Interaction of Arsenic Species with Organic Ligands: Competitive Removal from Water by Coagulation-Flocculation-Sedimentation (C/F/S)
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical Reagents and Stock Solutions Preparation
2.2. Experimental Procedures
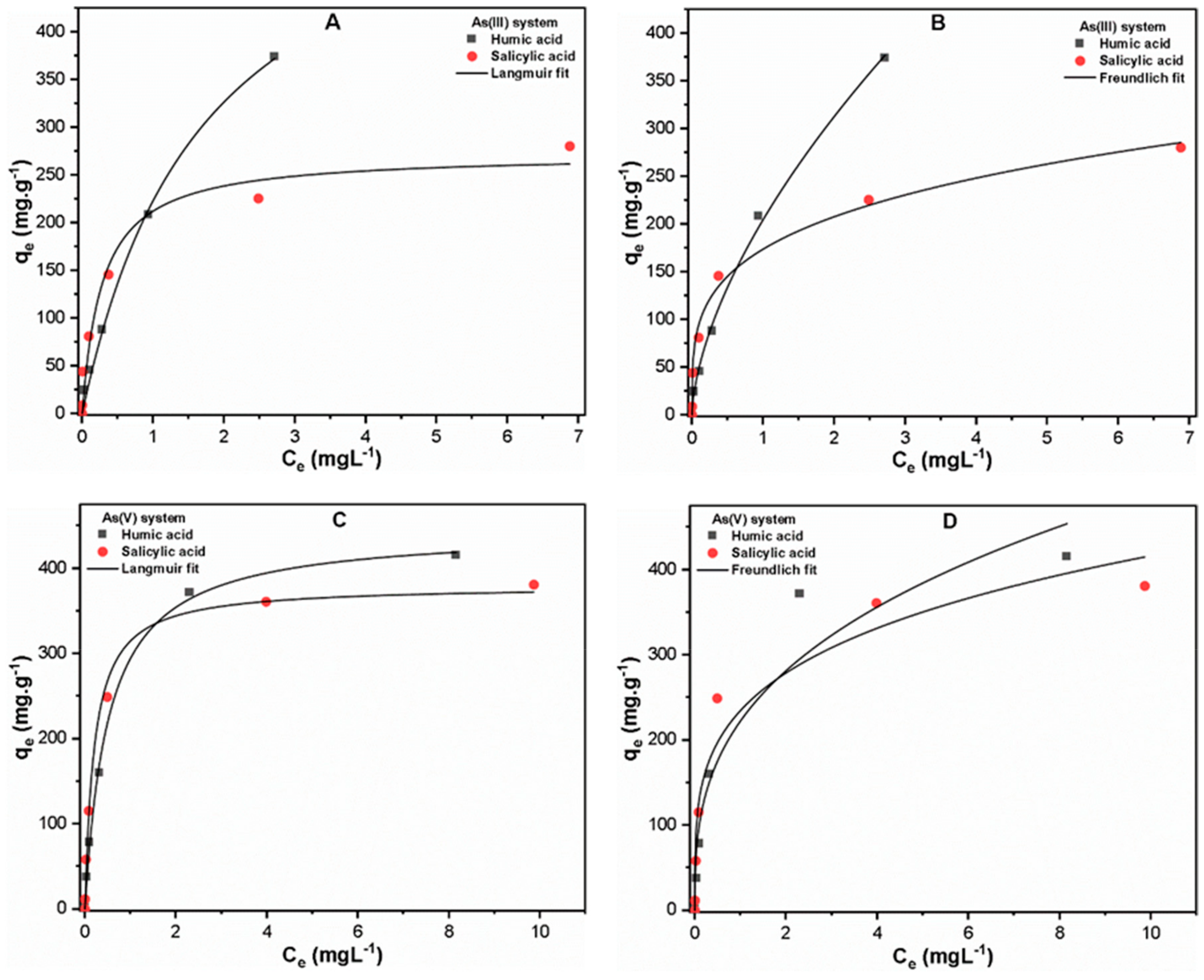
2.3. Adsorption Isotherm Study
2.4. Analytical Procedures
3. Results and Discussion
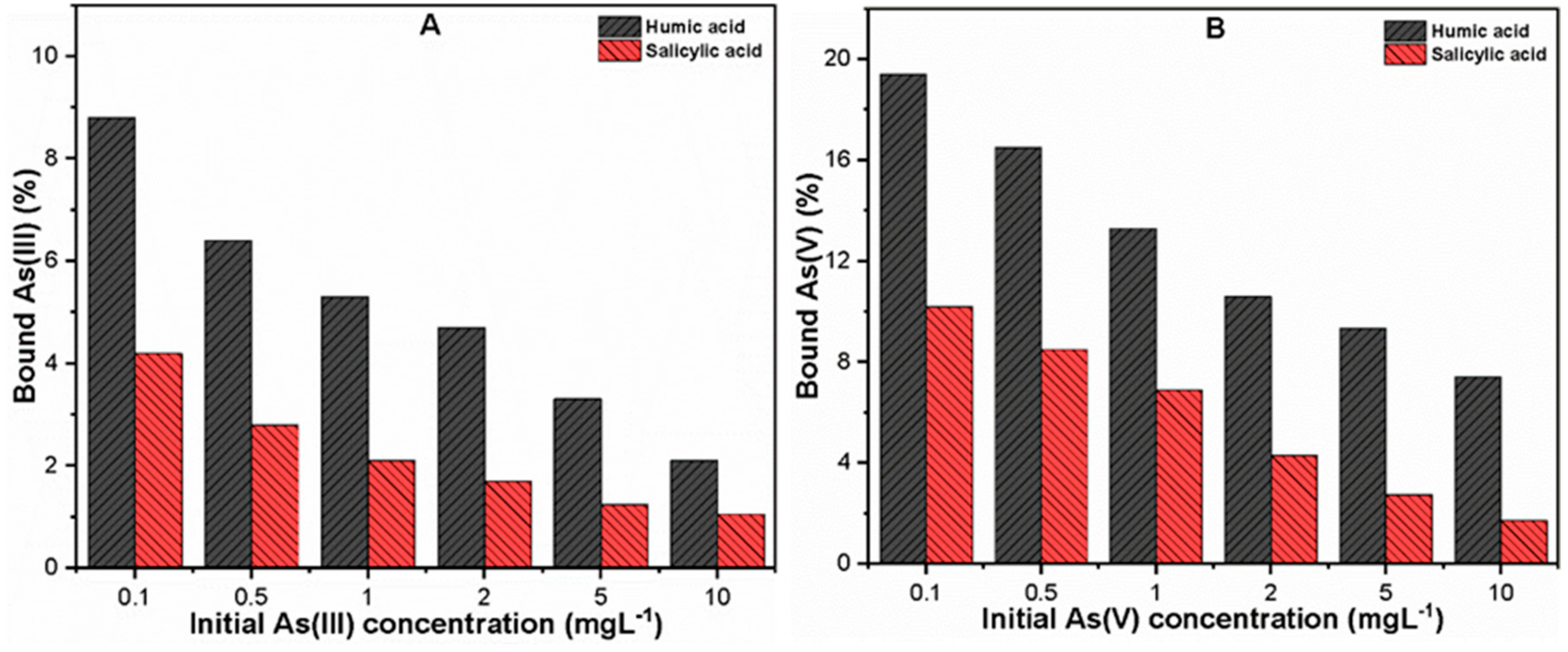
3.1. As Binding to Organic Ligands
3.2. Effects of Organic Ligands on As(III, V) and TOC Removal by C/F/S
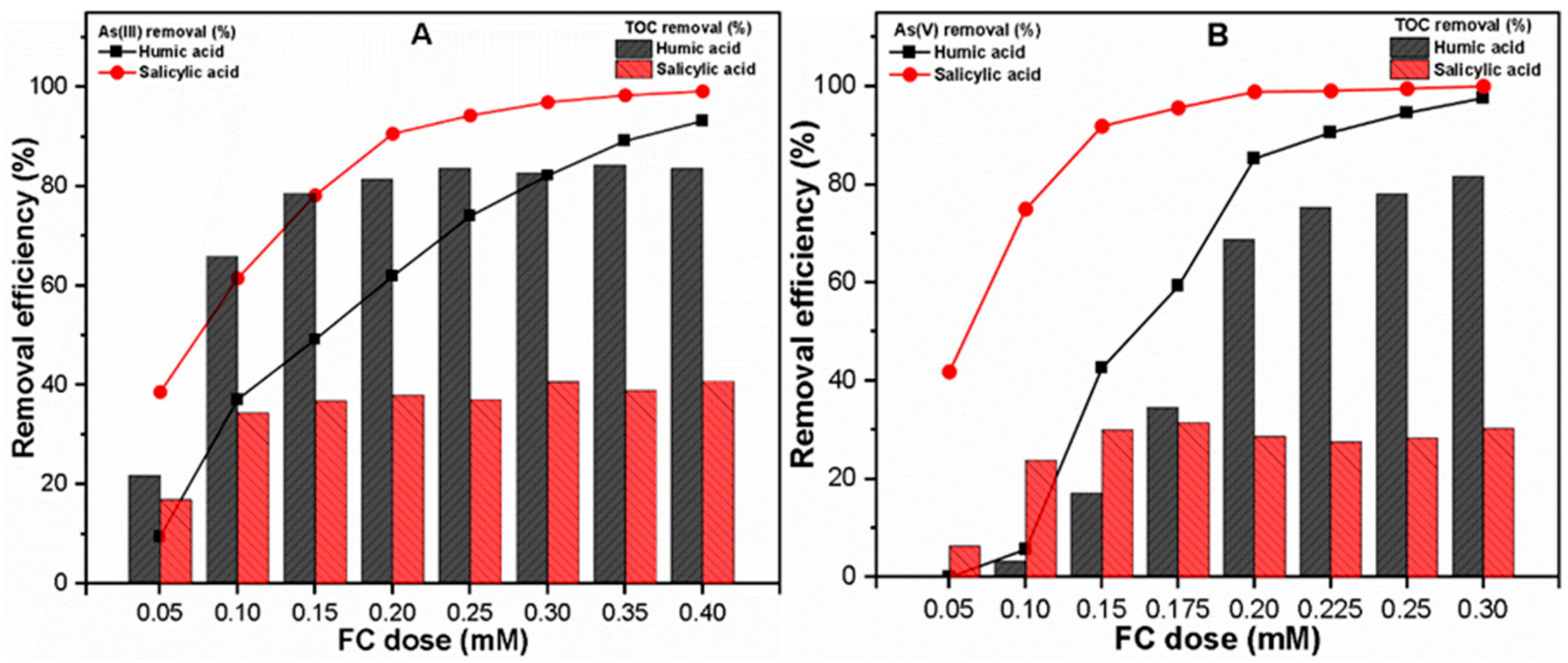
3.2.1. Varying FC Dose
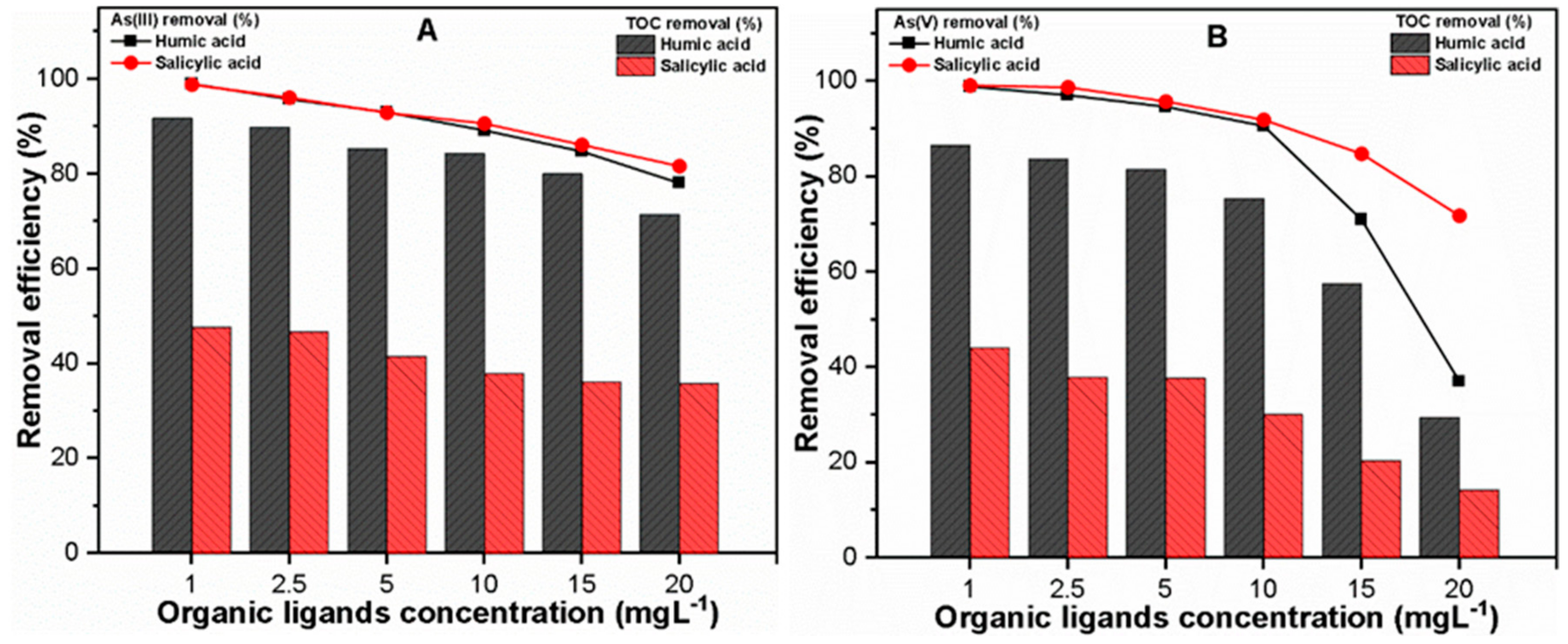
3.2.2. Varying Organic Ligands Concentration
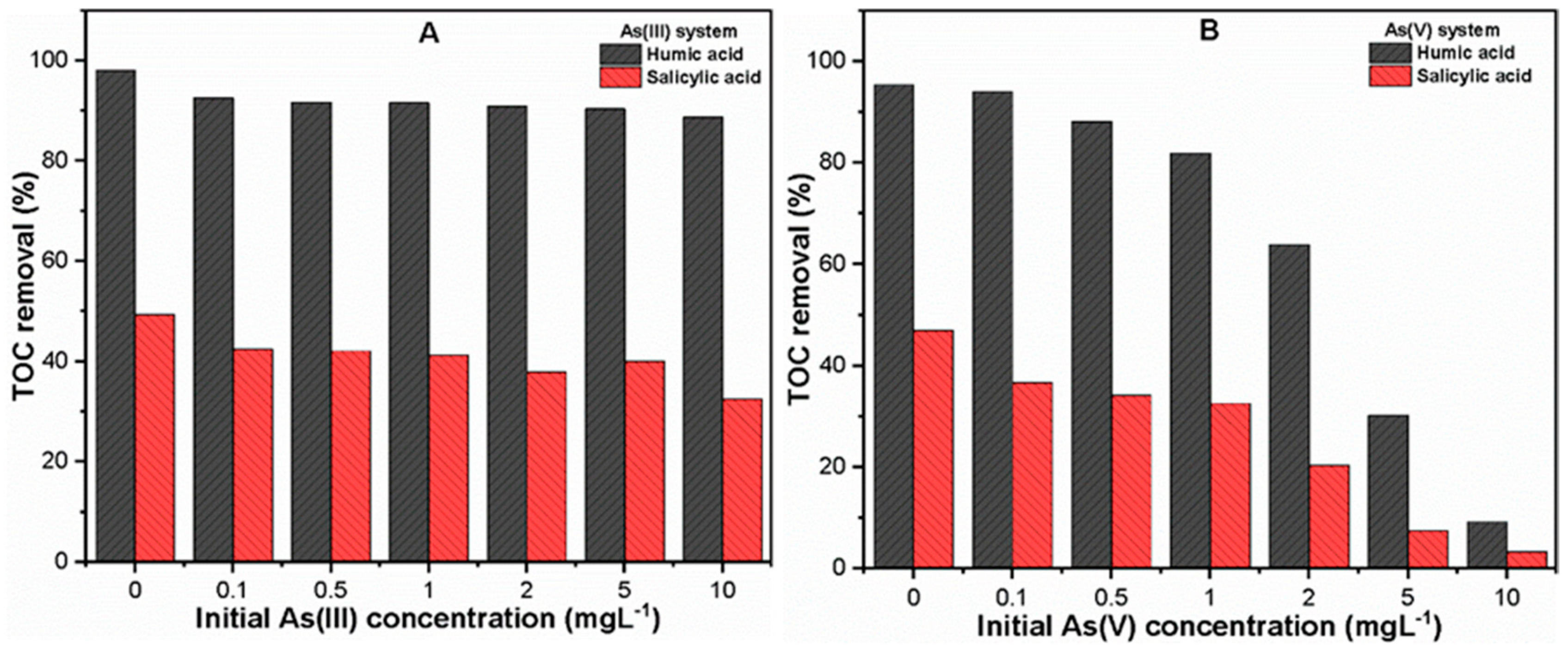
3.2.3. Varying As(III, V) Concentration
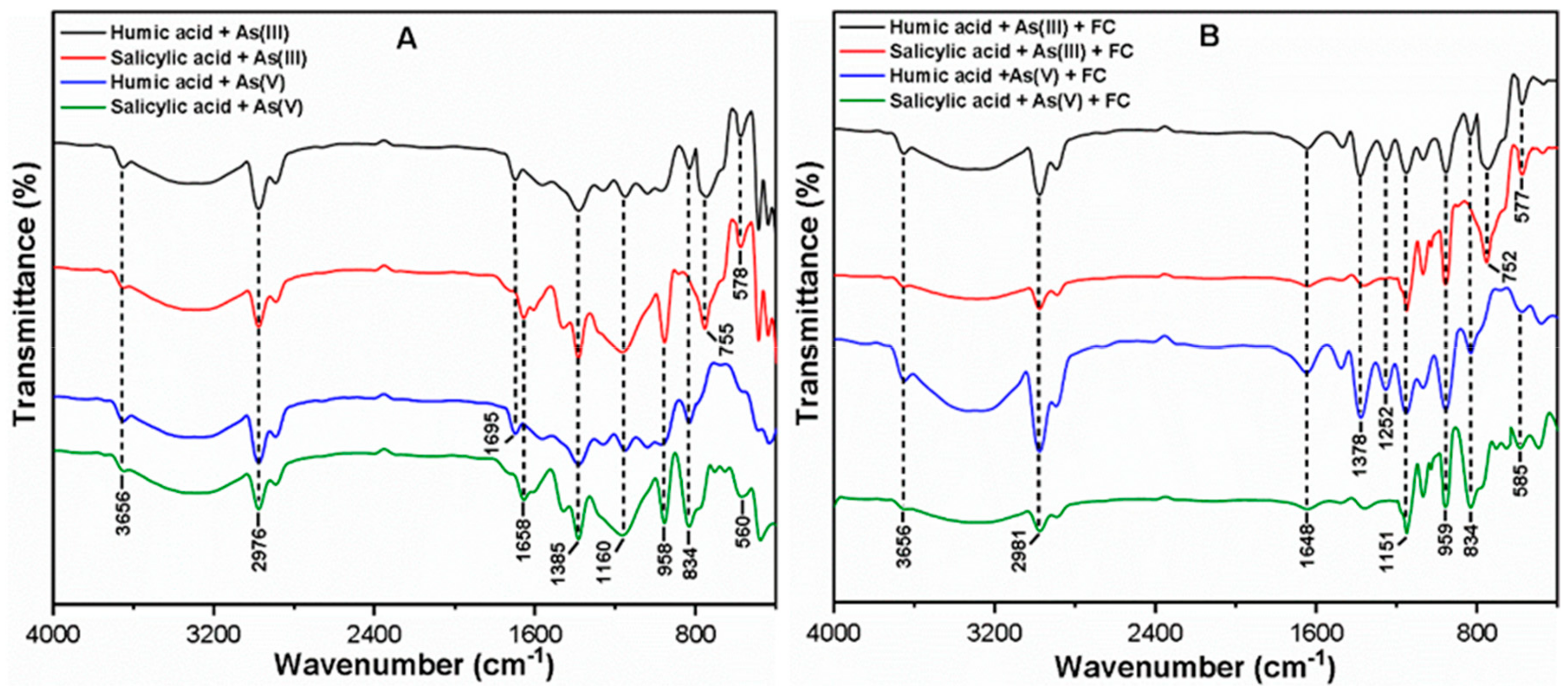
3.3. Interaction and Removal Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hughes, M.F. Arsenic toxicity and potential mechanisms of action. Toxicol. Lett. 2002, 133, 1–16. [Google Scholar] [CrossRef]
- Inam, M.A.; Khan, R.; Park, D.R.; Ali, B.A.; Uddin, A.; Yeom, I.T. Influence of pH and Contaminant Redox Form on the Competitive Removal of Arsenic and Antimony from Aqueous Media by Coagulation. Minerals 2018, 8, 574. [Google Scholar] [CrossRef]
- Ungureanu, G.; Santos, S.; Boaventura, R.; Botelho, C. Arsenic and antimony in water and wastewater: Overview of removal techniques with special reference to latest advances in adsorption. J. Environ. Manag. 2015, 151, 326–342. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, V.J.; Ilgen, A.G.; Mueller, S.H.; Trainor, T.P.; Goldfarb, R.J. Mobility and chemical fate of antimony and arsenic in historic mining environments of the Kantishna Hills district, Denali National Park and Preserve, Alaska. Chem. Geol. 2013, 335, 172–188. [Google Scholar] [CrossRef]
