Mechanism of Reactions of 1-Substituted Silatranes and Germatranes, 2,2-Disubstituted Silocanes and Germocanes, 1,1,1-Trisubstituted Hyposilatranes and Hypogermatranes with Alcohols (Methanol, Ethanol): DFT Study
Abstract
1. Introduction
2. Results and Discussion
2.1. Methodology
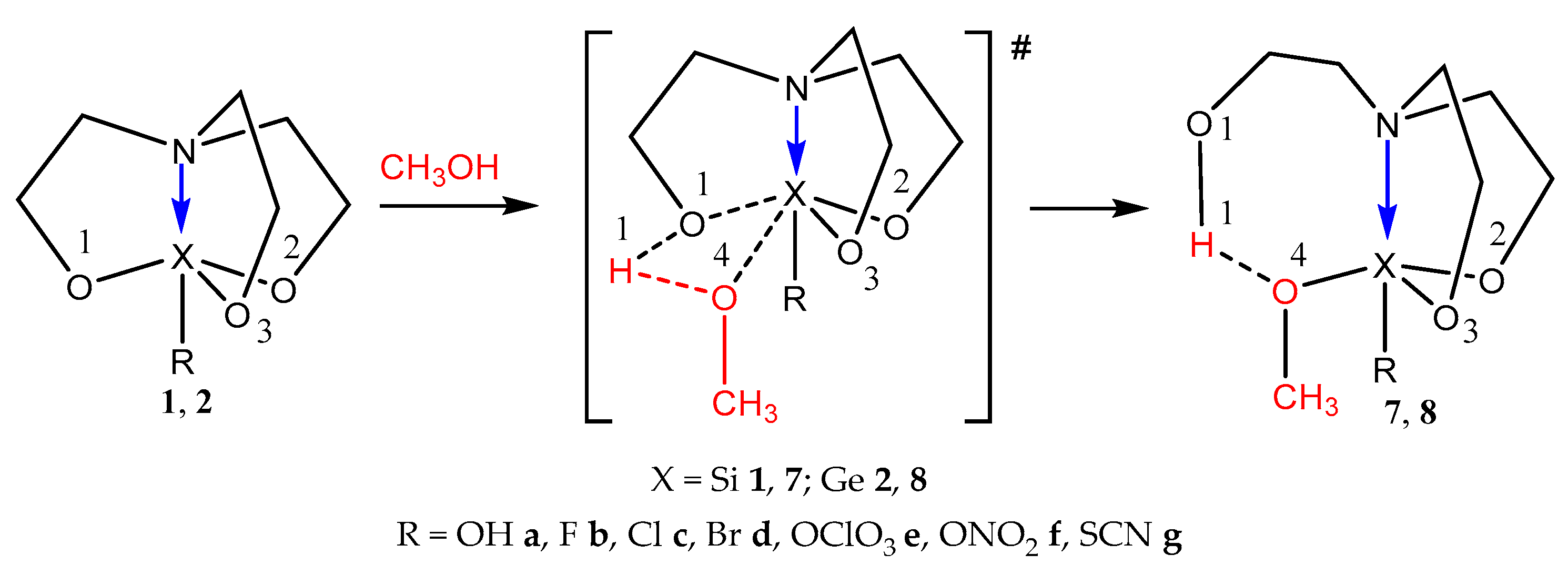
2.2. Reactions of 1-Substituted Atranes with Methanol
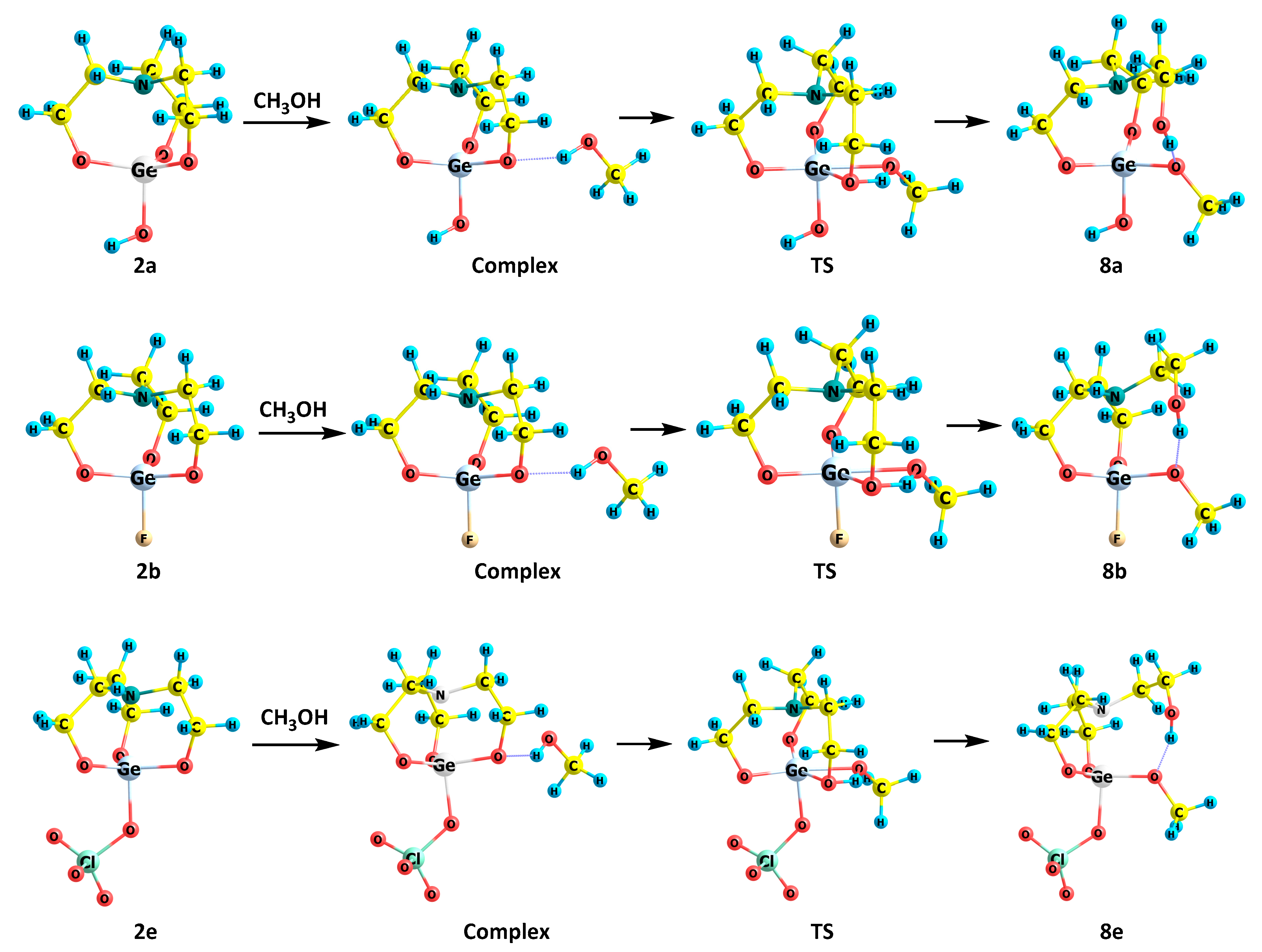
2.3. Reactions of 2,2-Disubstituted Ocanes with Methanol
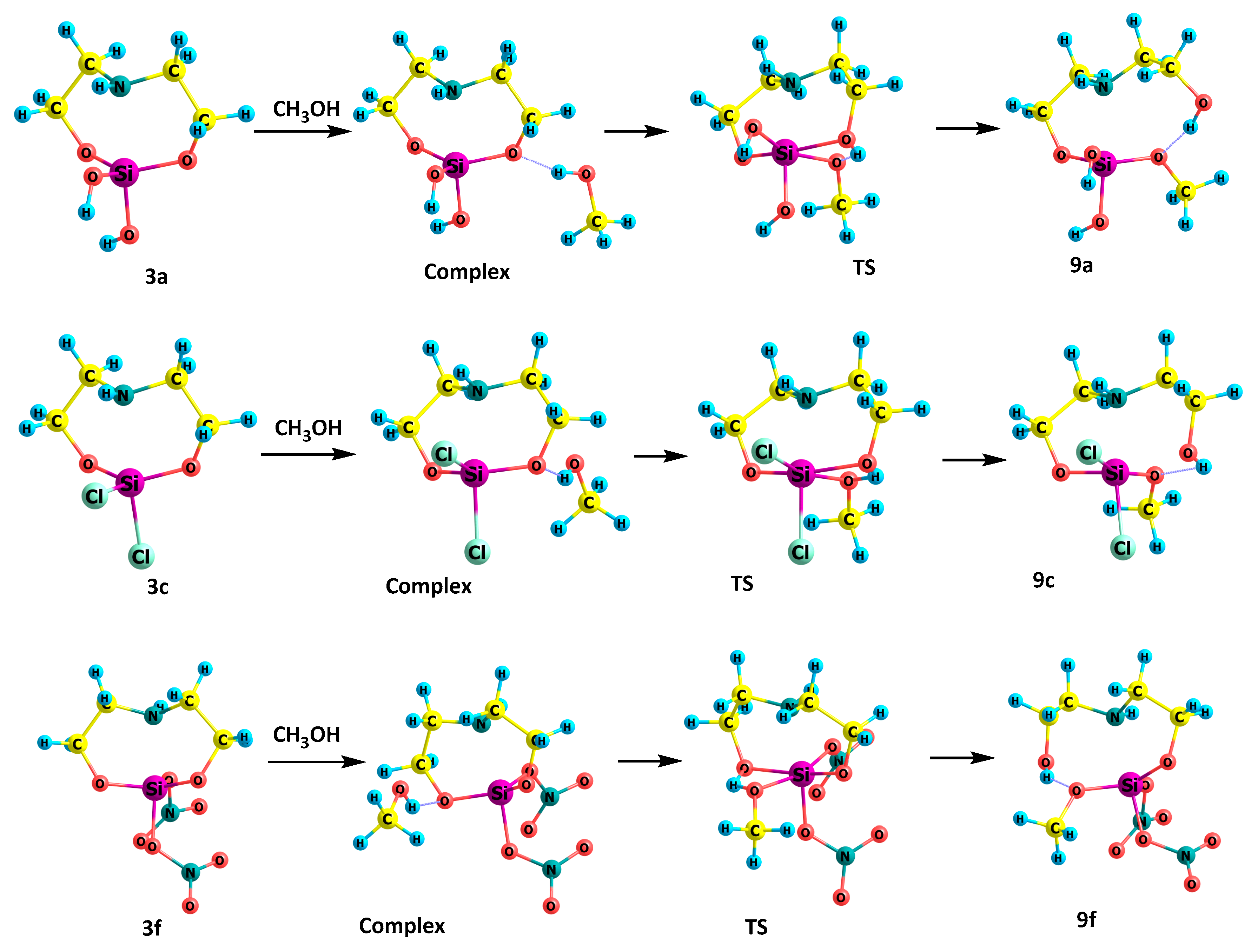
2.4. Reactions of 1,1,1-Trisubstituted Hypoatranes with Methanol
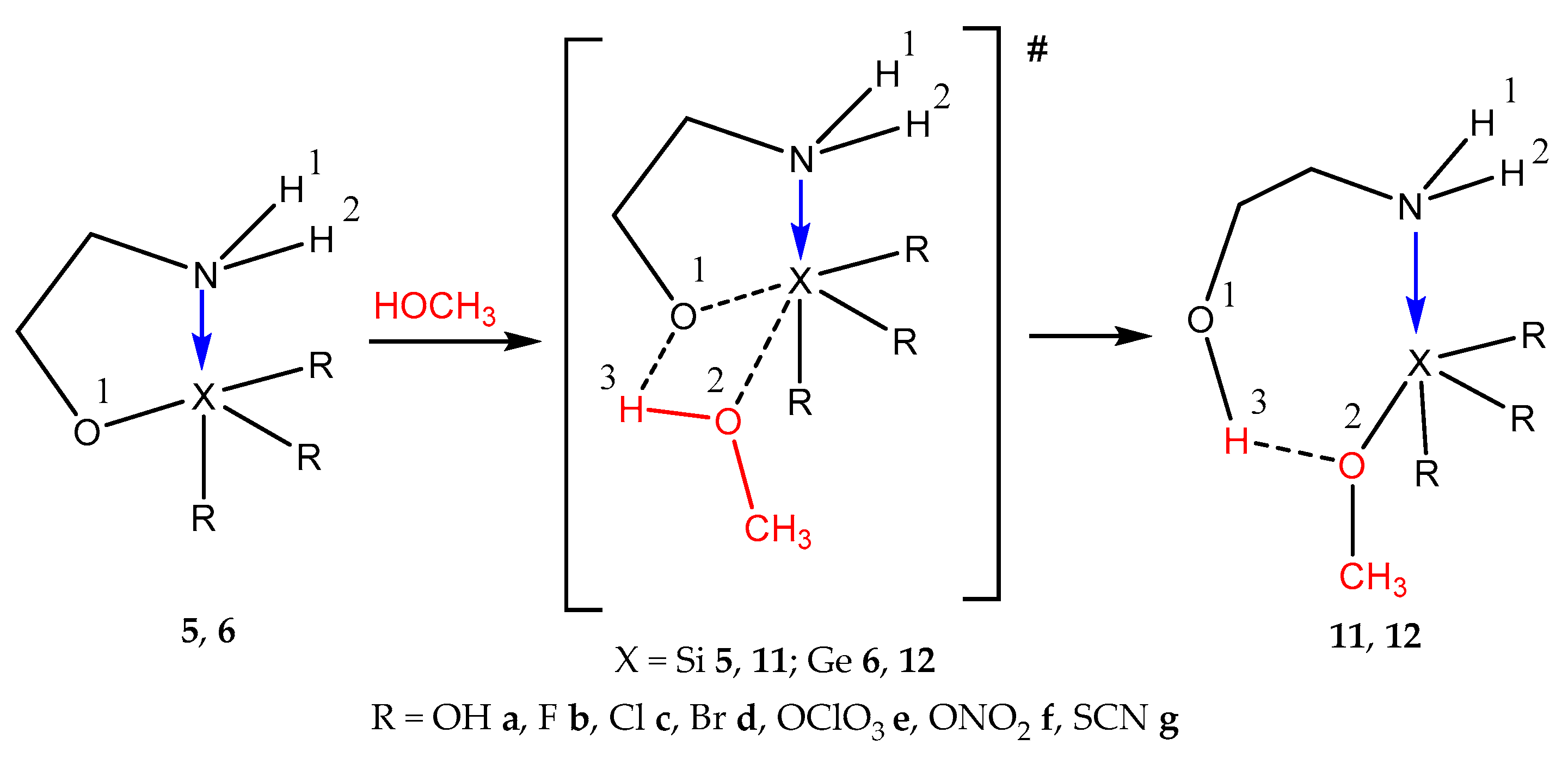
2.5. Reactions of Atranes, Ocanes, and Hypoatranes with Ethanol
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Voronkov, M.G.; Zelčans, G.; Mažeika, I.; Baltkajs, J.; Jegorov, J.; Pestunovich, V. Silatranes: Intracomplex heterocyclic compounds of pentacoordinated silicon. Pure Appl. Chem. 1966, 13, 35–59. [Google Scholar] [CrossRef]
- Verkade, J.G. Main group atranes: Chemical and structural features. Coord. Chem. Rev. 1994, 137, 233–295. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Baryshok, V.P. Antitumor activity of silatranes (a review). Pharm. Chem. J. 2004, 38, 3–9. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Baryshok, V.P. Atranes as a new generation of biologically active substances. Herald Russ. Acad. Sci. 2010, 80, 514–521. [Google Scholar] [CrossRef]
- Menchikov, L.G.; Ignatenko, M.A. Biological activity of organogermanium compounds (a review). Pharm. Chem. J. 2013, 46, 635–638. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Baryshok, V.P. Silatranes in medicine and agriculture; Publishing House SB RAS: Novosibirsk, Russia, 2005; p. 255. [Google Scholar]
- Garabadzhiu, A.V.; Voronkov, M.G.; Nyanikova, G.G.; Samokhin, G.S.; Vrazhnov, D.V.; Kochina, T.A. The influence of silatranes, germatranes, protatranes, and triethanolamine on vital functions of microorganisms. Dokl. Biol. Sci. 2011, 439, 264–266. [Google Scholar] [CrossRef]
- Singh, G.; Saroa, A.; Girdhar, S.; Rani, S.; Sahoo, S.; Choquesillo-Lazarte, D. Synthesis, characterization, electronic absorption and antimicrobial studies of N-(silatranylpropyl)phthalimide derived from phthalic anhydride. Inorg. Chim. Acta 2015, 427, 232–239. [Google Scholar] [CrossRef]
