Activity Dependence of a Novel Lectin Family on Structure and Carbohydrate-Binding Properties
Abstract
1. Introduction
2. Novel Lectin Family Structure
3. Glycan Specificity
4. Effect on Bacteria and Fungi
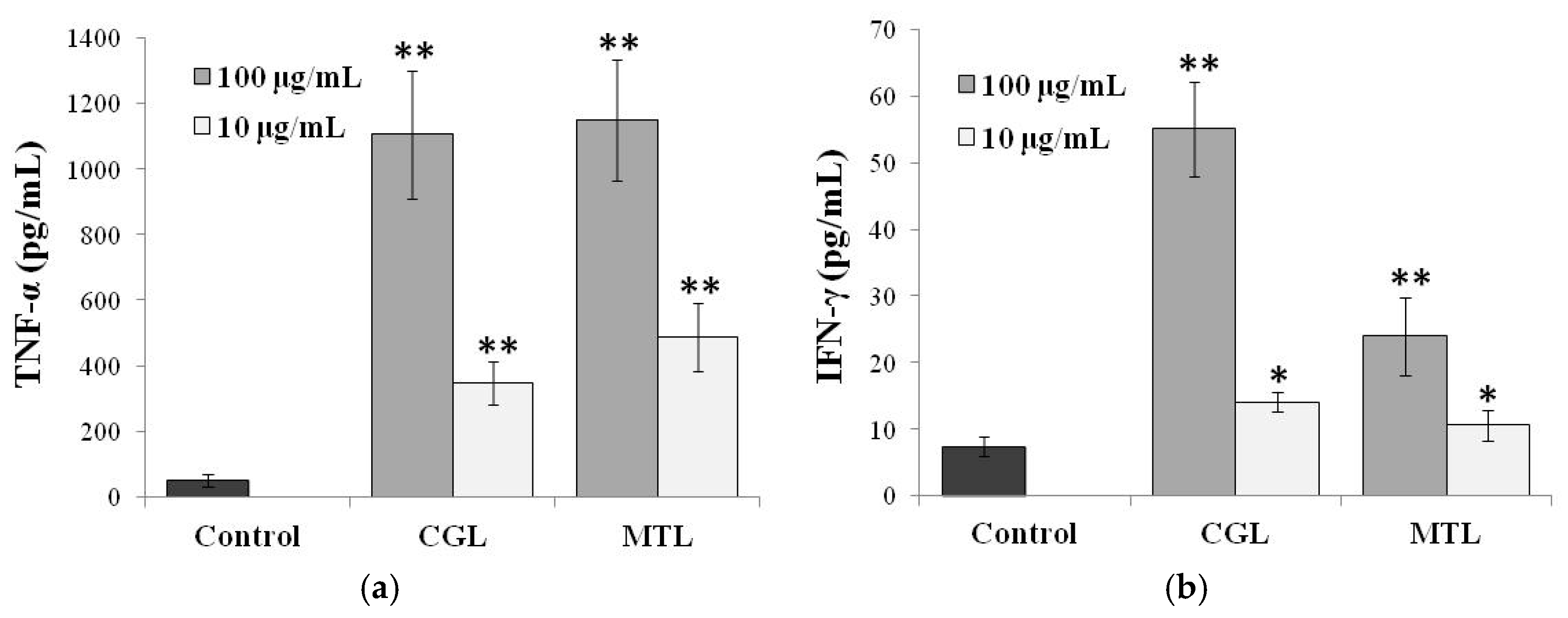
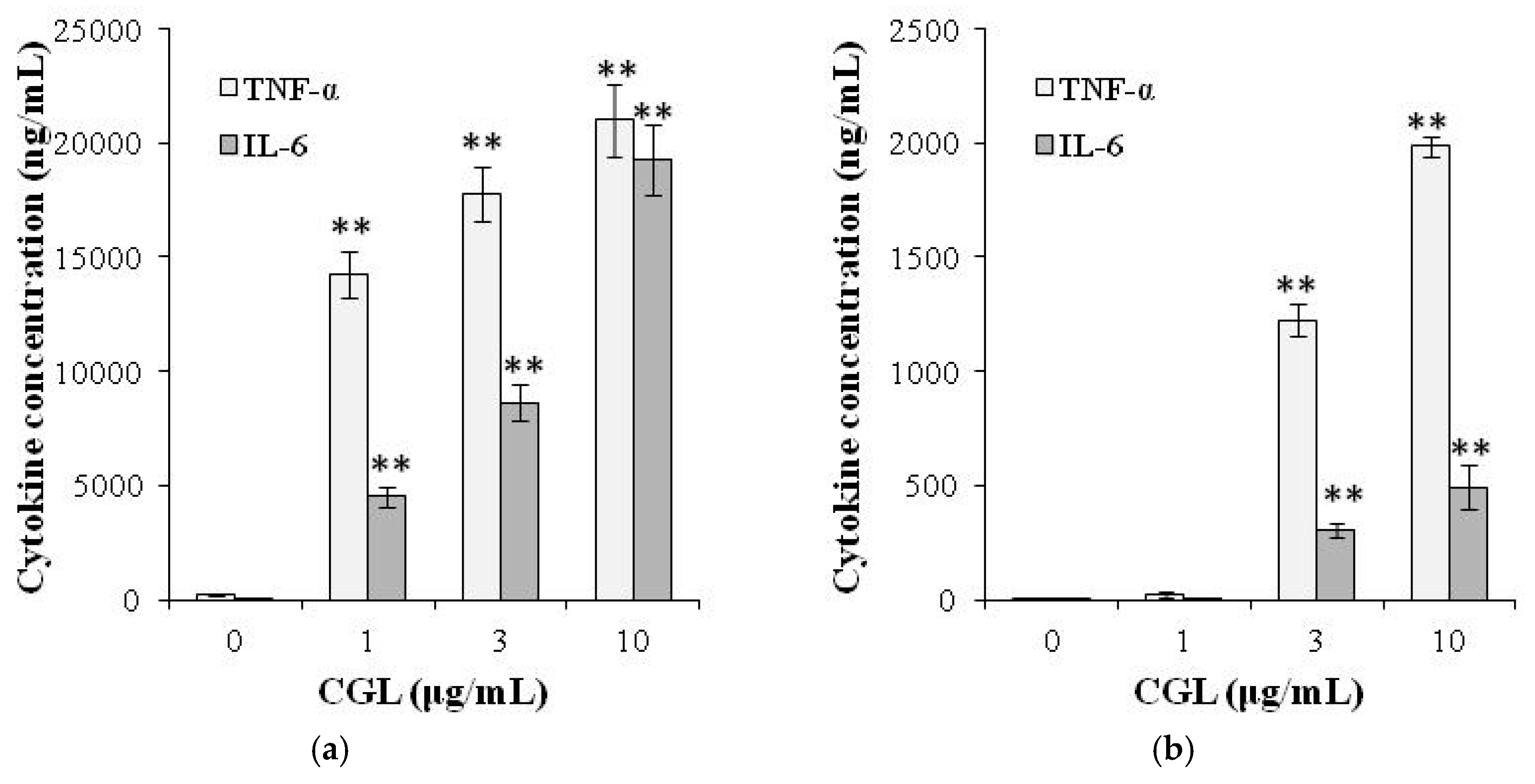
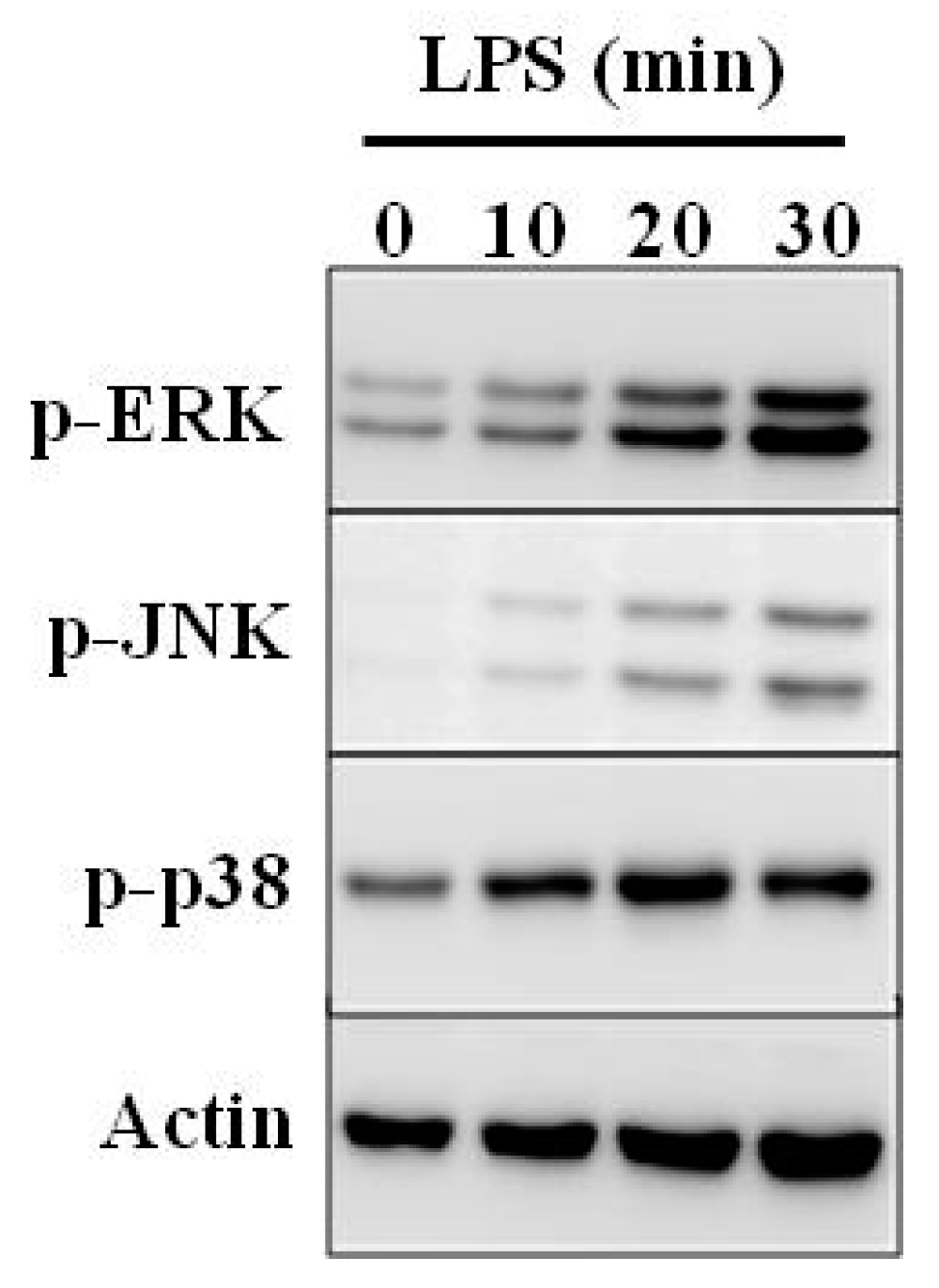
5. Immunomodulatory Activity
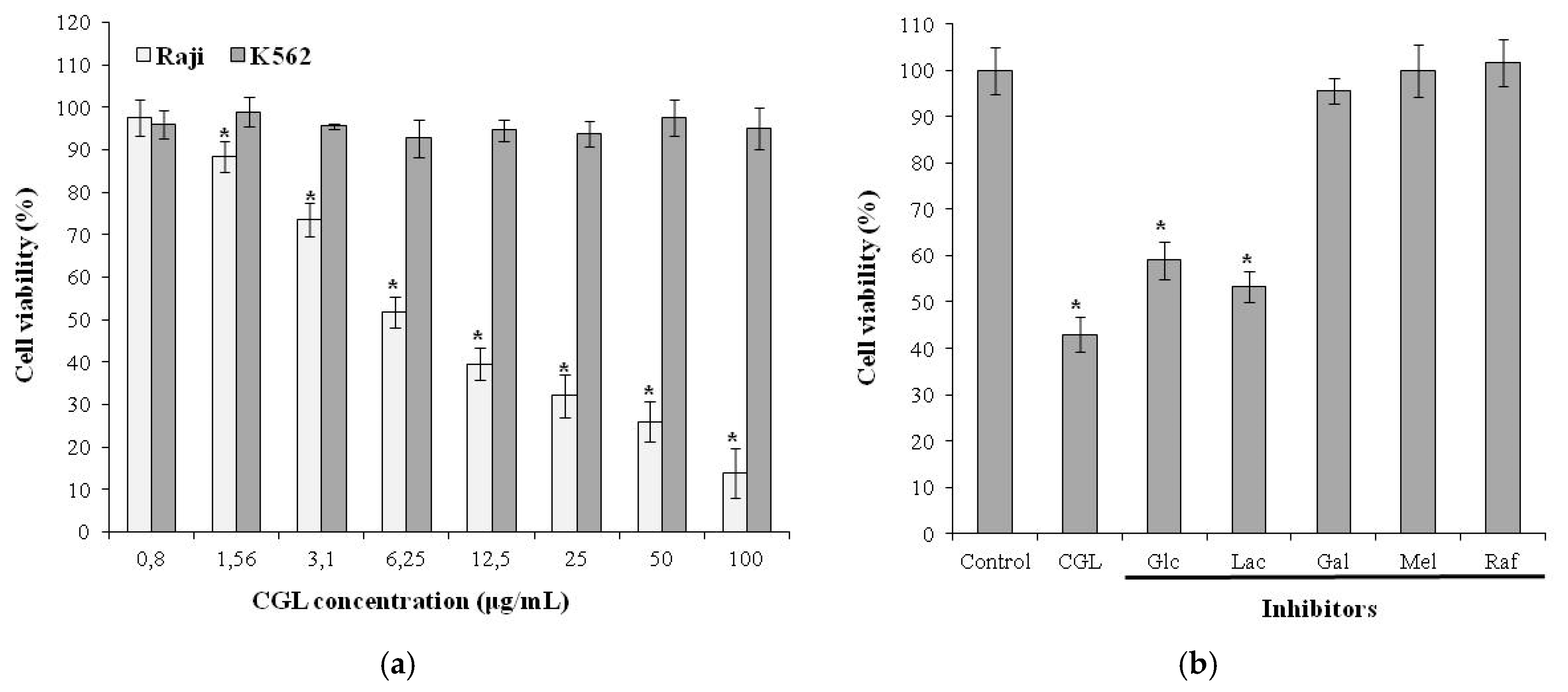
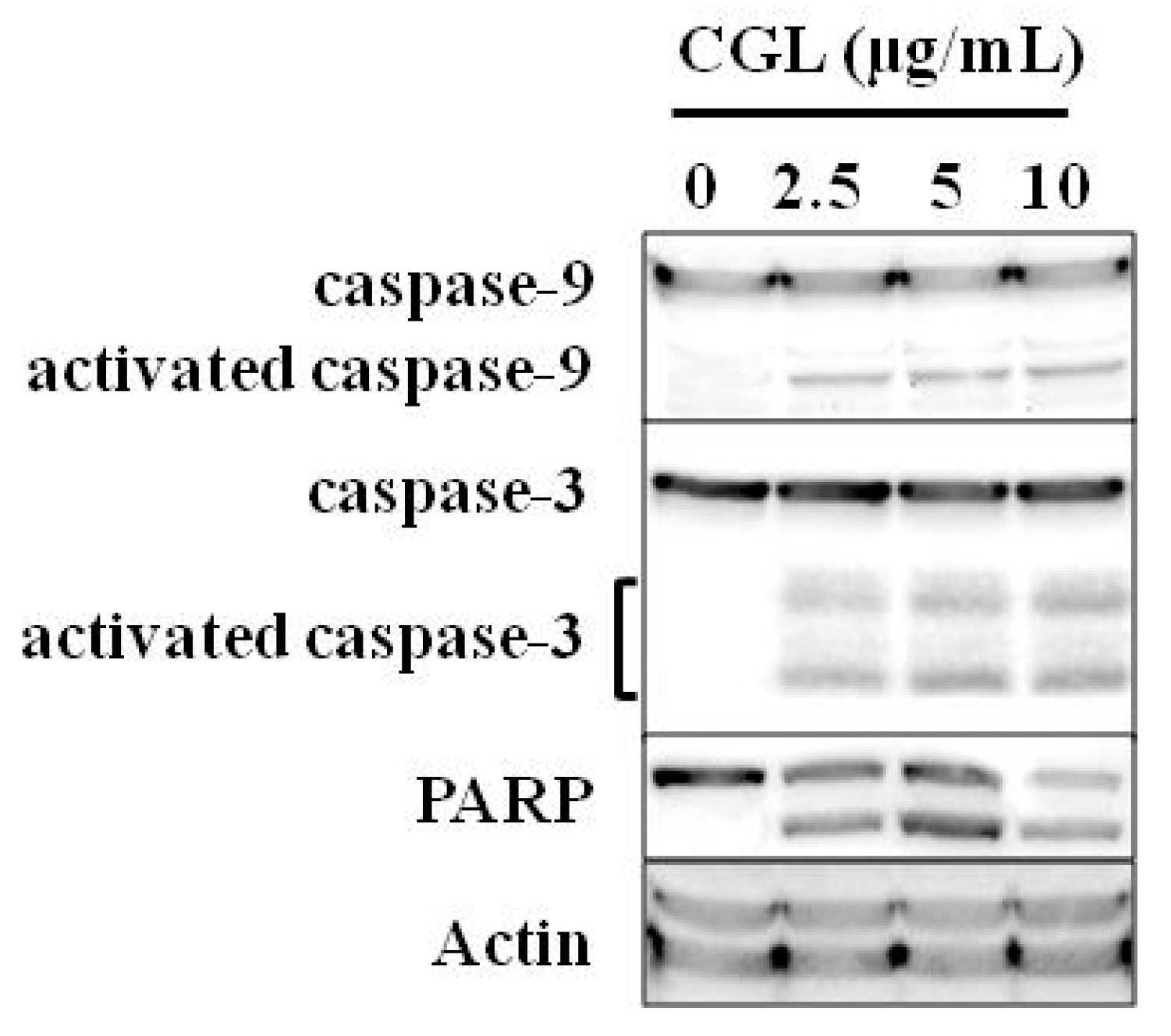
6. Effect on Tumor Cells
7. Conclusions
Funding
Conflicts of Interest
References
- Sharon, N.; Lis, H. Lectins; Springer: Dordrecht, The Netherlands, 2007; ISBN 978-1-4020-6605-4. [Google Scholar]
- McGreal, E.; Martinez-Pomares, L.; Gordon, S. Divergent roles for C-type lectins expressed by cells of the innate immune system. Mol. Immunol. 2004, 41, 1109–1121. [Google Scholar] [CrossRef] [PubMed]
- Sharon, N. Lectins: Carbohydrate-specific Reagents and Biological Recognition Molecules. J. Biol. Chem. 2007, 282, 2753–2764. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Luo, Y.; Zhou, T.-T.; Zhang, W.-Z. Could plant lectins become promising anti-tumour drugs for causing autophagic cell death? Cell Prolif. 2013, 46, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.R.D.; Mota, C.M.; Ribeiro, D.P.; Noleto, P.G.; Andrade, W.B.F.; Souza, M.A.; Silva, N.M.; Mineo, T.W.P.; Mineo, J.R.; Silva, D.A.O. Adjuvant and immunostimulatory effects of a D-galactose-binding lectin from Synadenium carinatum latex (ScLL) in the mouse model of vaccination against neosporosis. Vet. Res. 2012, 43, 76. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-H.; Kong, J.; Li, W.; Molchanova, V.; Chikalovets, I.; Belogortseva, N.; Luk’yanov, P.; Zheng, Y.-T. A beta-galactose-specific lectin isolated from the marine worm Chaetopterus variopedatus possesses anti-HIV-1 activity. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2006, 142, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Allen, H.J.; Kisailus, E.C. Glycoconjugates: Composition, Structure, and Function; Dekker: New York, NY, USA, 1992; ISBN 9780824784317. [Google Scholar]
- Chatterjee, B.P.; Adhya, M. Lectins with Varying Specificity and Biological Activity from Marine Bivalves. In Marine Proteins and Peptides; John Wiley & Sons, Ltd: Chichester, UK, 2013; pp. 41–68. ISBN 9781118375082. [Google Scholar]
