Triazolopyridopyrimidine: A New Scaffold for Dual-Target Small Molecules for Alzheimer’s Disease Therapy
Abstract
1. Introduction
2. Results
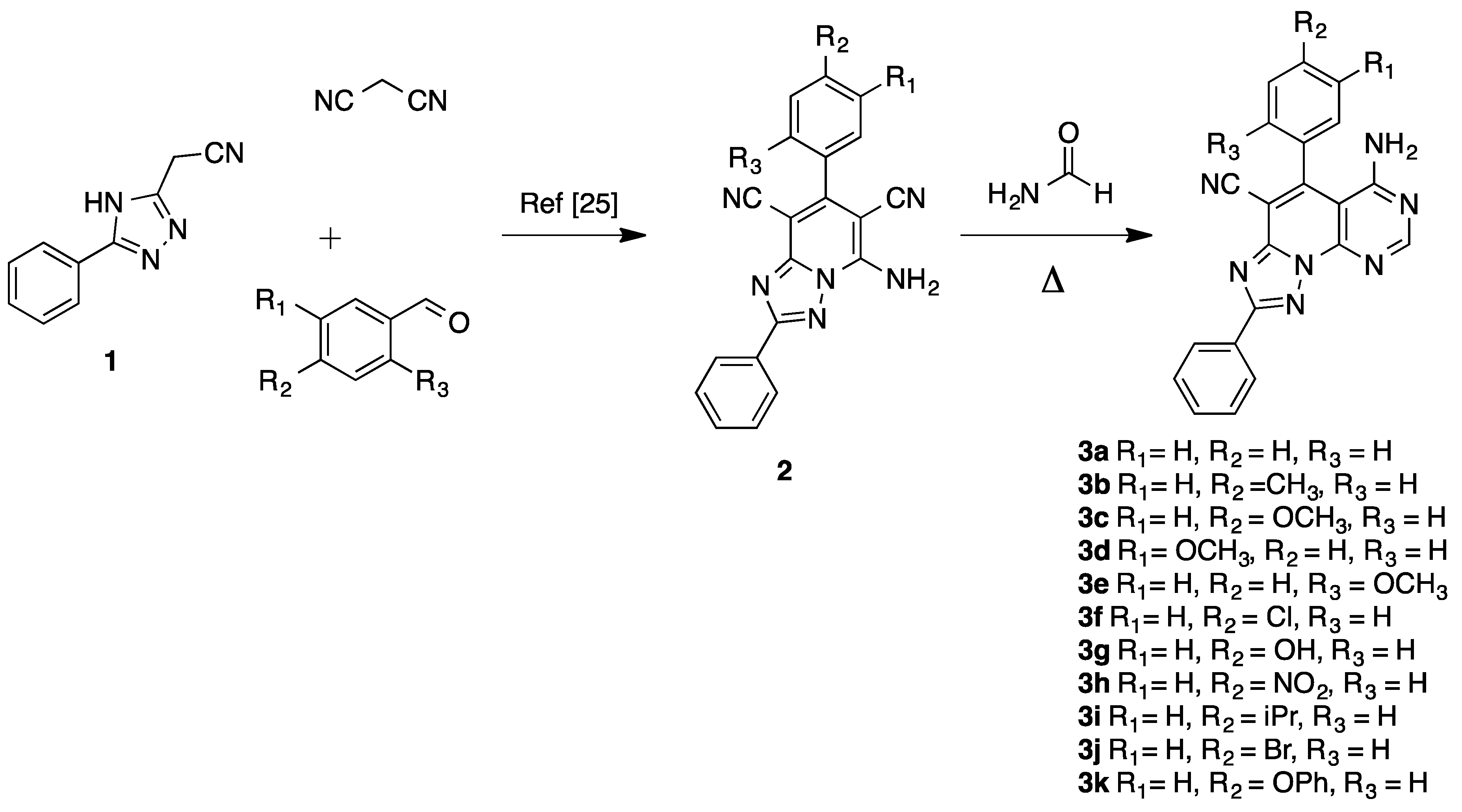
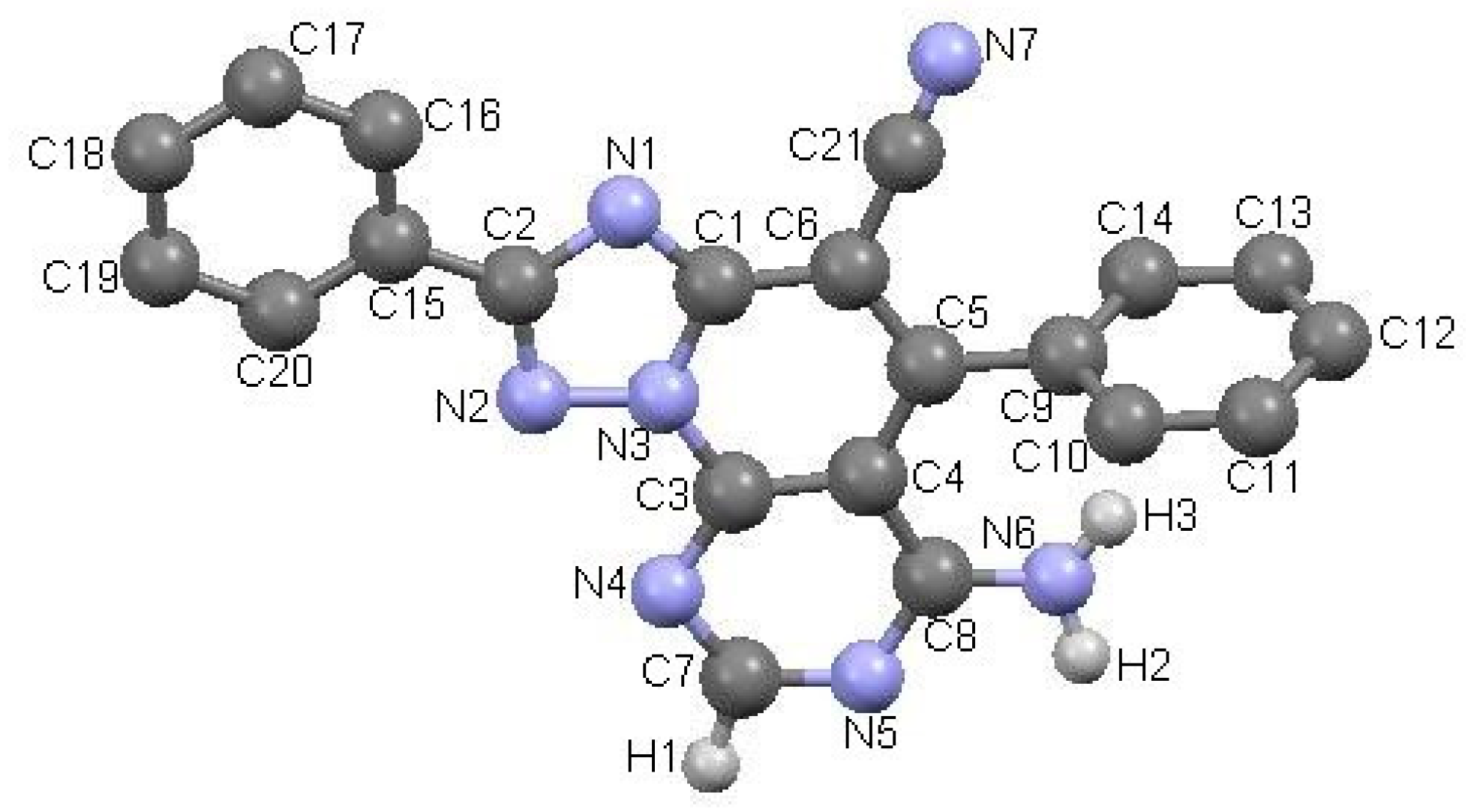
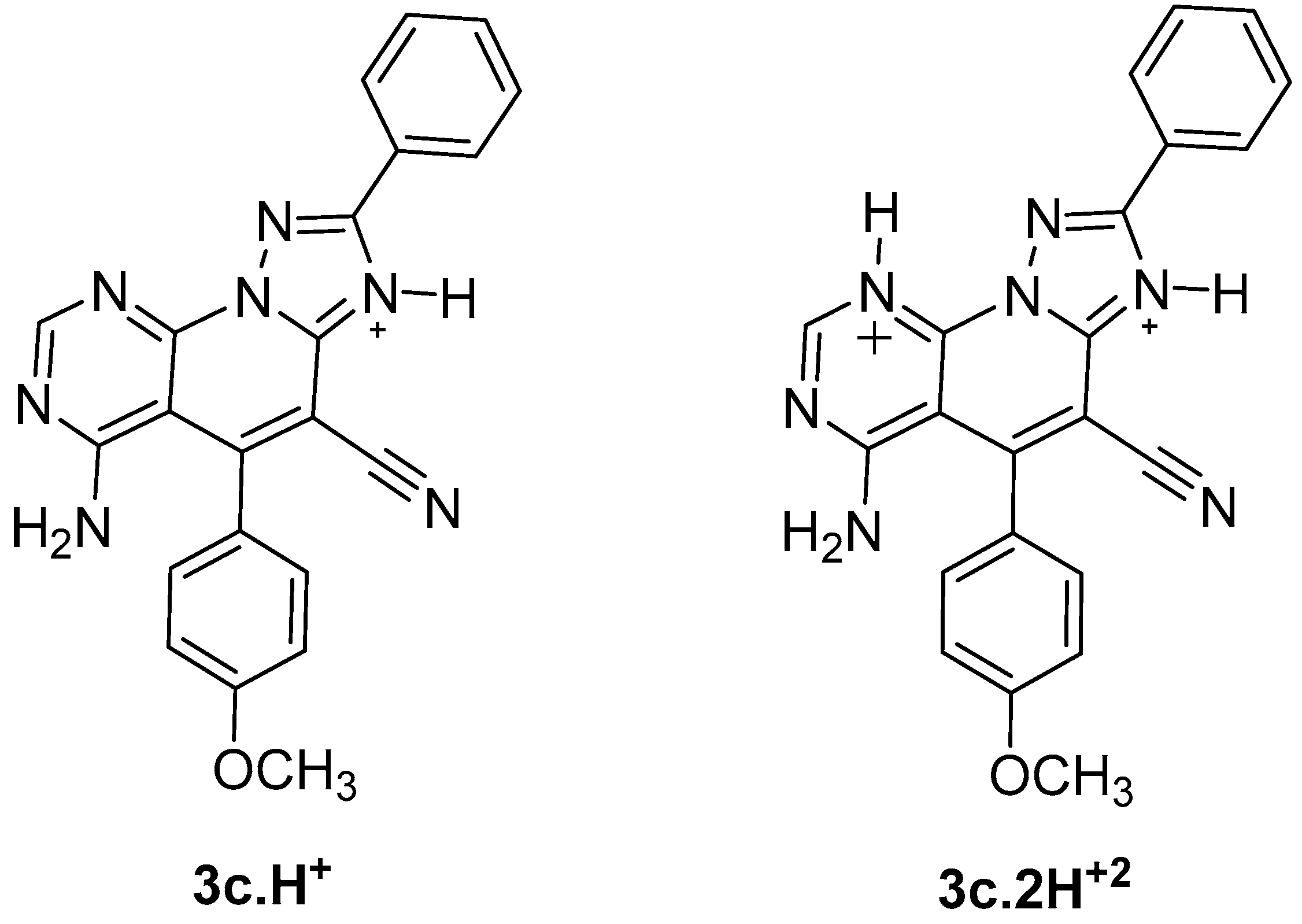
2.1. Synthesis
2.2. Biological Evaluation
2.2.1. Antioxidant Analysis
2.2.2. Inhibition of EeAChE/eqBuChE
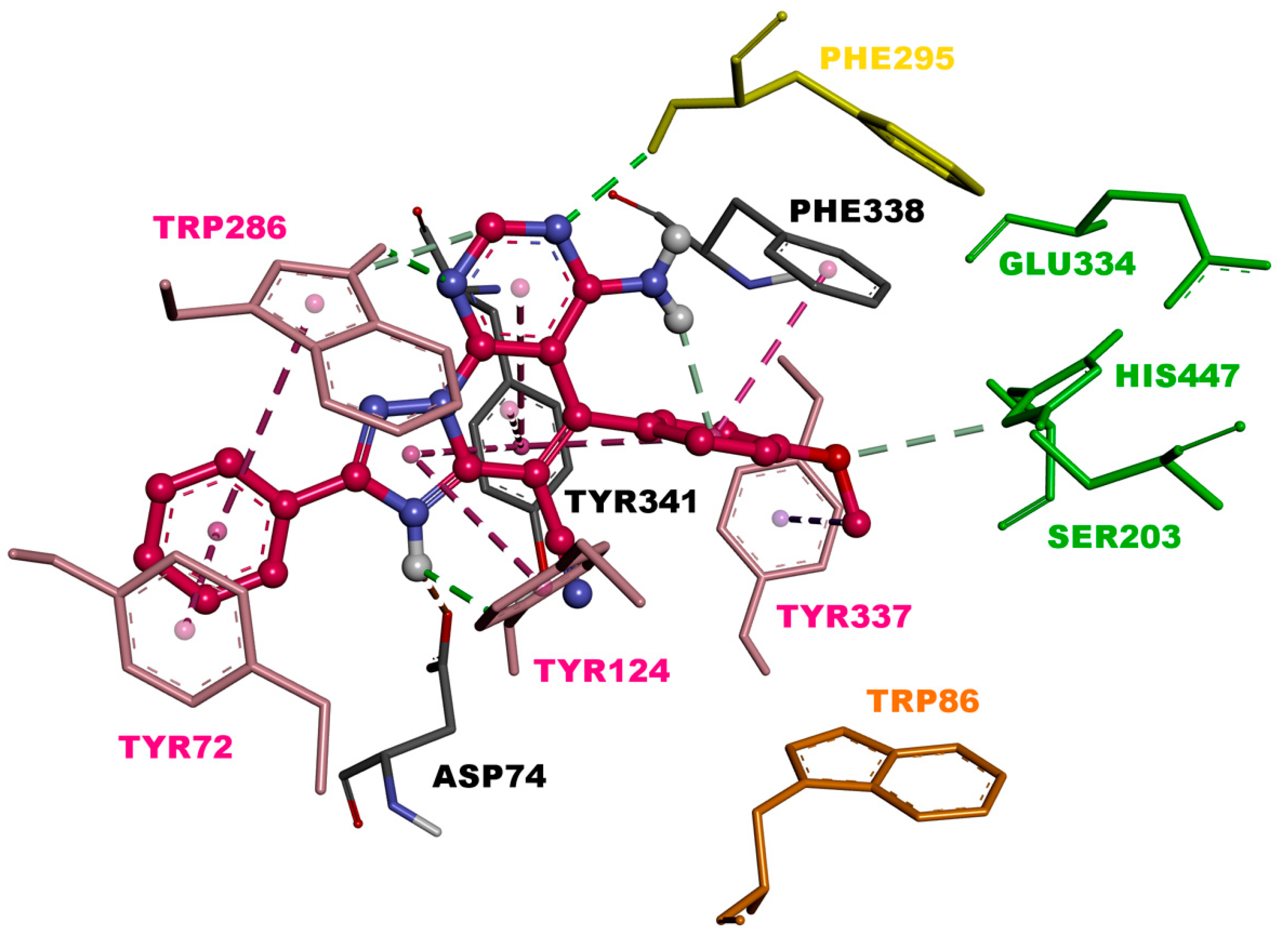
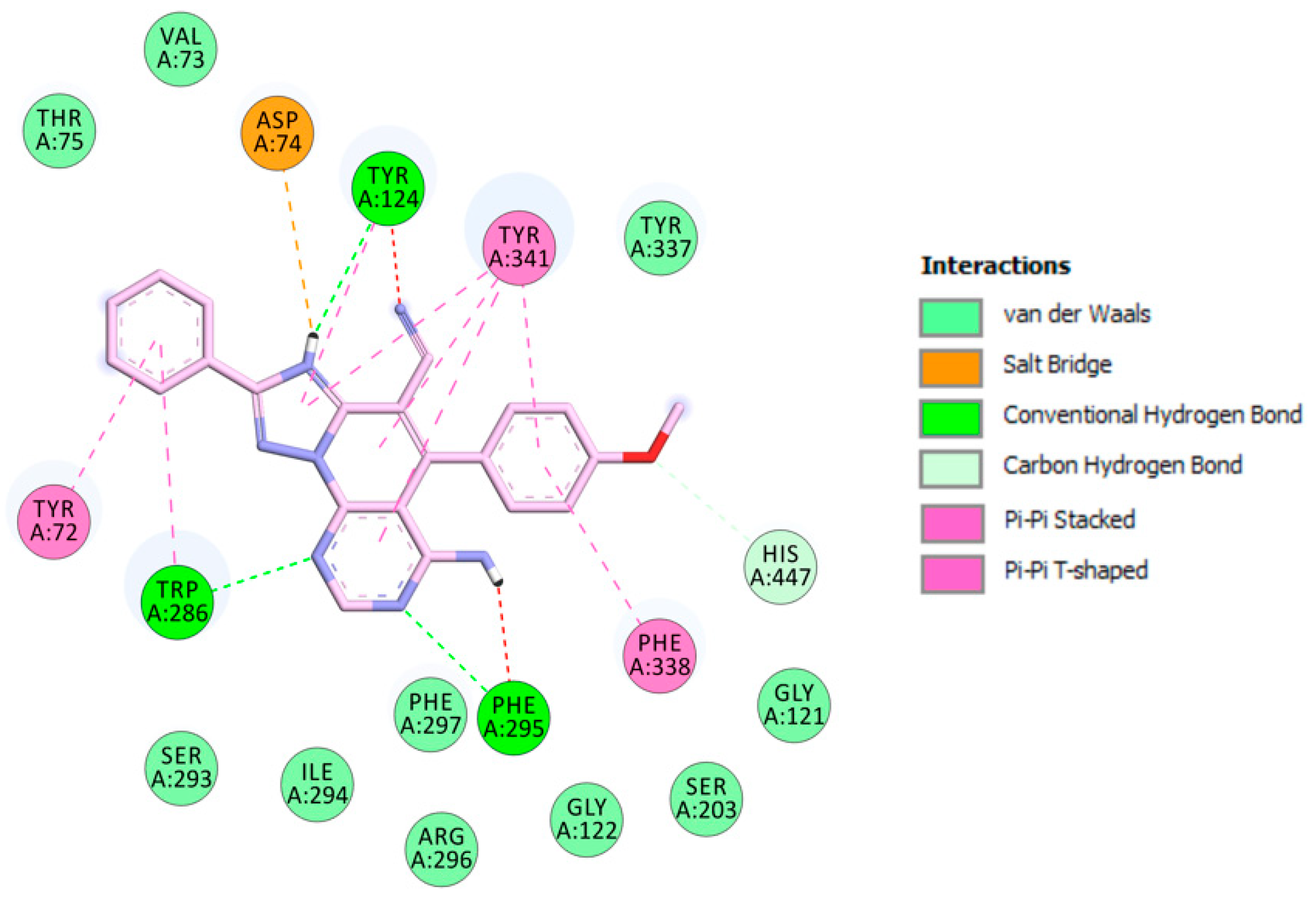
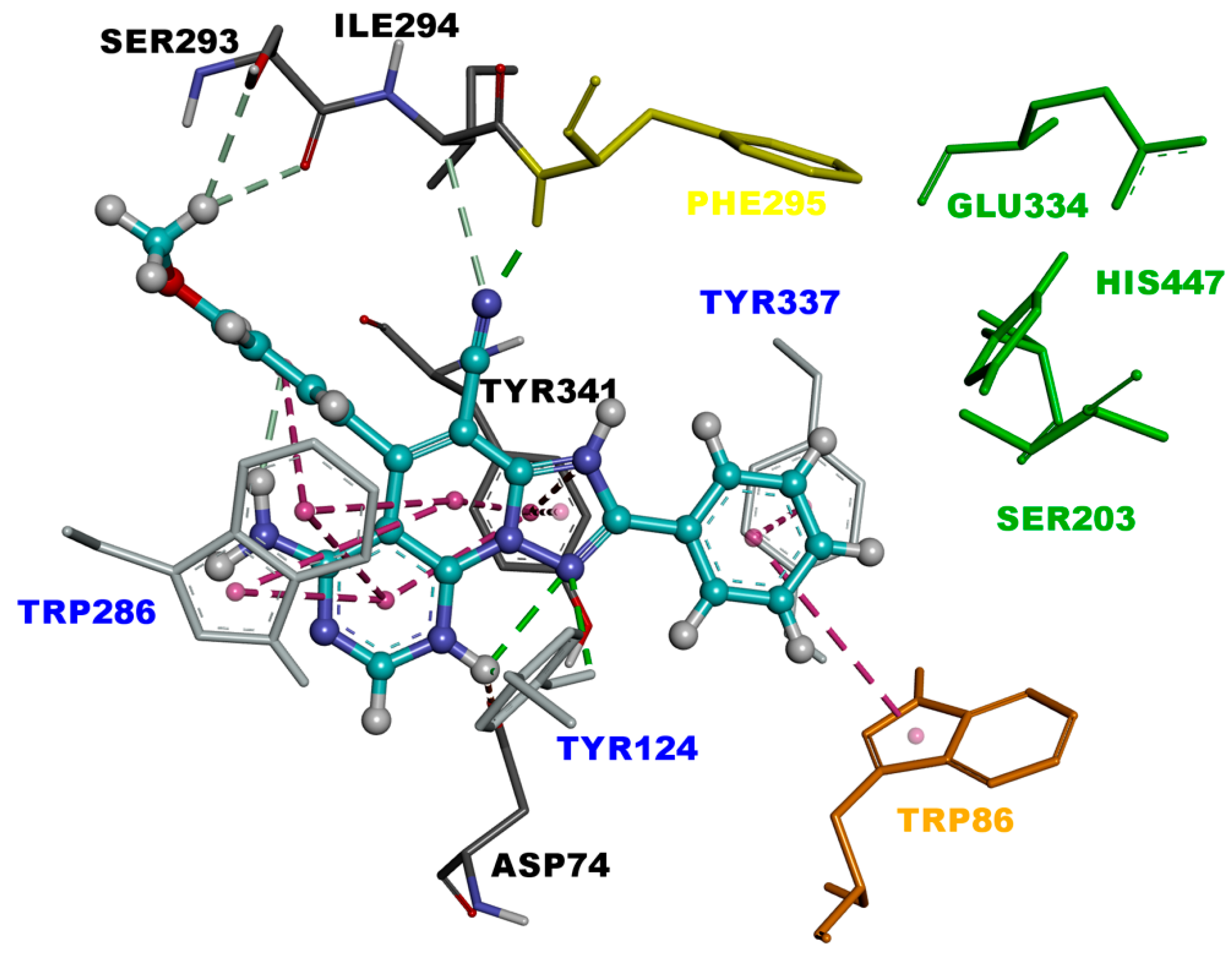
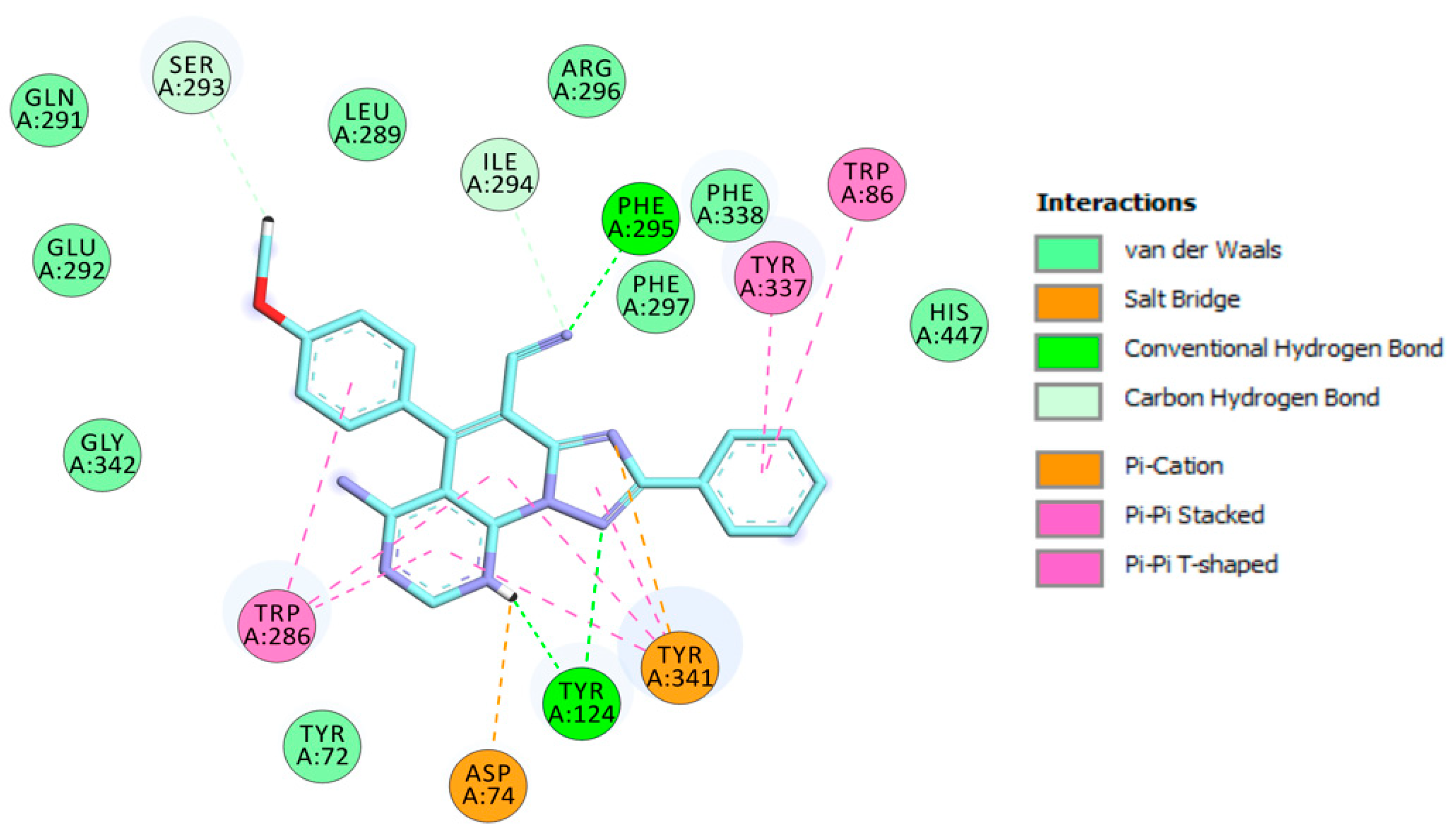
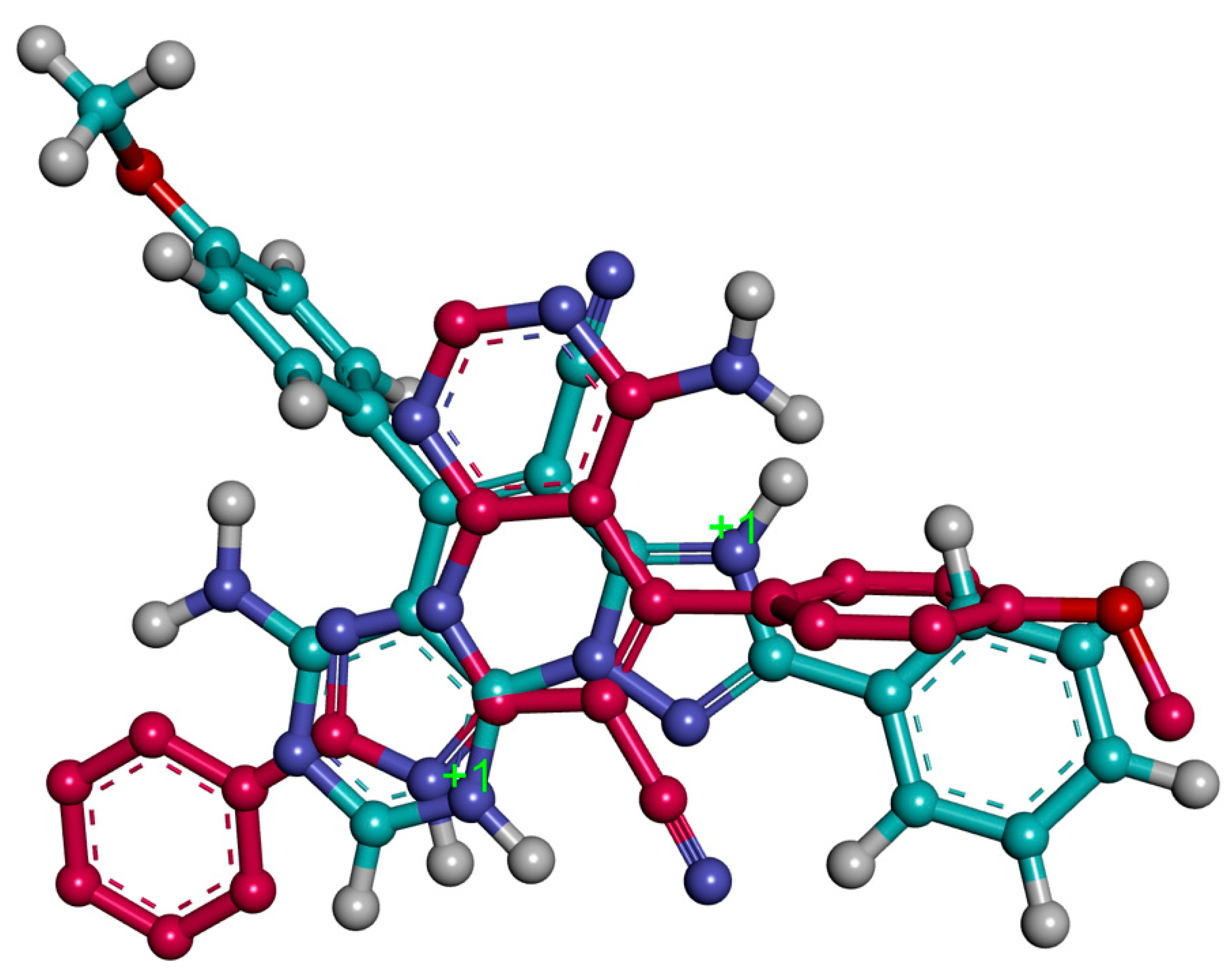
2.3. Molecular Modeling
3. Materials and Methods
3.1. Synthesis of Compounds 3a–k
3.2. Biological Evaluation
3.2.1. Oxygen Radical Absorbance Capacity Assay
3.2.2. Inhibition of EeAChE and eqBuChE
3.2.3. Molecular Modeling
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ahmad, H.; Ahmad, S.; Ali, M.; Latif, A.; Shah, S.A.A.; Naz, H.; ur Rahman, N.; Shaheen, F.; Wadood, A.; Khan, H.U.; et al. Norditerpenoid alkaloids of Delphinium denudatum as cholinesterase inhibitors. Bioorg. Chem. 2018, 78, 427–435. [Google Scholar] [CrossRef]
- Ulrich, J.D.; Holtzman, D.M. TREM2 Function in Alzheimer’s Disease and Neurodegeneration. ACS Chem. Neurosci. 2016, 7, 420–427. [Google Scholar] [CrossRef]
