Advances in Molecular Mechanisms of Wheat Allergenicity in Animal Models: A Comprehensive Review
Abstract
1. Introduction
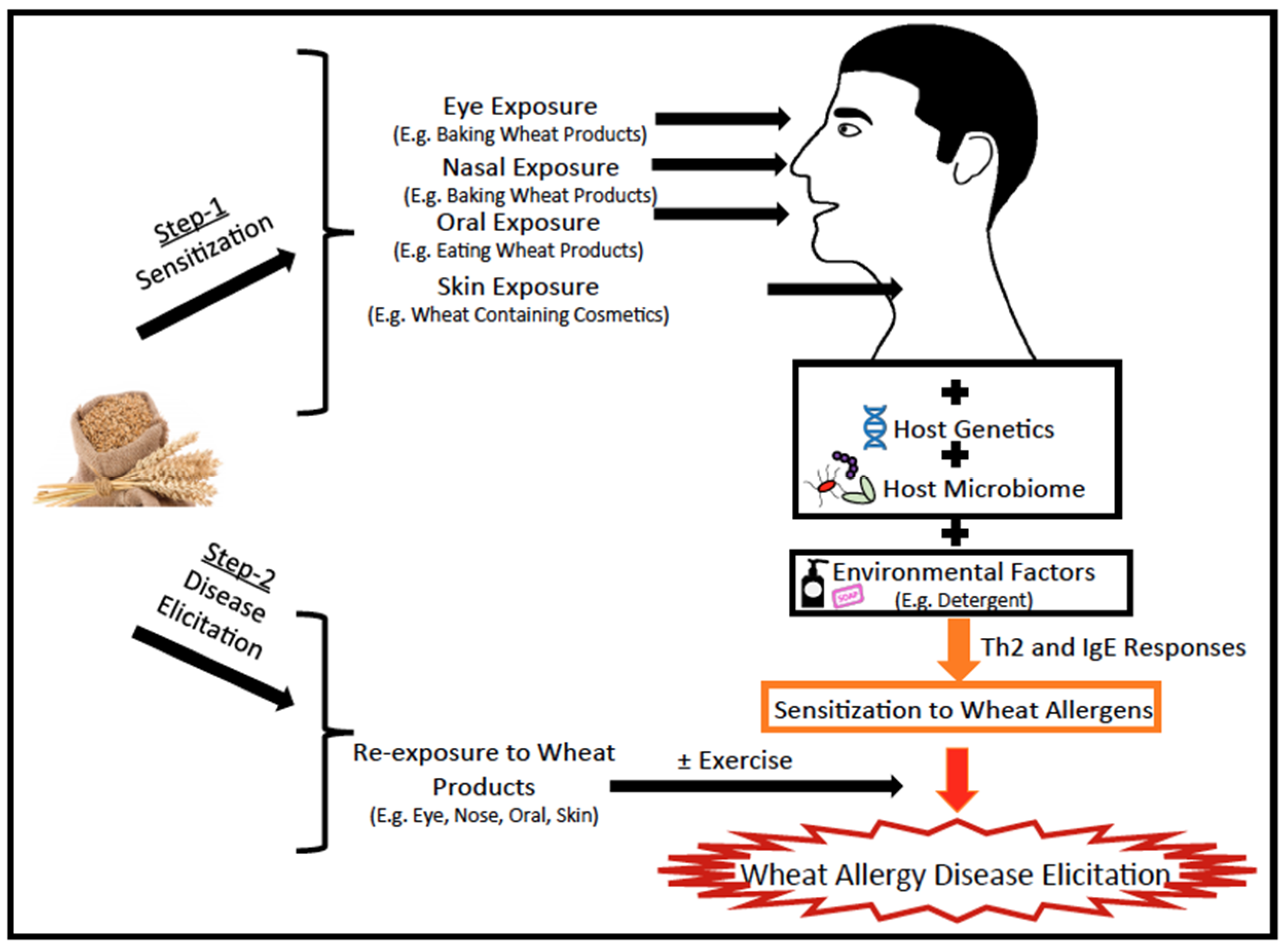
2. Insights in to the Molecular Mechanisms of Wheat Allergenicity as Revealed by Animal Models
2.1. Animal Models of Wheat Allergy: How Many Are There and What Lessons Have They Taught us?
2.1.1. Lessons from the Dog Model of Wheat Allergenicity
2.1.2. Lessons Learnt from the Mouse Models of Wheat Allergenicity
2.1.3. Lessons from the Rat Models of Sensitization to Wheat Allergens
2.2. Animal Models of Wheat Allergy: What Are the Current Challenges and Opportunities?
2.2.1. Current Challenges Facing Wheat Allergy Animal Models
Species, Sex, Age, and Strain of the Animals Used in Model Development
Routes of Sensitization and Elicitation of Disease
To Use or Not-to-Use Adjuvants for Wheat Allergenicity Testing?