- Adhya, M.; Singha, B. Gal/GalNAc specific multiple lectins in marine bivalve Anadara granosa. Fish Shellfish Immunol. 2016, 50, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Belogortseva, N.I.; Molchanova, V.I.; Kurika, A.V.; Skobun, A.S.; Glazkova, V.E. Isolation and characterization of new GalNAc/Gal-specific lectin from the sea mussel Crenomytilus grayanus. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1998, 119, 45–50. [Google Scholar] [CrossRef]
- Chikalovets, I.V.; Kovalchuk, S.N.; Litovchenko, A.P.; Molchanova, V.I.; Pivkin, M.V.; Chernikov, O.V. A new Gal/GalNAc-specific lectin from the mussel Mytilus trossulus: Structure, tissue specificity, antimicrobial and antifungal activity. Fish Shellfish Immunol. 2016, 50, 27–33. [Google Scholar] [CrossRef]
- Fujii, Y.; Dohmae, N.; Takio, K.; Kawsar, S.M.; Matsumoto, R.; Hasan, I.; Koide, Y.; Kanaly, R.A.; Yasumitsu, H.; Ogawa, Y.; et al. A lectin from the mussel Mytilus galloprovincialis has a highly novel primary structure and induces glycan-mediated cytotoxicity of globotriaosylceramide-expressing lymphoma cells. J. Biol. Chem. 2012, 287, 44772–44783. [Google Scholar] [CrossRef]
- Chikalovets, I.V.; Mizgina, T.O.; Molchanova, V.I.; Ovcharenko, Y.S.; Chernikov, O.V. Isolation and Characterization of Lectin from the Scallop Patinopecten yessoensis. Chem. Nat. Compd. 2017, 53, 717–721. [Google Scholar] [CrossRef]
- Hasan, I.; Gerdol, M.; Fujii, Y.; Rajia, S.; Koide, Y.; Yamamoto, D.; Kawsar, S.M.A.; Ozeki, Y. CDNA and Gene Structure of MytiLec-1, A Bacteriostatic R-Type Lectin from the Mediterranean Mussel (Mytilus galloprovincialis). Mar. Drugs 2016, 14, 92. [Google Scholar] [CrossRef] [PubMed]
- Kovalchuk, S.N.; Chikalovets, I.V.; Chernikov, O.V.; Molchanova, V.I.; Li, W.; Rasskazov, V.A.; Lukyanov, P.A. CDNA cloning and structural characterization of a lectin from the mussel Crenomytilus grayanus with a unique amino acid sequence and antibacterial activity. Fish Shellfish Immunol. 2013, 35, 1320–1324. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.D.; Coggill, P.; Eberhardt, R.Y.; Eddy, S.R.; Mistry, J.; Mitchell, A.L.; Potter, S.C.; Punta, M.; Qureshi, M.; Sangrador-Vegas, A.; et al. The Pfam protein families database: Towards a more sustainable future. Nucleic Acids Res. 2016, 44, D279–D285. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y.; Gerdol, M.; Hasan, I.; Koide, Y.; Matsuzaki, R.; Ikeda, M.; Rajia, S.; Ogawa, Y.; Kawsar, S.M.A.; Ozeki, Y. Phylogeny and Properties of a Novel Lectin Family with β-Trefoil Folding in Mussels. Trends Glycosci. Glycotechnol. 2018, 30, J155–J168. [Google Scholar] [CrossRef]
