Green and Sustainable Valorization of Bioactive Phenolic Compounds from Pinus By-Products
Abstract
1. Introduction
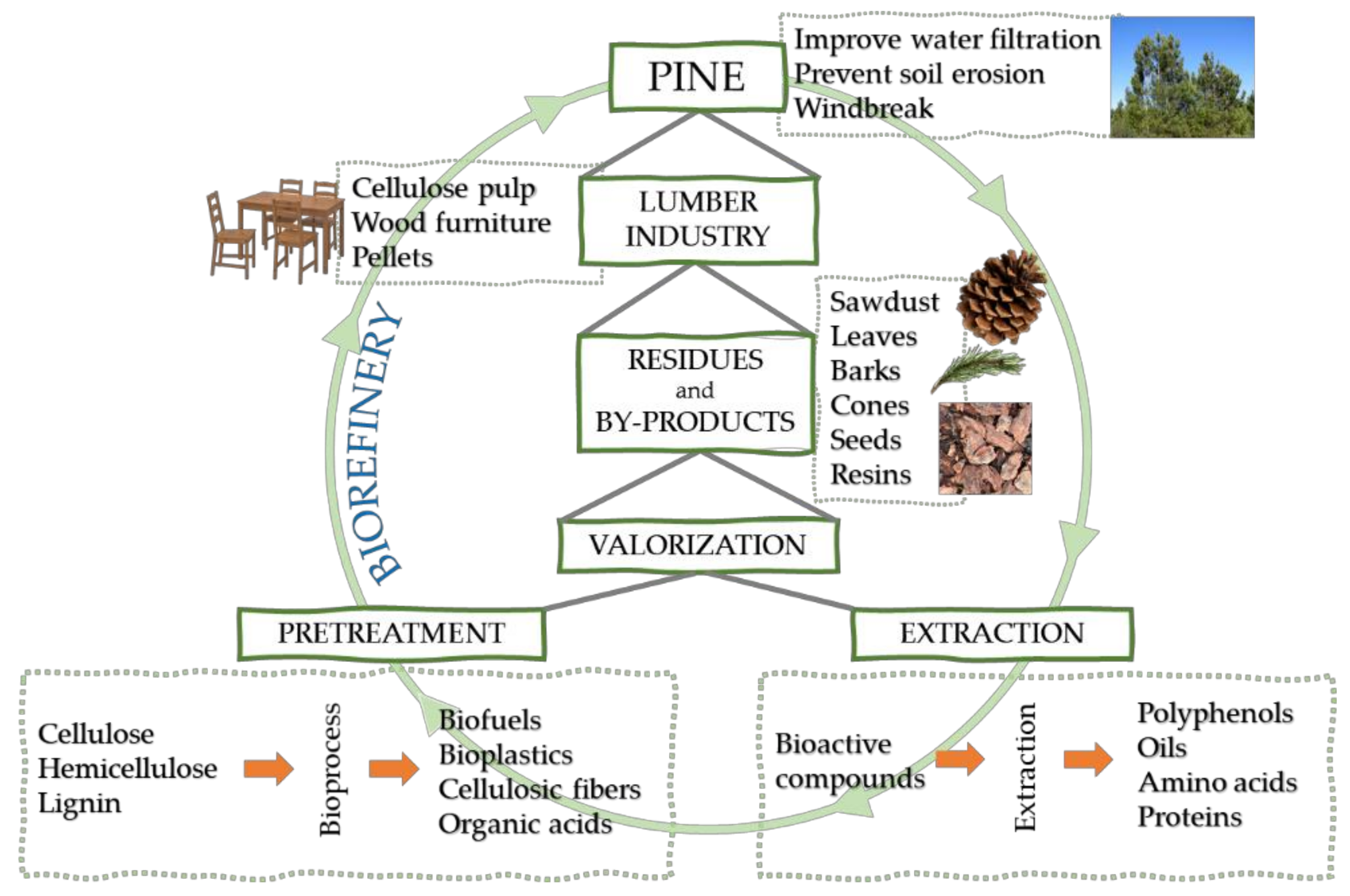
2. Biorefinery and Lignocellulosic By-Products
3. Pine as Feedstocks
Pine Applications
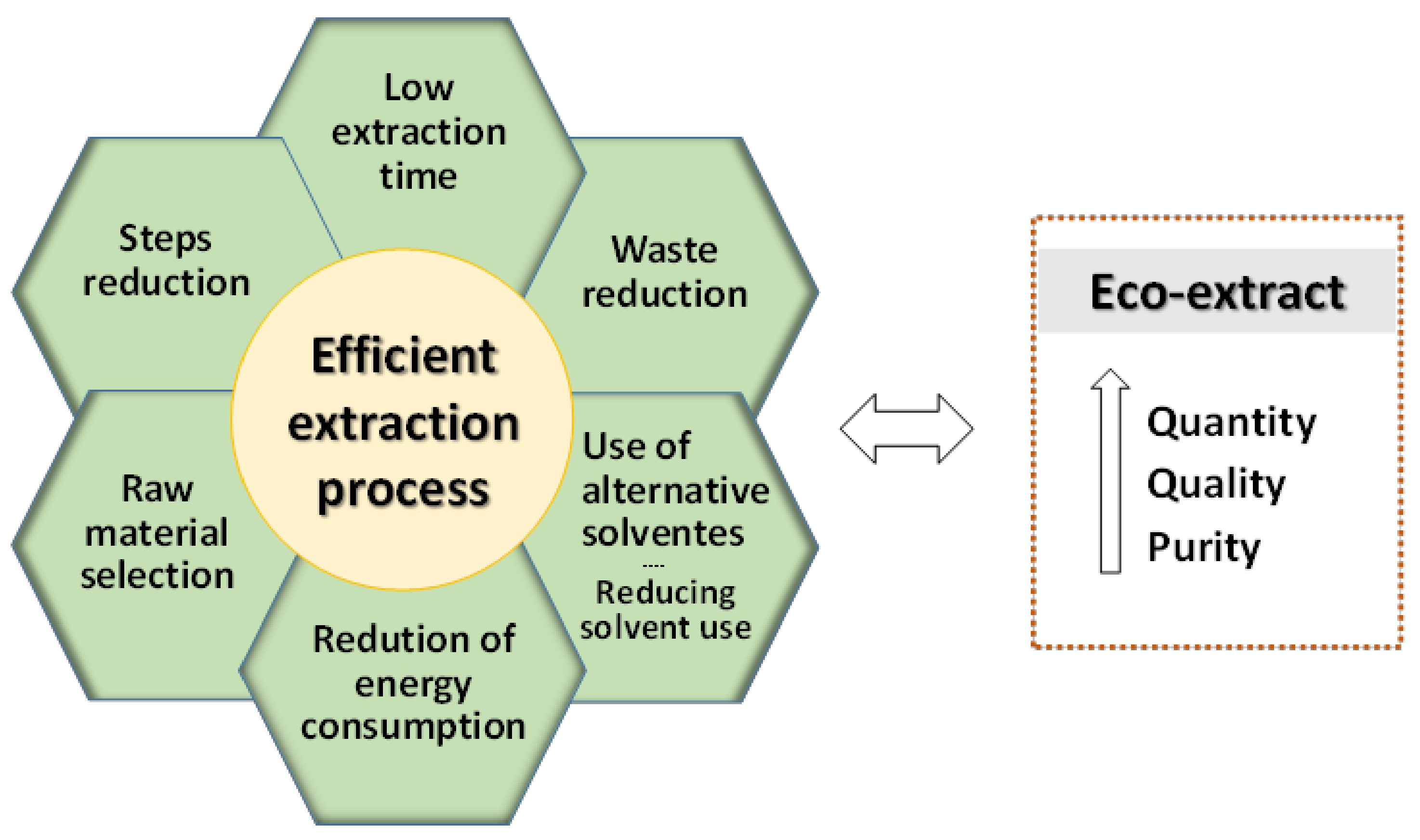
4. Extraction Processes for Phenolic Compounds Recovery
4.1. Extraction Solvents
4.2. Extraction Technologies
4.2.1. Ultrasound-Assisted Extraction
4.2.2. Microwave Assisted Extraction
4.2.3. Supercritical Fluid Extraction
4.2.4. Pressurized Liquid Extraction
4.2.5. Ohmic Heating Extraction
5. Polyphenols as Extracted Biocompounds
6. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- European Commission. Going Climate-Neutral by 2050: A Strategic Long-Term Vision for a Prosperous, Modern, Competitive and Climate-Neutral EU Economy. Available online: https://ec.europa.eu/clima/sites/clima/files/long_term_strategy_brochure_en.pdf (accessed on 21 January 2020).
- 2050 Long-Term Strategy | Energy. Available online: https://ec.europa.eu/energy/en/topics/energy-strategy-and-energy-union/2050-long-term-strategy (accessed on 21 January 2020).
