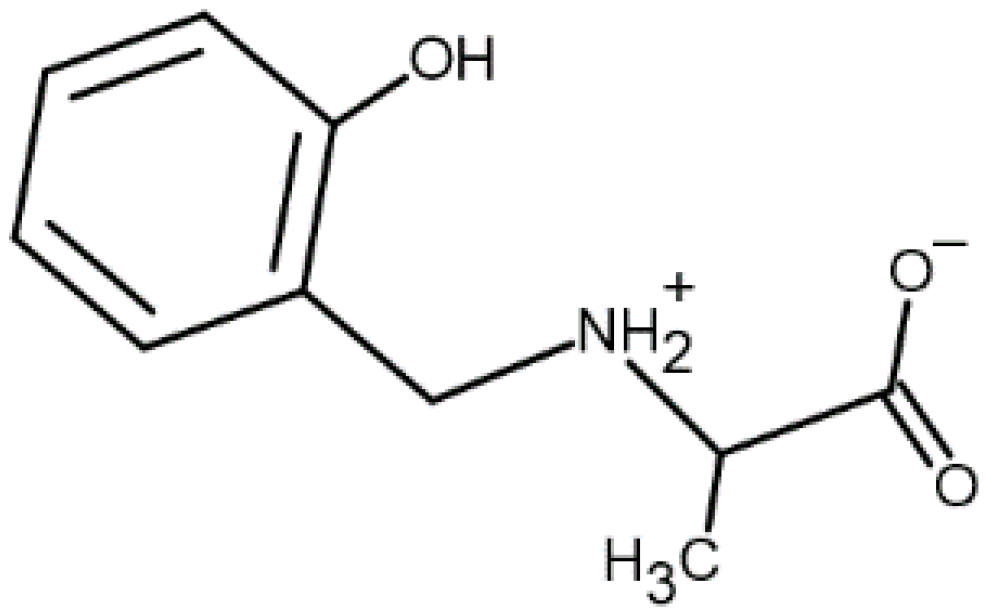
Equilibria in Aqueous Cobalt(II)—Reduced Schiff Base N-(2-hydroxybenzyl)alanine System: Chemical Characterization, Kinetic Analysis, Antimicrobial and Cytotoxic Properties
Abstract
1. Introduction
2. Results and Discussions
2.1. Protonation and Complex Formation Equilibria
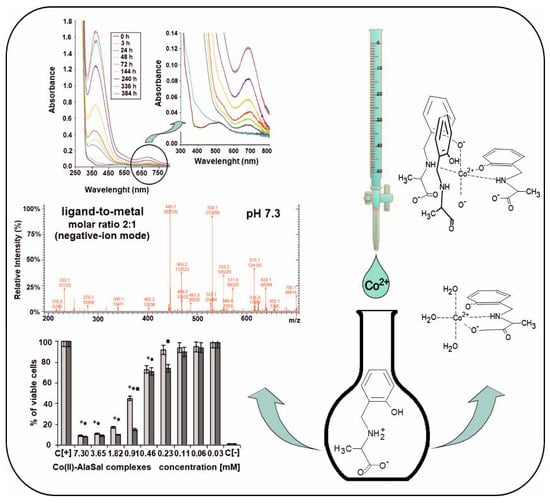
2.2. ESI-MS Results
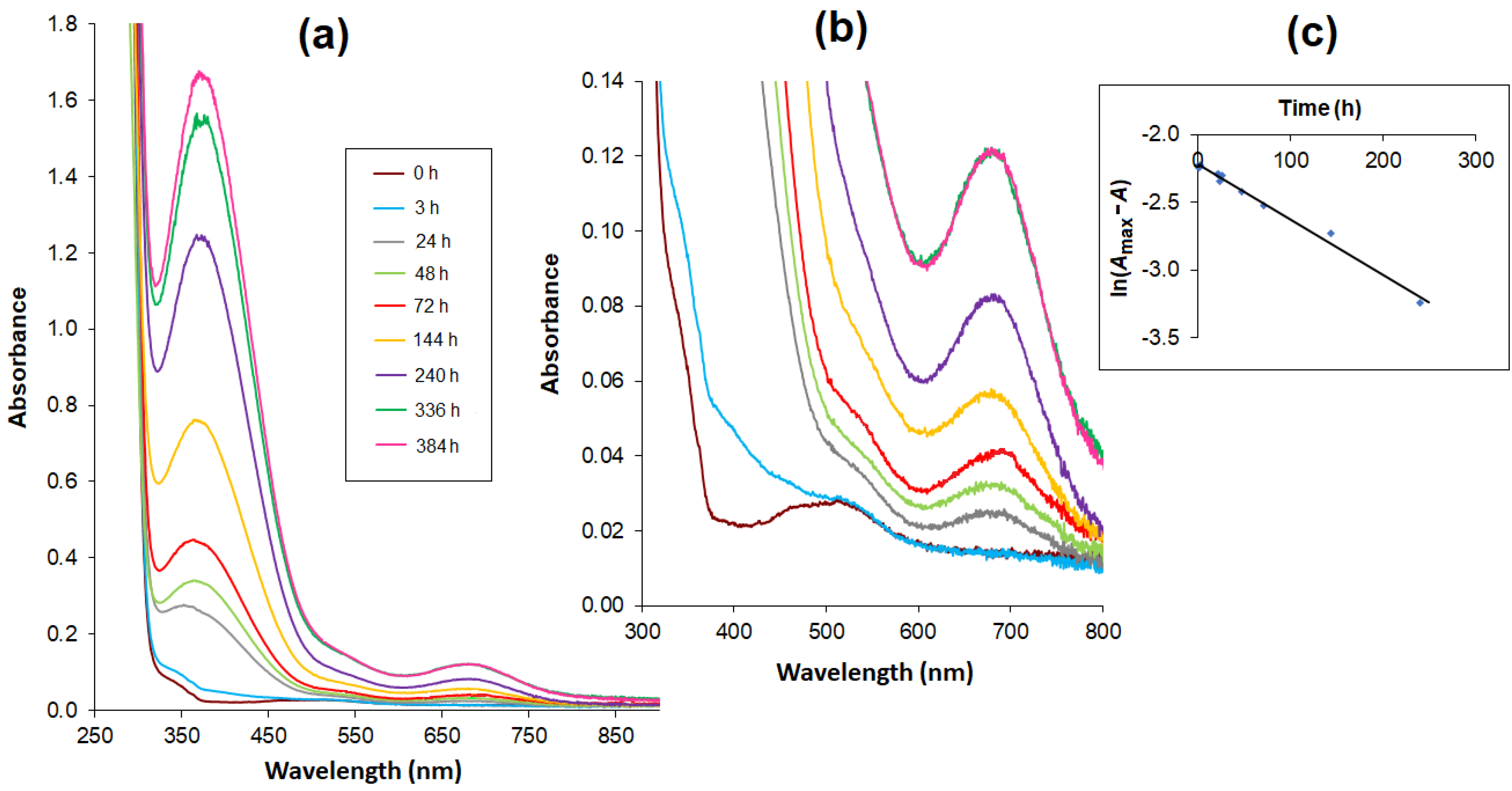
2.3. UV/Vis Spectra
2.4. Kinetic Analysis
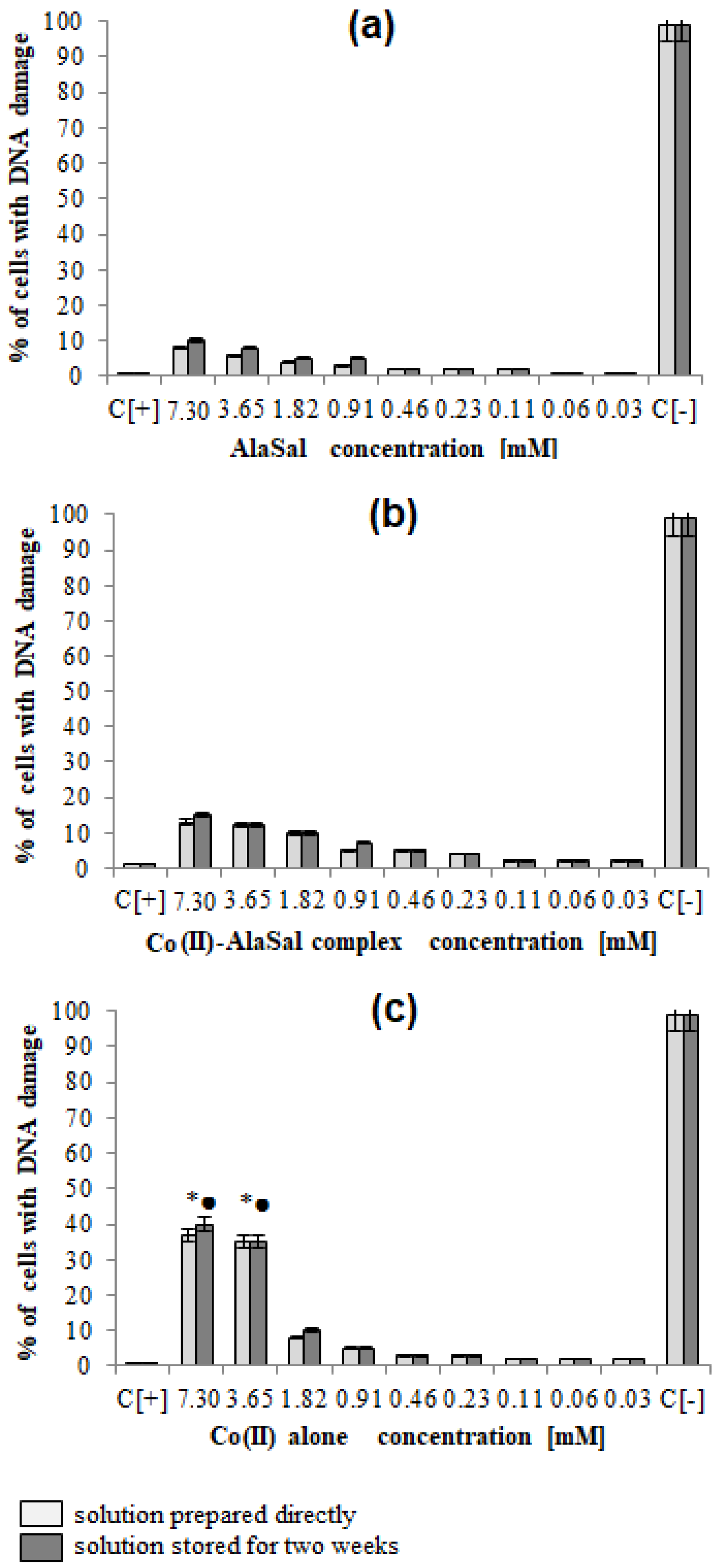
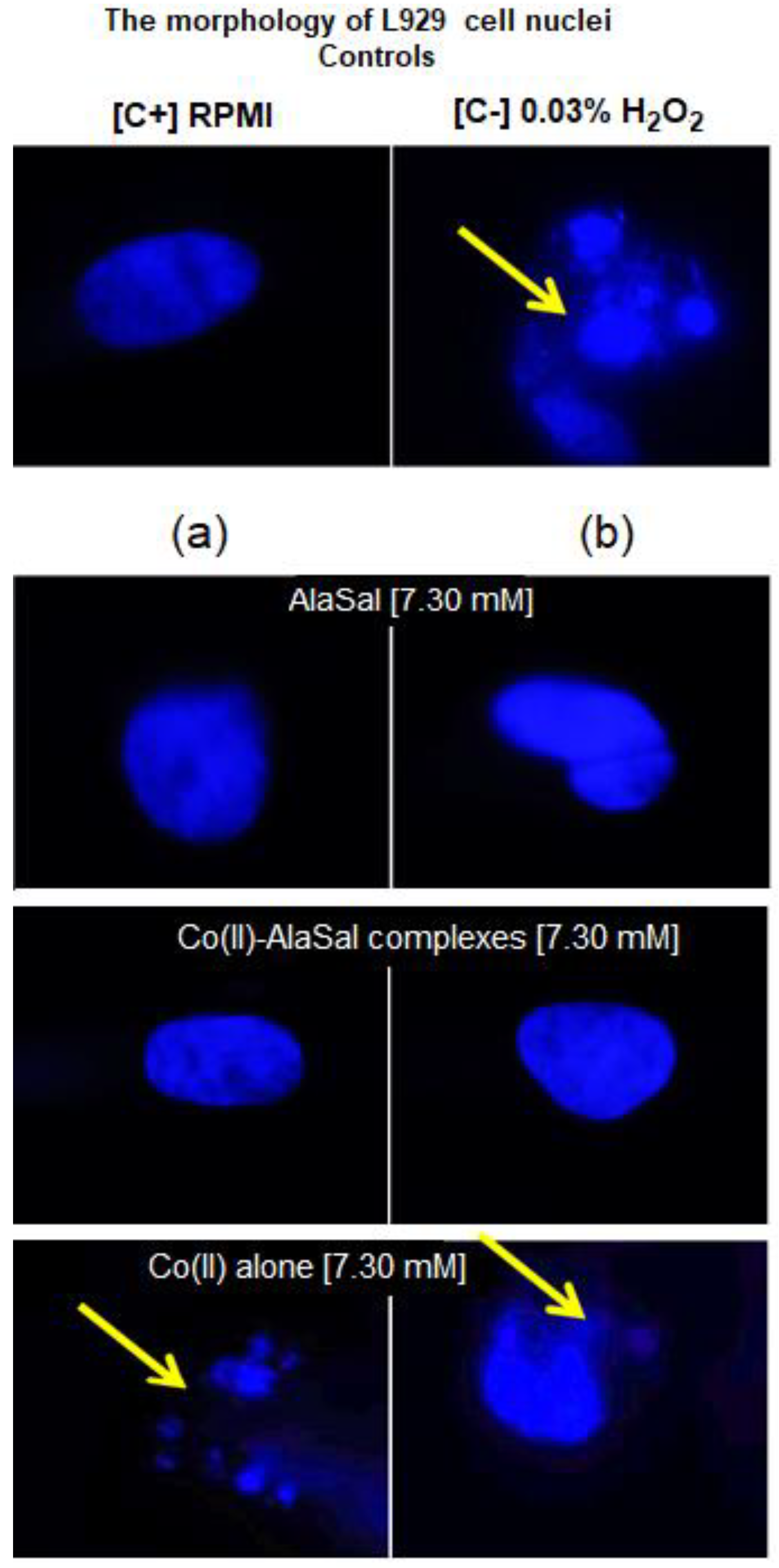
2.5. Biological Activity
3. Materials and Methods
3.1. Materials
3.2. pH-Metric Titrations
3.3. Electrospray-Ionization Mass Spectrometry (ESI-MS) Measurements
3.4. Spectrophotometric Measurements
3.5. Kinetics
3.6. Biological Assays
3.6.1. Investigation of Antimicrobial Properties
3.6.2. Cell Cultures
Measurements of Cellular Metabolic. Activity and Global Growth Inhibition
DAPI Staining of Cell Nuclei
3.6.3. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, X.; Hamon, J.-R. Recent developments in penta-, hexa- and heptadentate Schiff base ligands and their metal complexes. Coord. Chem. Rev. 2019, 389, 94–118. [Google Scholar] [CrossRef]
- Sutradhar, M.; Pombeiro, A.J.L. Coordination chemistry of non-oxido, oxido and dioxidovanadium(IV/V) complexes with azine fragment ligands. Coord. Chem. Rev. 2014, 265, 89–124. [Google Scholar] [CrossRef]
- Ganguly, R.; Sreenivasulu, B.; Vittal, J.J. Amino acid-containing reduced Schiff bases as the building blocks for metallasupramolecular structures. Coord. Chem. Rev. 2008, 252, 1027–1050. [Google Scholar] [CrossRef]
- Lintnerová, L.; Valentová, J.; Herich, P.; Kožišek, J.; Devinsky, F. Synthesis and antiradical activity of novel copper (II) complexes of long chain reduced Schiff base ligands. Monatsh. Chem. 2018, 149, 901–911. [Google Scholar] [CrossRef]
- Zdrada, J.; Odrzywołek, W.; Deda, A.; Wilczyński, S. A split-face comparative study to evaluate the efficacy of 50% pyruvic acid against a mixture of glycolic and salicylic acids in the treatment of acne vulgaris. J. Cosmet. Dermatol. 2020, 1–7. [Google Scholar] [CrossRef]
- Kong, J.; Zhang, Y.; Ju, J.; Xie, Y.; Guo, Y.; Cheng, Y.; Qian, H.; Quek, S.Y.; Yao, W. Antifungal effects of thymol and salicylic acid on cell membrane and mitochondria of Rhizopus stolonifera and their application in postharvest preservation of tomatoes. Food Chem. 2019, 285, 380–388. [Google Scholar] [CrossRef]
- Bougherra, H.; Berradj, O.; Adkhis, A.; Amrouche, T. Synthesis, characterization, electrochemical and biological activities of mixed ligand copper (II) complexes with dimethylglyoxime and amino acids. J. Mol. Struct. 2018, 1173, 280–290. [Google Scholar] [CrossRef]
