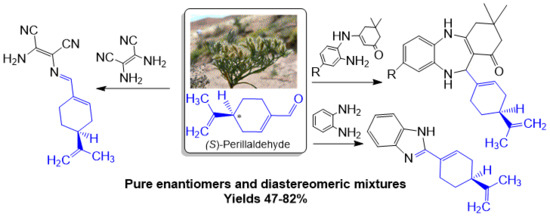
Hemi-Synthesis of Chiral Imine, Benzimidazole and Benzodiazepines from Essential Oil of Ammodaucus leucotrichus subsp. leucotrichus
Abstract
1. Introduction
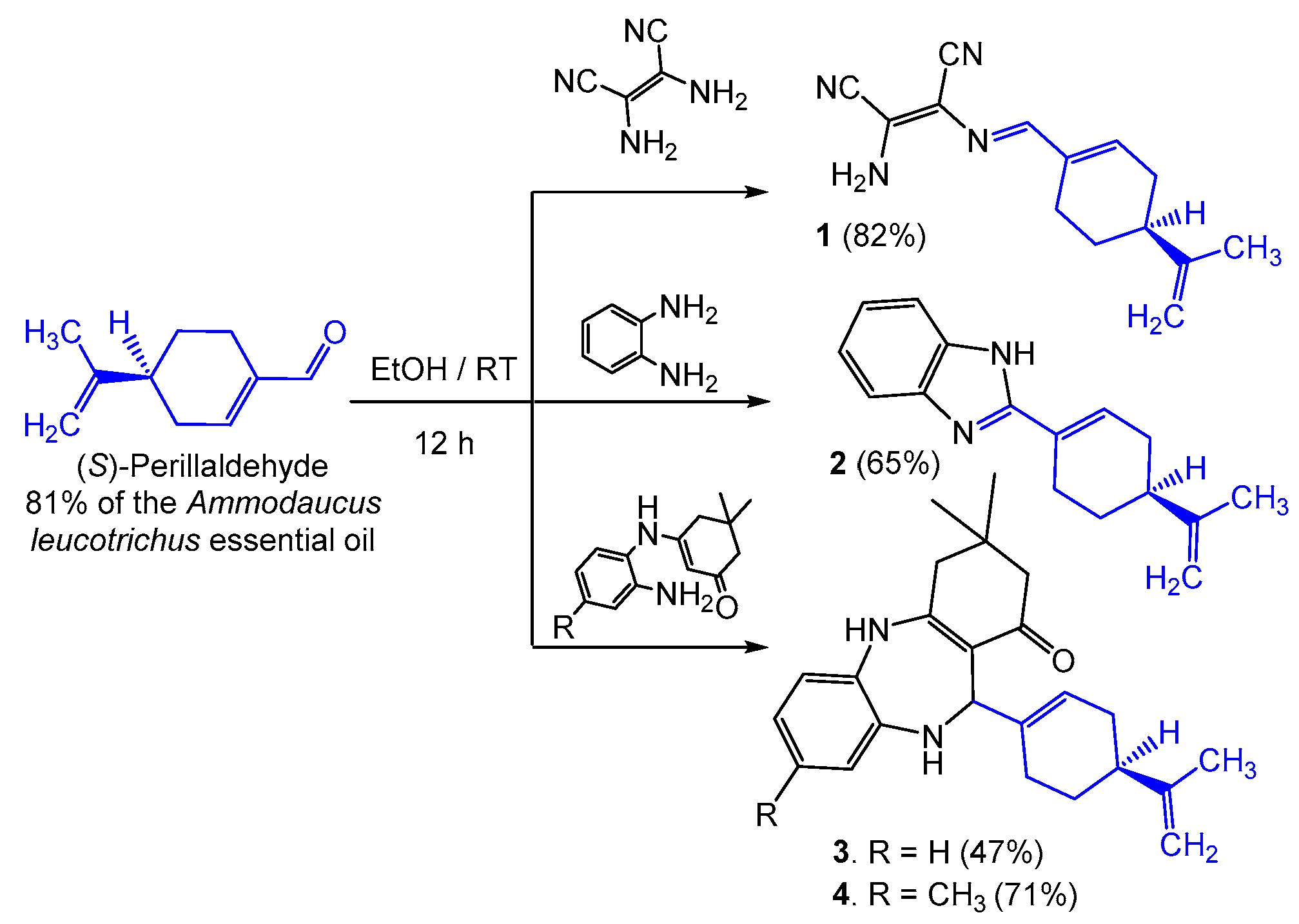
2. Results and Discussion