- Terada, D.; Kawai, F.; Noguchi, H.; Unzai, S.; Hasan, I.; Fujii, Y.; Park, S.-Y.; Ozeki, Y.; Tame, J.R.H. Crystal structure of MytiLec, a galactose-binding lectin from the mussel Mytilus galloprovincialis with cytotoxicity against certain cancer cell types. Sci. Rep. 2016, 6, 28344. [Google Scholar] [CrossRef]
- Liao, J.H.; Chien, C.T.; Wu, H.Y.; Huang, K.F.; Wang, I.; Ho, M.R.; Tu, I.F.; Lee, I.M.; Li, W.; Shih, Y.L.; et al. A Multivalent Marine Lectin from Crenomytilus grayanus Possesses Anti-cancer Activity through Recognizing Globotriose Gb3. J. Am. Chem. Soc. 2016, 138, 4787–4795. [Google Scholar] [CrossRef]
- Gerdol, M.; Venier, P. An updated molecular basis for mussel immunity. Fish Shellfish Immunol. 2015, 46, 17–38. [Google Scholar] [CrossRef]
- Chikalovets, I.V.; Kondrashina, A.S.; Chernikov, O.V.; Molchanova, V.I.; Luk’yanov, P.A. Isolation and general characteristics of lectin from the mussel Mytilus trossulus. Chem. Nat. Compd. 2013, 48, 1058–1061. [Google Scholar] [CrossRef]
- Chichvarkhin, A.I.; Kartavtsev, I.F.; Kafanov, A.I. Genetic connections between some species of Mytilidae (Mollusca: Bivalvia) from the northern part of the Pacific Ocean. Genetika 2000, 36, 1206–1220. [Google Scholar]
- Houser, J.; Komárek, J.; Kostlánová, N.; Cioci, G.; Imberty, A.; Wimmerová, M. Protein oligomerization in Aleuria aurantia lectin family—Importance and difficulties. Mater. Struct. Chem. Biol. Phys. Technol. 2012, 19, 20–21. [Google Scholar]
- Chikalovets, I.V.; Molchanova, V.I.; Chernikov, O.V.; Luk’Yanov, P.A. Domain organization of lectin from the mussel Crenomytilus grayanus. Chem. Nat. Compd. 2014, 50, 706–709. [Google Scholar] [CrossRef]
- Cho, M.; Cummings, R.D. Galectin-1, a beta-galactoside-binding lectin in Chinese hamster ovary cells. I. Physical and chemical characterization. J. Biol. Chem. 1995, 270, 5198–5206. [Google Scholar] [CrossRef] [PubMed]
- Terada, D.; Voet, A.R.D.; Noguchi, H.; Kamata, K.; Ohki, M.; Addy, C.; Fujii, Y.; Yamamoto, D.; Ozeki, Y.; Tame, J.R.H.; et al. Computational design of a symmetrical β-trefoil lectin with cancer cell binding activity. Sci. Rep. 2017, 7, 5943. [Google Scholar] [CrossRef] [PubMed]
- Chernikov, O.; Kuzmich, A.; Chikalovets, I.; Molchanova, V.; Hua, K.-F. Lectin CGL from the sea mussel Crenomytilus grayanus induces Burkitt’s lymphoma cells death via interaction with surface glycan. Int. J. Biol. Macromol. 2017, 104, 508–514. [Google Scholar] [CrossRef]
- Oyelaran, O.; Gildersleeve, J.C. Glycan arrays: Recent advances and future challenges. Curr. Opin. Chem. Biol. 2009, 13, 406–413. [Google Scholar] [CrossRef]