- Abendrot, M.; Chęcińska, L.; Kusz, J.; Lisowska, K.; Zawadzka, K.; Felczak, A.; Kalinowska-Lis, U. Zinc(II) complexes with amino acids for potential use in dermatology: Synthesis, crystal structures, and antibacterial activity. Molecules 2020, 25, 951. [Google Scholar] [CrossRef]
- Pająk, M.; Woźniczka, M.; Vogt, A.; Kufelnicki, A. Reversible uptake of molecular oxygen by heteroligand Co (II)-L-α-amino acid-imidazole systems: Equilibrium models at full mass balance. Chem. Cent. J. 2017, 11, 90. [Google Scholar] [CrossRef]
- Li, A.; Liu, Y.-H.; Yuan, L.-Z.; Ma, Z.-Y.; Zhao, C.-L.; Xie, C.-Z.; Bao, W.-G.; Xu, J.-Y. Association of structural modifications with bioactivity in three new copper (II) complexes of Schiff base ligands derived from 5-chlorosalicylaldehyde and amino acids. J. Inorg. Biochem. 2015, 146, 52–60. [Google Scholar] [CrossRef]
- Shukla, S.N.; Gaur, P.; Raidas, M.L.; Chaurasia, B. Tailored synthesis of unsymmetrical tetradentate ONNO schiff base complexes of Fe (IIl), Co (II) and Ni (II): Spectroscopic characterization, DFT optimization, oxygen-binding study, antibacterial and anticorrosion activity. J. Mol. Struct. 2020, 1202, 127362. [Google Scholar] [CrossRef]
- Chohan, Z.H.; Arif, M.; Akhtar, M.A.; Supuran, C.T. Metal-based antibacterial and antifungal agents: Synthesis, characterization, and in vitro biological evaluation of Co (II), Cu (II), Ni (II), and Zn (II) complexes with amino acid-derived compounds. Bioinorg. Chem. Appl. 2006. [Google Scholar] [CrossRef]
- Dar, O.A.; Lone, S.A.; Malik, M.A.; Wani, M.Y.; Talukdar, M.I.A.; Al-Bogami, A.S.; Hashmi, A.A.; Ahmad, A. Heteroleptic transition metal complexes of Schiff-base-derived ligands exert their antifungal activity by disrupting membrane integrity. Appl. Organometal. Chem. 2019, 33, e5128. [Google Scholar] [CrossRef]
- Sutradhar, M.; Alegria, E.C.B.A.; Ferretti, F.; Raposo, L.R.; Guedes da Silva, M.F.C.; Baptista, P.V.; Fernandes, A.R.; Pombeiro, A.J.L. Antiproliferative activity of heterometallic sodium and potassium-dioxidovanadium(V) polymers. J. Inorg. Biochem. 2019, 200, 110811. [Google Scholar] [CrossRef] [PubMed]
- Sutradhar, M.; Fernandes, A.R.; Silva, J.; Mahmudov, K.T.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Water soluble heterometallic potassium-dioxidovanadium(V) complexes as potential antiproliferative agents. J. Inorg. Biochem. 2016, 155, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Sutradhar, M.; Rajeshwari; Barman, T.R.; Fernandes, A.R.; Paradinha, F.; Roma-Rodrigues, C.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Mixed ligand aroylhydrazone and N-donor heterocyclic Lewis base Cu (II) complexes as potential antiproliferative agents. J. Inorg. Biochem. 2017, 175, 267–275. [Google Scholar] [CrossRef]
