Corrosion Assessment of Myo-Inositol Sugar Alcohol as a Phase Change Material in Storage Systems Connected to Fresnel Solar Plants
Abstract
1. Introduction
2. Results and Discussion
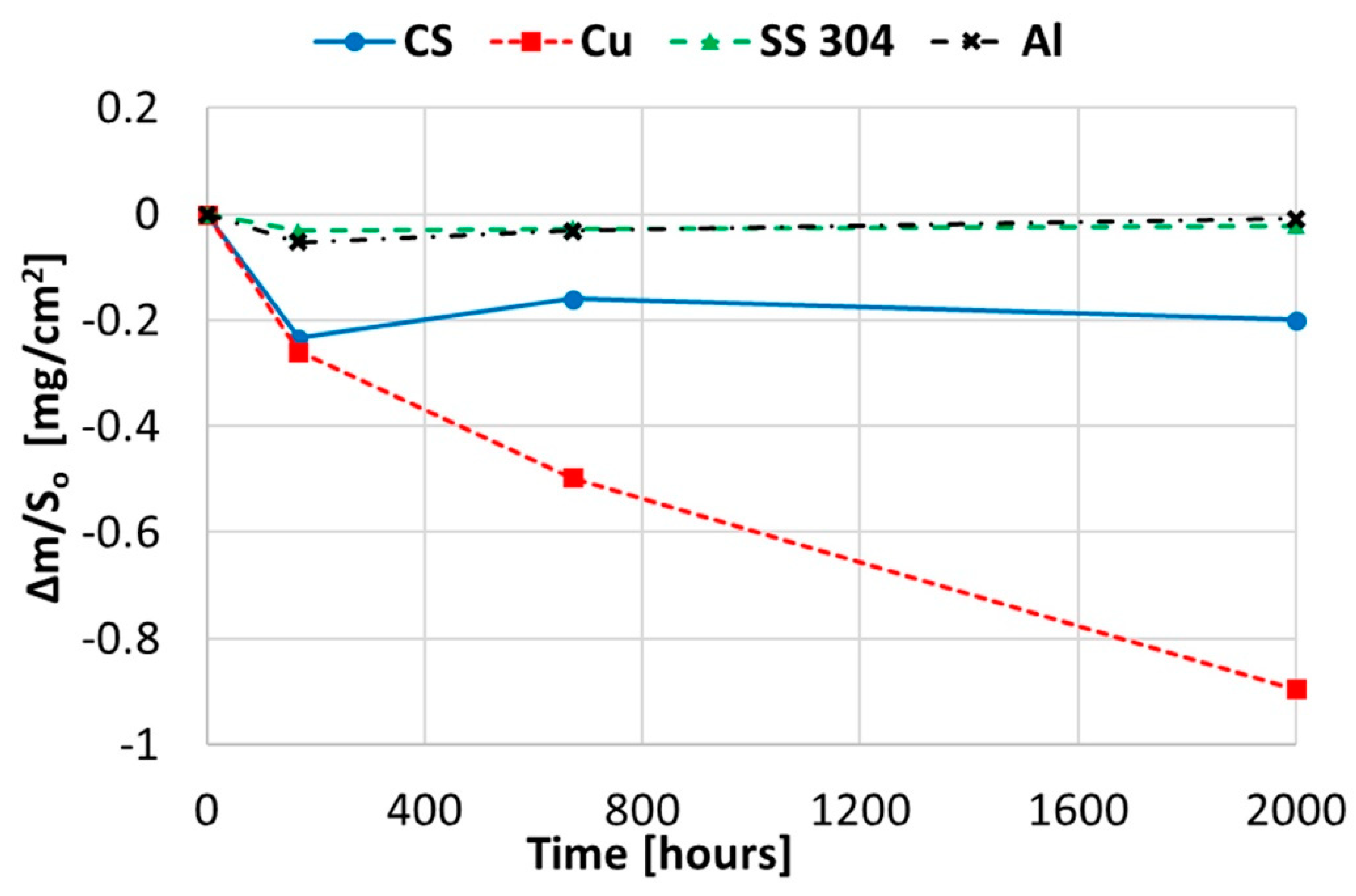
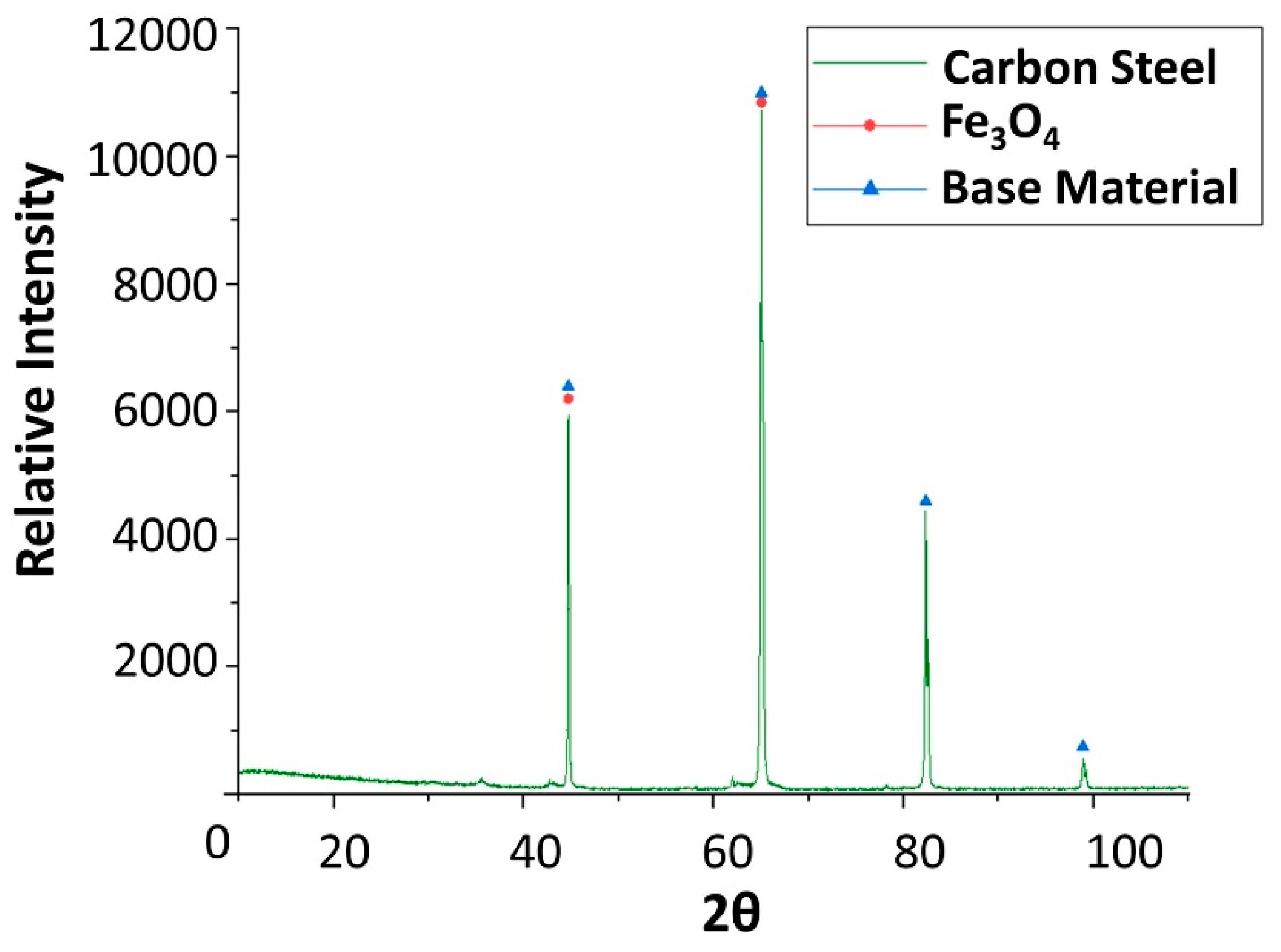
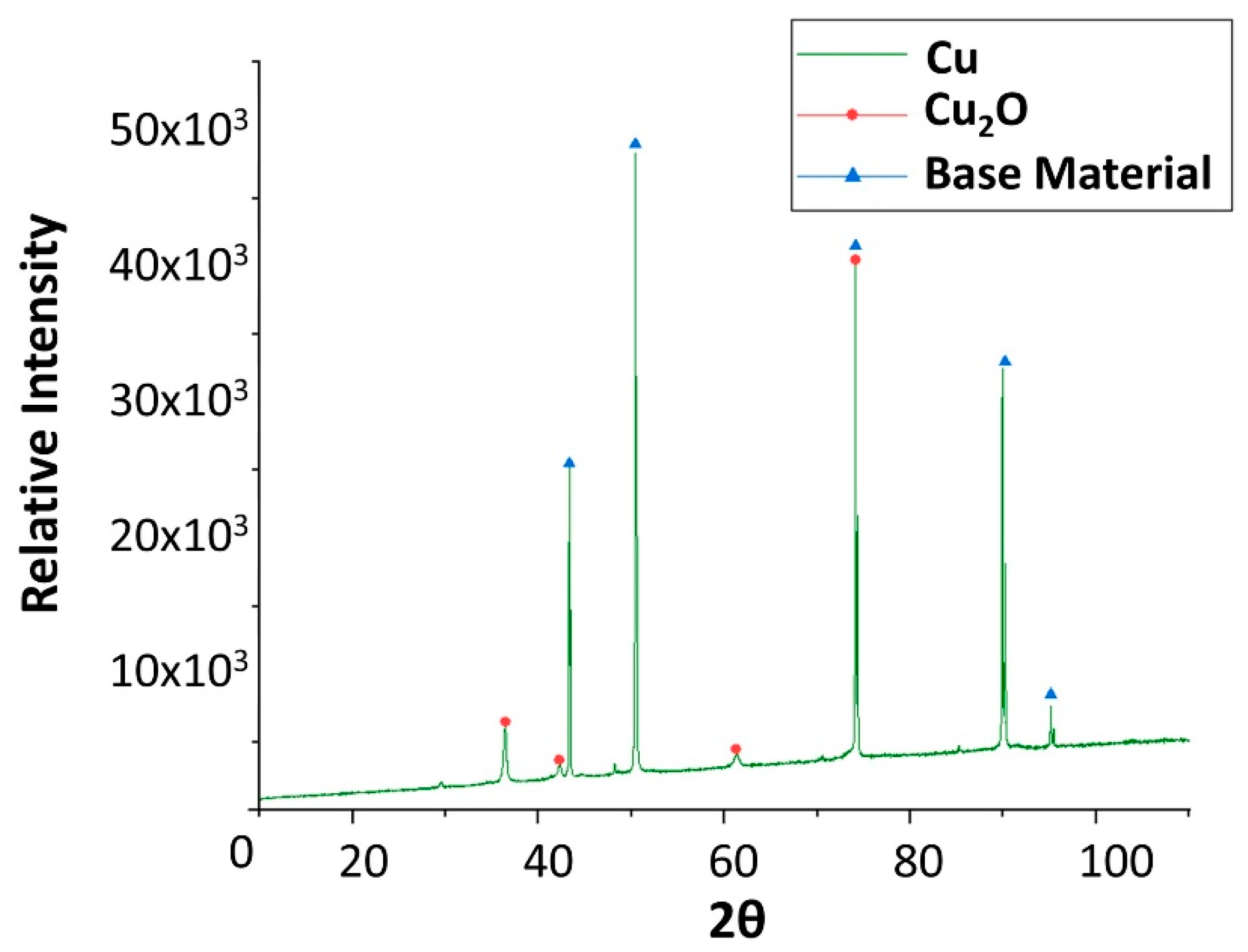
2.1. Corrosion Produced by Solar Salt
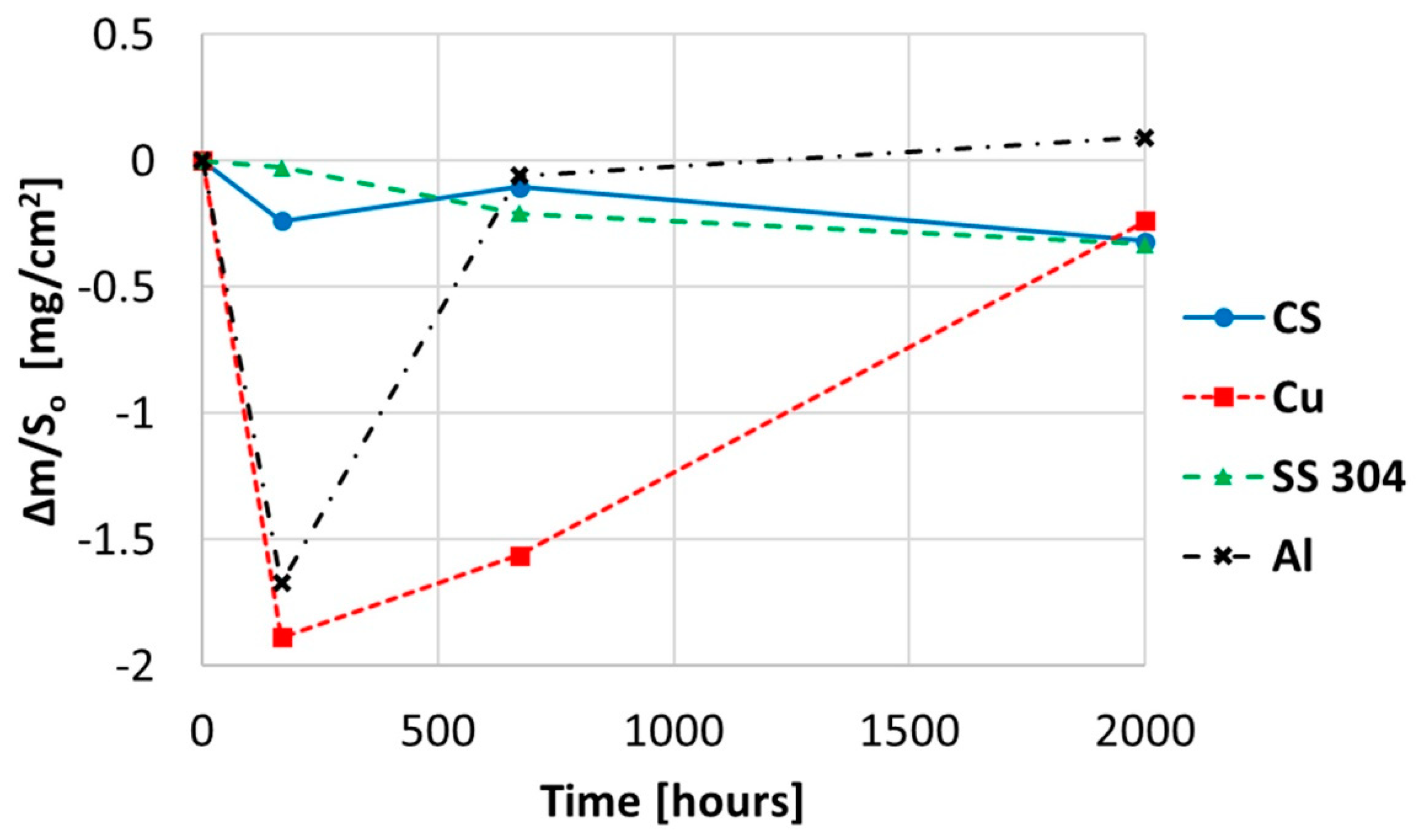
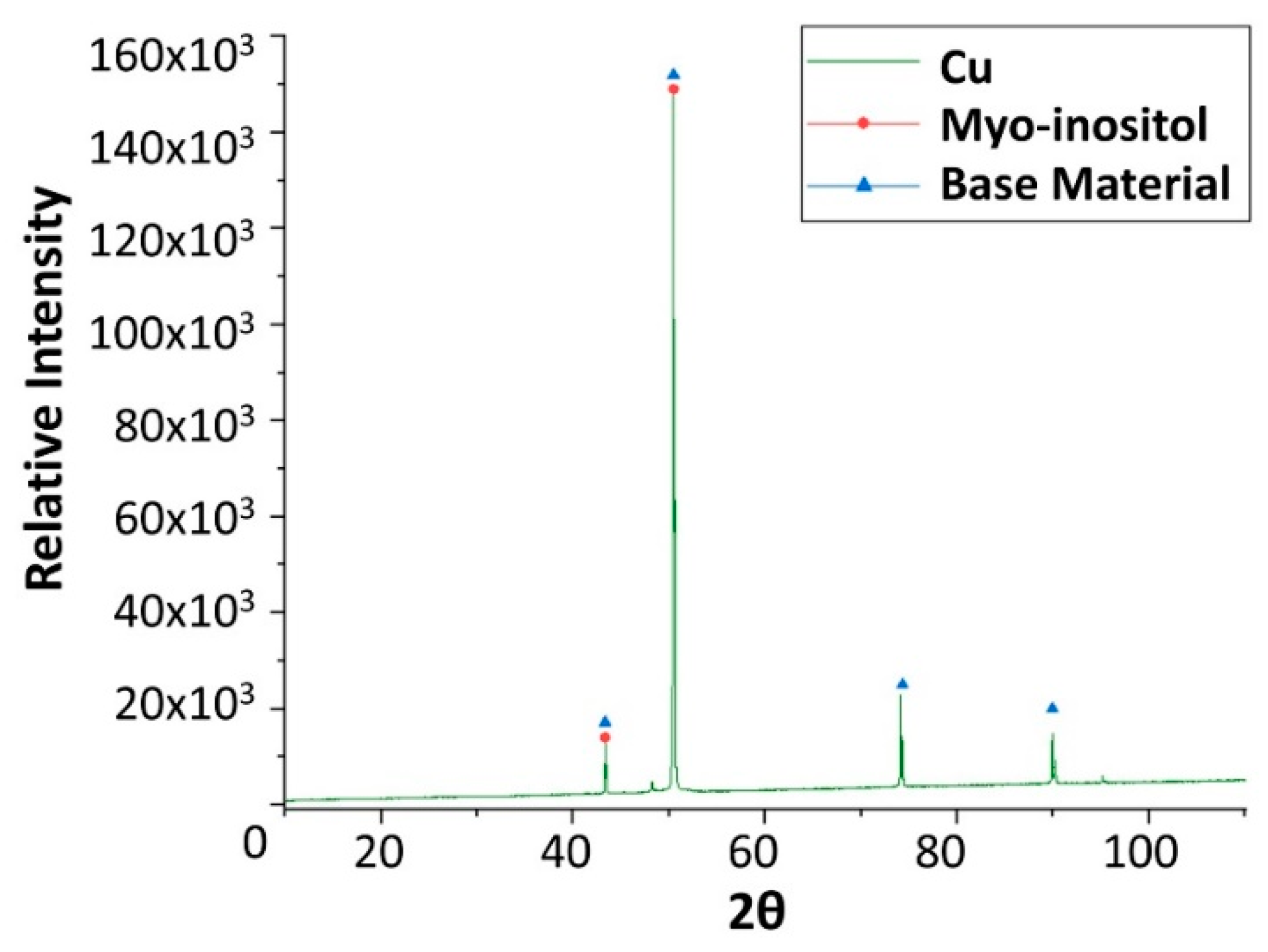
2.2. Corrosion Produced by Myo-Inositol (C6H12O6)
2.3. Uncertainties Analysis
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The Global Risks Report 2018, 13th ed.; World Economic Forum: Geneva, Switzerland, 2018; ISBN 978-1-944835-15-6.
- Kost, T.; Mayer, J.N.; Thomsen, J.; Hartmann, N.; Senkpiel, C.; Philipps, S.P.; Nold, S.; Lude, S.; Saad, N.; Schmid, J.; et al. Study: Levelized Cost of Electricity- Renewable Energy Technologies. Available online: https://www.ise.fraunhofer.de/en/publications/studies/cost-of-electricity (accessed on 6 March 2018).
- Fernández, A.G.; Galleguillos, H.; Fuentealba, E.; Pérez, F.J. Thermal characterization of HITEC molten salt for energy storage in solar linear concentrated technology. J. Therm. Anal. Calorim. 2015, 122, 3–9. [Google Scholar] [CrossRef]