- Nath, B.; Stüben, D.; Mallik, S.B.; Chatterjee, D.; Charlet, L. Mobility of arsenic in West Bengal aquifers conducting low and high groundwater arsenic. Part I: Comparative hydrochemical and hydrogeological characteristics. Appl. Geochemistry 2008, 23, 977–995. [Google Scholar] [CrossRef]
- Sánchez-Rodas, D.; Gómez-Ariza, J.L.; Giráldez, I.; Velasco, A.; Morales, E. Arsenic speciation in river and estuarine waters from southwest Spain. Sci. Total Environ. 2005, 345, 207–217. [Google Scholar] [CrossRef]
- Hiller, E.; Lalinská, B.; Chovan, M.; Jurkovič, Ľ.; Klimko, T.; Jankulár, M.; Hovorič, R.; Šottník, P.; Fľaková, R.; Ženišová, Z. Arsenic and antimony contamination of waters, stream sediments and soils in the vicinity of abandoned antimony mines in the Western Carpathians, Slovakia. Appl. Geochemistry 2012, 27, 598–614. [Google Scholar] [CrossRef]
- Sharma, V.K.; Sohn, M. Aquatic arsenic: Toxicity, speciation, transformations and remediation. Environ. Int. 2009, 35, 743–759. [Google Scholar] [CrossRef]
- Buschmann, J.; Kappeler, A.; Lindauer, U.; Kistler, D.; Berg, M.; Sigg, L. Arsenite and arsenate binding to dissolved humic acids: Influence of pH, type of humic acid and aluminum. Environ. Sci. Technol. 2006, 40, 6015–6020. [Google Scholar] [CrossRef] [PubMed]
- Pallier, V.; Feuillade-Cathalifaud, G.; Serpaud, B.; Bollinger, J.-C. Effect of organic matter on arsenic removal during coagulation/flocculation treatment. J. Colloid Interface Sci. 2010, 342, 26–32. [Google Scholar] [CrossRef]