- Cheung, R.C.; Wong, J.H.; Pan, W.; Chan, Y.S.; Yin, C.; Dan, X.; Ng, T.B. Marine lectins and their medicinal applications. Appl. Microbiol. Biotechnol. 2015, 99, 3755–3773. [Google Scholar] [CrossRef]
- Toubiana, M.; Gerdol, M.; Rosani, U.; Pallavicini, A.; Venier, P.; Roch, P. Toll-like receptors and MyD88 adaptors in Mytilus: Complete cds and gene expression levels. Dev. Comp. Immunol. 2013, 40, 158–166. [Google Scholar] [CrossRef]
- Kubler-Kielb, J.; Lai, W.-T.; Schneerson, R.; Vinogradov, E. The structure of the Escherichia coli O148 lipopolysaccharide core region and its linkage to the O-specific polysaccharide. Carbohydr. Res. 2011, 346, 150–152. [Google Scholar] [CrossRef]
- Poxton, I.R. Teichoic Acids, Lipoteichoic Acids and Other Secondary Cell Wall and Membrane Polysaccharides of Gram-Positive Bacteria. In Molecular Medical Microbiology; Academic Press: Cambridge, MA, USA, 2015; pp. 91–103. ISBN 9780123971692. [Google Scholar]
- Vollmer, W. Peptidoglycan. In Molecular Medical Microbiology; Academic Press: Cambridge, MA, USA, 2015; pp. 105–124. ISBN 9780123971692. [Google Scholar]
- Gaudet, R.G.; Sintsova, A.; Buckwalter, C.M.; Leung, N.; Cochrane, A.; Li, J.; Cox, A.D.; Moffat, J.; Gray-Owen, S.D. Cytosolic detection of the bacterial metabolite HBP activates TIFA-dependent innate immunity. Science 2015, 348, 1251–1255. [Google Scholar] [CrossRef]
- Wollin, R.; Creeger, E.S.; Rothfield, L.I.; Stocker, B.A.; Lindberg, A.A. Salmonella typhimurium mutants defective in UDP-D-galactose:lipopolysaccharide alpha 1,6-D-galactosyltransferase. Structural, immunochemical, and enzymologic studies of rfaB mutants. J. Biol. Chem. 1983, 258, 3769–3774. [Google Scholar]
- Klein, R.A.; Hartmann, R.; Egge, H.; Behr, T.; Fischer, W. The aqueous solution structure of a lipoteichoic acid from Streptococcus pneumoniae strain R6 containing 2,4-diamino-2,4,6-trideoxy-galactose: Evidence for conformational mobility of the galactopyranose ring. Carbohydr. Res. 1996, 281, 79–98. [Google Scholar] [CrossRef]
- Valiente, E.; Jiménez, N.; Merino, S.; Tomás, J.M.; Amaro, C. Vibrio vulnificus biotype 2 serovar E gne but not galE is essential for lipopolysaccharide biosynthesis and virulence. Infect. Immun. 2008, 76, 1628–1638. [Google Scholar] [CrossRef] [PubMed]
- Maldonado-Aguayo, W.; Teneb, J.; Gallardo-Escárate, C. A galectin with quadruple-domain from red abalone Haliotis rufescens involved in the immune innate response against to Vibrio anguillarum. Fish Shellfish Immunol. 2014, 40, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Shen, H.; Zhou, H.; Dong, Y.; Lin, Z. A tandem-repeat galectin from blood clam Tegillarca granosa and its induced mRNA expression response against bacterial challenge. Genes Genom. 2013, 35, 733–740. [Google Scholar] [CrossRef]
