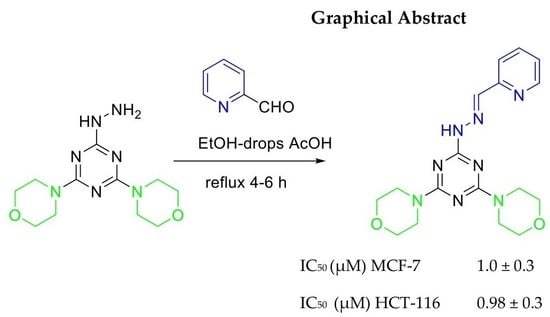
Synthesis and Characterization of New Series of 1,3-5-Triazine Hydrazone Derivatives with Promising Antiproliferative Activity
Abstract
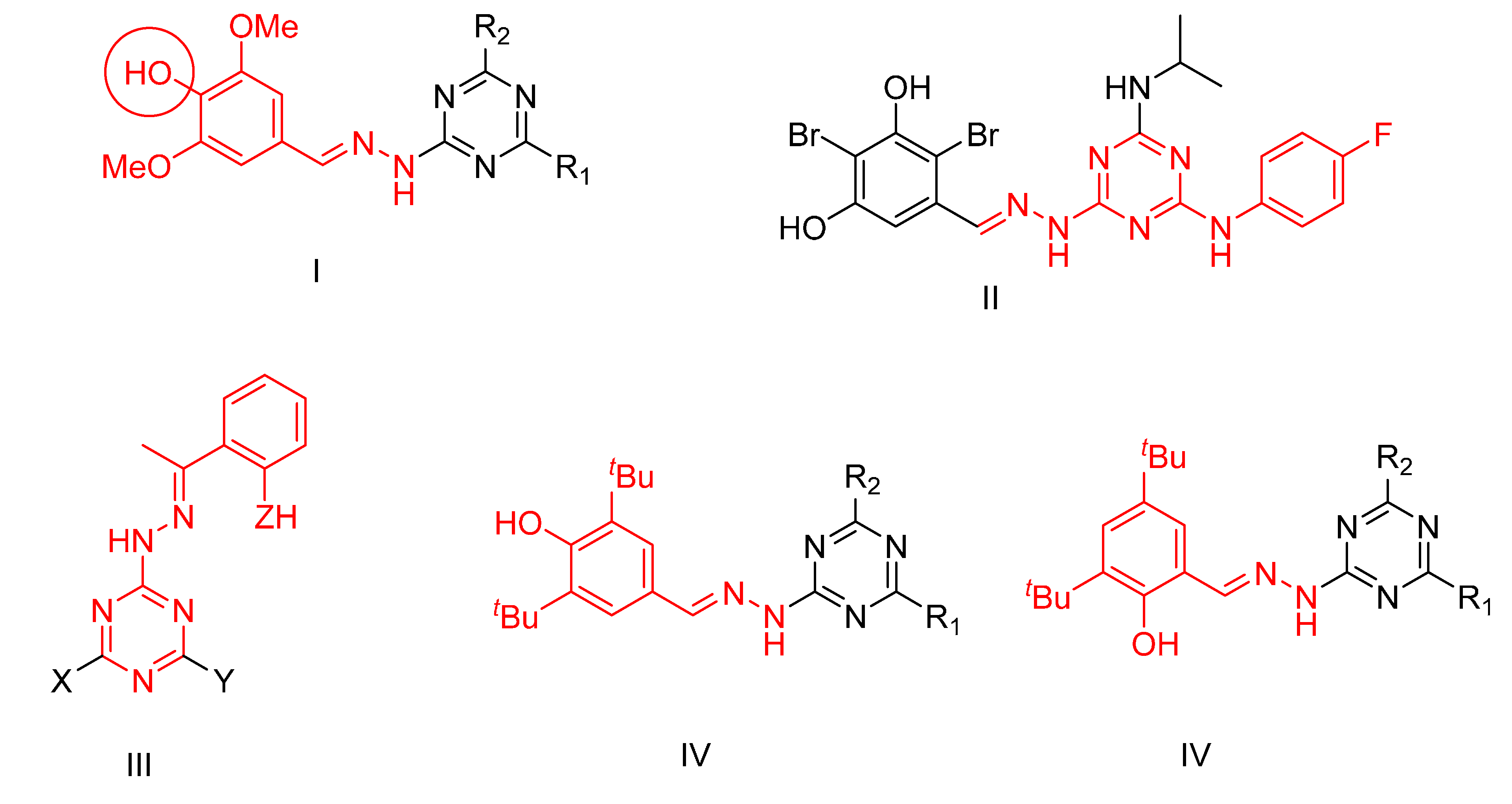
1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Biology
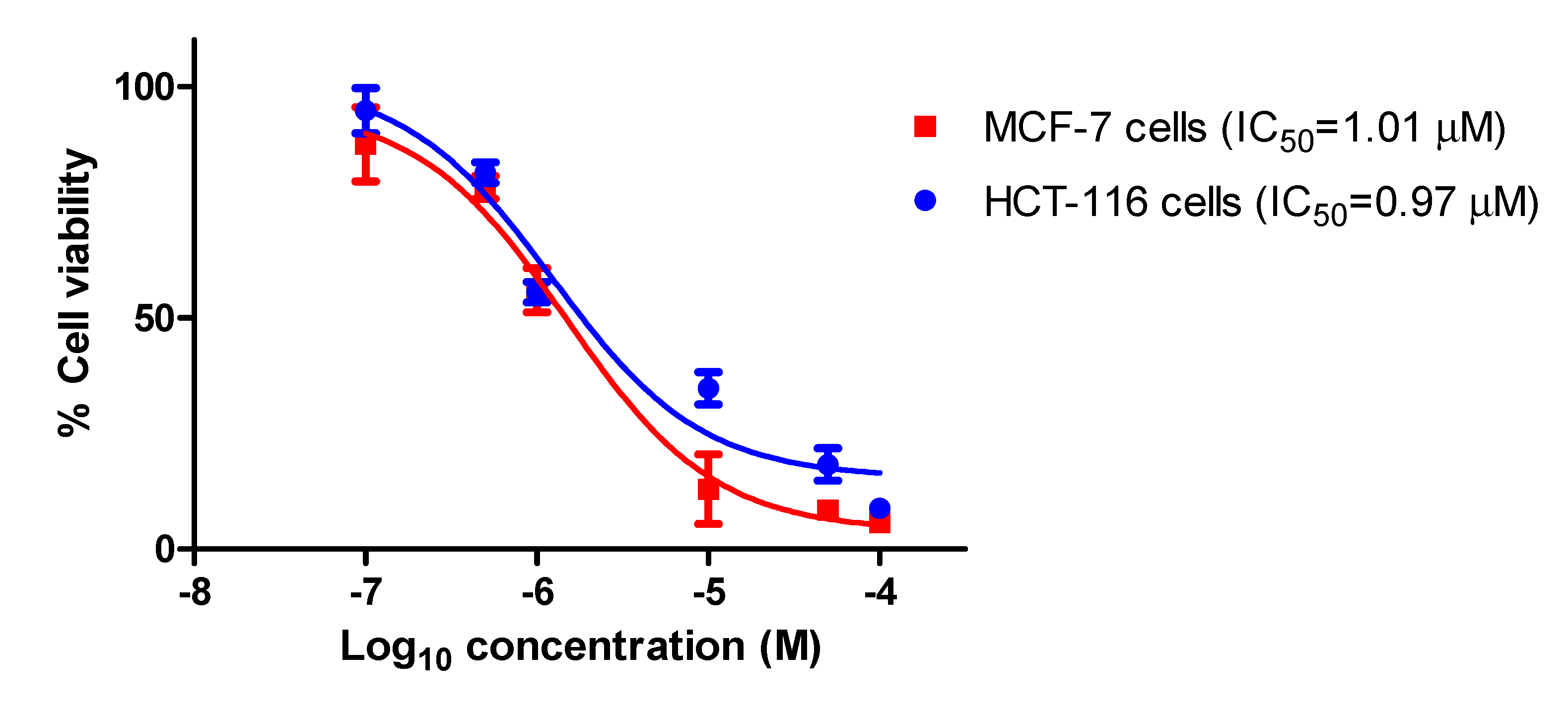
2.2.1. Antiproliferative Activity
3. Experimental Section
3.1. Materials and Methods
3.2. General Method for the Synthesis of 1,3,5-Triazine-Hydrazone Derivatives
3.2.1. 2-(2-Benzylidenehydrazinyl)-4,6-di(piperidin-1-yl)-1,3,5-triazine, 7a
3.2.2. 2-(2-(4-Chlorobenzylidene)hydrazinyl)-4,6-di(piperidin-1-yl)-1,3,5-triazine, 7b
3.2.3. 2-(2-(4-Bromobenzylidene)hydrazinyl)-4,6-di(piperidin-1-yl)-1,3,5-triazine, 7c
3.2.4. 4-((2-(4,6-Di(piperidin-1-yl)-1,3,5-triazin-2-yl)hydrazono)methyl)phenol, 7d
3.2.5. 2-(2-(4-Fluorobenzylidene)hydrazinyl)-4,6-di(piperidin-1-yl)-1,3,5-triazine, 7e
3.2.6. 2-(2-(4-Methoxybenzylidene)hydrazinyl)-4,6-di(piperidin-1-yl)-1,3,5-triazine, 7f
3.2.7. 4,4’-(6-(2-(4-Fluorobenzylidene)hydrazinyl)-1,3,5-triazine-2,4-diyl)dimorpholine, 8e
3.2.8. 4-(4-(2-Benzylidenehydrazinyl)-6-(piperidin-1-yl)-1,3,5-triazin-2-yl)morpholine, 9a
3.2.9. 4-(4-(2-(4-Chlorobenzylidene)hydrazinyl)-6-(piperidin-1-yl)-1,3,5-triazin-2- yl)morpholine, 9b
3.2.10. 4-(4-(2-(4-Bromobenzylidene)hydrazinyl)-6-(piperidin-1-yl)-1,3,5-triazin-2-yl)morpholine, 9c
3.2.11. 4-((2-(4-Morpholino-6-(piperidin-1-yl)-1,3,5-triazin-2-yl)hydrazono)methyl)phenol, 9d
3.2.12. 4-(4-(2-(4-Fluorobenzylidene)hydrazinyl)-6-(piperidin-1-yl)-1,3,5-triazin-2-yl)morpholine, 9e
3.2.13. 4-(4-(2-(4-Methoxybenzylidene)hydrazinyl)-6-(piperidin-1-yl)-1,3,5-triazin-2-yl)morpholine, 9f
3.2.14. 2,4-Di(piperidin-1-yl)-6-(2-(pyridin-2-ylmethylene)hydrazinyl)-1,3,5-triazine, 10
