Eco-Friendly Ca-Montmorillonite Grafted by Non-Acidic Ionic Liquid Used as A Solid Acid Catalyst in Cellulose Hydrolysis to Reducing Sugars
Abstract
1. Introduction
2. Results and Discussion
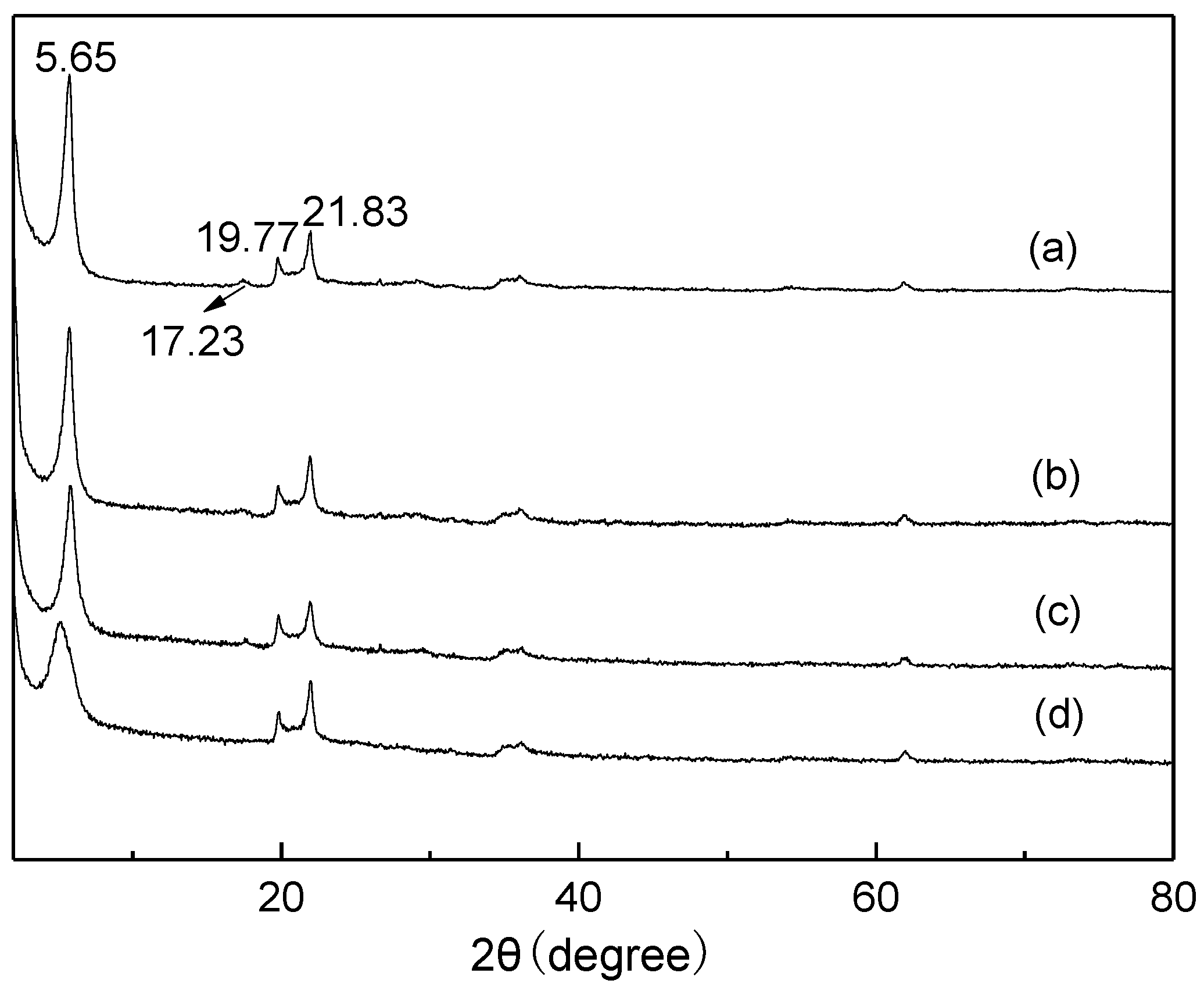
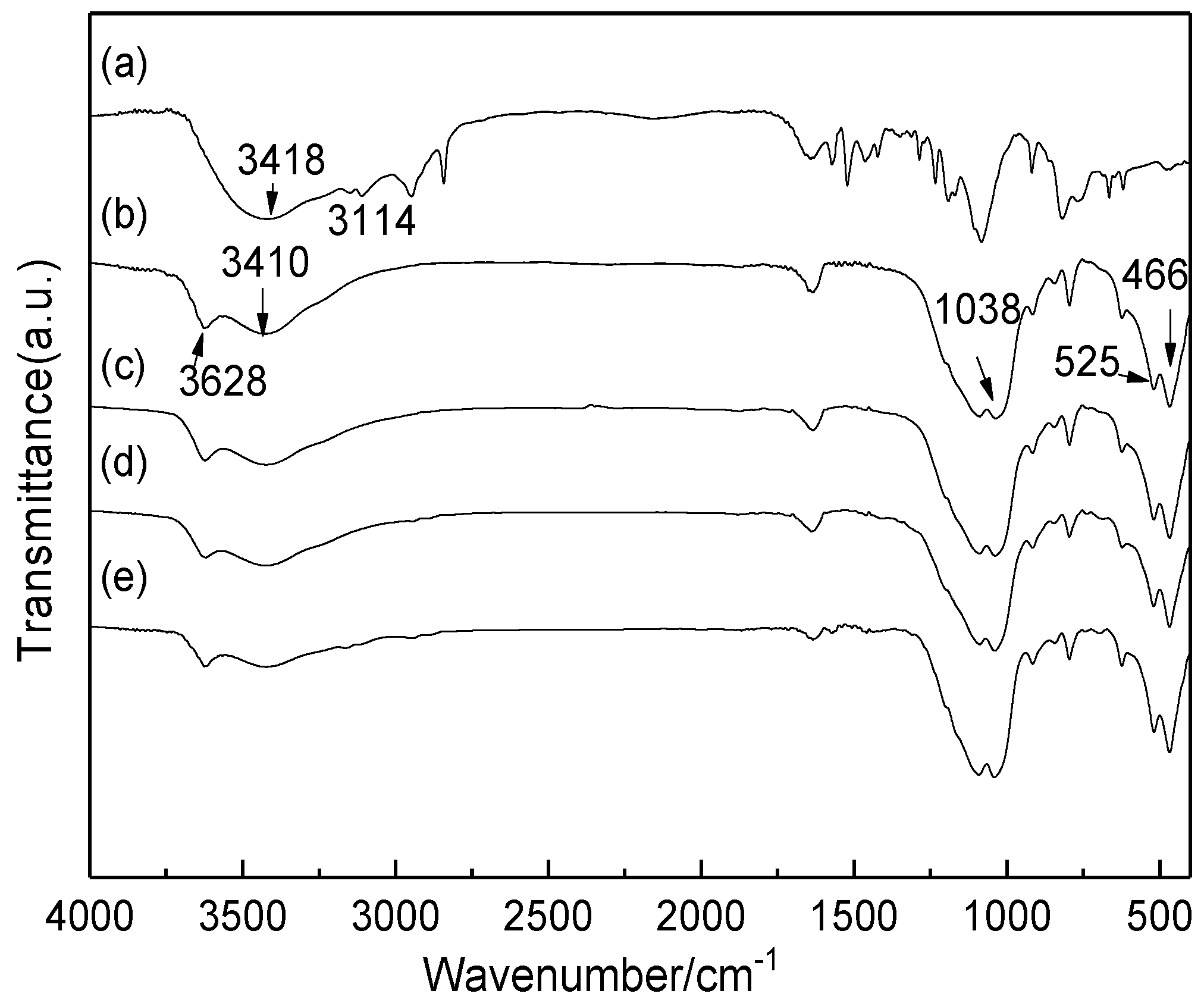
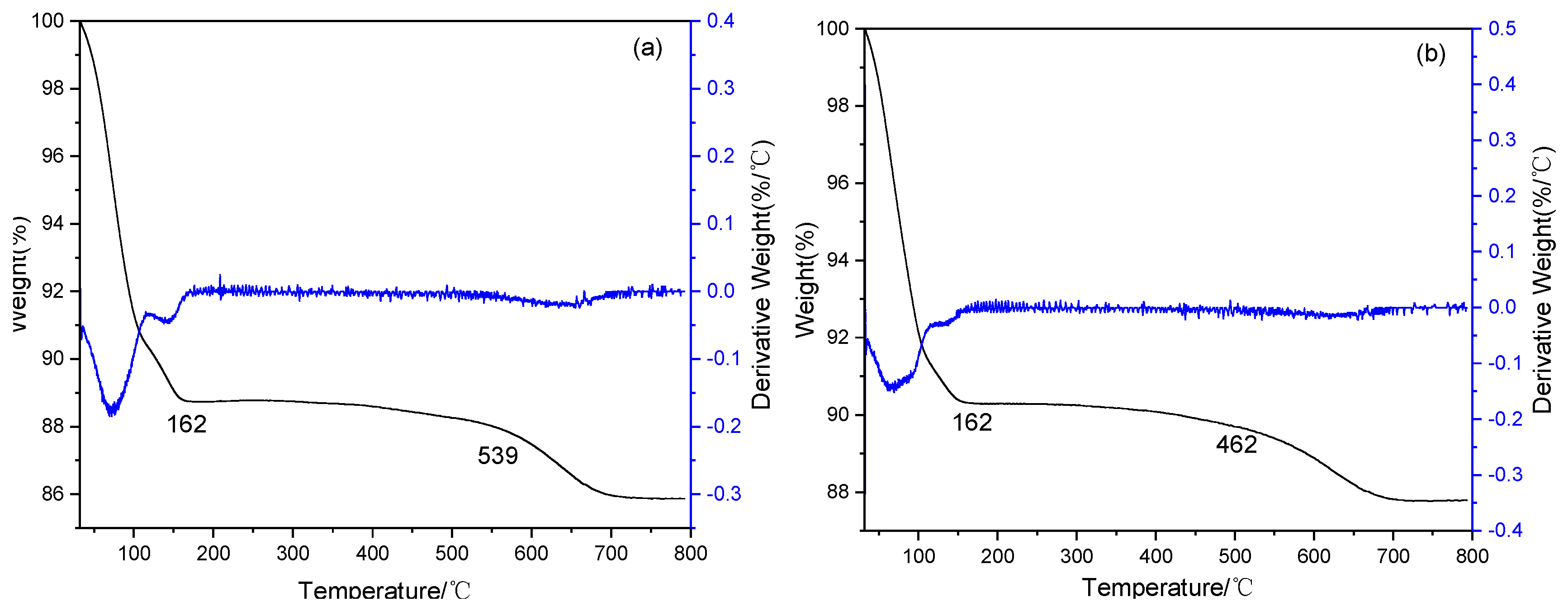
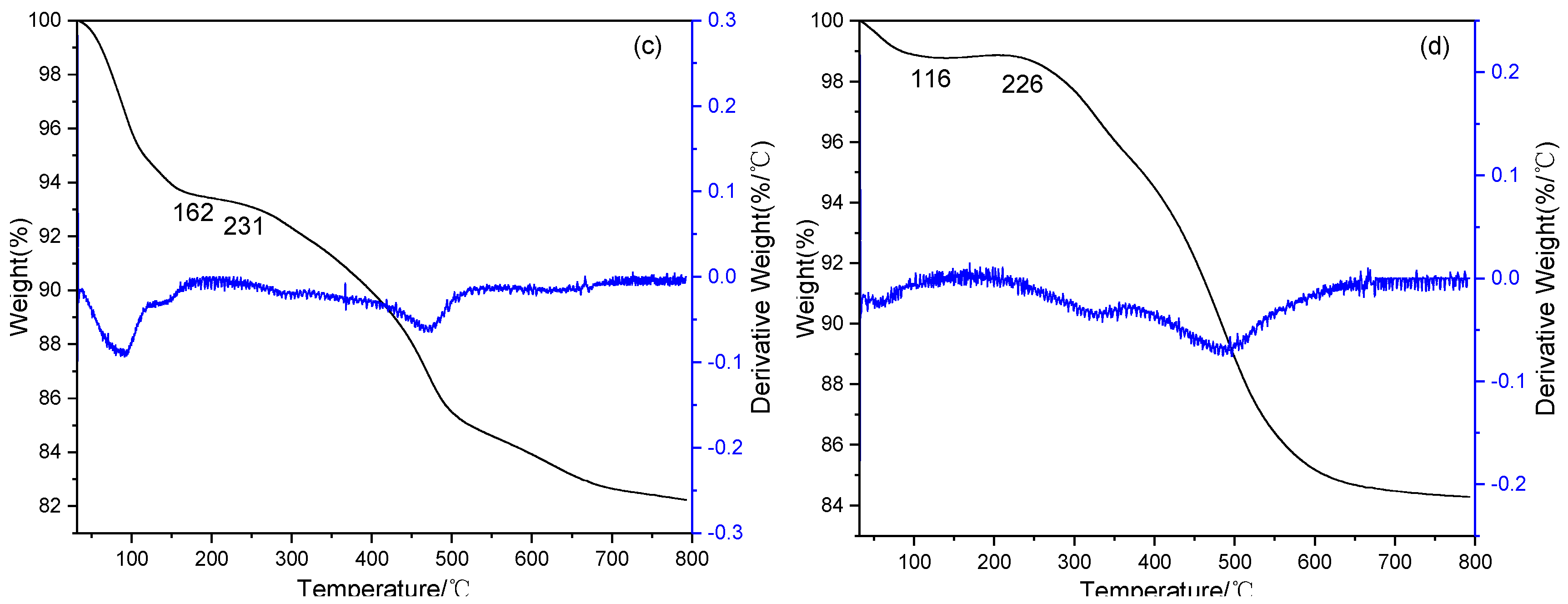
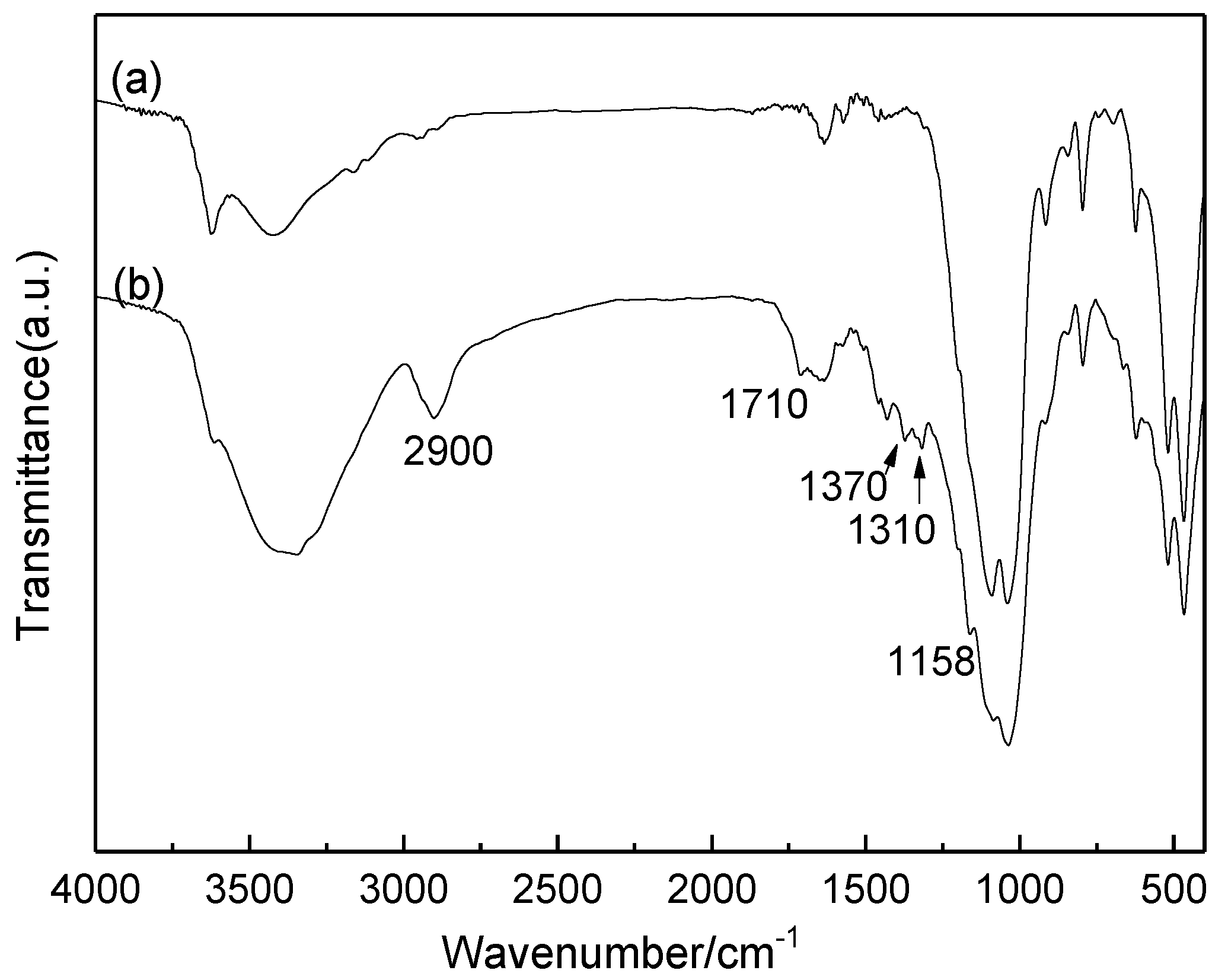
2.1. Characterization of Catalysts
2.2. Hydrolysis of Cellulose
2.2.1. Effect of Mt Modified by Different Functional Groups in Cellulose Hydrolysis
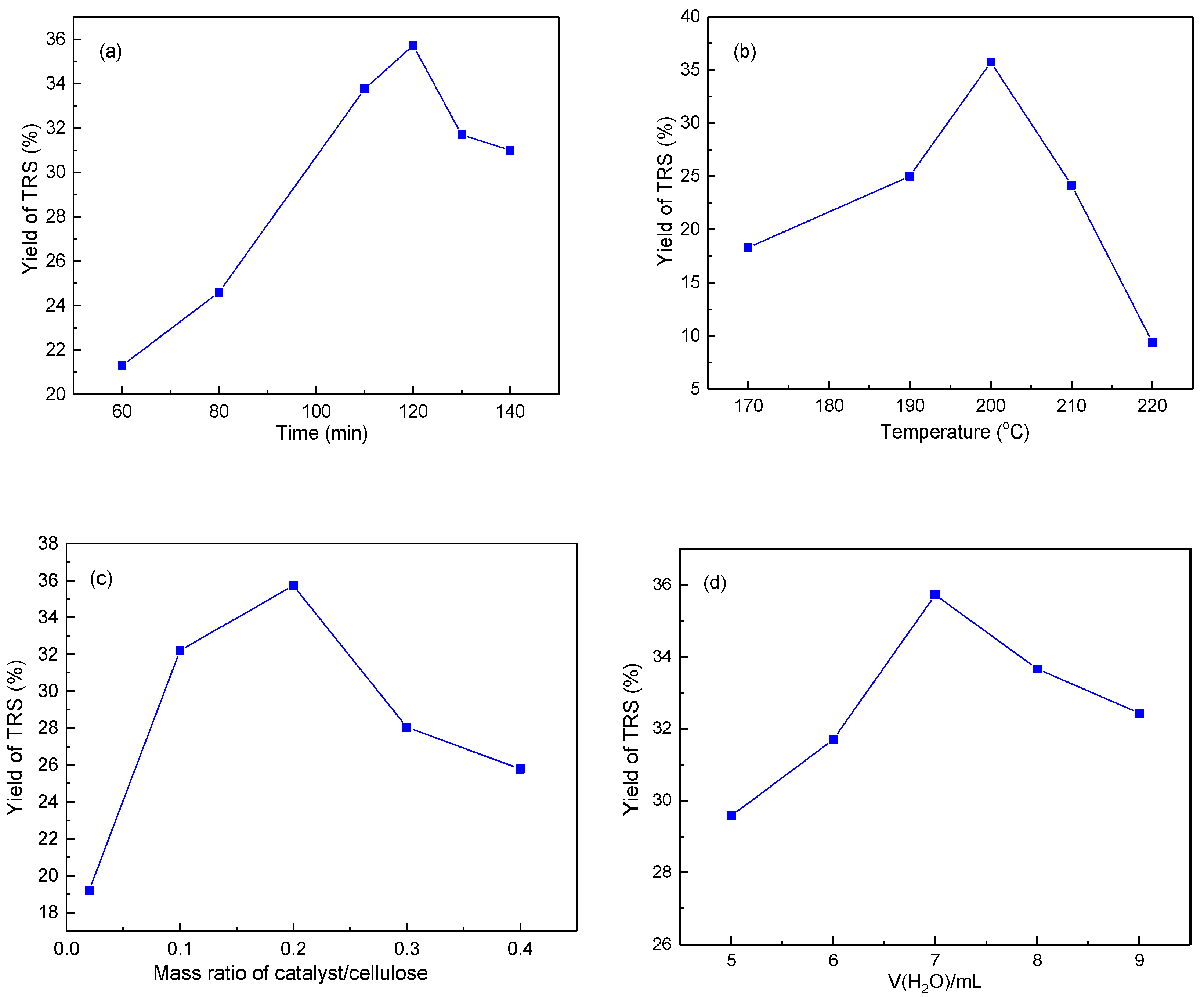
2.2.2. Effect of Reaction Conditions on Cellulose Hydrolysis over Mt-IL
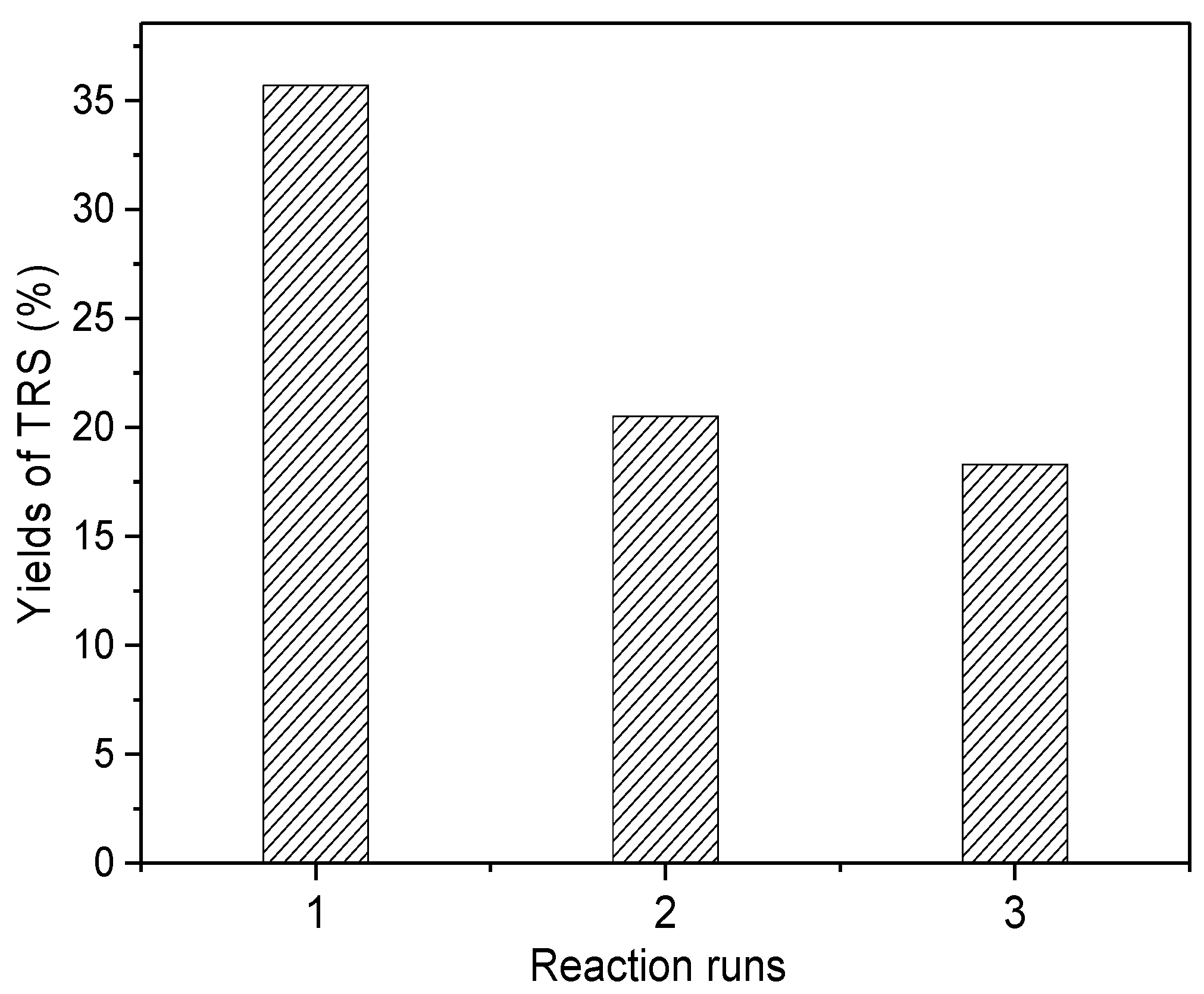
2.2.3. Reuse of Mt-IL in Cellulose Hydrolysis
3. Materials and Methods
3.1. Materials
3.2. Catalyst Preparation
3.2.1. Preparation of Acid-Activated Mt
3.2.2. Preparation of Mt-SO3H
3.2.3. Preparation of Mt-IL
3.3. Catalytic Conversion of Cellulose
3.4. Catalyst Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhou, C.H.; Xia, X.; Lin, C.X.; Tong, D.S.; Beltramini, J. Catalytic conversion of lignocellulosic biomass to fine chemicals and fuels. Chem. Soc. Rev. 2011, 40, 5588–5617. [Google Scholar] [CrossRef]
- Sweygers, N.; Alewaters, N.; Dewil, R.; Appels, L. Microwave effects in the dilute acid hydrolysis of cellulose to 5-hydroxymethylfurfural. Sci. Rep. 2018, 8, 11–15. [Google Scholar] [CrossRef]
- Yu, J.; Wang, J.Y.; Wang, Z.; Zhou, M.D.; Wang, H.Y. Hydrolysis of cellulose promoted by silicalite-1 modified HY zeolite in 1-ethyl-3-methylimidazolium chloride. Cellulose 2018, 25, 1607–1615. [Google Scholar] [CrossRef]
- Chen, Y.D.; Ai, X.L.; Huang, B.; Huang, M.J.; Huang, Y.; Lu, Y. Consecutive preparation of hydrochar catalyst functionalized in situ with sulfonic groups for efficient cellulose hydrolysis. Cellulose 2017, 24, 2743–2752. [Google Scholar] [CrossRef]
- Li, O.L.; Ikura, R.; Ishizaki, T. Hydrolysis of cellulose to glucose over carbon catalysts sulfonated via a plasma process in dilute acids. Green Chem. 2017, 19, 4774–4777. [Google Scholar] [CrossRef]