- Ye, F.; Song, X.; Liu, J.; Xu, X.; Wang, Y.; Hu, L.; Wang, Y.; Liang, G.; Guo, P.; Xie, Z. Design, synthesis, and biological evaluation of γ-aminopropyl silatrane–acyclovir hybrids with immunomodulatory effects. Chem. Biol. Drug Des. 2015, 86, 905–910. [Google Scholar] [CrossRef]
- Singh, G.; Arora, A.; Mangat, S.S.; Rani, S.; Kaur, H.; Goyal, K.; Sehgal, R.; Maurya, I.K.; Tewari, R.; Choquesillo-Lazarte, D.; et al. Design, synthesis and biological evaluation of chalconyl blended triazole allied organosilatranes as giardicidal and trichomonacidal agents. Eur. J. Med. Chem. 2016, 108, 287–300. [Google Scholar] [CrossRef]
- Singh, R.; Mutneja, R.; Kaur, V.; Wagler, J.; Kroke, E. Derivatization of 3-aminopropylsilatrane to introduce azomethine linkage in the axial chain: Synthesis, characterization and structural studies. J. Organomet. Chem. 2013, 724, 186–191. [Google Scholar] [CrossRef]
- Brennan, B.J.; Gust, D.; Brudvig, G.W. Organosilatrane building blocks. Tetrahedron Lett. 2014, 55, 1062–1064. [Google Scholar] [CrossRef]
- Mutneja, R.; Singh, R.; Kaur, V.; Wagler, J.; Felsc, S.; Kroke, E. Schiff base tailed silatranes for the fabrication of functionalized silica based magnetic nano-cores possessing active sites for the adsorption of copper ions. New J. Chem. 2016, 40, 1640–1648. [Google Scholar] [CrossRef]
- Vlasova, N.N.; Sorokin, M.S.; Oborina, E.N. Carbofunctional sulfur-containing organosilicon compounds. Appl. Organomet. Chem. 2017, 31, e3668. [Google Scholar] [CrossRef]
- Singh, G.; Arora, A.; Rani, S.; Kalra, P.; Aulakh, D.; Wriedt, M. A family of silatrane-armed triazole-encapped salicylaldehyde-derived Schiff bases: Synthesis, spectral analysis, and antimicrobial and quantum chemical evaluation. Appl. Organomet. Chem. 2017, 31, e3728. [Google Scholar] [CrossRef]
- Krause, J.; Richter, S. New catalysts and use thereof in the production of polyurethanes. Patent DE 10 2008 021 980 A1, 18 April 2009. [Google Scholar]
