Selective Adsorption-Based Separation of Flue Gas and Natural Gas in Zirconium Metal-Organic Frameworks Nanocrystals
Abstract
1. Introduction
2. Results
2.1. Synthesis and Characterization
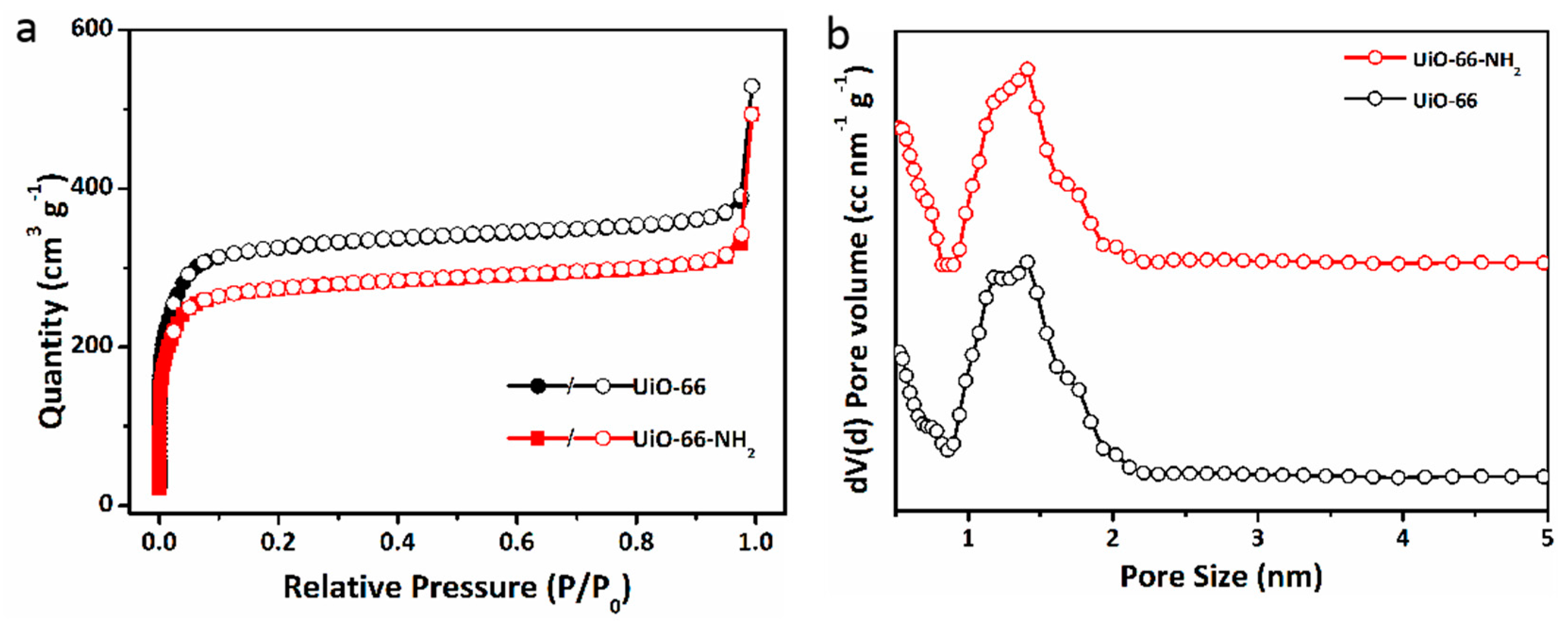
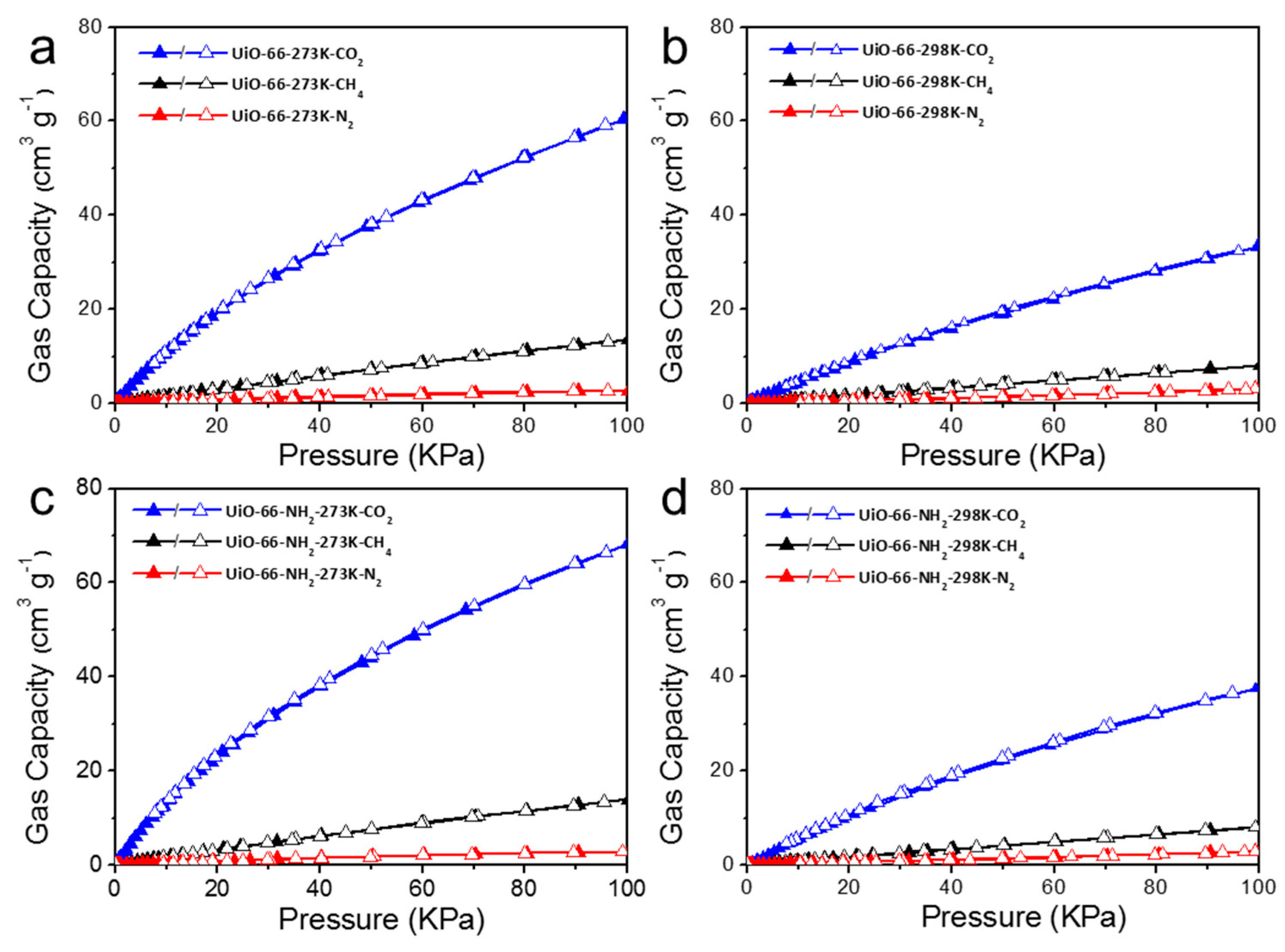
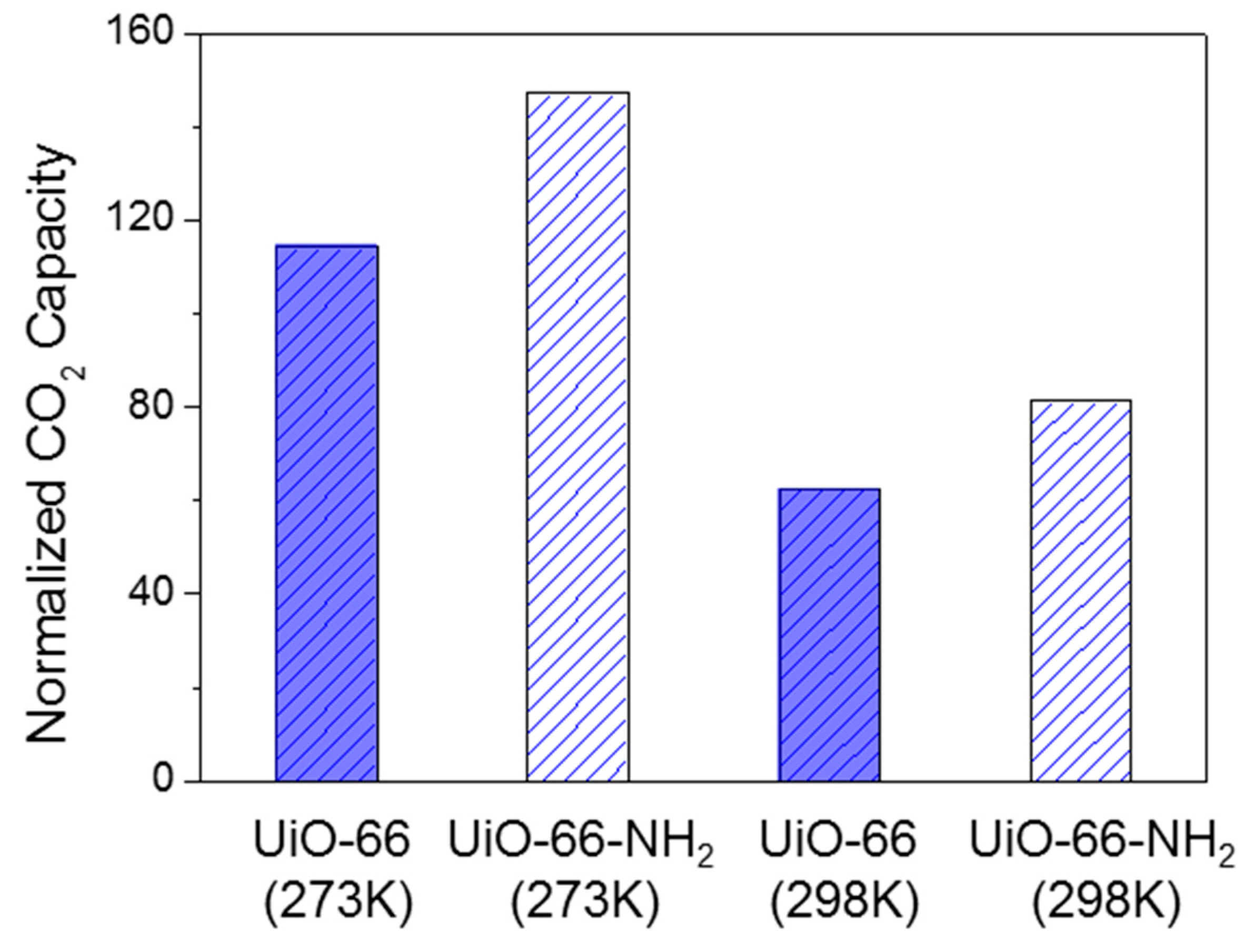