3.2.15. 4,4’-(6-(2-(Pyridin-2-ylmethylene)hydrazinyl)-1,3,5-triazine-2,4-diyl)dimorpholine, 11
3.2.16. 4-(4-(Piperidin-1-yl)-6-(2-(pyridin-2-ylmethylene)hydrazinyl)-1,3,5-triazin-2-yl)morpholine, 12
3.3. Biology
3.3.1. In Vitro Antiproliferative Assay
4. Conclusion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sharma, A.; El-Faham, A.; de la Torre, B.G.; Albericio, F. Exploring the orthogonal chemoselectivity of 2,4,6-trichloro-1,3,5-triazine (TCT) as a trifunctional linker with different nucleophiles: Rules of the game. Front. Chem. 2018, 6, 516. [Google Scholar] [CrossRef]
- Huy, P.H.; Mbouhom., C. Formamide catalyzed activation of carboxylic acids–versatile and cost-efficient amidation and esterification. Chem. Sci. 2019, 10, 7399–7406. [Google Scholar] [CrossRef] [PubMed]
- Blotny, G. Recent applications of 2, 4, 6-trichloro-1, 3, 5-triazine and its derivatives in organic synthesis. Tetrahedron 2006, 62, 9507–9522. [Google Scholar] [CrossRef]
- Zhou, C.; Min, J.; Liu, Z.; Young, A.; Deshazer, H.; Gao, T.; Kallenbach, N.R. Synthesis and biological evaluation of novel 1, 3, 5-triazine derivatives as antimicrobial agents. Bioorg. Med. Chem. Lett. 2008, 18, 1308–1311. [Google Scholar] [CrossRef] [PubMed]
- Bahar, A.A.; Liu, Z.; Garafalo, M.; Kallenbach, N.; Ren, D. Controlling persister and biofilm cells of gram-negative bacteria with a new 1, 3, 5-triazine derivative. Pharmaceuticals 2015, 8, 696–710. [Google Scholar] [CrossRef] [PubMed]
- Sarmah, K.N.; Sarmah, N.K.; Kurmi, K.B.; Patel, T.V. Synthesis and studies of antifungal activity of 2, 4, 6-trisubstituted 1, 3, 5-triazines. Adv. Appl. Sci. Res. 2012, 3, 1459–1462. [Google Scholar]