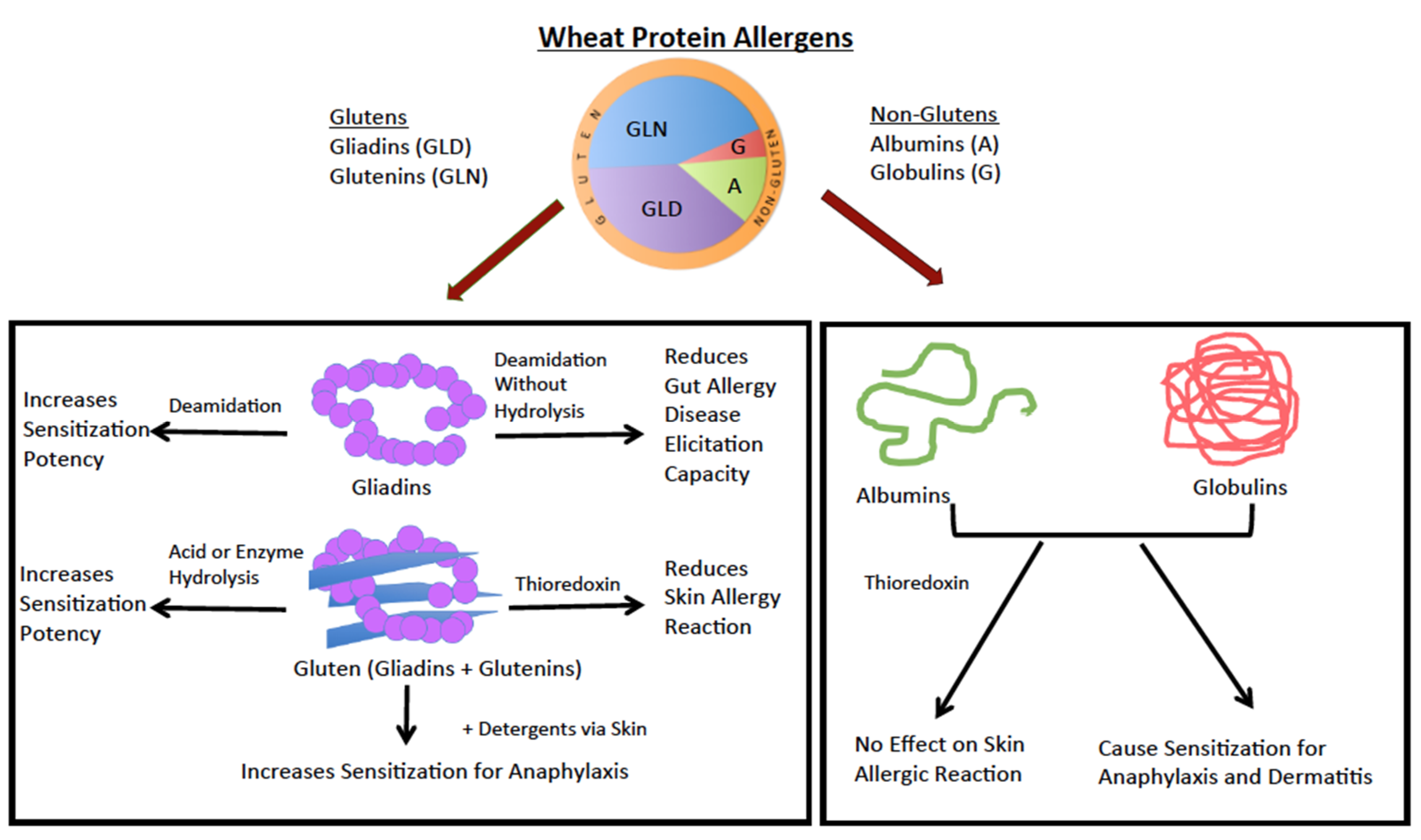
Wheat Proteins to Use in Animal Testing: Which Ones?
2.3. Opportunities for Improvement of Current Models and Development of New Models
2.4. What More Can Animal Models of Wheat Allergies Teach us? Anticipated Lessons
2.4.1. Determination of Genetic Susceptibility Factors for Wheat Allergy
2.4.2. Identification of Environmental Factors in Wheat Allergies
2.4.3. Determination of the Impact of Food Processing Methods on Wheat Allergenicity
2.4.4. Allergenicity Testing of Genetically Modified Wheat
2.4.5. Pre-Clinical Testing of Novel Pharmaceuticals and Vaccines for the Prevention and Treatment of Wheat Allergies
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Renz, H.; Allen, K.J.; Sicherer, S.H.; Sampson, H.A.; Lack, G.; Beyer, K.; Oettgen, H.C. Food allergy. Nat. Rev. Dis. Primers 2018, 4, 17098. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Sampson, H.A. Food allergy: A review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J. Allergy Clin. Immunol. 2018, 141, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Platts-Mills, T.A. The allergy epidemics: 1870-2010. J. Allergy Clin. Immunol. 2015, 136, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Gangur, V.; Kelly, C.; Navuluri, L. Sesame allergy: A growing food allergy of global proportions? Ann. Allergy Asthma Immunol. 2005, 95, 4–11, quiz 11-3, 44. [Google Scholar] [CrossRef]
- Gupta, R.S.; Springston, E.E.; Warrier, M.R.; Smith, B.; Kumar, R.; Pongracic, J.; Holl, J.L. The prevalence, severity, and distribution of childhood food allergy in the United States. Pediatrics 2011, 128, e9–e17. [Google Scholar] [CrossRef]
- Gupta, R.; Holdford, D.; Bilaver, L.; Dyer, A.; Holl, J.L.; Meltzer, D. The economic impact of childhood food allergy in the United States. JAMA Pediatr. 2013, 167, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- United States Food and Drug Administration Food Allergies: What You Need to Know. Available online: https://www.fda.gov/food/resourcesforyou/consumers/ucm079311.htm (accessed on 16 August 2018).
- Altman, D.R.; Chiaramonte, L.T. Public perception of food allergy. J. Allergy Clin. Immunol. 1996, 97, 1247–1251. [Google Scholar] [CrossRef]
- Vierk, K.A.; Koehler, K.M.; Fein, S.B.; Street, D.A. Prevalence of self-reported food allergy in American adults and use of food labels. J. Allergy Clin. Immunol. 2007, 119, 1504–1510. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations Wheat. Available online: http://www.fao.org/land-water/databases-and-software/crop-information/wheat/en/ (accessed on 20 August 2018).
- United States Department of Agriculture Wheat’s Role in the US Diet. Available online: https://www.ers.usda.gov/topics/crops/wheat/wheats-role-in-the-us-diet/ (accessed on 20 August 2018).
