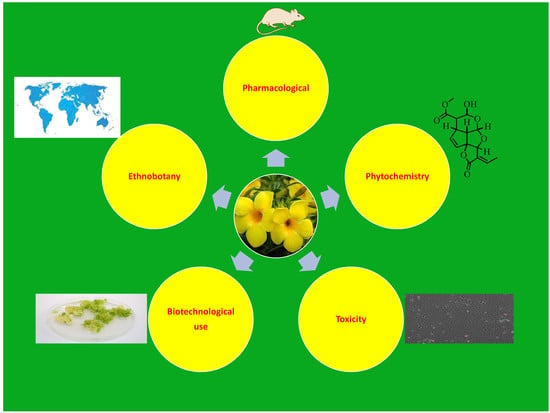
Allamanda cathartica: A Review of the Phytochemistry, Pharmacology, Toxicology, and Biotechnology
Abstract
1. Introduction
2. Ethnobotany
2.1. Botanical Characterization
2.2. Distribution
2.3. Synonyms
2.4. Traditional Medical Use
3. Phytochemistry
3.1. Hydrocarbons
3.2. Alcohol, Ester, Ether, Aldehyde, and Ketone
3.3. Fatty Acids and Phospholipids
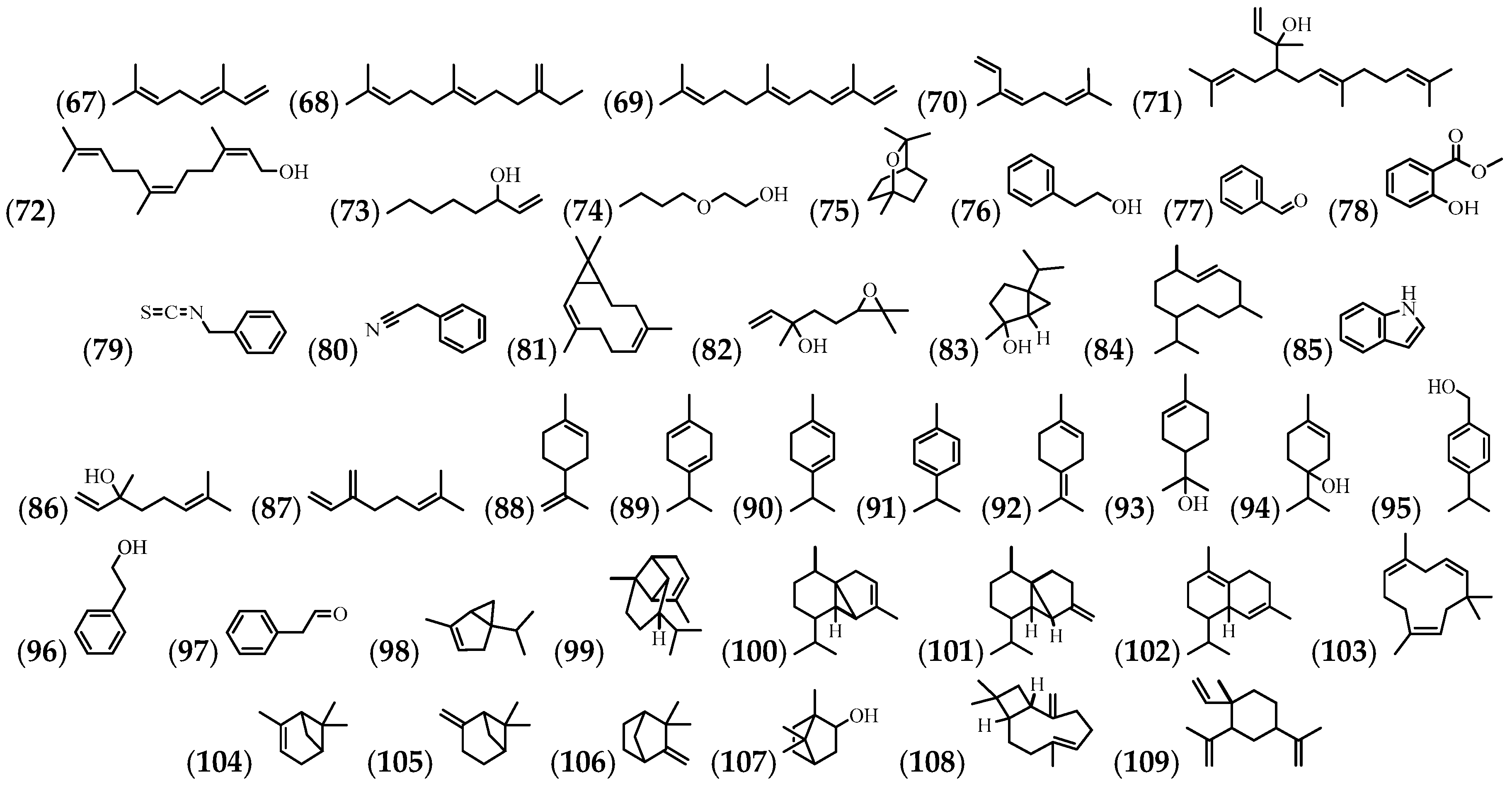
3.4. Volatile Compounds
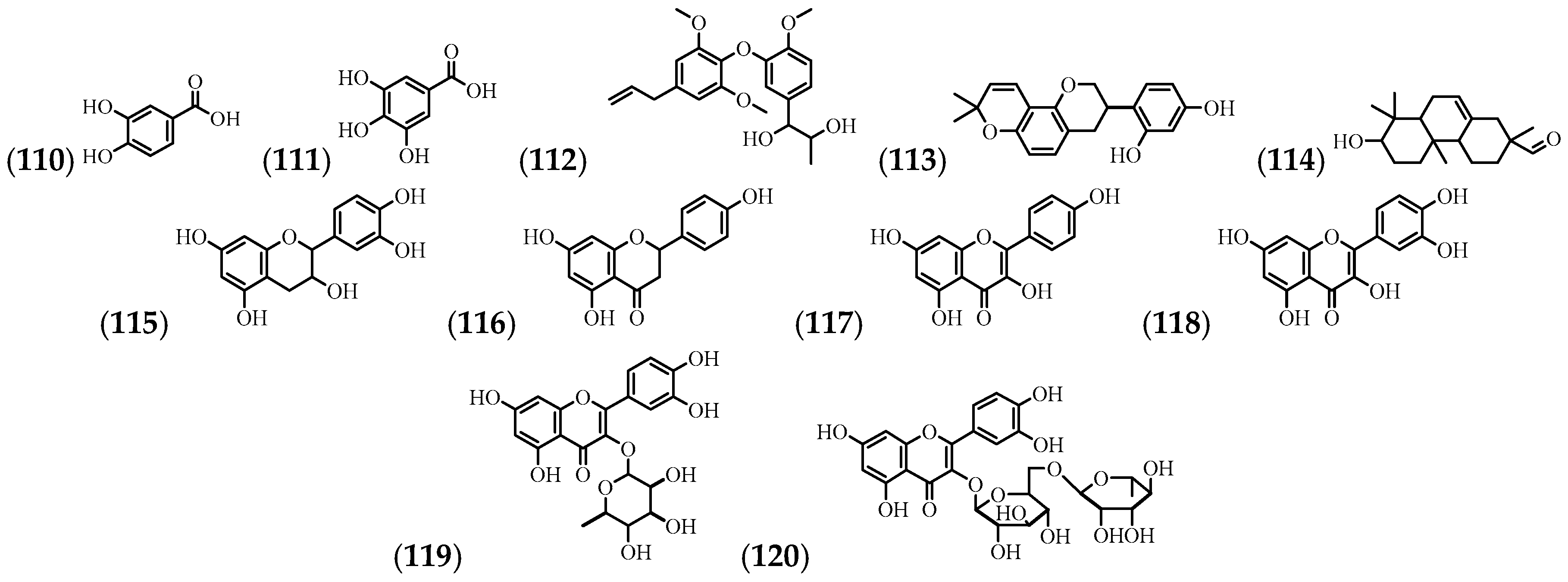
3.5. Phenolic Compounds and Flavonoids
3.6. Alkaloids
3.7. Steroids and Terpenes
3.8. Lactones
3.9. Carbohydrates
4. Pharmacological Activity
4.1. Analgesic
4.2. Anti-Inflammatory
4.3. Antidepressant
4.4. Antidiabetic
4.5. Antihyperlipidaemic
4.6. Antifertility
4.7. Wound Healing
4.8. Thrombolysis
4.9. Purgative Effect
4.10. Tyrosinase
4.11. Amylase
4.12. Antiviral
4.13. Antimicrobial
4.14. Antimalarial
4.15. Nematicide
4.16. Pesticidal
4.17. Antihaemorrhagic
4.18. Cytotoxicity
4.19. Antioxidants
5. Toxicity
6. Biotechnological Use
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- The Plants List. Allamanda [Internet]. 2019. Available online: http://www.theplantlist.org./tpl1.1/search?q=Allamanda (accessed on 15 February 2019).