- Zöller, T.; Iovkova-Berends, L.; Dietz, C.; Berends, T.; Jurkschat, K. On the reaction of elemental tin with alcohols: A straightforward approach to tin(II) and tin(IV) alkoxides and related tinoxo clusters. Chem. Eur. J. 2011, 17, 2361–2364. [Google Scholar] [CrossRef]
- Karlov, S.S.; Zaitseva, G.S. Germatranes and their analogs. Synthesis, structure, and reactivity. Chem. Heterocycl. Comp. 2001, 37, 1325–1357. [Google Scholar] [CrossRef]
- Puri, J.K.; Singh, R.; Chahal, V.K. Silatranes: A review on their synthesis, structure, reactivity and applications. Chem. Soc. Rev. 2011, 40, 1791–1840. [Google Scholar] [CrossRef]
- Adamovich, S.N. New atranes and similar ionic complexes. Synthesis, structure, properties. Appl Organomet. Chem. 2019, 33, e4940. [Google Scholar] [CrossRef]
- Milov, A.A.; Minyaev, R.M.; Minkin, V.I. Hypervalent intramolecular X←N (X = C, Si, Ge) coordination in atranes: Quantum-chemical study. Russ. J. Org. Chem. 2003, 39, 340–347. [Google Scholar] [CrossRef]
- Chernyshev, E.A.; Knyazev, S.P.; Kirin, V.N.; Vasilev, I.M.; Alekseev, N.V. Structural features of silatranes and germatranes. Russ. J. Gen. Chem. 2004, 74, 58–65. [Google Scholar] [CrossRef]
- Zabalov, M.V.; Karlov, S.S.; Zaitseva, G.S.; Lemenovskii, D.A. The molecular and electronic structure features of silatranes, germatranes, and their carbon analogs. Russ. Chem. Bull. 2006, 55, 464–476. [Google Scholar] [CrossRef]
- Feshin, V.P.; Feshina, E.V. Nature of coordination bond in silatranes and its formation dynamics according to the ab initio calculations. Russ. J. Gen. Chem. 2014, 84, 70–74. [Google Scholar] [CrossRef]
- Korlyukov, A.A. Coordination compounds of tetravalent silicon, germanium and tin: The structure, chemical bonding and intermolecular interactions in them. Russ. Chem. Rev. 2015, 84, 422–440. [Google Scholar] [CrossRef]
- Marín-Luna, M.; Alkorta, I.; Elguero, J. Theoretical study of the geometrical, energetic and NMR properties of atranes. J. Organomet. Chem. 2015, 794, 206–215. [Google Scholar] [CrossRef]
- Karlov, S.S.; Tyurin, D.A.; Zabalov, M.V.; Churakov, A.V.; Zaitseva, G.S. Quantum chemical study of group 14 elements pentacoordinated derivatives–metallatranes. J. Mol. Struct. THEOCHEM. 2005, 724, 31–37. [Google Scholar] [CrossRef]
