Characterization of a New DyP-Peroxidase from the Alkaliphilic Cellulomonad, Cellulomonas bogoriensis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Novel DyP Enzyme Identification from C. bogoriensis
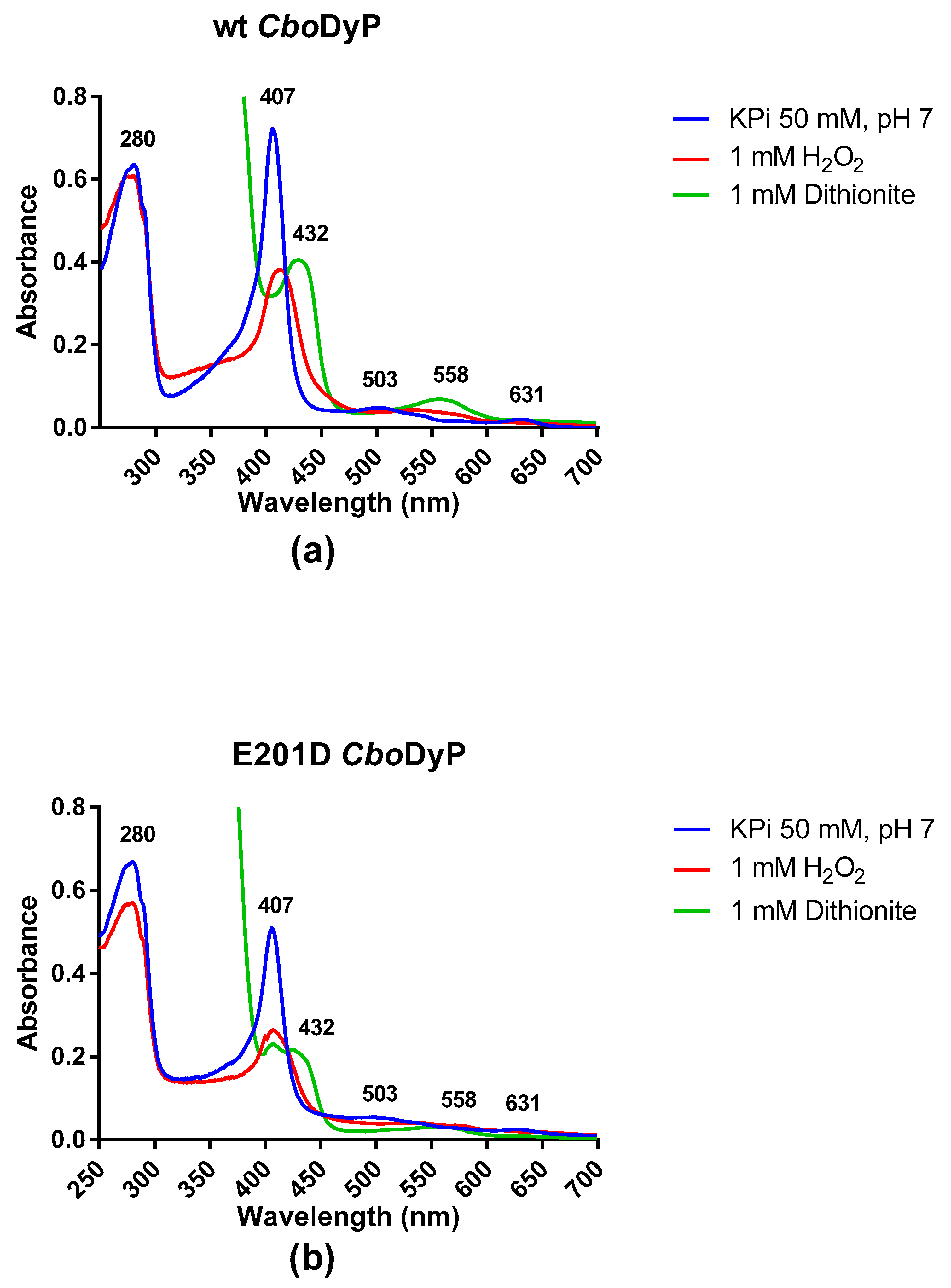
2.2. Spectral Properties of CboDyP
2.3. Activity of Wild-Type (wt) and E201D CboDyP with Different Dyes
2.4. pH Profile Determination for wt and E201D CboDyP
2.5. Steady State Kinetic Measurements for Peroxidase Activity Determination
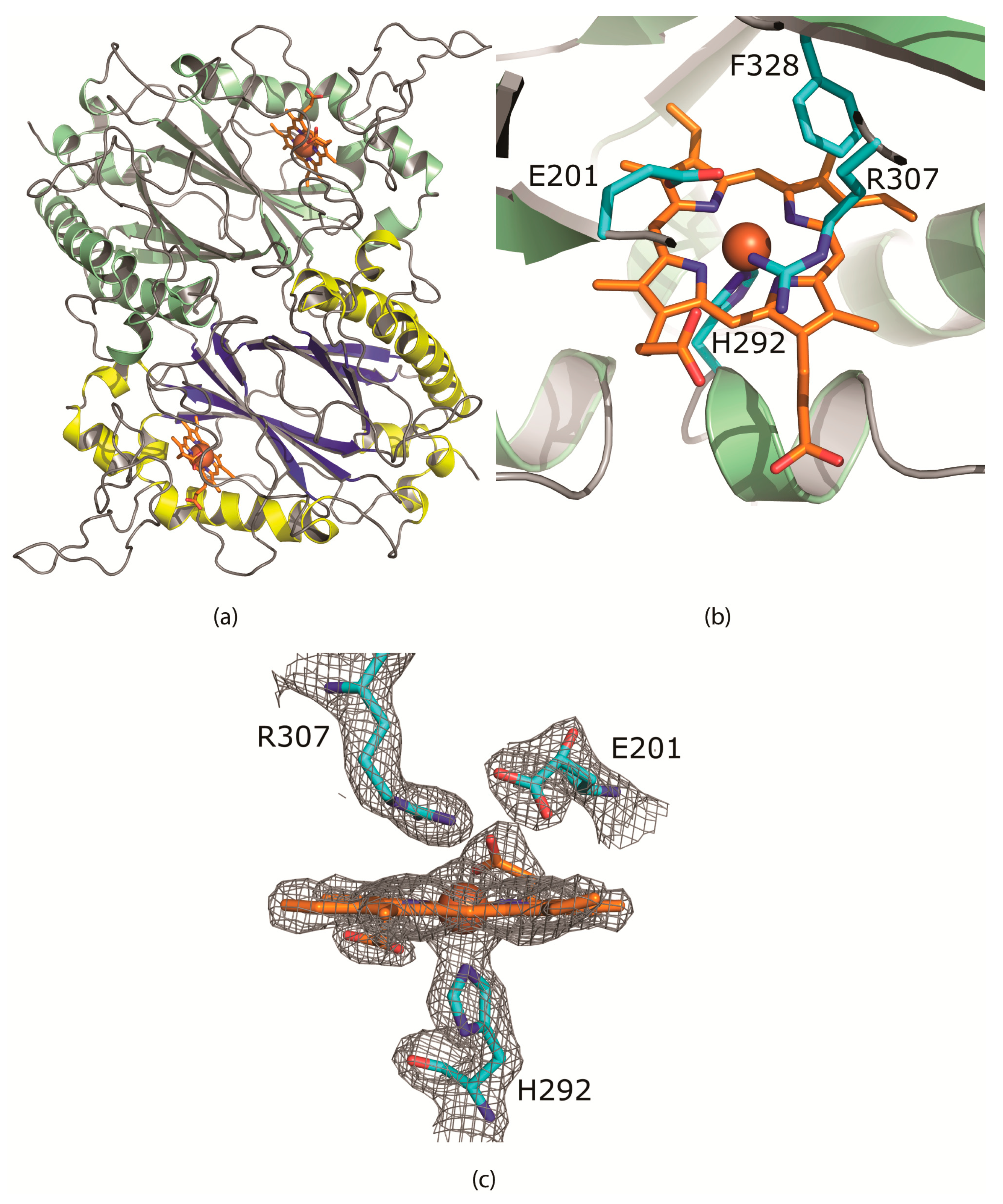
2.6. Structural Characterization of CboDyP
2.7. Thermal Stability of CboDyP
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Enzyme Cloning
3.3. Enzyme Expression and Purification
3.4. Determination of Enzyme Concentration
3.5. Determination of the Activity of CboDyP with Different Dyes
3.6. pH Profile for CboDyP wt and CboDyP E201D Mutant
3.7. Steady-State Kinetics Measurements
3.8. Spectral Properties of CboDyP Wild-Type and E201D Mutant
3.9. Preparation of CboDyP for X-Ray Crystallography
3.10. Crystallization, Data Collection, Structure Determination and Refinement
Substrate Channel Calculation
3.11. Thermal Stability of CboDyP
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Bloois, E.; Torres Pazmiño, D.E.; Winter, R.T.; Fraaije, M.W. A Robust and Extracellular Heme-Containing Peroxidase from Thermobifida fusca as Prototype of a Bacterial Peroxidase Superfamily. Appl. Microbiol. Biotechnol. 2010, 86, 1419–1430. [Google Scholar] [CrossRef]
- Jones, B.E.; Grant, W.D.; Duckworth, A.W.; Schumann, P.; Weiss, N.; Stackebrandt, E. Cellulomonas bogoriensis Sp. Nov., an Alkaliphilic Cellulomonad. Int. J. Syst. Evol. Microbiol. 2005, 55, 1711–1714. [Google Scholar] [CrossRef] [PubMed]