- Chu, S.; Yang, L.N.; Guo, X.C.; Dong, L.L.; Chen, X.F.; Li, Y.R.; Mu, X.D. The influence of pore structure and Si/Al ratio of HZSM-5 zeolites on the product distributions of alpha-cellulose hydrolysis. Mol. Catal. 2018, 445, 240–247. [Google Scholar] [CrossRef]
- Dutta, S.; Pal, S. Promises in direct conversion of cellulose and lignocellulosic biomass to chemicals and fuels: Combined solvent-nanocatalysis approach for biorefinary. Biomass Bioenerg. 2014, 62, 182–197. [Google Scholar] [CrossRef]
- Liu, X.X.; Xu, Q.; Liu, J.Y.; Yin, D.L.; Su, S.P.; Ding, H. Hydrolysis of cellulose into reducing sugars in ionic liquids. Fuel 2016, 164, 46–50. [Google Scholar] [CrossRef]
- Song, J.L.; Fan, H.L.; Ma, J.; Han, B.X. Conversion of glucose and cellulose into value-added products in water and ionic liquids. Green Chem. 2013, 15, 2619–2635. [Google Scholar] [CrossRef]
- Zhang, M.; Wu, M.H.; Liu, Q.; Wang, X.; Lv, T.; Jia, L.S. Graphene oxide mediated cellulose-derived carbon as a highly selective catalyst for the hydrolysis of cellulose to glucose. Appl. Catal. A-Gen. 2017, 543, 218–224. [Google Scholar] [CrossRef]
- Weingarten, R.; Rodriguez-Beuerman, A.; Cao, F.; Luterbacher, J.S.; Alonso, D.M.; Dumesic, J.A.; Huber, G.W. Selective Conversion of cellulose to hydroxymethylfurfural in polar aprotic solvents. Chem. Cat. Chem. 2014, 6, 2229–2234. [Google Scholar] [CrossRef]
- Chen, G.Z.; Wang, X.C.; Jiang, Y.J.; Mu, X.D.; Liu, H.C. Insights into deactivation mechanism of sulfonated carbonaceous solid acids probed by cellulose hydrolysis. Catal. Today 2019, 319, 25–30. [Google Scholar] [CrossRef]
- Palkovits, R.; Tajvidi, K.; Procelewska, J.; Rinaldi, R.; Ruppert, A. Hydrogenolysis of cellulose combining mineral acids and hydrogenation catalysts. Green Chem. 2010, 12, 972–978. [Google Scholar] [CrossRef]