- Belogolova, E.F.; Sidorkin, V.F. Correlation among the gas-phase, solution, and solid-phase geometrical and NMR parameters of dative bonds in the pentacoordinate silicon compounds. 1-Substituted silatranes. J. Phys. Chem. A. 2013, 117, 5365–5376. [Google Scholar] [CrossRef]
- Ignatyev, I.S.; Samokhin, G.S.; Kochina, T.A.; Belyaeva, V.V.; Khaikin, S.Y.; Montejo, M.; López González, J.J.; Voronkov, M.G. Vibrational spectra and electronic structure of germatranols (HO)4-n Ge(OCH2CH2)nNR3-n (R = H; n = 1–3) with transannular Ge…N bonding. J. Organomet. Chem. 2013, 747, 62–68. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Zel’bst, E.A.; Belyaeva, V.V. Intramolecular inductive through-space interaction between nitrogen and oxygen atoms in silatranes, quasisilatranes, protatranes, triethanolamine, and diethanolamine. Dokl. Chem. 2014, 433, 167–170. [Google Scholar] [CrossRef]
- Glowacki, B.; Lutter, M.; Schollmeyer, D.; Hiller, W.; Jurkschat, K. Novel stannatrane N(CH2CMe2O)2(CMe2CH2O)SnO-t-Bu and related oligonuclear tin(IV) oxoclusters. Two isomers in one crystal. Inorg. Chem. 2016, 55, 10218–10228. [Google Scholar] [CrossRef]
- Lermontova, E.K.; Selina, A.A.; Karlov, S.S.; Churakov, A.V.; Howard, J.A.K.; Oprunenko, Y.F.; Antipin, M.Y.; Sundermeyer, J.; Zaitseva, G.S. The transannular interaction germanium–nitrogen in germocanes: The influence of substituents. J. Organomet. Chem. 2006, 691, 5710–5724. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Korlyukov, A.A.; Zelbst, E.A.; Grebneva, E.A.; Trofimova, O.M.; Antipin, M.Y. Molecular structure of 1,1-difluoroquasisilatrane (N→Si) F2Si(OCH2CH2)2NH. Dokl. Chem. 2008, 418, 27–29. [Google Scholar] [CrossRef]
- Ignat’ev, I.S.; Voronkov, M.G.; Kochina, T.A.; Vrazhnov, D.V. Quantum-chemical study of the stereoelectronic structure of 1-fluorosilatrane, 1,1-difluoroquasisilatrane, 1,1,1-trifluorohyposilatrane, and cations formed thereof. Russ. J. Gen. Chem. 2010, 80, 2274–2282. [Google Scholar] [CrossRef]
