Enhanced Supercapacitor Performance Based on CoAl Layered Double Hydroxide-Polyaniline Hybrid Electrodes Manufactured Using Hydrothermal-Electrodeposition Technology
Abstract
1. Introduction
2. Results and Discussion
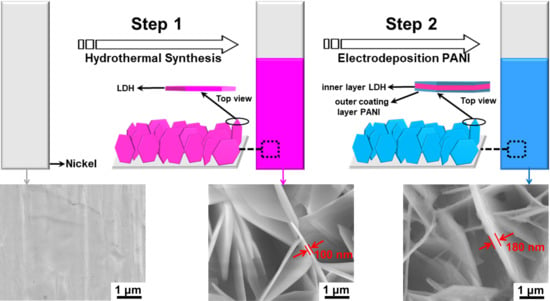
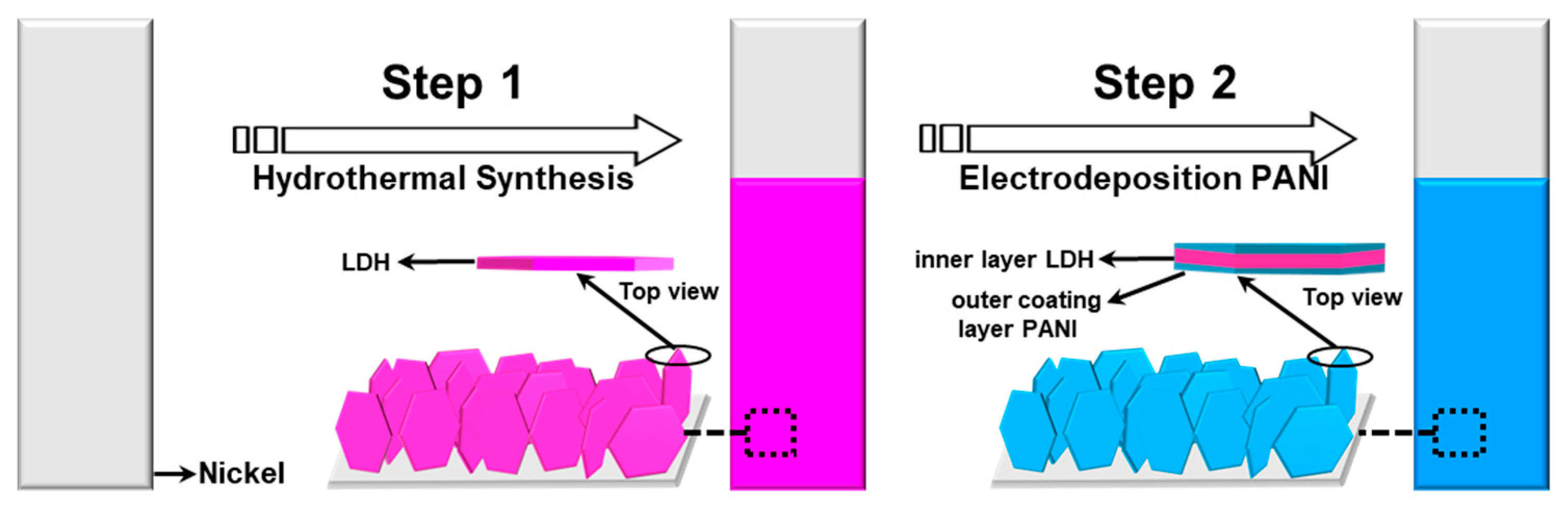
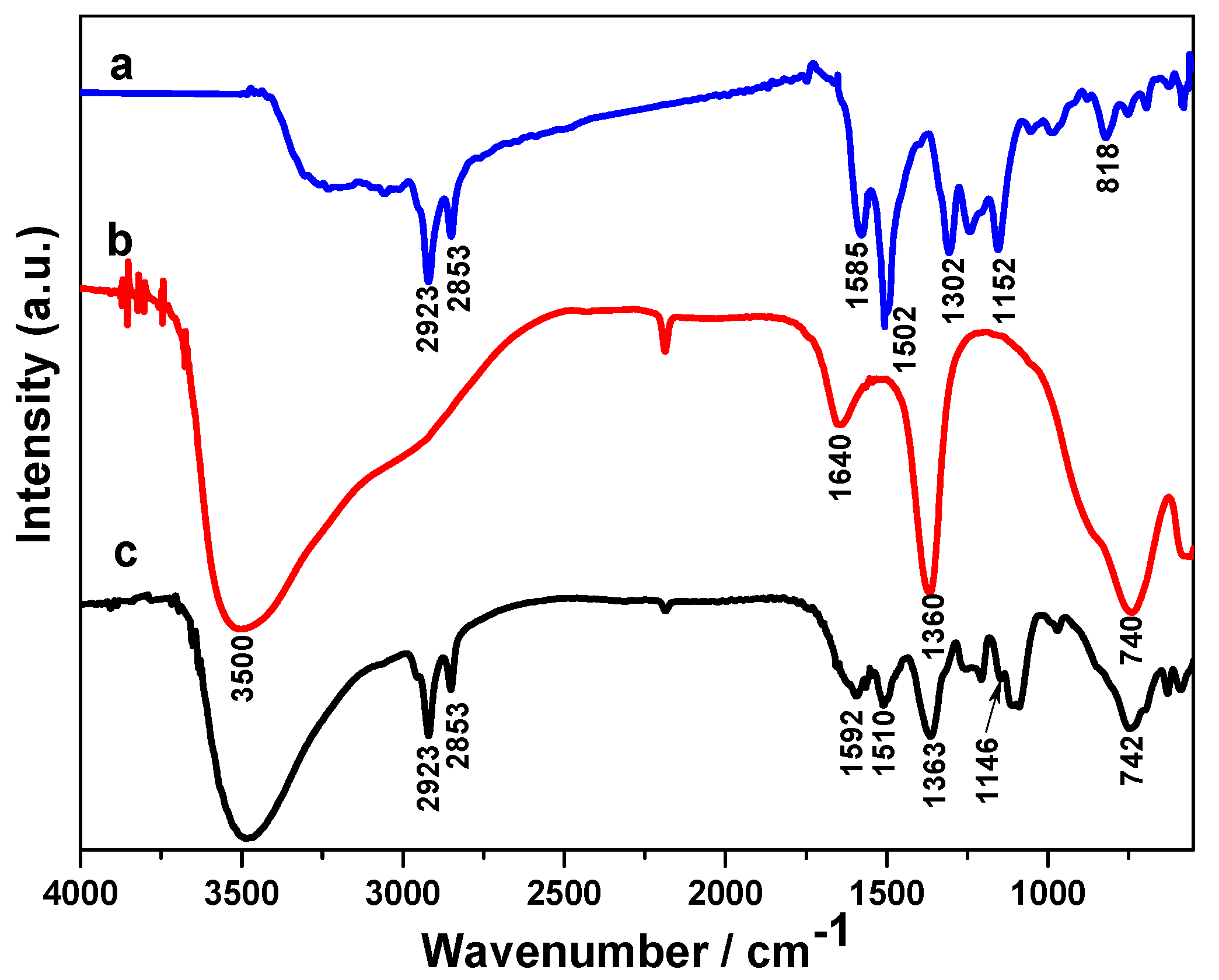
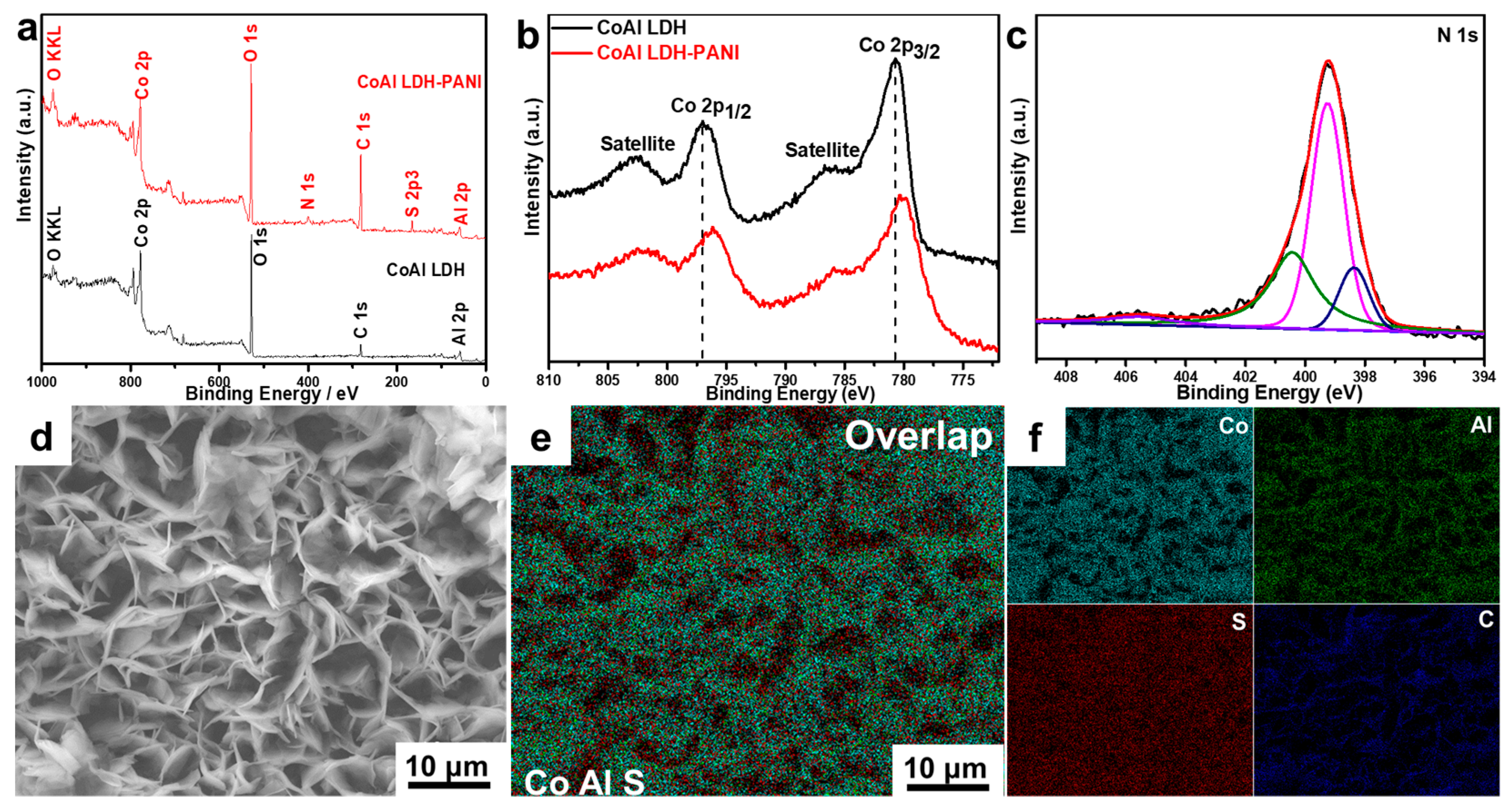
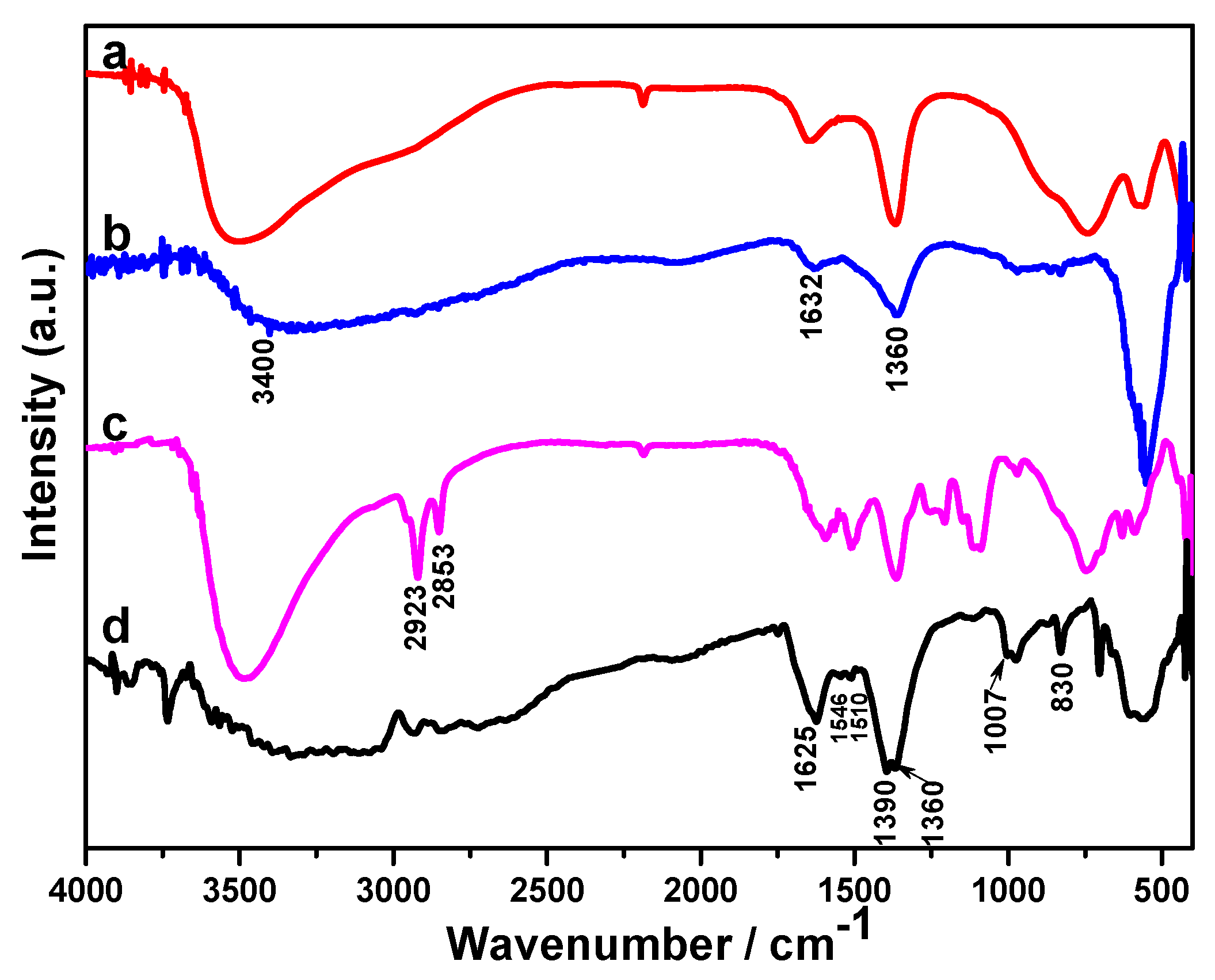
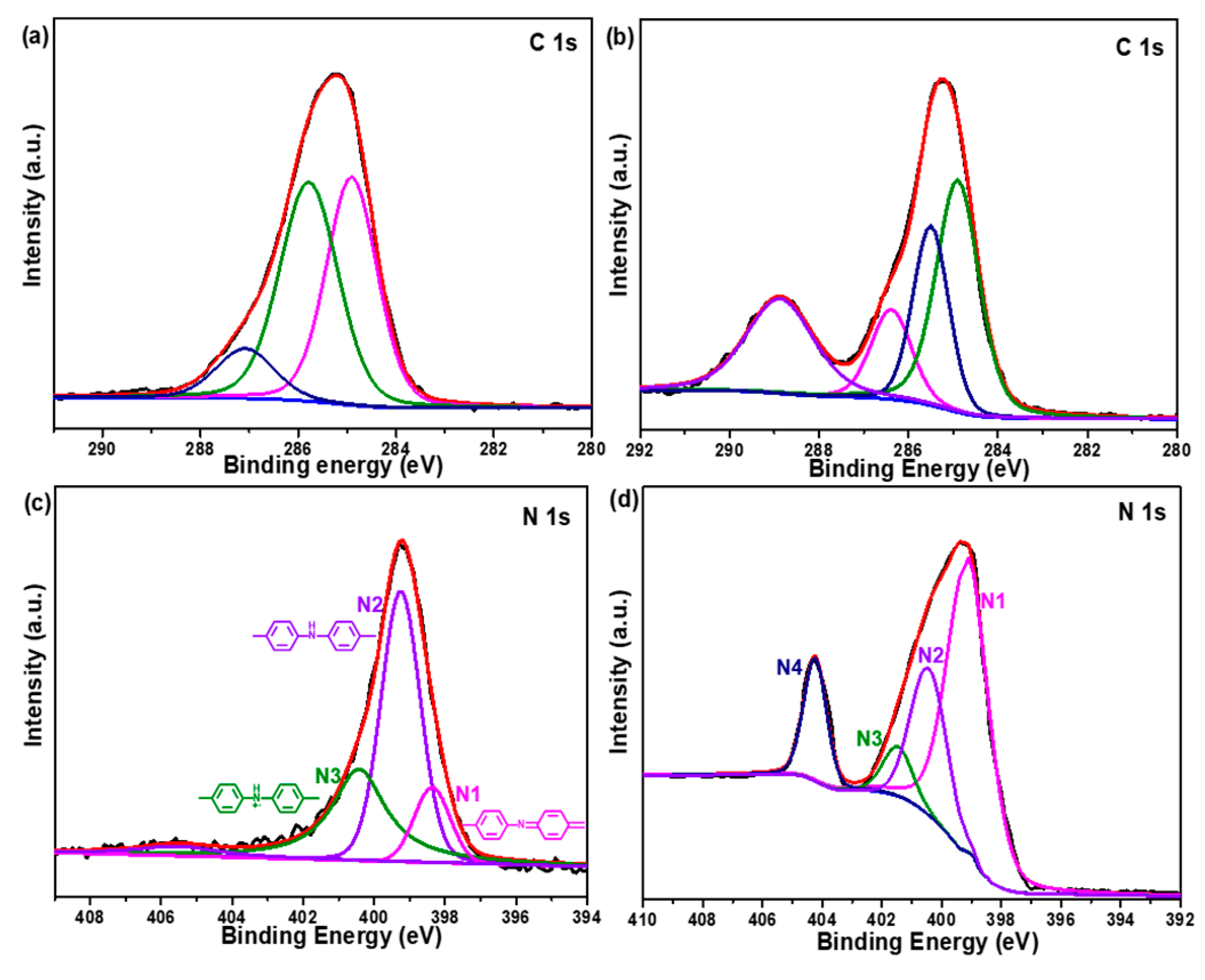
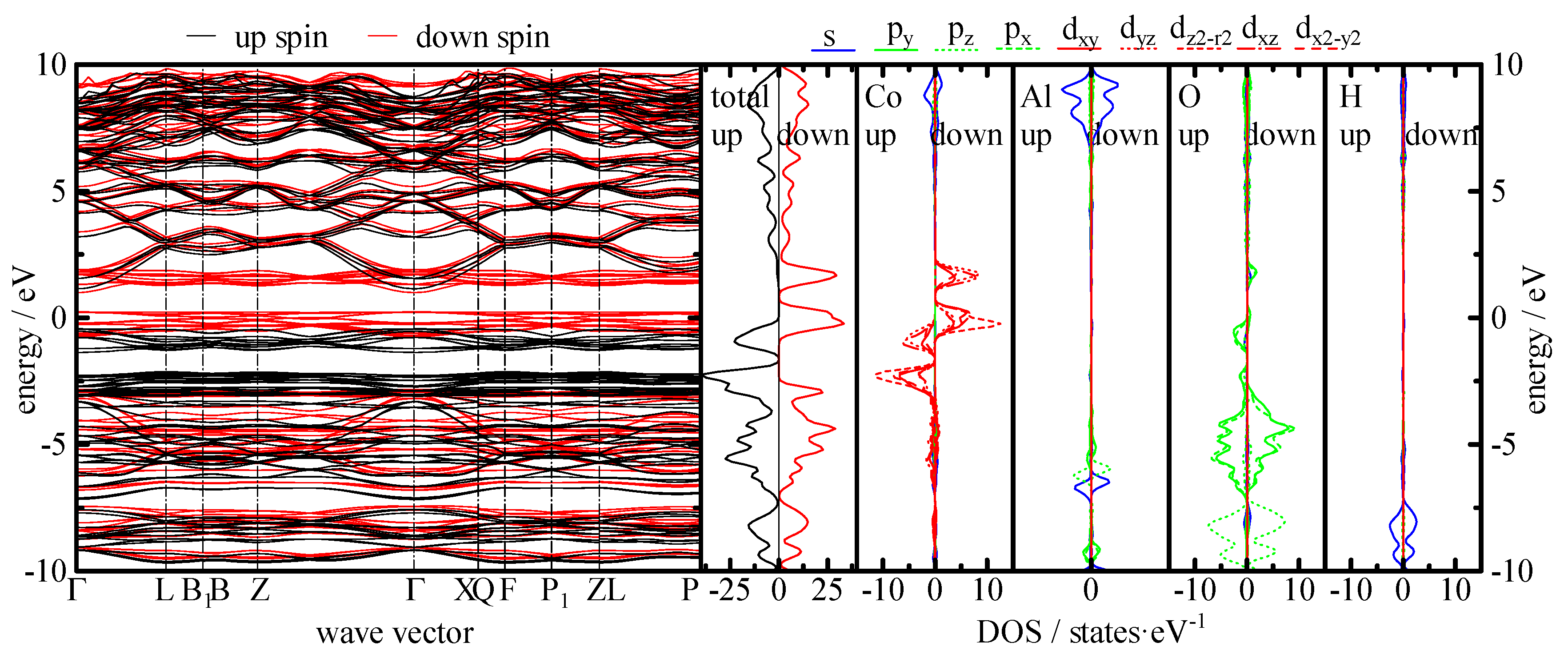
2.1. Structural Study
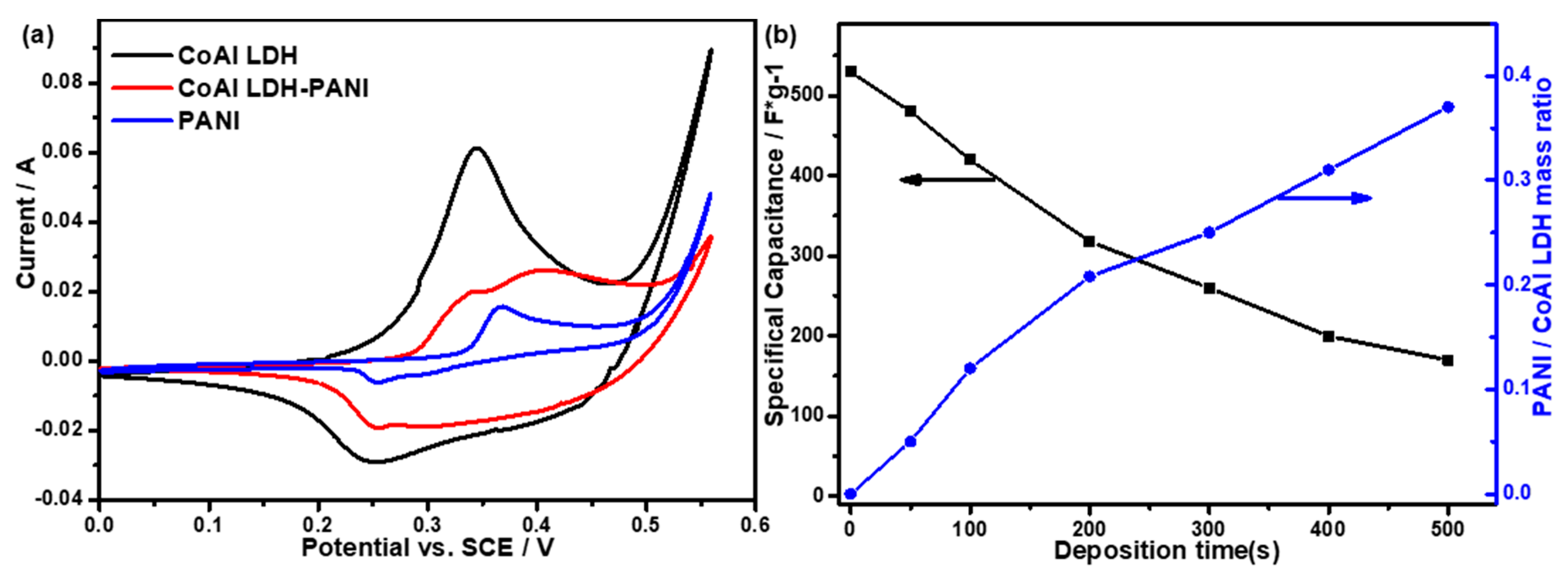
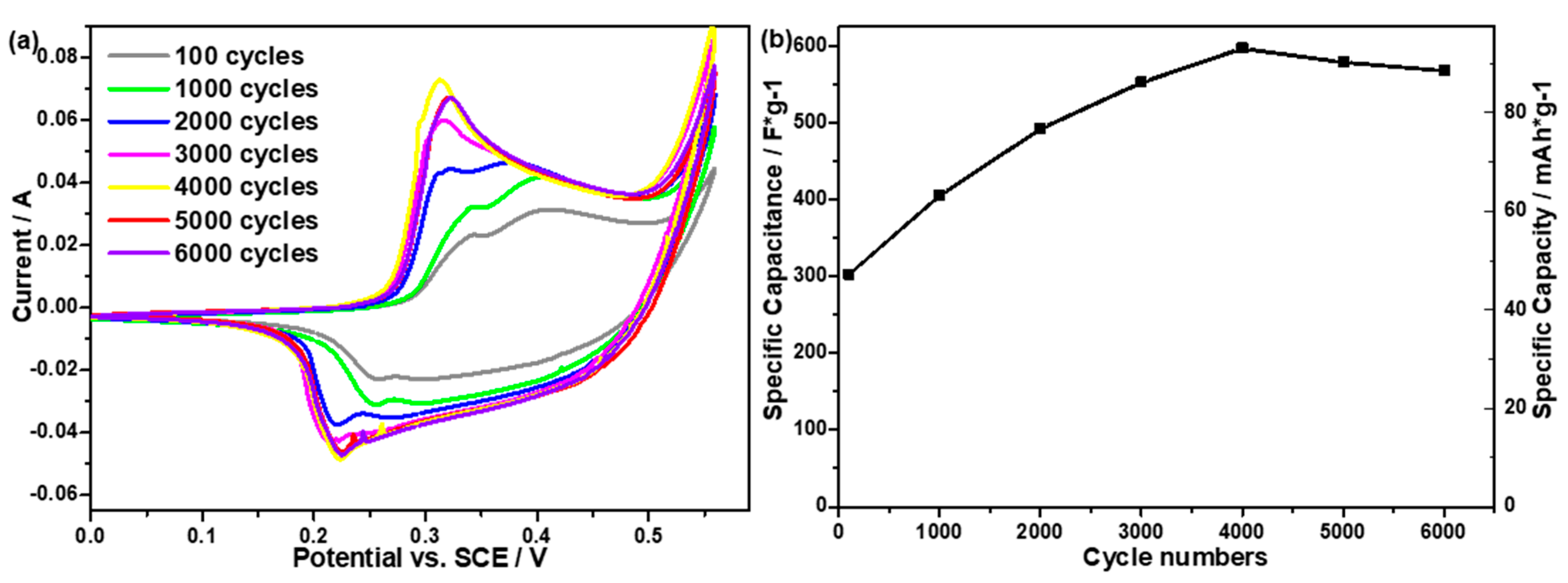
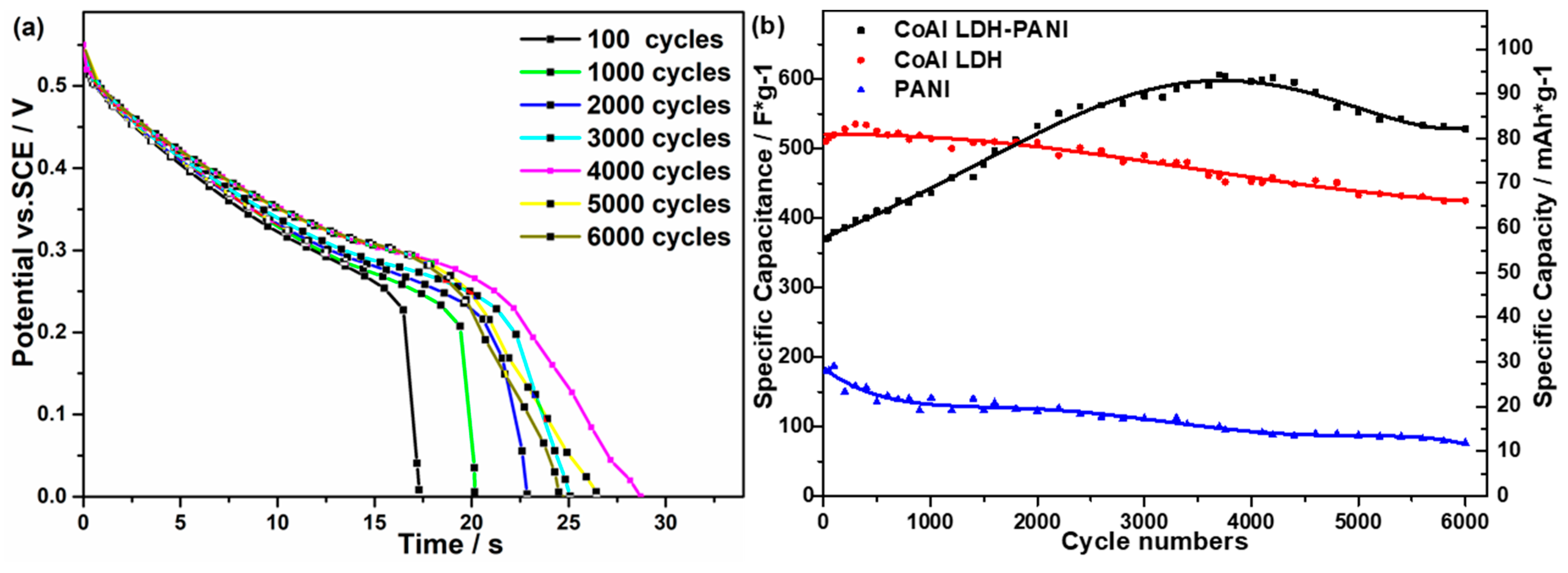
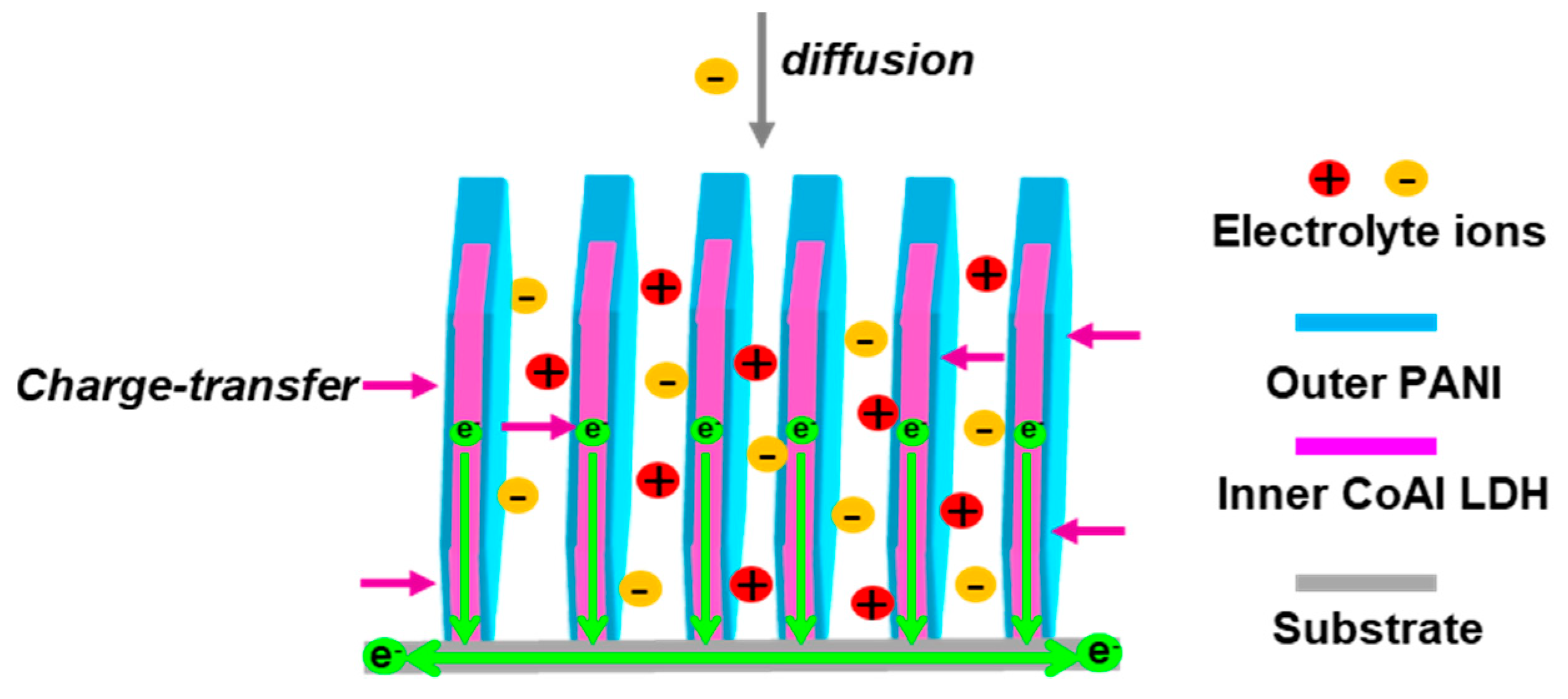
2.2. Electrochemical Performance
3. Materials and Methods
3.1. Synthesis of the CoAl LDH Nanosheet Structures
3.2. Synthesis of the CoAl LDH-PANI Nanocomposites
3.3. Structural Characterization, Theoretical Calculations and Electrochemical Performance Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lu, X.; Wang, G.; Zhai, T.; Yu, M.; Xie, S.; Ling, Y.; Liang, C.; Tong, Y.; Li, Y. Stabilized TiN nanowire arrays for high-performance and flexible supercapacitors. Nano Lett. 2012, 12, 5376–5381. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Peng, X.; Liu, B.; Wu, C.; Xie, Y.; Yu, G. Ultrathin two-dimensional MnO2/graphene hybrid nanostructures for high-performance, flexible planar supercapacitors. Nano Lett. 2013, 13, 2151–2157. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.; Song, M.K.; Park, Y.J.; Kim, J.M.; Liu, M.; Wang, Z.L. Fiber supercapacitors made of nanowire-fiber hybrid structures for wearable/flexible energy storage. Angew, Chem. Int. Ed. 2011, 50, 1683–1687. [Google Scholar] [CrossRef] [PubMed]