- Moreira, R.; Balseiro, P.; Romero, A.; Dios, S.; Posada, D.; Novoa, B.; Figueras, A. Gene expression analysis of clams Ruditapes philippinarum and Ruditapes decussatus following bacterial infection yields molecular insights into pathogen resistance and immunity. Dev. Comp. Immunol. 2012, 36, 140–149. [Google Scholar] [CrossRef]
- Chikalovets, I.V.; Chernikov, O.V.; Pivkin, M.V.; Molchanova, V.I.; Litovchenko, A.P.; Li, W.; Lukyanov, P.A. A lectin with antifungal activity from the mussel Crenomytilus grayanus. Fish Shellfish Immunol. 2015, 42, 503–507. [Google Scholar] [CrossRef]
- Van Parijs, J.; Joosen, H.M.; Peumans, W.J.; Geuns, J.M.; Van Laere, A.J. Effect of the Urtica dioica agglutinin on germination and cell wall formation of Phycomyces blakesleeanus Burgeff. Arch. Microbiol. 1992, 158, 19–25. [Google Scholar] [CrossRef]
- Zvereva, L.V.; Vysotskaya, M.A. Filamentous Fungi Associated with Bivalve Mollusks from Polluted Biotopes of Ussuriiskii Bay, Sea of Japan. Russ. J. Mar. Biol. 2005, 31, 382–385. [Google Scholar] [CrossRef]
- Kang, H.K.; Lee, H.H.; Seo, C.H.; Park, Y. Antimicrobial and immunomodulatory properties and applications of marine-derived proteins and peptides. Mar. Drugs 2019, 17, 350. [Google Scholar] [CrossRef]
- Malagoli, D.; Sacchi, S.; Ottaviani, E. Lectins and cytokines in celomatic invertebrates: Two tales with the same end. Invertebr. Surviv. J. 2010, 7, 1–10. [Google Scholar]
- Hosono, M.; Sugawara, S.; Matsuda, A.; Tatsuta, T.; Koide, Y.; Hasan, I.; Ozeki, Y.; Nitta, K. Binding profiles and cytokine-inducing effects of fish rhamnose-binding lectins on Burkitt’s lymphoma Raji cells. Fish Physiol. Biochem. 2014, 40, 1559–1572. [Google Scholar] [CrossRef] [PubMed]
- Yamanishi, T.; Yamamoto, Y.; Hatakeyama, T.; Yamaguchi, K.; Oda, T. CEL-I, an invertebrate N-acetylgalactosamine-specific C-type lectin, induces TNF-alpha and G-CSF production by mouse macrophage cell line RAW264.7 cells. J. Biochem. 2007, 142, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.H.; Mun, S.H.; Ko, N.Y.; Lee, J.H.; Jun, M.H.; Seo, J.Y.; Kim, Y.M.; Choi, W.S.; Her, E. The synergistic effect of phytohemagglutinin and interferon-gamma on the expression of tumor necrosis factor-alpha from RAW 264.7 cells. Immunol. Lett. 2005, 98, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Chernikov, O.V.; Wong, W.-T.; Li, L.-H.; Chikalovets, I.V.; Molchanova, V.I.; Wu, S.-H.; Liao, J.-H.; Hua, K.-F. A GalNAc/Gal-specific lectin from the sea mussel Crenomytilus grayanus modulates immune response in macrophages and in mice. Sci. Rep. 2017, 7, 6315. [Google Scholar] [CrossRef] [PubMed]