- Wullings, B.A.; Bakker, G.; van der Kooij, D. Concentration and diversity of uncultured Legionella spp. in two unchlorinated drinking water supplies with different concentrations of natural organic matter. Appl. Environ. Microbiol. 2011, 77, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.D.; Ternes, T.A. Water analysis: Emerging contaminants and current issues. Anal. Chem. 2014, 86, 2813–2848. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.A.; Tubić, A.; Agbaba, J.; Nikić, J.; Maletić, S.; Jazić, J.M.; Dalmacija, B. Response surface methodology investigation into the interactions between arsenic and humic acid in water during the coagulation process. J. Hazard. Mater. 2016, 312, 150–158. [Google Scholar] [CrossRef]
- Santschi, P.H.; Xu, C.; Zhang, S.; Schwehr, K.A.; Lin, P.; Yeager, C.M.; Kaplan, D.I. Recent advances in the detection of specific natural organic compounds as carriers for radionuclides in soil and water environments, with examples of radioiodine and plutonium. J. Environ. Radioact. 2017, 171, 226–233. [Google Scholar] [CrossRef]
- Knauer, K.; Homazava, N.; Junghans, M.; Werner, I. The influence of particles on bioavailability and toxicity of pesticides in surface water. Integr. Environ. Assess. Manag. 2017, 13, 585–600. [Google Scholar] [CrossRef] [PubMed]
- Matilainen, A.; Gjessing, E.T.; Lahtinen, T.; Hed, L.; Bhatnagar, A.; Sillanpää, M. An overview of the methods used in the characterisation of natural organic matter (NOM) in relation to drinking water treatment. Chemosphere 2011, 83, 1431–1442. [Google Scholar] [CrossRef]
- Fakour, H.; Lin, T.-F. Experimental determination and modeling of arsenic complexation with humic and fulvic acids. J. Hazard. Mater. 2014, 279, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Blodau, C. Mobilization of arsenic by dissolved organic matter from iron oxides, soils and sediments. Sci. Total Environ. 2006, 354, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Mulligan, C.N. Effect of natural organic matter on arsenic release from soils and sediments into groundwater. Environ. Geochem. Health 2006, 28, 197–214. [Google Scholar] [CrossRef]