- He, Y.B.; Li, G.R.; Wang, Z.L.; Su, C.Y.; Tong, Y.X. Single-crystal ZnO nanorod/amorphous and nanoporous metal oxide shell composites: Controllable electrochemical synthesis and enhanced supercapacitor performances. Energy Environ. Sci. 2011, 4, 1288–1292. [Google Scholar] [CrossRef]
- Li, J.; Yang, M.; Wei, J.; Zhou, Z. Preparation and electrochemical performances of doughnut-like Ni(OH)2-Co(OH)2 composites as pseudocapacitor materials. Nanoscale 2012, 4, 4498–4503. [Google Scholar] [CrossRef] [PubMed]
- Mai, L.Q.; Yang, F.; Zhao, Y.L.; Xu, X.; Xu, L.; Luo, Y.Z. Hierarchical MnMoO4/CoMoO4 heterostructured nanowires with enhanced supercapacitor performance. Nat. Commun. 2011, 2, 381. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Casalongue, H.S.; Liang, Y.; Dai, H. Ni(OH)2 nanoplates grown on graphene as advanced electrochemical pseudocapacitor materials. J. Am. Chem. Soc. 2010, 132, 7472–7477. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Li, Z.J.; Li, R.Y.; Ning, Q.; Kong, H.; Niu, Y.L.; Liu, J.K. Nickel-cobalt double hydroxides microspheres with hollow interior and hedgehog-like exterior structures for supercapacitors. J. Mater. Chem. 2012, 22, 23587–23592. [Google Scholar]
- Pan, G.X.; Xia, X.H.; Luo, J.S.; Cao, F.; Yang, Z.H.; Fan, H.J. Preparation of CoAl layered double hydroxide nanoflake arrays and their high supercapacitance performance. Appl. Clay Sci. 2014, 102, 28–32. [Google Scholar]
- Zhang, Y.; Feng, H.; Wu, X.; Wang, L.; Zhang, A.; Xia, T.; Dong, H.; Li, X.; Zhang, L. Progress of electrochemical capacitor electrode materials: A review. Int. J. Hydrogen Energy 2009, 34, 4889–4899. [Google Scholar] [CrossRef]
- Huang, J.; Lei, T.; Wei, X.; Liu, X.; Liu, T.; Cao, D.; Yin, J.; Wang, G. Effect of Al-Doped β-Ni (OH)2 nanosheets on electrochemical behaviors for high performance supercapacitor application. J. Power Sources 2013, 232, 370–375. [Google Scholar] [CrossRef]
