Vitamin D Signaling in Inflammation and Cancer: Molecular Mechanisms and Therapeutic Implications
Abstract
:1. Introduction
2. Epidemiology
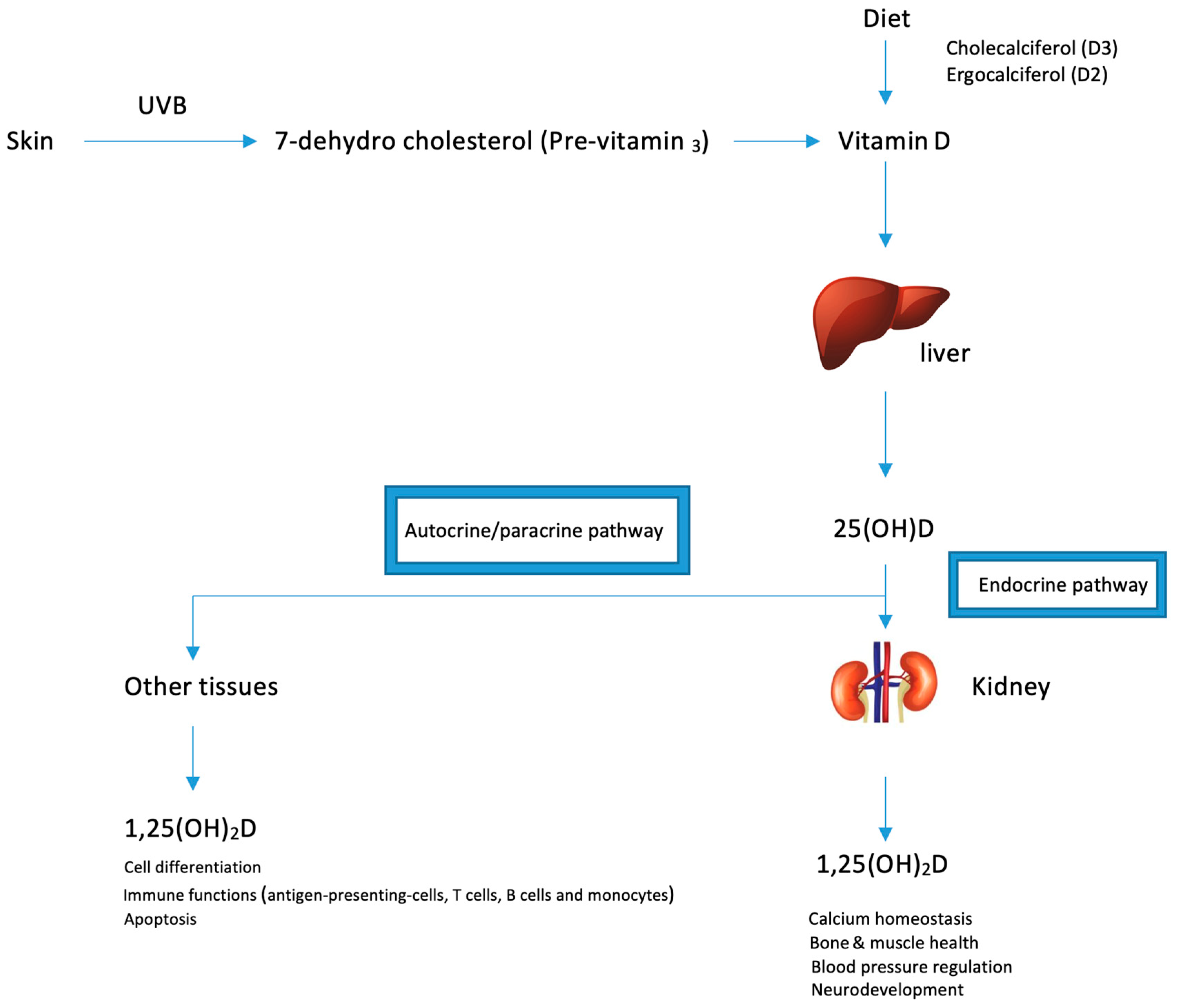
3. Metabolism
4. Vitamin D Status
5. Vitamin D Signaling
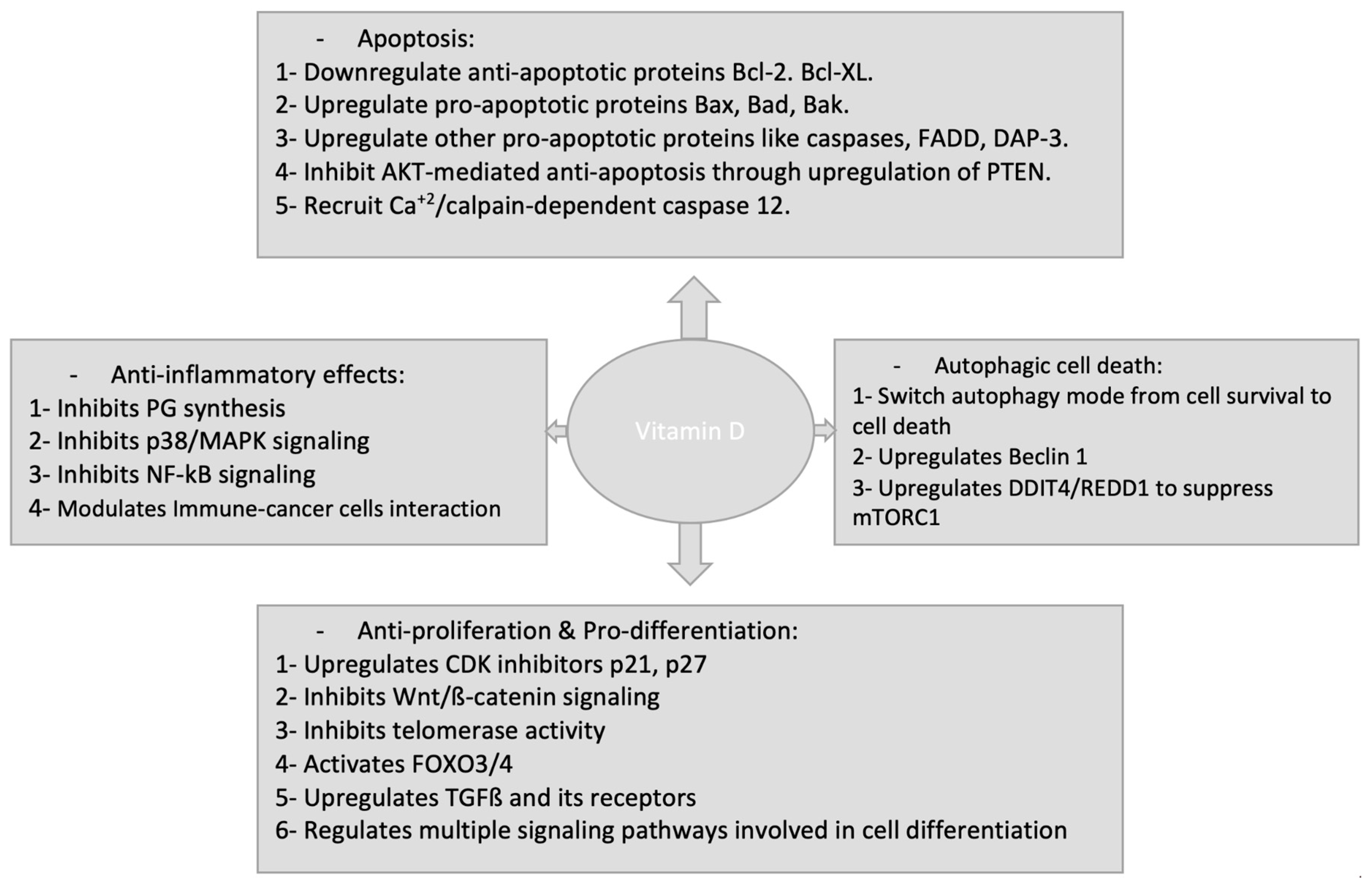
6. Inflammation
6.1. Vitamin D and MAP Kinase Phosphatase 5
6.2. Vitamin D and NF-κB
6.3. Vitamin D and Prostaglandins
7. Vitamin D signaling Within Tumour Cells
7.1. Regulation of Proliferation
7.2. Induction of Differentiation
7.3. Induction of Apoptosis
7.4. Inhibition of Angiogenesis and Metastasis
7.5. Induction of Autophagy
8. Therapeutic Potential and Future Perspectives
8.1. Analogues of 1,25(OH)2D3 (Calcitriol)
8.2. Calcitriol-Mediated Anticancer Properties in Animal Models
Calcitriol Combination Regimens in Animal Studies
8.3. Vitamin D Clinical Studies
8.3.1. Trials of Calcitriol as a Single Agent
8.3.2. Other Calcitriol Analogues
8.3.3. Calcitriol Combinational Studies
- (1)
- The asymmetric design of ASCENT II (it was designed as a randomized study comparing the FDA approved docetaxel regimen (75 mg/m2/3 weeks) + prednisone (10 mg/day) + placebo versus docetaxel (36 mg/m2/week (this regimen was shown to be inferior to the weekly every 3 weeks docetaxel regimen) + prednisone (10 mg/day) + calcitriol (DN-101, 0.5 μg/kg 1 day before docetaxel) violates one of the essential tenets of a randomized trial design; that is, to eliminate all variables between standard and experimental arms, except one.
- (2)
- Lack of data defining either the optimal or the maximal dose of oral calcitriol. Conveniently, 0.5 μg/kg weekly oral dose was used in ASCENT II. However, an intravenously dose of approximately 77 μg (>1 μg/kg in a 70-kg patient) is required to achieve the AUC (area under the curve) associated with the antitumor activities seen in mice.
8.3.4. Vitamin D and Omega-3 Trial (VITAL)
- (1)
- Daily vitamin D and omega-3;
- (2)
- Daily vitamin D and omega-3 placebo;
- (3)
- Daily vitamin D placebo and omega-3
- (4)
- Daily vitamin D placebo and omega-3 placebo.
- (1)
- Only one dose was tested in the intervention group (2000 IU/day);
- (2)
- Approximately 40% of participants with no records of serum 25(OH)D levels at baseline;
- (3)
- Lack of assessment of vitamin D status during the follow-up in most participants;
- (4)
- Lack of information about sun exposure, indoor and outdoor physical activity and body-covering habits of participants;
- (5)
- No increase in vitamin D doses according to BMI (2000 IU/day: low dose for overweight/ obese individuals)
- (6)
- Low number of participants (12.7%) with serum 25(OH) levels <20 ng/mL at baseline
- (7)
- The median period of follow-up was 5.3 years
9. Summary
- (1)
- There is a need to define MTD, phase II dose of calcitriol as a single agent or in combination with chemotherapeutics and the definition of biologically optimal dose of these agents.
- (2)
- Designation and conduction of randomized phase III trials with the analogue be the only variable.