- Fang, Z.; Yan, J.; Yu, W.; Zhang, N.; Zhang, S. Three Schiff base complexes based on diethylenetriamine: Synthesis, structure, DNA binding and cleavage, and in vitro cytotoxicity. Transit. Met. Chem. 2019, 44, 463–474. [Google Scholar] [CrossRef]
- Mohsen, A.; Saby, C.; Collery, P.; Sabry, G.M.; Hassan, R.E.; Badawi, A.; Jeannesson, P.; Desmaële, D.; Morjani, H. Biological study of the effect of water soluble [N-(2-hydroxybenzyl)-L-aspartato] gallium complexes on breast carcinoma and fibrosarcoma cells. J. Biol. Inorg. Chem. 2016, 21, 837–849. [Google Scholar] [CrossRef] [PubMed]
- Mondal, M.; Ghosh, S.; Maity, S.; Giri, S.; Ghosh, A. In situ transformation of a tridentate to a tetradentate unsymmetric Schiff base ligand via deaminative coupling in Ni (II) complexes: Crystal structures, magnetic properties and catecholase activity study. Inorg. Chem. Front. 2020, 7, 247–259. [Google Scholar] [CrossRef]
- Sheoran, M.; Bhar, K.; Jain, S.; Rana, M.; Khan, T.A.; Sharma, A.K. Phenoxo-bridged dicopper complexes: Syntheses, characterizations, crystal structures and catecholase activity. Polyhedron 2019, 161, 169–178. [Google Scholar] [CrossRef]
- Bhunia, A.; Bertolasi, V.; Manna, S.C. Tridentate Schiff base and 4,4’-bipyridine coordinated di/polynuclear Cu (II) complexes: Synthesis, crystal structure, DNA/protein binding and catecholase activity. Appl. Organometal. Chem. 2020, e5424. [Google Scholar] [CrossRef]
- Kumar, M.; Parsekar, S.U.; Duraipandy, N.; Kiran, M.S.; Koley, A.P. Synthesis, DNA binding and in vitro cytotoxicity studies of a mononuclear copper (II) complex containing N2S (thiolate) Cu core and 1,10-phenanthroline as a coligand. Inorg. Chim. Acta 2019, 484, 219–226. [Google Scholar] [CrossRef]
- Tarinc, D.; Muslu, H.; Cesme, M.; Gölcü, A.; Tumer, M.; Ozkan, S.A. Synthesis, structural characterization and electrochemical evaluation of Schiff base transition metal complexes with ceftazidime. Curr. Anal. Chem. 2013, 9, 319–332. [Google Scholar] [CrossRef]
- Harriott, M.M.; Noverr, M.C. Candida albicans and Staphylococcus aureus form polymicrobial biofilms: Effects on antimicrobial resistance. Antimicrob. Agents Chemother. 2009, 53, 3914–3922. [Google Scholar] [CrossRef] [PubMed]
- Harriott, M.M.; Noverr, M.C. Ability of Candida albicans mutants to induce Staphylococcus aureus vancomycin resistance during polymicrobial biofilm formation. Antimicrob. Agents Chemother. 2010, 54, 3746–3755. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Thammavongsa, V.; Schneewind, O.; Missiakas, D. Recurrent infections and immune evasion strategies of Staphylococcus aureus. Curr. Opin. Microbiol. 2012, 15, 92–99. [Google Scholar] [CrossRef]