- Monroy-Ortiz, C.; Monroy, R. Las Plantas, Compañeras de Siempre: La Experiencia en Morelos; UAEM, Centro de Investigaciones Biológicas de la CONABIO CONANP: Cuernavaca, Mexico, 2006; 582p. [Google Scholar]
- David, W.N. Poisonous Plants and Animals of Florida and the Caribbean; Sing Cheong Print Co Ltd.: Hong Kong, China, 1997; p. 138. [Google Scholar]
- Wong, S.K.; Lim, Y.Y.; Chan, E.W.C. Botany, uses, phytochemistry and pharmacology of selected Apocynaceae species: A review. Pharmacogn. Commun. 2013, 3, 2. [Google Scholar]
- Morales, J. La familia Apocynaceae (Apocynoideae, Rauvolfioideae) en Guatemala. Darwiniana Nueva Ser. 2009, 47, 140–184. [Google Scholar]
- Morales, J. Estudios en las Apocynaceae Neotropicales XXVIII: La familia Apocynaceae (Apocynoideae, Rauvolfioideae) de El Salvador, Centroamérica. Darwiniana Nueva Ser. 2006, 44, 453–489. [Google Scholar]
- Carballeira, N.M.; Cruz, C. 5,9-Nonadecadienoic acids in Malvaviscus arboreus and Allamanda cathartica. Phytochemistry 1998, 49, 1253–1256. [Google Scholar] [CrossRef]
- Nayak, S.; Nalabothu, P.; Sandiford, S.; Bhogadi, V.; Adogwa, A. Evaluation of wound healing activity of Allamanda cathartica L. and Laurus nobilis L. extracts on rats. BMC Complement. Altern. Med. 2006, 6, 12. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Kader, M.S.; Wisse, J.; Evans, R.; van der Werff, H.; Kingston, D.G. Bioactive iridoids and a new lignan from Allamanda cathartica and Himatanthus fallax from the Suriname rainforest. J. Nat. Prod. 1997, 60, 1294–1297. [Google Scholar] [CrossRef]
- Báez, D.; Pino, J.A.; Morales, D. Scent composition from flowers of Allamanda cathartica L. from Cuba. J Essent. Oil Bear Plants 2012, 15, 12–14. [Google Scholar]
- Warrell, D.A. Researching nature’s venoms and poisons. Trans. R. Soc. Trop. Med Hyg. 2009, 103, 860–866. [Google Scholar] [CrossRef]
- Otero, R.; Nunez, V.; Barona, J.; Fonnegra, R.; Jimenez, S.L.; Osorio, R.G.; Saldarriaga, M.; Diaz, A. Snakebites and ethnobotany in the northwest region of Colombia. Part III: Neutralization of the haemorrhagic effect of Bothrops atrox venom. J. Ethnopharmacol. 2000, 73, 233–241. [Google Scholar] [CrossRef]
- Swanholm, C.E.; St John, H.; Scheuer, P.J. A survey for alkaloids in Hawaiian plants. I. Pac. Sci. 1959, 8, 295–300. [Google Scholar]
- Arthur, H.R.; Hui, W.H. Products from some plants of Hong Kong. J. Chem. Soc. 1954, 2782–2784. [Google Scholar] [CrossRef]
- Manogaran, S.; Sulochana, N. Extraction and characterization of Allamanda cathartica. Asian J. Chem. 2005, 17, 1955. [Google Scholar]
- Suprapta, D.N.; Khalimi, K. Anti-fungal activities of selected tropical plants from Bali Island. Phytopharmacology 2012, 2, 265–270. [Google Scholar]
- Hameed, A.; Nawaz, G.; Gulzar, T. Chemical composition, antioxidant activities and protein profiling of different parts of Allamanda cathartica. Nat. Prod. Res. 2014, 28, 2066–2071. [Google Scholar] [CrossRef]
- Coppen, J.J.W.; Cobb, A.L. The occurrence of iridoids in Plumeria and Allamanda. Phytochemistry 1983, 22, 125–128. [Google Scholar] [CrossRef]
- Alen, Y.; Nakajima, S.; Nitoda, T.; Baba, N.; Kanzaki, H.; Kawazu, K. Antinematodal activity of some tropical rainforest plants against the pinewood nematode, Bursaphelenchus xylophilus. Z. Naturforsch C 2000, 55, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.K.; Datta, P.C. Pharmacognosy of Allamanda bark drugs. Int. J. Crude Drug. Res. 1982, 20, 43–52. [Google Scholar] [CrossRef]
- Tinoi, J.; Rakariyatham, N.; Deming, R.L. Determination of major carotenoid constituents in petal extracts of eight selected flowering plants in the north of Thailand. Chiang Mai J. Sci. 2006, 33, 327–334. [Google Scholar]
- Nguyen, P.Q.T.; Luu, T.T.; Bai, Y.; Nguyen, G.K.T.; Pervushin, K.; Tam, J.P. Allotides: Proline-rich cystine knot alpha-amylase inhibitors from Allamanda cathartica. J. Nat. Prod. 2015, 78, 695–704. [Google Scholar] [CrossRef]
- Mahbubur Rahman, A.H.M.; Akter, M. Taxonomy and traditional medicinal uses of apocynaceae (Dogbane) family of Rajshahi district, Bangladesh. Int. J. Bot. Stud. 2016, 1, 5–13. [Google Scholar]
- Li, A.-N.; Li, S.; Li, H.-B.; Xu, D.-P.; Xu, X.-R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Radford, D.J.; Cheung, K.; Urech, R.; Gollogly, J.R.; Duffy, P. Immunological detection of cardiac glycosides in plants. Aust. Vet. J. 1994, 71, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Bhat, N.R.; Suleiman, M.K.; Abdal, M. Selection of crops for sustainable utilization of land and water resources in Kuwait. World J. Agric. Sci. 2009, 5, 201–206. [Google Scholar]
- Gurib-Fakim, A.; Gueho, J.; Sewraj-Bissoondoyal, M. The medicinal plants of Mauritius—Part 1. Int. J. Pharmacogn. 1997, 35, 237–254. [Google Scholar] [CrossRef]
- Akah, P.A.; Offiah, V.N. Gastrointestinal effects of Allamanda cathartica leaf extracts. Int. J. Pharmacogn. 1992, 30, 213–217. [Google Scholar] [CrossRef]
- Maroyi, A. Garden Plants in Zimbabwe: Their ethnomedicinal uses and reported toxicity. Ethnobot. Res. Appl. 2012, 10, 45–57. [Google Scholar] [CrossRef]
- Sarker, R.; Sharmin, T.; Chowdhury, S.R.; Islam, F. Thrombolytic activity and preliminary cytotoxicity of five different fractions of methanol extract of Allamanda cathartica leaf. J. Appl. Pharm. Sci. 2012, 2, 129–132. [Google Scholar] [CrossRef]