- Lambeth, J.D. NOX enzymes and the biology of reactive oxygen. Nat. Rev. Immunol. 2004, 4, 181–189. [Google Scholar] [CrossRef]
- West, A.P.; Brodsky, I.E.; Rahner, C.; Woo, D.K.; Erdjument-Bromage, H.; Tempst, P.; Walsh, M.C.; Choi, Y.; Shadel, G.S.; Ghosh, S. TLR signalling augments macrophage bactericidal activity through mitochondrial ROS. Nature 2011, 472, 476–480. [Google Scholar] [CrossRef]
- Fan, H.; Cook, J.A. Molecular mechanisms of endotoxin tolerance. J. Endotoxin Res. 2004, 10, 71–84. [Google Scholar] [CrossRef]
- Wheeler, D.S.; Lahni, P.M.; Denenberg, A.G.; Poynter, S.E.; Wong, H.R.; Cook, J.A.; Zingarelli, B. Induction of endotoxin tolerance enhances bacterial clearance and survival in murine polymicrobial sepsis. Shock 2008, 30, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Furtak, V.A.; Kurika, A.V.; Belogortseva, N.I.; Chikalovets, I.V.; Kleshch, Y. Cell localization of mucin-type receptors assayed with novel GalNac/Gal-specific lectin from sea mussel Crenomytilus grayanus in human colon tumors. Bull. Exp. Biol. Med. 1999, 128, 1039–1041. [Google Scholar] [CrossRef]
- Nudelman, E.; Kannagi, R.; Hakomori, S.; Parsons, M.; Lipinski, M.; Wiels, J.; Fellous, M.; Tursz, T. A glycolipid antigen associated with Burkitt lymphoma defined by a monoclonal antibody. Science 1983, 220, 509–511. [Google Scholar] [CrossRef]
- Cao, Y.; Merling, A.; Karsten, U.; Goletz, S.; Punzel, M.; Kraft, R.; Butschak, G.; Schwartz-Albiez, R. Expression of CD175 (Tn), CD175s (sialosyl-Tn) and CD176 (Thomsen-Friedenreich antigen) on malignant human hematopoietic cells. Int. J. Cancer 2008, 123, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Geiger, P.; Mayer, B.; Wiest, I.; Schulze, S.; Jeschke, U.; Weissenbacher, T. Binding of galectin-1 to breast cancer cells MCF7 induces apoptosis and inhibition of proliferation in vitro in a 2D- and 3D- cell culture model. BMC Cancer 2016, 16, 870. [Google Scholar] [CrossRef]
- Freire, T.; Bay, S.; von Mensdorff-Pouilly, S.; Osinaga, E. Molecular Basis of Incomplete O-Glycan Synthesis in MCF-7 Breast Cancer Cells: Putative Role of MUC6 in Tn Antigen Expression. Cancer Res. 2005, 65, 7880–7887. [Google Scholar] [CrossRef]