- El-Ayaan, U.; Abdel-Aziz, A.A.M. Synthesis, antimicrobial activity and molecular modeling of cobalt and nickel complexes containing the bulky ligand: Bis[N-(2,6-diisopropylphenyl)imino] acenaphthene. Eur. J. Med. Chem. 2005, 40, 1214–1221. [Google Scholar] [CrossRef]
- Correia, I.; Adão, P.; Roy, S.; Wahba, M.; Matos, C.; Maurya, M.R.; Marques, F.; Pavan, F.R.; Leite, C.Q.F.; Avecilla, F.; et al. Hydroxyquinoline derived vanadium (IV and V) and copper (II) complexes as potential anti-tuberculosis and anti-tumor agents. J. Inorg. Biochem. 2014, 141, 83–93. [Google Scholar] [CrossRef]
- Woźniczka, M.; Świątek, M.; Pająk, M.; Gądek-Sobczyńska, J.; Chmiela, M.; Gonciarz, W.; Lisiecki, P.; Pasternak, B.; Kufelnicki, A. Complexes in aqueous cobalt(II)–2-picolinehydroxamic acid system: Formation equilibria, DNA-binding ability, antimicrobial and cytotoxic properties. J. Inorg. Biochem. 2018, 187, 62–72. [Google Scholar] [CrossRef]
- Chang, E.L.; Simmers, C.; Knight, D.A. Cobalt complexes as antiviral and antibacterial agents. Pharmaceuticals 2010, 3, 1711–1728. [Google Scholar] [CrossRef]
- Ritsma, J.H. Stereoselectivity and proton binding in some transition-metal complexes of N-(o-hydroxybenzyl)alanine. Recl. J. R. Neth. Chem. Soc. 1975, 94, 174–178. [Google Scholar] [CrossRef]
- Ozawa, T.; Jitsukawa, K.; Masuda, H.; Einaga, H. Thermodynamic stabilities and formation mechanisms of iron (III) complexes with Nα-carboxymethyl-Nα-salicyl-L-phenylalanine and Nα-salicyl-L-phenylalanine: Enhanced thermodynamic stability by additional methylcarboxylate group. Polyhedron 1994, 13, 2343–2351. [Google Scholar] [CrossRef]
- Taylor, D.P.; Choppin, G.R. Rare earth complexation with 1,3,5-trideoxy-1,3,5-tris(2-hydroxybenzyl)amino-cis-inositol. Inorg. Chim. Acta 2007, 360, 3712–3718. [Google Scholar] [CrossRef]
- Al-Farhan, B.S.; Gouda, G.A.; Farghaly, O.A.; EL Khalafawy, A.K. Potentiometeric study of new Schiff base complexes bearing morpholine in ethanol-water medium with some metal ions. Int. J. Electrochem. Sci. 2019, 14, 3350–3362. [Google Scholar] [CrossRef]
- Patel, P.; Bhattacharya, P.K. Study of binary and ternary complexes of some binucleating ligands. Indian J. Chem. 1995, 34A, 196–200. [Google Scholar]
- Yang, C.-T.; Moubaraki, B.; Murray, K.S.; Vittal, J.J. Synthesis, characterization and properties of ternary copper (II) complexes containing reduced Schiff base N-(2-hydroxybenzyl)-α-amino acids and 1,10-phenanthroline. Dalton Trans. 2003, 5, 880–889. [Google Scholar] [CrossRef]
- Liu, H.-Y.; Guan, Q.-H.; Tian, J.; Du, P.; Chen, H. Synthesis, crystal structures, and antibacterial studies of silver(I) complexes with reduced Schiff base amino acid ligands. Transit. Met. Chem. 2016, 41, 615–622. [Google Scholar] [CrossRef]
