazinyl-β-cyclodextrin finishes improve antistatic properties of cotton/polyester blend and polyester fabrics. Carbohydr. Polym. 2010, 82, 202–208. [Google Scholar] [CrossRef]
- Shi, H.; Wang, Y.; Hua, R. Acid-catalyzed carboxylic acid esterification and ester hydrolysis mechanism: Acylium ion as a sharing active intermediate via a spontaneous trimolecular reaction based on density functional theory calculation and supported by electrospray ionization-mass spectrometry. Phys. Chem. Chem. Phys. 2015, 17, 30279–30291. [Google Scholar] [CrossRef]
- Radu, C.-D.; Salariu, M.; Avadanei, M.; Ghiciuc, C.; Foia, L.; Lupusoru, E.C.; Ferri, A.; Ulea, E.; Lipsa, F. Cotton-made cellulose support for anti-allergic pajamas. Carbohydr. Polym. 2013, 95, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Chakraborty, J.N. Optimization of monochlorotriazine β-cyclodextrin grafting on cotton and assessment of release behavior of essential oils from functionalized fabric. Fash. Text. 2017, 4, 6. [Google Scholar] [CrossRef]
- Ibrahim, N.A.; Abdalla, W.A.; El-Zairy, E.M.R.; Khalil, H.M. Utilization of monochloro-triazine β-cyclodextrin for enhancing printability and functionality of wool. Carbohydr. Polym. 2013, 92, 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- Martel, B.; Weltrowski, M.; Ruffin, D.; Morcellet, M. Polycarboxylic acids as crosslinking agents for grafting cyclodextrins onto cotton and wool fabrics: Study of the process parameters. J. Appl. Polym. Sci. 2002, 83, 1449–1456. [Google Scholar] [CrossRef]
- Liu, S. Bio-Functional Textiles. In Handbook of Medical Textiles; Textiles; Woodhead Publishing: Cambridge, UK, 2011; pp. 336–359. ISBN 978-0-85709-369-1. [Google Scholar]
- Blanchemain, N.; Karrout, Y.; Tabary, N.; Neut, C.; Bria, M.; Siepmann, J.; Hildebrand, H.F.; Martel, B. Methyl-β-cyclodextrin modified vascular prosthesis: Influence of the modification level on the drug delivery properties in different media. Acta Biomater. 2011, 7, 304–314. [Google Scholar] [CrossRef]
- Abdel-Halim, E.S.; Al-Deyab, S.S.; Alfaifi, A.Y.A. Cotton fabric finished with β-cyclodextrin: Inclusion ability toward antimicrobial agent. Carbohydr. Polym. 2014, 102, 550–556. [Google Scholar] [CrossRef]
- Bajpai, M.; Gupta, P.; Bajpai, S.K. Silver (I) ions loaded cyclodextrin-grafted-cotton fabric with excellent antimicrobial property. Fibers Polym. 2010, 11, 8–13. [Google Scholar] [CrossRef]
- Hebeish, A.; El-Shafei, A.; Sharaf, S.; Zaghloul, S. In situ formation of silver nanoparticles for multifunctional cotton containing cyclodextrin. Carbohydr. Polym. 2014, 103, 442–447. [Google Scholar] [CrossRef]
- Rukmani, A.; Sundrarajan, M. Inclusion of antibacterial agent thymol on β-cyclodextrin-grafted organic cotton. J. Ind. Text. 2011, 42, 132–144. [Google Scholar] [CrossRef]
- Selvam, S.; Gandhi, R.R.; Suresh, J.; Gowri, S.; Ravikumar, S.; Sundrarajan, M. Antibacterial effect of novel synthesized sulfated β-cyclodextrin crosslinked cotton fabric and its improved antibacterial activities with ZnO, TiO2 and Ag nanoparticles coating. Int. J. Pharm. 2012, 434, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cai, Z. Incorporation of the antibacterial agent, miconazole nitrate into a cellulosic fabric grafted with β-cyclodextrin. Carbohydr. Polym. 2008, 72, 695–700. [Google Scholar] [CrossRef]
- Cabrales, L.; Abidi, N.; Hammond, A.; Hamood, A. Cotton Fabric Functionalization with Cyclodextrins. Surfaces 2012, 6, 14. [Google Scholar]