- Ng, H.L.; Ma, X.; Chew, E.H.; Chui, W.K. Design, synthesis, and biological evaluation of coupled bioactive scaffolds as potential anticancer agents for dual targeting of dihydrofolate reductase and thioredoxin reductase. J. Med. Chem. 2017, 60, 1734–1745. [Google Scholar] [CrossRef]
- Nishimura, N.; Kato, A.; Isamu, M. Synthesis of pyrrolo[2,1-f][1,2,4]triazine C-nucleosides. Isosteres of sangivamycin, tubercidin, and toyocamycin. Carbohydr. Res. 2001, 331, 77–82. [Google Scholar] [CrossRef]
- Zacharie, B.; Abbott, S.D.; Bienvenu, J.F.; Cameron, A.D.; Cloutier, J.; Duceppe, J.S.; Ezzitouni, A.; Fortin, D.; Houde, K.; Lauzon, C. 2, 4, 6-trisubstituted triazines as protein a mimetics for the treatment of autoimmune diseases. J. Med. Chem. 2010, 53, 1138–1145. [Google Scholar] [CrossRef]
- Klenke, B.; Stewart, M.; Barrett, M.P.; Brun, R.; Gilbert, I.H. Synthesis and biological evaluation of s-triazine substituted polyamines as potential new anti-trypanosomal drugs. J. Med. Chem. 2001, 44, 3440–3452. [Google Scholar] [CrossRef]
- Mibu, N.; Yokomizo, K.; Aki, H.; Ota, N.; Fujii, H.; Yuzuriha, A.; Saneyoshi, S.; Tanaka, A.; Koga, A.; Zhou, J. Synthesis and antiviral evaluation of some C3-symmetrical trialkoxy substituted 1, 3, 5-triazines and their molecular geometry. Chem. Pharm. Bull. 2015, 63, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.R.; Modh, R.P.; Chikhalia, K.H. Privileged s-triazines: Structure and pharmacological applications. Future. Med. Chem. 2014, 6, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Sun, T.; Zhou, Z.; Du, L. A systematic review on antitumor agents with 1, 3, 5-triazines. Med. Chem. 2015, 5, 131–148. [Google Scholar] [CrossRef]
- Zhao, H.; Liu, Y.; Cui, Z.; Beattie, D.; Gu, Y.; Wang, Q. Design, synthesis, and biological activities of arylmethylamine substituted chlorotriazine and methylthiotriazine compounds. J. Agric. Food. Chem. 2011, 59, 11711–11717. [Google Scholar] [CrossRef]