- Zalba, B.; Marín, J.M.; Cabeza, L.F.; Mehling, H. Review on thermal energy storage with phase change: Materials, heat transfer analysis and applications. Appl. Therm. Eng. 2003, 23, 251–283. [Google Scholar] [CrossRef]
- Haillot, D.; Bauer, T.; Kröner, U.; Tamme, R. Thermal analysis of phase change materials in the temperature range 120–150 °C. Thermochim. Acta 2011, 513, 49–59. [Google Scholar] [CrossRef]
- Bayón, R.; Coco, S.; Barcenilla, M.; Espinet, P.; Imbuluzqueta, G.; Hidalgo, J.; Rojas, E. Feasibility of storing latent heat with liquid crystals. Proof of concept at lab scale. Appl. Sci. 2016, 6, 121. [Google Scholar] [CrossRef]
- Miró, L.; Barreneche, C.; Ferrer, G.; Solé, A.; Martorell, I.; Cabeza, L.F. Health hazard, cycling and thermal stability as key parameters when selecting a suitable phase change material (PCM). Thermochim. Acta 2016, 627, 39–47. [Google Scholar] [CrossRef]
- Gasia, J.; Martin, M.; Solé, A.; Barreneche, C.; Cabeza, L.F. Phase Change Material Selection for Thermal Processes Working under Partial Load Operating Conditions in the Temperature Range between 120 and 200 °C. Appl. Sci. 2017, 7, 722. [Google Scholar] [CrossRef]
- Maldonado, J.M.; Fullana-Puig, M.; Martín, M.; Solé, A.; Fernández, Á.G.; De Gracia, A.; Cabeza, L.F. Phase change material selection for thermal energy storage at high temperature range between 210 °C and 270 °C. Energies 2018, 11, 861. [Google Scholar] [CrossRef]
- Walczak, M.; Pineda, F.; Fernández, Á.G.; Mata-Torres, C.; Escobar, R.A. Materials corrosion for thermal energy storage systems in concentrated solar power plants. Renew. Sustain. Energy Rev. 2018, 86, 22–44. [Google Scholar] [CrossRef]
- Fernández, A.G.; Lasanta, M.I.; Pérez, F.J. Molten salt corrosion of stainless steels and low-Cr steel in CSP plants. Oxid. Met. 2012, 78, 329–348. [Google Scholar] [CrossRef]
- Fernández, A.G.; Muñoz-Sánchez, B.; Nieto-Maestre, J.; García-Romero, A. High temperature corrosion behavior on molten nitrate salt-based nanofluids for CSP plants. Renew. Energy 2019, 130, 902–909. [Google Scholar] [CrossRef]