- Pfanzagl, V.; Nys, K.; Bellei, M.; Michlits, H.; Mlynek, G.; Battistuzzi, G.; Djinovic-Carugo, K.; Van Doorslaer, S.; Furtmuller, P.G.; Hofbauer, S.; et al. Roles of distal aspartate and arginine of B-class dye-decolorizing peroxidase in heterolytic hydrogen peroxide cleavage. J. Biol. Chem. 2018, 293, 14823–14838. [Google Scholar] [CrossRef]
- Singh, R.; Grigg, J.; Armstrong, Z.; Murphy, M.E.P.; Eltis, L.D. Distal heme pocket residues of B-type dye-decolorizing peroxidase: Argninine but not aspartate is essential for peroxidase activity. J. Biol. Chem. 2012, 287, 10623–10630. [Google Scholar] [CrossRef]
- Colpa, D.I.; Fraaije, M.W. High overexpression of dye decolorizing peroxidase TfuDyP leads to the incorporation of heme precursor protoporphyrin IX. J. Mol. Catal. B-Enzym. 2016, 134, 372–377. [Google Scholar] [CrossRef]
- Lončar, N.; Colpa, D.I.; Fraaije, M.W. Exploring the Biocatalytic Potential of a DyP-Type Peroxidase by Profiling the Substrate Acceptance of Thermobifida fusca DyP Peroxidase. Tetrahedron 2016, 72, 7276–7281. [Google Scholar] [CrossRef]
- Yu, W.; Liu, W.; Huang, H.; Zheng, F.; Wang, X.; Wu, Y.; Li, K.; Xie, X.; Jin, Y. Application of a Novel Alkali-Tolerant Thermostable DyP-Type Peroxidase from Saccharomonospora viridis DSM 43017 in Biobleaching of Eucalyptus Kraft Pulp. PLoS ONE 2014, 9, e110319. [Google Scholar] [CrossRef] [PubMed]
- Rahmanpour, R.; Rea, D.; Jamshidi, S.; Fulop, V.; Bugg, T.D.H. Structure of Thermobifida fusca DyP-Type Peroxidase and Activity towards Kraft Lignin and Lignin Model Compounds. Arch. Biochem. Biophys. 2016, 594, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, R.; Chen, X.; Ramyar, K.X.; Hayati, Z.; Carlson, E.A.; Bossmann, S.H.; Song, L.; Geisbrecht, B.V.; Li, P. Identification of Surface-Exposed Protein Radicals and a Substrate Oxidation Site in A-Class Dye-Decolorizing Peroxidase from Thermomonospora curvata. ACS Catal. 2016, 6, 8036–8047. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Shrestha, R.; Jia, K.; Gao, P.F.; Geisbrecht, B.V.; Bossmann, S.H.; Shi, J.; Li, P. Characterization of Dye-Decolorizing Peroxidase (DyP) from Thermomonospora curvata Reveals Unique Catalytic Properties of A-Type DyPs. J. Biol. Chem. 2015, 290, 23447–23463. [Google Scholar] [CrossRef]
- Kekilli, D.; Moreno-Chicano, T.; Chaplin, A.K.; Horrell, S.; Dworkowski, F.S.N.; Worrall, J.A.R.; Strange, R.W.; Hough, M.A. Photoreduction and Validation of Haem-Ligand Intermediate States in Protein Crystals by in Situ Single-Crystal Spectroscopy and Diffraction. IUCrJ 2017, 4, 263–270. [Google Scholar] [CrossRef]
- Liu, X.; Du, Q.; Wang, Z.; Zhu, D.; Huang, Y.; Li, N.; Wei, T.; Xu, S.; Gu, L. Crystal Structure and Biochemical Features of EfeB/YcdB from Escherichia coli O157: ASP235 Plays Divergent Roles in Different Enzyme-Catalyzed Processes. J. Biol. Chem. 2011, 286, 14922–14931. [Google Scholar] [CrossRef]
- Yoshida, T.; Ogola, H.; Amano, Y.; Hisabori, T.; Ashida, H.; Sawa, Y.; Tsuge, H.; Sugano, Y. Anabaena sp. DyP-type peroxidase is a tetramer consisting of two asymmetric dimers. Proteins. 2016, 84, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Gasteiger, E.; Hoogland, C.; Gattiker, A.; Duvaud, S.; Wilkins, M.R.; Appel, R.D.; Bairoch, A. Protein Identification and Analysis Tools on the ExPASy Server. In The Proteomics Protocols Handbook; Walker, J.M., Ed.; Humana Press: Totowa, NJ, USA, 2005; pp. 571–607. [Google Scholar]
- Powell, H.R.; Johnson, O.; Leslie, A.G.W. Autoindexing Diffraction Images with IMosflm. Acta Crystallogr. D Biol. Crystallogr. 2013, 69, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Winn, M.D.; Ballard, C.C.; Cowtan, K.D.; Dodson, E.J.; Emsley, P.; Evans, P.R.; Keegan, R.M.; Krissinel, E.B.; Leslie, A.G.W.; McCoy, A.; et al. Overview of the CCP4 Suite and Current Developments. Acta Crystallogr. D Biol. Crystallogr. 2011, 67, 235–242. [Google Scholar] [CrossRef]