- Matsumura, Y.; Niitsuma, T.; Ito, H. A study of factors contributing to bakers’ allergy symptoms. Arerugi 1994, 43, 625–633. [Google Scholar]
- Baur, X.; Degens, P.O.; Sander, I. Baker’s asthma: Still among the most frequent occupational respiratory disorders. J. Allergy Clin. Immunol. 1998, 102 (6 Pt 1), 984–997. [Google Scholar] [CrossRef]
- Salcedo, G.; Quirce, S.; Diaz-Perales, A. Wheat allergens associated with Baker’s asthma. J. Investig. Allergol. Clin. Immunol. 2011, 21, 81–92. [Google Scholar]
- Mimura, T.; Yamagami, S.; Noma, H.; Kamei, Y.; Goto, M.; Kondo, A.; Matsubara, M. Specific IgE for wheat in tear fluid of patients with allergic conjunctivitis. Cutan. Ocul. Toxicol. 2015, 34, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Beaudouin, E.; Renaudin, J.M.; Morisset, M.; Codreanu, F.; Kanny, G.; Moneret-Vautrin, D.A. Food-dependent exercise-induced anaphylaxis--update and current data. Eur. Ann. Allergy Clin. Immunol. 2006, 38, 45–51. [Google Scholar]
- Morita, E.; Matsuo, H.; Chinuki, Y.; Takahashi, H.; Dahlstrom, J.; Tanaka, A. Food-dependent exercise-induced anaphylaxis -importance of omega-5 gliadin and HMW-glutenin as causative antigens for wheat-dependent exercise-induced anaphylaxis. Allergol. Int. 2009, 58, 493–498. [Google Scholar] [CrossRef]
- Scherf, K.A.; Brockow, K.; Biedermann, T.; Koehler, P.; Wieser, H. Wheat-dependent exercise-induced anaphylaxis. Clin. Exp. Allergy 2016, 46, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Furuta, G.T.; Katzka, D.A. Eosinophilic Esophagitis. N. Engl. J. Med. 2015, 373, 1640–1648. [Google Scholar] [CrossRef] [PubMed]
- Kottyan, L.C.; Rothenberg, M.E. Genetics of eosinophilic esophagitis. Mucosal Immunol. 2017, 10, 580–588. [Google Scholar] [CrossRef]
- Rochman, M.; Azouz, N.P.; Rothenberg, M.E. Epithelial origin of eosinophilic esophagitis. J. Allergy Clin. Immunol. 2018, 142, 10–23. [Google Scholar] [CrossRef]
- Leonard, M.M.; Sapone, A.; Catassi, C.; Fasano, A. Celiac Disease and Nonceliac Gluten Sensitivity: A Review. JAMA 2017, 318, 647–656. [Google Scholar] [CrossRef]
- Costes, L.M.; Meresse, B.; Cerf-Bensussan, N.; Samsom, J.N. The role of animal models in unravelling therapeutic targets in coeliac disease. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 437–450. [Google Scholar] [CrossRef]
- Ju, J.M.; Marietta, E.V.; Murray, J.A. Generating Transgenic Mouse Models for Studying Celiac Disease. Methods Mol. Biol. 2015, 1326, 23–33. [Google Scholar]
- Korneychuk, N.; Meresse, B.; Cerf-Bensussan, N. Lessons from rodent models in celiac disease. Mucosal Immunol. 2015, 8, 18–28. [Google Scholar] [CrossRef]
- Buchanan, B.B.; Adamidi, C.; Lozano, R.M.; Yee, B.C.; Momma, M.; Kobrehel, K.; Ermel, R.; Frick, O.L. Thioredoxin-linked mitigation of allergic responses to wheat. Proc. Natl. Acad. Sci. USA 1997, 94, 5372–5377. [Google Scholar] [CrossRef]
- Kozai, H.; Yano, H.; Matsuda, T.; Kato, Y. Wheat-dependent exercise-induced anaphylaxis in mice is caused by gliadin and glutenin treatments. Immunol. Lett. 2006, 102, 83–90. [Google Scholar] [CrossRef]
- Tanaka, M.; Nagano, T.; Yano, H.; Matsuda, T.; Ikeda, T.M.; Haruma, K.; Kato, Y. Impact of omega-5 gliadin on wheat-dependent exercise-induced anaphylaxis in mice. Biosci. Biotechnol. Biochem. 2011, 75, 313–317. [Google Scholar] [CrossRef]
- Denery-Papini, S.; Bodinier, M.; Pineau, F.; Triballeau, S.; Tranquet, O.; Adel-Patient, K.; Moneret-Vautrin, D.A.; Bakan, B.; Marion, D.; Mothes, T.; et al. Immunoglobulin-E-binding epitopes of wheat allergens in patients with food allergy to wheat and in mice experimentally sensitized to wheat proteins. Clin. Exp. Allergy 2011, 41, 1478–1492. [Google Scholar] [CrossRef]
- Bodinier, M.; Leroy, M.; Ah-Leung, S.; Blanc, F.; Tranquet, O.; Denery-Papini, S.; Wal, J.M.; Adel-Patient, K. Sensitization and elicitation of an allergic reaction to wheat gliadins in mice. J. Agric. Food Chem. 2009, 57, 1219–1225. [Google Scholar] [CrossRef]
- Adel-Patient, K.; Nahori, M.A.; Proust, B.; Lapa e Silva, J.R.; Creminon, C.; Wal, J.M.; Vargaftig, B.B. Elicitation of the allergic reaction in beta-lactoglobulin-sensitized Balb/c mice: Biochemical and clinical manifestations differ according to the structure of the allergen used for challenge. Clin. Exp. Allergy 2003, 33, 376–385. [Google Scholar] [CrossRef]
- Gourbeyre, P.; Denery-Papini, S.; Larre, C.; Gaudin, J.C.; Brossard, C.; Bodinier, M. Wheat gliadins modified by deamidation are more efficient than native gliadins in inducing a Th2 response in Balb/c mice experimentally sensitized to wheat allergens. Mol. Nutr. Food Res. 2012, 56, 336–344. [Google Scholar] [CrossRef]