- Soleimani Dorcheh, A.; Durham, R.N.; Galetz, M.C. Corrosion behavior of stainless and low-chromium steels and IN625 in molten nitrate salts at 600 °C. Sol. Energy Mater. Sol. Cells 2016, 144, 109–116. [Google Scholar] [CrossRef]
- Ferrer, G.; Solé, A.; Barreneche, C.; Martorell, I.; Cabeza, L.F. Corrosion of metal containers for use in PCM energy storage. Renew. Energy 2015, 76, 465–469. [Google Scholar] [CrossRef]
- Solé, A.; Miró, L.; Barreneche, C.; Martorell, I.; Cabeza, L.F. Corrosion of metals and salt hydrates used for thermochemical energy storage. Renew. Energy 2015, 75, 519–523. [Google Scholar] [CrossRef]
- Vasu, A.; Hagos, F.Y.; Noor, M.M.; Mamat, R.; Azmi, W.H.; Abdullah, A.A.; Ibrahim, T.K. Corrosion effect of phase change materials in solar thermal energy storage application. Renew. Sustain. Energy Rev. 2017, 76, 19–33. [Google Scholar] [CrossRef]
- Solé, A.; Neumann, H.; Niedermaier, S.; Martorell, I.; Schossig, P.; Cabeza, L.F. Stability of sugar alcohols as PCM for thermal energy storage. Sol. Energy Mater. Sol. Cells 2014, 126, 125–134. [Google Scholar] [CrossRef]