- Woźniczka, M.; Vogt, A.; Kufelnicki, A. Equilibria in cobalt(II)-amino acid-imidazole system under oxygen-free conditions. Effect of side groups on mixed-ligand systems with selected L-α-amino acids. Chem. Cent. J. 2016, 10, 14. [Google Scholar] [CrossRef]
- Koh, L.L.; Ranford, J.O.; Robinson, W.T.; Svensson, J.O.; Tan, A.L.C.; Wu, D. Model for the reduced Schiff base intermediate between amino acids and pyridoxal: Copper(II) complexes of N-(2-hydroxybenzyl)amino acids with nonpolar side chains and the crystal structures of [Cu(N-(2-hydroxybenzyl)-D,L-alanine)(phen)]·H2O and [Cu(N-(2-hydroxybenzyl)-D,L-alanine)(imidazole)]. Inorg. Chem. 1996, 35, 6466–6472. [Google Scholar] [CrossRef]
- Lou, B.-Y.; Yuan, D.-Q.; Wang, R.-H.; Xu, Y.; Wu, B.-L.; Han, L.; Hong, M.-C. Two supramolecular architectures constructed from dinuclear zinc (II) unit. J. Mol. Struct. 2004, 698, 87–91. [Google Scholar] [CrossRef]
- Zhao, Z.-G.; Wu, X.-Y.; Zhai, Q.-G.; Chen, L.-J.; Zhang, Q.-Z.; Li, W.; Xie, Y.-M.; Lu, C.-Z. Syntheses, structures, and properties of mono- and tetranuclear nickel(II) complexes derived from a tridentate Schiff base ligand. Z. Anorg. Allg. Chem. 2008, 634, 288–294. [Google Scholar] [CrossRef]
- Fekri, R.; Salehi, M.; Asadi, A.; Kubicki, M. Synthesis, characterization, anticancer and antibacterial evaluation of Schiff base ligands derived from hydrazine and their transition metal complexes. Inorg. Chim. Acta 2019, 484, 245–254. [Google Scholar] [CrossRef]
- Patel, P.; Bhattacharya, P.K. Stereochemical factors affectin intramolecular interligand interaction in mixed ligand complexes. Indian J. Chem. 1993, 32A, 506–510. [Google Scholar]
- Lou, B.-Y.; Xu, Y.; Yuan, D.-Q.; Han, L.; Hong, M.-C. Bis[N-(4-hydroxybenzyl)-D,L-alaninato]-copper(II) tetrahydrate. Acta Crystallogr. Sect. E Struct. Rep. Online 2004, 60, m522–m523. [Google Scholar] [CrossRef]
- Faizi, M.S.H.; Dege, N.; Pogrebetsky, J.; Iskenderov, T.S. Structural characterization and DFT study of bis{(S)-2-[(2-hydroxybenzyl)amino]-3-(4-hydroxyphenyl)propanoato-κ2 N,O}(1,10-phenanthroline- κ2 N,N’)cadmium(II) tetrahydrate. Acta Crystallogr. Sect. E Crystallogr. Commun. 2018, E74, 1339–1343. [Google Scholar] [CrossRef] [PubMed]
- Kiss, T. Complexes of amino acids. In Biocoordination Chemistry: Coordination Equilibria in Biologically Active Systems; Burger, K., Ed.; Ellis Horwood Ltd.: Chichester, UK, 1990; pp. 56–134. [Google Scholar]
- Lever, A.B.P. Inorganic Electronic Spectroscopy, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 1984. [Google Scholar]
- Luigi, C.; Gullotti, M. Synthesis, stereochemistry, and oxygenation of cobalt (II)-pyridoxal model complexes. A new family of chiral dioxygen carriers. Inorg. Chem. 1986, 25, 1293–1303. [Google Scholar] [CrossRef]