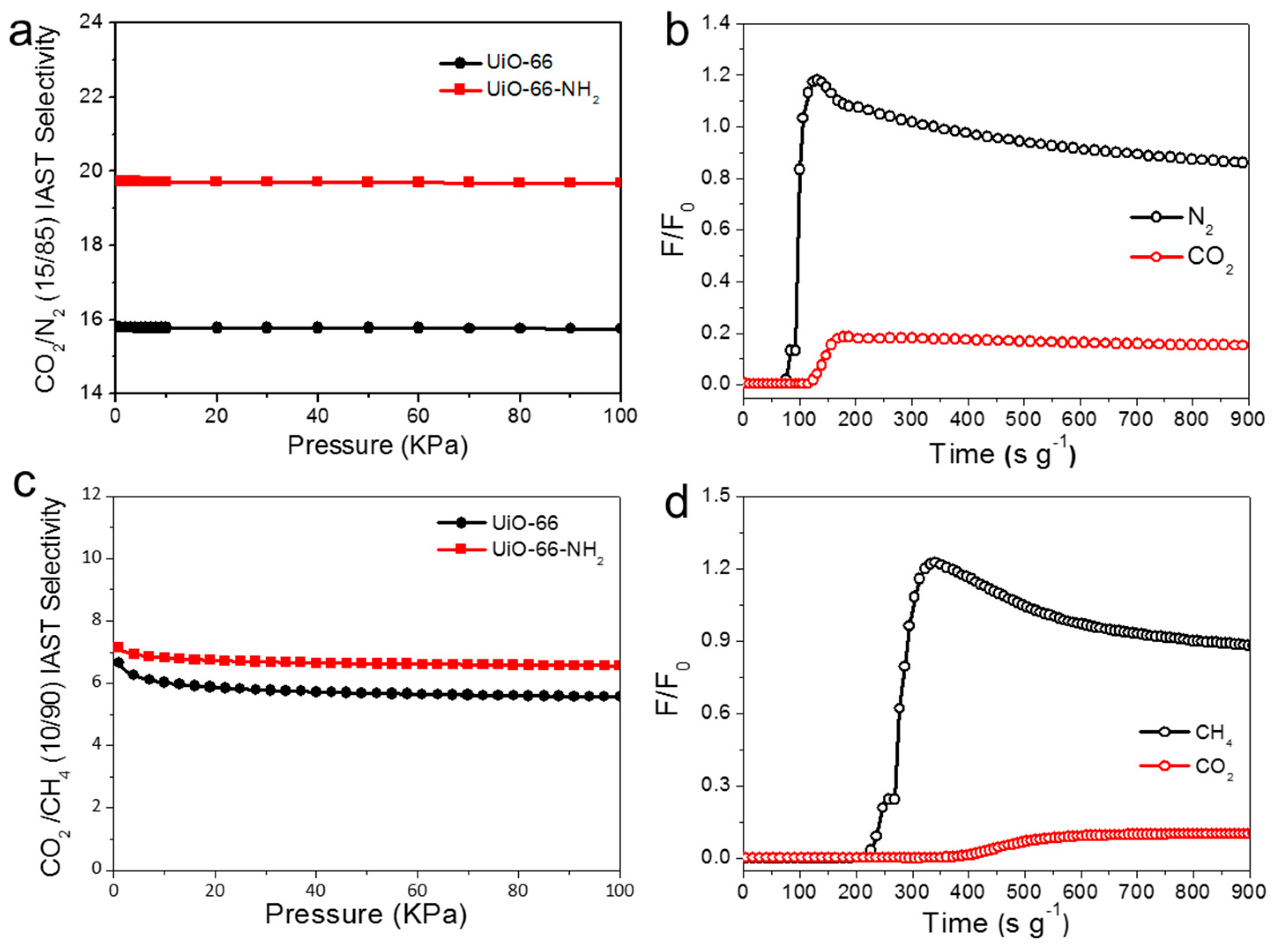
2.2. Pore Structure and Gas Selective Adsorption
2.3. Dynamic Separation of Flue Gas and Natural Gas
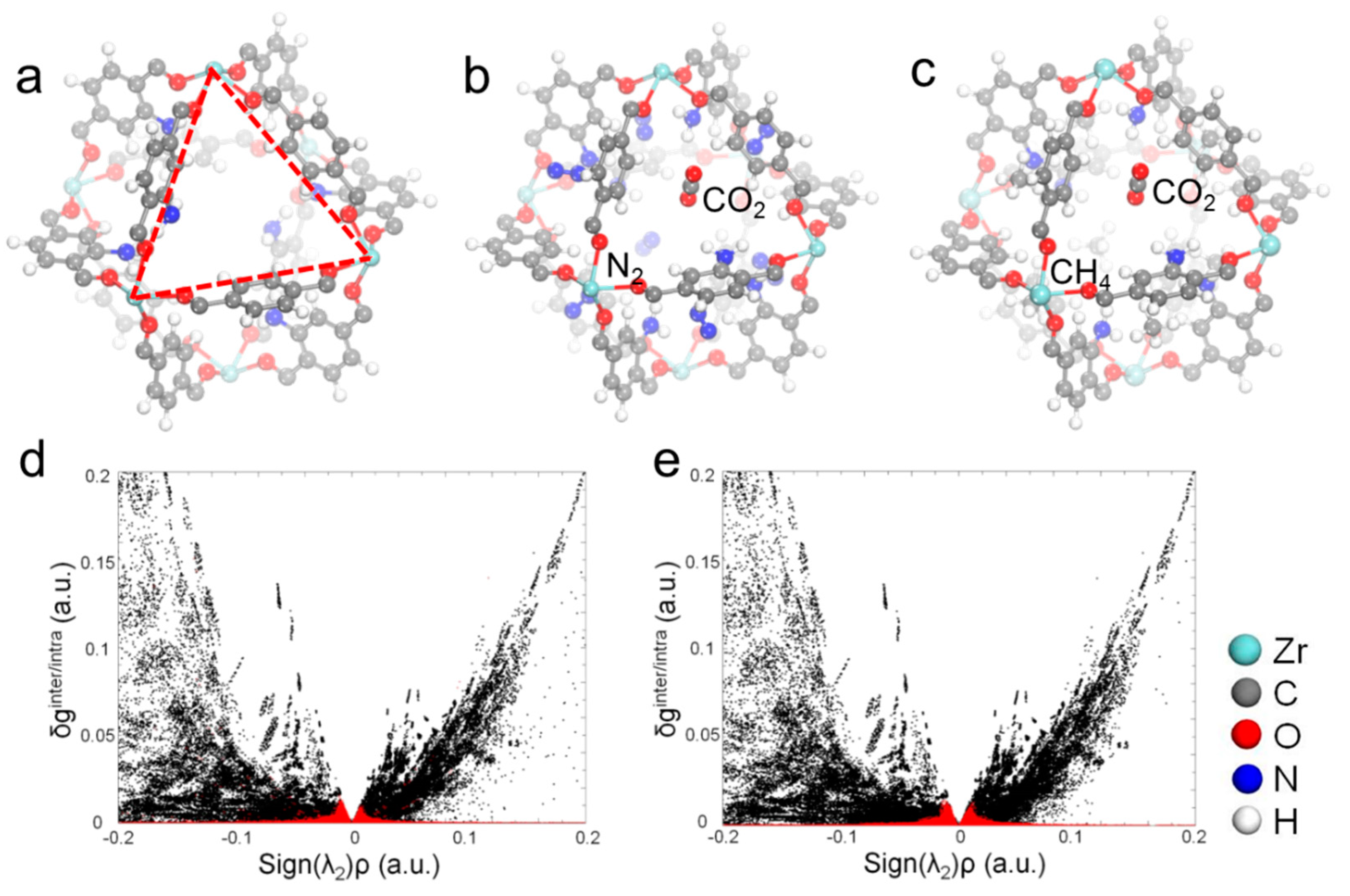
2.4. Monte Carlo Simulation of Gas Selective Adsorption
3. Materials and Methods
3.1. Chemicals and Characterizations
3.2. Synthesis of UiO-66 and UiO-66-NH2
3.3. Acid-Base Titrations
3.4. Gas Adsorption Measurement
3.5. Breakthrough Experiments
3.6. DFT Calculations
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davis, S.J.; Lewis, N.S.; Shaner, M.; Aggarwal, S.; Arent, D.; Azevedo, I.L.; Benson, S.M.; Bradley, T.; Brouwer, J.; Chiang, Y.-M.; et al. Net-zero emissions energy systems. Science 2018, 360, eaas9793. [Google Scholar] [CrossRef]
- The State of Greenhouse Gases in the Atmosphere Based on Global Observations through 2016. Available online: https://meetingorganizer.copernicus.org/EGU2017/EGU2017-8824-1.pdf (accessed on 8 May 2019).