- Bouza, C.; Martínez-Alés, G.; López-Cuadrado, T. Effect of dementia on the incidence, short-term outcomes, and resource utilization of invasive mechanical ventilation in the elderly: A nationwide population-based study. Crit. Care 2019, 23, 291. [Google Scholar] [CrossRef]
- Frölich, L. The cholinergic pathology in Alzheimer’s disease--discrepancies between clinical experience and pathophysiological findings. J. Neural Transm (Vienna) 2002, 109, 1003–1013. [Google Scholar] [CrossRef]
- Tumiatti, V.; Minarini, A.; Bolognesi, M.L.; Milelli, A.; Rosini, M.; Melchiorre, C. Tacrine derivatives and Alzheimer’s disease. Curr. Med. Chem. 2010, 17, 1825–1838. [Google Scholar] [CrossRef]
- Rosini, M.; Simoni, E.; Milelli, A.; Minarini, A.; Melchiorre, C. Oxidative stress in Alzheimer’s disease: Are we connecting the dots? J. Med. Chem. 2014, 57, 2821–2831. [Google Scholar] [CrossRef]
- Bush, A.I. Drug development based on the metals hypothesis of Alzheimer’s disease. J. Alzheimers Dis. 2008, 15, 223–240. [Google Scholar] [CrossRef]
- Bond, M.; Rogers, G.; Peters, J.; Anderson, R.; Hoyle, M.; Miners, A.; Moxham, T.; Davis, S.; Thokala, P.; Wailoo, A.; et al. The effectiveness and cost-effectiveness of donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer’s disease (review of Technology Appraisal No. 111): A systematic review and economic model. HTA 2012, 16. [Google Scholar] [CrossRef]
- Villarroya, M.; García, A.G.; Marco-Contelles, J.; López, M.G. An update on the pharmacology of galantamine. Expert Opin. Investig. Drugs 2007, 16, 1987–1998. [Google Scholar] [CrossRef]