- Mehta, S.; Roy, S.; Chowdhary, A. Use of rapid fluorescent focus inhibition test (RFFIT) for in vitro evaluation of anti-rabies activity. Virus Dis. 2017, 28, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Ashrafuzzaman, M.; Ali, H.; Liza, L.N.; Zinnah, K.M.A. Antimicrobial activity of some medicinal plants against multi drug resistant human pathogens. Adv. Biosci. Bioeng. 2013, 1, 1–24. [Google Scholar]
- Haque, M.M.; Choudhury, M.S.; Hossain, M.S.; Haque, M.A.; Seraj, S.; Rahmatullah, M. Ethnographic information and medicinal formulations of a Mro community of Gazalia Union in the Bandarbans district of Bangladesh. Am. Eur. J. Sustain. Agric. 2012, 6, 162–171. [Google Scholar]
- Scio, E.; Mendes, R.F.; Motta, E.V.S.; Bellozi, P.M.Q.; Aragão, D.M.O.; Mello, J.; Fabri, R.L.; Moreira, J.R.; de Assis, I.V.L.; Bouzada, M.L.M. Antimicrobial and Antioxidant Activities of Some Plant Extracts. In Phytochemicals as Nutraceuticals—Global Approaches to Their Role in Nutrition and Health; InTech: London, UK, 2012. [Google Scholar]
- Maia, J.G.S.; das Zoghbi, M.G.B.; Andrade, E.H.A.; Carreira, L.M.M. Volatiles from Flowers of Thevetia peruviana (Pers.) K. Schum. and Allamanda cathartics Linn. (Apocynaceae). J. Essent. Oil Res. 2000, 1, 322–324. [Google Scholar] [CrossRef]
- Hirschhorn, H.H. Botanical remedies of south and central America, and the Caribbean: An archival analysis. Part I. J. Ethnopharmacol. 1981, 4, 129–158. [Google Scholar] [CrossRef]
- Hirschhorn, H.H. Botanical remedies of South and Central America, and the Caribbean: An archival analysis. Part II. Conclusion. J. Ethnopharmacol. 1982, 5, 163–180. [Google Scholar] [CrossRef]
- Bharath Kumar, R.; Asha, S.; Babu, B.S. A note on phytodiversity and phytochemistry of important plant species of Vignan University Campus, Vadlamudi, Andhra Pradesh. Int. J. Pharm. Bio-Sci. 2014, 5, 373–386. [Google Scholar]
- Dutta, M.L. Plants used as ethnomedicine by the Thengal Kacharies of Assam, India. Asian J. Plant Sci. Res. 2017, 7, 7–8. [Google Scholar]
- Singha, I.M.; Unni, B.G.; Kakoty, Y.; Das, J.; Wann, S.B.; Singh, L.; Kalita, M.C. Evaluation of in vitro antifungal activity of medicinal plants against phytopathogenic fungi. Arch. Phytopathol. Plant Prot. 2011, 44, 1033–1040. [Google Scholar] [CrossRef]
- Joshi, S.C.; Sharma, A.; Chaturvedi, M. Antifertility potential of some medicinal plants in males: An overview. Int. J. Pharm. Pharm. Sci. 2011, 3, 204–217. [Google Scholar]
- Haron, F.F.; Sijam, K.; Omar, D.; Rahmani, M. Bioassay-guided isolation of antifungal plumericin from Allamanda species (Apocynaceae). J. Biol. Sci. 2013, 13, 158–162. [Google Scholar]
- Fasola, T.R.; Iyamah, P.C. The use of ethnobotanicals in the management of inflammation in Nigeria: A review. Int. J. Environ. 2015, 4, 1–18. [Google Scholar] [CrossRef]
- Nwambie, A.I.; Akah, P.A. Preliminary studies on some Nigerian herbal purgative recipes. Int. J. Pharmacogn. 1993, 31, 278–282. [Google Scholar] [CrossRef]
- Iyamah, P.C.; Idu, M. Ethnomedicinal survey of plants used in the treatment of malaria in Southern Nigeria. J. Ethnopharmacol. 2015, 173, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.F.; Taha, R.M. The effect of 2,4-dichlorophenoxyacetic acid and 6-benzylaminopurine on callus induction and plant regeneration of Allamanda cathartica—A valuable medicinal plant. Res. J. Biotecnol. 2012, 7, 75. [Google Scholar]
- Pawar, K.P.; Bhitre, M.J.; Kalamkar, P.V.; Kale, M.K. Pharmacognostical studies on leaves of Allamanda cathartica with detail physicochemical and phytochemical evaluation. Res. J. Pharmacogn. Phytochem. 2015, 7, 69. [Google Scholar] [CrossRef]
- Chaithra Amin, B.; Satish, S.; Abhishek, N.; Ajay Kumar, K. An investigation on anti-diabetic activity in aqueous extract of aerial parts of Allamanda cathartica Linn in streptozotocin induced diabetic rats. Int. J. Pharm. Chem. Res. 2017, 3, 242–247. [Google Scholar]
- Blasco, F.A.; De Guzman, G.Q.; Alejandro, G.J.D. A survey of ethnomedicinal plants in Surigao del Sur Mountain Range, Philippines. Int. J. Pure Appl. Biosci. 2014, 2, 166–172. [Google Scholar]
- Essiett, U.A.; Udo, E. Comparative phytochemical screening and nutritional potentials of the stems, leaves and flowers of Allamanda cathartica (Apocynaceae). Int. J. Sci. Technol. 2015, 4, 248–253. [Google Scholar]
- Savithramma, N.; Linga Rao, M.; Suhrulatha, D. Qualitative and quantification analysis of phytochemicals from leaf aqueous extract of Allamanda cathartica L. and Terminalia paniculata Roth. Int. J. 2013, 1, 821–825. [Google Scholar]
- Augustus, G.D.P.S.; Seiler, G.J. Phytochemicals of selected plant species of the Apocynaceae and Asclepiadaceae from Western Ghats, Tamil Nadu, India. Biomass Bioenergy 2011, 35, 3012–3017. [Google Scholar] [CrossRef]
- Joselin, J.; Brintha, T.S.S.; Florence, A.R.; Jeeva, S. Screening of select ornamental flowers of the family Apocynaceae for phytochemical constituents. Asian Pac. J. Trop. Dis. 2012, 2, S260–S264. [Google Scholar] [CrossRef]
- Mukherjee, K.; Ray, L.N. Phytochemical screening of some Indian medicinal plant species part II. Int. J. Crude Drug Res. 1986, 24, 187–205. [Google Scholar] [CrossRef]
- Marvin. MarvinSketch. Available online: http://www.chemaxon.com (accessed on 18 April 2018).