- Hammerer, F.; Loots, L.; Do, J.L.; Therien, J.P.D.; Nickels, C.W.; Friscic, T.; Auclair, K. Solvent-free enzyme activity: Quick, high-yielding mechanoenzymatic hydrolysis of cellulose into glucose. Angew. Chem.-Int. Edit. 2018, 57, 2621–2624. [Google Scholar] [CrossRef]
- Buffiere, J.; Ahvenainen, P.; Borrega, M.; Svedstrom, K.; Sixta, H. Supercritical water hydrolysis: A green pathway for producing low-molecular-weight cellulose. Green Chem. 2016, 18, 6516–6525. [Google Scholar] [CrossRef]
- Huang, Y.B.; Fu, Y. Hydrolysis of cellulose to glucose by solid acid catalysts. Green Chem. 2013, 15, 1095–1111. [Google Scholar] [CrossRef]
- Van, D.V.S.; Geboers, J.; Jacobs, P.A.; Sels, B.F. Recent advances in the catalytic conversion of cellulose. Chem. Cat. Chem. 2011, 3, 82–94. [Google Scholar]
- Chang, J.K.W.; Duret, X.; Berberi, V.; Zahedi-Niaki, H.; Lavoie, J.M. Two-Step Thermochemical Cellulose Hydrolysis With Partial Neutralization for Glucose Production. Front. Chem. 2018, 6, 117–128. [Google Scholar] [CrossRef]
- Shen, F.; Smith, R.L.; Li, L.Y.; Yan, L.L.; Qi, X.H. Eco-friendly method for efficient conversion of cellulose into levulinic acid in pure water with cellulase-mimetic solid acid catalyst. ACS Sustain. Chem. Eng. 2017, 5, 2421–2427. [Google Scholar] [CrossRef]
- Lanzafame, P.; Temi, D.M.; Perathoner, S.; Spadaro, A.N.; Centi, G. Direct conversion of cellulose to glucose and valuable intermediates in mild reaction conditions over solid acid catalysts. Catal. Today 2012, 179, 178–184. [Google Scholar] [CrossRef]
- Tian, J.; Wang, J.H.; Zhao, S.; Jiang, C.Y.; Zhang, X.; Wang, X.H. Hydrolysis of cellulose by the heteropoly acid H3PW12O40. Cellulose 2010, 17, 587–594. [Google Scholar] [CrossRef]
- Almohalla, M.; Rodriguez-Ramos, I.; Ribeiro, L.S.; Orfao, J.J.; Pereira, M.F.R.; Guerrero-Ruiz, A. Cooperative action of heteropolyacids and carbon supported Ru catalysts for the conversion of cellulose. Catal. Today 2018, 301, 65–71. [Google Scholar] [CrossRef]