- Abe, R.; Shimizu, S.; Yasuda, K.; Sugai, M.; Okada, Y.; Chiba, K.; Akao, M.; Kumagai, H.; Kumagai, H. Evaluation of reduced allergenicity of deamidated gliadin in a mouse model of wheat-gliadin allergy using an antibody prepared by a peptide containing three epitopes. J. Agric. Food Chem. 2014, 62, 2845–2852. [Google Scholar] [CrossRef]
- Lauriere, M.; Pecquet, C.; Bouchez-Mahiout, I.; Snegaroff, J.; Bayrou, O.; Raison-Peyron, N.; Vigan, M. Hydrolysed wheat proteins present in cosmetics can induce immediate hypersensitivities. Contact Dermat. 2006, 54, 283–289. [Google Scholar] [CrossRef]
- Fukutomi, Y.; Itagaki, Y.; Taniguchi, M.; Saito, A.; Yasueda, H.; Nakazawa, T.; Hasegawa, M.; Nakamura, H.; Akiyama, K. Rhinoconjunctival sensitization to hydrolyzed wheat protein in facial soap can induce wheat-dependent exercise-induced anaphylaxis. J. Allergy Clin. Immunol. 2011, 127, 531–533. [Google Scholar] [CrossRef] [PubMed]
- Pelkonen, A.S.; Makinen-Kiljunen, S.; Hilvo, S.; Siltanen, M.; Makela, M.J. Severe allergic reaction to gluten hydrolysate without reaction to wheat. Ann. Allergy Asthma Immunol. 2011, 106, 343–344. [Google Scholar] [CrossRef] [PubMed]
- Chinuki, Y.; Morita, E. Wheat-dependent exercise-induced anaphylaxis sensitized with hydrolyzed wheat protein in soap. Allergol. Int. 2012, 61, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Adachi, R.; Nakamura, R.; Sakai, S.; Fukutomi, Y.; Teshima, R. Sensitization to acid-hydrolyzed wheat protein by transdermal administration to BALB/c mice, and comparison with gluten. Allergy 2012, 67, 1392–1399. [Google Scholar] [CrossRef] [PubMed]
- Navuluri, L.; Parvataneni, S.; Hassan, H.; Birmingham, N.P.; Kelly, C.; Gangur, V. Allergic and anaphylactic response to sesame seeds in mice: Identification of Ses i 3 and basic subunit of 11s globulins as allergens. Int. Arch. Allergy Immunol. 2006, 140, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Birmingham, N.P.; Parvataneni, S.; Hassan, H.M.; Harkema, J.; Samineni, S.; Navuluri, L.; Kelly, C.J.; Gangur, V. An adjuvant-free mouse model of tree nut allergy using hazelnut as a model tree nut. Int. Arch. Allergy Immunol. 2007, 144, 203–210. [Google Scholar] [CrossRef]
- Gonipeta, B.; Parvataneni, S.; Tempelman, R.J.; Gangur, V. An adjuvant-free mouse model to evaluate the allergenicity of milk whey protein. J. Dairy Sci. 2009, 92, 4738–4744. [Google Scholar] [CrossRef]
- Parvataneni, S.; Gonipeta, B.; Tempelman, R.J.; Gangur, V. Development of an adjuvant-free cashew nut allergy mouse model. Int. Arch. Allergy Immunol. 2009, 149, 299–304. [Google Scholar] [CrossRef]
- Gonipeta, B.; Parvataneni, S.; Paruchuri, P.; Gangur, V. Long-term characteristics of hazelnut allergy in an adjuvant-free mouse model. Int. Arch. Allergy Immunol. 2010, 152, 219–225. [Google Scholar] [CrossRef]
- Cianferoni, A. Wheat allergy: Diagnosis and management. J. Asthma Allergy 2016, 9, 13–25. [Google Scholar] [CrossRef]
- Jin, Y.; Ebaugh, S.; Martens, A.; Gao, H.; Olson, E.; Ng, P.K.W.; Gangur, V. A Mouse Model of Anaphylaxis and Atopic Dermatitis to Salt-Soluble Wheat Protein Extract. Int. Arch. Allergy Immunol. 2017, 174, 7–16. [Google Scholar] [CrossRef]
- Khodoun, M.V.; Strait, R.; Armstrong, L.; Yanase, N.; Finkelman, F.D. Identification of markers that distinguish IgE- from IgG-mediated anaphylaxis. Proc. Natl. Acad. Sci. USA 2011, 108, 12413–12418. [Google Scholar] [CrossRef]
- Kroghsbo, S.; Rigby, N.M.; Johnson, P.E.; Adel-Patient, K.; Bogh, K.L.; Salt, L.J.; Mills, E.N.; Madsen, C.B. Assessment of the sensitizing potential of processed peanut proteins in Brown Norway rats: Roasting does not enhance allergenicity. PLoS ONE 2014, 9, e96475. [Google Scholar] [CrossRef]
- Ballegaard, A.R.; Madsen, C.B.; Bogh, K.L. An Animal Model for Wheat Allergy Skin Sensitization: A Comparative Study in Naive versus Tolerant Brown Norway Rats. Int. Arch. Allergy Immunol. 2019, 178, 106–118. [Google Scholar] [CrossRef]
- Gonipeta, B.; Kim, E.; Gangur, V. Mouse models of food allergy: How well do they simulate the human disorder? Crit. Rev. Food Sci. Nutr. 2015, 55, 437–452. [Google Scholar] [CrossRef]
- Parvataneni, S.; Gonipeta, B.; Acharya, H.G.; Gangur, V. An Adjuvant-Free Mouse Model of Transdermal Sensitization and Oral Elicitation of Anaphylaxis to Shellfish. Int. Arch. Allergy Immunol. 2015, 168, 269–276. [Google Scholar] [CrossRef]