- Khanna, S.; Sharma, S.; Chakraborty, J.N. Performance assessment of fragrance finished cotton with cyclodextrin assisted anchoring hosts. Fash. Text. 2015, 2, 19. [Google Scholar] [CrossRef]
- Abdel-Mohdy, F.A.; Fouda, M.M.G.; Rehan, M.F.; Aly, A.S. Repellency of controlled-release treated cotton fabrics based on cypermethrin and prallethrin. Carbohydr. Polym. 2008, 73, 92–97. [Google Scholar] [CrossRef]
- Radu, C.D.; Parteni, O.; Popa, M.; Muresan, I.E.; Ochiuz, L.; Bulgariu, L.; Munteanu, C.; Istrate, B.; Ulea, E. Comparative Study of a Drug Release from a Textile to Skin. J. Pharm. Drug Deliv. Res. 2016, 2015, 1–8. [Google Scholar] [CrossRef]
- El-Ghoul, Y.; Blanchemain, N.; Laurent, T.; Campagne, C.; El Achari, A.; Roudesli, S.; Morcellet, M.; Martel, B.; Hildebrand, H.F. Chemical, biological and microbiological evaluation of cyclodextrin finished polyamide inguinal meshes. Acta Biomater. 2008, 4, 1392–1400. [Google Scholar] [CrossRef]
- Scalia, S.; Tursilli, R.; Bianchi, A.; Nostro, P.L.; Bocci, E.; Ridi, F.; Baglioni, P. Incorporation of the sunscreen agent, octyl methoxycinnamate in a cellulosic fabric grafted with β-cyclodextrin. Int. J. Pharm. 2006, 308, 155–159. [Google Scholar] [CrossRef]
- Blanchemain, N.; Karrout, Y.; Tabary, N.; Bria, M.; Neut, C.; Hildebrand, H.F.; Siepmann, J.; Martel, B. Comparative study of vascular prostheses coated with polycyclodextrins for controlled ciprofloxacin release. Carbohydr. Polym. 2012, 90, 1695–1703. [Google Scholar] [CrossRef]
- Shlar, I.; Droby, S.; Rodov, V. Antimicrobial coatings on polyethylene terephthalate based on curcumin/cyclodextrin complex embedded in a multilayer polyelectrolyte architecture. Colloids Surf. B Biointerfaces 2018, 164, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Tabary, N.; Leclercq, L.; Junthip, J.; Degoutin, S.; Aubert-Viard, F.; Cazaux, F.; Lyskawa, J.; Janus, L.; Bria, M.; et al. Multilayered textile coating based on a β-cyclodextrin polyelectrolyte for the controlled release of drugs. Carbohydr. Polym. 2013, 93, 718–730. [Google Scholar] [CrossRef] [PubMed]
- Martel, B.; Morcellet, M.; Ruffin, D.; Vinet, F.; Weltrowski, L. Capture and Controlled Release of Fragrances by CD Finished Textiles. J. Incl. Phenom. 2002, 44, 439–442. [Google Scholar] [CrossRef]
- Castriciano, M.A.; Zagami, R.; Casaletto, M.P.; Martel, B.; Trapani, M.; Romeo, A.; Villari, V.; Sciortino, M.T.; Grasso, L.; Guglielmino, S.; et al. Poly(carboxylic acid)-Cyclodextrin/Anionic Porphyrin Finished Fabrics as Photosensitizer Releasers for Antimicrobial Photodynamic Therapy. Biomacromolecules 2017, 18, 1134–1144. [Google Scholar] [CrossRef]
- Yao, T.; Wang, J.; Xue, Y.; Yu, W.; Gao, Q.; Ferreira, L.; Ren, K.-F.; Ji, J. A photodynamic antibacterial spray-coating based on the host–guest immobilization of the photosensitizer methylene blue. J. Mater. Chem. B 2019, 7, 5089–5095. [Google Scholar] [CrossRef]
- Jiang, H.-L.; Xu, M.-Y.; Xie, Z.-W.; Hai, W.; Xie, X.-L.; He, F.-A. Selective adsorption of anionic dyes from aqueous solution by a novel β-cyclodextrin-based polymer. J. Mol. Struct. 2020, 1203, 127373. [Google Scholar] [CrossRef]
- Chen, J.; Liu, M.; Pu, Y.; Wang, C.; Han, J.; Jiang, M.; Liu, K. The preparation of thin-walled multi-cavities β-cyclodextrin polymer and its static and dynamic properties for dyes removal. J. Environ. Manag. 2019, 245, 105–113. [Google Scholar] [CrossRef]
- Afkhami, A.; Saber-Tehrani, M.; Bagheri, H. Modified maghemite nanoparticles as an efficient adsorbent for removing some cationic dyes from aqueous solution. Desalination 2010, 263, 240–248. [Google Scholar] [CrossRef]