- United Nations. Transforming our world: The 2030 Agenda for Sustainable Development. Available online: https://sustainabledevelopment.un.org/post2015/transformingourworld (accessed on 21 May 2020).
- Torres-Valenzuela, L.S.; Ballesteros-Gómez, A.; Rubio, S. Green solvents for the extraction of high added-value compounds from agri-food waste. Food Eng. Rev. 2019, 12, 83–100. [Google Scholar] [CrossRef]
- Naidu, D.S.; Hlangothi, S.P.; John, M.J. Bio-based products from xylan: A review. Carbohydr. Polym. 2018, 179, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Jablonský, M.; Škulcová, A.; Malvis, A.; Šima, J. Extraction of value-added components from food industry based and agro-forest biowastes by deep eutectic solvents. J. Biotechnol. 2018, 282, 46–66. [Google Scholar] [CrossRef] [PubMed]
- Cvjetko Bubalo, M.; Vidović, S.; Radojčić Redovniković, I.; Jokić, S. New perspective in extraction of plant biologically active compounds by green solvents. Food Bioprod. Process. 2018, 109, 52–73. [Google Scholar] [CrossRef]
- Chemat, F.; Abert-Vian, M.; Fabiano-Tixier, A.S.; Strube, J.; Uhlenbrock, L.; Gunjevic, V.; Cravotto, G. Green extraction of natural products. Origins, current status, and future challenges. Trac Trends Anal. Chem. 2019, 118, 248–263. [Google Scholar] [CrossRef]
- Herrero, M.; Ibañez, E. Green extraction processes, biorefineries and sustainability: Recovery of high added-value products from natural sources. J. Supercrit. Fluids 2018, 134, 252–259. [Google Scholar] [CrossRef]
- Gielen, D.; Boshell, F.; Saygin, D. Climate and energy challenges for materials science. Nat. Publ. Gr. 2016, 15, 117–120. [Google Scholar] [CrossRef]
- Wenger, J.; Stern, T. Reflection on the research on and implementation of biorefinery systems—A systematic literature review with a focus on feedstock. Biofuels Bioprod. Bior. 2019, 13, 1347–1364. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Conrad, M.; Sun, S.-N.; Sanchez, A.; Rocha, G.J.M.; Romaní, A.; Castro, E.; Torres, A.; Rodríguez-Jasso, R.M.; Andrade, L.P.; et al. Engineering aspects of hydrothermal pretreatment: From batch to continuous operation, scale-up and pilot reactor under biorefinery concept. Bioresour. Technol. 2020, 299, 122685. [Google Scholar] [CrossRef]
- Pachapur, V.L.; Kaur Brar, S.; Le Bihan, Y. Integrated wood biorefinery: Improvements and tailor-made two-step strategies on hydrolysis techniques. Bioresour. Technol. 2020, 299, 122632. [Google Scholar] [CrossRef]
- Pino, M.S.; Rodríguez-Jasso, R.M.; Michelin, M.; Flores-Gallegos, A.C.; Morales-Rodriguez, R.; Teixeira, J.A.; Ruiz, H.A. Bioreactor design for enzymatic hydrolysis of biomass under the biorefinery concept. Chem. Eng. J. 2018, 347, 119–136. [Google Scholar] [CrossRef]
- Avelino Gonçaves, F.; Ruiz, H.A.; Silvino dos Santos, E.; Teixeira, J.A.; Ribeiro de Macedo, G. Bioethanol production by Saccharomyces cerevisiae, Pichia stipitis and Zymomonas mobilis from delignified coconut fibre mature and lignin extraction according to biorefinery concept. Renew. Energy 2016, 94, 353–365. [Google Scholar] [CrossRef]
- Akhtar, N.; Gupta, K.; Goyal, D.; Goyal, A. Recent advances in pretreatment technologies for efficient hydrolysis of lignocellulosic biomass. Env.. Prog. Sustain. Energy 2016, 35, 498–511. [Google Scholar] [CrossRef]
- Sorokina, K.N.; Taran, O.P.; Medvedeva, T.B.; Samoylova, Y.V.; Piligaev, A.V.; Parmon, V.N. Cellulose biorefinery based on a combined catalytic and biotechnological approach for rroduction of 5-HMF and ethanol. ChemSusChem 2017, 10, 562–574. [Google Scholar] [CrossRef] [PubMed]
- Brinchi, L.; Cotana, F.; Fortunati, E.; Kenny, J.M. Production of nanocrystalline cellulose from lignocellulosic biomass: Technology and applications. Carbohydr. Polym. 2013, 94, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Nuruddin, M.; Hosur, M.; Uddin, M.J.; Baah, D.; Jeelani, S. A novel approach for extracting cellulose nanofibers from lignocellulosic biomass by ball milling combined with chemical treatment. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Luo, Y.; Li, Z.; Li, X.; Liu, X.; Fan, J.; Clark, J.H. The production of furfural directly from hemicellulose in lignocellulosic biomass: A review. Catal. Today 2019, 319, 14–24. [Google Scholar] [CrossRef]
- Clauser, N.M.; Gutiérrez, S.; Area, M.C.; Felissia, F.E.; Vallejos, M.E. Techno-economic assessment pellet production in a pine sawdust. BiofuelsBioprod. Biorefining 2018, 12, 997–1012. [Google Scholar] [CrossRef]
- Safari, A.; Karimi, K.; Shafiei, M. Dilute alkali pretreatment of softwood pine: A biorefinery approach. Bioresour. Technol. 2017, 234, 67–76. [Google Scholar] [CrossRef]
- Charisteidis, I.; Lazaridis, P.; Fotopoulos, A.; Pachatouridou, E.; Matsakas, L.; Rova, U.; Christakopoulos, P.; Triantafyllidis, K. Catalytic fast pyrolysis of lignin isolated by hybrid organosolv — Steam explosion pretreatment of hardwood and softwood biomass for the production of phenolics and aromatics. Catalyst 2019, 9, 935. [Google Scholar] [CrossRef]
- Ragauskas, A.J.; Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science. 2014, 344, 1246843. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.-Q.; Xu, F.; Sun, R.-C. Role of lignin in a biorefinery: Separation characterization and valorization. J. Chem. Technol. Biotechnol. 2013, 88, 346–352. [Google Scholar] [CrossRef]
- Paone, E.; Tabanelli, T.; Mauriello, F. The rise of lignin biorefinery. Curr. Opin. Green Sustain. Chem. 2019, 24, 1–6. [Google Scholar] [CrossRef]
- Chen, J.; Yuan, Z.; Zanuso, E.; Trajano, H.L. Response of biomass species to hydrothermal pretreatment. In Hydrothermal Processing in Biorefineries: Production of Bioethanol and High Added-Value Compounds of Second and Third Generation Biomass; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar]
- Forests, Forestry and Logging - Statistics Explained. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/Forests,_forestry_and_logging (accessed on 21 January 2020).
- Cambero, C.; Sowlati, T.; Pavel, M. Economic and life cycle environmental optimization of forest-based biorefinery supply chains for bioenergy and biofuel production. Chem. Eng. Res. Des. 2016, 107, 218–235. [Google Scholar] [CrossRef]
- Vega, L.Y.; López, L.; Valdés, C.F.; Chejne, F. Assessment of energy potential of wood industry wastes through thermochemical conversions. Waste Manag. 2019, 87, 108–118. [Google Scholar] [CrossRef]
- Belt, T.; Keplinger, T.; Hänninen, T.; Rautkari, L. Cellular level distributions of Scots pine heartwood and knot heartwood extractives revealed by Raman spectroscopy imaging. Ind. Crop. Prod. 2017, 108, 327–335. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Species - EUFORGEN European Forest Genetic Resources Programme. Available online: http://www.euforgen.org/species/ (accessed on 27 March 2020).
- IFN6 — ICNF. Available online: http://www2.icnf.pt/portal/florestas/ifn/ifn6 (accessed on 27 March 2020).