- Xiong, Y.; Zhang, Z.; Wang, X.; Liu, B.; Lin, J. Hydrolysis of cellulose in ionic liquids catalyzed by a magnetically-recoverable solid acid catalyst. Chem. Eng. J. 2014, 235, 349–355. [Google Scholar] [CrossRef]
- Hu, L.; Wu, Z.; Xu, J.; Zhou, S.; Tang, G. Efficient hydrolysis of cellulose over a magnetic lignin-derived solid acid catalyst in 1-butyl-3-methylimidazolium chloride. Korean J. Chem. Eng. 2016, 33, 1232–1238. [Google Scholar] [CrossRef]
- Shrotri, A.; Kobayashi, H.; Fukuoka, A. Cellulose Depolymerization over heterogeneous catalysts. Accounts Chem. Res. 2018, 51, 761–768. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, B.; Douthwaite, M.; Liu, Q.; Zhang, C.; Wu, Q.; Hutchings, G. Macroporous–mesoporous carbon supported Ni catalysts for the conversion of cellulose to polyols. Green Chem. 2018, 20, 3634–3642. [Google Scholar] [CrossRef]
- Li, H.X.; Zhang, X.H.; Wang, Q.; Zhang, K.; Cao, Q.; Jin, L.E. Preparation of the recycled and regenerated mesocarbon microbeads-based solid acid and its catalytic behaviors for hydrolysis of cellulose. Bioresour. Technol. 2018, 270, 166–171. [Google Scholar] [CrossRef]
- Shen, F.; Guo, T.M.; Bai, C.X.; Qiu, M.; Qi, X.H. Hydrolysis of cellulose with one-pot synthesized sulfonated carbonaceous solid acid. Fuel Process. Technol. 2018, 169, 244–247. [Google Scholar] [CrossRef]
- Su, J.L.; Qiu, M.; Shen, F.; Qi, X.H. Efficient hydrolysis of cellulose to glucose in water by agricultural residue-derived solid acid catalyst. Cellulose 2018, 25, 17–22. [Google Scholar] [CrossRef]
- Zhang, D.; Zhou, C.H.; Lin, C.X.; Tong, D.S.; Yu, W.H. Synthesis of clay minerals. Appl. Clay Sci. 2010, 50, 1–11. [Google Scholar] [CrossRef]
- Saikia, P.K.; Sarmah, P.P.; Borah, B.J.; Saikia, L.; Saikia, K.; Dutta, D.K. Stabilized Fe3O4 magnetic nanoparticles into nanopores of modified montmorillonite clay: A highly efficient catalyst for the Baeyer-Villiger oxidation under solvent free conditions. Green Chem. 2016, 18, 2843–2850. [Google Scholar] [CrossRef]