- Rizvi, M.A.; Yasmeen, K.; Ali, S.A.; Iqbal, G. Detection of trace elements in medicinal flowers of Pakistan. Int. J. Adv. Res. 2014, 2, 195–203. [Google Scholar]
- Prabhadevi, V.; Sahaya, S.S.; Johnson, M.; Venkatramani, B.; Janakiraman, N. Phytochemical studies on Allamanda cathartica L. using GC–MS. Asian Pac. J. Trop. Biomed. 2012, 2 (Suppl. 2), S550–S554. [Google Scholar] [CrossRef]
- Mathew, S.; Sreekumar, S.; Biju, C.K. Identification of lead compounds against human hepatitis B viral capsid protein in three medicinal plants through in silico method. IOSR J. Pharm. Biol. Sci. 2016, 11, 1–6. [Google Scholar]
- Yamauchi, K.; Mitsunaga, T.; Batubara, I. Isolation, identification and tyrosinase inhibitory activities of the extractives from Allamanda cathartica. Nat. Resour. 2011, 2, 167. [Google Scholar] [CrossRef]
- Hema, K.; Sukumar, D. Isolation and phytochemical studies of quercetin and quercetin 3-O-rhamnoside. Int. J. Pharm. Bio-Sci. 2013, 4, 519–524. [Google Scholar]
- Bonomini, T.J.; Holzmann, I.; Thiesen, L.C.; Fratoni, E.; Muller, A.F.F.; Lucinda-Silva, R.M.; Yunes, R.A.; Malheiros, A.; Gonçalves, A.E.; Dalmagro, A.P. Neuropharmacological and acute toxicological evaluation of ethanolic extract of Allamanda cathartica L. flowers and plumieride. Regul. Toxicol. Pharmacol. 2017, 91, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Ohmiya, A. Diversity of carotenoid composition in flower petals. Jpn. Agric. Res. Q 2011, 45, 163–171. [Google Scholar] [CrossRef]
- Gupta, N.C.; Singh, B.; Bhakuni, D.S. Steroids and triterpenes from Alangium lamarckii, Allamanda cathartica, Abrus precatorius and Holoptelea integrifolia. Phytochemistry 1969, 8, 791–792. [Google Scholar] [CrossRef]
- Bonomini, T.J.; Góes, J.A.; Machado, M.D.; da Silva, R.M.L.; Malheiros, A. Development and optimization of a microwave-assisted extraction of plumieride from Allamanda cathartica L. Flowers. Quim. Nova 2018, 41, 36–42. [Google Scholar] [CrossRef]
- Kupchan, S.M.; Dessertine, A.L.; Blaylock, B.T.; Bryan, R.F. Isolation and structural elucidation of allamandin, an antileukemic iridoid lactone from Allamanda cathartica. J. Org. Chem. 1974, 39, 2477–2482. [Google Scholar] [CrossRef] [PubMed]
- Coppen, J.J.W. Iridoids with algicidal properties from Allamanda cathartica. Phytochemistry 1983, 22, 179–182. [Google Scholar] [CrossRef]
- Thomas, V. Structure and biology of floral nectary in Allamanda cathartica L. (Apocynaceae). Feddes Repert. 1992, 103, 357–361. [Google Scholar] [CrossRef]
- Osborn, E.M. On the occurrence of antibacterial substances in green plants. Br. J. Exp. Pathol. 1943, 24, 227. [Google Scholar]
- Sarker, R.; Sharmin, T.; Islam, F.; Chowdhury, S.R. In vitro antioxidant, total phenolic, membrane stabilizing and antimicrobial activity of Allamanda cathartica L.: A medicinal plant of Bangladesh. J. Med. Plants Res. 2014, 8, 63–67. [Google Scholar]
- Hema, K. In vitro anti-inflammatory activity of quercitrin isolated from Allamanda cathartica. Int. J. Pharm. Bio-Sci. 2014, 5, 440–445. [Google Scholar]
- Boeing, T.; de Souza, P.; Bonomini, T.J.; Mariano, L.N.B.; Somensi, L.B.; Lucinda, R.M.; Malheiros, A.; da Silva, L.M.; Andrade, S.F. Antioxidant and anti-inflammatory effect of plumieride in dextran sulfate sodium-induced colitis in mice. Biomed. Pharmacother. 2018, 99, 697–703. [Google Scholar] [CrossRef]
- Singh, A.; Singh, S.K. Reversible antifertility effect of aqueous leaf extract of Allamanda cathartica L. in male laboratory mice. Andrologia 2008, 40, 337–345. [Google Scholar]
- Gupta, R.S.; Bhatnager, A.K.; Joshi, Y.C.; Sharma, R.; Sharma, A. Effects of plumieride, an iridoid on spermatogenesis in male albino rats. Phytomedicine 2004, 11, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Fartyal, M.; Kumar, P. Evaluation of antimicrobial efficacy of alkaloids, flavonoids and steroids of Allamanda cathartica Linn. against some pathogenic bacteria. Int. J. Adv. Pharm. Biol. Chem. 2016, 5, 303–313. [Google Scholar]
- Islam, M.R.; Ahamed, R.; Rahman, M.O.; Akbar, M.A.; Al-Amin, M.; Alam, K.D.; Lyzu, F. In vitro antimicrobial activities of four medicinally important plants in Bangladesh. Eur. J. Sci. Res. 2010, 39 (Suppl. 2), 199–206. [Google Scholar]
- Mannan, M.A.; Alam, M.S.; Mustari, F.; Kudrat-E-Zahan, M.; Ali, R.; Haque, A.H.; Zaman, S.; Talukder, D. In vitro antioxidant, antimicrobial, insecticidal and cytotoxic activities of the medicinal plants: Allamanda cathartica and Mimusops elengi. Eur. J. Med. Plants 2017, 20, 1–12. [Google Scholar] [CrossRef]
- Rao, M.L.; Bhumi, G.; Savithramma, N. Green synthesis of silver nanoparticles by Allamanda cathartica L. leaf extract and evaluation for antimicrobial activity. Int. J. Pharm. Sci. Nanotechnol. 2013, 6, 2260–2268. [Google Scholar]
- Karunakaran, G.; Jagathambal, M.; Gusev, A.; Kolesnikov, E.; Mandal, A.R.; Kuznetsov, D. Allamanda cathartica flower’s aqueous extract-mediated green synthesis of silver nanoparticles with excellent antioxidant and antibacterial potential for biomedical application. MRS Commun. 2016, 6, 41–46. [Google Scholar] [CrossRef]
- Hema, K.; Krishnaveni, R. Antibacterial and antifungal activities of Allamanda cathartica linn. Int. J. Pharm. Bio-Sci. 2014, 5, 588–593. [Google Scholar]
- Okwubie, L.; Senior, C.C. Evaluation of the antimicrobial activity of the crude root extracts of Allamanda cathartica L (Apocynaceae). Pharm. Innov. J. 2017, 6, 88–92. [Google Scholar]
- Rajamanickam, K.; Sudha, S.S. In vitro antimicrobial activity and in vivo toxicity of Moringa oleifera and Allamanda cathartica against multiple drug resistant clinical pathogens. Int. J. Pharm. Bio-Sci. 2013, 4, 768–775. [Google Scholar]
- Chusri, S.; Siriyong, T.; Na-Phatthalung, P.; Voravuthikunchai, S.P. Synergistic effects of ethnomedicinal plants of Apocynaceae family and antibiotics against clinical isolates of Acinetobacter baumannii. Asian Pac. J. Trop. Med. 2014, 7, 456–461. [Google Scholar] [CrossRef]
- Fartyal, M. Allamanda cathartica linn.: Extraction and pharmaceutical evaluation of various extracts of leaves and flowers. Int. J. Curr. Pharm. Res. 2016, 8, 28–32. [Google Scholar]
- Shukla, R.; Singh, P.; Prakash, B.; Dubey, N.K. Antifungal, aflatoxin inhibitory and free radical-scavenging activities of some medicinal plants extracts. J. Food Qual. 2012, 35, 182–189. [Google Scholar] [CrossRef]
- Arundhina, E. Aktivitas, Ekstrak Etanol daun Alamanda (Allamanda cathartica L.) Sebagai Antijamur Terhadap Candida Albicans dan Pityrosporum ovale Secara in vitro. J. Teknobiol. Available online: http://e-journal.uajy.ac.id/6530/1/jurnal%20BL01139.pdf (accessed on 25 March 2019).