- (3)
- Defining Vitamin D response-dependent biomarkers, this could facilitate selection of an active dose to therapeutically study vitamin D and help in targeting patients with a higher likelihood of response to vitamin D compounds.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Severo, M.; Lopes, C.; Lucas, R.; Barros, H. Development of a tool for the assessment of calcium and vitamin D intakes in clinical settings. Osteoporos. Int. 2009, 20, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Antonucci, R.; Locci, C.; Clemente, M.G.; Chicconi, E.; Antonucci, L. Vitamin D deficiency in childhood: Old lessons and current challenges. J. Pediatr. Endocrinol. Metab. 2018, 31, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Uday, S.; Högler, W. Prevention of rickets and osteomalacia in the UK: Political action overdue. Arch. Dis. Child. 2018, 103, 901–906. [Google Scholar] [CrossRef] [PubMed]
- D’Amelio, P.; Cristofaro, M.A.; De Vivo, E.; Ravazzoli, M.; Grosso, E.; Di Bella, S.; Aime, M.; Cotto, N.; Silvagno, F.; Isaia, G.C.; et al. Platelet vitamin D receptor is reduced in osteoporotic patients. Panminerva Med. 2012, 54, 225–231. [Google Scholar]
- Wei, R.; Christakos, S. Mechanisms Underlying the regulation of innate and adaptive immunity by vitamin D. Nutrients 2015, 7, 8251–8260. [Google Scholar] [CrossRef] [Green Version]
- Balkwill, F.; Mantovani, A. Inflammation and cancer: Back to Virchow? Lancet 2001, 357, 539–545. [Google Scholar] [CrossRef]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Fujiki, H. Gist of Dr. Katsusaburo Yamagiwa’s papers entitled “Experimental study on the pathogenesis of epithelial tumors” (I to VI reports). Cancer Sci. 2014, 105, 143–149. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Hu, W.; Lu, L.; Zhao, Y.; Zhou, Y.; Xiao, Z.; Zhang, L.; Zhang, H.; Li, X.; Li, W.; et al. Repurposing vitamin D for treatment of human malignancies via targeting tumor microenvironment. Acta Pharm. Sin. B 2019, 9, 203–219. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C. Do sun-light and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 2006, 35, 217–220. [Google Scholar] [CrossRef] [Green Version]
- Colston, K.; Colston, M.J.; Feldman, D. 1,25-dihydroxy vitamin D3 and malignant melanoma: The presence of receptors and inhibition of cell growth in culture. Endocrinology 1981, 108, 1083–1086. [Google Scholar] [CrossRef] [PubMed]
- Abe, E.; Miyaura, C.; Sakagami, H.; Takeda, M.; Konno, K.; Yamazaki, T.; Yoshiki, S.; Suda, T. Differentiation of mouse myeloid leukemia cells induced by 1 α, 25-dihydroxyvitamin D3. Proc. Natl. Acad. Sci. USA 1981, 78, 4990–4994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheung, F.S.; Lovicu, F.J.; Reichardt, J.K. Current progress in using vitamin D and its analogs for cancer prevention and treatment. Expert Rev. Anticancer Ther. 2012, 12, 811–837. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, A.V.; Feldman, D. Mechanisms of the anticancer and anti-inflammatory actions of vitamin D. Annu. Rev. Pharm. Toxicol. 2011, 51, 311–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leyssens, C.; Verlinden, L.; Verstuyf, A. Antineoplastic effects of 1,25(OH)2D3 and its analogs in breast, prostate and colorectal cancer. Endocr. Relat. Cancer 2013, 20, R31–R47. [Google Scholar] [CrossRef] [Green Version]
- Mehta, R.G.; Peng, X.; Alimirah, F.; Murillo, G.; Mehta, R. Vitamin D and breast cancer: Emerging concepts. Cancer Lett. 2013, 334, 95–100. [Google Scholar] [CrossRef]
- Pereira, F.; Larriba, M.J.; Munoz, A. Vitamin D and colon cancer. Endocr. Relat. Cancer 2012, 19, R51–R71. [Google Scholar] [CrossRef] [Green Version]
- Rosen, C.J.; Adams, J.S.; Bikle, D.D.; Black, D.M.; Demay, M.B.; Manson, J.E.; Murad, M.H.; Kovacs, C.S. The non-skeletal effects of vitamin D: An Endocrine Society scientific statement. Endocr. Rev. 2012, 33, 456–492. [Google Scholar] [CrossRef] [Green Version]
- Feldman, D.; Krishnan, A.V.; Swami, S. Osteoporosis; Marcus, R., Feldman, D., Dempster, D., Luckey, M., Cauley, J., Eds.; Elsevier Academic Press: Amsterdam, The Netherlands, 2013; pp. 283–329. [Google Scholar]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef]
- Haussler, M.R.; Whitfield, G.K.; Kaneko, I.; Haussler, C.A.; Hsieh, D.; Hsieh, J.C.; Jurutka, P.W. Molecular mechanisms of vitamin D action. Calcif. Tissue Int. 2013, 92, 77–98. [Google Scholar] [CrossRef]
- Fleet, J.C.; De Smet, M.; Johnson, R.; Li, Y. Vitamin D and cancer: A review of molecular mechanisms. Biochem. J. 2012, 441, 61–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Zhu, J.; DeLuca, H.F. Where is the vitamin D receptor? Arch. Biochem. Biophys. 2012, 523, 123–133. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.M.; Sandler, D.P.; Taylor, J.A.; Weinberg, C.R. Serum Vitamin D and Risk of Breast Cancer within Five Years. Environ. Health Perspect. 2017, 125, 077004. [Google Scholar] [CrossRef] [PubMed]
- Vaughan-Shaw, P.G.; O’sullivan, F.; Farrington, S.; Theodoratou, E.; Campbell, H.; Dunlop, M.G.; Zgaga, L. The impact of vitamin D pathway genetic variation and circulating 25-hydroxyvitamin D on cancer outcome: Systematic review and meta-analysis. Br. J. Cancer 2017, 116, 1092–1110. [Google Scholar] [CrossRef] [PubMed]
- Lappe, J.; Garland, C.; Gorham, E. Vitamin D Supplementation and Cancer Risk. JAMA 2017, 318, 299–300. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.M.; Shin, E.A. Exploring vitamin D metabolism and function in cancer. Exp. Mol. Med. 2018, 50, 20. [Google Scholar] [CrossRef] [Green Version]
- Gil, A.; Plaza-Diaz, J.; Mesa, M.D. Vitamin D: Classic and Novel Actions. Ann. Nutr. Metab. 2018, 72, 87–95. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, L.; Xu, H.J.; Li, Y.; Hu, C.M.; Yang, J.Y.; Sun, M.Y. The Anti-Inflammatory Effects of Vitamin D in Tumorigenesis. Int. J. Mol. Sci. 2018, 19, 2736. [Google Scholar] [CrossRef] [Green Version]
- So, J.Y.; Suh, N. Targeting cancer stem cells in solid tumors by vitamin D. J. Steroid Biochem. Mol. Biol. 2015, 148, 79–85. [Google Scholar] [CrossRef] [Green Version]
- Tretli, S.; Schwartz, G.G.; Torjesen, P.A.; Robsahm, T.E. Serum levels of 25-hydroxyvitamin D and survival in Norwegian patients with cancer of breast, colon, lung, and lymphoma: A population-based study. Cancer Causes Control 2012, 23, 363–370. [Google Scholar] [CrossRef] [Green Version]
- Tagliabue, E.; Raimondi, S.; Gandini, S. Vitamin D, Cancer Risk, and Mortality. Adv. Food Nutr. Res. 2015, 75, 1–52. [Google Scholar] [PubMed]
- Ordonez Mena, J.M.; Brenner, H. Vitamin D and cancer: An overview on epidemiological studies. Adv. Exp. Med. Biol. 2014, 810, 17–32. [Google Scholar] [PubMed]
- Pilz, S.; Tomaschitz, A.; Obermayer-Pietsch, B.; Dobnig, H.; Pieber, T.R. Epidemiology of vitamin D insufficiency and cancer mortality. Anticancer Res. 2009, 29, 3699–3704. [Google Scholar]
- Cho, M.; Peddi, P.F.; Ding, K.; Chen, L.; Thomas, D.; Wang, J.; Lockhart, A.C.; Tan, B.; Wang-Gillam, A. Vitamin D deficiency and prognostics among patients with pancreatic adenocarcinoma. J. Transl. Med. 2013, 11, 206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budhathoki, S.; Hidaka, A.; Yamaji, T.; Sawada, N.; Tanaka-Mizuno, S.; Kuchiba, A.; Charvat, H.; Goto, A.; Kojima, S.; Sudo, N.; et al. Plasma 25-hydroxyvitamin D concentration and subsequent risk of total and site specific cancers in Japanese population: Large case-cohort study within Japan Public Health Center-based Prospective Study cohort. BMJ 2018, 360, k671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Chen, P.; Li, J.; Chu, R.; Xie, D.; Wang, H. Review: The impacts of circulating 25-hydroxyvitamin D levels on cancer patient outcomes: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 2327–2336. [Google Scholar] [CrossRef] [Green Version]
- Goyal, H.; Perisetti, A.; Rahman, M.R.; Levin, A.; Lippi, G. Vitamin D and Gastrointestinal Cancers: A Narrative Review. Dig. Dis. Sci. 2018. [Google Scholar] [CrossRef]
- Gorham, E.D.; Garland, C.F.; Garland, F.C.; Grant, W.B.; Mohr, S.B.; Lipkin, M.; Newmark, H.L.; Giovannucci, E.; Wei, M.; Holick, M.F. Vitamin D and prevention of colorectal cancer. J. Steroid. Biochem. Mol. Biol. 2005, 97, 179–194. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, P.; Wang, F.; Yang, J.; Liu, Z.; Qin, H. Association between vitamin D and risk of colorectal cancer: A systematic review of prospective studies. J. Clin. Oncol. 2011, 29, 3775–3782. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Sun, X.; Lu, S.; Liu, S. Vitamin intake and pancreatic cancer risk reduction A meta-analysis of observational studies. Medicine (Baltimore) 2018, 97, e0114. [Google Scholar] [CrossRef]
- Wu, D.B.; Wang, M.L.; Chen, E.Q.; Tang, H. New insights into the role of vitamin D in hepatocellular carcinoma. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Moukayed, M.; Grant, W.B. The roles of UVB and vitamin D in reducing risk of cancer incidence and mortality: A review of the epidemiology, clinical trials, and mechanisms. Rev. Endocr. Metab. Disord. 2017, 18, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. The epidemiology of vitamin D and cancer incidence and mortality: A review (United States). Cancer Causes Control 2005, 16, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.Y.; Yao, Q.; Zhuo, Z.; Ma, Z.; Chen, G. Circulating vitamin D level and mortality in prostate cancer patients: A dose-response meta-analysis. Endocr. Connect. 2018, 7, R294–R303. [Google Scholar] [CrossRef] [Green Version]
- Gao, J.; Wei, W.; Wang, G.; Zhou, H.; Fu, Y.; Liu, N. Circulating vitamin D concentration and risk of prostate cancer: A dose–response meta-analysis of prospective studies. Ther. Clin. Risk Manag. 2018, 14, 95–104. [Google Scholar] [CrossRef] [Green Version]
- Narvaez, C.J.; Matthews, D.; LaPorta, E.; Simmons, K.M.; Beaudin, S.; Welsh, J. The impact of vitamin D in breast cancer: Genomics, pathways, metabolism. Front. Physiol. 2014, 5, 213. [Google Scholar] [CrossRef]
- Kim, Y.; Je, Y. Vitamin D intake, blood 25(OH)D levels, and breast cancer risk or mortality: A meta-analysis. Br. J. Cancer 2014, 110, 2772–2784. [Google Scholar] [CrossRef] [Green Version]
- Hu, K.; Callen, D.F.; Li, J.; Zheng, H. Circulating Vitamin D and Overall Survival in Breast Cancer Patients: A Dose-Response Meta-Analysis of Cohort Studies. Integr. Cancer Ther. 2017, 17, 217–225. [Google Scholar] [CrossRef] [Green Version]
- Welsh, J. Vitamin D and breast cancer: Past and present. J. Steroid Biochem. Mol. Biol. 2017, 177, 15–20. [Google Scholar] [CrossRef]
- Silva, M.C.; Furlanetto, T.W. Intestinal absorption of vitamin D: A systematic review. Nutr. Rev. 2018, 76, 60–76. [Google Scholar] [CrossRef]
- Mulligan, G.B.; Licata, A. Taking vitamin D with the largest meal improves absorption and results in higher serum levels of 25-hydroxyvitamin D. J. Bone Min. Res. 2010, 25, 928–930. [Google Scholar] [CrossRef] [PubMed]
- Brannon, P.M.; Fleet, J.C. Vitamin D. In Advances in Nutrition; Issue 4; Oxford Academics: Oxford, UK, 2011; Volume 2, pp. 365–367. [Google Scholar]
- Wacker, M.; Holick, M.F. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol 2013, 5, 51–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, N.; Zhang, H. CYP24A1 depletion facilitates the antitumor effect of vitamin D3 on thyroid cancer cells. Exp. Ther. Med. 2018, 16, 2821–2830. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Wang, C.; Hao, M.; Sun, R.; Wang, Y.; Liu, T.; Cong, X.; Liu, Y. CYP24A1 is a potential biomarker for the progression and prognosis of human colorectal cancer. Hum. Pathol. 2016, 50, 101–108. [Google Scholar] [CrossRef]
- Meyer, M.B.; Goetsch, P.D.; Pike, J.W. VDR/RXR and TCF4/beta-catenin cistromes in colonic cells of colorectal tumour origin: Impact on c-FOS and c-MYC gene expression. Mol. Endocrinol. 2012, 26, 37–51. [Google Scholar] [CrossRef]
- Shiratsuchi, H.; Wang, Z.; Chen, G.; Ray, P.; Lin, J.; Zhang, Z.; Zhao, L.; Beer, D.; Ray, D.; Ramnath, N. Oncogenic potential of CYP24A1in lung adenocarcinoma. J. Thorac. Oncol. 2017, 12, 269–280. [Google Scholar] [CrossRef] [Green Version]
- Osanai, M.; Lee, G.H. CYP24A1-induced vitamin D insufficiency promotes breast cancer growth. Oncol. Rep. 2016, 36, 2755–2762. [Google Scholar] [CrossRef] [Green Version]
- Luo, W.; Yu, W.D.; Ma, Y.; Chernov, M.; Trump, D.L.; Johnson, C.S. Inhibition of protein kinase CK2 reduces Cyp24a1 expression and enhances 1,25-dihydroxy vitamin D3 antitumor activity in human prostate cancer cells. Cancer Res. 2013, 73, 2289–2297. [Google Scholar] [CrossRef] [Green Version]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Endocrine Society Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [Green Version]
- Cesareo, R.; Attanasio, R.; Caputo, M.; Castello, R.; Chiodini, I.; Falchetti, A.; Guglielmi, R.; Papini, E.; Santonati, A.; Scillitani, A.; et al. Italian Association of Clinical Endocrinologists (AME) and Italian chapter of the American Association of Clinical Endocrinologists (AACE) position statement: Clinical management of vitamin D deficiency in adults. Nutrients 2018, 10, 546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valcour, A.; Blocki, F.; Hawkins, D.M.; Rao, S.D. Effects of age and serum 25-OH-vitamin D on serum parathyroid hormone levels. J. Clin. Endocrinol. Metab. 2012, 97, 3989–3995. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, E.; Chou, R.; Zakher, B.; Daeges, M.; Pappas, M. Screening for Vitamin D Deficiency: Systematic Review for the U.S. Preventive Services Task Force Recommendation; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2014.