- Alekseev, N.V.; Chernyshev, E.A. A quantum chemical study of the electronic structure of substituted germocanes. J. Struct. Chem. 2010, 51, 419–427. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Korlyukov, A.A.; Samokhin, G.S.; Vrazhnov, D.V.; Kochina, T.A. Germatranes and their quasi and hypo analogs with highly electronegative substituent at the Ge atom. Russ. Chem. Bull. 2012, 61, 992–998. [Google Scholar] [CrossRef]
- Vereshchagina, Y.A.; Alimova, A.Z.; Chachkov, D.V.; Ishmaeva, E.A.; Kochina, T.A.; Voronkov, M.G. Polarity and structure of 1,3,5,11-tetraoxa-8-aza-4-germaspiro [3,7]undecan-2-one. Russ. J. Org. Chem. 2014, 50, 1225–1226. [Google Scholar] [CrossRef]
- Vereshchagina, Y.A.; Alimova, A.Z.; Chachkov, D.V.; Ishmaeva, E.A.; Kochina, T.A. Polarity and structure of 1,1-dihalo-2,8-dioxa-5-azagermocanes. Russ. J. Org. Chem. 2015, 51, 750–752. [Google Scholar] [CrossRef]
- Ignatyev, I.S.; Kochina, T.A.; Avrorin, V.V.; Gurzhiy, V.V.; Fundamensky, V.S. Molecular and crystal structures of 2-phenyl-2-hydro-6-methyl-1,3-dioxa-6-aza-2-silacyclooctane. J. Mol. Struct. 2015, 1094, 169–173. [Google Scholar] [CrossRef]
- Samokhin, G.S.; Vrazhnov, D.V.; Kochina, T.A.; Voronkov, M.G. Quasigermatranes and their monocyclic analogs with highly electronegative substituents at the germanium atom: A new synthesis strategy. Glass Phys. Chem. 2010, 36, 623–627. [Google Scholar] [CrossRef]
- Samokhin, G.S. A new path to Ge-substituted germatranes, quasigermatranes, and hypogermatranes. Ph.D. Thesis, I.V. Grebenshchikov Institute of Silicate Chemistry of RAS, Saint-Petersburg, Russia, 2011. [Google Scholar]
- Voronkov, M.G.; Samokhin, G.S.; Vrazhnov, D.V.; Kochina, T.A. New intracomplex compounds of pentacoordinate germanium. Hypogermatranes, Ge-substituted (N→Ge) 2-oxa-5-aza-1-germacyclopentanes. Russ. J. Gen. Chem. 2012, 82, 170–171. [Google Scholar] [CrossRef]
- Vereshchagina, Y.A.; Ismagilova, R.R.; Chachkov, D.V. Mechanism of hydrolysis of 1,1,1-trisubstituted hyposilatranes and hypogermatranes. Russ. J. Org. Chem. 2019, 55, 227–233. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Belyaeva, V.V.; Abzaeva, K.A. Basicity of silatranes (Review). Chem. Heterocycl. Comp. 2012, 47, 1330–1338. [Google Scholar] [CrossRef]