- Tümen, İ.; Akkol, E.K.; Taştan, H.; Süntar, I.; Kurtca, M. Research on the antioxidant, wound healing, and anti-inflammatory activities and the phytochemical composition of maritime pine (Pinus pinaster Ait). J. Ethnopharmacol. 2018, 211, 235–246. [Google Scholar] [CrossRef]
- del Río, P.G.; Gomes-Dias, J.S.; Rocha, C.M.R.; Romaní, A.; Garrote, G.; Domingues, L. Recent trends on seaweed fractionation for liquid biofuels production. Bioresour. Technol. 2019, 299, 122613. [Google Scholar] [CrossRef]
- Räisänen, T.; Athanassiadis, D. Basic chemical composition of the biomass components of pine, spruce and birch. Sci. Res. 2013. [Google Scholar]
- Shemfe, M.B.; Gu, S.; Ranganathan, P. Techno-economic performance analysis of biofuel production and miniature electric power generation from biomass fast pyrolysis and bio-oil upgrading. Fuel 2015, 143, 361–372. [Google Scholar] [CrossRef]
- Akgül, M.; Çöpür, Y.; Temiz, S. A comparison of kraft and kraft-sodium borohydrate brutia pine pulps. Build. Env. 2007, 42, 2586–2590. [Google Scholar] [CrossRef]
- Chaula, Z.; Said, M.; John, G.; Manyele, S.; Mhilu, C. Modelling the Suitability of Pine Sawdust for Energy Production via Biomass Steam Explosion. Smart Grid Renew. Energy 2014, 5, 1–7. [Google Scholar] [CrossRef]
- Gulsoy, S.K.; Ozturk, F. Kraft pulping properties of european black pine cone. Maderas Cienc. Y Tecnol. 2016, 17, 875–882. [Google Scholar] [CrossRef]
- Cotana, F.; Cavalaglio, G.; Gelosia, M.; Nicolini, A.; Coccia, V.; Petrozzi, A. Production of bioethanol in a second generation prototype from pine wood chips. Energy Procedia. 2014, 45, 42–51. [Google Scholar] [CrossRef]
- Miranda, I.; Mirra, I.; Gominho, J.; Pereira, H. Fractioning of bark of Pinus pinea by milling and chemical characterization of the different fractions. Maderas. Cienc. Y Tecnol. 2017, 19, 185–194. [Google Scholar] [CrossRef]
- Fradinho, D.M.; Neto, C.P.; Evtuguin, D.; Jorge, F.C.; Irle, M.A.; Gil, M.H.; Pedrosa de Jesus, J. Chemical characterisation of bark and of alkaline bark extracts from maritime pine grown in Portugal. Ind. Crop. Prod. 2002, 16, 23–32. [Google Scholar] [CrossRef]
- Ferreira-Santos, P.; Genisheva, Z.; Botelho, C.; Santos, J.; Ramos, C.; Teixeira, J.A.; Rocha, C.M.R. Unravelling the biological potential of pinus pinaster bark extracts. Antioxidants 2020, 9, 334. [Google Scholar] [CrossRef]
- Miranda, I.; Gominho, J.; Mirra, I.; Pereira, H. Chemical characterization of barks from Picea abies and Pinus sylvestris after fractioning into different particle sizes. Ind. Crop. Prod. 2012, 36, 395–400. [Google Scholar] [CrossRef]
- Vieito, C.; Pires, P.; Fernandes, É.; Velho, M. Chemical characterization of pine bark (Pinus pinaster Aiton subsp. atlantica), antioxidant properties and phenolic profile of its extracts. Millenium J. Educ. Technol. Heal. 2019, 8, 79–87. [Google Scholar] [CrossRef]
- Khan, I.U.; Shah, A.A.; Sahibzada, F.A.; Hayyat, A.; Nazar, M.; Mobashar, M.; Tariq, A.; Sultana, N. Carcass characteristics and serum biochemical profile of Japanese quail by the supplementation of pine needles and vitamin E powder. Biologia (Bratisl). 2019, 74, 993–1000. [Google Scholar] [CrossRef]
- Ramay, M.S.; Yalçın, S. Effects of supplemental pine needles powder (Pinus brutia) on growth performance, breast meat composition, and antioxidant status in broilers fed linseed oil-based diets. Poult. Sci. 2019, 2019.99, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Dönmez, I.E.; Hafizoǧlu, H.; Kilic, A.; Tümen, I.; Sivrikaya, H. Chemical composition of fourteen different coniferous species cones growing naturally in Turkey. Wood Res. 2012, 57, 339–344. [Google Scholar]
- López-Mata, L. Proteins, amino acids and fatty acids composition of nuts from the mexican endemic rarity, Pinus maximartinezii, and its conservation implications. Interciencia 2001, 26, 606–610. [Google Scholar]
- Cheikh-Rouhou, S.; Hentati, B.; Besbes, S.; Blecker, C.; Deroanne, C.; Attia, H. Chemical composition and lipid fraction characteristics of Aleppo pine (pinus halepensis Mill.) seeds cultivated in Tunisia. Food Sci. Technol. Int. 2006, 12, 407–416. [Google Scholar] [CrossRef]
- Tukan, S.K.; Al-Ismail, K.; Ajo, R.Y.; Al-Dabbas, M.M. Seeds and seed oil compositions of Aleppo pine (Pinus halepensis Mill.) grown in Jordan. Riv. Ital. Delle Sostanze Grasse 2013, 90, 87–93. [Google Scholar]
- Rodrigues-Corrêa, K.C.d.S.; de Lima, J.C.; Fett-Neto, A.G. Pine oleoresin: Tapping green chemicals, biofuels, food protection, and carbon sequestration from multipurpose trees. Food Energy Secur. 2012, 1, 81–93. [Google Scholar]
- Wiyono, B.; Tachibana, S.; Tinambunan, D. Chemical compositions of pine resin, rosin and turpentine oil from west java. Indones. J. Res. 2006, 3, 7–17. [Google Scholar] [CrossRef]
- Wang, Z.; Lei, T.; Yan, X.; Chen, G.; Xin, X.; Yang, M.; Guan, Q.; He, X.; Gupta, A.K. Common characteristics of feedstock stage in life cycle assessments of agricultural residue-based biofuels. Fuel 2019, 253, 1256–1263. [Google Scholar] [CrossRef]
- Mitchell, G.R.; Biscaia, S.; Mahendra, V.S.; Mateus, A. High value materials from the forests. Adv. Mater. Phys. Chem. 2016, 6, 54–60. [Google Scholar] [CrossRef]
- Ehn, M.; Thornton, J.A.; Kleist, E.; Sipilä, M.; Junninen, H.; Pullinen, I.; Springer, M.; Rubach, F.; Tillmann, R.; Lee, B.; et al. A large source of low-volatility secondary organic aerosol. Nature 2014, 506, 476–479. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Santos, P.; Genisheva, Z.; Pereira, R.N.; Teixeira, J.A.; Rocha, C.M.R. Moderate electric fields as a potential tool for sustainable recovery of phenolic compounds from pinus pinaster bark. Acs Sustain. Chem. Eng. 2019, 7, 8816–8826. [Google Scholar] [CrossRef]
- Silva, B.; Martins, M.; Rosca, M.; Rocha, V.; Lago, A.; Neves, I.C.; Tavares, T. Waste-based biosorbents as cost-effective alternatives to commercial adsorbents for the retention of fluoxetine from water. Sep. Purif. Technol. 2020, 235. [Google Scholar] [CrossRef]
- Kim, K.H.; Daugaard, T.J.; Smith, R.; Mba-Wright, M.; Brown, R.C. Recovery of resin acids from fast pyrolysis of pine. J. Anal. Appl. Pyrolysis 2019, 138, 132–136. [Google Scholar] [CrossRef]
- Shi, J.; Nawaz, H.; Pohorly, J.; Mittal, G.; Kakuda, Y.; Jiang, Y. Extraction of polyphenolics from plant material for functional foods - Engineering and technology. Food Rev. Int. 2005, 21, 139–166. [Google Scholar] [CrossRef]
- Wen, L.; Zhang, Z.; Sun, D.W.; Sivagnanam, S.P.; Tiwari, B.K. Combination of emerging technologies for the extraction of bioactive compounds. Crit. Rev. Food. Sci. Nutr. 2020, 60, 1826–1841. [Google Scholar] [CrossRef]
- Chemat, F.; Fabiano-tixier, A.S.; Abert, M.; Allaf, T.; Vorobiev, E. Trends in analytical chemistry solvent-free extraction of food and natural products. Trends Anal. Chem. 2015, 71, 157–168. [Google Scholar] [CrossRef]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of natural plant origins: From sources to food industry applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef]
- Maqsood, S.; Adiamo, O.; Ahmad, M.; Mudgil, P. Bioactive compounds from date fruit and seed as potential nutraceutical and functional food ingredients. Food Chem. 2019, 308, 125522. [Google Scholar] [CrossRef]