- LeBlanc, E.S.; Zakher, B.; Daeges, M.; Pappas, M.; Chou, R. Screening for vitamin D deficiency: A systematic review for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2015, 162, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, L.D.; Robertson, M.C.; Gillespie, W.J.; Sherrington, C.; Gates, S.; Clemson, L.M.; Lamb, S.E. Interventions for preventing falls in older people living in the community. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Bhattoa, H.P.; Konstantynowicz, J.; Laszcz, N.; Wojcik, M.; Pludowski, P. Vitamin D: Musculoskeletal health. Rev. Endocr. Metab. Disord. 2017, 18, 363–371. [Google Scholar] [CrossRef]
- Sanders, K.M.; Stuart, A.L.; Williamson, E.J.; Simpson, J.A.; Kotowicz, M.A.; Young, D.; Nicholson, G.C. Annual high-dose oral vitamin D and falls and fractures in older women: A randomized controlled trial. JAMA 2010, 303, 1815–1822. [Google Scholar] [CrossRef] [Green Version]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Orav, E.J.; Staehelin, H.B.; Meyer, O.W.; Theiler, R.; Dick, W.; Willett, W.C.; Egli, A. Monthly high-dose vitamin D Treatment for the prevention of functional decline: A randomized clinical trial. JAMA Intern. Med. 2016, 176, 175–183. [Google Scholar] [CrossRef]
- Amrein, K.; Quraishi, S.A.; Litonjua, A.A.; Gibbons, F.K.; Pieber, T.R.; Camargo, C.A.; Giovannucci, E.; Christopher, K.B. Evidence for a U-shaped relationship between prehospital vitamin D status and mortality: A cohort study. J. Clin. Endocrinol. Metab. 2014, 99, 1461–1469. [Google Scholar] [CrossRef] [Green Version]
- Fleet, J.C. Rapid, membrane-initiated actions of 1,25 dihydroxyvitamin D: What are they and what do they mean? J. Nutr. 2004, 134, 3215–3218. [Google Scholar] [CrossRef]
- Doroudi, M.; Schwartz, Z.; Boyan, B.D. Membrane-mediated actions of 1,25-dihydroxy vitamin D3: A review of the roles of phospholipase A2 activating protein and Ca(2+)/calmodulin-dependent protein kinase II. J. Steroid Biochem. Mol. Biol. 2015, 147, 81–84. [Google Scholar] [CrossRef] [Green Version]
- Dwivedi, P.P.; Gao, X.H.; Tan, J.C.; Evdokiou, A.; Ferrante, A.; Morris, H.A.; May, B.K.; Hii, C.S. A role for the phosphatidylinositol 3-kinase—Protein kinase C zeta—Sp1 pathway in the 1,25-dihydroxyvitamin D3 induction of the 25-hydroxyvitamin D3 24-hydroxylase gene in human kidney cells. Cell. Signal. 2010, 22, 543–552. [Google Scholar] [CrossRef]
- Norman, A.W. Minireview: Vitamin D receptor: New assignments for an already busy receptor. Endocrinology 2006, 147, 5542–5548. [Google Scholar] [CrossRef] [Green Version]
- Friedrich, M.; Axt-Fliedner, R.; Villena-Heinsen, C.; Tilgen, W.; Schmidt, W.; Reichrath, J. Analysis of vitamin D-receptor(VDR)and retinoid X-receptor alpha in breas tcancer. Histochem. J. 2002, 34, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Izkhakov, E.; Somjen, D.; Sharon, O.; Knoll, E.; Aizic, A.; Fliss, D.M.; Limor, R.; Stern, N. Vitamin D receptor expression isl inked to potential markers of human thyroid papillary carcinoma. J. Steroid Biochem. Mol. Biol. 2016, 159, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Zhalehjoo, N.; Shakiba, Y.; Panjehpour, M. Gene expression profiles of CYP24A1 and CYP27B1 in malignant and normal breast tissues. Mol. Med. Rep. 2017, 15, 467–473. [Google Scholar] [CrossRef] [Green Version]
- Khadzkou, K.; Buchwald, P.; Westin, G.; Dralle, H.; Akerstrom, G.; Hellman, P. 25-hydroxy vitamin D3 1 alpha-hydroxylase and vitamin D receptor expression in papillary thyroid carcinoma. J. Histochem. Cytochem. 2006, 54, 355–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hendrickson, W.K.; Flavin, R.; Kasperzyk, J.L.; Fiorentino, M.; Fang, F.; Lis, R.; Fiore, C.; Penney, K.L.; Ma, J.; Kantoff, P.W.; et al. Vitamin D receptor protein expression in tumor tissue and prostate cancer progression. J. Clin. Oncol. 2011, 29, 2378–2385. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.H.; Chen, G.; King, A.N.; Jeon, C.K.; Christensen, P.J.; Zhao, L.; Simpson, R.U.; Thomas, D.G.; Giordano, T.J.; Brenner, D.E.; et al. Characterization of vitamin D receptor (VDR)in lung adenocarcinoma. Lung Cancer 2012, 77, 265–271. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.; Dong, M.; Sheng, W.; Liu, Q.; Yu, D.; Dong, Q.; Li, Q.; Wang, J. Expression of vitamin D receptor as a potential prognostic factor and therapeutic target in pancreatic cancer. Histopathology 2015, 67, 386–397. [Google Scholar] [CrossRef]
- Momen-Heravi, F.; Masugi, Y.; Qian, Z.R.; Nishihara, R.; Liu, L.; Smith-Warner, S.A.; Keum, N.; Zhang, L.; Tchrakian, N.; Nowak, J.A.; et al. Tumor expression of calcium sensing receptor and colorectal cancer survival: Results from the nurses ‘health study and health professionals follow-up study. Int. J. Cancer 2017, 141, 2471–2479. [Google Scholar] [CrossRef] [Green Version]
- Ferrer-Mayorga, G.; Gómez-López, G.; Barbáchano, A.; Fernández-Barral, A.; Peña, C.; Pisano, D.G.; Cantero, R.; Rojo, F.; Muñoz, A.; Larriba, M.J.; et al. Vitamin D receptor expression and associated gene signature in tumour stromal fibroblasts predict clinical outcome in colorectal cancer. Gut 2017, 66, 1449–1462. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, L.; Zhang, L.; Hu, W.; Shen, J.; Xiao, Z.; Wu, X.; Chan, F.L.; Cho, C.H. 1,25-Dihydroxyvitamin D3 suppresses gastric cancer cell growth through VDR- and mutant p53-mediated induction of p21. Life Sci. 2017, 179, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Ordóñez-Morán, P.; Larriba, M.J.; Pálmer, H.G.; Valero, R.A.; Barbáchano, A.; Duñach, M.; de Herreros, A.G.; Villalobos, C.; Berciano, M.T.; Lafarga, M.; et al. RhoA-ROCK and p38 MAPK-MSK1 mediate vitamin D effects on gene expression, phenotype, and Wnt pathway in colon cancer cells. J. Cell Biol. 2008, 183, 697–710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Yu, W.D.; Kong, R.X.; Trump, D.L.; Johnson, C.S. Role of non-genomic activation of phosphatidylinositol3-kinase/Akt and mitogen-activated protein kinase/extracellular signal-regulated kinase kinase/extracellular signal-regulated kinase1/2 pathways in 1,25D3-mediated apoptosis in squamous cell carcinoma cells. Cancer Res. 2006, 66, 8131–8138. [Google Scholar]
- Zheng, Y.; Trivedi, T.; Lin, R.C.; Fong-Yee, C.; Nolte, R.; Manibo, J.; Chen, Y.; Hossain, M.; Horas, K.; Dunstan, C.; et al. Loss of the vitamin D receptor in human breast and prostate cancers strongly induces cell apoptosis through downregulation of Wnt/β-catenin signalling. Bone Res. 2017, 5, 17023. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Fouad, Y.A.; Aanei, C. Revisiting the hallmarks of cancer. Am. J. Cancer Res. 2017, 7, 1016–1036. [Google Scholar]
- Atsumi, T.; Singh, R.; Sabharwal, L.; Bando, H.; Meng, J.; Arima, Y.; Yamada, M.; Harada, M.; Jiang, J.J.; Kamimura, D.; et al. Inflammation amplifier, a new paradigm in cancer biology. Cancer Res. 2014, 74, 8–14. [Google Scholar] [CrossRef] [Green Version]
- Gagliani, N.; Hu, B.; Huber, S.; Elinav, E.; Flavell, R.A. The fire within: Microbes inflame tumors. Cell 2014, 157, 776–783. [Google Scholar] [CrossRef] [Green Version]
- Murata, M. Inflammation and cancer. Environ. Health Prev. Med. 2018, 23, 50. [Google Scholar] [CrossRef] [Green Version]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014, 15, e493–e503. [Google Scholar] [CrossRef]
- Han, Y.; Zhang, Y.; Jia, T.; Sun, Y. Molecular mechanism underlying the tumor-promoting functions of carcinoma-associated fibroblasts. Tumour Biol. 2015, 36, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Piazuelo, E.; Lanas, A. NSAIDS and gastrointestinal cancer. Prostaglandins Other Lipid Mediat. 2015, 120, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Taylor, B.V.; Körner, H. Genomic Effects of the Vitamin D Receptor: Potentially the Link between Vitamin D, Immune Cells, and Multiple Sclerosis. Front. Immunol. 2018, 9, 477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barragan, M.; Good, M.; Kolls, J.K. Regulation of Dendritic Cell Function by Vitamin D. Nutrients 2015, 7, 8127–8151. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Choi, E.J. Pathological roles of MAPK signalling pathways in human diseases. Biochim. Biophys. Acta 2010, 1802, 396–405. [Google Scholar] [CrossRef] [Green Version]
- Nonn, L.; Peng, L.; Feldman, D.; Peehl, D.M. Inhibition of p38 by vitamin D reduces interleukin-6 production in normal prostate cells via mitogen-activated protein kinase phosphatase 5: Implications for prostate cancer prevention by vitamin D. Cancer Res. 2006, 66, 4516–4524. [Google Scholar] [CrossRef] [Green Version]
- Miraghazadeh, B.; Cook, M.C. Nuclear Factor-kappaB in Autoimmunity: Man and Mouse. Front. Immunol. 2018, 9, 613. [Google Scholar] [CrossRef] [Green Version]
- Xia, Y.; Shen, S.; Verma, I.M. NF-B, an active player in human cancers. Cancer Immunol. Res. 2014, 2, 823–830. [Google Scholar] [CrossRef] [Green Version]
- Yu, X.P.; Bellido, T.; Manolagas, S.C. Down-regulation of NF-kappa B protein levels in activated human lymphocytes by 1,25-dihydroxyvitamin D3. Proc. Natl. Acad. Sci. USA 1995, 92, 10990–10994. [Google Scholar] [CrossRef] [Green Version]