- Sidorkin, V.F.; Shagun, V.A.; Pestunovich, V.A. Stereoelectronic effects and the problem of the choice of model compounds for organic derivatives of a pentacoordinated silicon atom (taking silatranes as an example). Russ. Chem Bul. 1999, 48, 1049–1053. [Google Scholar] [CrossRef]
- Yashikawa, A.; Gordon, M.S.; Sidorkin, V.F.; Pestunovich, V.A. Proton affinities of the silatranes and their analogues. Organometallics 2001, 20, 927–931. [Google Scholar] [CrossRef][Green Version]
- Belyaeva, V.V.; Frolov, Y.L.; Abumov, P.F.; Voronkov, M.G. Quantum-chemical study of the electron-donating ability of endocyclic nitrogen and oxygen atoms in silicon-substituted silatranes. Dokl. Phys. Chem. 2004, 397, 150–153. [Google Scholar] [CrossRef]
- Sok, S.; Gordon, M.S. A dash of protons: A theoretical study on the hydrolysis mechanism of 1-substituted silatranes and their protonated analogs. Comput. Theor. Chem. 2012, 987, 2–15. [Google Scholar] [CrossRef]
- Zoöller, T.; Jurkschat, K. Novel trialkanolamine derivatives of tin of the type [N(CH2CMe2O)2(CH2)nOSnOR]m (m = 1, 2; n = 2, 3; R = t-Bu, 2,6-Me2C6H3) and related tri- and pentanuclear tin(IV) oxoclusters. Syntheses and molecular structures. Inorg. Chem. 2013, 52, 1872–1882. [Google Scholar] [CrossRef]
- Vereshchagina, Y.A.; Chachkov, D.V.; Alimova, A.Z.; Ishmaeva, E.A. Mechanism of the hydrolysis reactions of 1-hydroxysilatrane and 1-hydroxygermatrane, 2,2-dihydroxysilocane and 2,2-dihydroxygermocane. Phosphorus, Sulfur Silicon Relat. Elem. 2016, 191, 496–501. [Google Scholar] [CrossRef]
- Ignatyev, I.S.; Montejo, M.; Rodriguez Ortega, P.G.; Kochina, T.A.; González, J.J.L. DFT study of the hydrolysis reaction in atranes and ocanes: The influence of transannular bonding. J. Mol. Model. 2016, 22, 3. [Google Scholar] [CrossRef]
- Vereshchagina, Y.A.; Chachkov, D.V.; Ismagilova, R.R.; Vedeneeva, E.A. Mechanism of hydrolysis of 2,2-disubstituted silocanes and germocanes and 1-substituted silatranes and germatranes. Russ. J. Org. Chem. 2018, 54, 490–499. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Wang, Y. Generalized gradient approximation for the exchange-correlation hole of a many-electron system. Phys. Rev. B 1996, 54, 16533–16539. [Google Scholar] [CrossRef] [PubMed]