- Tiwari, T.N.; Pandey, V.B.; Dubey, N.K. Plumieride from Allamanda cathartica as an antidermatophytic agent. Phyther. Res. 2002, 16, 393–394. [Google Scholar] [CrossRef]
- Mone, M.; Saieed, A.U.; Dastogeer, K.M.G.; Ali, M.A.; Meah, M.B. Plumieride from Allamanda cathartica as an inhibitory compound to plant pathogenic fungi. Arch. Phytopathol. Plant Prot. 2014, 47, 1311–1326. [Google Scholar] [CrossRef]
- Conrad, O.A.; Dike, I.P.; Agbara, U. In vivo antioxidant assessment of two antimalarial plants—Allamanda cathartica and Bixa orellana. Asian Pac. J. Trop. Biomed. 2013, 3, 388–394. [Google Scholar] [CrossRef]
- Fabiyi, O.A.; Olatunji, G.A.; Omoyele, A.A. Nematicidal and quantitative phytochemical analysis of the chromatographic fractions from the leaf and stem of Allamanda cathartica (L). Ethiop. J. Environ. Stud. Manag. 2014, 7, 253–257. [Google Scholar] [CrossRef]
- Radhakrishnan, B.; Prabhakaran, P. Biocidal activity of certain indigenous plant extracts against red spider mite, Oligonychus coffeae (Nietner) infesting tea. J. Biopestic. 2014, 7, 29. [Google Scholar]
- Das Nelaturi, P.; Sriramaia, N.H.; Nagaraj, S.; Kotakadi, V.S.; Kutty, M.; Veeran, A.V.; Kiranmayee, P. An in vitro cytotoxic and genotoxic properties of Allamanda cathartica L. latex green NPs on human peripheral blood mononuclear cells. Nano Biomed. Eng. 2017, 9, 314–323. [Google Scholar]
- Omonhinmin, C.A.; Dike, I.P.; Rotimi, S.O. Phytochemical, cytotoxicity and antioxidant activities of five anti-malaria plants. Res. J. Med. Plant 2015, 9, 181–189. [Google Scholar] [CrossRef]
- Victor, O.N.; Emeka, A.G.; Chukwuka, A.J.; Victor, A.O.; Simeon, E.I.; Victor, A.C.; Patience, O.N. Preliminary in vitro assessment of some phytochemical constituents and radical scavenging activity of methanol extracts of five flowers varieties. Annu. Res. Rev. Biol. 2015, 5, 357. [Google Scholar] [CrossRef]
- Chaveeracha, A.; Taneeb, T.; Patarapadungkitb, N.; Khamwachirapithakb, P.; Sudmoonb, R. Cytotoxicity and genotoxicity of Allamanda and Plumeria species. Sci. Asia 2016, 42, 375–381. [Google Scholar] [CrossRef]


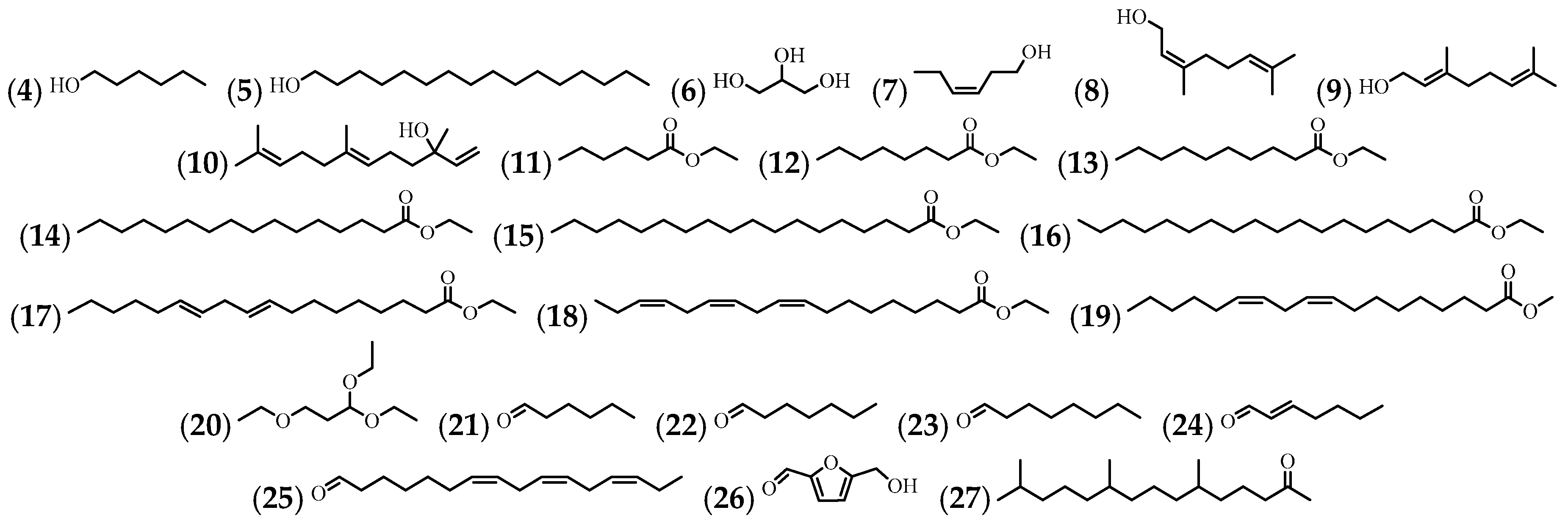


| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
| (1) | n-Heneicosane | Flowers | [10] |
| (2) | n-Tricosane | Flowers | [10] |
| (3) | n-Pentacosane | Flowers | [10] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
| (4) | 1-Hexanol | Flowers | [10] |
| (5) | 1-Hexadecanol | Flowers | [10] |
| (6) | Glycerin | Leaves and stem | [57] |
| (7) | (Z)-3-Hexenol | Flowers | [10] |
| (8) | Nerol | Flowers | [35] |
| (9) | Geraniol | Flowers | [35] |
| (10) | (E)-Nerolidol | Flowers | [35] |
| (11) | Hexanoic acid, ethyl ester | Leaves and stem | [57] |
| (12) | Octanoic acid, ethyl ester | Leaves and stem | [57] |
| (13) | Decanoic acid, ethyl ester | Leaves and stem | [57] |
| (14) | Hexadecanoic acid, ethyl ester | Leaves and stem | [57] |
| (15) | Octadecanoic acid, ethyl ester | Leaves and stem | [57] |
| (16) | Nonadecanoic acid, ethyl ester | Leaves | [57] |
| (17) | 9,12-Octadecadienoic acid, ethyl ester | Leaves and stem | [57] |
| (18) | 9,12,15-octadecatrienoic acid, ethyl ester, (Z,Z,Z)- | Leaves and stem | [43,57] |
| (19) | Methyl linoleate | Flowers | [10] |
| (20) | Propane, 1,1,3-triethoxy- | Leaves and stem | [57] |
| (21) | Hexanal | Flowers | [10] |
| (22) | Heptanal | Flowers | [10] |
| (23) | Octanal | Flowers | [10] |
| (24) | (E)-2-Heptenal | Flowers | [10] |
| (25) | Cis,cis,cis-7,10,13-hexadecatrienal | Leaves | [57] |
| (26) | 2-furancarboxaldehyde, 5-(hydroxymethyl)- | Stem | [57] |
| (27) | 6,10,14-Trimethyl-2-pentadecanone | Flowers | [10] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
| (28) | Dodecanoic acid | Flowers, leaves, and stem | [52,57] |
| (29) | Tetradecanoic acid | Flowers, leaves, and stem | [7,52,57] |