- Bao, B.Y.; Yao, J.; Lee, Y.F. 1Alpha, 25-dihydroxyvitamin D3 suppresses interleukin-8-mediated prostate cancer cell angiogenesis. Carcinogenesis 2006, 27, 1883–1893. [Google Scholar] [CrossRef] [PubMed]
- Schwab, M.; Reynders, V.; Loitsch, S.; Steinhilber, D.; Stein, J.; Schroder, O. Involvement of different nuclear hormone receptors in butyrate-mediated inhibition of inducible NF kappa B signalling. Mol. Immunol. 2007, 44, 3625–3632. [Google Scholar] [CrossRef] [PubMed]
- Tse, A.K.W.; Zhu, G.Y.; Wan, C.K.; Shen, X.L.; Yu, Z.L.; Fong, W.F. 1,25-Dihydroxyvitamin D3 inhibits transcriptional potential of nuclear factor B in breast cancer cells. Mol. Immunol. 2010, 47, 1728–1738. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Kong, J.; Duan, Y.; Szeto, F.L.; Liao, A.; Madara, J.L.; Li, Y.C. Increased NFkappaB activity in fibroblasts lacking the vitamin D receptor. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E315–E322. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Dubois, R.N. Eicosanoids and cancer. Nat. Rev. Cancer 2010, 10, 181–193. [Google Scholar] [CrossRef]
- Menter, D.G.; DuBois, R.N. Prostaglandins in Cancer Cell Adhesion, Migration, and Invasion. Int. J. Cell Biol. 2012, 2012, 723419. [Google Scholar] [CrossRef] [Green Version]
- Hawk, T.E.; Viner, L.J.; Dannenberg, A.; DuBois, N.R. COX-2 in cancer—A player that’s defining the rules. J. Natl. Cancer Inst. 2002, 94, 545–546. [Google Scholar] [CrossRef] [Green Version]
- Vanoirbeek, E.; Krishnan, A.; Eelen, G.; Verlinden, L.; Bouillon, R.; Feldman, D.; Verstuyf, A. The anti-cancer and anti-inflammatory actions of 1,25(OH)2D3. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 593–604. [Google Scholar] [CrossRef] [Green Version]
- Moreno, J.; Krishnan, A.V.; Swami, S.; Nonn, L.; Peehl, D.M.; Feldman, D. Regulation of prostaglandin metabolism by calcitriol attenuates growth stimulation in prostate cancer cells. Cancer Res. 2005, 65, 7917–7925. [Google Scholar] [CrossRef] [Green Version]
- Krishnan, A.V.; Swami, S.; Peng, L.; Wang, J.; Moreno, J.; Feldman, D. Tissue selective regulation of aromatase expression by calcitriol: Implications for breast cancer therapy. Endocrinology 2010, 151, 32–42. [Google Scholar] [CrossRef] [Green Version]
- Yuan, L.; Jiang, R.; Yang, Y.; Ding, S.; Deng, H. 1,25-Dihydroxyvitamin D3 inhibits growth of the breast cancer cell line MCF-7 and downregulates cytochrome P4501B1 through the COX-2/PGE2 pathway. Oncol. Rep. 2012, 28, 2131–2137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trump, D.L. Calcitriol and cancer therapy: A missed opportunity. Bone Rep. 2018, 9, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Saramaki, A.; Banwell, C.M.; Campbell, M.J.; Carlberg, C. Regulation of the human p21(WAF1/CIP1) gene promoter via multiple binding sites for p53 and the vitamin D3 receptor. Nucleic Acids Res. 2006, 34, 543–554. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Lee, M.H.; Cohen, M.; Bommakanti, M.; Freedman, L.P. Transcriptional activation of the CDK inhibitor p21 by vitamin D3 leads to the induced differentiation of the myelomonocytic cell line U937. Genes Dev. 1996, 10, 142–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Umar, M.; Sastry, K.S.; Chouchane, A.I. Role of Vitamin D Beyond the Skeletal Function: A Review of the Molecular and Clinical Studies. Int. J. Mol. Sci. 2018, 19, 1618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, W.; Zhang, X.; Zanello, L.P. 1alpha,25-Dihydroxyvitamin D (3) anti-proliferative actions involve vitamin D receptor-mediated activation of MAPK pathways and AP-1/p21(waf1) upregulation in human osteosarcoma. Cancer Lett. 2007, 254, 75–86. [Google Scholar] [CrossRef] [Green Version]
- Al Bitar, S.; Gali-Muhtasib, H. The Role of the Cyclin Dependent Kinase Inhibitor p21cip1/waf1 in Targeting Cancer: Molecular Mechanisms and Novel Therapeutics. Cancers 2019, 11, 1475. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.C.; Chen, J.Y.; Hung, W.C. Vitamin D3 receptor/Sp1 complex is required for the induction of p27Kip1 expression by vitamin D3. Oncogene 2004, 23, 4856–4861. [Google Scholar] [CrossRef] [Green Version]
- Yang, E.S.; Burnstein, K.L. Vitamin D inhibits G1 to S progression in LNCaP prostate cancer cells through p27Kip1 stabilization and Cdk2 mislocalization to the cytoplasm. J. Biol. Chem. 2003, 278, 46862–46868. [Google Scholar] [CrossRef] [Green Version]
- Li, P.; Li, C.; Zhao, X.; Zhang, X.; Nicosia, S.V.; Bai, W. p27Kip1. Stabilization and G1 Arrest by 1,25-Dihydroxyvitamin D3 in Ovarian Cancer Cells Mediated through Down-regulation of Cyclin E/Cyclin-dependent Kinase 2 and Skp1-Cullin-F-box Protein/Skp2 Ubiquitin Ligase. J. Biol. Chem. 2004, 279, 25260–25267. [Google Scholar] [CrossRef] [Green Version]
- Chiang, K.C.; Chen, T.C. The anti-cancer actions of vitamin D. Anticancer Agents Med. Chem. 2013, 13, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Freedman, L.P. Transcriptional targets of the vitamin D3 receptor-mediating cell cycle arrest and differentiation. J. Nutr. 1999, 129, 581S–586S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nowak, D.; Stewart, D.; Koeffler, H.P. Differentiation therapy of leukemia: 3 decades of development. Blood 2009, 113, 3655–3665. [Google Scholar] [CrossRef] [PubMed]
- Miyaura, C.; Abe, E.; Kuribayashi, T.; Tanaka, H.; Konno, K.; Nishii, Y.; Suda, T. 1a,25-Dihydroxy vitamin D3 induces differentiation of human myeloid leukemia cells. Biochem. Biophys. Res. Commun. 1981, 102, 937–943. [Google Scholar] [CrossRef]
- Hlubek, F.; Brabletz, T.; Budczies, J.; Pfeiffer, S.; Jung, A.; Kirchner, T. Heterogeneous expression of Wnt/b-catenin target genes within colorectal cancer. Int. J. Cancer 2007, 121, 1941–1948. [Google Scholar] [CrossRef]
- Larriba, M.J.; Gonzalez-Sancho, J.M.; Barbachano, A.; Niell, N.; Ferrer-Mayorga, G.; Munoz, A. Vitamin D is a multilevel repressor of Wnt/β-catenin signalling in cancer cells. Cancers 2013, 5, 1242–1260. [Google Scholar] [CrossRef] [Green Version]
- Shah, S.; Islam, M.N.; Dakshanamurthy, S.; Rizvi, I.; Rao, M.; Herrell, R.; Zinser, G.; Valrance, M.; Aranda, A.; Moras, D.; et al. The molecular basis of vitamin D receptor and β-catenin cross regulation. Mol. Cell 2006, 21, 799–809. [Google Scholar] [CrossRef]
- Xin, Y.; He, L.; Luan, Z.; Lv, H.; Yang, H.; Zhou, Y.; Zhao, X.; Zhou, W.; Yu, S.; Tan, B.; et al. E-cadherin mediates the preventive effect of vitamin D3 in colitis-associated carcinogenesis. Inflamm. Bowel Dis. 2017, 23, 1535–1543. [Google Scholar] [CrossRef] [Green Version]
- Aguilera, O.; Peña, C.; García, J.M.; Larriba, M.J.; Ordóñez-Morán, P.; Navarro, D.; Barbáchano, A.; López de Silanes, I.; Ballestar, E.; Fraga, M.F.; et al. The Wnt antagonist DICKKOPF-1 gene is induced by 1a,25-dihydroxy vitamin D3 associated to the differentiation of human colon cancer cells. Carcinogenesis 2007, 28, 1877–1884. [Google Scholar] [CrossRef]
- Pendás-Franco, N.; García, J.M.; Peña, C.; Valle, N.; Pálmer, H.G.; Heinäniemi, M.; Carlberg, C.; Jimenez, B.; Bonilla, F.; Munoz, A.; et al. DICKKOPF-4 is induced by TCF/b-catenin and upregulated in human colon cancer, promotes tumour cell invasion and angiogenesis and is repressed by1a,25-dihydroxy vitamin D3. Oncogene 2008, 27, 4467–4477. [Google Scholar] [CrossRef] [Green Version]
- Pendas-Franco, N.; Aguilera, O.; Pereira, F.; Gonzalez-Sancho, J.M.; Munoz, A. Vitamin D and Wnt/b-catenin pathway in colon cancer: Role and regulation of DICKKOPF genes. Anticancer Res. 2008, 28, 2613–2623. [Google Scholar] [PubMed]
- Pálmer, H.G.; González-Sancho, J.M.; Espada, J.; Berciano, M.T.; Puig, I.; Baulida, J.; Quintanilla, M.; Cano, A.; de Herreros, A.G.; Lafarga, M.; et al. Vitamin D3 promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of b-catenin signaling. J. Cell Biol. 2001, 154, 369–387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diaz, G.D.; Paraskeva, C.; Thomas, M.G.; Binderup, L.; Hague, A. Apoptosis is induced by the active metabolite of vitamin D3 and its analogue EB1089 in colorectal adenoma and carcinoma cells: Possible implications for prevention and therapy. Cancer Res. 2000, 60, 2304–2312. [Google Scholar] [PubMed]
- James, S.Y.; Mackay, A.G.; Colston, K.W. Effects of 1,25 dihydroxy vitamin D3 and its analogues on induction of apoptosis in breast cancer cells. J. Steroid Biochem. Mol. Biol. 1996, 58, 395–401. [Google Scholar] [CrossRef]
- Giammanco, M.; Di Majo, D.; La Guardia, M.; Aiello, S.; Crescimannno, M.; Flandina, C.; Tumminello, F.M.; Leto, G. Vitamin D in cancer chemoprevention. Pharm. Biol. 2015, 53, 1399–1434. [Google Scholar] [CrossRef] [Green Version]
- Narvaez, C.J.; Welsh, J. Role of mitochondria and caspases in vitamin D-mediated apoptosis of MCF-7 breast cancer cells. J. Biol. Chem. 2001, 276, 9101–9107. [Google Scholar] [CrossRef] [Green Version]
- Simboli-Campbell, M.; Narvaez, C.J.; Tenniswood, M.; Welsh, J. 1,25-Dihydroxy vitamin D3 induces morphological and biochemical markers of apoptosis in MCF-7 breast cancer cells. J. Steroid Biochem. Mol. Biol. 1996, 58, 367–376. [Google Scholar] [CrossRef]
- Guzey, M.; Kitada, S.; Reed, J.C. Apoptosis induction by 1a,25-dihydroxy vitamin D3 in prostate cancer. Mol. Cancer Ther. 2002, 1, 667–677. [Google Scholar]