- Zare, K.; Gupta, V.K.; Moradi, O.; Makhlouf, A.S.H.; Sillanpää, M.; Nadagouda, M.N.; Sadegh, H.; Shahryari-ghoshekandi, R.; Pal, A.; Wang, Z.; et al. A comparative study on the basis of adsorption capacity between CNTs and activated carbon as adsorbents for removal of noxious synthetic dyes: A review. J. Nanostruct. Chem. 2015, 5, 227–236. [Google Scholar] [CrossRef]
- Sansuk, S.; Srijaranai, S.; Srijaranai, S. A New Approach for Removing Anionic Organic Dyes from Wastewater Based on Electrostatically Driven Assembly. Environ. Sci. Technol. 2016, 50, 6477–6484. [Google Scholar] [CrossRef]
- Debnath, S.; Ballav, N.; Maity, A.; Pillay, K. Competitive adsorption of ternary dye mixture using pine cone powder modified with β-cyclodextrin. J. Mol. Liq. 2017, 225, 679–688. [Google Scholar] [CrossRef]
- Abd-Elhamid, A.I.; El-Aassar, M.R.; El Fawal, G.F.; Soliman, H.M.A. Fabrication of polyacrylonitrile/β-cyclodextrin/graphene oxide nanofibers composite as an efficient adsorbent for cationic dye. Environ. NanoTechnol. Monit. Manag. 2019, 11, 100207. [Google Scholar] [CrossRef]
- Batista, L.M.B.; dos Santos, A.J.; da Silva, D.R.; de Melo Alves, A.P.; Garcia-Segura, S.; Martínez-Huitle, C.A. Solar photocatalytic application of NbO2OH as alternative photocatalyst for water treatment. Sci. Total Environ. 2017, 596–597, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Basha, C.A.; Selvakumar, K.V.; Prabhu, H.J.; Sivashanmugam, P.; Lee, C.W. Degradation studies for textile reactive dye by combined electrochemical, microbial and photocatalytic methods. Sep. Purif. Technol. 2011, 79, 303–309. [Google Scholar] [CrossRef]
- Chollom, M.N.; Rathilal, S.; Pillay, V.L.; Alfa, D. The applicability of nanofiltration for the treatment and reuse of textile reactive dye effluent. Water SA 2015, 41, 398–405. [Google Scholar] [CrossRef]
- Yeap, K.L.; Teng, T.T.; Poh, B.T.; Morad, N.; Lee, K.E. Preparation and characterization of coagulation/flocculation behavior of a novel inorganic–organic hybrid polymer for reactive and disperse dyes removal. Chem. Eng. J. 2014, 243, 305–314. [Google Scholar] [CrossRef]
- Gosavi, V.D.; Sharma, S. A General Review on Various Treatment Methods for Textile Wastewater. Available online: /paper/A-General-Review-on-Various-Treatment-Methods-for-Gosavi-Sharma/e8d649881280265b4bf146b25f6e5eb2ddb99afb (accessed on 14 June 2020).
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.; Thomaidis, N.S.; Xu, J. Progress in the biological and chemical treatment technologies for emerging contaminant removal from wastewater: A critical review. J. Hazard. Mater. 2017, 323, 274–298. [Google Scholar] [CrossRef]
- Lin, Q.; Gao, M.; Chang, J.; Ma, H. Adsorption properties of crosslinking carboxymethyl cellulose grafting dimethyldiallylammonium chloride for cationic and anionic dyes. Carbohydr. Polym. 2016, 151, 283–294. [Google Scholar] [CrossRef]
- Crini, G. Kinetic and equilibrium studies on the removal of cationic dyes from aqueous solution by adsorption onto a cyclodextrin polymer. Dye. Pigment. 2008, 77, 415–426. [Google Scholar] [CrossRef]
- Pellicer, J.A.; Rodríguez-López, M.I.; Fortea, M.I.; Lucas-Abellán, C.; Mercader-Ros, M.T.; López-Miranda, S.; Gómez-López, V.M.; Semeraro, P.; Cosma, P.; Fini, P.; et al. Adsorption Properties of β- and Hydroxypropyl-β-Cyclodextrins Cross-Linked with Epichlorohydrin in Aqueous Solution. A Sustainable Recycling Strategy in Textile Dyeing Process. Polymers 2019, 11, 252. [Google Scholar] [CrossRef]