- Hasan, I.; Sugawara, S.; Fujii, Y.; Koide, Y.; Terada, D.; Iimura, N.; Fujiwara, T.; Takahashi, K.G.; Kojima, N.; Rajia, S.; et al. MytiLec, a Mussel R-Type Lectin, Interacts with Surface Glycan Gb3 on Burkitt’s Lymphoma Cells to Trigger Apoptosis through Multiple Pathways. Mar. Drugs 2015, 13, 7377–7389. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.Y.; Yang, X. Proteases for cell suicide: Functions and regulation of caspases. Microbiol. Mol. Biol. Rev. 2000, 64, 821–846. [Google Scholar] [CrossRef] [PubMed]
- Stennicke, H.R.; Salvesen, G.S. Properties of the caspases. Biochim. Biophys. Acta 1998, 1387, 17–31. [Google Scholar] [CrossRef]
- Dias, N.; Bailly, C. Drugs targeting mitochondrial functions to control tumor cell growth. Biochem. Pharmacol. 2005, 70, 1–12. [Google Scholar] [CrossRef]


| Lectin | MW (Da) | Thermal Stability | pH Dependence | Metal Ion Dependence | Localization in Mussel Tissue | Reference |
|---|---|---|---|---|---|---|
| CGL | 16,953 | Stable until 50 °C | 8–10 | Not dependent | Mantle | [10,15] |
| MTL | 16,492 | Stable until 50 °C | 9–10 | Not dependent | Mantle | [11,21] |
| MytiLec-1 | 16,812 | N.D. 1 | N.D. | Not dependent | Mantle | [12] |
| Carbohydrate | Minimum Inhibitory Concentration, mM | ||
|---|---|---|---|
| CGL [10] | MTL [21] | MytiLec-1 [12] | |
| N-Acetyl-d-galactosamine | 1.4 | 0.7 | 1.6 |
| N-Acetyl-d-glucosamine | No inhibition at 80 | >50 | No inhibition at 100 |
| d-Galactose | 5.4 | 1.7 | 3.1 |
| d-Glucose | No inhibition at 80 | >50 | >50 |
| d-Fucose | No inhibition at 80 | >50 | >50 |
| d-Talose | 5.4 | N.D. 1 | 25 |
| Lactose | No inhibition at 80 | 29.2 | 50 |
| Melibiose | 2.0 | N.D. | 1.6 |
| Raffinose | 1.8 | N.D. | N.D. |
| Glycoproteins | Minimum Inhibitory Concentration, mg/mL | ||
| BSM | 0.007 | 0.0156 | No inhibition at 2 |
| Asialo-BSM | 0.0017 | N.D. | 0.2 |
| Fetuin | 2 | 0.0156 | No inhibition at 2 |
| Asialofetuin | 0.03 | 0.0156 | 0.2 |
| Glycan # | Compound | Fluorescence (%) 1 | |
|---|---|---|---|
| CGL [27] | MTL 2 | ||
| 559 | Galα1-3Galβ1-4GlcNAcβ1-2Mana1-6(Galα1-3Galβ1-4GlcNAcβ1-2Manα1-3)Manβ1-4GlcNAcβ1-4GlcNAc-Sp24 | 100 | 87.7 |
| 587 | Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-6(Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-2)Manα1-6(Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-2Manα1-3)Manβ1-4GlcNAcβ1-4(Fucα1-6)GlcNAcβ-Sp24 | 98.7 | 100 |
| 362 | Galα1-3Galβ1-4GlcNAcβ1-2Manα1-6(Galα1-3Galβ1-4GlcNAcβ1-2Manα1-3)Manβ1-4GlcNAcβ1-4GlcNAcβ-Sp20 | 96.4 | 91.5 |