- Fidelis, M.; De Moura, C.; Kabbas, T.; Pap, N.; Mattila, P.; Mäkinen, S.; Putnik, P.; Kovačević, D.B.; Tian, Y.; Yang, B.; et al. Fruit seeds as sources of bioactive compounds: Sustainable production of high value-added ingredients from by-products within circular economy. Molecules 2019, 24, 3854. [Google Scholar] [CrossRef]
- Tanase, C.; Cosarcă, S.; Muntean, D.L. A critical review of phenolic compounds extracted from the bark of woody vascular plants and their potential biological activity. Molecules 2019, 24, 1182. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Goyal, R.; Sharma, L. Potential biological efficacy of Pinus plant species against oxidative, inflammatory and microbial disorders. Bmc Complement. Altern. Med. 2016, 16, 1–11. [Google Scholar] [CrossRef]
- Ku, C.S.; Jang, J.P.; Mun, S.P. Exploitation of polyphenol-rich pine barks for potent antioxidant activity. J. Wood Sci. 2007, 53, 524–528. [Google Scholar] [CrossRef]
- Yesil-Celiktas, O.; Otto, F.; Parlar, H. A comparative study of flavonoid contents and antioxidant activities of supercritical CO2 extracted pine barks grown in different regions of Turkey and Germany. Eur. Food Res. Technol. 2009, 229, 671–677. [Google Scholar] [CrossRef]
- Mellouk, H.; Meullemiestre, A.; Maache-Rezzoug, Z.; Bejjani, B.; Dani, A.; Rezzoug, S.A. Valorization of industrial wastes from French maritime pine bark by solvent free microwave extraction of volatiles. J. Clean. Prod. 2016, 112, 4398–4405. [Google Scholar] [CrossRef]
- Iravani, S.; Zolfaghari, B. Pharmaceutical and nutraceutical effects of Pinus pinaster bark extract. Res. Pharm. Sci. 2011, 6, 1–11. [Google Scholar] [PubMed]
- Mármol, I.; Quero, J.; Jiménez-Moreno, N.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. A systematic review of the potential uses of pine bark in food industry and health care. Trends Food Sci. Technol. 2019, 88, 558–566. [Google Scholar] [CrossRef]
- Maimoona, A.; Naeem, I.; Saddiqe, Z.; Jameel, K. A review on biological, nutraceutical and clinical aspects of French maritime pine bark extract. J. Ethnopharmacol. 2011, 133, 261–277. [Google Scholar] [CrossRef]
- Meullemiestre, A.; Petitcolas, E.; Maache-Rezzoug, Z.; Chemat, F.; Rezzoug, S.A. Impact of ultrasound on solid-liquid extraction of phenolic compounds from maritime pine sawdust waste. Kinetics, optimization and large scale experiments. Ultrason. Sonochem. 2016, 28, 230–239. [Google Scholar] [CrossRef]
- Egenberg, I.M.; Aasen, J.A.B.; Holtekjølen, A.K.; Lundanes, E. Characterisation of traditionally kiln produced pine tar by gas chromatography-mass spectrometry. J. Anal. Appl. Pyrolysis 2002, 62, 143–155. [Google Scholar] [CrossRef]
- Stacey, R.J.; Dunne, J.; Brunning, S.; Devièse, T.; Mortimer, R.; Ladd, S.; Parfitt, K.; Evershed, R.; Bull, I. Birch bark tar in early Medieval England – Continuity of tradition or technological revival? J. Archaeol. Sci. Rep. 2020, 29, 102118. [Google Scholar] [CrossRef] [PubMed]
- Hoon, Y.L.; Choo, C.; Watawana, M.I.; Jayawardena, N.; Waisundara, V.Y. Short communications Evaluation of the total antioxidant capacity and antioxidant compounds of different solvent extracts of Chilgoza pine nuts ( Pinus gerardiana ). J. Funct. Foods 2015, 18, 1014–1021. [Google Scholar] [CrossRef]
- Lin, S.; Liang, R.; Xue, P.; Zhang, S.; Liu, Z.; Dong, X. Antioxidant activity improvement of identified pine nut peptides by pulsed electric field (PEF) and the mechanism exploration. LWT Food Sci. Technol. 2017, 75, 366–372. [Google Scholar] [CrossRef]
- Hou, L.; Li, C.; Qiu, J. Comparison of the physicochemical characteristics of Pinus koraiensis L. nut oils from different extraction technologies. Grain Oil Sci. Technol. 2018, 1, 113–118. [Google Scholar]
- Silva, S.; Costa, E.M.; Calhau, C.; Morais, R.M.; Pintado, M.E. Anthocyanin extraction from plant tissues: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 3072–3083. [Google Scholar] [CrossRef]
- da Silva, R.P.F.F.; Rocha-Santos, T.A.P.; Duarte, A.C. Supercritical fluid extraction of bioactive compounds. Trac - Trends Anal. Chem. 2016, 76, 40–51. [Google Scholar] [CrossRef]
- Marcheafave, G.G.; Tormena, C.D.; Pauli, E.D.; Rakocevic, M.; Bruns, R.E.; Scarminio, I.S. Experimental mixture design solvent effects on pigment extraction and antioxidant activity from Coffea arabica L. leaves. Microchem. J. 2019, 146, 713–721. [Google Scholar] [CrossRef]
- Venkatesan, T.; Choi, Y.W.; Kim, Y.K. Impact of different extraction solvents on phenolic content and antioxidant potential of pinus densiflora bark extract. Biomed Res. Int. 2019, 2019, 1–14. [Google Scholar] [CrossRef]
- Mokrani, A.; Madani, K. Effect of solvent, time and temperature on the extraction of phenolic compounds and antioxidant capacity of peach (Prunus persica L.) fruit. Sep. Purif. Technol. 2016, 162, 68–76. [Google Scholar] [CrossRef]
- Fierascu, R.C.; Fierascu, I.; Avramescu, S.M.; Sieniawska, E. Recovery of Natural Antioxidants from Agro-Industrial Side Streams through Advanced Extraction Techniques. Molecules 2019, 24, 4212. [Google Scholar] [CrossRef]
- Hadzri, H.M.; Yunus, M.A.C.; Zhari, S.; Rithwan, F. The effects of solvents and extraction methods on the antioxidant activity of P. niruri. J. Teknol. Sci. Eng. 2014, 68, 47–52. [Google Scholar] [CrossRef][Green Version]
- Prat, D.; Wells, A.; Hayler, J.; Sneddon, H.; McElroy, C.R.; Abou-Shehada, S.; Dunn, P.J. CHEM21 selection guide of classical- and less classical-solvents. Green Chem. 2015, 18, 288–296. [Google Scholar] [CrossRef]
- Domínguez, E.; Romaní, A.; Domingues, L.; Garrote, G. Evaluation of strategies for second generation bioethanol production from fast growing biomass Paulownia within a biorefinery scheme. Appl. Energy 2017, 187, 777–789. [Google Scholar] [CrossRef]
- Liu, Y.; Friesen, J.B.; McAlpine, J.B.; Lankin, D.C.; Chen, S.N.; Pauli, G.F. Natural deep eutectic solvents: Properties, applications, and perspectives. J. Nat. Prod. 2018, 81, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.A.R.; Pinho, S.P.; Coutinho, J.A.P. Insights into the nature of eutectic and deep eutectic mixtures. J. Solut. Chem. 2019, 48, 962–982. [Google Scholar] [CrossRef]
- Ventura, S.P.M.E.; Silva, F.A.; Quental, M.V.; Mondal, D.; Freire, M.G.; Coutinho, J.A.P. Ionic-liquid-mediated extraction and separation processes for bioactive compounds: Past, present, and future trends. Chem. Rev. 2017, 117, 6984–7052. [Google Scholar] [CrossRef]
- Martins, P.L.G.; Braga, A.R.; de Rosso, V.V. Can ionic liquid solvents be applied in the food industry? Trends Food Sci. Technol. 2017, 66, 117–124. [Google Scholar] [CrossRef]
- Murador, D.C.; de Souza Mesquita, L.M.; Vannuchi, N.; Braga, A.R.C.; de Rosso, V.V. Bioavailability and biological effects of bioactive compounds extracted with natural deep eutectic solvents and ionic liquids: Advantages over conventional organic solvents. Curr. Opin. Food Sci. 2019, 26, 25–34. [Google Scholar] [CrossRef]
- Passos, H.; Freire, M.G.; Coutinho, J.A.P. Ionic liquid solutions as extractive solvents for value-added compounds from biomass. Green Chem. 2014, 16, 4786–4815. [Google Scholar] [CrossRef]
- da Costa Lopes, A.M.; Brenner, M.; Falé, P.; Roseiro, L.B.; Bogel-Łukasik, R. Extraction and purification of phenolic compounds from lignocellulosic biomass assisted by ionic liquid, polymeric resins, and supercritical CO2. Acs Sustain. Chem. Eng. 2016, 4, 3357–3367. [Google Scholar] [CrossRef]