- Yang, C.-T.; Vetrichelvan, M.; Yang, X.; Moubaraki, B.; Murray, K.S.; Vittal, J.J. Syntheses, structural properties and catecholase activity of copper(II) complexes with reduced Schiff base N-(2-hydroxybenzyl)-amino acids. J. Chem. Soc. Dalton Trans. 2004, 4, 113–121. [Google Scholar] [CrossRef]
- Wagner, M.R.; Walker, F.A. Spectroscopic study of 1:1 copper (II) complexes with Schiff base ligands derived from salicylaldehyde and L-histidine and its analogs. Inorg. Chem. 1983, 22, 3021–3028. [Google Scholar] [CrossRef]
- Nair, M.S.; Joseyphus, R.S. Synthesis and characterization of Co (II), Ni (II), Cu (II) and Zn (II) complexes of tridentate Schiff base derived from vanillin and DL-α-aminobutyric acid. Spectrochim. Acta Part. A 2008, 70, 749–753. [Google Scholar] [CrossRef]
- Belz, T.; Ihmaid, S.; Al-Rawi, J.; Petrovski, S. Synthesis characterization and antibacterial, antifungal activity of N-(benzyl carbamoyl or carbamothioyl)-2-hydroxy substituted benzamide and 2-benzyl amino-substituted benzoxazines. Int. J. Med. Chem. 2013, 436397. [Google Scholar] [CrossRef]
- Ohi, N.; Aoki, B.; Shinozaki, T.; Moro, K.; Noto, T.; Nehashi, T.; Okazaki, H.; Matsunaga, I. Semisynthetic β-lactam antibiotics I. Synthesis and antibacterial activity of new ureidopenicillin derivatives having catechol moieties. J. Antibiot. 1986, 39, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Stec, J.; Huang, Q.; Pieroni, M.; Kaiser, M.; Fomovska, A.; Mui, E.; Witola, W.H.; Bettis, S.; McLeod, R.; Brun, R.; et al. Synthesis, biological evaluation, and structure-activity relationships of N-benzoyl-2-hydroxybenzamides as agents active against P. falciparum (K1 strain), Trypanosomes, and Leishmanial. J. Med. Chem. 2012, 55, 3088–3100. [Google Scholar] [CrossRef] [PubMed]
- Patole, J.; Shingnapurkar, D.; Padhye, S.; Ratledge, C. Schiff base conjugates of p-aminosalicylic acid as antimycobacterial agents. Bioorg. Med. Chem. Lett. 2006, 16, 1514–1517. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Bi, C.; Fan, Y.; Liu, S.; Zhang, X.; Zhang, D.; Wang, Y.; Zhu, R. Synthesis, characterization and bioactivity of Schiff base copper (II) complexes derived from L-glutamine and L-asparagine. J. Coord. Chem. 2009, 62, 3029–3039. [Google Scholar] [CrossRef]
- Muslu, H.; Golcu, A.; Ozkan, S.A. Electrochemical study of ceftazidime-copper (II) complex: Synthesis, characterization, biological activity and analytical application to pharmaceutical dosage forms. Curr. Anal. Chem. 2010, 6, 299–309. [Google Scholar] [CrossRef]
- Liu, L.; Yao, W.D.; Rao, Y.F.; Lu, X.Y.; Gao, J.Q. pH-Responsive carriers for oral drug delivery: Challenges and opportunities of current platforms. Drug Deliv. 2017, 24, 569–581. [Google Scholar] [CrossRef]
- Gans, P.; O’Sullivan, B. GLEE, a new computer program for glass electrode calibration. Talanta 2000, 51, 33–37. [Google Scholar] [CrossRef]