- McLean, A.D.; Chandler, G.S. Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z = 11–18. J. Chem. Phys. 1980, 72, 5639–5648. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A. Gaussian 09; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Medvedev, M.G.; Bushmarinov, I.S.; Sun, J.; Perdew, J.P.; Lyssenko, K.A. Density functional theory is straying from the path toward the exact functional. Science 2017, 355, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Anglada, J.M.; Bo, C.; Bofill, J.M.; Crehuet, R.; Poblet, J.M. Inductive effects in neutral pentacoordinated silicon compounds containing a Si←N dative bond. A theoretical study. Organometallics 1999, 18, 5584–5593. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


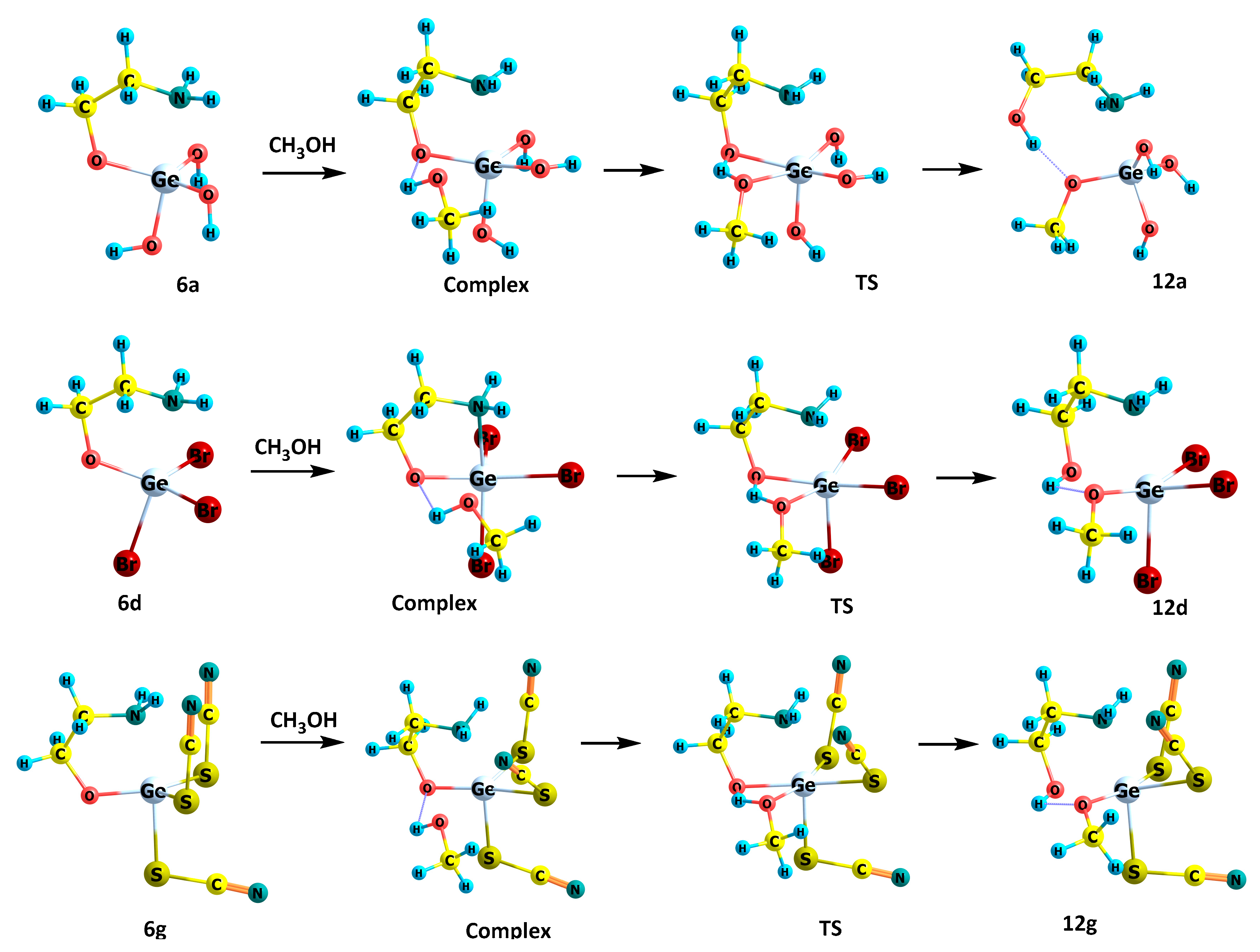
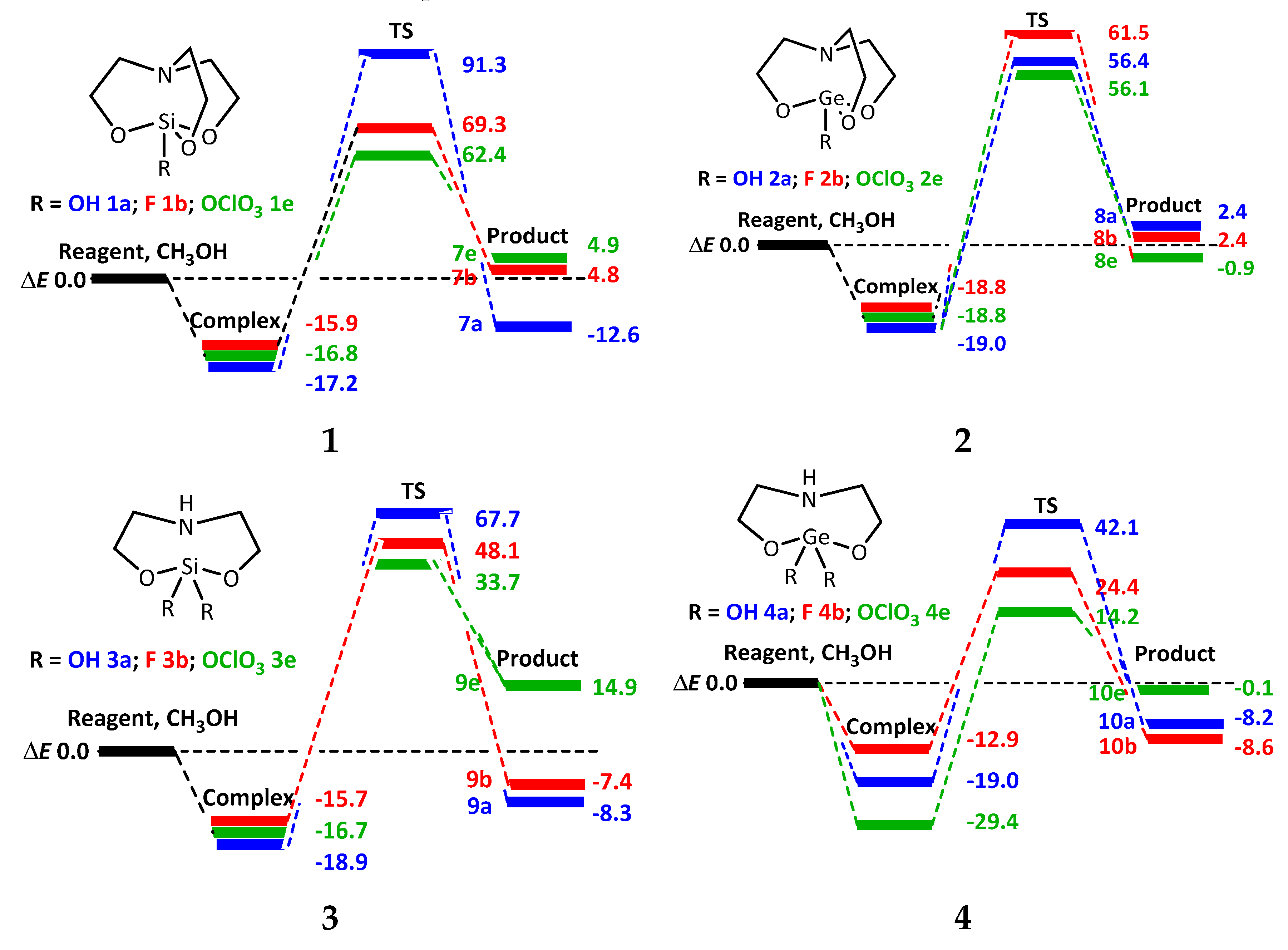
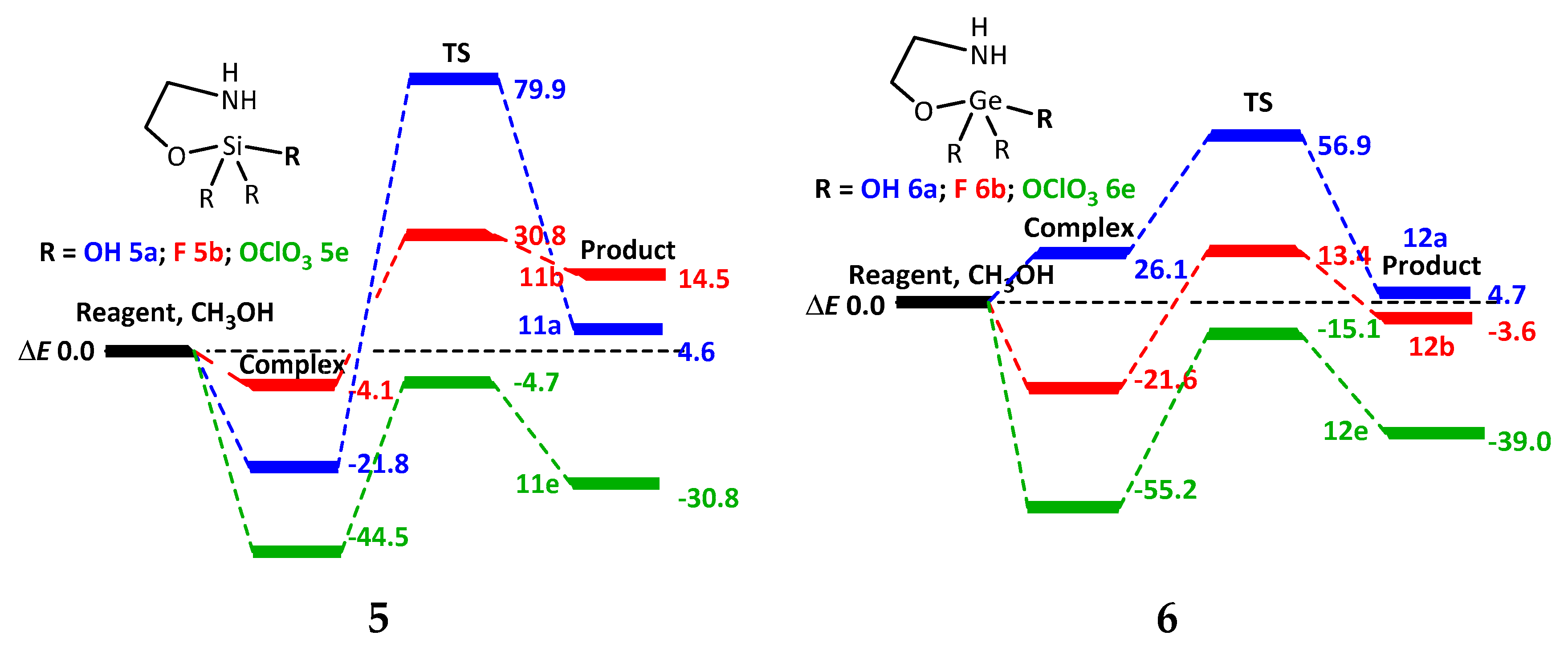


| ΔE # | ΔG # | ν1, cm−1 | |
|---|---|---|---|
| 1a | 91.3 | 149.4 | 877i |
| 1b | 69.3 | 128.1 | 910i |
| 1c | 72.4 | 131.9 | 951i |
| 1d | 73.2 | 132.8 | 966i |
| 1e | 62.4 | 120.4 | 992i |
| 1f | 71.9 | 129.1 | 969i |
| 1g | 78.8 | 135.8 | 972i |
| 2a | 56.4 | 113.2 | 821i |
| 2b | 61.5 | 115.6 | 803i |
| 2c | 67.3 | 121.4 | 815i |
| 2d | 68.7 | 122.7 | 819i |
| 2e | 56.1 | 110.9 | 891i |
| 2f | 66.3 | 119.4 | 851i |
| 2g | 72.4 | 124.8 | 822i |
| ΔE # | ΔG # | ν1, cm−1 | |
|---|---|---|---|
| 3a | 67.7 | 125.6 | 835i |
| 3b | 48.1 | 106.0 | 942i |
| 3c | 55.1 | 114.0 | 990i |
| 3d | 55.8 | 113.4 | 1020i |
| 3e | 33.7 | 93.3 | 1041i |
| 3f | 48.6 | 104.6 | 1000i |
| 3g | 49.9 | 107.6 | 1035i |
| 4a | 42.1 | 97.3 | 802i |
| 4b | 24.4 | 79.8 | 922i |
| 4c | 38.0 | 93.9 | 917i |
| 4d | 41.5 | 97.3 | 928i |
| 4e | 14.2 | 70.8 | 1010i |
| 4f | 37.6 | 92.6 | 950i |
| 4g | 38.5 | 95.3 | 952i |
| ΔE # | ΔG # | ν1, cm−1 | |
|---|---|---|---|
| 5a | 79.9 | 136.0 | 963i |
| 5b | 30.8 | −95.1 | 1020i |
| 5c | 35.5 | 92.9 | 1049i |
| 5d | 39.3 | 96.3 | 1064i |
| 5e | −4.7 | 56.0 | 1145i |
| 5f | 13.9 | 72.6 | 1074i |
| 5g | 28.4 | 83.9 | 1103i |
| 6a | 56.9 | 110.7 | 925i |
| 6b | 13.4 | 68.3 | 1000i |
| 6c | 27.7 | 84.2 | 995i |
| 6d | 33.4 | 89.9 | 994i |
| 6e | −15.1 | 44.8 | 1120i |
| 6f | 3.6 | 61.2 | 1037i |
| 6g | 21.7 | 79.2 | 1034i |