| (30) | Pentadecanoic acid | Leaves and flowers | [7,57] |
| (31) | Hexadecanoic acid | Flowers, leaves, and stem | [7,43,52,57] |
| (32) | Heptadecanoic acid | Flowers | [7] |
| (33) | Octadecanoic acid | Flowers and leaves | [7,52] |
| (34) | Nonadecanoic acid | Flowers | [7] |
| (35) | Eicosanoic acid | Flowers and leaves | [7,52] |
| (36) | Heneicosanoic acid | Flowers | [7] |
| (37) | Docosanoic acid | Flowers | [7] |
| (38) | Tetracosanoic acid | Flowers | [7] |
| (39) | Pentacosanoic acid | Flowers | [7] |
| (40) | Hexacosanoic acid | Flowers | [7] |
| (41) | 2-Hydroxyhexadecanoic acid | Flowers | [7] |
| (42) | 2-Hydroxyoctadecanoic acid | Flowers | [7] |
| (43) | 2-Hydroxyeicosanoic acid | Flowers | [7] |
| (44) | 2-Hydroxydocosanoic acid | Flowers | [7] |
| (45) | 2-Hydroxytricosanoic acid | Flowers | [7] |
| (46) | 2-Hydroxytetracosanoic acid | Flowers | [7] |
| (47) | 2-Hydroxydocosenoic acid | Flowers | [7] |
| (48) | 2-Hydroxytetracosenoic acid | Flowers | [7] |
| (49) | 7-Eicosenoic acid | Flowers | [7] |
| (50) | 9-Hexadecenoic acid | Flowers | [7] |
| (51) | 9-Octadecenoic acid | Flowers, leaves, and stem | [7,52,57] |
| (52) | 9-Nonadecenoic acid | Flowers | [7] |
| (53) | 11-Octadecenoic acid | Flowers | [7] |
| (54) | 11-Eicosenoic acid | Flowers | [7] |
| (55) | 13-Eicosenoic acid | Flowers | [7] |
| (56) | 13-Docosenoic acid | Flowers | [7] |
| (57) | 15-Docosenoic acid | Flowers | [7] |
| (58) | 5,9-Nonadecadienoic acid | Flowers | [7] |
| (59) | 17-Methyl-5,9-octadecadienoic acid * | Flowers | [7] |
| (60) | 11,14-Eicosadienoic acid | Flowers | [7] |
| (61) | 9,12-Octadecadienoic acid | Flowers and leaves | [7,52] |
| (62) | 9,12-Octadecadienoic acid (Z,Z)- | Stem | [57] |
| (63) | 9,12,15-Octadecatrienoic acid | Flowers | [7] |
| (64) | 9,12,15-Octadecatrienoic acid (Z,Z,Z)- | Leaves and Stem | [44,57] |
| (65) | Phosphatidylinositol | Flowers | [7] |
| (66) | Phosphatidycholine | Flowers | [7] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
| (67) | (E)-β-ocineme | Flowers | [10] |
| (68) | (E)-β-Farnesene | Flowers | [10] |
| (69) | (E,E)-α-Farnesene | Flowers | [10] |
| (70) | (Z)-β-ocimene | Flowers | [10] |
| (71) | (E,E)-Geranyl linaool | Flowers | [10] |
| (72) | (Z,Z)-Farnesol | Flowers | [10] |
| (73) | 1-Octen-3-ol | Flowers | [10] |
| (74) | 2-Butooxyethanol | Flowers | [10] |
| (75) | 1,8-cineole | Flowers | [10] |
| (76) | 2-Phenylethanol | Flowers | [10] |
| (77) | Benzaldehyde | Flowers | [10] |
| (78) | Benzoic acid, 2-hydroxy-, methyl ester | Leaves | [57] |
| (79) | Benzyl isothiocyanate | Flowers | [35] |
| (80) | Phenylacetonitrile | Flowers | [35] |
| (81) | Bicyclogermacrene | Flowers | [35] |
| (82) | Trans-Linalool oxide | Flowers | [35] |
| (83) | Cis-sabinehydrate | Flowers | [10] |
| (84) | Germacrene D | Flowers | [35] |
| (85) | Indole | Flowers | [10] |
| (86) | Linalool | Flowers | [35] |
| (87) | Myrcene | Flowers | [10] |
| (88) | Limonene | Flowers | [10] |
| (89) | γ-Terpinene | Flowers | [10] |
| (90) | α-Terpinene | Flowers | [10] |
| (91) | p-cyneme | Flowers | [10] |
| (92) | Terpinolene | Flowers | [10] |
| (93) | α-Terpineol | Flowers | [10,35] |
| (94) | Terpinen-4-ol | Flowers | [10] |
| (95) | 3,7,11,15-tetramethyl-2-hexadecen-1-ol | N.R. | [58] |
| (96) | Cumin alcohol | Flowers | [35] |
| (97) | Phenylacetaldehyde | Flowers | [10,35] |
| (98) | α-Thujene | Flowers | [10] |
| (99) | α-Copaene | Flowers | [35] |
| (100) | α-Cubebene | Flowers | [35] |
| (101) | β-Cubebene | Flowers | [35] |
| (102) | δ-Cadinene | Flowers | [35] |
| (103) | α-Humulene | Flowers | [35] |
| (104) | α-Pinene | Flowers | [10] |
| (105) | β-Pinene | Flowers | [10] |
| (106) | Camphene | Flowers | [10] |
| (107) | Isoborneol | Flowers | [10] |
| (108) | β-Caryophyllene | Flowers | [10,35] |