- McCoy, A.J.; Grosse-Kunstleve, R.W.; Adams, P.D.; Winn, M.D.; Storoni, L.C.; Read, R.J. Phaser Crystallographic Software. J. Appl. Crystallogr. 2007, 40, 658–674. [Google Scholar] [CrossRef] [PubMed]
- Vagin, A.; Teplyakov, A. Molecular Replacement with MOLREP. Acta Crystallogr. D Biol. Crystallogr. 2010, 66, 22–25. [Google Scholar] [CrossRef]
- Matthews, B.W. Solvent content of protein crystals. J. Mol. Biol. 1968, 33, 491–497. [Google Scholar] [CrossRef]
- Murshudov, G.N.; Skubak, P.; Lebedev, A.A.; Pannu, N.S.; Steiner, R.A.; Nicholls, R.A.; Winn, M.D.; Long, F.; Vagin, A.A. REFMAC5 for the Refinement of Macromolecular Crystal Structures. Acta Crystallogr. D Biol. Crystallogr. 2011, 67, 355–367. [Google Scholar] [CrossRef]
- Emsley, P.; Lohkamp, B.; Scott, W.G.; Cowtan, K. Features and Development of Coot. Acta Crystallogr. D Biol. Crystallogr. 2010, 66, 486–501. [Google Scholar] [CrossRef] [PubMed]
- Joosten, R.P.; Long, F.; Murshudov, G.N.; Perrakis, A. The PDB_REDO Server for Macromolecular Structure Model Optimization. IUCrJ 2014, 1, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Chen, V.B.; Arendall, W.B., 3rd; Headd, J.J.; Keedy, D.A.; Immormino, R.M.; Kapral, G.J.; Murray, L.W.; Richardson, J.S.; Richardson, D.C. MolProbity: All-Atom Structure Validation for Macromolecular Crystallography. Acta Crystallogr. D Biol. Crystallogr. 2010, 66, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Chovancova, E.; Pavelka, A.; Benes, P.; Strnad, O.; Brezovsky, J.; Kozlikova, B.; Gora, A.; Sustr, V.; Klvana, M.; Medek, P.; et al. CAVER 3.0: A Tool for the Analysis of Transport Pathways in Dynamic Protein Structures. PLoS Comput. Biol. 2012, 8, e1002708. [Google Scholar] [CrossRef] [PubMed]