- Li, X.M.; Serebrisky, D.; Lee, S.Y.; Huang, C.K.; Bardina, L.; Schofield, B.H.; Stanley, J.S.; Burks, A.W.; Bannon, G.A.; Sampson, H.A. A murine model of peanut anaphylaxis: T- and B-cell responses to a major peanut allergen mimic human responses. J. Allergy Clin. Immunol. 2000, 106 (1 Pt 1), 150–158. [Google Scholar] [CrossRef]
- Li, X.M.; Kleiner, G.; Huang, C.K.; Lee, S.Y.; Schofield, B.; Soter, N.A.; Sampson, H.A. Murine model of atopic dermatitis associated with food hypersensitivity. J. Allergy Clin. Immunol. 2001, 107, 693–702. [Google Scholar] [CrossRef]
- Ganeshan, K.; Neilsen, C.V.; Hadsaitong, A.; Schleimer, R.P.; Luo, X.; Bryce, P.J. Impairing oral tolerance promotes allergy and anaphylaxis: A new murine food allergy model. J. Allergy Clin. Immunol. 2009, 123, 231–238.e4. [Google Scholar] [CrossRef]
- Dearman, R.J.; Kimber, I. A mouse model for food allergy using intraperitoneal sensitization. Methods 2007, 41, 91–98. [Google Scholar] [CrossRef]
- Dearman, R.J.; Kimber, I. Animal models of protein allergenicity: Potential benefits, pitfalls and challenges. Clin. Exp. Allergy 2009, 39, 458–468. [Google Scholar] [CrossRef]
- Ladics, G.S.; Selgrade, M.K. Identifying food proteins with allergenic potential: Evolution of approaches to safety assessment and research to provide additional tools. Regul. Toxicol. Pharmacol. 2009, 54 (Suppl. 3), S2–S6. [Google Scholar] [CrossRef]
- Selgrade, M.K.; Bowman, C.C.; Ladics, G.S.; Privalle, L.; Laessig, S.A. Safety assessment of biotechnology products for potential risk of food allergy: Implications of new research. Toxicol. Sci. 2009, 110, 31–39. [Google Scholar] [CrossRef]
- Buchanan, B.B.; Frick, O.L. The dog as a model for food allergy. Ann. N. Y. Acad. Sci. 2002, 964, 173–183. [Google Scholar] [CrossRef]
- Shewry, P.R. Do ancient types of wheat have health benefits compared with modern bread wheat? J. Cereal. Sci. 2018, 79, 469–476. [Google Scholar] [CrossRef]
- Kohno, K.; Takahashi, H.; Endo, T.R.; Matsuo, H.; Shiwaku, K.; Morita, E. Characterization of a hypoallergenic wheat line lacking omega-5 gliadin. Allergol. Int. 2016, 65, 400–405. [Google Scholar] [CrossRef]
- Larre, C.; Lupi, R.; Gombaud, G.; Brossard, C.; Branlard, G.; Moneret-Vautrin, D.A.; Rogniaux, H.; Denery-Papini, S. Assessment of allergenicity of diploid and hexaploid wheat genotypes: Identification of allergens in the albumin/globulin fraction. J. Proteom. 2011, 74, 1279–1289. [Google Scholar] [CrossRef]
- Xu, H.; Feely, S.L.; Wang, X.; Liu, D.X.; Borda, J.T.; Dufour, J.; Li, W.; Aye, P.P.; Doxiadis, G.G.; Khosla, C.; et al. Gluten-sensitive enteropathy coincides with decreased capability of intestinal T cells to secrete IL-17 and IL-22 in a macaque model for celiac disease. Clin. Immunol. 2013, 147, 40–49. [Google Scholar] [CrossRef]
- Sestak, K.; Thwin, H.; Dufour, J.; Aye, P.P.; Liu, D.X.; Moehs, C.P. The effects of reduced gluten barley diet on humoral and cell-mediated systemic immune responses of gluten-sensitive rhesus macaques. Nutrients 2015, 7, 1657–1671. [Google Scholar] [CrossRef]
- Mohan, M.; Chow, C.T.; Ryan, C.N.; Chan, L.S.; Dufour, J.; Aye, P.P.; Blanchard, J.; Moehs, C.P.; Sestak, K. Dietary Gluten-Induced Gut Dysbiosis Is Accompanied by Selective Upregulation of microRNAs with Intestinal Tight Junction and Bacteria-Binding Motifs in Rhesus Macaque Model of Celiac Disease. Nutrients 2016, 8. [Google Scholar] [CrossRef]
- Iga, N.; Tanizaki, H.; Endo, Y.; Egawa, G.; Fujisawa, A.; Tanioka, M.; Miyachi, Y.; Kabashima, K. Hydrolyzed wheat protein-containing facial soap-induced wheat-dependent exercise-induced anaphylaxis in a patient without filaggrin mutations. J. Dermatol. 2013, 40, 494–495. [Google Scholar] [CrossRef]
- Mizuno, O.; Nomura, T.; Ohguchi, Y.; Suzuki, S.; Nomura, Y.; Hamade, Y.; Hoshina, D.; Sandilands, A.; Akiyama, M.; McLean, W.H.; et al. Loss-of-function mutations in the gene encoding filaggrin underlie a Japanese family with food-dependent exercise-induced anaphylaxis. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 805–808. [Google Scholar] [CrossRef]
- Ruiz-Ortiz, E.; Montraveta, M.; Cabre, E.; Herrero-Mata, M.J.; Pujol-Borrell, R.; Palou, E.; Faner, R. HLA-DQ2/DQ8 and HLA-DQB1*02 homozygosity typing by real-time polymerase chain reaction for the assessment of celiac disease genetic risk: Evaluation of a Spanish celiac population. Tissue Antigens 2014, 84, 545–553. [Google Scholar] [CrossRef]