- Abushrenta, N.; Wu, X.; Wang, J.; Liu, J.; Sun, X. Hierarchical Co-based porous layered double hydroxide arrays derived via alkali etching for high-performance supercapacitors. Sci. Rep. 2015, 5, 13082. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.H.; Lim, T.T.; Dong, Z. Application of layered double hydroxides for removal of oxyanions: A review. Water Res. 2008, 42, 1343–1368. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.; Wang, J.; Niu, L.; Sun, J.; Gong, P.; Yang, S. Controllable synthesis of CoAl LDH@Ni(OH)2 nanosheet arrays as binder-free electrode for supercapacitor applications. J. Alloys Compd. 2014, 608, 297–303. [Google Scholar] [CrossRef]
- Yang, J.; Yu, C.; Fan, X.; Ling, Z.; Qiu, J.; Gogotsi, Y. Facile fabrication of MWCNT-doped NiCoAl-layered double hydroxide nanosheets with enhanced electrochemical performances. J. Mater. Chem. A 2013, 1, 1963–1968. [Google Scholar] [CrossRef]
- Wang, Y.G.; Cheng, L.; Xia, Y.Y. Electrochemical profile of nano-particle CoAl double hydroxide/active carbon supercapacitor using KOH electrolyte solution. J. Power Sources 2006, 153, 191–196. [Google Scholar] [CrossRef]
- Malak-Polaczyk, A.; Vix-Guterl, C.; Frackowiak, E. Carbon/layered double hydroxide (LDH) composites for supercapacitor application. Energy Fuels 2010, 24, 3346–3351. [Google Scholar] [CrossRef]
- Chen, Z.; Augustyn, V.; Jia, X.; Xiao, Q.; Dunn, B.; Lu, Y. High-performance sodium-ion pseudocapacitors based on hierarchically porous nanowire composites. ACS Nano 2012, 6, 4319–4327. [Google Scholar] [CrossRef] [PubMed]
- Rakhi, R.; Chen, W.; Cha, D.; Alshareef, H.N. Substrate dependent self-organization of mesoporous cobalt oxide nanowires with remarkable pseudocapacitance. Nano Lett. 2012, 12, 2559–2567. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Duay, J.; Lee, S.B. Redox exchange induced MnO2 nanoparticle enrichment in poly (3, 4-ethylenedioxythiophene) nanowires for electrochemical energy storage. ACS Nano 2010, 4, 4299–4307. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lu, Z.; Shao, M.; Yan, D.; Wei, M.; Evans, D.G.; Duan, X. Flexible hierarchical nanocomposites based on MnO2 nanowires/CoAl hydrotalcite/carbon fibers for high-performance supercapacitors. RSC Adv. 2013, 3, 1045–1049. [Google Scholar] [CrossRef]
- Shao, M.; Ning, F.; Zhao, Y.; Zhao, J.; Wei, M.; Evans, D.G.; Duan, X. Core-shell layered double hydroxide microspheres with tunable interior architecture for supercapacitors. Chem. Mater. 2012, 24, 1192–1197. [Google Scholar] [CrossRef]
- Han, J.; Dou, Y.; Zhao, J.; Wei, M.; Evans, D.G.; Duan, X. Flexible CoAl LDH@PEDOT core/shell nanoplatelet array for high-performance energy storage. Small 2013, 9, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.; Li, Z.; Zhang, R. Hierarchical Conducting Polymer@Clay Core–Shell Arrays for Flexible All-Solid-State Supercapacitor Devices. Small 2005, 11, 3530–3538. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, S.; Khastgir, D.; Singha, N.K.; Lee, J.H. Progress in preparation, processing and applications of polyaniline. Prog. Polym. Sci. 2009, 34, 783–810. [Google Scholar] [CrossRef]
- MacDiarmid, A.G. “Synthetic metals”: A novel role for organic polymers (Nobel lecture). Angew. Chem. Int. Ed. 2001, 40, 2581–2590. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, L.L.; Zhao, X.S. Graphene/polyaniline nanofiber composites as supercapacitor electrodes. Chem. Mater. 2010, 22, 1392–1401. [Google Scholar] [CrossRef]
- Snook, G.A.; Kao, P.; Best, A.S. Conducting-polymer-based supercapacitor devices and electrodes. J. Power Sources 2011, 196, 1–12. [Google Scholar] [CrossRef]