- Sanz-Perez, E.S.; Murdock, C.R.; Didas, S.A.; Jones, C.W. Direct capture of CO2 from ambient air. Chem. Rev. 2016, 116, 11840–11876. [Google Scholar] [CrossRef] [PubMed]
- Bui, M.; Adjiman, C.S.; Bardow, A.; Anthony, E.J.; Boston, A.; Brown, S.; Fennell, P.S.; Fuss, S.; Galindo, A.; Hackett, L.A.; et al. Carbon capture and storage (CCS): The way forward. Energy Environ. Sci. 2018, 11, 1062–1176. [Google Scholar]
- Olajossy, A. Effective recovery of methane from coal mine methane gas by vacuum pressure swing adsorption: A pilot scale case study. Chem. Eng. Sci. 2013, 1, 46–54. [Google Scholar] [CrossRef]
- Nanda, S.; Reddy, S.N.; Mitra, S.K.; Kozinski, J.A. The progressive routes for carbon capture and sequestration. Energy Sci. Eng. 2016, 4, 99–122. [Google Scholar] [CrossRef]
- Sholl, D.S.; Lively, R.P. Seven chemical separations to change the world. Nature 2016, 532, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Baena-Moreno, F.M.; Rodriguez-Galan, M.; Vega, F.; Alonso-Farinas, B.; Vilches Arenas, L.F.; Navarrete, B. Carbon capture and utilization technologies: A literature review and recent advances. Energy Sources Part A 2019, 41, 1403–1433. [Google Scholar] [CrossRef]
- Wilberforce, T.; Baroutaji, A.; Soudan, B.; Al-Alami, A.H.; Olabi, A.G. Outlook of carbon capture technology and challenges. Sci. Total Environ. 2019, 657, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, H.P.; Liao, C.-Y.; Zhao, X.; Hsiung, T.-L.; Liu, S.-H.; Chang, S.-G. Dual alkali solvent system for CO2 capture from flue gas. Environ. Sci. Technol. 2017, 51, 8824–8831. [Google Scholar] [CrossRef]
- Cachaza, A.; Gomez-Diaz, D.; Montans, A.; Navaza, J.M.; Rumbo, A. Carbon dioxide chemical absorption by solvents based on diamine and amines blend. AICHE J. 2018, 64, 2702–2710. [Google Scholar] [CrossRef]
- Yu, J.; Xie, L.-H.; Li, J.-R.; Ma, Y.; Seminario, J.M.; Balbuena, P.B. CO2 capture and separations using MOFs: Computational and experimental studies. Chem. Rev. 2017, 117, 9674–9754. [Google Scholar] [CrossRef] [PubMed]
- Oschatz, M.; Antonietti, M. A search for selectivity to enable CO2 capture with porous adsorbents. Energy Environ. Sci. 2018, 11, 57–70. [Google Scholar] [CrossRef]
- Li, J.-R.; Kuppler, R.J.; Zhou, H.-C. Selective gas adsorption and separation in metal-organic frameworks. Chem. Soc. Rev. 2009, 38, 1477–1504. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Sun, Y.; Che, S.; Yang, X.; Wang, X.; Bosch, M.; Wang, Q.; Li, H.; Smith, M.; Yuan, S.; et al. Porous organic polymers for post-combustion carbon capture. Adv. Mater. 2017, 29, 1700229. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhang, Y.-B. Crystallization of covalent organic frameworks for gas storage applications. Molecules 2017, 22, 1149. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-R.; Ma, Y.; McCarthy, M.C.; Sculley, J.; Yu, J.; Jeong, H.-K.; Balbuena, P.B.; Zhou, H.-C. Carbon dioxide capture-related gas adsorption and separation in metal-organic frameworks. Coord. Chem. Rev. 2011, 255, 1791–1823. [Google Scholar] [CrossRef]