- Holman, J. Experimental Methods for Engineers, 8th ed.; McGraw-Hill Companies: New York, NY, USA, 2012. [Google Scholar]
- ASTM Committee G-1 on Corrosion of Metals. Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimes; ASTM International: West Conshohocken, PA, USA, 2017. [Google Scholar] [CrossRef]
Sample Availability: Myo-inositol, NaNO3, KNO3 and tested materials, copper (Cu), aluminium (Al), carbon steel (AISI 1090) and stainless steel (AISI 304). |






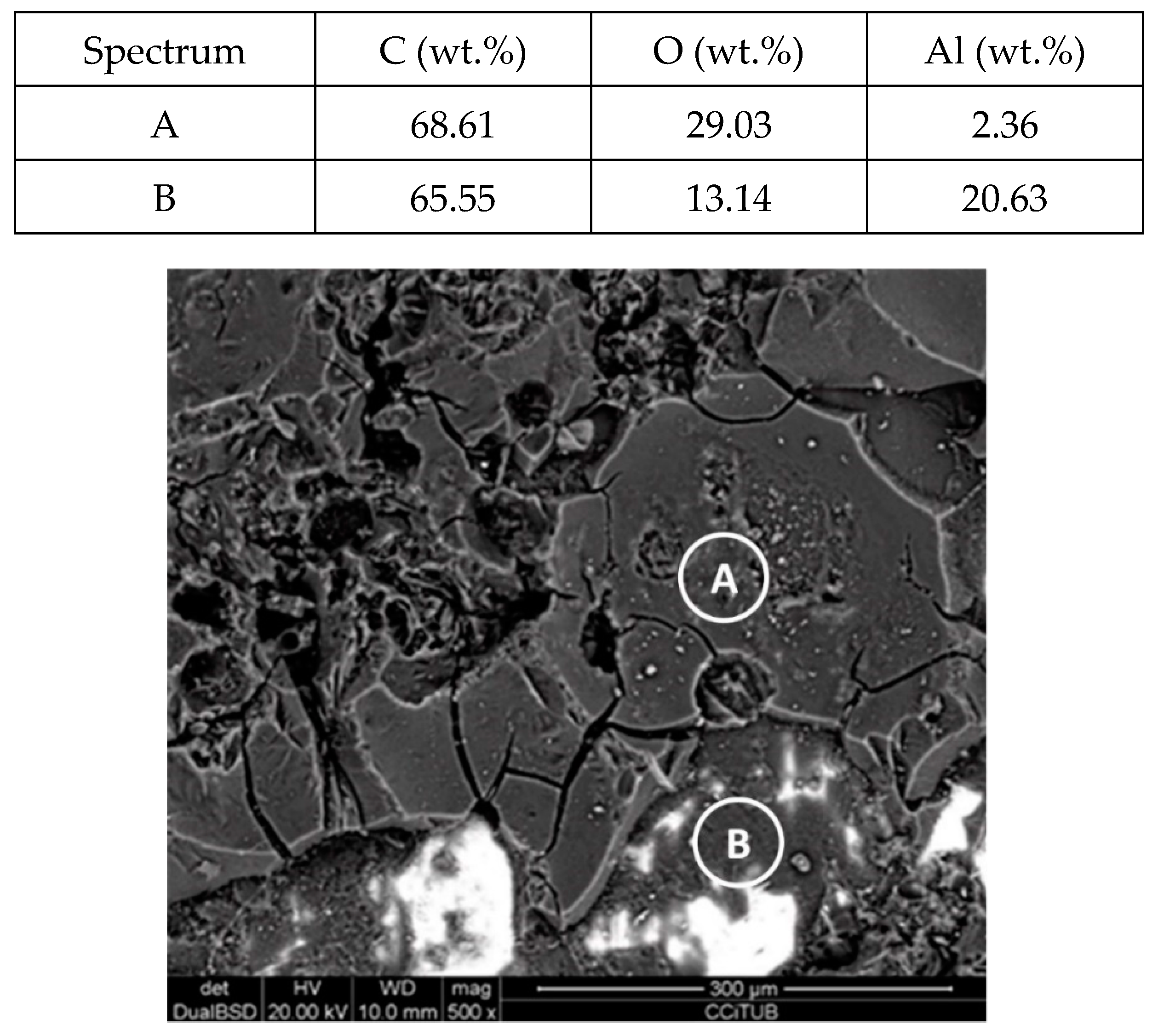







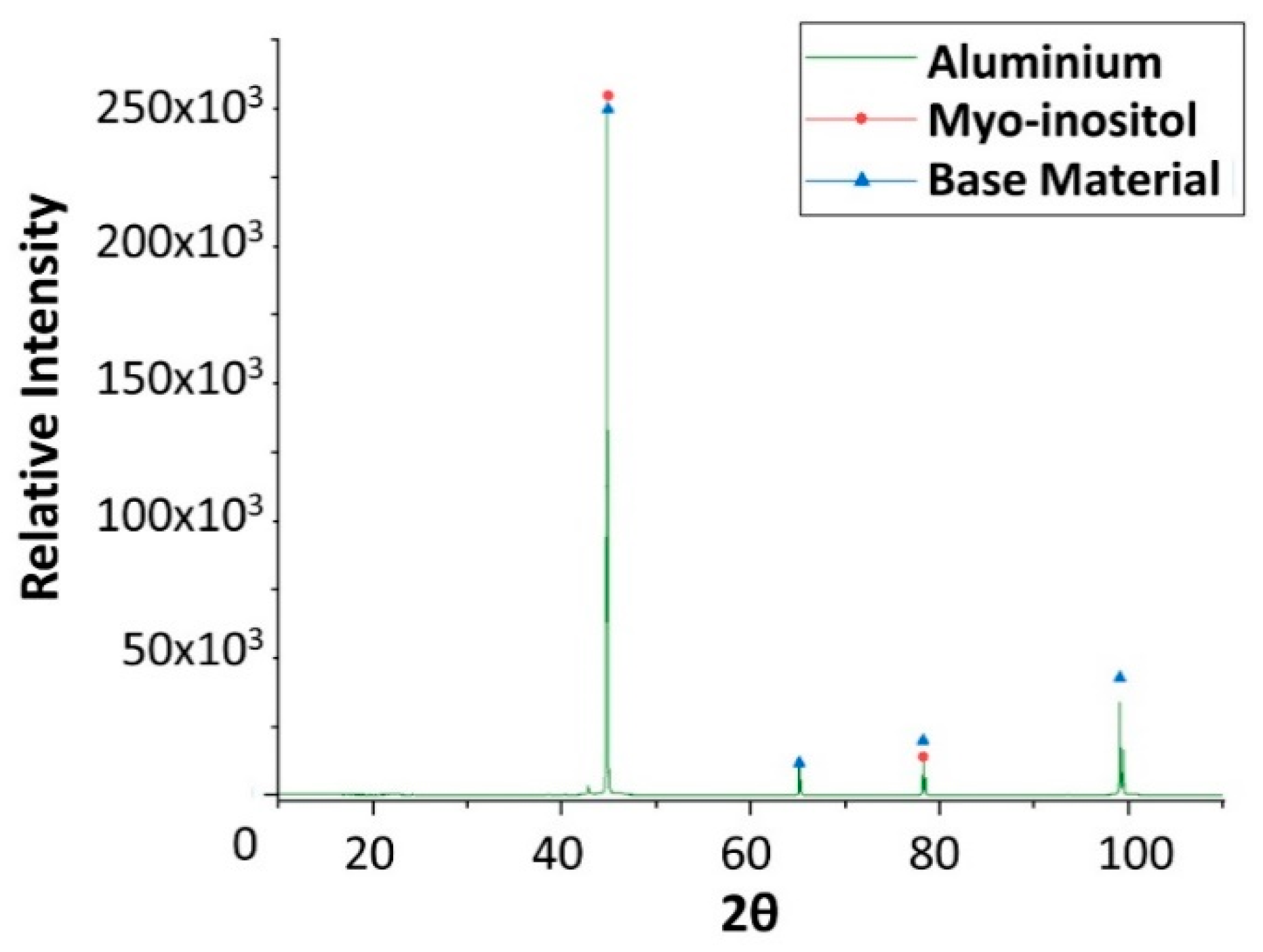
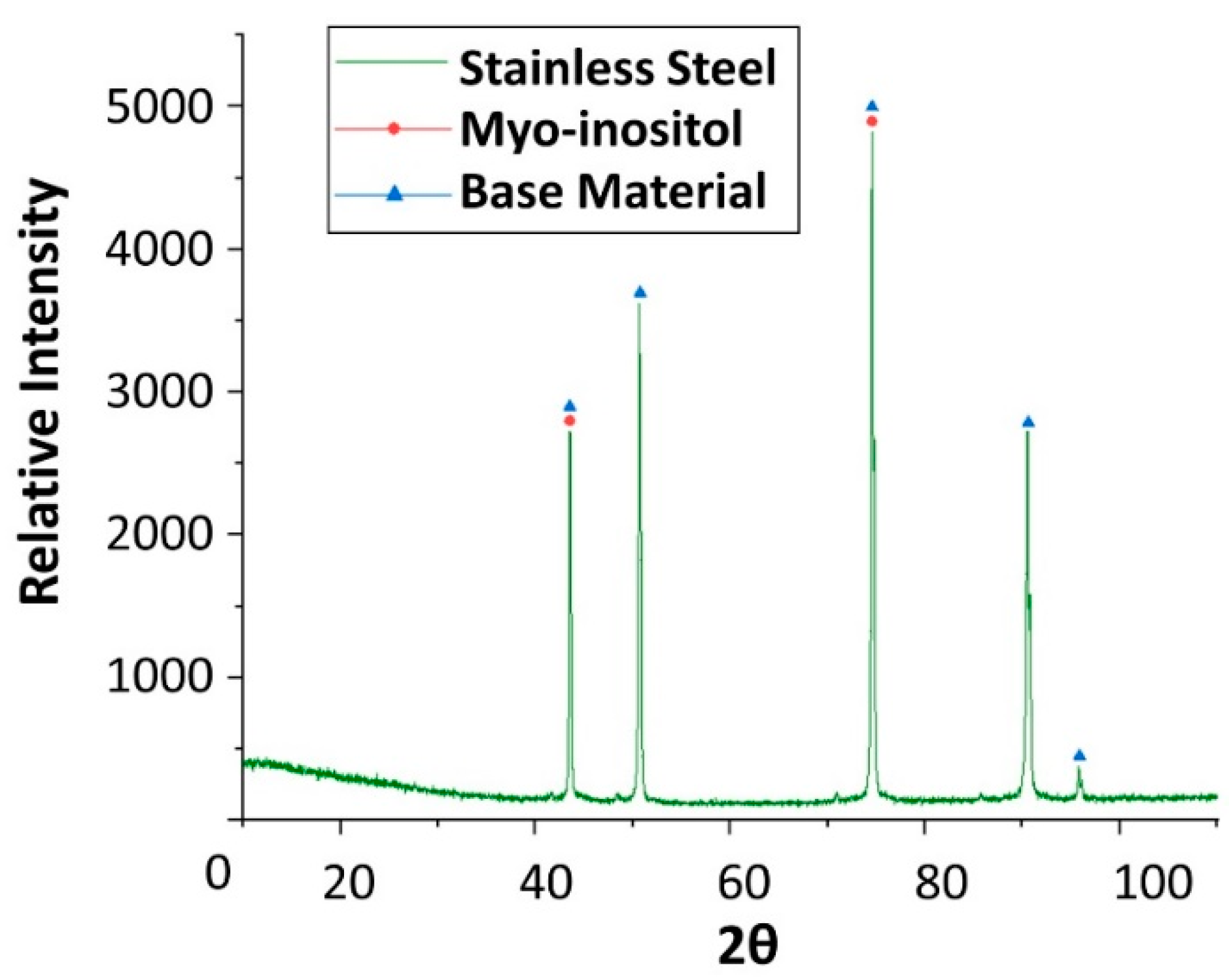
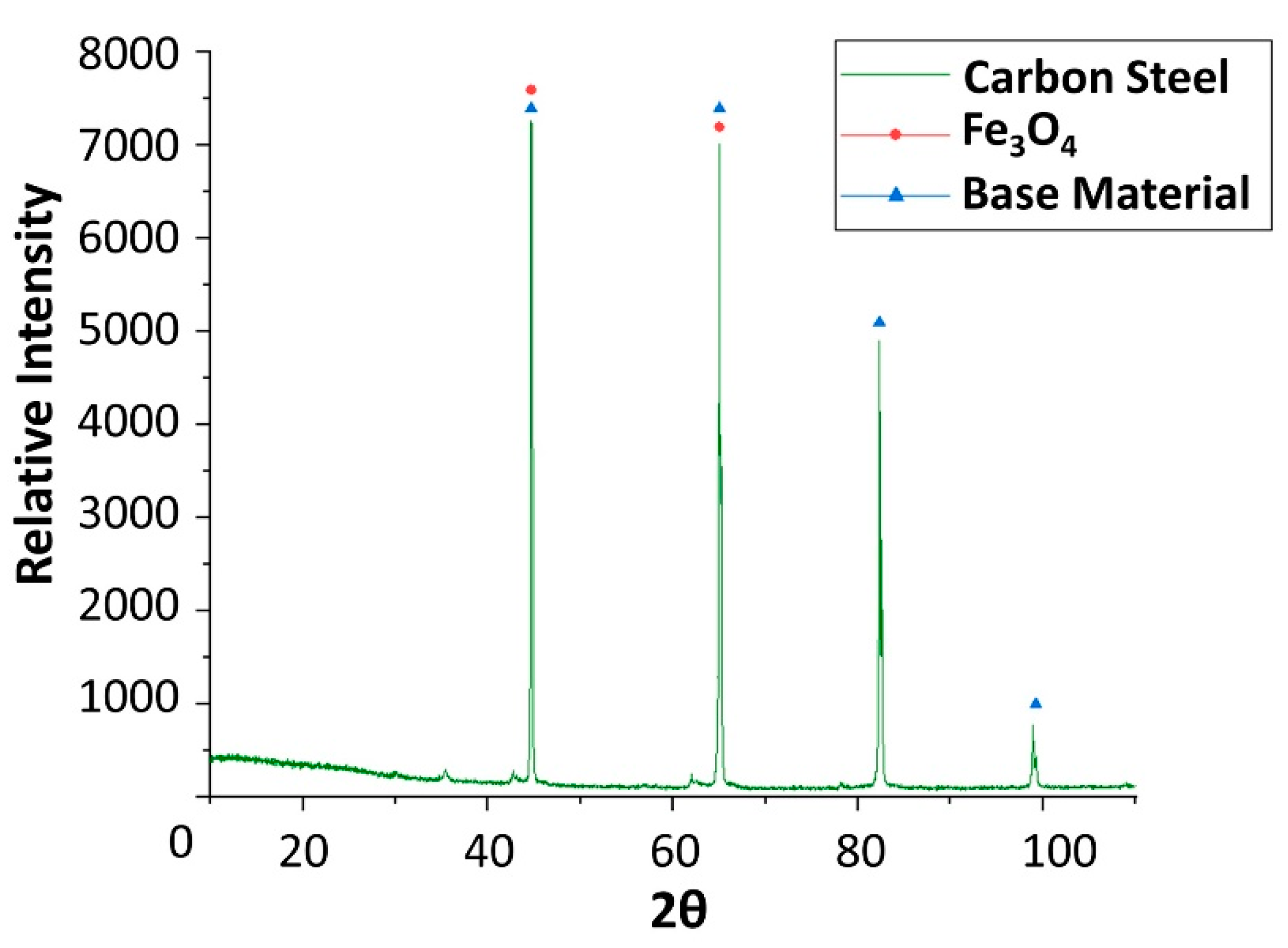


| Material | Price [€/kg] | Melting Temperature [°C] | Melting Enthalpy [J/g] | Ref. |
|---|---|---|---|---|
| Myo-inositol (C6H12O6) | 8–10 | 220 | 190–223 | [9,17] |
| Solar salt (40 wt.% KNO3/ 60 wt.% NaNO3) | 33 | 222 | 94–100 | [9,11] |
| Solar Salt | Myo-inositol | |
|---|---|---|
| Corrosion Rate [mm/year] | ||
| Aluminium | 0.0015 | 0.0150 |
| Stainless Steel AISI 304 | 0.0012 | 0.0182 |
| Carbon Steel (AISI 1090) | 0.0111 | 0.0178 |
| Copper | 0.0444 | 0.0116 |
| Parameter | Units | Sensor | Accuracy |