- Crini, G. Recent developments in polysaccharide-based materials used as adsorbents in wastewater treatment. Prog. Polym. Sci. 2005, 30, 38–70. [Google Scholar] [CrossRef]
- Eli, W.; Chen, W.; Xue, Q. The association of anionic surfactants with β -cyclodextrin. An isothermal titration calorimeter study. J. Chem. Thermodyn. 1999, 31, 1283–1296. [Google Scholar] [CrossRef]
- Zhao, R.; Wang, Y.; Li, X.; Sun, B.; Jiang, Z.; Wang, C. Water-insoluble sericin/β-cyclodextrin/PVA composite electrospun nanofibers as effective adsorbents towards methylene blue. Colloids Surf. B Biointerfaces 2015, 136, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.; Yilmaz, E.; Yilmaz, M.; Bartsch, R.A. Removal of azo dyes from aqueous solutions using calix[4]arene and β-cyclodextrin. Dye. Pigment. 2007, 74, 54–59. [Google Scholar] [CrossRef]
- Liu, J.; Liu, G.; Liu, W. Preparation of water-soluble β-cyclodextrin/poly(acrylic acid)/graphene oxide nanocomposites as new adsorbents to remove cationic dyes from aqueous solutions. Chem. Eng. J. 2014, 257, 299–308. [Google Scholar] [CrossRef]
- Mohammadi, A.; Veisi, P. High adsorption performance of β-cyclodextrin-functionalized multi-walled carbon nanotubes for the removal of organic dyes from water and industrial wastewater. J. Environ. Chem. Eng. 2018, 6, 4634–4643. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Lazaridis, N.K.; Bikiaris, D.N. Optimization of chitosan and β-cyclodextrin molecularly imprinted polymer synthesis for dye adsorption. Carbohydr. Polym. 2013, 91, 198–208. [Google Scholar] [CrossRef]
- Ozmen, E.Y.; Sezgin, M.; Yilmaz, A.; Yilmaz, M. Synthesis of β-cyclodextrin and starch based polymers for sorption of azo dyes from aqueous solutions. Bioresour. Technol. 2008, 99, 526–531. [Google Scholar] [CrossRef]
- Yilmaz, E.; Memon, S.; Yilmaz, M. Removal of direct azo dyes and aromatic amines from aqueous solutions using two β-cyclodextrin-based polymers. J. Hazard. Mater. 2010, 174, 592–597. [Google Scholar] [CrossRef]
- Li, X.; Zhou, M.; Jia, J.; Jia, Q. A water-insoluble viologen-based β-cyclodextrin polymer for selective adsorption toward anionic dyes. React. Funct. Polym. 2018, 126, 20–26. [Google Scholar] [CrossRef]
- Kekes, T.; Tzia, C. Adsorption of indigo carmine on functional chitosan and β-cyclodextrin/chitosan beads: Equilibrium, kinetics and mechanism studies. J. Environ. Manag. 2020, 262, 110372. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-Y.; Tung, S.-H. One-Step Electrospinning to Produce Nonsolvent-Induced Macroporous Fibers with Ultrahigh Oil Adsorption Capability. Macromolecules 2017, 50, 2528–2534. [Google Scholar] [CrossRef]
- Akinyeye, O.J.; Ibigbami, T.B.; Odeja, O. Effect of Chitosan Powder Prepared from Snail Shells to Remove Lead (II) Ion and Nickel (II) Ion from Aqueous Solution and Its Adsorption Isotherm Model. Am. J. Appl. Chem. 2016, 4, 146. [Google Scholar] [CrossRef]
- Chatterjee, S.; Lee, D.S.; Lee, M.W.; Woo, S.H. Nitrate removal from aqueous solutions by cross-linked chitosan beads conditioned with sodium bisulfate. J. Hazard. Mater. 2009, 166, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Cestari, A.R.; Vieira, E.F.S.; Mota, J.A. The removal of an anionic red dye from aqueous solutions using chitosan beads—The role of experimental factors on adsorption using a full factorial design. J. Hazard. Mater. 2008, 160, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zou, Z.; Ren, R.; Sui, X.; Mao, Z.; Xu, H.; Zhong, Y.; Zhang, L.; Wang, B. Chitosan adsorbent reinforced with citric acid modified β-cyclodextrin for highly efficient removal of dyes from reactive dyeing effluents. Eur. Polym. J. 2018, 108, 212–218. [Google Scholar] [CrossRef]