- D’Amato, R.; Donnadio, A.; Carta, M.; Sangregorio, C.; Tiana, D.; Vivani, R.; Taddei, M.; Costantino, F. Water-based synthesis and enhanced CO2 capture performance of perfluorinated cerium-based metal-organic frameworks with UiO-66 and MIL-140 topology. ACS Sustainable Chem. Eng. 2019, 7, 394–402. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, Z.; Kundu, T.; Cheng, Y.; Dong, J.; Qian, Y.; Zhai, L.; Zhao, D. Metal-organic frameworks with reduced hydrophilicity for postcombustion CO2 capture from wet flue gas. ACS Sustainable Chem. Eng. 2018, 6, 11904–11912. [Google Scholar] [CrossRef]
- Cavka, J.H.; Jakobsen, S.; Olsbye, U.; Guillou, N.; Lamberti, C.; Bordiga, S.; Lillerud, K.P. A new zirconium inorganic building brick forming metal organic frameworks with exceptional stability. J. Am. Chem. Soc. 2008, 130, 13850–13851. [Google Scholar] [CrossRef]
- Peterson, G.W.; Mahle, J.J.; DeCoste, J.B.; Gordon, W.O.; Rossin, J.A. Extraordinary NO2 removal by the metal-organic framework UiO-66-NH2. Angew. Chem. Int. Ed. 2016, 55, 6235–6238. [Google Scholar] [CrossRef] [PubMed]
- Rapti, S.; Pournara, A.; Sarma, D.; Papadas, I.T.; Armatas, G.S.; Hassan, Y.S.; Alkordi, M.H.; Kanatzidis, M.G.; Manos, M.J. Rapid, green and inexpensive synthesis of high quality UiO-66 amino-functionalized materials with exceptional capability for removal of hexavalent chromium from industrial waste. Inorg. Chem. Front. 2016, 3, 635–644. [Google Scholar] [CrossRef]
- Zhan, X.-Q.; Tsai, F.-C.; Xie, L.; Zhang, K.-D.; Liu, H.-L.; Ma, N.; Shi, D.; Jiang, T. Ligands-coordinated Zr-based MOF for wastewater treatment. Nanomaterials 2018, 8, 655. [Google Scholar] [CrossRef]
- Liu, X.L.; Demir, N.K.; Wu, Z.T.; Li, K. Highly water-stable zirconium metal organic framework UiO-66 membranes supported on alumina hollow fibers for desalination. J. Am. Chem. Soc. 2015, 137, 6999–7002. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.; Wan, L.; Caro, J. Microwave-assisted synthesis of well-shaped UiO-66-NH2 with high CO2 adsorption capacity. Mater. Res. Bull. 2018, 98, 308–313. [Google Scholar] [CrossRef]
- Cam Loc, L.; Thi Thuy Van, N.; Tri, N.; Tien Cuong, H. Synthesis, characterization and adsorption ability of UiO-66-NH2. Adv. Nat. Sci. Nanosci. Nanotec. 2015, 6, 2. [Google Scholar]
- Cmarik, G.E.; Kim, M.; Cohen, S.M.; Walton, K.S. Tuning the adsorption properties of UiO-66 via ligand functionalization. Langmuir 2012, 28, 15606–15613. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Feng, Y.; Wang, Z.; Jia, M.; Yao, J. Fabrication of cellulose nanofibrils/UiO-66-NH2 composite membrane for CO2/N2 separation. J. Membrane Sci. 2018, 568, 10–16. [Google Scholar] [CrossRef]
- Nguyen, T.-B.; Rodrigue, D.; Kaliaguine, S. In-situ cross interface linking of PIM-1 polymer and UiO-66-NH2 for outstanding gas separation and physical aging control. J. Membrane Sci. 2018, 548, 429–438. [Google Scholar]
- Valenzano, L.; Civalleri, B.; Chavan, S.; Bordiga, S.; Nilsen, M.H.; Jakobsen, S.; Lillerud, K.P.; Lamberti, C. Disclosing the complex structure of UiO-66 metal organic framework: A synergic combination of experiment and theory. Chem. Mater. 2011, 23, 1700–1718. [Google Scholar] [CrossRef]