| (109) | β-Elemene | Flowers | [35] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
| (110) | Protocatechuic acid | Flowers | [24] |
| (111) | Gallic acid | Flowers | [24] |
| (112) | 1-(3-(4-Allyl-2,6-dimethoxyphenoxy)-4-methoxyphenyl)propane-1,2,diol | Stem | [59] |
| (113) | Glabridin | Stem | [59] |
| (114) | 2-phenanthrenecarboxaldehyde, 1,2,3,4,4a,4b,5,6,7,8,8a,9-dodecahydro-7-hydroxy-2,4b,8,8-tetramethyl- | Leaves and stem | [57] |
| (115) | Epicatechin | Flowers | [24] |
| (116) | Naringenin | Stem | [59] |
| (117) | Kaempferol | Stem | [59] |
| (118) | Quercetin | Flowers | [60] |
| (119) | Quercitrin | Flowers | [60] |
| (120) | Rutin | Flowers | [61] |
| No. | Compound’s name | Parts Used | Reference |
|---|---|---|---|
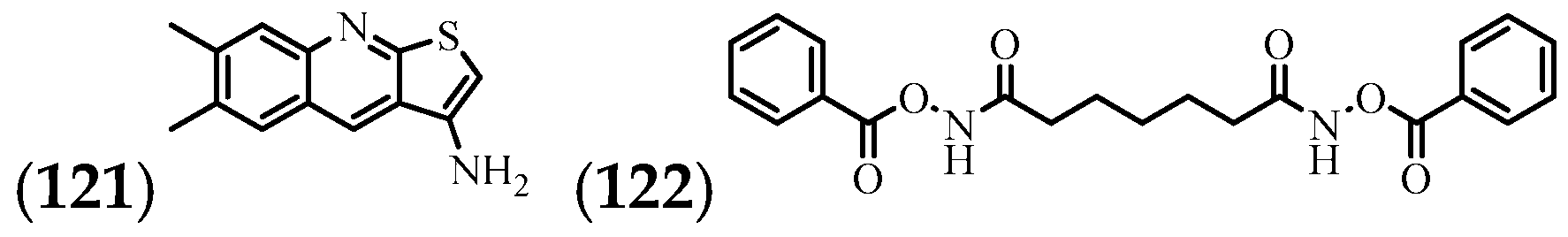
| (121) | 6,7-dimethylthieno(2,3-b) quinolin-3-ylamine | Stem | [57] |
| (122) | Heptanediamide, N,N′-di-benzoyloxy- | Stem | [57] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
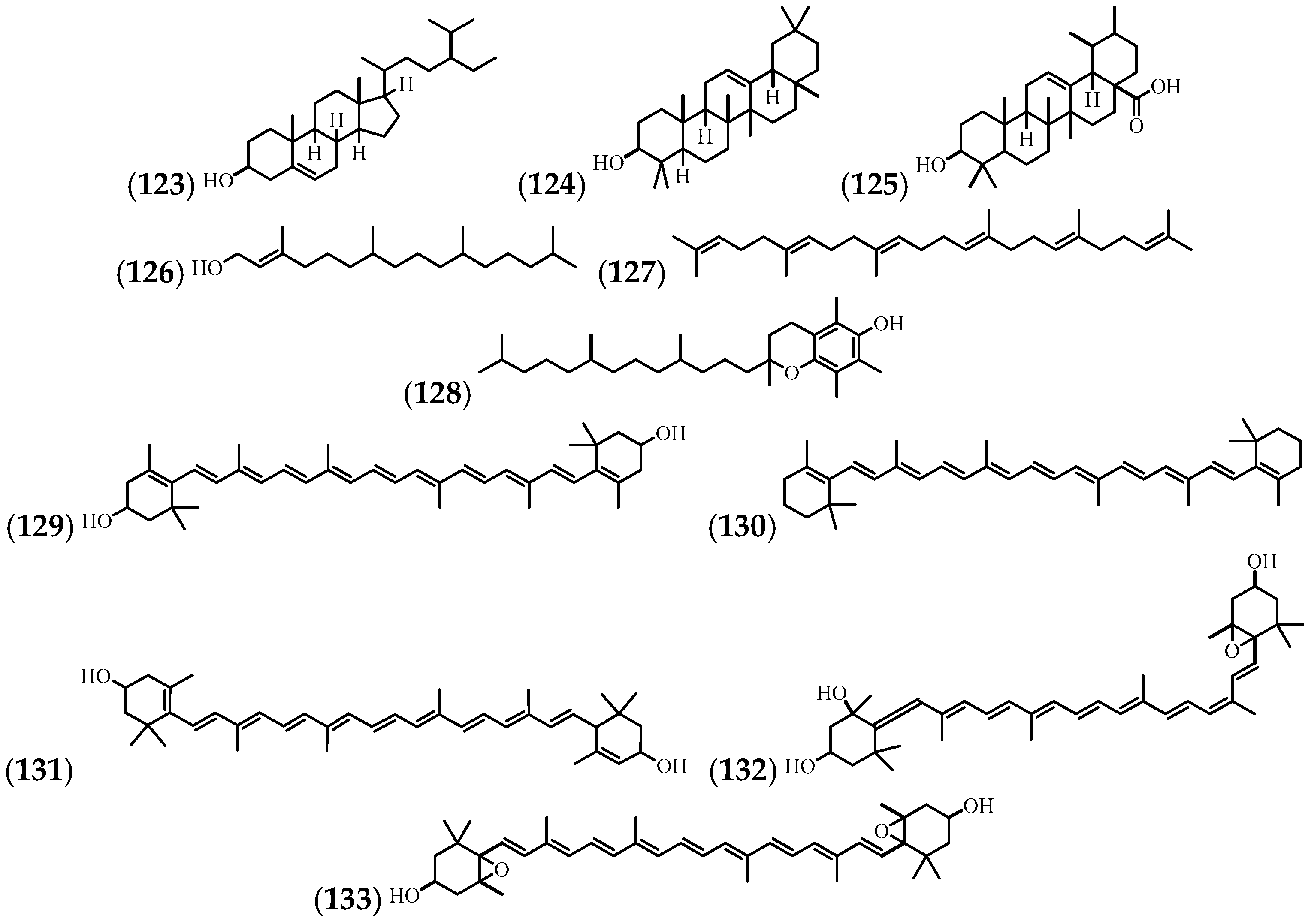
| (123) | β-sitosterol | Leaves and stem | [63] |
| (124) | β-Amyrin | Leaves and stem | [63] |
| (125) | Ursolic acid | Leaves and stem | [14,63] |
| (126) | Phytol | Flowers, leaves, and stem | [10,57] |
| (127) | Squalene | Leaves | [57] |
| (128) | Vitamine E | Leaves | [57] |
| (129) | Zeaxanthin | Flowers | [21] |
| (130) | b-Carotene | Flowers | [21] |
| (131) | Lutein | Flowers | [21] |
| (132) | Neoxanthin | Flowers | [21] |
| (133) | Violaxanthin | Flowers | [21] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
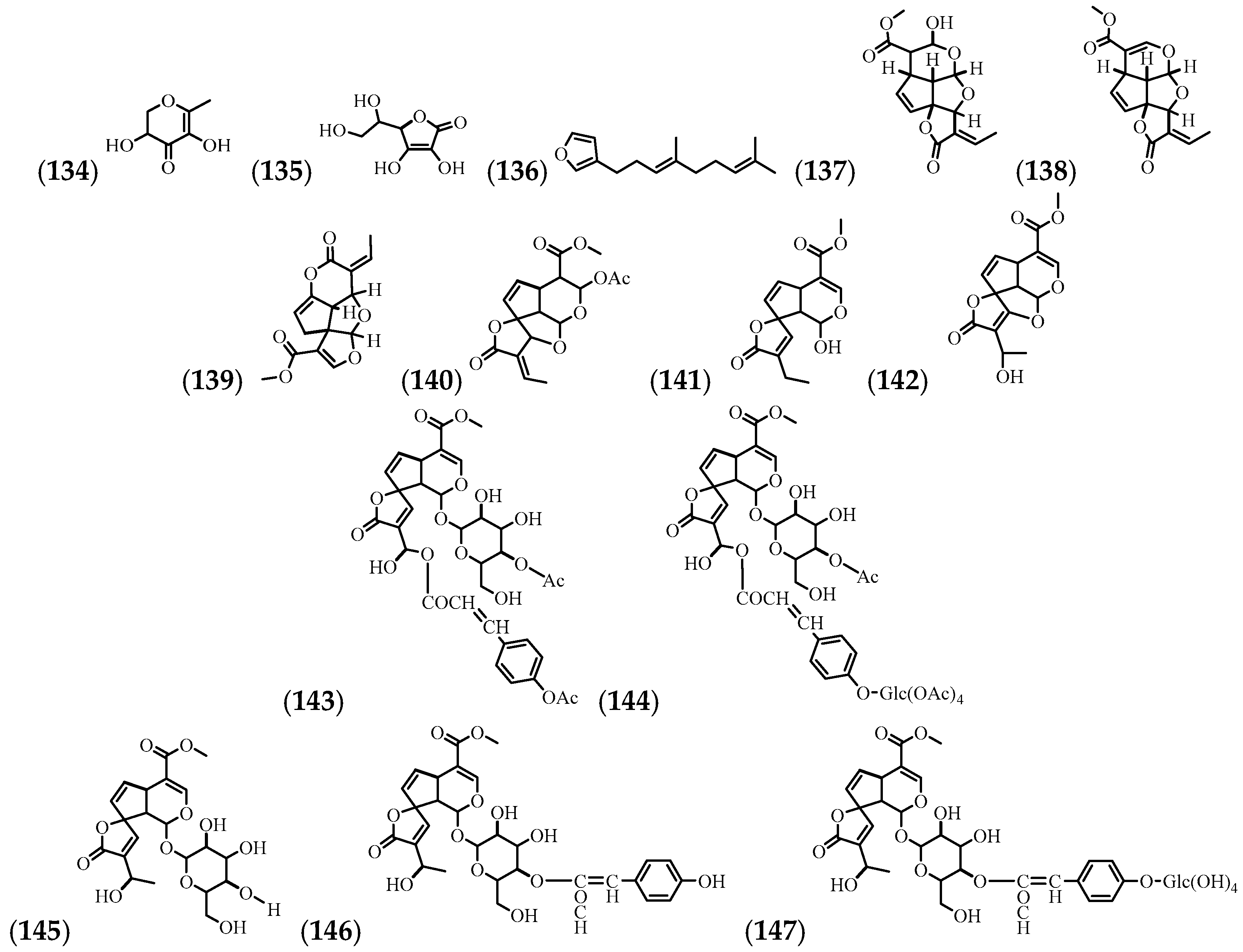
| (134) | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | Leaves and stem | [57] |
| (135) | Vitamine C | Leaves | [14] |
| (136) | Dendrolasin | Flowers | [35] |
| (137) | Allamandin | Root bark | [65] |
| (138) | Plumericin | Leaves, root, stem, leaves, flowers, bark, and root bark | [9,18,65,66] |
| (139) | Isoplumericin | Leaves, root, root bark, stem, and bark | [9,18,65,66] |
| (140) | Acetylallamandin | Root bark | [65] |
| (141) | Allamdin | Root bark | [65] |
| (142) | Allamandicin | Root bark | [65] |
| (143) | Penta-acetylplumieride coumarate | Root | [66] |
| (144) | Octa-acetylplumieride coumarate | Root | [66] |
| (145) | Plumieride | Root, stem, leaves, flowers, bark, and bark (inner part) | [18] |
| (146) | Plumieride coumarate | Root, stem, leaves, flowers, bark, and bark (inner part) | [18,66] |
| (147) | Plumieride coumarate glucoside | Root, stem, leaves, flowers, bark, and bark (inner part) | [18,66] |
| No. | Compound’s Name | Parts Used | Reference |
|---|---|---|---|
| (148) | 1-Deoxy-d-mannitol | Leaves | [57] |
| (149) | 3-O-methyl-d-glucose | Leaves and stem | [43,57] |
| (150) | Glucose | Nectar | [67] |
| (151) | Rhamnose | Nectar | [15] |
| (152) | Fructose | Nectar | [67] |
| (153) | β-l-arabinopyranoside, methyl | Leaves | [57] |
| Microorganism | Used Part | Extract/Fraction | Reference |
|---|---|---|---|
| Gram Positive | |||
| Agrobacterium tumefaciens | Flowers and leaves | Bound and free flavonoids, steroids, and alkaloids | [74] |
| Bacillus cereus | Leaves | TCM | [75] |
| EtOAc | [69] | ||
| MeOH, PE, TCM, EtOAc, and Dia-Ion | [76] | ||
| Bacillus megaterium | Leaves | TCM | [75] |
| EtOAc | [69] | ||
| Bacillus subtilis | Flowers and Leaves | Bound and free flavonoids and steroids | [74] |
| Leaves | TCM | [75] | |
| Water * | [77] | ||
| Sarcina lutea | Leaves | TCM | [75] |
| Staphylococcus aureus | Flowers | Water * | [78] |
| MeOH 90% | [79] | ||
| Flowers and leaves | Free flavonoids, alkaloids, bound flavonoids, and steroids | [74] | |
| Leaves | MeOH, PE, TCM, EtOAc, and Dia-Ion | [76] | |
| TCM | [77] | ||
| Root | MeOH, EtOAc, and PE | [80] | |
| All plant | N.E. | [68] | |
| Staphylococcus aureus ** | Leaves | MeOH, EtOH, EtOAc, TCM, and PE | [81] |