- Saito, T.; Koopal, L.K.; van Riemsdijk, W.H.; Nagasaki, S.; Tanaka, S. Adsorption of humic acid on goethite: Isotherms, charge adjustments and potential profiles. Langmuir 2004, 20, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Vermeer, A.W.P.; van Riemsdijk, W.H.; Koopal, L.K. Adsorption of Humic Acid to Mineral Particles. 1. Specific and Electrostatic Interactions. Langmuir 1998, 14, 2810–2819. [Google Scholar] [CrossRef]
- Gu, B.; Schmitt, J.; Chen, Z.; Liang, L.; McCarthy, J.F. Adsorption and desorption of natural organic matter on iron oxide: Mechanisms and models. Environ. Sci. Technol. 1994, 28, 38–46. [Google Scholar] [CrossRef]
- Luo, L.; Zhang, S.; Shan, X.-Q.; Zhu, Y.-G. Effects of oxalate and humic acid on arsenate sorption by and desorption from a Chinese red soil. Water. Air. Soil Pollut. 2006, 176, 269–283. [Google Scholar] [CrossRef][Green Version]
- Rao, P.; Mak, M.S.H.; Liu, T.; Lai, K.C.K.; Lo, I.M.C. Effects of humic acid on arsenic (V) removal by zero-valent iron from groundwater with special references to corrosion products analyses. Chemosphere 2009, 75, 156–162. [Google Scholar] [CrossRef]
- Wang, Y.; Duan, J.; Liu, S.; Li, W.; van Leeuwen, J.; Mulcahy, D. Removal of As (III) and As (V) by ferric salts coagulation—Implications of particle size and zeta potential of precipitates. Sep. Purif. Technol. 2014, 135, 64–71. [Google Scholar] [CrossRef]
- Warwick, P.; Inam, E.; Evans, N. Arsenic’s interaction with humic acid. Environ. Chem. 2005, 2, 119–124. [Google Scholar] [CrossRef]