- Kutlu, B.; Leuteritz, A.; Boldt, R.; Jehnichen, D.; Wagenknecht, U.; Heinrich, G. PANI-LDH prepared by polymerization-adsorption method and processing to conductive compounds. Appl. Clay Sci. 2013, 72, 91–95. [Google Scholar] [CrossRef]
- Gao, H.; Jiang, T.; Han, B.; Wang, Y.; Du, J.; Liu, Z.; Zhang, J. Aqueous/ionic liquid interfacial polymerization for preparing polyaniline nanoparticles. Polymer 2004, 45, 3017–3019. [Google Scholar] [CrossRef]
- Trchová, M.; Stejskal, J. Polyaniline: The infrared spectroscopy of conducting polymer nanotubes (IUPAC Technical Report). Pure Appl. Chem. 2011, 83, 1803–1817. [Google Scholar] [CrossRef]
- Leng, W.; Zhou, S.; Gu, G.; Wu, L. Wettability switching of SDS-doped polyaniline from hydrophobic to hydrophilic induced by alkaline/reduction reactions. J. Colloid Interface Sci. 2012, 369, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Salaün, F.; Campagne, C. The influence of 1-butanol and trisodium citrate ion on morphology and chemical properties of chitosan-based microcapsules during rigidification by alkali treatment. Mar. Drugs 2014, 12, 5801–5816. [Google Scholar] [CrossRef] [PubMed]
- Kloprogge, J.T.; Hickey, L.; Frost, R.L. FT-Raman and FT-IR spectroscopic study of synthetic Mg/Zn/Al-hydrotalcites. J. Raman Spectrosc. 2004, 35, 967–974. [Google Scholar] [CrossRef]
- Han, J.B.; Lu, J.; Wei, M.; Wang, Z.L.; Duan, X. Heterogeneous ultrathin films fabricated by alternate assembly of exfoliated layered double hydroxides and polyanion. Chem. Commun. 2008, 41, 5188–5190. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Angelopoulos, M.; Epstein, A.J. Concentration dependence of aggregation of polyaniline in NMP solution and properties of resulting cast films. Macromolecules 1997, 30, 7634–7637. [Google Scholar] [CrossRef]
- Sinha, S.; Bhadra, S.; Khastgir, D. Effect of dopant type on the properties of polyaniline. J. Appl. Polym. Sci. 2009, 112, 3135–3140. [Google Scholar] [CrossRef]