- Ivanović, M.; Alañón, M.E.; Arráez-Román, D.; Segura-Carretero, A. Enhanced and green extraction of bioactive compounds from Lippia citriodora by tailor-made natural deep eutectic solvents. Food Res. Int. 2018, 111, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Hou, K.; Bao, M.; Wang, L.; Zhang, H.; Yang, L.; Zhao, H.; Wang, Z. Aqueous enzymatic pretreatment ionic liquid–lithium salt based microwave–assisted extraction of essential oil and procyanidins from pinecones of Pinus koraiensis. J. Clean. Prod. 2019, 236, 117581. [Google Scholar] [CrossRef]
- Rocha, C.M.R.; Genisheva, Z.; Ferreira-Santos, P.; Rodrigues, R.; Vicente, A.A.; Teixeira, J.A.; Pereira, R.N. Electric field-based technologies for valorization of bioresources. Bioresour. Technol. 2018, 254, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Heleno, S.A.; Diz, P.; Prieto, M.A.; Barros, L.; Rodrigues, A.; Barreiro, M.F.; Ferreira, I.C.F.R. Optimization of ultrasound-assisted extraction to obtain mycosterols from Agaricus bisporus L. by response surface methodology and comparison with conventional Soxhlet extraction. Food Chem. 2016, 197, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Alara, O.R.; Abdurahman, N.H.; Ukaegbu, C.I.; Azhari, N.H. Vernonia cinerea leaves as the source of phenolic compounds, antioxidants, and anti-diabetic activity using microwave-assisted extraction technique. Ind. Crop. Prod. 2018, 122, 533–544. [Google Scholar] [CrossRef]
- Goula, A.M.; Ververi, M.; Adamopoulou, A.; Kaderides, K. Green ultrasound-assisted extraction of carotenoids from pomegranate wastes using vegetable oils. Ultrason. Sonochem. 2017, 34, 821–830. [Google Scholar] [CrossRef]
- Okolie, C.L.; Akanbi, T.O.; Mason, B.; Udenigwe, C.C.; Aryee, A.N.A. Influence of conventional and recent extraction technologies on physicochemical properties of bioactive macromolecules from natural sources: A review. Food Res. Int. 2019, 116, 827–839. [Google Scholar] [CrossRef]
- Bouras, M.; Chadni, M.; Barba, F.J.; Grimi, N.; Bals, O.; Vorobiev, E. Optimization of microwave-assisted extraction of polyphenols from Quercus bark. Ind. Crop. Prod. 2015, 77, 590–601. [Google Scholar] [CrossRef]
- Barba, F.J.; Zhu, Z.; Koubaa, M.; Sant’Ana, A.S.; Orlien, V. Green alternative methods for the extraction of antioxidant bioactive compounds from winery wastes and by-products: A review. Trends Food Sci. Technol. 2016, 49, 96–109. [Google Scholar] [CrossRef]
- Vinatoru, M.; Mason, T.J.; Calinescu, I. Ultrasonically assisted extraction (UAE) and microwave assisted extraction (MAE) of functional compounds from plant materials. Trac Trends Anal. Chem. 2017, 97, 159–178. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, Y.; Padilla-Zakour, O.; Yang, G. Polyphenols, antioxidant and antimicrobial activities of leaf and bark extracts of Solidago canadensis L. Ind. Crop. Prod. 2015, 74, 803–809. [Google Scholar] [CrossRef]
- Ghitescu, R.-E.; Volf, I.; Carausu, C.; Bühlmann, A.-M.; Gilca, A.; Popa, V.I. Optimization of ultrasound-assisted extraction of polyphenols from spruce wood bark. Ultrason. Sonochem. 2015, 22, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Contreras, M.d.M.; Lama-Muñoz, A.; Espínola, F.; Moya, M.; Romero, I.; Castro, E. Valorization of olive mill leaves through ultrasound-assisted extraction. Food Chem. 2020, 314, 126218. [Google Scholar] [CrossRef]
- Liazid, A.; Schwarz, M.; Varela, R.M.; Palma, M.; Guillén, D.A.; Brigui, J.; Macías, F.A.; Barroso, C.G. Evaluation of various extraction techniques for obtaining bioactive extracts from pine seeds. Food Bioprod. Process. 2010, 88, 247–252. [Google Scholar] [CrossRef]
- Aspé, E.; Fernández, K. The effect of different extraction techniques on extraction yield, total phenolic, and anti-radical capacity of extracts from Pinus radiata Bark. Ind. Crop. Prod. 2011, 34, 838–844. [Google Scholar] [CrossRef]
- Karapandzova, M.; Stefkov, G.; Cvetkovikj, I.; Stanoeva, J.P.; Stefova, M.; Kulevanova, S. Flavonoids and other phenolic compounds in needles of pinus peuce and other pine species from the macedonian flora. Nat. Prod. Commun. 2015, 10, 987–990. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ding, J.; Ren, N. Recent advances in microwave-assisted extraction of trace organic pollutants from food and environmental samples. Trac Trends Anal. Chem. 2016, 75, 197–208. [Google Scholar] [CrossRef]
- Chupin, L.; Maunu, S.L.; Reynaud, S.; Pizzi, A.; Charrier, B.; Charrier-EL Bouhtoury, F. Microwave assisted extraction of maritime pine (Pinus pinaster) bark: Impact of particle size and characterization. Ind. Crop. Prod. 2015, 65, 142–149. [Google Scholar] [CrossRef]
- Wrona, O.; Rafińska, K.; Mozenski, C.; Byszewski, B. Supercritical fluid extraction of bioactive compounds from plant materials. J. Aoac Int. 2017, 100, 1624–1635. [Google Scholar] [CrossRef]
- Ferrentino, G.; Morozova, K.; Mosibo, O.K.; Ramezani, M.; Scampicchio, M. Biorecovery of antioxidants from apple pomace by supercritical fluid extraction. J. Clean. Prod. 2018, 186, 253–261. [Google Scholar] [CrossRef]
- Braga, M.E.M.; Santos, R.M.S.; Seabra, I.J.; Facanali, R.; Marques, M.O.M.; de Sousa, H.C. Fractioned SFE of antioxidants from maritime pine bark. J. Supercrit. Fluids 2008, 47, 37–48. [Google Scholar] [CrossRef]
- Seabra, I.J.; Dias, A.M.A.; Braga, M.E.M.; de Sousa, H.C. High pressure solvent extraction of maritime pine bark: Study of fractionation, solvent flow rate and solvent composition. J. Supercrit. Fluids 2012, 62, 135–148. [Google Scholar] [CrossRef]
- Ghoreishi, S.M.; Hedayati, A.; Mohammadi, S. Optimization of periodic static-dynamic supercritical CO2 extraction of taxifolin from pinus nigra bark with ethanol as entrainer. J. Supercrit. Fluids 2016, 113, 53–60. [Google Scholar] [CrossRef]
- Soquetta, M.B.; Terra, L.d.M.; Bastos, C.P. Green technologies for the extraction of bioactive compounds in fruits and vegetables. Cyta - J. Food 2018, 16, 400–412. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Chang, C.M.J.; Deng, T.S. Enzymatic hot pressurized fluids extraction of polyphenolics from Pinus taiwanensis and Pinus morrisonicola. J. Taiwan Inst. Chem. Eng. 2009, 40, 136–142. [Google Scholar] [CrossRef]
- Jaeger, H.; Roth, A.; Toepfl, S.; Holzhauser, T.; Engel, K.H.; Knorr, D.; Vogel, R.F.; Bandick, N.; Kulling, S.; Heinz, V.; et al. Opinion on the use of ohmic heating for the treatment of foods. Trends Food Sci. Technol. 2016, 55, 84–97. [Google Scholar] [CrossRef]
- Pereira, R.N.; Vicente, A.A. Environmental impact of novel thermal and non-thermal technologies in food processing. Food Res. Int. 2010, 43, 1936–1943. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Meullemiestre, A.; Turk, M.; Périno, S.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Review of green food processing techniques. Preservation, transformation, and extraction. Innov. Food Sci. Emerg. Technol. 2017, 41, 357–377. [Google Scholar] [CrossRef]
- Sastry, S. Ohmic heating and moderate electric field processing. Food Sci. Technol. Int. 2008, 14, 419–422. [Google Scholar] [CrossRef]
- Lebovka, N.I.; Praporscic, I.; Ghnimi, S.; Vorobiev, E. Does electroporation occur during the ohmic heating of food? J. Food Sci. 2005, 70, E308–E311. [Google Scholar] [CrossRef]
- Pereira, R.N.; Rodrigues, R.M.; Genisheva, Z.; Oliveira, H.; de Freitas, V.; Teixeira, J.A.; Vicente, A.A. Effects of ohmic heating on extraction of food-grade phytochemicals from colored potato. LWT Food Sci. Technol. 2016, 74, 493–503. [Google Scholar] [CrossRef]