- Lundin, K.E.; Qiao, S.W.; Snir, O.; Sollid, L.M. Coeliac disease - from genetic and immunological studies to clinical applications. Scand. J. Gastroenterol. 2015, 50, 708–717. [Google Scholar] [CrossRef]
- Sollid, L.M. The roles of MHC class II genes and post-translational modification in celiac disease. Immunogenetics 2017, 69, 605–616. [Google Scholar] [CrossRef]
- Rockwell, C.E.; Zhang, M.; Fields, P.E.; Klaassen, C.D. Th2 skewing by activation of Nrf2 in CD4(+) T cells. J. Immunol. 2012, 188, 1630–1637. [Google Scholar] [CrossRef]
- Tobar, S.; Tordesillas, L.; Berin, M.C. Triclosan promotes epicutaneous sensitization to peanut in mice. Clin. Transl. Allergy 2016, 6, 13. [Google Scholar] [CrossRef]
- Marshall, N.B.; Lukomska, E.; Nayak, A.P.; Long, C.M.; Hettick, J.M.; Anderson, S.E. Topical application of the anti-microbial chemical triclosan induces immunomodulatory responses through the S100A8/A9-TLR4 pathway. J. Immunotoxicol. 2017, 14, 50–59. [Google Scholar] [CrossRef]
- Walters, G.I.; Robertson, A.S.; Moore, V.C.; Burge, P.S. Occupational asthma caused by sensitization to a cleaning product containing triclosan. Ann. Allergy Asthma Immunol. 2017, 118, 370–371. [Google Scholar] [CrossRef]
- Kourosh, A.; Luna, R.A.; Balderas, M.; Nance, C.; Anagnostou, A.; Devaraj, S.; Davis, C.M. Fecal microbiome signatures are different in food-allergic children compared to siblings and healthy children. Pediatr. Allergy Immunol. 2018, 29, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Lee-Sarwar, K.; Hauser, R.; Calafat, A.M.; Ye, X.; O’Connor, G.T.; Sandel, M.; Bacharier, L.B.; Zeiger, R.S.; Laranjo, N.; Gold, D.R.; et al. Prenatal and early-life triclosan and paraben exposure and allergic outcomes. J. Allergy Clin. Immunol. 2018, 142, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhong, Y.; Shen, X.; Yang, K.; Cai, W. Maternal and early-life vitamin D deficiency enhances allergic reaction in an ovalbumin-sensitized BALB/c mouse model. Food Nutr. Res. 2018, 62. [Google Scholar] [CrossRef] [PubMed]
- Verhoeckx, K.C.M.; Vissers, Y.M.; Baumert, J.L.; Faludi, R.; Feys, M.; Flanagan, S.; Herouet-Guicheney, C.; Holzhauser, T.; Shimojo, R.; van der Bolt, N.; et al. Food processing and allergenicity. Food Chem. Toxicol. 2015, 80, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, T.; Para, R.; Gonipeta, B.; Reitmeyer, M.; He, Y.; Srkalovic, I.; Ng, P.K.; Gangur, V. Effect of extrusion processing on immune activation properties of hazelnut protein in a mouse model. Int. J. Food Sci. Nutr. 2016, 67, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Vanga, S.K.; Singh, A.; Raghavan, V. Review of conventional and novel food processing methods on food allergens. Crit. Rev. Food Sci. Nutr. 2017, 57, 2077–2094. [Google Scholar] [CrossRef]
- Phromraksa, P.; Nagano, H.; Boonmars, T.; Kamboonruang, C. Identification of proteolytic bacteria from thai traditional fermented foods and their allergenic reducing potentials. J. Food Sci. 2008, 73, M189–M195. [Google Scholar] [CrossRef]
- Tanabe, S. Analysis of food allergen structures and development of foods for allergic patients. Biosci. Biotechnol. Biochem. 2008, 72, 649–659. [Google Scholar] [CrossRef]
- Kobayashi, T.; Ito, T.; Kawakami, H.; Fuzishiro, K.; Hirano, H.; Okubo, Y.; Tsuboi, R. Eighteen cases of wheat allergy and wheat-dependent exercise-induced urticaria/anaphylaxis sensitized by hydrolyzed wheat protein in soap. Int. J. Dermatol. 2015, 54, e302–e305. [Google Scholar] [CrossRef]
- Lombardo, C.; Bolla, M.; Chignola, R.; Senna, G.; Rossin, G.; Caruso, B.; Tomelleri, C.; Cecconi, D.; Brandolini, A.; Zoccatelli, G. Study on the Immunoreactivity of Triticum monococcum (Einkorn) Wheat in Patients with Wheat-Dependent Exercise-Induced Anaphylaxis for the Production of Hypoallergenic Foods. J. Agric. Food Chem. 2015, 63, 8299–8306. [Google Scholar] [CrossRef]
- Stefanska, I.; Piasecka-Jozwiak, K.; Kotyrba, D.; Kolenda, M.; Stecka, K.M. Selection of lactic acid bacteria strains for the hydrolysis of allergenic proteins of wheat flour. J. Sci. Food Agric. 2016, 96, 3897–3905. [Google Scholar] [CrossRef]
- Magishi, N.; Yuikawa, N.; Kobayashi, M.; Taniuchi, S. Degradation and removal of soybean allergen in Japanese soy sauce. Mol. Med. Rep. 2017, 16, 2264–2268. [Google Scholar] [CrossRef]
- Mishra, A.; Arora, N. Allergenicity Assessment of Transgenic Wheat Lines In Silico. Methods Mol. Biol. 2017, 1679, 97–111. [Google Scholar]
- Rey, M.D.; Calderon, M.C.; Rodrigo, M.J.; Zacarias, L.; Alos, E.; Prieto, P. Novel Bread Wheat Lines Enriched in Carotenoids Carrying Hordeum chilense Chromosome Arms in the ph1b Background. PLoS ONE 2015, 10, e0134598. [Google Scholar] [CrossRef]