- Fan, G.; Wang, H.; Xiang, X.; Li, F. Co-Al mixed metal oxides/carbon nanotubes nanocomposite prepared via a precursor route and enhanced catalytic property. J. Solid State Chem. 2013, 197, 14–22. [Google Scholar] [CrossRef]
- Nguyen, V.H.; Shim, J.J. Green synthesis and characterization of carbon nanotubes/polyaniline nanocomposites. J. Spectrosc. 2015, 2015. [Google Scholar] [CrossRef]
- Augustyn, V.; Simon, P.; Dunn, B. Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ. Sci. 2014, 4, 1597–1614. [Google Scholar] [CrossRef]
- Cooper, S.J.; Bertei, A.; Finegan, D.P. Simulated impedance of diffusion in porous media. Electrochim. Acta 2017, 251, 681–689. [Google Scholar] [CrossRef]
- Chamaani, A.; Safa, M.; Chawla, N. Stabilizing effect of ion complex formation in lithium–oxygen battery electrolytes. J. Electroanal. Chem. 2018, 815, 143–150. [Google Scholar] [CrossRef]
- Lu, X.; Zheng, D.; Zhai, T. Facile synthesis of large-area manganese oxide nanorod arrays as a high-performance electrochemical supercapacitor. Energy Environ. Sci. 2011, 4, 2915–2921. [Google Scholar] [CrossRef]
- Yuan, L.; Yao, B.; Hu, B. Polypyrrole-coated paper for flexible solid-state energy storage. Energy Environ. Sci. 2013, 6, 470–476. [Google Scholar] [CrossRef]
- Wu, J.; Wang, J.; Huang, X. A self-assembly route to porous polyaniline/reduced graphene oxide composite materials with molecular-level uniformity for high-performance supercapacitors. Energy Environ. Sci. 2018, 11, 1280–1286. [Google Scholar] [CrossRef]
- Wang, J.; Wu, J.; Bai, H. Degradation-induced capacitance: A new insight into the superior capacitive performance of polyaniline/graphene composites. Energy Environ. Sci. 2017, 10, 2372–2382. [Google Scholar]