|---|---|---|---|
| Weight | g | Mettler Toledo AG135 | ±0.00001 g |
| Length | mm | Digital caliper | ±0.01 mm |
| Width | mm | Digital caliper | ±0.01 mm |
| Thickness | mm | Digital caliper | ±0.01 mm |
| Weight Difference | Area | Mass Loss | |
|---|---|---|---|
| [g] | [mm2] | [g/mm2] | |
| Carbon Steel (AISI 1090) | 1.33 × 10−3 | 569.125 | 2.337 × 10−6 |
| 1.05 × 10−3 | 658.436 | 1.595 × 10−6 | |
| 1.33 × 10−3 | 667.290 | 1.993 × 10−6 | |
| Copper | 1.55 × 10−3 | 595.823 | 2.601 × 10−6 |
| 3.18 × 10−3 | 640.614 | 4.964 × 10−6 | |
| 5.62 × 10−3 | 627.983 | 8.949 × 10−6 | |
| Stainless steel (AISI 304) | 0.18 × 10−3 | 552.278 | 3.259 × 10−6 |
| 0.18 × 10−3 | 656.986 | 2.740 × 10−6 | |
| 0.14 × 10−3 | 638.872 | 2.191 × 10−6 | |
| Aluminium | 0.3 × 10−3 | 569.180 | 5.271 × 10−6 |
| 0.21 × 10−3 | 643.462 | 3.264 × 10−6 | |
| 0.06 × 10−3 | 647.667 | 9.264 × 10−6 |
| Uncertainty | Weight Difference | Area | Mass Loss | |||
|---|---|---|---|---|---|---|
| Steels | [± g] | [± %] | [± mm2] | [± %] | [± g/mm2] | [± %] |
| Carbon Steel (AISI 1090) | 0.00001 | 0.752 | 0.069 | 0.012 | 1.757 × 10−8 | 0.752 |