- Wilkinson, D.; Wirth, Y.; Goebel, C. Memantine in Patients with Moderate to Severe Alzheimer’s Disease: Meta-Analyses Using Realistic Definitions of Response. Dement. Geriatr. Cogn. Disord. 2014, 37, 71–85. [Google Scholar] [CrossRef]
- Cavalli, A.; Bolognesi, M.L.; Minarini, A.; Rosini, M.; Tumiatti, V.; Recanatini, M.; Melchiorre, C. Multi-target-Directed Ligands To Combat Neurodegenerative Diseases. J. Med. Chem. 2008, 51, 347–372. [Google Scholar] [CrossRef]
- Morphy, R.; Rankovic, Z. Designing Multiple Ligands – Medicinal Chemistry Strategies and Challenges. Curr. Pharm. Des. 2009, 15, 587–600. [Google Scholar] [CrossRef] [PubMed]
- Agis-Torres, A.; Sollhuber, M.; Fernandez, M.; Sanchez-Montero, J.M. Multi-Target-Directed Ligands and other Therapeutic Strategies in the Search of a Real Solution for Alzheimer’s Disease. Curr. Neuropharmacol. 2014, 12, 2–36. [Google Scholar] [CrossRef] [PubMed]
- León, R.; Garcia, A.G.; Marco-Contelles, J. Recent advances in the multitarget-directed ligands approach for the treatment of Alzheimer’s disease. Med. Res. Rev. 2013, 33, 139–189. [Google Scholar] [CrossRef] [PubMed]
- Ismaili, L.; Refouvelet, B.; Benchekroun, M.; Brogi, S.; Brindisi, M.; Gemma, S.; Campiani, G.; Filipic, S.; Agbaba, D.; Esteban, G.; et al. Multitarget compounds bearing tacrine- and donepezil-like structural and functional motifs for the potential treatment of Alzheimer’s disease. Prog. Neurobiol. 2017, 151, 4–34. [Google Scholar] [CrossRef] [PubMed]