- Irving, H.; Miles, M.G.; Pettit, L.D. A study of some problems in determining the stoicheiometric proton dissociation constants of complexes by potentiometric titrations using a glass electrode. Anal. Chim. Acta 1967, 38, 475–488. [Google Scholar] [CrossRef]
- Gans, P.; Sabatini, A.; Vacca, A. Investigation of equilibria in solution. Determination of equilibrium constants with the Hyperquad suite of programs. Talanta 1996, 43, 1739–1753. [Google Scholar] [CrossRef]
- Woźniczka, M.; Szajdzinska-Pietek, E.; Jezierska, J.; Pasternak, B.; Gądek-Sobczyńska, J.; Kufelnicki, A. The complexing properties of oxalodihydrazide, acethydrazide and formic hydrazide with Cu (II) in aqueous solution. Inorg. Chim. Acta 2017, 455, 659–665. [Google Scholar] [CrossRef]
- Pettit, L.D.; Powell, K.J. Stability Constants Database, IUPAC, Academic Software; Royal Society of Chemistry: London, UK, 1993–2000. [Google Scholar]
- Alderighi, L.; Gans, P.; Ienco, A.; Peters, D.; Sabatini, A.; Vacca, A. Hyperquad simulation and speciation (HySS): A utility program for the investigation of equilibria involving soluble and partially soluble species. Coordin. Chem. Rev. 1999, 184, 311–318. [Google Scholar] [CrossRef]
- Kostiainen, R.; Kauppila, T.J. Effect of eluent on the ionization process in liquid chromatography–mass spectrometry. J. Chromatogr. A 2009, 1216, 685–699. [Google Scholar] [CrossRef] [PubMed]
- Mnich, E.; Kowalewicz-Kulbat, M.; Sicińska, P.; Hinc, K.; Obuchowski, M.; Gajewski, A.; Moran, A.P.; Chmiela, M. Impact of Helicobacter pylori on the healing process of the gastrin barrier. World J. Gastroenterol. 2016, 22, 7536–7558. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
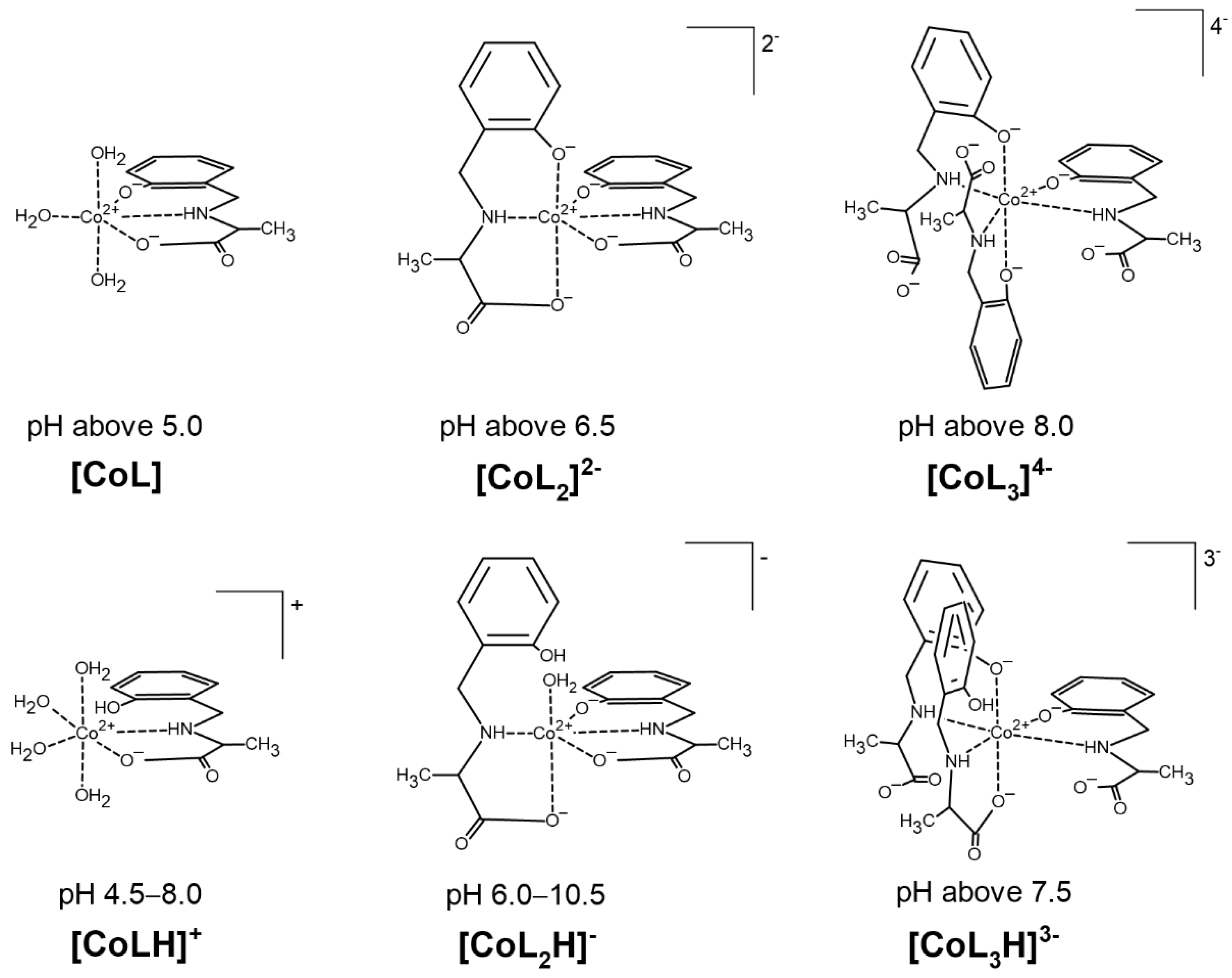
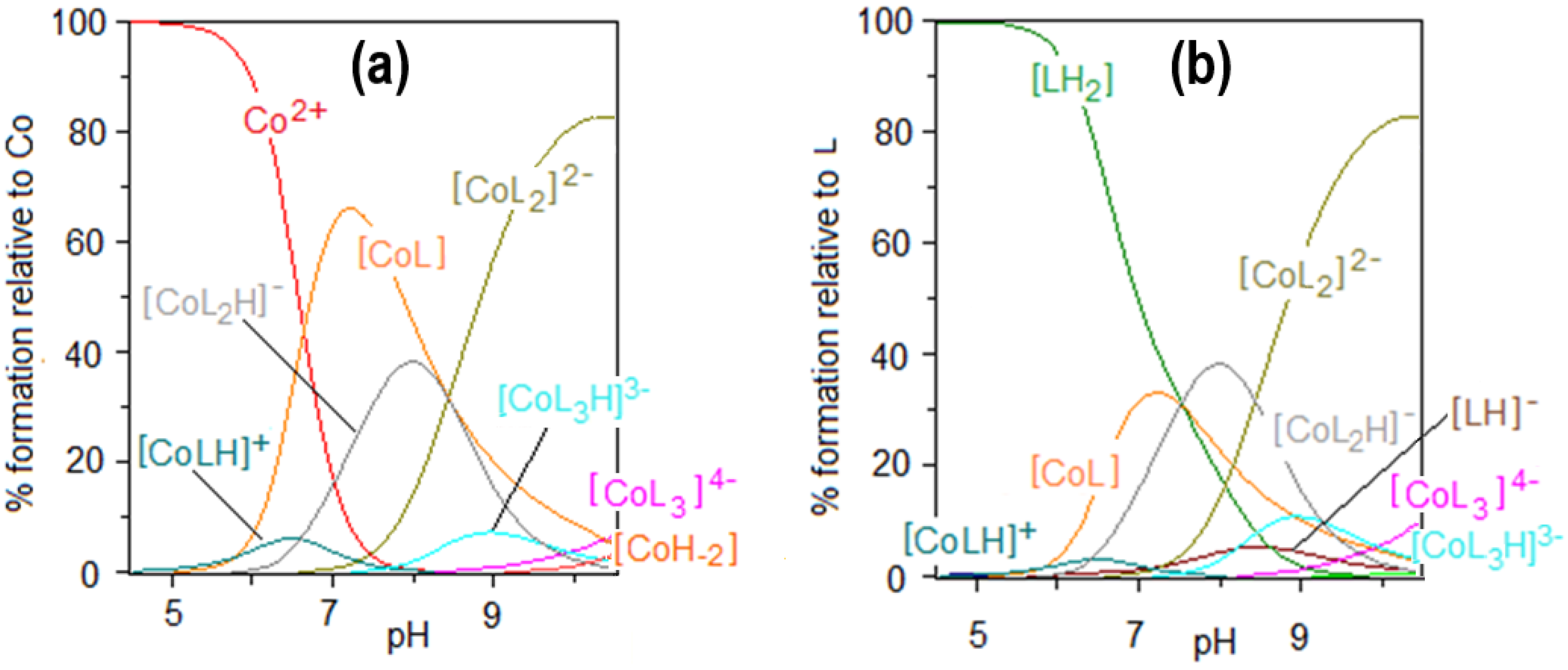

| Species | log10 βmlh | Stepwise Dissociation Constants | Related Constants | λmax (εmax) |
|---|---|---|---|---|
| [L]2− | 236 (8.2 × 103) | |||
| 292 (3.5 × 103) | ||||
| [LH]− | 10.73(1) (OH) | pKa3 10.73 | 237 (6.1 × 103) | |
| 291 (2.6 × 103) | ||||
| [LH2] | 19.36(1) (NH2+) | pKa2 8.63 2 | ~240sh (4.1 × 103) | |
| 274 (2.1 × 103) | ||||
| [LH3]+ | 21.66(2) (COOH) | pKa1 2.30 3 | ~298sh (1.6 × 103) | |
| σ; n1 | 5.82; 804 | |||
| [CoL] | 7.98(1) | 508 (21) | ||
| [CoL2]2− | 13.35(2) | 483 (31) | ||
| [CoL3]4− | 16.39(4) | |||
| [CoLH]+ | 13.64(10) | 2.92 4 | ||
| 21.78(2) | 11.06 5 | |||
| [CoL2H]− | 26.35(4) | 15.63 6 | ||
| [CoL3H]3− | ||||
| σ; n1 | 5.85; 455 |
| Microorganism | MIC/MBC (mM) | MIC = MBC (mM) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| AlaSal | Co(II) Alone | Co(II)–AlaSal Complexes | Gentamicin | Amphotericin B | Amoxicillin | ||||
| MIC | MBC | MIC | MBC | MIC | MBC | ||||
| Gram-negative bacteria | |||||||||