- Ruiz-Hitzky, E.; Sobral, M.M.C.; Gomez-Aviles, A.; Nunes, C.; Ruiz-Garcia, C.; Ferreira, P.; Aranda, P. Clay-graphene nanoplatelets functional conducting composites. Adv. Funct. Mater. 2016, 26, 7394–7405. [Google Scholar] [CrossRef]
- Wang, Q.Y.; Wang, Y.X.; Chen, L.Y. A green composite hydrogel based on cellulose and clay as efficient absorbent of colored organic effluent. Carbohydr. Polym. 2019, 210, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Biswick, T.T.; Choy, J.H. Layered nanomaterials for green materials. J. Mater. Chem. 2009, 19, 2553–2563. [Google Scholar] [CrossRef]
- Kaur, M.; Arshad, M.; Ullah, A. In-situ nanoreinforced green bionanomaterials from natural keratin and montmorillonite (MMT)/cellulose nanocrystais (CNC). ACS Sustain. Chem. Eng. 2018, 6, 1977–1987. [Google Scholar] [CrossRef]
- Zhou, C.H.; Zhang, D.; Tong, D.S.; Wu, L.M.; Yu, W.H.; Ismadji, S. Paper-like composites of cellulose acetate-organo-montmorillonite for removal of hazardous anionic dye in water. Chem. Eng. J. 2012, 209, 223–234. [Google Scholar] [CrossRef]
- Bonacci, S.; Nardi, M.; Costanzo, P.; De, N.A.; Di, G.M.L.; Oliverio, M.; Procopio, A. Montmorillonite K10-catalyzed solvent-free conversion of furfural into cyclopentenones. Catalysts 2019, 9, 301. [Google Scholar] [CrossRef]
- Mokhtarani, B.; Musavi, J.; Parvini, M. Extraction of toluene from alkane using BMIM NO3 or OMIM NO3 ionic liquid at 298.15 K and atmospheric pressure. Fluid Phase Equilib. 2014, 363, 41–47. [Google Scholar] [CrossRef]
- Tong, D.S.; Xia, X.; Luo, X.P.; Wu, L.M.; Lin, C.X.; Yu, W.H.; Zhou, C.H.; Zhong, Z.K. Catalytic hydrolysis of cellulose to reducing sugar over acid-activated montmorillonite catalysts. Appl. Clay Sci. 2013, 74, 147–153. [Google Scholar] [CrossRef]
- Nino, D.A.; Merino, P.; Algieri, V.; Nardi, M.; Gioia, D.M.L.; Russo, B.; Tallarida, M.A.; Maiuolo, L. Synthesis of 1,5-functionalized 1,2,3-triazoles using ionic liquid/iron(III) chloride as an efficient and reusable homogeneous catalyst. Catalysts 2018, 8, 364–377. [Google Scholar]