- Benchekroun, M.; Ismaili, L.; Pudlo, M.; Luzet, V.; Gharbi, T.; Refouvelet, B.; Marco-Contelles, J. Donepezil–ferulic acid hybrids as anti-Alzheimer drugs. Future. Med. Chem. 2015, 7, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Benchekroun, M.; Romero, A.; Egea, J.; León, R.; Michalska, P.; Buendía, I.; Jimeno, M.L.; Jun, D.; Janockova, J.; Sepsova, V.; et al. The Antioxidant Additive Approach for Alzheimer’s Disease Therapy: New Ferulic (Lipoic) Acid Plus Melatonin Modified Tacrines as Cholinesterases Inhibitors, Direct Antioxidants, and Nuclear Factor (Erythroid-Derived 2)-Like 2 Activators. J. Med. Chem. 2016, 59, 9967–9973. [Google Scholar] [CrossRef]
- Ismaili, L.; do Carmo Carreiras, M. Multicomponent Reactions for Multitargeted Compounds for Alzheimer`s Disease. Curr. Top. Med. Chem. 2018, 17, 3319–3327. [Google Scholar] [CrossRef]
- Martorana, A.; Giacalone, V.; Bonsignore, R.; Pace, A.; Gentile, C.; Pibiri, I.; Buscemi, S.; Lauria, A.; Piccionello, A.P. Heterocyclic Scaffolds for the Treatment of Alzheimer’s Disease. Curr. Pharm. Des. 2016, 22, 3971–3995. [Google Scholar] [CrossRef]