- Liu, G.; Cai, Y. Complexation of arsenite with dissolved organic matter: Conditional distribution coefficients and apparent stability constants. Chemosphere 2010, 81, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Redman, A.D.; Macalady, D.L.; Ahmann, D. Natural organic matter affects arsenic speciation and sorption onto hematite. Environ. Sci. Technol. 2002, 36, 2889–2896. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Inam, M.; Park, D.; Zam Zam, S.; Shin, S.; Khan, S.; Akram, M.; Yeom, I. Influence of Organic Ligands on the Colloidal Stability and Removal of ZnO Nanoparticles from Synthetic Waters by Coagulation. Processes 2018, 6, 170. [Google Scholar] [CrossRef]
- Hong, S.; Elimelech, M. Chemical and physical aspects of natural organic matter (NOM) fouling of nanofiltration membranes. J. Memb. Sci. 1997, 132, 159–181. [Google Scholar] [CrossRef]
- Inam, M.A.; Khan, R.; Park, D.R.; Khan, S.; Uddin, A.; Yeom, I.T. Complexation of Antimony with Natural Organic Matter: Performance Evaluation during Coagulation-Flocculation Process. Int. J. Environ. Res. Public Health 2019, 16, 1092. [Google Scholar] [CrossRef]
- Inam, M.A.; Khan, R.; Park, D.R.; Lee, Y.W.; Yeom, I.T. Removal of Sb(III) and Sb(V) by ferric chloride coagulation: Implications of Fe solubility. Water 2018, 10, 418. [Google Scholar] [CrossRef]
- Jabłońska-Czapla, M. Antimony, arsenic and chromium speciation studies in Biała Przemsza River (Upper Silesia, Poland) water by HPLC-ICP-MS. Int. J. Environ. Res. Public Health 2015, 12, 4739–4757. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Ofner, J.; Kappler, A. Formation of binary and ternary colloids and dissolved complexes of organic matter, Fe and As. Environ. Sci. Technol. 2010, 44, 4479–4485. [Google Scholar] [CrossRef] [PubMed]