| 402 | Galα1-4Galβ1-3GlcNAcβ1-2Manα1-6(Galα1-4Galβ1-3GlcNAcβ1-2Manα1-3)Manβ1-4GlcNAcβ1-4GlcNAcβ-Sp19 | 78.2 | 72.5 |
| 583 | Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-6(Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-2)Manα1-6(Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-3Galβ1-4GlcNAcβ1-2Manα1-3)Manβ1-4GlcNAcβ1-4(Fucα1-6)GlcNAcβ-Sp24 | 66.5 | 72.4 |
| 122 | Galα1-4Galβ1-4Glcβ-Sp0 | 24.5 | 23.3 |
| 72 | Fucα1-2Galβ1-4(Fucα1-3)GlcNAcβ-Sp8 | 0.15 | 0.03 |
| Bacteria | Binding Activity 1 | Agglutination 2 | Growth Suppressive Activity (%) | |||||
|---|---|---|---|---|---|---|---|---|
| CGL | MTL | CGL | MTL | MytiLec-1 | CGL | MTL | MytiLec-1 | |
| Candida albicans | 0.65 ± 0.01 | 0.44 ± 0.05 | ++ | +++ | N.D. 3 | - 4 | - | N.D. |
| Vibrio proteolyticus | 1.42 ± 0.04 | 0.37 ± 0.14 | +++ | ++ | N.D. | 39.9 ± 5 | - | N.D. |
| Escherichia coli | 1.63 ± 0.09 | 0.48 ± 0.06 | ++ | + | ++ | 46 ± 5 | 10.6 ± 7 | 58 ± 5 |
| Bacillus subtilis | 0.74 ± 0.07 | 0.26 ± 0.07 | +++ | ++ | ++ | 85 ± 8 | 62 ± 6 | 74 ± 8 |
| Staphylococcus aureus | 0.39 ± 0.06 | 0.49 ± 0.01 | ++ | ++ | + | 68 ± 6 | - | 61 ± 36 |
| CGL (μg/mL) | CFU (×106) | Killed Bacteria (×106) | |
|---|---|---|---|
| 1 h after Infection | 24 h after Infection | ||
| 0 | 1680 ± 370 | 724 ± 196 | 956 |
| 10 | 1984 ± 792 | 448 ± 166 | 1536 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chikalovets, I.; Filshtein, A.; Molchanova, V.; Mizgina, T.; Lukyanov, P.; Nedashkovskaya, O.; Hua, K.-F.; Chernikov, O. Activity Dependence of a Novel Lectin Family on Structure and Carbohydrate-Binding Properties. Molecules 2020, 25, 150. https://doi.org/10.3390/molecules25010150
Chikalovets I, Filshtein A, Molchanova V, Mizgina T, Lukyanov P, Nedashkovskaya O, Hua K-F, Chernikov O. Activity Dependence of a Novel Lectin Family on Structure and Carbohydrate-Binding Properties. Molecules. 2020; 25(1):150. https://doi.org/10.3390/molecules25010150
Chicago/Turabian StyleChikalovets, Irina, Alina Filshtein, Valentina Molchanova, Tatyana Mizgina, Pavel Lukyanov, Olga Nedashkovskaya, Kuo-Feng Hua, and Oleg Chernikov. 2020. "Activity Dependence of a Novel Lectin Family on Structure and Carbohydrate-Binding Properties" Molecules 25, no. 1: 150. https://doi.org/10.3390/molecules25010150
APA StyleChikalovets, I., Filshtein, A., Molchanova, V., Mizgina, T., Lukyanov, P., Nedashkovskaya, O., Hua, K.-F., & Chernikov, O. (2020). Activity Dependence of a Novel Lectin Family on Structure and Carbohydrate-Binding Properties. Molecules, 25(1), 150. https://doi.org/10.3390/molecules25010150