- Zhang, Y.T.; Du, H.B.; Qian, X.H.; Chen, E.Y.X. Ionic liquid-water mixtures: Enhanced K-w for efficient cellulosic biomass conversion. Energy Fuels 2010, 24, 2410–2417. [Google Scholar] [CrossRef]
- Moreau, C.; Finiels, A.; Vanoye, L. Dehydration of fructose and sucrose into 5-hydroxymethylfurfural in the presence of 1-H-3-methyl imidazolium chloride acting both as solvent and catalyst. J. Mol. Catal. A-Chem. 2006, 253, 165–169. [Google Scholar] [CrossRef]
- Vu, A.; Wickramasinghe, S.R.; Qian, X.H. Polymeric solid acid catalysts for lignocellulosic biomass fractionation. Ind. Eng. Chem. Res. 2018, 57, 4514–4525. [Google Scholar] [CrossRef]
- Gioia, D.M.L.; Costanzo, P.; Nino, D.A.; Maiuolo, L.; Nardi, M.; Olivito, F.; Procopio, A. Simple and efficient Fmoc removal in ionic liquid. Rsc. Adv. 2017, 7, 36482–36491. [Google Scholar] [CrossRef]
- Moyer, P.; Smith, M.D.; Abdoulmoumine, N.; Chmely, S.C.; Smith, J.C.; Petridis, L.; Labbe, N. Relationship between lignocellulosic biomass dissolution and physicochemical properties of ionic liquids composed of 3-methylimidazolium cations and carboxylate anions. Phys. Chem. Chem. Phys. 2018, 20, 2508–2516. [Google Scholar] [CrossRef]
- Yu, J.L.; Maliutina, K.; Tahmasebi, A. A review on the production of nitrogen-containing compounds from microalgal biomass via pyrolysis. Bioresour. Technol. 2018, 270, 689–701. [Google Scholar] [CrossRef]
- Lopes, A.M.D.; Bogel-Lukasik, R. Acidic ionic liquids as sustainable approach of cellulose and lignocellulosic biomass conversion without additional catalysts. ChemSusChem 2015, 8, 947–965. [Google Scholar] [CrossRef]
- Shuai, L.; Pan, X.J. Hydrolysis of cellulose by cellulase-mimetic solid catalyst. Energy Environ. Sci. 2012, 5, 6889–6894. [Google Scholar] [CrossRef]
- Chipera, S.J.; Bish, D.L. Baseline studies of the clay minerals society source clays: Powder X-ray diffraction analyses. Clay Clay Min. 2001, 49, 398–409. [Google Scholar] [CrossRef]