- Blutt, S.E.; McDonnell, T.J.; Polek, T.C.; Weigel, N.L. Calcitriol-induced apoptosis in LNCaP cells is blocked by overexpression of Bcl-2. Endocrinology 2000, 141, 10–17. [Google Scholar] [CrossRef]
- Jiang, F.; Bao, J.; Li, P.; Nicosia, S.V.; Bai, W. Induction of ovarian cancer cell apoptosis by1,25-dihydroxy vitamin D3 through the down-regulation of telomerase. J. Biol. Chem. 2004, 279, 53213–53221. [Google Scholar] [CrossRef] [Green Version]
- Kasiappan, R.; Shen, Z.; Anfernee, K.W.; Jinwal, U.; Tang, J.; Lungchukiet, P.; Sun, Y.; Kruk, P.; Nicosia, S.V.; Zhang, X.; et al. 1,25-Dihydroxy vitamin D3 suppresses telomerase expression and human cancer growth through micro RNA-498. J. Biol. Chem. 2012, 287, 41297–41309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, W.D.; Ma, Y.; Flynn, G.; Muindi, J.R.; Kong, R.X.; Trump, D.L.; Johnson, C.S. Calcitriol enhances gemcitabine anti-tumor activity in vitro and in vivo by promoting apoptosis in a human pancreatic carcinoma model system. Cell Cycle 2010, 9, 3022–3029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hershberger, P.A.; Yu, W.D.; Modzelewski, R.A.; Rueger, R.M.; Johnson, C.S.; Trump, D.L. Calcitriol (1,25-dihydroxy cholecalciferol) enhances paclitaxel antitumor activity in vitro and in vivo and accelerates paclitaxel-induced apoptosis. Clin. Cancer Res. 2001, 7, 1043–1051. [Google Scholar] [PubMed]
- Hershberger, P.A.; McGuire, T.F.; Yu, W.D.; Zuhowski, E.G.; Schellens, J.H.; Egorin, M.J.; Trump, D.L.; Johnson, C.S. Cisplatin potentiates1,25-dihydroxy vitamin D3- induced apoptosis in association with increased mitogen-activated protein kinase kinase kinase1(MEKK-1) expression. Mol. Cancer Ther. 2002, 1, 821–829. [Google Scholar]
- Bernardi, R.J.; Trump, D.L.; Yu, W.D.; McGuire, T.F.; Hershberger, P.A.; Johnson, C.S. Combination of 1a,25-dihydroxy vitaminD3 with Dexamethasone enhances cell cycle arrest and apoptosis: Role of nuclear receptor cross-talk and Erk/Akt signaling. Clin. Cancer Res. 2001, 7, 4164–4173. [Google Scholar]
- Bao, A.; Li, Y.; Tong, Y.; Zheng, H.; Wu, W.; Wei, C. 1,25-Dihydroxy vitamin D3 and cisplatin synergistically induce apoptosis and cell cycle arrest in gastric cancer cells. Int. J. Mol. Med. 2014, 3, 1177–1184. [Google Scholar] [CrossRef] [Green Version]
- Pan, L.; Matloob, A.F.; Du, J.; Pan, H.; Dong, Z.; Zhao, J.; Feng, Y.; Zhong, Y.; Huang, B.; Lu, J.; et al. Vitamin D stimulates apoptosis in gastric cancer cells in synergy with trichostatin A/sodium butyrate-induced and 5-aza-20-deoxycytidine-induced PTEN upregulation. FEBS J. 2010, 277, 989–999. [Google Scholar] [CrossRef]
- Yang, J.; Yan, J.; Liu, B. Targeting VEGF/VEGFR to Modulate Antitumor Immunity. Front. Immunol. 2018, 9, 978. [Google Scholar] [CrossRef] [Green Version]
- Moukayed, M.; Grant, W.B. Molecular link between vitamin D and cancer prevention. Nutrients 2013, 5, 3993–4021. [Google Scholar] [CrossRef] [Green Version]
- Ben-Shoshan, M.; Amir, S.; Dang, D.T.; Dang, L.H.; Weisman, Y.; Mabjeesh, N.J. 1alpha,25-Dihydroxy vitamin D3 (calcitriol) inhibits hypoxia-inducible factor-1/vascular endothelial growth factor pathway in human cancer cells. Mol. Cancer Ther. 2007, 6, 1433–1439. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Jia, Z.; Gao, Y.; Xie, D.; Wei, D.; Cui, J.; Mishra, L.; Huang, S.; Zhang, Y.; Xie, K. Activation of vitamin D receptor signaling down regulates the expression of nuclear FOXM1 protein and suppresses pancreatic cancer cell stemness. Clin. Cancer Res. 2015, 21, 844–853. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, Y.F.; Gao, S.H.; Wang, P.; Zhang, H.M.; Liu, L.Z.; Ye, M.X.; Zhou, G.M.; Zhang, Z.L.; Li, B.Y. 1alpha,25(OH)(2)D(3) suppresses the migration of ovarian cancer SKOV-3 cells through the inhibition of epithelial-mesenchymal transition. Int. J. Mol. Sci. 2016, 17, 1285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Dijk, A.; Hedegaard, C.J.; Haagsman, H.P.; Heegaard, P.M.H. The potential for immunoglobulins and host defense peptides (HDPs) to reduce the use of antibiotics in animal production. Vet. Res. 2018, 49, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Lian, H.; Zhao, Y.; Kauss, M.A.; Spindel, S. Vitamin D3 induces autophagy of human myeloid leukemia cells. J. Biol. Chem. 2008, 283, 25596–25605. [Google Scholar] [CrossRef] [Green Version]
- Hoyer-Hansen, M.; Basthol, M.L.; Mathiasen, I.S.; Elling, F.; Jaattela, M. Vitamin D analog EB1089 triggers dramatic lysosomal changes and beclin 1-mediated autophagic cell death. Cell Death Differ. 2005, 12, 1297–1309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuk, J.M.; Shin, D.M.; Lee, H.M.; Yang, C.S.; Jin, H.S.; Kim, K.K.; Lee, Z.W.; Lee, S.H.; Kim, J.M.; Jo, E.K. Vitamin D3 induces autophagy in human monocytes/macrophages via cathelicidin. Cell Host Microbe 2009, 6, 231–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Høyer-Hansen, M.; Bastholm, L.; Szyniarowski, P.; Campanella, M.; Szabadkai, G.; Farkas, T.; Bianchi, K.; Fehrenbacher, N.; Elling, F.; Rizzuto, R.; et al. Control of macroautophagy by calcium, calmodulin-dependent kinase kinase-beta, and Bcl-2. Mol. Cell 2007, 25, 193–205. [Google Scholar] [CrossRef]
- Tavera-Mendoza, L.; Wang, T.T.; Lallemant, B.; Zhang, R.; Nagai, Y.; Bourdeau, V.; Ramirez-Calderon, M.; Desbarats, J.; Mader, S.; White, J.H.; et al. Convergence of vitamin D and retinoic acid signalling at a common hormone response element. EMBO Rep. 2006, 7, 180–185. [Google Scholar] [CrossRef] [Green Version]
- Tavera-Mendoza, L.E.; Westerling, T.; Libby, E.; Marusyk, A.; Cato, L.; Cassani, R.; Cameron, L.A.; Ficarro, S.B.; Marto, J.A.; Klawitter, J.; et al. Vitamin D receptor regulates autophagy in the normal mammary gland and in luminal breast cancer cells. Proc. Natl. Acad. Sci. USA 2017, 114, E2186–E2194. [Google Scholar] [CrossRef] [Green Version]
- Demasters, G.; Di, X.; Newsham, I.; Shiu, R.; Gewirtz, D.A. Potentiation of radiation sensitivity in breast tumor cells by the vitamin D3 analogue, EB 1089, through promotion of autophagy and interference with Proliferative recovery. Mol. Cancer Ther. 2006, 5, 2786–2797. [Google Scholar] [CrossRef] [Green Version]
- Sharma, K.; Goehe, R.W.; Di, X.; Hicks, M.A.; Torti, S.V.; Torti, F.M.; Harada, H.; Gewirtz, D.A. A novel cytostatic form of autophagy in sensitization of non- small cell lung cancer cells to radiation by vitamin D and the vitamin D analog, EB1089. Autophagy 2014, 10, 2346–2361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Trump, D.L.; Johnson, C.S. Vitamin D in combination cancer treatment. J. Cancer 2010, 1, 101–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tebben, P.J.; Singh, R.J.; Kumar, R. Vitamin D-Mediated Hypercalcemia: Mechanisms, Diagnosis, and Treatment. Endocrine Rev. 2016, 37, 521–547. [Google Scholar] [CrossRef] [PubMed]
- Trump, D.L.; Deeb, K.K.; Johnson, C.S. Vitamin D: Considerations in the continued development as an agent for cancer prevention and therapy. Cancer J. 2010, 16, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.C.; Kittaka, A. Novel vitamin D analogs for prostate cancer therapy. ISRN Urol. 2011, 2011, 301490. [Google Scholar] [CrossRef] [Green Version]
- Byers, S.W.; Rowlands, T.; Beildeck, M.; Bong, Y.S. Mechanism of action of vitamin D and the vitamin D receptor in colorectal cancer prevention and treatment. Rev. Endocr. Metab. Disord. 2012, 13, 31–38. [Google Scholar] [CrossRef] [Green Version]
- Skowronski, R.J.; Peehl, D.M.; Feldman, D. Actions of vitamin D3, analogs on human prostate cancer cell lines: Comparison with 1,25-dihydroxyvitamin D3. Endocrinology 1995, 136, 20–26. [Google Scholar] [CrossRef]
- Campbell, M.J.; Reddy, G.S.; Koeffler, H.P. Vitamin D3 analogs and their 24-oxo metabolites equally inhibit clonal proliferation of a variety of cancer cells but have differing molecular effects. J. Cell. Biochem. 1997, 66, 413–425. [Google Scholar] [CrossRef]
- Berkovich, L.; Sintov, A.C.; Ben-Shabat, S. Inhibition of cancer growth and induction of apoptosis by BGP-13 and BGP-15, new calcipotriene-derived vitamin D3 analogs, in-vitro and in-vivo studies. Investig. New Drugs 2013, 31, 247–255. [Google Scholar] [CrossRef] [Green Version]
- Okamoto, R.; Delansorne, R.; Wakimoto, N.; Doan, N.B.; Akagi, T.; Shen, M.; Ho, Q.H.; Said, J.W.; Koeffler, H.P. Inecalcitol, an analog of 1alpha,25(OH)(2) D(3), induces growth arrest of androgendependent prostate cancer cells. Int. J. Cancer 2012, 130, 2464–2473. [Google Scholar] [CrossRef]
- Verlinden, L.; Verstuyf, A.; Van Camp, M.; Marcelis, S.; Sabbe, K.; Zhao, X.Y.; De Clercq, P.; Vandewalle, M.; Bouillon, R. Two novel 14-Epi-analogues of 1,25-dihydroxyvitamin D3 inhibit the growth of human breast cancer cells in vitro and in vivo. Cancer Res. 2000, 60, 2673–2679. [Google Scholar] [PubMed]