- Hu, Z.; Ma, T.; Chen, Z.; Ye, Z.; Zhang, G.; Lou, Y.; Yu, Y. Solid-phase synthesis and antitumor evaluation of 2, 4-diamino-6-aryl-1, 3, 5-triazines. J. Comb. Chem. 2009, 11, 267–273. [Google Scholar] [CrossRef]
- Zheng, M.; Xu, C.; Ma, J.; Sun, Y.; Du, F.; Liu, H.; Lin, L.; Li, C.; Ding, J.; Chen, K. Synthesis and antitumor evaluation of a novel series of triamino-triazine derivatives. Bioorg. Med. Chem. 2007, 15, 1815–1827. [Google Scholar] [CrossRef]
- Sączewski, F.; Bułakowska, A.; Bednarski, P.; Grunert, R. Synthesis, structure and anticancer activity of novel 2, 4-diamino-1, 3, 5-triazine derivatives. Eur. J. Med. Chem. 2006, 41, 219–225. [Google Scholar] [CrossRef]
- Sączewski, F.; Bułakowska, A. Synthesis, structure and anticancer activity of novel alkenyl-1, 3, 5-triazine derivatives. Eur. J. Med. Chem. 2006, 41, 611–615. [Google Scholar] [CrossRef]
- Perspicace, E.; Jouan-Hureaux, V.; Ragno, R.; Ballante, F.; Sartini, S.; La Motta, C.; Da Settimo, F.; Chen, B.; Kirsch, G.; Schneider, S. Design, synthesis and biological evaluation of new classes of thieno [3, 2-d] pyrimidinone and thieno [1,2,3] triazine as inhibitor of vascular endothelial growth factor receptor-2 (VEGFR-2). Eur. J. Med. Chem. 2013, 63, 765–781. [Google Scholar] [CrossRef]
- Srivastava, G.K.; Alonso-Alonso, M.L.; Fernandez-Bueno, I.; Garcia-Gutierrez, M.T.; Rull, F.; Medina, J.; Coco, R.M.; Pastor, J.C. Comparison between direct contact and extract exposure methods for PFO cytotoxicity evaluation. Sci. Rep. 2018, 8, 1425. [Google Scholar] [CrossRef]