- Rastegari, A.; Nadri, H.; Mahdavi, M.; Moradi, A.; Mirfazli, S.S.; Edraki, N.; Moghadam, F.H.; Larijani, B.; Akbarzadeh, T.; Saeedi, M. Design, synthesis and anti-Alzheimer’s activity of novel 1,2,3-triazole-chromenone carboxamide derivatives. Bioorg. Chem. 2019, 83, 391–401. [Google Scholar] [CrossRef]
- Xu, M.; Peng, Y.; Zhu, L.; Wang, S.; Ji, J.; Rakesh, K.P. Triazole derivatives as inhibitors of Alzheimer’s disease: Current developments and structure-activity relationships. Eur. J. Med. Chem. 2019, 180, 656–672. [Google Scholar] [CrossRef] [PubMed]
- Jalili-Baleh, L.; Nadri, H.; Forootanfar, H.; Samzadeh-Kermani, A.; Küçükkılınç, T.T.; Ayazgok, B.; Rahimifard, M.; Baeeri, M.; Doostmohammadi, M.; Firoozpour, L.; et al. Novel 3-phenylcoumarin–lipoic acid conjugates as multi-functional agents for potential treatment of Alzheimer’s disease. Bioorg. Chem. 2018, 79, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated Docking with Selective Receptor Flexibility. J. Comput Chem 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Dehbi, O.; Tikad, A.; Bourg, S.; Bonnet, P.; Lozach, O.; Meijer, L.; Aadil, M.; Akssira, M.; Guillaumet, G.; Routier, S. Synthesis and optimization of an original V-shaped collection of 4-7-disubstituted Pyrido[3,2-d]pyrimidines as CDK5 and DYRK1A inhibitors. Eur. J. Med. Chem. 2014, 80, 352–363. [Google Scholar] [CrossRef]
- Zribi, L.; Zribi, F.; Marco-Contelles, J.; Chabchoub, F.; Ismaili, L. Facile one-pot synthesis of new [1,2,4]triazolo[1,5-a]pyridine derivatives by ultrasonic irradiation. Syn. Comm. 2017, 47, 1934–1939. [Google Scholar] [CrossRef]