- Krause, R.W.M.; Mamba, B.B.; Bambo, F.M.; Malefetse, T.J. Cyclodextrin polymers: Synthesis and Application in Water Treatment. In Cyclodextrins: Chemistry and Physics; Transworld Research Network: Kerala, India, 2010; ISBN 978-81-7895-430-1. [Google Scholar]
- Vahedi, S.; Tavakoli, O.; Khoobi, M.; Ansari, A.; Faramarzi, M.A. Application of novel magnetic β-cyclodextrin-anhydride polymer nano-adsorbent in cationic dye removal from aqueous solution. J. Taiwan Inst. Chem. Eng. 2017, 80, 452–463. [Google Scholar] [CrossRef]
- Massaro, M.; Colletti, C.G.; Lazzara, G.; Guernelli, S.; Noto, R.; Riela, S. Synthesis and Characterization of Halloysite–Cyclodextrin Nanosponges for Enhanced Dyes Adsorption. ACS Sustain. Chem. Eng. 2017, 5, 3346–3352. [Google Scholar] [CrossRef]
- Ozmen, E.Y.; Yilmaz, M. Use of β-cyclodextrin and starch based polymers for sorption of Congo red from aqueous solutions. J. Hazard. Mater. 2007, 148, 303–310. [Google Scholar] [CrossRef]
- Jiang, H.-L.; Lin, J.-C.; Hai, W.; Tan, H.-W.; Luo, Y.-W.; Xie, X.-L.; Cao, Y.; He, F.-A. A novel crosslinked β-cyclodextrin-based polymer for removing methylene blue from water with high efficiency. Colloids Surf. A Physicochem. Eng. Asp. 2019, 560, 59–68. [Google Scholar] [CrossRef]
- Zhao, F.; Repo, E.; Yin, D.; Meng, Y.; Jafari, S.; Sillanpää, M. EDTA-Cross-Linked β-Cyclodextrin: An Environmentally Friendly Bifunctional Adsorbent for Simultaneous Adsorption of Metals and Cationic Dyes. Environ. Sci. Technol. 2015, 49, 10570–10580. [Google Scholar] [CrossRef] [PubMed]



| Properties | α-CD | β-CD | γ-CD |
|---|---|---|---|
| Empirical formula | C36H60O30 | C42H70O35 | C48H80O40 |
| Molecular weight (g/mol) | 972 | 1135 | 1297 |
| Glucopyranose units | 6 | 7 | 8 |
| Cavity diameter (nm) | 0.47–0.57 | 0.60–0.78 | 0.83–0.95 |
| Internal cavity volume (nm3) | 1740 | 2620 | 4720 |
| Number of water molecules in the cavity | 6 | 11 | 17 |
| Aqueous solubility (g/L) | 129.5 | 18.4 | 249.2 |
| Temperature of degradation (°C) | 278 | 298 | 267 |
| Application of Cyclodextrins | Fiber | Dye | Reference |
|---|---|---|---|
| Auxiliary agent | Polyester | Disperse | [73] |
| Synthetic | [85] | ||
| Disperse Orange 30, Disperse Red 167, Disperse Blue 79 | [86] | ||
| Methylene Blue | [84] | ||
| Polyamide 6 | Disperse Red 60 | [87] | |
| Disperse | [88] | ||
| Synthetic | [32,89,90] | ||
| Nylon, polyester and cotton | Synthetic, reactive and disperse dye | [91] | |
| Cellulose Acetate | Azo disperse | [92] | |
| Polyacrylic | Basic Blue 4 | [62] | |
| Cotton | Direct | [82] | |
| Wool | Natural (Bixa orellana) | [93] | |
| Chemical modification | Polyester | Pigment inks carbon black, magenta, yellow and cyan | [94] |
| Disperse Red 60, Disperse Yellow, Disperse Blue 56, Disperse Red 343 | [83] | ||
| Cellulose Acetate | Disperse Red 60 and 82 | [75] | |
| Vinylon fibre | Reactive Red 2 | [95] | |
| Cotton | Acid | [96] | |
| Cotton and cotton/polyester | Basic Red 14, Basic Blue 3, Basic Yellow 24 and 13 | [97] | |
| Polyester/Wool | Disperse Red 54 and 167, Disperse blue 183 | [98] | |
| Polypropylene | Disperse, acid and reactive | [99] |
| Fiber | Effect | Active Molecule | Reference |
|---|---|---|---|
| Cotton | Antimicrobial | Octenidine dihydrochloride | [138] |
| Silver | [139,140] | ||