- Planes, G.A.; Rodriguez, J.L.; Miras, M.C.; Garcia, G.; Pastor, E.; Barbero, C.A. Spectroscopic evidence for intermediate species formed during aniline polymerization and polyaniline degradation. Phys. Chem. Chem. Phys. 2010, 12, 10584–10593. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Wang, T.; Guo, J. Electrochemical capacity fading of polyaniline electrode in supercapacitor: An XPS analysis. Prog. Nat. Sci. Mater. Int. 2017, 27, 257–260. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, S.; Wu, X. One-Step Electrodeposition of MnO2@NiAl Layered Double Hydroxide Nanostructures on the Nickel Foam for High-Performance Supercapacitors. J. Electrochem. Soc. 2017, 164, 56–62. [Google Scholar] [CrossRef]
- Nakagaki, R.; Frost, D.C.; McDowell, C.A. X-ray photoelectron spectroscopy of nitroanilines and their derivatives. J. Electron Spectrosc. Relat. Phenom. 1981, 22, 289–296. [Google Scholar] [CrossRef]
- Nakagaki, R.; Frost, D.C.; McDowell, C.A. The intramolecular charge-transfer interaction in X-ray photoelectron spectroscopy: The charge-transfer satellites observed in p-nitroaniline and related compounds. J. Electron Spectrosc. Relat. Phenom. 1980, 19, 355–370. [Google Scholar] [CrossRef]
- Nakayama, M.; Saeki, S.; Ogura, K. In situ observation of electrochemical formation and degradation processes of polyaniline by fourier-transform infrared spectroscopy. Anal. Sci. 1999, 15, 259–263. [Google Scholar] [CrossRef]
- Yang, X.; Xie, Q.; Yao, S. A comparative study on polyaniline degradation by an electrochemical quartz crystal impedance system: Electrode and solution effects. Synth. Met. 2004, 143, 119–128. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmuller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B. 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented- wave method. Phys. Rev. 1999, 59, 1758–1775. [Google Scholar] [CrossRef]