- Dávalos, A.; Gómez-Cordovés, C.; Bartolomé, B. Extending applicability of the oxygen radical absorbance capacity (ORAC-fluorescein) assay. J. Agric. Food Chem. 2004, 52, 48–54. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Malek, R.; Arribas, R.L.; Palomino-Antolin, A.; Totoson, P.; Demougeot, C.; Kobrlova, T.; Soukup, O.; Iriepa, I.; Moraleda, I.; Diez-Iriepa, D.; et al. New Dual Small Molecules for Alzheimer’s Disease Therapy Combining Histamine H3 Receptor (H3R) Antagonism and Calcium Channels Blockade with Additional Cholinesterase Inhibition. J. Med. Chem. 2019, 62, 11416–11422. [Google Scholar] [CrossRef] [PubMed]
- Benchekroun, M.; Bartolini, M.; Egea, J.; Romero, A.; Soriano, E.; Pudlo, M.; Luzet, V.; Andrisano, V.; Jimeno, M.-L.; López, M.G.; et al. Novel Tacrine-Grafted Ugi Adducts as Multipotent Anti-Alzheimer Drugs: A Synthetic Renewal in Tacrine-Ferulic Acid Hybrids. ChemMedChem 2015, 10, 523–539. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtney, K.D.; Andres jr., V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Knez, D.; Sova, M.; Košak, U.; Gobec, S. Dual inhibitors of cholinesterases and monoamine oxidases for Alzheimer’s disease. Future Med. Chem. 2017, 9, 811–832. [Google Scholar] [CrossRef] [PubMed]