- Puértolas, E.; Barba, F.J. Electrotechnologies applied to valorization of by-products from food industry: Main findings, energy and economic cost of their industrialization. Food Bioprod. Process. 2016, 100, 172–184. [Google Scholar] [CrossRef]
- Bouras, M.; Grimi, N.; Bals, O.; Vorobiev, E. Impact of pulsed electric fields on polyphenols extraction from Norway spruce bark. Ind. Crop. Prod. 2016, 80, 50–58. [Google Scholar] [CrossRef]
- Andreou, V.; Dimopoulos, G.; Dermesonlouoglou, E.; Taoukis, P. Application of pulsed electric fields to improve product yield and waste valorization in industrial tomato processing. J. Food Eng. 2020, 270, 109778. [Google Scholar] [CrossRef]
- Frontuto, D.; Carullo, D.; Harrison, S.M.; Brunton, N.P.; Ferrari, G.; Lyng, J.G.; Pataro, G. Optimization of pulsed electric fields-assisted extraction of polyphenols from potato peels using response surface methodology. Food Bioprocess Technol. 2019, 12, 1708–1720. [Google Scholar] [CrossRef]
- Conde, E.; Hemming, J.; Smeds, A.; Reinoso, B.D.; Moure, A.; Willför, S.; Domínguez, H.; Parajó, J.C. Extraction of low-molar-mass phenolics and lipophilic compounds from Pinus pinaster wood with compressed CO2. J. Supercrit. Fluids 2013, 81, 193–199. [Google Scholar] [CrossRef]
- Xu, C.C.; Wang, B.; Pu, Y.Q.; Tao, J.S.; Zhang, T. Advances in extraction and analysis of phenolic compounds from plant materials. Chin. J. Nat. Med. 2017, 15, 721–731. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Kalisz, S.; Oszmiański, J.; Kolniak-Ostek, J.; Grobelna, A.; Kieliszek, M.; Cendrowski, A. Effect of a variety of polyphenols compounds and antioxidant properties of rhubarb (Rheum rhabarbarum). Lwt 2020, 118, 108775. [Google Scholar] [CrossRef]
- Yan, Z.; Zhong, Y.; Duan, Y.; Chen, Q.; Li, F. Antioxidant mechanism of tea polyphenols and its impact on health benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Reis Giada, M.d.L. Food phenolic compounds: main classes, sources and their antioxidant power. In Oxidative Stress and Chronic Degenerative Diseases - A Role for Antioxidants; InTechOpen: London, UK, 2013. [Google Scholar]
- Minatel, I.O.; Borges, C.V.; Ferreira, M.I.; Gomez, H.A.G.; Chen, C.-Y.O.; Lima, G.P.P. Phenolic compounds: Functional properties, impact of processing and bioavailability. In Phenolic Compounds - Biological Activity; InTechOpen: London, UK, 2017. [Google Scholar]
- Spínola, V.; Llorent-Martínez, E.J.; Castilho, P.C. Inhibition of α-amylase, α-glucosidase and pancreatic lipase by phenolic compounds of Rumex maderensis (Madeira sorrel). Influence of simulated gastrointestinal digestion on hyperglycaemia-related damage linked with aldose reductase activity and protein glacation. LWT 2020, 118, 108727. [Google Scholar]
- Castro, M.C.; Villagarcía, H.; Nazar, A.; Arbeláez, L.G.; Massa, M.L.; Del Zotto, H.; Ríos, J.L.; Schinella, G.R.; Francini, F. Cacao extract enriched in polyphenols prevents endocrine-metabolic disturbances in a rat model of prediabetes triggered by a sucrose rich diet. J. Ethnopharmacol. 2020, 247, 112263. [Google Scholar] [CrossRef] [PubMed]
- Dzah, C.S.; Duan, Y.; Zhang, H.; Wen, C.; Zhang, J.; Chen, G.; Ma, H. The effects of ultrasound assisted extraction on yield, antioxidant, anticancer and antimicrobial activity of polyphenol extracts: A review. Food Biosci. 2020, 35, 100547. [Google Scholar] [CrossRef]
- Jesus, M.S.; Ballesteros, L.F.; Pereira, R.N.; Genisheva, Z.; Carvalho, A.C.; Pereira-Wilson, C.; Teixeira, J.A.; Domingues, L. Ohmic heating polyphenolic extracts from vine pruning residue with enhanced biological activity. Food Chem. 2020, 316, 126298. [Google Scholar] [CrossRef]
- Zhang, H.; Qi, R.; Mine, Y. The impact of oolong and black tea polyphenols on human health. Food Biosci. 2019, 29, 55–61. [Google Scholar] [CrossRef]
- Cabral, F.L.; Bernardes, V.M.; Passos, D.F.; de Oliveira, J.S.; Doleski, P.H.; Silveira, K.L.; Horvarth, M.C.; Bremm, J.M.; Barbisan, F.; Azzolin, V.F.; et al. Astrocaryum aculeatum fruit improves inflammation and redox balance in phytohemagglutinin-stimulated macrophages. J. Ethnopharmacol. 2020, 247, 112274. [Google Scholar] [CrossRef]
- Fei, P.; Ali, M.A.; Gong, S.; Sun, Q.; Bi, X.; Liu, S.; Guo, L. Antimicrobial activity and mechanism of action of olive oil polyphenols extract against Cronobacter sakazakii. Food Control 2018, 94, 289–294. [Google Scholar] [CrossRef]
- Gascón, S.; Jiménez-Moreno, N.; Jiménez, S.; Quero, J.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. Nutraceutical composition of three pine bark extracts and their antiproliferative effect on Caco-2 cells. J. Funct. Foods 2018, 48, 420–429. [Google Scholar] [CrossRef]
- Ustun, O.; Senol, F.S.; Kurkcuoglu, M.; Orhan, I.E.; Kartal, M.; Baser, K.H.C. Investigation on chemical composition, anticholinesterase and antioxidant activities of extracts and essential oils of Turkish Pinus species and pycnogenol. Ind. Crop. Prod. 2012, 38, 115–123. [Google Scholar] [CrossRef]
- Ince, I.; Yesil-Celiktas, O.; Karabay-Yavasoglu, N.U.; Elgin, G. Effects of Pinus brutia bark extract and Pycnogenol® in a rat model of carrageenan induced inflammation. Phytomedicine 2009, 16, 1101–1104. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, P.; Julkunen-Tiitto, R.; Junninen, K.; Kouki, J. Phenolic compounds in scots pine heartwood: Are kelo trees a unique woody substrate? Can. J. Res. 2015, 46, 225–233. [Google Scholar] [CrossRef]
- Yen, G.C.; Der Duh, P.; Huang, D.W.; Hsu, C.L.; Fu, T.Y.C. Protective effect of pine (Pinus morrisonicola Hay.) needle on LDL oxidation and its anti-inflammatory action by modulation of iNOS and COX-2 expression in LPS-stimulated RAW 264.7 macrophages. Food Chem. Toxicol. 2008, 46, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Pauli, E.D.; Malta, G.B.; Sanchez, P.M.; Moreira, I.C.; Scarminio, I.S. Mixture design analysis of solvent extractor effects on epicatechin, epigallocatechin gallate, epigallocatechin and antioxidant activities of the Camellia sinensis L. leaves. Anal. Chem. Res. 2014, 2, 23–29. [Google Scholar] [CrossRef]
- Latha, R.C.R.; Daisy, P. Insulin-secretagogue, antihyperlipidemic and other protective effects of gallic acid isolated from Terminalia bellerica Roxb. in streptozotocin-induced diabetic rats. Chem. Biol. Interact. 2011, 189, 112–118. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.; Yousef, A.I.; Abd El-Twab, S.M.; Abdel Reheim, E.S.; Ashour, M.B. Gallic acid and p-coumaric acid attenuate type 2 diabetes-induced neurodegeneration in rats. Metab. Brain Dis. 2017, 32, 1279–1286. [Google Scholar] [CrossRef]
- Taofiq, O.; Heleno, S.A.; Calhelha, R.C.; Fernandes, I.P.; Alves, M.J.; Barros, L.; González-Paramás, A.M.; Ferreira, I.C.F.R.; Barreiro, M.F. Phenolic acids, cinnamic acid, and ergosterol as cosmeceutical ingredients: Stabilization by microencapsulation to ensure sustained bioactivity. Microchem. J. 2019, 147, 469–477. [Google Scholar] [CrossRef]
- Lantto, T.A.; Dorman, H.J.D.; Shikov, A.N.; Pozharitskaya, O.N.; Makarov, V.G.; Tikhonov, V.P.; Hiltunen, R.; Raasmaja, A. Chemical composition, antioxidative activity and cell viability effects of a Siberian pine (Pinus sibirica Du Tour) extract. Food Chem. 2009, 112, 936–943. [Google Scholar] [CrossRef]