| ΔE # | ΔG # | ν1, cm−1 | |
|---|---|---|---|
| 1a | 76.4 | 137.4 | 893i |
| 1b | 71.4 | 130.5 | 917i |
| 1c | 74.8 | 134.5 | 956i |
| 1d | 75.7 | 134.7 | 973i |
| 2a | 58.3 | 115.5 | 833i |
| 2b | 63.3 | 117.9 | 814i |
| 2c | 69.5 | 124.1 | 820i |
| 2d | 71.1 | 125.6 | 828i |
| 3a | 74.1 | 130.9 | 857i |
| 3b | 50.0 | 108.1 | 951i |
| 3c | 57.1 | 116.1 | 998i |
| 3d | 57.9 | 116.5 | 1028i |
| 4a | 53.4 | 107.9 | 905i |
| 4b | 33.4 | 88.3 | 976i |
| 4c | 43.4 | 99.1 | 968i |
| 4d | 47.6 | 103.1 | 965i |
| 5a | 78.4 | 131.9 | 914i |
| 5b | 32.1 | 88.8 | 1030i |
| 5c | 36.8 | 93.6 | 1056i |
| 5d | 40.6 | 97.4 | 1072i |
| 6a | 54.8 | 106.6 | 883i |
| 6b | 14.6 | 69.7 | 1012i |
| 6c | 29.3 | 86.1 | 1003i |
| 6d | 35.2 | 91.9 | 1001i |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chachkov, D.; Ismagilova, R.; Vereshchagina, Y. Mechanism of Reactions of 1-Substituted Silatranes and Germatranes, 2,2-Disubstituted Silocanes and Germocanes, 1,1,1-Trisubstituted Hyposilatranes and Hypogermatranes with Alcohols (Methanol, Ethanol): DFT Study. Molecules 2020, 25, 2803. https://doi.org/10.3390/molecules25122803
Chachkov D, Ismagilova R, Vereshchagina Y. Mechanism of Reactions of 1-Substituted Silatranes and Germatranes, 2,2-Disubstituted Silocanes and Germocanes, 1,1,1-Trisubstituted Hyposilatranes and Hypogermatranes with Alcohols (Methanol, Ethanol): DFT Study. Molecules. 2020; 25(12):2803. https://doi.org/10.3390/molecules25122803
Chicago/Turabian StyleChachkov, Denis, Rezeda Ismagilova, and Yana Vereshchagina. 2020. "Mechanism of Reactions of 1-Substituted Silatranes and Germatranes, 2,2-Disubstituted Silocanes and Germocanes, 1,1,1-Trisubstituted Hyposilatranes and Hypogermatranes with Alcohols (Methanol, Ethanol): DFT Study" Molecules 25, no. 12: 2803. https://doi.org/10.3390/molecules25122803
APA StyleChachkov, D., Ismagilova, R., & Vereshchagina, Y. (2020). Mechanism of Reactions of 1-Substituted Silatranes and Germatranes, 2,2-Disubstituted Silocanes and Germocanes, 1,1,1-Trisubstituted Hyposilatranes and Hypogermatranes with Alcohols (Methanol, Ethanol): DFT Study. Molecules, 25(12), 2803. https://doi.org/10.3390/molecules25122803