- Altenbach, S.B.; Tanaka, C.K.; Pineau, F.; Lupi, R.; Drouet, M.; Beaudouin, E.; Morisset, M.; Denery-Papini, S. Assessment of the Allergenic Potential of Transgenic Wheat (Triticum aestivum) with Reduced Levels of omega5-Gliadins, the Major Sensitizing Allergen in Wheat-Dependent Exercise-Induced Anaphylaxis. J. Agric. Food Chem. 2015, 63, 9323–9332. [Google Scholar] [CrossRef]
- Su, Y.; Romeu-Bonilla, E.; Heiland, T. Next generation immunotherapy for treepollen allergies. Hum. Vaccines Immunother. 2017, 13, 2402–2415. [Google Scholar] [CrossRef]
- McGonigle, P.; Ruggeri, B. Animal models of human disease: Challenges in enabling translation. Biochem. Pharmacol. 2014, 87, 162–171. [Google Scholar] [CrossRef]
| Model/Developers | Wheat Protein Used | Sensitization (Route, Dose, Age, Gender, Adjuvant) | Elicitation of Reaction (Route, Dose and Age) | Immune Markers | Disease Phenotype |
|---|---|---|---|---|---|
| Dog model Spaniel/basenji inbred dog colony/Buchanan et al. (1997) | Wheat + cow’s milk + beef extract | SC injection (on days of age: days 1, 22, 29, 50, 57, 78, and 85) 1 ug each of the food allergens + 0.2 mL alum; SC injection distemper-hepatitis vaccine on days 21, 49, and 77; Booster at bimonthly intervals with 10 ug each of the food allergens; Bleedings at 3, 4 months | At 6 months: Feeding challenge with 200 g wheat flour gruel or cow’s milk | Specific IgE, Skin prick test | Vomiting and/or diarrhea (increased number of loose or watery stools for 2–4 days after the feeding challenge) |
| Rat models Brown Norway inbred rats (bred on gluten-free diet for three generations)/Kroghsbo et al. (2014) Brown Norway inbred rats/Bellegaard et al. (2019) | Gluten (Unmodified, acid hydrolyzed, Enzyme hydrolyzed) Native gluten vs. acid hydrolyzed gluten | IP sensitization: day 0: 200 ug adsorbed on Alhydrogel/rat in PBS; Days 14, 21, and 28: 20 ug in 0.9% NaCl; 0.2 mL volume/bleeding on day 35 | None | Specific IgE, IgG Rat Basophilic Leukemia cell degranulation in vitro | None |
| Oral sensitization: Female BN rats; Days 1 to 35: gavage with 0.2, 2, and 20 mg suspension in 0.5 mL PBS; Bleeding on days 0, 14, 28, and 42 | None | Specific IgE, IgG Rat Basophilic Leukemia cell degranulation in vitro | None | ||
| Skin sensitization: damage to skin then apply gluten without adjuvant 3 times per week for 3–5 weeks | None | Specific IgE, IgG antibodies | None |
| Model/Developers | Wheat Protein Used | Sensitization (Route, Dose, Age, Gender, Adjuvant) | Elicitation of Reaction (Route, Dose and Age) | Immune Markers | Disease Phenotype |
|---|---|---|---|---|---|
| B10.A model by Kozai et al. (2006) | Water/saline- soluble protein extract, Alcohol-soluble protein extract, alkali-soluble protein | IP (day 0, 14, 28, and 42) 10 ug + 1 mg alum/mouse Female B10.A | 20 mg/0.5 mL/mouse oral feeding plus acute or moderate exercise | Specific IgE | Time to exhaustion, mucosal lesions in the small intestine, wheat protein leakage into the liver |
| Balb/cJ B10.A C3H/HeJ model by Bodinier et al. (2009) | Gliadin (Hardi) | IP (day 0, 10, 20, and 30) 10 or 20 ug + 1 mg alum/mouse 3-week females Balb/CJ 4–5-week females B10.A, C3H/HeJ | Nasal administration (10 ug on day 40) | Specific IgE, IgG1; IL-4, IL-5, IL-10, GM-CSF, IL-12 in lungs; cell counts in lung fluids | Eosinophil influx to lungs upon challenge |
| B10.A model by Tanaka et al. (2011) | Gliadin, purified ω5-gliadin | Gliadin 100 ug/mouse first IP injection; 50 ug/mouse for next 5 injections at weekly interval + 1 mg alum/mouse Female B10.A 5 weeks age | Gliadin at 0.1 and 0.8 mg/mouse/0.5 mL acetic acid; ω5 gliadin at 0.1 mg/mouse/0.5 mL acetic acid Oral feeding plus acute exercise | Specific IgE | Anaphylaxis by hypothermia shock response, voluntary exercise performance, leakage of wheat proteins into the blood |
| Balb/cJ model by Denery-Papini, et al. (2011) | Gliadins extract (Hardi) LTP1 | IP (day 0, 10, 20, and 30) 10 ug of gliadins or LTP1 + 1 mg alum/mouse 3-week females | As in Bodinier et al. (2009) | Specific IgE | None reported |
| Balb/cJ model by Gourbeyre et al. (2012) | Deamidated gliadins (acid hydrolysis) (Hardi) | IP (day 0, 10, 20, and 30) 10 ug + 1 mg alum/mouse, 6-week females | IP injection with 1 mg + 1 mg alum on day 38 | Total IgE, specific IgG1, IgG2a | None reported |