| 0.00001 | 0.952 | 0.069 | 0.011 | 1.519 × 10−8 | 0.953 | |
| 0.00001 | 0.752 | 0.069 | 0.010 | 1.499 × 10−8 | 0.752 | |
| Copper | 0.00001 | 0.645 | 0.069 | 0.012 | 1.679 × 10−8 | 0.645 |
| 0.00001 | 0.314 | 0.069 | 0.011 | 1.562 × 10−8 | 0.315 | |
| 0.00001 | 0.178 | 0.069 | 0.011 | 1.595 × 10−8 | 0.178 | |
| Stainless steel (AISI 304) | 0.00001 | 5.556 | 0.069 | 0.013 | 1.81 × 10−8 | 5.556 |
| 0.00001 | 5.556 | 0.069 | 0.011 | 1.522 × 10−8 | 5.556 | |
| 0.00001 | 7.143 | 0.069 | 0.011 | 1.565 × 10−8 | 7.143 | |
| Aluminum | 0.00001 | 3.333 | 0.069 | 0.012 | 1.757 × 10−8 | 3.333 |
| 0.00001 | 4.762 | 0.069 | 0.011 | 1.554 × 10−8 | 4.762 | |
| 0.00001 | 16.667 | 0.069 | 0.011 | 1.544 × 10−8 | 16.667 | |
| Steels | Weight (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Al | Mn | Ni | Cr | P | C | S | Fe | Cu | Mo | |
| Stainless steel (AISI 304) | - | 1.7 | 8.04 | 18.28 | - | - | - | Balance | - | 0.27 |
| Carbon steel (AISI 1090) | - | 0.6–0.9 | - | - | 0.04 | 0.85–0.98 | 0.05 | Balance | - | - |
| Aluminium | 100 | - | - | - | - | - | - | - | - | - |