- Gaskill, A., Jr.; Byrd, J.T.; Shuman, M.S. Fractionation and trace metal content of a commercial humic acid. J. Environ. Sci. Heal. Part A 1977, 12, 95–103. [Google Scholar] [CrossRef]
- Liu, N.; Liu, C.; Zhang, J.; Lin, D. Removal of dispersant-stabilized carbon nanotubes by regular coagulants. J. Environ. Sci. 2012, 24, 1364–1370. [Google Scholar] [CrossRef]
- Stone, A.T.; Torrents, A.; Smolen, J.; Vasudevan, D.; Hadley, J. Adsorption of organic compounds possessing ligand donor groups at the oxide/water interface. Environ. Sci. Technol. 1993, 27, 895–909. [Google Scholar] [CrossRef]
- Stumm, W. Reactivity at the mineral-water interface: Dissolution and inhibition. Colloids Surfaces A Physicochem. Eng. Asp. 1997, 120, 143–166. [Google Scholar] [CrossRef]
- Stone, A.T. Reactions of extracellular organic ligands with dissolved metal ions and mineral surfaces. Rev. Mineral. Geochemistry 1997, 35, 309–344. [Google Scholar]
- Jiang, J. The role of coagulation in water treatment. Curr. Opin. Chem. Eng. 2015, 8, 36–44. [Google Scholar] [CrossRef]
- Joseph, L.; Flora, J.R.V.; Park, Y.; Badawy, M.; Saleh, H.; Yoon, Y. Removal of natural organic matter from potential drinking water sources by combined coagulation and adsorption using carbon nanomaterials. Sep. Purif. Technol. 2012, 95, 64–72. [Google Scholar] [CrossRef]
- Ncibi, M.C.; Matilainen, A. Removal of natural organic matter in drinking water treatment by coagulation: A comprehensive review. Chemosphere 2018, 190, 54–71. [Google Scholar] [CrossRef]
- Sharp, E.L.; Parson, S.A.; Jefferson, B. Coagulation of NOM: Linking character to treatment. Water Sci. Technol. 2006, 53, 67–76. [Google Scholar] [CrossRef]
- Stumm, W. The Inner-Sphere Surface Complex. Aquat. Chem. 1995, 244, 1–32. [Google Scholar]