| Balb/c model by Adachi et al. (2012) | Acid hydrolyzed gluten | Skin sensitization (days 1–3, 8–10, 15–17, and 22–24) 0.5 mg | IP injection with 1 mg on days 18 or 25 | Specific IgE, IgG1, plasma histamine levels | Hypothermia shock response |
| Balb/c model by Abe et al. (2014) | Native gliadin and deamidated gliadin by carboxylated cation exchange resin | IP (day 0, day 14) 50 ug of native gluten with 1 mg alum/mouse 5-week males | Intra-gastric administration of deamidated gliadin, 10 mg on days 28, 30, 32, 34, 36, 38, and 40 | Specific IgE; peritoneal mast cells, histamine (gut and plasma) | Intestinal permeability, mast cell degranulation |
| Balb/cJ model by Jin et al. (2017) | Saline-soluble wheat protein (duram) | IP (days 0, 10, 24, and 40), 10 ug + 1 mg alum, 6–8-week females | IP injection with 0.5 mg, 1 week after last sensitization and repeated | Specific IgE, IgG1, total IgE murine mast cell protease-1, correlation analysis among readouts, cytokines, chemokines, adhesion molecule in skin lesion | Hypothermia shock response, atopic dermatitis, skin mast cell degranulation, mucosal mast cell mediator release |
| Protein | Pathogenic IgE Binding Peptide Epitopes |
|---|---|
| Salt-soluble protein | |
| LTP1 * | (1) QARSQSDRQS; (2) GIARGIHNLN |
| Alcohol-soluble proteins | |
| α-gliadin | (1) PLVQQQ; (2) QQQFPGQQQQ #; (3) YLQLQLP #; (4) YPQQQPQYLQ; (5) SFQQPQQQYP |
| ω2-gliadin | (1) FPTPQQQFPE; (2) QQSFPLQPQQ #; (3) QQLFPELQ |
| ω5-gliadin | (1) QQFPQQQ #; (2) QQLPQQQ #; (3) QQSPQQQ #; (4) QQEFPQQQ; (5) QQQFPQQEFP |
| Species | Wheat Allergen | Exposure Route | Sensitization | Elicitation of Reaction | |
|---|---|---|---|---|---|
| Dog | Gliadins | IP | IgE | ND | |
| Glutenins Albumins Globulins |  | Oral | ND | Vomiting, Diarrhea | |
| Skin | ND | Skin Reaction | |||
| Thioredoxin Modified Gliadins & Glutenins | Skin | ND | Reduced Skin Reaction | ||
| Mouse | Gliadins | IP | IgE | Anaphylaxis, EIA | |
| Gluten | Skin | No IgE | No Anaphylaxis | ||
| Gluten + Detergent | Skin | IgE | Anaphylaxis | ||
| Acid-Hydrolyzed gluten (AHG) | Skin | IgE | Anaphylaxis | ||
| AHG + Detergent | Skin | Increased IgE | Increased Anaphylaxis | ||
| Deamidated-Gliadins (DG) | IP | Increased IgE | ND | ||
| DG | Oral | ND | Reduced Gut Reactions | ||
| Albumins + Globulins | IP | IgE | Anaphylaxis | ||
| Dermatitis (Th1, Th2, Th17 Cytokines + Allergenic Chemokines) | |||||
| Rat | Gluten and AHG | IP | IgE | ND | |
| Skin | IgE | ND | |||
| Enzyme Hydrolyzed Gluten | Oral | IgE | ND | ||
| IP | IgE | ND | |||
| Oral | IgE | ND | |||
| Model Characteristics | Dog | Rat | Mouse |
|---|---|---|---|
| Strengths | |||
| Protocol used in the model development is relatively simple | No | Yes | Yes |
| Used wheat flour for exposure | Yes | No | No |
| Used purified wheat allergens/extracts for exposure | No | Yes | Yes |
| Studied immune response to all four general groups of wheat allergens | Yes | No | No (most) |
| Used oral route to induce sensitization | No | Yes | No |
| Studied clinical reactions after oral exposure | Yes | No | No |
| Limitations | |||
| Used adjuvant to induce sensitization | Yes | No | Yes |
| Used injection to induce sensitization | Yes | No | Yes |
| Used injection to elicit clinical reaction | Yes (skin) | No | Yes |
| Limited availability of animal breed/strain | Yes (Limited) | Commercially Available | Commercially Available |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, Y.; Acharya, H.G.; Acharya, D.; Jorgensen, R.; Gao, H.; Secord, J.; Ng, P.K.W.; Gangur, V. Advances in Molecular Mechanisms of Wheat Allergenicity in Animal Models: A Comprehensive Review. Molecules 2019, 24, 1142. https://doi.org/10.3390/molecules24061142
Jin Y, Acharya HG, Acharya D, Jorgensen R, Gao H, Secord J, Ng PKW, Gangur V. Advances in Molecular Mechanisms of Wheat Allergenicity in Animal Models: A Comprehensive Review. Molecules. 2019; 24(6):1142. https://doi.org/10.3390/molecules24061142
Chicago/Turabian StyleJin, Yining, Harini G. Acharya, Devansh Acharya, Rick Jorgensen, Haoran Gao, James Secord, Perry K. W. Ng, and Venugopal Gangur. 2019. "Advances in Molecular Mechanisms of Wheat Allergenicity in Animal Models: A Comprehensive Review" Molecules 24, no. 6: 1142. https://doi.org/10.3390/molecules24061142
APA StyleJin, Y., Acharya, H. G., Acharya, D., Jorgensen, R., Gao, H., Secord, J., Ng, P. K. W., & Gangur, V. (2019). Advances in Molecular Mechanisms of Wheat Allergenicity in Animal Models: A Comprehensive Review. Molecules, 24(6), 1142. https://doi.org/10.3390/molecules24061142