| Copper | - | - | - | - | - | - | - | - | 100 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maldonado, J.M.; Fernández, Á.G.; Cabeza, L.F. Corrosion Assessment of Myo-Inositol Sugar Alcohol as a Phase Change Material in Storage Systems Connected to Fresnel Solar Plants. Molecules 2019, 24, 1383. https://doi.org/10.3390/molecules24071383
Maldonado JM, Fernández ÁG, Cabeza LF. Corrosion Assessment of Myo-Inositol Sugar Alcohol as a Phase Change Material in Storage Systems Connected to Fresnel Solar Plants. Molecules. 2019; 24(7):1383. https://doi.org/10.3390/molecules24071383
Chicago/Turabian StyleMaldonado, José Miguel, Ángel G. Fernández, and Luisa F. Cabeza. 2019. "Corrosion Assessment of Myo-Inositol Sugar Alcohol as a Phase Change Material in Storage Systems Connected to Fresnel Solar Plants" Molecules 24, no. 7: 1383. https://doi.org/10.3390/molecules24071383
APA StyleMaldonado, J. M., Fernández, Á. G., & Cabeza, L. F. (2019). Corrosion Assessment of Myo-Inositol Sugar Alcohol as a Phase Change Material in Storage Systems Connected to Fresnel Solar Plants. Molecules, 24(7), 1383. https://doi.org/10.3390/molecules24071383