- Zhang, G.; Liu, H.; Liu, R.; Qu, J. Removal of phosphate from water by a Fe–Mn binary oxide adsorbent. J. Colloid Interface Sci. 2009, 335, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Edzwald, J.K.; Tobiason, J.E. Enhanced coagulation: US requirements and a broader view. Water Sci. Technol. 1999, 40, 63–70. [Google Scholar] [CrossRef]
- Alberts, J.J.; Filip, Z. Metal Binding in Estuarine Humic and Fulvic Acids: FTIR Analysis of Humic Acid-Metal Complexes. Environ. Technol. 2010, 3330. [Google Scholar] [CrossRef]
- Carbonaro, R.F.; Atalay, Y.B.; Di, D.M. Linear free energy relationships for metal–ligand complexation: Bidentate binding to negatively-charged oxygen donor atoms. Geochim. Cosmochim. Acta 2011, 75, 2499–2511. [Google Scholar] [CrossRef]
- Vlachos, N.; Skopelitis, Y.; Psaroudaki, M.; Konstantinidou, V.; Chatzilazarou, A.; Tegou, E. Applications of Fourier transform-infrared spectroscopy to edible oils. Anal. Chim. Acta 2006, 573–574, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Buschmann, J.; Sigg, L. Antimony (III) binding to humic substances: Influence of pH and type of humic acid. Environ. Sci. Technol. 2004, 38, 4535–4541. [Google Scholar] [CrossRef] [PubMed]
- Matrajt, G.; Borg, J.; Raynal, P.I.; Djouadi, Z.; Hendecourt, L.; Flynn, G.; Debo, D. Astrophysics FTIR and Raman analyses of the Tagish Lake meteorite: Relationship with the aliphatic hydrocarbons observed. Astron. Astrophys. 2004, 990, 983–990. [Google Scholar] [CrossRef]
- Khan, R.; Inam, M.A.; Zam, S.Z.; Park, D.R.; Yeom, I.T. Assessment of Key Environmental Factors Influencing the Sedimentation and Aggregation Behavior of Zinc Oxide Nanoparticles in Aquatic Environment. Water 2018, 10, 660. [Google Scholar] [CrossRef]