- Cummings, M.D.; Farnum, M.A.; Nelen, M.I. Universal Screening Methods and Applications of ThermoFluor®. J. Biomol. Screen. 2006, 11, 854–863. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available. |
| Dye Type | Dye | λmax (nm) | Conc. (µM) | kobs at pH 4 (s−1) | Hemin | ||
|---|---|---|---|---|---|---|---|
| wt CboDyP | E201D CboDyP | TfuDyP b | |||||
| Anthraquinone | Reactive Blue 19 | 595 | 50 | 0.22 a | 2.88 a | 1.7 | N.D. c |
| Anthraquinone | Disperse Blue 1 | 577 | 50 | 2.26 ± 0.20 | 2.13 ± 0.02 | 10 | N.D. |
| Azo dye | Acid Red 14 | 516 | 25 | 0.029 ± 0.001 | 0.34 ± 0.01 | 0.047 | 0.027 ± 0.038 |
| Indigoid dye | Indigotetrasulfonate | 590 | 50 | 0.051 ± 0.02 | 0.129 ± 0.004 | 0.023 | 0.040 ± 0.015 |
| Phthalocyanine dye | Copper phthalocyanine-3,4′,4″,4‴-tetrasulfonic acid | 616 | 25 | - | 0.137 ± 0.022 | 0.85 | 0.008 ± 0.002 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Habib, M.H.; Rozeboom, H.J.; Fraaije, M.W. Characterization of a New DyP-Peroxidase from the Alkaliphilic Cellulomonad, Cellulomonas bogoriensis. Molecules 2019, 24, 1208. https://doi.org/10.3390/molecules24071208
Habib MH, Rozeboom HJ, Fraaije MW. Characterization of a New DyP-Peroxidase from the Alkaliphilic Cellulomonad, Cellulomonas bogoriensis. Molecules. 2019; 24(7):1208. https://doi.org/10.3390/molecules24071208
Chicago/Turabian StyleHabib, Mohamed H., Henriëtte J. Rozeboom, and Marco W. Fraaije. 2019. "Characterization of a New DyP-Peroxidase from the Alkaliphilic Cellulomonad, Cellulomonas bogoriensis" Molecules 24, no. 7: 1208. https://doi.org/10.3390/molecules24071208
APA StyleHabib, M. H., Rozeboom, H. J., & Fraaije, M. W. (2019). Characterization of a New DyP-Peroxidase from the Alkaliphilic Cellulomonad, Cellulomonas bogoriensis. Molecules, 24(7), 1208. https://doi.org/10.3390/molecules24071208