- Adachi, H.; Tsukuda, M.; Satoko, C. Discrete variational Xα cluster calculations. I. Application to metal clusters. J. Phys. Soc. Jpn. 1978, 45, 875–883. [Google Scholar] [CrossRef]
- Muralidharan, N.; Westover, A.S.; Sun, H. From the Junkyard to the Power Grid: Ambient Processing of Scrap Metals into Nanostructured Electrodes for Ultrafast Rechargeable Batteries. ACS Energy Lett. 2016, 1, 1034–1041. [Google Scholar] [CrossRef]
- Mai, L.Q.; Minhas-Khan, A.; Tian, X.; Hercule, K.M.; Zhao, Y.L.; Lin, X.; Xu, X. Synergistic interaction between redox-active electrolyte and binder-free functionalized carbon for ultrahigh supercapacitor performance. Nat. Commun. 2013, 4, 2923. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, G.; Takei, T.; Yanagida, S.; Kumada, N. Enhanced Supercapacitor Performance Based on CoAl Layered Double Hydroxide-Polyaniline Hybrid Electrodes Manufactured Using Hydrothermal-Electrodeposition Technology. Molecules 2019, 24, 976. https://doi.org/10.3390/molecules24050976
Yang G, Takei T, Yanagida S, Kumada N. Enhanced Supercapacitor Performance Based on CoAl Layered Double Hydroxide-Polyaniline Hybrid Electrodes Manufactured Using Hydrothermal-Electrodeposition Technology. Molecules. 2019; 24(5):976. https://doi.org/10.3390/molecules24050976
Chicago/Turabian StyleYang, Guoshen, Takahiro Takei, Sayaka Yanagida, and Nobuhiro Kumada. 2019. "Enhanced Supercapacitor Performance Based on CoAl Layered Double Hydroxide-Polyaniline Hybrid Electrodes Manufactured Using Hydrothermal-Electrodeposition Technology" Molecules 24, no. 5: 976. https://doi.org/10.3390/molecules24050976
APA StyleYang, G., Takei, T., Yanagida, S., & Kumada, N. (2019). Enhanced Supercapacitor Performance Based on CoAl Layered Double Hydroxide-Polyaniline Hybrid Electrodes Manufactured Using Hydrothermal-Electrodeposition Technology. Molecules, 24(5), 976. https://doi.org/10.3390/molecules24050976