- Zhang, G.-S.; Qu, J.-H.; Liu, H.-J.; Liu, R.-P.; Li, G.-T. Removal mechanism of As (III) by a novel Fe–Mn binary oxide adsorbent: Oxidation and sorption. Environ. Sci. Technol. 2007, 41, 4613–4619. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Organic Ligands | As Type | Langmuir Fitting | Freundlich Fitting | ||||
|---|---|---|---|---|---|---|---|
| KL (L.mg−1) | qm (mg.g−1) | R2 | KF (mg.g−1) (L.mg−1) | n | R2 | ||
| Humic acid | As(III) | 0.579 | 608.36 | 0.995 | 204.72 | 1.64 | 0.997 |
| As(V) | 1.954 | 445.11 | 0.998 | 222.54 | 2.94 | 0.933 | |
| Salicylic acid | As(III) | 3.531 | 272.29 | 0.956 | 173.79 | 3.88 | 0.992 |
| As(V) | 4.844 | 379.87 | 0.992 | 234.79 | 4.02 | 0.944 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inam, M.A.; Khan, R.; Akram, M.; Khan, S.; Park, D.R.; Yeom, I.T. Interaction of Arsenic Species with Organic Ligands: Competitive Removal from Water by Coagulation-Flocculation-Sedimentation (C/F/S). Molecules 2019, 24, 1619. https://doi.org/10.3390/molecules24081619
Inam MA, Khan R, Akram M, Khan S, Park DR, Yeom IT. Interaction of Arsenic Species with Organic Ligands: Competitive Removal from Water by Coagulation-Flocculation-Sedimentation (C/F/S). Molecules. 2019; 24(8):1619. https://doi.org/10.3390/molecules24081619
Chicago/Turabian StyleInam, Muhammad Ali, Rizwan Khan, Muhammad Akram, Sarfaraz Khan, Du Ri Park, and Ick Tae Yeom. 2019. "Interaction of Arsenic Species with Organic Ligands: Competitive Removal from Water by Coagulation-Flocculation-Sedimentation (C/F/S)" Molecules 24, no. 8: 1619. https://doi.org/10.3390/molecules24081619
APA StyleInam, M. A., Khan, R., Akram, M., Khan, S., Park, D. R., & Yeom, I. T. (2019). Interaction of Arsenic Species with Organic Ligands: Competitive Removal from Water by Coagulation-Flocculation-Sedimentation (C/F/S). Molecules, 24(8), 1619. https://doi.org/10.3390/molecules24081619