- Menear, K.A.; Gomez, S.; Malagu, K.; Bailey, C.; Blackburn, K.; Cockcroft, X.L.; Sebastian, L.; Ewen, S.; Fundo, A.; le Gall, A.; et al. Identification and optimisation of novel and selective small molecular weight kinase inhibitors of mTOR. Bioorg. Med. Chem. Lett. 2009, 19, 5898–5901. [Google Scholar] [CrossRef] [PubMed]
- Bai, F.; Liu, H.; Tong, L.; Zhou, W.; Liu, L.; Zhao, Z.; Liu, X.; Jiang, H.; Wang, X.; Xie, H. Discovery of novel selective inhibitors for EGFR-T790M/L858R. Bioorg. Med. Chem. Lett. 2012, 22, 1365–1370. [Google Scholar] [CrossRef] [PubMed]
- El-Faham, A.; Soliman, S.M.; Ghabbour, H.A.; Elnakady, Y.A.; Mohaya, T.A.; Siddiqui, M.R.; Albericio, F. Ultrasonic promoted synthesis of novel s-triazine-Schiff base derivatives; molecular structure, spectroscopic studies and their preliminary anti-proliferative activities. J. Mol. Str. 2016, 1125, 121–135. [Google Scholar] [CrossRef]
- Barakat, A.; El-Senduny, F.F.; Almarhoon, Z.; Al-Rasheed, H.H.; Badria, F.A.; Al-Majid, A.M.; El-Faham, A. Synthesis, X-Ray Crystal Structures, and Preliminary Antiproliferative Activities of New s-Triazine-hydroxybenzylidene Hydrazone Derivatives. J. Chem. 2019, 2019. [Google Scholar] [CrossRef]
- Kaur, S.; Kumari, P.; Singh, G.; Bhatti, R.; Singh, P. Design and synthesis of aza-/oxa heterocycle-based conjugates as novel anti-Inflammatory agents targeting cyclooxygenase-2. ACS Omega 2018, 3, 5825–5845. [Google Scholar] [CrossRef]
- Sharma, A.; Ghabbour, H.; Khan, S.T.; Beatriz, G.; Albericio, F.; El-Faham, A. Novel pyrazolyl-s-triazine derivatives, molecular structure and antimicrobial activity. J. Mol. Str. 2017, 1145, 244–253. [Google Scholar] [CrossRef]
- El-Faham, A.; Elnakady, Y. Synthesis, characterization of novel morpholino-1, 3, 5-triazinyl amino acid Ester derivatives and their anti-proliferation activities. Lett. Org. Chem. 2015, 12, 753–758. [Google Scholar] [CrossRef]