| Pseudomonas aeruginosa ATCC 27853 | 1.82 | 1.82 | 0.91 | 1.82 | 1.82 | 1.82 | <0.008 | - | - |
| Escherichia coli ATCC 25922 | 7.30 | >7.30 | 1.82 | 1.82 | 7.30 | >7.30 | <0.004 | - | - |
| Helicobacter pylori CCUC 17874 | 7.30 | >7.30 | 1.82 | >7.30 | 3.65 | 3.65 | - | - | <0.001 |
| Helicobacter pylori ATCC 700392 | 7.30 | >7.30 | 1.82 | >7.30 | 3.65 | 3.65 | - | - | <0.001 |
| Gram-positive bacteria | |||||||||
| Enterococcus faecalis ATCC 29212 | 7.30 | >7.30 | 1.82 | 1.82 | 1.82 | 3.65 | <0.26 | - | - |
| Staphylococcus aureus ATCC 29213 | 7.30 | >7.30 | 0.91 | 0.91 | 1.82 | 3.65 | <0.002 | - | - |
| Staphylococcus aureus ATCC 6538 | 7.30 | >7.30 | 0.91 | 0.91 | 1.82 | 3.65 | <0.002 | - | - |
| Staphylococcus epidermidis ATCC 12228 | 7.30 | >7.30 | 0.91 | 0.91 | 1.82 | 3.65 | <0.002 | - | - |
| Fungi | |||||||||
| Candida albicans ATTC 10231 | 3.65 | >7.30 | 0.23 | 3.65 | 1.82 | 3.65 | - | <0.001 | - |
| Candida glabrata ATCC 2001 | 3.65 | >7.30 | 0.23 | 3.65 | 1.82 | 3.65 | - | <0.001 | - |
| Candida parapsilosis ATCC 22019 | 3.65 | >7.30 | 0.23 | 7.30 | 1.82 | 3.65 | - | <0.001 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woźniczka, M.; Sutradhar, M.; Pombeiro, A.J.L.; Świątek, M.; Pająk, M.; Gądek-Sobczyńska, J.; Chmiela, M.; Gonciarz, W.; Pasternak, B.; Kufelnicki, A. Equilibria in Aqueous Cobalt(II)—Reduced Schiff Base N-(2-hydroxybenzyl)alanine System: Chemical Characterization, Kinetic Analysis, Antimicrobial and Cytotoxic Properties. Molecules 2020, 25, 3462. https://doi.org/10.3390/molecules25153462
Woźniczka M, Sutradhar M, Pombeiro AJL, Świątek M, Pająk M, Gądek-Sobczyńska J, Chmiela M, Gonciarz W, Pasternak B, Kufelnicki A. Equilibria in Aqueous Cobalt(II)—Reduced Schiff Base N-(2-hydroxybenzyl)alanine System: Chemical Characterization, Kinetic Analysis, Antimicrobial and Cytotoxic Properties. Molecules. 2020; 25(15):3462. https://doi.org/10.3390/molecules25153462
Chicago/Turabian StyleWoźniczka, Magdalena, Manas Sutradhar, Armando J. L. Pombeiro, Mirosława Świątek, Marek Pająk, Joanna Gądek-Sobczyńska, Magdalena Chmiela, Weronika Gonciarz, Beata Pasternak, and Aleksander Kufelnicki. 2020. "Equilibria in Aqueous Cobalt(II)—Reduced Schiff Base N-(2-hydroxybenzyl)alanine System: Chemical Characterization, Kinetic Analysis, Antimicrobial and Cytotoxic Properties" Molecules 25, no. 15: 3462. https://doi.org/10.3390/molecules25153462
APA StyleWoźniczka, M., Sutradhar, M., Pombeiro, A. J. L., Świątek, M., Pająk, M., Gądek-Sobczyńska, J., Chmiela, M., Gonciarz, W., Pasternak, B., & Kufelnicki, A. (2020). Equilibria in Aqueous Cobalt(II)—Reduced Schiff Base N-(2-hydroxybenzyl)alanine System: Chemical Characterization, Kinetic Analysis, Antimicrobial and Cytotoxic Properties. Molecules, 25(15), 3462. https://doi.org/10.3390/molecules25153462