- Dgachi, Y.; Ismaili, L.; Knez, D.; Benchekroun, M.; Martin, H.; Szałaj, N.; Wehle, S.; Bautista-Aguilera, O.M.; Luzet, V.; Bonnet, A.; et al. Synthesis and Biological Assessment of Racemic Benzochromenopyrimidinimines as Antioxidant, Cholinesterase, and Aβ1−42 Aggregation Inhibitors for Alzheimer’s Disease Therapy. ChemMedChem 2016, 11, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Morreale, A.; Maseras, F.; Iriepa, I.; Gálvez, E. Ligand-receptor interaction at the neural nicotinic acetylcholine binding site: A theoretical model. J. Mol. Graph. Model. 2002, 21, 111–118. [Google Scholar]
Sample Availability: Samples of the compounds 3a–k are available from the authors. |

| Compound | R1 | R2 | R3 | ORAC a (TE) | % Inhibition of EeAChE b at 10 μM | IC50 (μM) c |
|---|---|---|---|---|---|---|
| 3a | H | H | H | 1.85 ± 0.1 | 40.27 ± 2.4 | nd |
| 3b | H | CH3 | H | 1.77 ± 0.1 | 73.13 ± 0.1 | 1.17 ± 0.0 |
| 3c | H | OCH3 | H | 4.01 ± 0.1 | 63.93 ± 1.8 | 1.32 ± 0.0 |
| 3d | OCH3 | H | H | 4.33 ± 0.1 | 31.23 ± 2.1 | nd |
| 3e | H | H | OCH3 | 3.79 ± 0.1 | 32.33 ± 2.4 | nd |
| 3f | H | Cl | H | 1.81 ± 0.1 | 43.90 ± 1.2 | nd |
| 3g | H | OH | H | 7.17 ± 0.2 | 36.43 ± 0.6 | nd |
| 3h | H | NO2 | H | 3.54 ± 0.1 | 44.63 ± 1.6 | nd |
| 3i | H | iPr | H | 1.89 ± 0.1 | 18.17 ± 1 | nd |
| 3j | H | Br | H | 2.57 ± 0.0 | 38.13 ± 1.1 | nd |
| 3k | H | OPh | H | 2.99 ± 0.2 | 18.60 ± 1.8 | nd |
| Melatonin | - | - | - | 2.45 ± 0.1 | - | - |
| Ferulic acid | - | - | - | 3.74 ± 0.2 | - | - |