| Streptococcus pneumonia | Root | MeOH, EtOAc | [80] |
| Gram Negative | |||
| Acinetobacter baumannii ** | Flowers | EtOH | [82] |
| Acinetobacter sp ** | Leaves | MeOH, EtOH, EtOAc, Water, and PE | [81] |
| Bacillus subtillis | Leaves | Bound flavonoids | [74] |
| Escherichia coli | Flowers | Water * | [78] |
| Flowers and leaves | Bound flavonoids and steroids | [74] | |
| Flowers | MeOH 90% | [79] | |
| Leaves | TCM | [75] | |
| MeOH, PE, TCM, EtOAc, and Dia-Ion | [76] | ||
| Root | EtOAc | [80] | |
| Escherichia coli ** | Leaves | Water and PE | [81] |
| Water | [32] | ||
| Klebsiella pneumoniae | Root | MeOH and EtOAc | [80] |
| Flowers | Water * | [78] | |
| Flowers and leaves | Bound and free flavonoids | [74] | |
| Leaves | Water * | [77] | |
| Klebsiella pneumoniae ** | Leaves | Water | [32] |
| Proteus mirabilis ** | Leaves | Water | [32] |
| Proteus sp ** | Leaves | PE | [81] |
| Proteus vulgaris | Leaves | MeOH, PE, TCM, EtOAc, and Dia-Ion | [76] |
| Pseudomonas aeruginosa | Leaves | TCM | [75] |
| Water * | [77] | ||
| Pseudomonas aeruginosa ** | Leaves | Water | [32] |
| MeOH, EtOAc, TCM, and PE | [81] | ||
| Salmonella paratyphi | Leaves | TCM | [75] |
| EtOAc | [69] | ||
| Salmonella typhi | Leaves | TCM | [75] |
| EtOAc | [69] | ||
| Salmonella typhimurium | Flowers | Water * | [78] |
| Shigella boydii | Leaves | TCM | [75] |
| Shigella dysenteriae | Leaves | TCM | [75] |
| Vibrio mimicus | Leaves | TCM | [75] |
| Vibrio parahemolyticus | Leaves | TCM | [75] |
| Fungi | |||
| Aspergillus flavus | Leave and Flowers | MeOH | [83] |
| Aspergillus flavus | Leaves | MeOH:Water (2:1 v/v) | [84] |
| Water * | [77] | ||
| Aspergillus niger | Leaves | TCM | [75] |
| Water * | [77] | ||
| Candida albicans | Leaves | EtOH 99.8% | [85] |
| TCM | [75] | ||
| MeOH | [34] | ||
| Leave and Flowers | MeOH | [83] | |
| Flowers | MeOH 90% | [79] | |
| Candida albicans ** | Leaves | EtOH | [81] |
| Carvularia lunata | Leaves | PE and TCM | [40] |
| Epidermophyton floccosum | Leaves | MeOH | [86] |
| Microsporum gypseum | Leaves | MeOH | [86] |
| Pityrosporum ovale | Leaves | EtOH 99.8% | [85] |
| Sacharomyces cerevaceae | Leaves | TCM | [75] |
| Plant Fungi | |||
| Colletotrichum gloeosporioides | Leaves | TCM | [42] |
| Colletotrichum lidemuthianum | Leaves | PE and TCM | [40] |
| Curvularia luunata | Leaves | Water * | [77] |
| Fusarium oxysporum | Leaves | PE and TCM | [40] |
| MeOH, EtOH, EtOAc, and EtOH 50% | [87] | ||
| Fusarium oxysporum f.sp. capsici | Leave | MeOH | [16] |
| Phomopsis vexans | Leaves | MeOH, EtOH, EtOAc, and EtOH 50% | [87] |
| Phytophthora capsici | Leaves | MeOH, EtOH, EtOAc, and EtOH 50% | [87] |
| Rhizopus arrhizus | Leaves | Water * | [77] |
| Rhizotonia solani | Leaves | MeOH, EtOH, EtOAc, and EtOH 50% | [87] |
| Sclerotium rolsfsii | Leaves | MeOH, EtOH, EtOAc, and EtOH 50% | [87] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petricevich, V.L.; Abarca-Vargas, R. Allamanda cathartica: A Review of the Phytochemistry, Pharmacology, Toxicology, and Biotechnology. Molecules 2019, 24, 1238. https://doi.org/10.3390/molecules24071238
Petricevich VL, Abarca-Vargas R. Allamanda cathartica: A Review of the Phytochemistry, Pharmacology, Toxicology, and Biotechnology. Molecules. 2019; 24(7):1238. https://doi.org/10.3390/molecules24071238
Chicago/Turabian StylePetricevich, Vera L., and Rodolfo Abarca-Vargas. 2019. "Allamanda cathartica: A Review of the Phytochemistry, Pharmacology, Toxicology, and Biotechnology" Molecules 24, no. 7: 1238. https://doi.org/10.3390/molecules24071238
APA StylePetricevich, V. L., & Abarca-Vargas, R. (2019). Allamanda cathartica: A Review of the Phytochemistry, Pharmacology, Toxicology, and Biotechnology. Molecules, 24(7), 1238. https://doi.org/10.3390/molecules24071238