- Gillis, E.P.; Eastman, K.J.; Hill, M.D.; Donnelly, D.J.; Meanwell, N.A. Applications of fluorine in medicinal chemistry. J. Med. Chem. 2015, 58, 8315–8359. [Google Scholar] [CrossRef]
- Swallow, S. Fluorine in medicinal chemistry. In Progress in Medicinal Chemistry, 1st ed.; Witty, D.R., Lawton, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 54, pp. 65–133. [Google Scholar] [CrossRef]
- Al-Rasheed, H.H.; Al Alshaikh, M.; Khaled, J.M.; Alharbi, N.S.; El-Faham, A. Ultrasonic irradiation: Synthesis, characterization, and preliminary antimicrobial activity of novel series of 4, 6-disubstituted-1, 3, 5-triazine containing hydrazone derivatives. J. Chem. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds.are not available from the authors. |
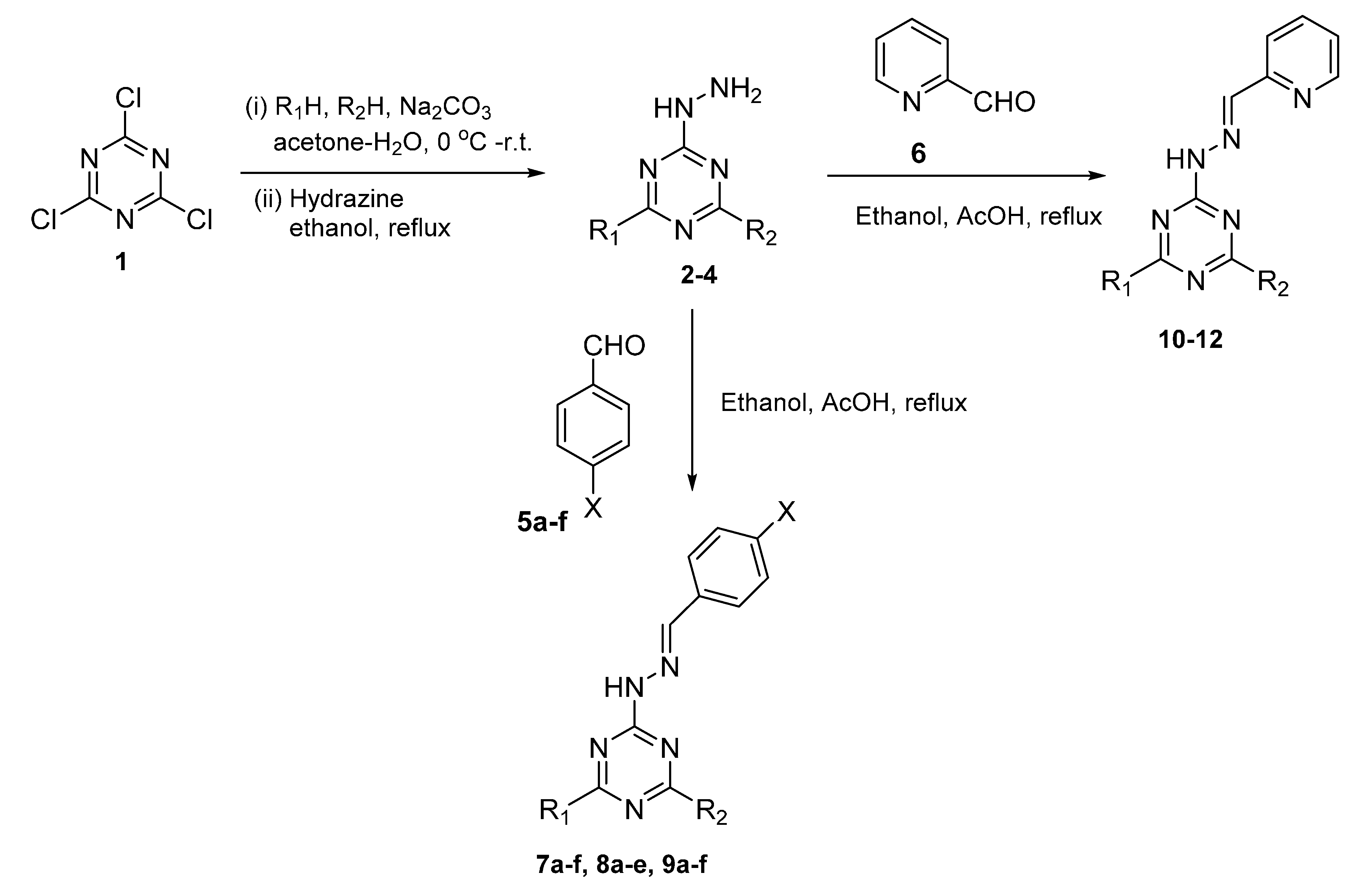
| Cpd No. | R1 | R2 | X | Cpd No. | R1 | R2 | X |
|---|---|---|---|---|---|---|---|
| 7a | piperidine | piperidine | H | 9a | morpholine | piperidine | H |
| 7b | piperidine | piperidine | Cl | 9b | morpholine | piperidine | Cl |
| 7c | piperidine | piperidine | Br | 9c | morpholine | piperidine | Br |
| 7d | piperidine | piperidine | OH | 9d | morpholine | piperidine | OH |
| 7e | piperidine | piperidine | F | 9e | morpholine | piperidine | F |
| 7f | piperidine | piperidine | OCH3 | 9f | morpholine | piperidine | OCH3 |
| 8a | morpholine | morpholine | H | 10 | piperidine | piperidine | |
| 8b | morpholine | morpholine | Cl | 11 | morpholine | morpholine | |
| 8c | morpholine | morpholine | Br | 12 | morpholine | piperidine | |
| 8d | morpholine | morpholine | OH | ||||
| 8e | morpholine | morpholine | F |
| Compound No. | IC50 (μM) MCF-7 | IC50 (μM) HCT-116 |
|---|---|---|
| 7a | 17.5 ± 5.9 | 12.6 ± 4.6 |
| 7b | 23.5 ± 8.0 | 10.8 ± 3.5 |