- Viani, A.; Gaultieri, A.F.; Artioli, G. The nature of disorder in montmorillonite by simulation of X-ray powder patterns. Am. Miner. 2002, 87, 966–975. [Google Scholar] [CrossRef]
- Zatta, L.; Ramos, L.P.; Wypych, F. Acid-activated montmorillonites as heterogeneous catalysts for the esterification of lauric acid acid with methanol. Appl. Clay Sci. 2013, 80, 236–244. [Google Scholar] [CrossRef]
- Zhang, C.; Fu, Z.H.; Liu, Y.C.; Dai, B.H.; Zou, Y.H.; Gong, X.L.; Wang, Y.L.; Deng, X.L.; Wu, H.T.; Xu, Q.; et al. Ionic liquid-functionalized biochar sulfonic acid as a biomimetic catalyst for hydrolysis of cellulose and bamboo under microwave irradiation. Green Chem. 2012, 14, 1928–1934. [Google Scholar] [CrossRef]
- Wilson, K.; Lee, A.F.; Macquarrie, D.J.; Clark, J.H. Structure and reactivity of sol-gel sulphonic acid silicas. Appl. Catal. A-Gen. 2002, 228, 127–133. [Google Scholar] [CrossRef]
- Cavani, F.; Trifiro, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Brandt, A.; Ray, M.J.; To, T.Q.; Leak, D.J.; Murphy, R.J.; Welton, T. Ionic liquid pretreatment of lignocellulosic biomass with ionic liquid-water mixtures. Green Chem. 2011, 13, 2489–2499. [Google Scholar] [CrossRef]
- Johns, W.D. Clay mineral catalysis and petroleum generatio. Annu. Rev. Earth Planet Sci. 1979, 7, 183–198. [Google Scholar] [CrossRef]
- Ramli, N.A.S.; Amin, N.A.S. Catalytic hydrolysis of cellulose and oil palm biomass in ionic liquid to reducing sugar for levulinic acid production. Fuel Process. Technol. 2014, 128, 490–498. [Google Scholar] [CrossRef]
- Chen, T.L.; Xiong, C.R.; Tao, Y.S. Enhanced Hydrolysis of cellulose in ionic liquid using mesoporous ZSM-5. Molecules 2018, 23, 10. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Wu, Z.; Xu, J.X.; Sun, Y.; Lin, L.; Liu, S.J. Zeolite-promoted transformation of glucose into 5-hydroxymethylfurfural in ionic liquid. Chem. Eng. J. 2014, 244, 137–144. [Google Scholar] [CrossRef]