- Van Belle, T.L.; Vanherwegen, A.S.; Feyaerts, D.; De Clercq, P.; Verstuyf, A.; Korf, H.; Gysemans, C.; Mathieu, C. 1,25-Dihydroxyvitamin D3 and its analog TX527 promote a stable regulatory T cell phenotype in T cells from type 1 diabetes patients. PLoS ONE 2014, 9, e109194. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, G.B.; Overbergh, L.; Verstuyf, A.; Mathieu, C. 1alpha,25- Dihydroxyvitamin D3 and its analogs as modulators of human dendritic cells: A comparison dose-titration study. J. Steroid Biochem. Mol. Biol. 2013, 136, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Verlinden, L.; Leyssens, C.; Beullens, I.; Marcelis, S.; Mathieu, C.; De Clercq, P.; Verstuyf, A. The vitamin D analog TX527 ameliorates disease symptoms in a chemically induced model of inflammatory bowel disease. J. Steroid Biochem. Mol. Biol. 2013, 136, 107–111. [Google Scholar] [CrossRef]
- Medioni, J.; Deplanque, G.; Ferrero, J.; Maurina, T.; Rodier, J.P.; Raymond, E.; Allyon, J.; Renaux, S.; Dufour-Lamartinie, J.; Oudard, S. Dose-finding and efficacy phase II study of inecalcitol, a new VDR agonist, in combination with docetaxel prednisone regimen for castration-resistant prostate cancer (CRPC) patients (pts). J. Clin. Oncol. 2011, 29, 142–146. [Google Scholar] [CrossRef]
- Medioni, J.; Deplanque, G.; Ferrero, J.M.; Maurina, T.; Rodier, J.M.P.; Raymond, E.; Allyon, J.; Maruani, G.; Houillier, P.; Mackenzie, S.; et al. Phase I safety and pharmacodynamic of inecalcitol, a novel VDR agonist with docetaxel in metastatic castration-resistant prostate cancer patients. Clin. Cancer Res. 2014, 20, 4471–4477. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Yu, W.D.; Hidalgo, A.A.; Luo, W.; Delansorne, R.; Johnson, C.S.; Trump, D.L. Inecalcitol, an analog of 1,25D3, displays enhanced antitumor activity through the induction of apoptosis in a squamous cell carcinoma model system. Cell Cycle 2013, 12, 743–752. [Google Scholar] [CrossRef] [Green Version]
- Kotlarz, A.; Przybyszewska, M.; Swoboda, P.; Miłoszewska, J.; Grygorowicz, M.A.; Kutner, A.; Markowicz, S. Differential interference of vitamin D analogs PRI-1906, PRI-2191, and PRI-2205 with the renewal of human colon cancer cells refractory to treatment with 5-fluorouracil. Tumor Biol. 2016, 37, 4699–4709. [Google Scholar] [CrossRef]
- Neska, J.; Swoboda, P.; Przybyszewska, M.; Kotlarz, A.; Bolla, N.R.; Miłoszewska, J.; Grygorowicz, M.A.; Kutner, A.; Markowicz, S. The Effect of Analogues of 1α,25-Dihydroxyvitamin D2 on the Regrowth and Gene Expression of Human Colon Cancer Cells Refractory to 5-Fluorouracil. Int. J. Mol. Sci. 2016, 17, 903. [Google Scholar] [CrossRef] [Green Version]
- Milczarek, M.; Rossowska, J.; Klopotowska, D.; Stachowicz, M.; Kutner, A.; Wietrzyk, J. Tacalcitol increases the sensitivity of colorectal cancer cells to 5-fluorouracil by downregulating the thymidylate synthase. J. Steroid Biochem. Mol. Biol. 2019, 190, 139–151. [Google Scholar] [CrossRef]
- Maj, E.; Filip-Psurska, B.; Milczarek, M.; Psurski, M.; Kutner, A.; Wietrzyk, J. Vitamin D derivatives potentiate the anticancer and anti-angiogenic activity of tyrosine kinase inhibitors in combination with cytostatic drugs in an A549 non-small cell lung cancer model. Int. J. Oncol. 2018, 52, 337–366. [Google Scholar] [PubMed]
- McElwain, M.C.; Dettelbach, M.A.; Modzelewski, R.A.; Russell, D.M.; Uskokovic, M.R.; Smith, D.C.; Trump, D.L.; Johnson, C.S. Antiproliferative effects in vitro and in vivo of 1,25-dihydroxyvitamin D3 and a vitamin D3 analog in a squamous cell carcinoma model system. Mol. Cell Differ. 1995, 3, 31–50. [Google Scholar]
- Zhang, X.; Jiang, F.; Li, P.; Li, C.; Ma, Q.; Nicosia, S.V.; Bai, W. Growth suppression of ovarian cancer xenografts in nude mice by vitamin D analogue EB1089. Clin. Cancer Res. 2005, 11, 323–328. [Google Scholar] [PubMed]
- Ma, Y.; Yu, W.D.; Trump, D.L.; Johnson, C.S. 1,25D3 enhances antitumor activity of gemcitabine and cisplatin in human bladder cancer models. Cancer 2010, 116, 3294–3303. [Google Scholar] [CrossRef] [Green Version]
- Colston, K.W.; Chander, S.K.; MaCkay, A.G.; Coombes, R.C. Effects of synthetic vitamin D analogues on breast cancer cell proliferation in vivo and in vitro. Biochem. Pharm. 1992, 44, 693–702. [Google Scholar] [CrossRef]
- Welsh, J.; Wietzke, J.A.; Zinser, G.M.; Byrne, B.; Smith, K.; Narvaez, C.J. Vitamin D-3 receptor as a target for breast cancer prevention. J. Nutr. 2003, 133 (Suppl. 7), 2425S–2433S. [Google Scholar] [CrossRef] [Green Version]
- Getzenberg, R.H.; Light, B.W.; Lapco, P.E.; Konety, B.R.; Nangia, A.K.; Acierno, J.S.; Dhir, R.; Shurin, Z.; Day, R.S.; Trump, D.L.; et al. Vitamin D inhibition of prostate adenocarcinoma growth and metastasis in the Dunning rat prostate model system. Urology 1997, 50, 999–1006. [Google Scholar] [CrossRef]
- Nakagawa, K.; Kawaura, A.; Kato, S.; Takeda, E.; Okano, T. 1 alpha,25-Dihydroxyvitamin D(3) is a preventive factor in the metastasis of lung cancer. Carcinogenesis 2005, 26, 429–440. [Google Scholar] [CrossRef]
- Colston, K.W.; James, S.Y.; Ofori-Kuragu, E.A.; Binderup, L.; Grant, A.G. Vitamin D receptors and antiproliferative effects of vitamin D derivatives in human pancreatic carcinoma cells in vivo and in vitro. Br. J. Cancer 1997, 76, 1017–1020. [Google Scholar] [CrossRef] [Green Version]
- Moore, T.B.; Koeffler, H.P.; Yamashiro, J.M.; Wada, R.K. Vitamin D3 analogs inhibit growth and induce differentiation in LA-N-5 human neuroblastoma cells. Clin. Exp. Metastasis 1996, 14, 239–245. [Google Scholar]
- Chung, I.; Han, G.; Seshadri, M.; Gillard, B.M.; Yu, W.D.; Foster, B.A.; Trump, D.L.; Johnson, C.S. Role of vitamin D receptor in the antiproliferative effects of calcitriol in tumor-derived endothelial cells and tumor angiogenesis in vivo. Cancer Res. 2009, 69, 967–975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, I.; Wong, M.K.; Flynn, G.; Yu, W.D.; Johnson, C.S.; Trump, D.L. Differential antiproliferative effects of calcitriol on tumor-derived and Matrigel-derived endothelial cells. Cancer Res. 2006, 66, 8565–8573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, I.; Karpf, A.R.; Muindi, J.R.; Conroy, J.M.; Nowak, N.J.; Johnson, C.S.; Trump, D.L. Epigenetic silencing of CYP24 in tumor-derived endothelial cells contributes to selective growth inhibition by calcitriol. J. Biol. Chem. 2007, 282, 8704–8714. [Google Scholar] [CrossRef] [Green Version]
- Ly, L.H.; Zhao, X.Y.; Holloway, L.; Feldman, D. Liarozole acts synergistically with 1alpha,25-dihydroxyvitamin D3 to inhibit growth of DU 145 human prostate cancer cells by blocking 24-hydroxylase activity. Endocrinology 1999, 140, 2071–2076. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.J.; Stapleton, G.E.; Hedlund, T.E.; Moffat, K.A. Vitamin D receptor expression, 24-hydroxylase activity, and inhibition of growth by 1alpha,25-dihydroxyvitamin D3 in seven human prostatic carcinoma cell lines. Clin. Cancer Res. 1995, 1, 997–1003. [Google Scholar] [PubMed]
- Peehl, D.M.; Seto, E.; Hsu, J.Y.; Feldman, D. Preclinical activity of ketoconazole in combination with calcitriol or the vitamin D analogue EB 1089 in prostate cancer cells. J. Urol. 2002, 168 Pt 1, 1583–1588. [Google Scholar] [CrossRef]
- Moffatt, K.A.; Johannes, W.U.; Miller, G.J. 1alpha,25-Dihydroxyvitamin D3 and platinum drugs act synergistically to inhibit the growth of prostate cancer cell lines. Clin. Cancer Res. 1999, 5, 695–703. [Google Scholar]
- Ma, Y.; Yu, W.D.; Hershberger, P.A.; Flynn, G.; Kong, R.X.; Trump, D.L.; Johnson, C.S. 1alpha,25-Dihydroxyvitamin D3 potentiates cisplatin antitumor activity by p73 induction in a squamous cell carcinoma model. Mol. Cancer Ther. 2008, 7, 3047–3055. [Google Scholar] [CrossRef] [Green Version]
- Trump, D.L.; Hershberger, P.A.; Bernardi, R.J.; Ahmed, S.; Muindi, J.; Fakih, M.; Yu, W.D.; Johnson, C.S. Anti-tumor activity of calcitriol: Pre-clinical and clinical studies. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 519–526. [Google Scholar] [CrossRef]
- Rochlitz, C.F.; Damon, L.E.; Russi, M.B.; Geddes, A.; Cadman, E.C. Cytotoxicity of ketoconazole in malignant cell lines. Cancer Chemother. Pharm. 1988, 21, 319–322. [Google Scholar] [CrossRef]
- Kahraman, M.; Sinishtaj, S.; Dolan, P.M.; Kensler, T.W.; Peleg, S.; Saha, U.; Chuang, S.S.; Bernstein, G.; Korczak, B.; Posner, G.H. Potent, selective and low-calcemic inhibitors of CYP24 hydroxylase: 24-sulfoximine analogues of the hormone 1alpha,25-dihydroxyvitamin D(3). J. Med. Chem. 2004, 47, 6854–6863. [Google Scholar] [CrossRef]
- Lechner, D.; Manhardt, T.; Bajna, E.; Posner, G.H.; Cross, H.S. A 24-phenylsulfone analog of vitamin D inhibits 1alpha,25-dihydroxyvitamin D (3) degradation in vitamin D metabolism-competent cells. J. Pharm. Exp. Ther. 2007, 320, 1119–1126. [Google Scholar] [CrossRef]
- Gallagher, J.C. Metabolic effects of synthetic calcitriol (Rocaltrol) in the treatment of postmenopausal osteoporosis. Metabolism 1990, 39 (Suppl. 1), 27–29. [Google Scholar] [CrossRef]
- Gallagher, J.C.; Goldgar, D. Treatment of postmenopausal osteoporosis with high doses of synthetic calcitriol. A randomized controlled study. Ann. Intern. Med. 1990, 113, 649–655. [Google Scholar] [CrossRef]
- Gross, C.; Stamey, T.; Hancock, S.; Feldman, D. Treatment of early recurrent prostate cancer with 1,25-dihydroxyvitamin D3 (calcitriol). J. Urol. 1998, 159, 2035–2039. [Google Scholar] [CrossRef]