| 7c | 39.8 ± 12.9 | 10.9 ± 3.6 |
| 7d | 18.2 ± 5.8 | 19.5 ± 6.2 |
| 7e | 11.5 ± 3.3 | 14.0 ± 4.4 |
| 7f | 39.9 ± 12.7 | 8.8 ± 2.5 |
| 8a | 29.3 ± 9.3 | >50 |
| 8b | 17.6 ± 5.7 | 19.2 ± 6.2 |
| 8c | 23.2 ± 7.3 | 38.7 ± 12.4 |
| 8d | 14.0 ± 4.8 | 29.9 ± 9.5 |
| 8e | 13.4 ± 4.0 | 18.3 ± 5.7 |
| 9a | 22.2 ± 7.9 | 44.2 ± 14.4 |
| 9b | 21.9 ± 6.0 | 30.0 ± 9.6 |
| 9c | 22.5 ± 3.9 | 28.2 ± 9.0 |
| 9d | 10.4 ± 3.1 | 25.4 ± 8.2 |
| 9e | 13.9 ± 4.7 | 22.0 ± 7.1 |
| 9f | 14.2 ± 4.5 | 23.4 ± 7.4 |
| 10 | 32.8 ± 10.4 | >50 |
| 11 | 1.0 ± 0.3 | 0.98 ± 0.3 |
| 12 | 17.7 ± 5.5 | 30.4 ± 9.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
H. Al Rasheed, H.H.A.; M. Malebari, A.M.; A. Dahlous, K.A.; El-Faham, A. Synthesis and Characterization of New Series of 1,3-5-Triazine Hydrazone Derivatives with Promising Antiproliferative Activity. Molecules 2020, 25, 2708. https://doi.org/10.3390/molecules25112708
H. Al Rasheed HHA, M. Malebari AM, A. Dahlous KA, El-Faham A. Synthesis and Characterization of New Series of 1,3-5-Triazine Hydrazone Derivatives with Promising Antiproliferative Activity. Molecules. 2020; 25(11):2708. https://doi.org/10.3390/molecules25112708
Chicago/Turabian StyleH. Al Rasheed, Hessa H. Al, Azizah M. M. Malebari, Kholood A. A. Dahlous, and Ayman El-Faham. 2020. "Synthesis and Characterization of New Series of 1,3-5-Triazine Hydrazone Derivatives with Promising Antiproliferative Activity" Molecules 25, no. 11: 2708. https://doi.org/10.3390/molecules25112708
APA StyleH. Al Rasheed, H. H. A., M. Malebari, A. M., A. Dahlous, K. A., & El-Faham, A. (2020). Synthesis and Characterization of New Series of 1,3-5-Triazine Hydrazone Derivatives with Promising Antiproliferative Activity. Molecules, 25(11), 2708. https://doi.org/10.3390/molecules25112708