- Khoshnam, S.E.; Sarkaki, A.; Rashno, M.; Farbood, Y. Memory deficits and hippocampal inflammation in cerebral hypoperfusion and reperfusion in male rats: Neuroprotective role of vanillic acid. Life Sci. 2018, 211, 126–132. [Google Scholar] [CrossRef]
- Sammeturi, M.; Shaik, A.H.; Bongu, S.B.R.; Cheemanapalli, S.; Mohammad, A.; Kodidhela, L.D. Protective effects of syringic acid, resveratrol and their combination against isoprenaline administered cardiotoxicity in wistar rats. SaudiJ. Biol. Sci. 2019, 26, 1429–1435. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y.; Chen, Y.; Zhou, B.; Shan, X.; Yang, G. Eriodictyol inhibits IL-1β-induced inflammatory response in human osteoarthritis chondrocytes. Biomed. Pharm. 2018, 107, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Chupin, L.; Motillon, C.; Charrier-El Bouhtoury, F.; Pizzi, A.; Charrier, B. Characterisation of maritime pine (Pinus pinaster) bark tannins extracted under different conditions by spectroscopic methods, FTIR and HPLC. Ind. Crop. Prod. 2013, 49, 897–903. [Google Scholar] [CrossRef]
- Hammerbacher, A.; Raguschke, B.; Wright, L.P.; Gershenzon, J. Gallocatechin biosynthesis via a flavonoid 3′,5′-hydroxylase is a defense response in Norway spruce against infection by the bark beetle-associated sap-staining fungus Endoconidiophora polonica. Phytochemistry 2018, 148, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Saija, A.; Tomaino, A.; Trombetta, D.; De Pasquale, A.; Uccella, N.; Barbuzzi, T.; Paolino, D.; Bonina, F. In vitro and in vivo evaluation of caffeic and ferulic acids as topical photoprotective agents. Int. J. Pharm. 2000, 199, 39–47. [Google Scholar] [CrossRef]
- Chung, Y.C.; Hsieh, F.C.; Lin, Y.J.; Wu, T.Y.; Lin, C.W.; Lin, C.T.; Tang, N.Y.; Jinn, T.R. Magnesium lithospermate B and rosmarinic acid, two compounds present in Salvia miltiorrhiza, have potent antiviral activity against enterovirus 71 infections. Eur. J. Pharm. 2015, 755, 127–133. [Google Scholar] [CrossRef]
- Adımcılar, V.; Kalaycıoğlu, Z.; Aydoğdu, N.; Dirmenci, T.; Kahraman, A.; Erim, F.B. Rosmarinic and carnosic acid contents and correlated antioxidant and antidiabetic activities of 14 Salvia species from Anatolia. J. Pharm. Biomed. Anal. 2019, 175, 112763. [Google Scholar] [CrossRef]
- Babich, H.; Zuckerbraun, H.L.; Weinerman, S.M. In vitro cytotoxicity of (-)-catechin gallate, a minor polyphenol in green tea. Toxicol. Lett. 2007, 171, 171–180. [Google Scholar] [CrossRef]
- Talmaciu, A.I.; Ravber, M.; Volf, I.; Knez, Ž.; Popa, V.I. Isolation of bioactive compounds from spruce bark waste using sub- and supercritical fluids. J. Supercrit. Fluids 2016, 117, 243–251. [Google Scholar] [CrossRef]
- Ban, J.Y.; Cho, S.O.; Jeon, S.Y.; Bae, K.H.; Song, K.S.; Seong, Y.H. 3,4-Dihydroxybenzoic acid from Smilacis chinae rhizome protects amyloid β protein (25-35)-induced neurotoxicity in cultured rat cortical neurons. Neurosci. Lett. 2007, 420, 184–188. [Google Scholar] [CrossRef]
- Lin, Z.; Lin, C.; Fu, C.; Lu, H.; Jin, H.; Chen, Q.; Pan, J. The protective effect of Ellagic acid (EA) in osteoarthritis: An in vitro and in vivo study. Biomed. Pharm. 2020, 125, 109845. [Google Scholar] [CrossRef]
- Silvan, S.; Manoharan, S. Apigenin prevents deregulation in the expression pattern of cell-proliferative, apoptotic, inflammatory and angiogenic markers during 7,12-dimethylbenz[a]anthracene-induced hamster buccal pouch carcinogenesis. Arch. Oral Biol. 2013, 58, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Szkudelska, K.; Okulicz, M.; Hertig, I.; Szkudelski, T. Resveratrol ameliorates inflammatory and oxidative stress in type 2 diabetic Goto-Kakizaki rats. Biomed. Pharm. 2020, 125, 110026. [Google Scholar] [CrossRef] [PubMed]
- Sajadimajd, S.; Bahramsoltani, R.; Iranpanah, A.; Kumar Patra, J.; Das, G.; Gouda, S.; Rahimi, R.; Rezaeiamiri, E.; Cao, H.; Giampieri, F.; et al. Advances on natural polyphenols as anticancer agents for skin cancer. Pharm. Res. 2020, 151, 104584. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Zhu, M.; Li, X.; Xu, Y.; Ma, X.; Shi, R.; Li, D.; Mu, C. Development of active rosmarinic acid-gelatin biodegradable films with antioxidant and long-term antibacterial activities. Food Hydrocoll. 2018, 83, 308–316. [Google Scholar] [CrossRef]
- Nguyen, D.M.C.; Seo, D.J.; Kim, K.Y.; Park, R.D.; Kim, D.H.; Han, Y.S.; Kim, T.H.; Jung, W.J. Nematicidal activity of 3,4-dihydroxybenzoic acid purified from Terminalia nigrovenulosa bark against Meloidogyne incognita. Microb. Pathog. 2013, 59–60, 52–59. [Google Scholar] [CrossRef]
- Muramatsu, D.; Uchiyama, H.; Kida, H.; Iwai, A. Cell cytotoxity and anti-glycation activity of taxifolin-rich extract from Japanese larch, Larix kaempferi. Heliyon 2019, 5, e02047. [Google Scholar] [CrossRef]
- Świeca, M.; Gawlik-Dziki, U.; Dziki, D.; Baraniak, B.; Czyz, J. The influence of protein-flavonoid interactions on protein digestibility in vitro and the antioxidant quality of breads enriched with onion skin. Food Chem. 2013, 141, 451–458. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Wang, H. Physicochemical properties, bioaccessibility and antioxidant activity of the polyphenols from pine cones of Pinus koraiensis. Int. J. Biol. Macromol. 2019, 126, 385–391. [Google Scholar] [CrossRef]
- Cesarone, M.R.; Belcaro, G.; Rohdewald, P.; Pellegrini, L.; Ledda, A.; Vinciguerra, G.; Ricci, A.; Ippolito, E.; Fano, F.; Dugall, M.; et al. Improvement of signs and symptoms of chronic venous insufficiency and microangiopathy with Pycnogenol®: A prospective, controlled study. Phytomedicine 2010, 17, 835–839. [Google Scholar] [CrossRef]
- Liu, X.; Wei, J.; Tan, F.; Zhou, S.; Würthwein, G.; Rohdewald, P. Antidiabetic effect of Pycnogenol® French maritime pine bark extract in patients with diabetes type II. Life Sci. 2004, 75, 2505–2513. [Google Scholar] [CrossRef]
- Segal, L.; Penman, M.G.; Piriou, Y. Evaluation of the systemic toxicity and mutagenicity of OLIGOPIN®, procyanidolic oligomers (OPC) extracted from French Maritime Pine Bark extract. Toxicol. Rep. 2018, 5, 531–541. [Google Scholar] [CrossRef] [PubMed]


| Latin Name | Common Name | Geographical Distribution |
|---|---|---|
| Pinus sylvestris | Scots pine | All countries of Europa and Asia |
| Pinus nigra | European black pine | Mountain areas of Europe, United States, and Asia Minor |
| Pinus brutia | Brutia pine | Eastern Coast of the Mediterranean (Turkey, Greece, Italy) |
| Pinus pinaster | Maritime pine | Western Mediterranean Sea, Central and Southern Europe, and North Africa |
| Pinus halepensis | Aleppo pine | Coastal areas of the Western Mediterranean region, Southern France and Italy, and North Africa |
| Pinus cembra | Swiss stone pine | Continental Alps and regions of the Carpathian Mountains |
| Pinus uncinata | Mountain pine | Mountains of Western Europe, Northern Europe, and Mediterranean |
| Pinus pinea | Stone pine | Mediterranean Basin, extending from Portugal to Syria |
| Pinus strobus | White pine | Eastern North America and Carpathian Mountains in Czech Republic and Southern Poland |
| Pinus mugo | Mountain pine | Mountains of Central and Eastern Europe |
| Pinus heldreichii | Bosnian pine | Southern and Western part of the Balkans, near the Mediterranean basin |
| Pinuscontorta | Lodgepole pine | Western North America, Europe, and New Zealand |
| Pinus peuce | Macedonian pine | Mountain areas of the Balkan Peninsula |