| Tacrine | - | - | - | 95.63 ± 2.1 | 0.03 ± 0.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zribi, L.; Pachòn-Angona, I.; Bautista-Aguilera, Ò.M.; Diez-Iriepa, D.; Marco-Contelles, J.; Ismaili, L.; Iriepa, I.; Chabchoub, F. Triazolopyridopyrimidine: A New Scaffold for Dual-Target Small Molecules for Alzheimer’s Disease Therapy. Molecules 2020, 25, 3190. https://doi.org/10.3390/molecules25143190
Zribi L, Pachòn-Angona I, Bautista-Aguilera ÒM, Diez-Iriepa D, Marco-Contelles J, Ismaili L, Iriepa I, Chabchoub F. Triazolopyridopyrimidine: A New Scaffold for Dual-Target Small Molecules for Alzheimer’s Disease Therapy. Molecules. 2020; 25(14):3190. https://doi.org/10.3390/molecules25143190
Chicago/Turabian StyleZribi, Lazhar, Irene Pachòn-Angona, Òscar M. Bautista-Aguilera, Daniel Diez-Iriepa, José Marco-Contelles, Lhassane Ismaili, Isabel Iriepa, and Fakher Chabchoub. 2020. "Triazolopyridopyrimidine: A New Scaffold for Dual-Target Small Molecules for Alzheimer’s Disease Therapy" Molecules 25, no. 14: 3190. https://doi.org/10.3390/molecules25143190
APA StyleZribi, L., Pachòn-Angona, I., Bautista-Aguilera, Ò. M., Diez-Iriepa, D., Marco-Contelles, J., Ismaili, L., Iriepa, I., & Chabchoub, F. (2020). Triazolopyridopyrimidine: A New Scaffold for Dual-Target Small Molecules for Alzheimer’s Disease Therapy. Molecules, 25(14), 3190. https://doi.org/10.3390/molecules25143190