- DeStefano, M.R.; Islamoglu, T.; Hupp, J.T.; Farha, O.K. Room-temperature synthesis of UiO-66 and thermal modulation of densities of defect sites. Chem. Mater. 2017, 29, 1357–1361. [Google Scholar] [CrossRef]
- Trickett, C.A.; Gagnon, K.J.; Lee, S.; Gandara, F.; Buergi, H.-B.; Yaghi, O.M. Definitive molecular level characterization of defects in UiO-66 crystals. Angew. Chem. Int. Ed. 2015, 54, 11162–11167. [Google Scholar] [CrossRef]
- Katz, M.J.; Brown, Z.J.; Colon, Y.J.; Siu, P.W.; Scheidt, K.A.; Snurr, R.Q.; Hupp, J.T.; Farha, O.K. A facile synthesis of UiO-66, UiO-67 and their derivatives. Chem. Commun. 2013, 49, 9449–9451. [Google Scholar] [CrossRef]
- Li, L.; Yang, J.; Li, J.; Chen, Y.; Li, J. Separation of CO2/CH4 and CH4/N2 mixtures by M/DOBDC: A detailed dynamic comparison with MIL-100(Cr) and activated carbon. Microporous Mesoporous Mater. 2014, 198, 236–246. [Google Scholar] [CrossRef]
- Ling, Y.; Yang, F.; Deng, M.; Chen, Z.; Liu, X.; Weng, L.; Zhou, Y. Novel iso-reticular Zn(II) metal-organic frameworks constructed by trinuclear-triangular and paddle-wheel units: Synthesis, structure and gas adsorption. Dalton Trans. 2012, 41, 4007–4011. [Google Scholar] [CrossRef]
- Millward, A.R.; Yaghi, O.M. Metal-organic frameworks with exceptionally high capacity for storage of carbon dioxide at room temperature. J. Am. Chem. Soc. 2005, 127, 17998–17999. [Google Scholar] [CrossRef]
- Ethiraj, J.; Albanese, E.; Civalleri, B.; Vitillo, J.G.; Bonino, F.; Chavan, S.; Shearer, G.C.; Lillerud, K.P.; Bordiga, S. Carbon dioxide adsorption in amine-functionalized mixed-ligand metal-organic frameworks of UiO-66 topology. ChemSusChem 2014, 7, 3382–3388. [Google Scholar] [CrossRef] [PubMed]
- Myers, A.L.; Prausnitz, J.M. Thermodynamics of mixed-gas adsorption. AICHE J. 1965, 11, 121–127. [Google Scholar] [CrossRef]
- Chen, Y.; Lv, D.; Wu, J.; Xiao, J.; Xi, H.; Xia, Q.; Li, Z. A new MOF-505@GO composite with high selectivity for CO2/CH4 and CO2/N-2 separation. Chem. Eng. J. 2017, 308, 1065–1072. [Google Scholar] [CrossRef]
- Lin, R.B.; Chen, D.; Lin, Y.Y.; Zhang, J.P.; Chen, X.M. A zeolite-like zinc triazolate framework with high gas adsorption and separation performance. Inorg. Chem. 2012, 51, 9950–9955. [Google Scholar] [CrossRef]
- Lefebvre, C.; Rubez, G.; Khartabil, H.; Boisson, J.-C.; Contreras-García, J.; Hénon, E. Accurately extracting the signature of intermolecular interactions present in the NCI plot of the reduced density gradient versus electron density. Phys. Chem. Chem. Phys. 2017, 19, 17928–17936. [Google Scholar] [CrossRef]
- Yao, K.X.; Chen, Y.; Lu, Y.; Zhao, Y.; Ding, Y. Ultramicroporous carbon with extremely narrow pore distribution and very high nitrogen doping for efficient methane mixture gases upgrading. Carbon 2017, 122, 258–265. [Google Scholar] [CrossRef]
- Lu, Y.; He, J.; Chen, Y.; Wang, H.; Zhao, Y.; Han, Y.; Ding, Y. Effective acetylene/ethylene separation at ambient conditions by a pigment-based covalent-triazine framework. Macromol. Rapid Commun. 2018, 39, 1700468. [Google Scholar] [CrossRef] [PubMed]
- Adsorption Locator; Version 7.0; For A Module in the Materials Studio Modeling Software Package; Accelrys Inc.: San Diego, CA, USA.