| Phenolic compounds | [76,141] | ||
| Ketoconazole | [115] | ||
| ZnO, TiO2 and Ag nanoparticles | [142] | ||
| Miconazole nitrate | [143] | ||
| Triclosan | [144] | ||
| Fragrance, antimicrobial | Essential Oils | [74,145] | |
| Insect repellent | Cypermethrin and Prallethrin | [146] | |
| Nocturnal regulation of sleep and antioxidant properties | Melatonin | [77] | |
| For coetaneous affections | Hydrocortisone acetate | [147] | |
| Polyamide | Perfume, moisturize and UV-protect. | 2-ethoxynaphtalene (neroline) | [119] |
| Antibiotics | Ciprofloxacin | [148] | |
| Tencel | Sunscreen | Octyl methoxycinnamate | [149] |
| Fragrance, antimicrobial and insect repellent | Vanillin, benzoic acid andIodine, N,N-diethyl-m-toluamide and dimethyl-phthalate | [44] | |
| Polyester | Antibiotics | Ciprofloxacin | [150] |
| Antimicrobial | Curcumin | [151] | |
| 4-tert-butylbenzoic acid | [152] | ||
| Wool | Insect repellent | Citronella essential oil | [78] |
| Cotton and Polyester | Insect repellent | Citronella essential oil | [38] |
| Cotton, wool and polyester | Fragrance | β-citronellol, camphor, menthol, cis-jasmone and benzyl acetate | [153] |
| Method | Dye | Reference |
|---|---|---|
| Cyclodextrin incorporated into a matrix | Crystal Violet | [162] |
| Reactive Black 5 | [80] | |
| Methylene Blue | [174] | |
| Methyl Orange | [175] | |
| Safranin O, Brilliant Green and Methylene Blue | [161] | |
| Methylene Blue and Safranine T | [176] | |
| Methylene Blue, Acid Blue 113, Methyl Orange and Disperse Red 1 | [177] | |
| Remazol Red 3BS, Remazol Blue RN, Remazol Yellow gelb 3RS 133 | [178] | |
| Methyl Blue | [169] | |
| β-cyclodextrin polymer | Acid Blue 25, Reactive Blue 19, Disperse Blue 3, Basic Blue 3 and Direct Red 81 | [81] |
| Basic Blue 3, Basic Violet 3 and Basic Violet 10 | [170] | |
| Direct Violent 51, Methyl Orange, And Tropaeolin 000 | [179] | |
| Congo Red and Methylene Blue | [41] | |
| Evans Blue, Chicago Sky Blue, Benzidine, P- hloroaniline | [180] | |
| Methylene Blue And Methyl Orange | [42] | |
| Congo Red, Methylene Blue, Methylene Orange | [156] | |
| Basic Orange 2, Rhodamine B, Methylene blue trihydrate, and Bisphenol A | [40] | |
| Methyl orange, Congo Red, Rhodamine B | [181] | |
| Direct Red 83:1 | [171] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bezerra, F.M.; Lis, M.J.; Firmino, H.B.; Dias da Silva, J.G.; Curto Valle, R.d.C.S.; Borges Valle, J.A.; Scacchetti, F.A.P.; Tessaro, A.L. The Role of β-Cyclodextrin in the Textile Industry—Review. Molecules 2020, 25, 3624. https://doi.org/10.3390/molecules25163624
Bezerra FM, Lis MJ, Firmino HB, Dias da Silva JG, Curto Valle RdCS, Borges Valle JA, Scacchetti FAP, Tessaro AL. The Role of β-Cyclodextrin in the Textile Industry—Review. Molecules. 2020; 25(16):3624. https://doi.org/10.3390/molecules25163624
Chicago/Turabian StyleBezerra, Fabricio Maestá, Manuel José Lis, Helen Beraldo Firmino, Joyce Gabriella Dias da Silva, Rita de Cassia Siqueira Curto Valle, José Alexandre Borges Valle, Fabio Alexandre Pereira Scacchetti, and André Luiz Tessaro. 2020. "The Role of β-Cyclodextrin in the Textile Industry—Review" Molecules 25, no. 16: 3624. https://doi.org/10.3390/molecules25163624
APA StyleBezerra, F. M., Lis, M. J., Firmino, H. B., Dias da Silva, J. G., Curto Valle, R. d. C. S., Borges Valle, J. A., Scacchetti, F. A. P., & Tessaro, A. L. (2020). The Role of β-Cyclodextrin in the Textile Industry—Review. Molecules, 25(16), 3624. https://doi.org/10.3390/molecules25163624