- Osborn, J.L.; Schwartz, G.G.; Smith, D.C.; Bahnson, R.; Day, R.; Trump, D.L. 1,25-Dihydroxyvitamin D (calcitriol) in hormone refractory prostate cancer. Urol. Oncol. 1995, 1, 195–198. [Google Scholar] [CrossRef]
- Muindi, J.R.; Peng, Y.; Potter, D.M.; Hershberger, P.A.; Tauch, J.S.; Capozzoli, M.J.; Egorin, M.J.; Johnson, C.S.; Trump, D.L. Pharmacokinetics of high-dose oral calcitriol: Results from a phase 1 trial of calcitriol and paclitaxel. Clin. Pharm. Ther. 2002, 72, 648–659. [Google Scholar] [CrossRef]
- Muindi, J.R.; Potter, D.M.; Peng, Y.; Johnson, C.S.; Trump, D.L. Pharmacokinetics of liquid calcitriol formulation in advanced solid tumor patients: Comparison with caplet formulation. Cancer Chemother. Pharm. 2005, 56, 492–496. [Google Scholar] [CrossRef]
- Beer, T.M.; Munar, M.; Henner, W.D. A phase I trial of pulse calcitriol in patients with refractory malignancies: Pulse dosing permits substantial dose escalation. Cancer 2001, 91, 2431–2439. [Google Scholar] [CrossRef]
- Fakih, M.G.; Trump, D.L.; Muindi, J.R.; Black, J.D.; Bernardi, R.J.; Creaven, P.J.; Schwartz, J.; Brattain, M.G.; Hutson, A.; French, R.; et al. A phase I pharmacokinetic and pharmacodynamic study of intravenous calcitriol in combination with oral gefitinib in patients with advanced solid tumors. Clin. Cancer Res. 2007, 13, 1216–1223. [Google Scholar] [CrossRef] [Green Version]
- Muindi, J.R.; Johnson, C.S.; Trump, D.L.; Christy, R.; Engler, K.L.; Fakih, M.G. A phase I and pharmacokinetics study of intravenous calcitriol in combination with oral dexamethasone and gefitinib in patients with advanced solid tumors. Cancer Chemother. Pharm. 2009, 65, 33–40. [Google Scholar] [CrossRef] [Green Version]
- Muindi, J.R.; Modzelewski, R.A.; Peng, Y.; Trump, D.L.; Johnson, C.S. Pharmacokinetics of 1alpha,25-dihydroxyvitamin D3 in normal mice after systemic exposure to effective and safe antitumor doses. Oncology 2004, 66, 62–66. [Google Scholar] [CrossRef]
- Beer, T.M.; Javle, M.M.; Ryan, C.W.; Garzotto, M.; Lam, G.N.; Wong, A.; Henner, W.D.; Johnson, C.S.; Trump, D.L. Phase I study of weekly DN-101, a new formulation of calcitriol, in patients with cancer. Cancer Chemother. Pharm. 2007, 59, 581–587. [Google Scholar] [CrossRef]
- Dalhoff, K.; Dancey, J.; Astrup, L.; Skovsgaard, T.; Hamberg, K.J.; Lofts, F.J.; Rosmorduc, O.; Erlinger, S.; Hansen, J.B.; Steward, W.P.; et al. A phase II study of the vitamin D analogue Seocalcitol in patients with inoperable hepatocellular carcinoma. Br. J. Cancer 2003, 89, 252–257. [Google Scholar] [CrossRef] [Green Version]
- Evans, T.R.J.; Colston, K.W.; Lofts, F.J.; Cunningham, D.; Anthoney, D.A.; Gogas, H.; De Bono, J.S.; Hamberg, K.J.; Skov, T.; Mansi, J.L. A phase II trial of the vitamin D analogue Seocalcitol (EB1089) in patients with inoperable pancreatic cancer. Br. J. Cancer 2002, 86, 680–685. [Google Scholar] [CrossRef]
- Gulliford, T.; English, J.; Colston, K.W.; Menday, P.; Moller, S.; Coombes, R.C. A phase I study of the vitamin D analogue EB 1089 in patients with advanced breast and colorectal cancer. Br. J. Cancer 1998, 78, 6–13. [Google Scholar] [CrossRef] [Green Version]
- Attia, S.; Eickhoff, J.; Wilding, G.; McNeel, D.; Blank, J.; Ahuja, H.; Jumonville, A.; Eastman, M.; Shevrin, D.; Glode, M.; et al. Randomized, double-blinded phase II evaluation of docetaxel with or without doxercalciferol in patients with metastatic, androgen-independent prostate cancer. Clin. Cancer Res. 2008, 14, 2437–2443. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Oettel, K.; Ripple, G.; Staab, M.J.; Horvath, D.; Alberti, D.; Arzoomanian, R.; Marnocha, R.; Bruskewitz, R.; Mazess, R.; et al. Phase I trial of 1alpha-hydroxyvitamin D (2) in patients with hormone refractory prostate cancer. Clin. Cancer Res. 2002, 8, 2820–2827. [Google Scholar]
- Liu, G.; Wilding, G.; Staab, M.J.; Horvath, D.; Miller, K.; Dresen, A.; Alberti, D.; Arzoomanian, R.; Chappell, R.; Bailey, H.H. Phase II study of 1alpha-hydroxyvitamin D (2) in the treatment of advanced androgen-independent prostate cancer. Clin. Cancer Res. 2003, 9, 4077–4083. [Google Scholar]
- Medioni, J.; Deplanque, G.; Maurina, T.; Ferrero, J.M.; Rodier, J.M.; Raymond, E.; Allyon, J.; Kalla, S.; Dufour-Lamartinie, J.F.; Oudard, S. Dose finding and safety analysis of inecalcitol in combination with a docetaxel-prednisone regimen in hormone-refractory prostate cancer (HRPC) patients. J. Clin. Oncol. 2009, 27, 5151. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Hall, M.C.; Stindt, D.; Patton, S.; Lovato, J.; Torti, F.M. Phase I/II study of 19-nor-1alpha-25-dihydroxyvitamin D2 (paricalcitol) in advanced, androgen-insensitive prostate cancer. Clin. Cancer Res. 2005, 11 Pt 1, 8680–8685. [Google Scholar] [CrossRef] [Green Version]
- Chan, J.S.; Beer, T.M.; Quinn, D.I.; Pinski, J.K.; Garzotto, M.; Sokoloff, M.; Dehaze, D.R.; Ryan, C.W. A phase II study of high-dose calcitriol combined with mitoxantrone and prednisone for androgen-independent prostate cancer. BJU Int. 2008, 102, 1601–1606. [Google Scholar] [CrossRef]
- Blanke, C.D.; Beer, T.M.; Todd, K.; Mori, M.; Stone, M.; Lopez, C. Phase II study of calcitriol-enhanced docetaxel in patients with previously untreated metastatic or locally advanced pancreatic cancer. Investig. New Drugs 2009, 27, 374–378. [Google Scholar] [CrossRef]
- Chadha, M.K.; Tian, L.; Mashtare, T.; Payne, V.; Silliman, C.; Levine, E.; Wong, M.; Johnson, C.; Trump, D.L. Phase 2 trial of weekly intravenous 1,25-dihydroxycholecalciferol (calcitriol) in combination with dexamethasone for castration-resistant prostate cancer. Cancer 2010, 116, 2132–2139. [Google Scholar]
- Scher, H.I.; Jia, X.; Chi, K.; de Wit, R.; Berry, W.R.; Albers, P.; Henick, B.; Waterhouse, D.; Ruether, D.J.; Rosen, P.J.; et al. Randomized, open-label phase III trial of docetaxel plus high-dose calcitriol versus docetaxel plus prednisone for patients with castration-resistant prostate cancer. J. Clinoncol. 2011, 29, 2191–2198. [Google Scholar] [CrossRef]
- Shams eddine, A.; Farhat, F.S.; Elias, E.; Khauli, R.B.; Saleh, A.; Bulbul, M.A. High-dose calcitriol, docetaxel and zoledronic acid in patients with castration-resistant prostate cancer: A phase II study. Urol. Int. 2013, 90, 56–61. [Google Scholar] [CrossRef]
- Wagner, D.; Trudel, D.; Van der Kwast, T.; Nonn, L.; Giangreco, A.A.; Li, D.; Dias, A.; Cardoza, M.; Laszlo, S.; Hersey, K.; et al. Randomized clinical trial of vitamin D3 doses on prostatic vitamin D metabolite levels and ki67 labeling in prostate cancer patients. J. Clin. Endocrinol. Metab. 2013, 98, 1498–1507. [Google Scholar] [CrossRef] [Green Version]
- Cheng, T.Y.D.; LaCroix, A.Z.; Beresford, S.A.; Goodman, G.E.; Thornquist, M.D.; Zheng, Y.; Chlebowski, R.T.; Ho, G.Y.; Neuhouser, M.L. Vitamin D intake and lung cancer risk in the Women’s Health Initiative. Am. J. Clin. Nutr. 2013, 98, 1002–1011. [Google Scholar] [CrossRef] [Green Version]
- Pommergaard, H.C.; Burcharth, J.; Rosenberg, J.; Raskov, H. Aspirin, calcitriol, and calcium donot prevent adenoma recurrence in a randomized controlled trial. Gastroenterology 2016, 150, 114–122. [Google Scholar] [CrossRef]
- Brinkhuizen, T.; Frencken, K.J.; Nelemans, P.J.; Hoff, M.L.; Kelleners-Smeets, N.W.; zur Hausen, A.; van der Horst, M.P.; Rennspiess, D.; Winnepenninckx, V.J.; van Steensel, M.A.; et al. The effect of topical diclofenac 3% and calcitriol 3 μg/g on superficial basal cell carcinoma (sBCC) and nodular basal cell carcinoma (nBCC):a phase II, randomized controlled trial. J. Am. Acad. Derm. 2016, 75, 126–134. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Barry, E.L.; Baron, J.A.; Rutherford, R.E.; Seabrook, M.E.; Bostick, R.M. Effects of supplemental calcium and vitamin D on the APC/b-catenin pathway in the normal colorectal mucosa of colorectal adenoma patients. Mol. Carcinog. 2017, 56, 412–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lappe, J.; Watson, P.; Travers-Gustafson, D.; Recker, R.; Garland, C.; Gorham, E.; Baggerly, K.; McDonnell, S.L. Effect of vitamin D and calcium supplementation on cancer incidence in older women: A randomized clinical trial. JAMA 2017, 317, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.L.; Peacock, J.L.; Rees, J.R.; Bostick, R.M.; Robertson, D.J.; Bresalier, R.S.; Baron, J.A. Vitamin D receptor genotype, vitamin D3 supplementation, and risk of colorectal adenomas: A randomized clinical trial. JAMA Oncol. 2017, 3, 628–635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manson, J.E.; Bassuk, S.S.; Lee, I.M.; Cook, N.R.; Albert, M.A.; Gordon, D.; Zaharris, E.; MacFadyen, J.G.; Danielson, E.; Lin, J.; et al. The VITamin D and OmegA-3 TriaL (VITAL): Rationale and design of a large randomized controlled trial of vitamin D and marine omega-3 fatty acid supplements for the primary prevention of cancer and cardiovascular disease. Contemp. Clin. Trials 2012, 33, 159–171. [Google Scholar] [CrossRef] [Green Version]
- Pradhan, A.D.; Manson, J.E. Update on the Vitamin D and OmegA-3 trial (VITAL). J. Steroid Biochem. Mol. Biol. 2016, 155 Pt B, 252–256. [Google Scholar] [CrossRef] [Green Version]