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Delley, B. An all-electron numerical method for solving the local density functional for polyatomic molecules. J. Chem. Phys. 1990, 92, 508–517. [Google Scholar] [CrossRef]
- Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 2000, 113, 7756–7764. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

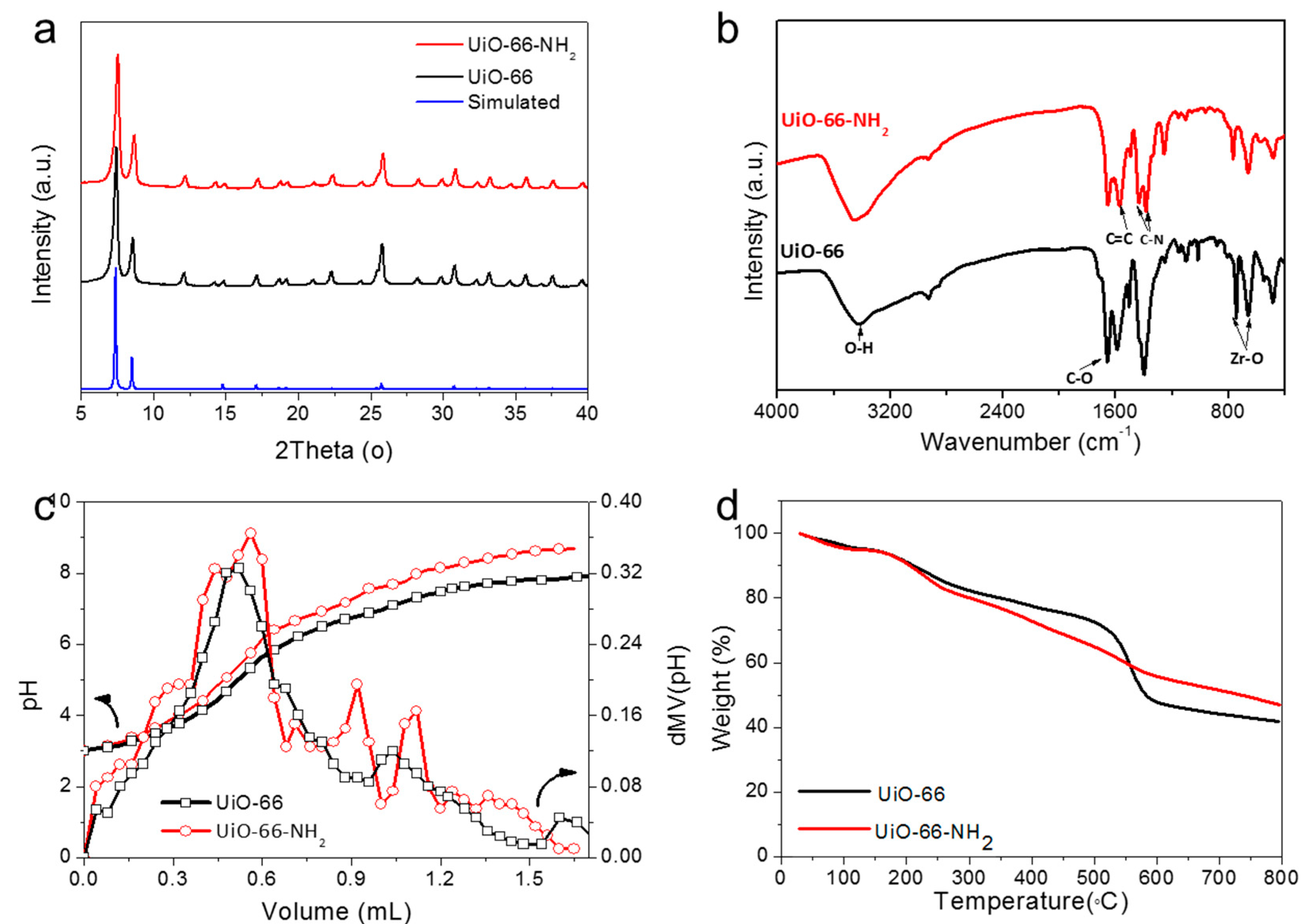
| MOFs | Bridging–OH | Acetate | Zr-OH2 | |||
|---|---|---|---|---|---|---|
| pKa | Equi. vol. (mL) | pKa | Equi. vol. (mL) | pKa | Equi. vol. (mL) | |
| UiO-66 | 3.47 | 0.52 | 5.04 | 1.04 | 6.47 | 1.60 |
| UiO-66-NH2 | 3.82 | 0.56 | 4.92 | 0.92 | 5.76 | 1.12 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, P.; Shen, Y.; Wang, D.; Chen, Y.; Zhao, Y. Selective Adsorption-Based Separation of Flue Gas and Natural Gas in Zirconium Metal-Organic Frameworks Nanocrystals. Molecules 2019, 24, 1822. https://doi.org/10.3390/molecules24091822
Li P, Shen Y, Wang D, Chen Y, Zhao Y. Selective Adsorption-Based Separation of Flue Gas and Natural Gas in Zirconium Metal-Organic Frameworks Nanocrystals. Molecules. 2019; 24(9):1822. https://doi.org/10.3390/molecules24091822
Chicago/Turabian StyleLi, Pengli, Yongli Shen, Dandan Wang, Yanli Chen, and Yunfeng Zhao. 2019. "Selective Adsorption-Based Separation of Flue Gas and Natural Gas in Zirconium Metal-Organic Frameworks Nanocrystals" Molecules 24, no. 9: 1822. https://doi.org/10.3390/molecules24091822
APA StyleLi, P., Shen, Y., Wang, D., Chen, Y., & Zhao, Y. (2019). Selective Adsorption-Based Separation of Flue Gas and Natural Gas in Zirconium Metal-Organic Frameworks Nanocrystals. Molecules, 24(9), 1822. https://doi.org/10.3390/molecules24091822