- Sun, Y.; Zhuang, J.P.; Lin, L.; Ouyang, P.K. Clean conversion of cellulose into fermentable glucose. Biotechnol. Adv. 2009, 27, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Tan, H.M. Structural characterization of cellulose with enzymatic treatment. J. Mol. Struct. 2004, 705, 189–193. [Google Scholar] [CrossRef]
- Oh, S.Y.; Yoo, D.I.; Shin, Y.; Kim, H.C.; Kim, H.Y.; Chung, Y.S.; Park, W.H.; Youk, J.H. Crystalline structure analysis of cellulose treated with sodium hydroxide and carbon dioxide by means of X-ray diffraction and FTIR spectroscopy. Carbohydr. Res. 2005, 340, 2376–2391. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

| Entry | Catalysts | Content of Acidic Sites (mmol·g−1) | TRS Yield (%) | 5-HMF Yield (%) |
|---|---|---|---|---|
| 1 | Mt | 0.012 | 7.9 | 0.4 |
| 2 | HMt | 0.320 | 14.4 | 1.0 |
| 3 | Mt-SO3H-1 | 0.236 | 19.3 | 1.6 |
| 4 | Mt-SO3H-2 | 0.296 | 24.5 | 1.7 |
| 5 | Mt-SO3H-3 | 0.532 | 24.6 | 1.8 |
| 6 | Mt-IL | 0.056 | 35.7 | 1.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Y.; Yang, M.; Tong, D.; Yang, H.; Fang, K. Eco-Friendly Ca-Montmorillonite Grafted by Non-Acidic Ionic Liquid Used as A Solid Acid Catalyst in Cellulose Hydrolysis to Reducing Sugars. Molecules 2019, 24, 1832. https://doi.org/10.3390/molecules24091832
Zhou Y, Yang M, Tong D, Yang H, Fang K. Eco-Friendly Ca-Montmorillonite Grafted by Non-Acidic Ionic Liquid Used as A Solid Acid Catalyst in Cellulose Hydrolysis to Reducing Sugars. Molecules. 2019; 24(9):1832. https://doi.org/10.3390/molecules24091832
Chicago/Turabian StyleZhou, Yang, Miao Yang, Dongshen Tong, Haiyan Yang, and Kai Fang. 2019. "Eco-Friendly Ca-Montmorillonite Grafted by Non-Acidic Ionic Liquid Used as A Solid Acid Catalyst in Cellulose Hydrolysis to Reducing Sugars" Molecules 24, no. 9: 1832. https://doi.org/10.3390/molecules24091832
APA StyleZhou, Y., Yang, M., Tong, D., Yang, H., & Fang, K. (2019). Eco-Friendly Ca-Montmorillonite Grafted by Non-Acidic Ionic Liquid Used as A Solid Acid Catalyst in Cellulose Hydrolysis to Reducing Sugars. Molecules, 24(9), 1832. https://doi.org/10.3390/molecules24091832