| Pinus radiata | Monterey pine | Central Coast of California, Australia, New Zealand, Mexico, Argentina, Chile, Uruguay, Kenya, Spain, and South Africa |
| Extraction Method | Pine Species | Part of Tree | Optimum Extraction Conditions | Foreseen Applications | Reference |
|---|---|---|---|---|---|
| Ultrasound-Assisted Extraction (UAE) | P. pinaster P. d’Alpes | seeds | Water; 75 °C; 20 min | Bioactive extracts for food supplements | [111] |
| P. radiata | bark | Acetone (70%, v/v); 25 °C; 6 min; 35 kHz/85 W | Nutraceutical action | [112] | |
| P. mugo P. nigra P. peuce P. sylvestris | needles | Methanol (70%, v/v); RT; 30 min | Medicinal and pharmaceutical | [113] | |
| P. pinaster | wood | Acidified water; 40 °C; 43 min; 0.67 W/cm2 | Diet supplement | [76] | |
| Microwave-Assisted Extraction (MAE) | P. pinaster P. d’Alpes | seeds | Water; 75 ºC; 20 min | Bioactive extracts for food supplements | [111] |
| P. radiata | bark | Acetone (70%, v/v); 25 °C; 1–2 min; 2450 MHz/900 W | Bioactive extracts as promising pharmaceutical and food applications | [112] | |
| P. pinaster | bark | Ethanol (80%, v/v); 3 min; 100W | Diet supplement | [115] | |
| P. pinaster | bark | 92.4 min; 803.5 W | Antioxidant essential oils | [72] | |
| P. koraiensis | cones | Enzymatic pretreatment; ionic liquid–lithium salt; 15.95 min; 581.49 W | Cosmetic or health-related applications | [99] | |
| Pressurised Liquid Extraction (PLE) | P. taiwanensis P. morrisonicola | needles | Enzymatic hydrolysis (ethanol 70% v/v); 70 °C; 180 min; 4.12 MPa | Food, cosmetic, or health applications | [123] |
| P. pinaster P. d’Alpes | seeds | Water; 100 °C; 20–23 min; 4 MPa | Bioactive extracts for food supplements | [111] | |
| P. koraiensis | seeds | n-butane; 21 °C; 0.5 MPa | Oils for cosmetology and pharmaceutical application | [81] | |
| Supercritical Fluid Extraction (SFE) | P. pinaster | bark | CO2 + ethanol (10%, v/v); 50 °C; 35 min; 25 MPa | Aroma/flavor, food, and pharmaceutical industries | [118] |
| P. pinaster | bark | CO2:ethanol (30:70, v/v); 30 °C; 360 min; 25 MPa | Antioxidant extracts | [119] | |
| P. pinea P. sylvestris P. nigra P. parviflora P. ponderosa | bark | CO2 + ethanol (3% v/v); 40 and 60 °C; 200 bar | Food, cosmetic, or health-related applications | [71] | |
| P. niruri | -- | CO2; 90 min; 60 °C; 30 MPa | Antioxidant extracts | [88] | |
| P. nigra | bark | CO2 + ethanol; 42.8 °C; 137.9 min; 19.3 MPa | Pharmaceutical industry | [120] | |
| P. pinaster | wood | CO2 + ethanol (10%, v/v); 50 °C; 35 min; 25 MPa | Antioxidant extracts for food and pharmaceutical applications | [134] | |
| Ohmic Heating Extraction (OH) | P. pinaster | bark | Ethanol (50%, v/v); 83 °C; 30 min; 5-15 V/cm | Antioxidant extracts for food and pharmaceutical applications | [59] |
| Name | Chemical Formula | Concentration Range (mg/g) | Bioactivities | Reference |
|---|---|---|---|---|
| NEEDLES | ||||
| Epicatechin | C15H14O6 | 1.5 | antioxidant | [152,153] |
| p-Coumaric Acid | C9H8O3 | 2.3 | antioxidant, anti-inflammatory, hepatoprotective and renoprotective, anti-neurodegenerative, anti-cholesterolemic, improve insulin resistance, anti-tyrosinase, antimicrobial | [152,154,155,156] |
| SEEDS | ||||
| Protocatechuic Acid | C7H6O4 | 0.5 | anti-tyrosinase, antimicrobial, and anti-inflammatory activities | [156,157] |
| Catechin | C15H14O6 | 0.5 | hepatoprotective activity | [145,157] |
| Epigallocatechin Gallate | C22H18O11 | 0.5 | antimicrobial, antioxidant, photoprotective | [145,157] |
| Vanillic Acid | C8H8O4 | 0.9 | anti-inflammatory, neuroprotective | [157,158] |
| Syringic Acid | C9H10O5 | 1.0 | cardioprotective, antioxidant, antimicrobial, anti-inflammatory, neuro and hepatoprotective activities | [157,159] |
| Epicatechin | C15H14O6 | 1.3 | antioxidant | [157] |
| Taxifolin | C15H14O7 | 1.7 | antioxidant, anticancer, anti-inflammatory | [157] |
| Cinnamic Acid | C9H8O2 | 0.1 | anti-tyrosinase, antimicrobial, and anti-inflammatory | [156,157] |
| Eriodictyol | C15H12O6 | 3.8 | anti-inflammatory | [157,160] |
| m-Coumaric Acid | C9H8O3 | traces | not found | [157] |
| BARK | ||||
| Gallic Acid | C7H6O5 | traces–5.5 | anti-inflammatory, antihyperlipidemic, antioxidant, antitumor, antihyperglycemic, and anti-neurodegenerative, cardioprotective | [59,154,155,159,161] |
| Gallocatechin | C15H14O7 | 0.07–0.95 | inhibitor of melanin biosynthesis | [45,59,162] |
| Epicatechin | C15H14O6 | 0.06–1.9 | antioxidant | [71,161] |
| Epicatechin Gallate | C22H18O10 | 0.3–0.9 | antioxidant | [161] |
| Catechin | C15H14O6 | 0.095–7.7 | antioxidant, anticancer, cardioprotective, antifungal | [45,59,71,161,162] |
| Vanillic Acid | C8H8O4 | 0.02–0.07 | neuroprotective, anti-inflammatory | [45,59,158] |
| Caffeic Acid | C9H8O4 | 0.03–0.2 | antioxidant, photoprotective | [45,59,163] |
| Rosmaniric Acid | C18H16O8 | 0.4–0.8 | antioxidant, antidiabetic, antibacterial, antiviral | [59,164,165] |
| Catechin Gallate | C22H18O10 | 0.002–1.5 | antioxidant, anticancer | [71,166] |
| Taxifolin | C15H12O7 | 0.01–4.7 | antioxidant, anticancer, anti-inflammatory | [45,59,148,167] |
| 3,4 Dihydroxy-Benzoic Acid | C9H10O4 | 0.08–0.8 | neuroprotective, antioxidant, nematicidal activity | [45,59,168] |
| Ellagic acid | C14H6O8 | 0.4–4.0 | anti-inflammatory, antioxidant | [45,59,169] |
| Naringin | C27H32O17 | 0.8–2.0 | not found | [45,59] |
| Apigenin | C15H10O5 | 0.3–0.5 | anticancer, antioxidant, anti-inflammatory | [45,59,170] |
| Resveratrol | C14H12O3 | 0.03–0.4 | antioxidant, anti-cancer, cardioprotective, anti-inflammatory | [45,59,159,171] |
| Ferulic acid | C10H10O4 | 0.06–0.5 | antioxidant, photoprotective | [45,59,163] |
| p-coumaric acid | C9H8O3 | n.q. | antioxidant, anti-inflammatory, hepatoprotective and renoprotective, anti-neurodegenerative, anti-cholesterolemic, improve insulin resistance, anti-tyrosinase, antimicrobial | [59,154,155,156] |
| Quercetin | C15H10O7 | 0.06–1.1 | inflammatory, antimicrobial, anticancer | [45,59] |
| Procyanidin A2 | C30H24O12 | n.q. | antioxidant | [148] |
| Procyanidin B1 | C30H26O12 | n.q. | antioxidant, neuroprotective, anti-proliferative activity | [148] |
| Procyanidin B2 | C30H26O12 | n.q. | antioxidant, anti-inflammatory, cardioprotective, neuroprotective, anti-proliferative activity | [148] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira-Santos, P.; Zanuso, E.; Genisheva, Z.; Rocha, C.M.R.; Teixeira, J.A. Green and Sustainable Valorization of Bioactive Phenolic Compounds from Pinus By-Products. Molecules 2020, 25, 2931. https://doi.org/10.3390/molecules25122931
Ferreira-Santos P, Zanuso E, Genisheva Z, Rocha CMR, Teixeira JA. Green and Sustainable Valorization of Bioactive Phenolic Compounds from Pinus By-Products. Molecules. 2020; 25(12):2931. https://doi.org/10.3390/molecules25122931
Chicago/Turabian StyleFerreira-Santos, Pedro, Elisa Zanuso, Zlatina Genisheva, Cristina M. R. Rocha, and José A. Teixeira. 2020. "Green and Sustainable Valorization of Bioactive Phenolic Compounds from Pinus By-Products" Molecules 25, no. 12: 2931. https://doi.org/10.3390/molecules25122931
APA StyleFerreira-Santos, P., Zanuso, E., Genisheva, Z., Rocha, C. M. R., & Teixeira, J. A. (2020). Green and Sustainable Valorization of Bioactive Phenolic Compounds from Pinus By-Products. Molecules, 25(12), 2931. https://doi.org/10.3390/molecules25122931