- Bassuk, S.S.; Manson, J.E.; Lee, I.M.; Cook, N.R.; Christen, W.G.; Bubes, V.Y.; Gordon, D.S.; Copeland, T.; Friedenberg, G.; D’Agostino, D.M.; et al. Baseline characteristics of participants in the VITamin D and OmegA-3 TriaL (VITAL). Contemp. Clin. Trials 2016, 47, 235–243. [Google Scholar] [CrossRef] [Green Version]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Albert, C.M.; Gordon, D.; Copeland, T.; et al. Marine n-3 Fatty Acids and Prevention of Cardiovascular Disease and Cancer. N. Engl. J. Med. 2019, 380, 23–32. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef]
- Beer, T.M.; Eilers, K.M.; Garzotto, M.; Egorin, M.J.; Lowe, B.A.; Henner, W.D. Weekly high-dose calcitriol and docetaxel in metastatic androgen-independent prostate cancer. J. Clin. Oncol. 2003, 21, 123–128. [Google Scholar] [CrossRef]
- Beer, T.M.; Ryan, C.W.; Venner, P.M.; Petrylak, D.P.; Chatta, G.S.; Ruether, J.D.; Redfern, C.H.; Fehrenbacher, L.; Saleh, M.N.; Waterhouse, D.M.; et al. Double-blinded randomized study of high-dose calcitriol plus docetaxel compared with placebo plus docetaxel in androgen-independent prostate cancer: A report from the ASCENT investigators. J. Clin. Oncol. 2007, 25, 669–674. [Google Scholar] [CrossRef]
- Beer, T.M.; Garzotto, M.; Katovic, N.M. High-dose calcitriol and carboplatin in metastatic androgen-independent prostate cancer. Am. J. Clin. Oncol. 2004, 27, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Flaig, T.W.; Barqawi, A.; Miller, G.; Kane, M.; Zeng, C.; Crawford, E.D.; Glodé, L. A phase II trial of dexamethasone, vitamin D, and carboplatin in patients with hormone-refractory prostate cancer. Cancer 2006, 107, 266–274. [Google Scholar] [CrossRef] [PubMed]
| Pathway | Experimental System Involved | Action of Vitamin D or Its Analogues |
|---|---|---|
| PI3KC3 | Human leukemia cell line, HL60 | PI3KC3 increased → induce autophagy [145]. |
| mTOR | HL60 | mTOR decreased → induce autophagy [145]. |
| Beclin 1 | HL60, MCF-7, human primary monocytes/macrophages, THP-1 | Beclin 1 increased → increase autophagy [145,146,147]. |
| Cathelicidin | THP-1 | Cathelicidin increased → Beclin-1 increased → increase autophagy (through promotion of fusion between lysosome and autophagosome) [147] |
| Bcl-2 | Human breast carcinoma cell line, MCF-7 | Decrease inhibition of Bcl-2 on Beclin 1 → induce autophagy [148]. Decrease endoplasmic reticulum → Bcl-2 → induce autophagy [148]. |
| Calcium | Human breast carcinoma (MCF-7, MCF10A) | Increase free cytosolic calcium → inhibit mTOR → induce autophagy [148] |
| Cyclin-dependent kinase (CDK) inhibitor, p19INK4D | Human head and neck squamous cell carcinoma (SCC25) | Decrease p19INK4D → induce autophagy [149] |
| Participants | Regimen | Aim | Main Results | Status | [Ref.] |
|---|---|---|---|---|---|
| 250 patients | Docetaxel (36 mg/m2, i.v. /week) for 4 weeks + oral DN-101 (45 μg) or placebo/day before docetaxel | Studying the safety and efficacy of the combination between DN-101 and docetaxel compared to docetaxel and placebo on AIPC | Oral DN-101 prolonged the survival of AIPC patients compared with placebo. | Completed | [216] |
| 19 patients | Oral DN-101 (180 μg) on the 1st day and i.v. mitoxantrone (12 mg/m2) on the 2nd day every 21 days + prednisone (10 mg orally) daily for 12 cycles maximally | Studying the safety and efficacy of the combination between DN-101, mitoxantrone and glucocorticoids in AIPC | Despite DN-101 addition doesn’t add significant activity to mitoxantrone and prednisone, it seems to reduce the toxicity of mitoxantrone in AIPC | Completed | [225] |
| 25 patients | Day 1: oral 1,25(OH)2D3 (0.5 mg/kg). Day 2: i.v. docetaxel (36 mg/m2)/ week for 3 consecutive weeks followed by 1-week with no treatment | Studying the safety and efficacy of the combination between the high-dose oral 1,25(OH)2D3 and docetaxel in patients with non-resectable, incurable pancreatic cancer | Using high-dose calcitriol with docetaxel may have activity in incurable pancreatic cancer, when compared to historical findings using single-agent docetaxel | Completed | [226] |
| 18 patients | i.v. 1,25(OH)2D3 (74 mg) weekly and dexamethasone in patients with CRPC | Prevention and treatment of CRPC | i.v. high-dose calcitriol combined with dexamethasone was well-tolerated but failed clinically to produce effects in CRPC patients | Completed | [227] |
| 935 patients | ASCENT: 45 μg DN-101, 36 mg/m2 docetaxel, and 24 mg dexamethasone weekly for 3 of every 4 weeks. Control: 5 mg prednisone twice daily with 75 mg/m2 docetaxel and 24 mg dexamethasone every 3 weeks. | Comparing the efficacy and safety of docetaxel + DN-101 to docetaxel + prednisone in a phase III trial | ASCENT treatment was associated with shorter survival than the control. the trial was halted due to more deaths in ASCENT arm | Stopped | [228] |
| 23 patients | Oral calcitriol (0.5 µg/kg) in 4 divided doses over 4 h on day 1 of each treatment week, i.v. docetaxel (36 mg/m2) on day 2 of each treatment week and i.v. zoledronic acid (4 mg) on day 2 of the first and fifth week of each cycle. Treatment was administered weekly for 6 consecutive weeks on an 8-week cycle. | Studying safety and efficacy of combining high dose calcitriol with docetaxel and zoledronic acid in CRPC | The regimen was tolerated and PSA response was detected in half of the CRPC patients | Completed | [229] |
| 63 patients | Daily oral supplementation of vitamin D (400, 10,000, or 40,000 IU per day) | Ki67 labeling in surgical prostate tissue, PSA assessment and vitamin D metabolite levels | Oral vitamin D3 lowered PSA levels, increased prostate calcitriol levels and lowered Ki67 expression levels | Completed | [230] |
| 128,779 participants | Oral 400 IU vitamin D plus 1 g calcium per day | The association between vitamin D intake and lung cancer | In never-smoking, postmenopausal women, Vitamin D intake was associated with a lower lung cancer risk | Completed | [231] |
| 54 patients | Daily high-dose inecalcitriol (40–8000 μg) + docetaxel | Prevention and treatment of prostate cancer | Addition of inecalcitriol to docetaxel encouraged a favored PSA response in prostate cancer | Completed | [179] |
| 1107 patients | 1,25(OH)2D3 (0.5 μg) + acetyl salicylic acid (75 mg) + calcium carbonate (1250 mg) (n = 209) or placebo (n = 218) | Adenoma recurrence after three-year treatment | Supplementing calcitriol didn’t reduce the risk of CRC recurrence | Completed | [232] |
| 64 cases + 64 controls | Topical diclofenac 3% + calcitriol 3 μg/g | BCC progression | Adding calcitriol to diclofenac treatment inhibited BCC proliferation | Completed | [233] |
| 104 patients with CRC | Daily vitamin D (1000 IU) alone or daily calcium (1200 mg) alone and in combination or placebo | APC/β-catenin pathway in normal colorectal mucosa | Vitamin D intake significantly suppressed APC/β-catenin pathway. | Completed | [234] |
| 2303 healthy postmenopausal women | Treatment group: 2000 IU/d of vitamin D and 1500 mg/d of calcium Placebo group: received identical placebos | all-type cancer risk (excluding non-melanoma skin cancers) | Supplementation of Vitamin D and calcium did not result in a significantly lower risk of all-type cancer | Completed | [235] |
| 2259 patients with colon adenoma | Daily oral vitamin D (1000 IU) alone or daily oral calcium carbonate (1200 mg) alone and in combination or placebo | colorectal adenoma recurrence | 1- Vitamin D prevent CC recurrence among individuals with AA genotype in VDR rs7969585 polymorphism. 2- Vitamin D3 supplementation for the prevention of advanced colorectal adenomas may vary according to vitamin D receptor genotype. | Completed | [236] |
| 25,871 participants | (1) Daily vitamin D3 (cholecalciferol, 2000 IU per day) + Omega-3 fatty acids (465 mg of eicosapentaenoic acid [EPA] and 375 mg of docosahexaenoic acid [DHA]) (2) Daily vitamin D3 (cholecalciferol, 2000 IU per day) + Placebo (omega-3 fatty acids) (3) Omega-3 fatty acids (465 mg of eicosapentaenoic acid [EPA] and 375 mg of docosahexaenoic acid [DHA]) + Placebo (vitamin D3) (4) Placebo (Vitamin D3) + Placebo (omega-3 fatty acids) | Risk for developing cancer and CVD | Supplementing vitamin D didn’t reduce the risk of development of invasive cancer or cardiovascular disease compared to placebo | Current | [219,220,221,222,223] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Sharkawy, A.; Malki, A. Vitamin D Signaling in Inflammation and Cancer: Molecular Mechanisms and Therapeutic Implications. Molecules 2020, 25, 3219. https://doi.org/10.3390/molecules25143219
El-Sharkawy A, Malki A. Vitamin D Signaling in Inflammation and Cancer: Molecular Mechanisms and Therapeutic Implications. Molecules. 2020; 25(14):3219. https://doi.org/10.3390/molecules25143219
Chicago/Turabian StyleEl-Sharkawy, Ahmed, and Ahmed Malki. 2020. "Vitamin D Signaling in Inflammation and Cancer: Molecular Mechanisms and Therapeutic Implications" Molecules 25, no. 14: 3219. https://doi.org/10.3390/molecules25143219
APA StyleEl-Sharkawy, A., & Malki, A. (2020). Vitamin D Signaling in Inflammation and Cancer: Molecular Mechanisms and Therapeutic Implications. Molecules, 25(14), 3219. https://doi.org/10.3390/molecules25143219