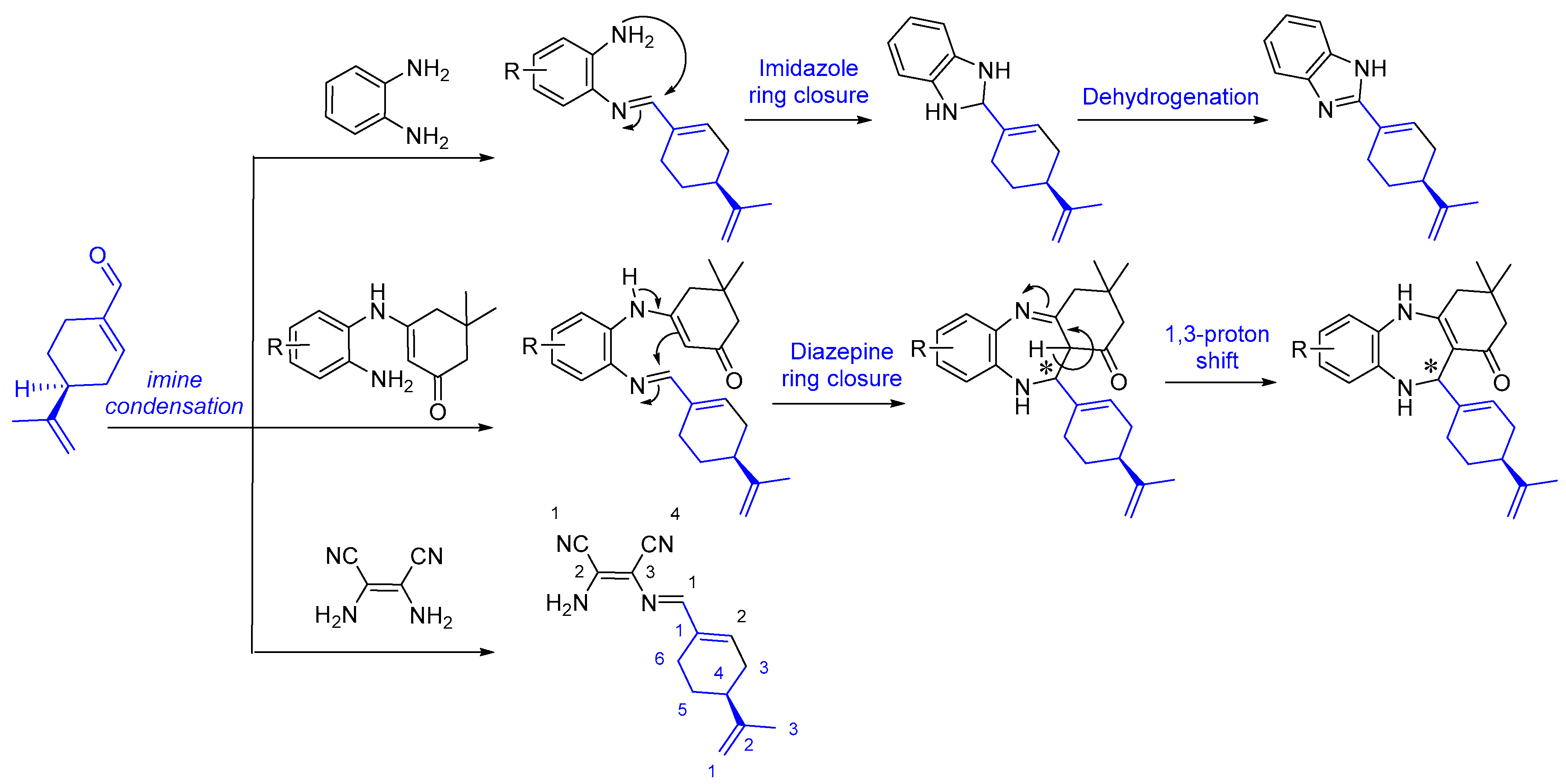
2.1. Chemistry and Mechanism
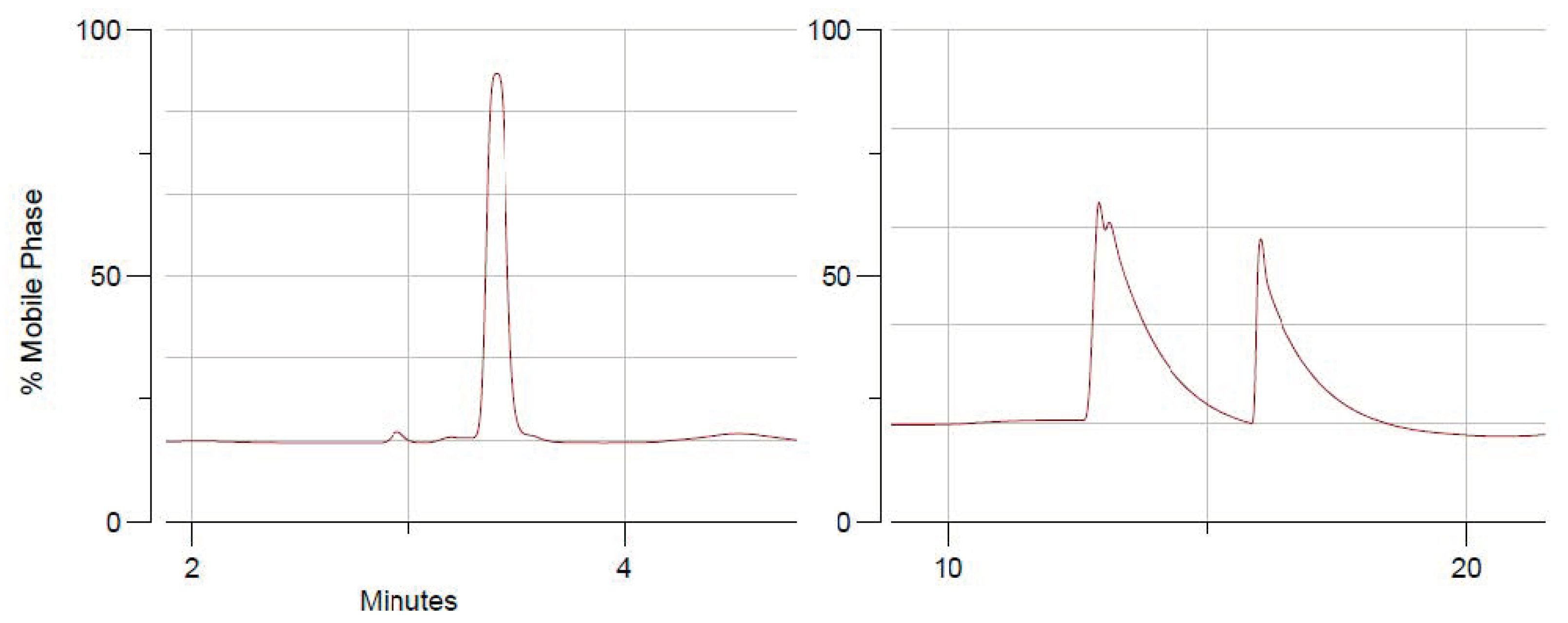
2.2. Chiral HPLC and Stereochemistry
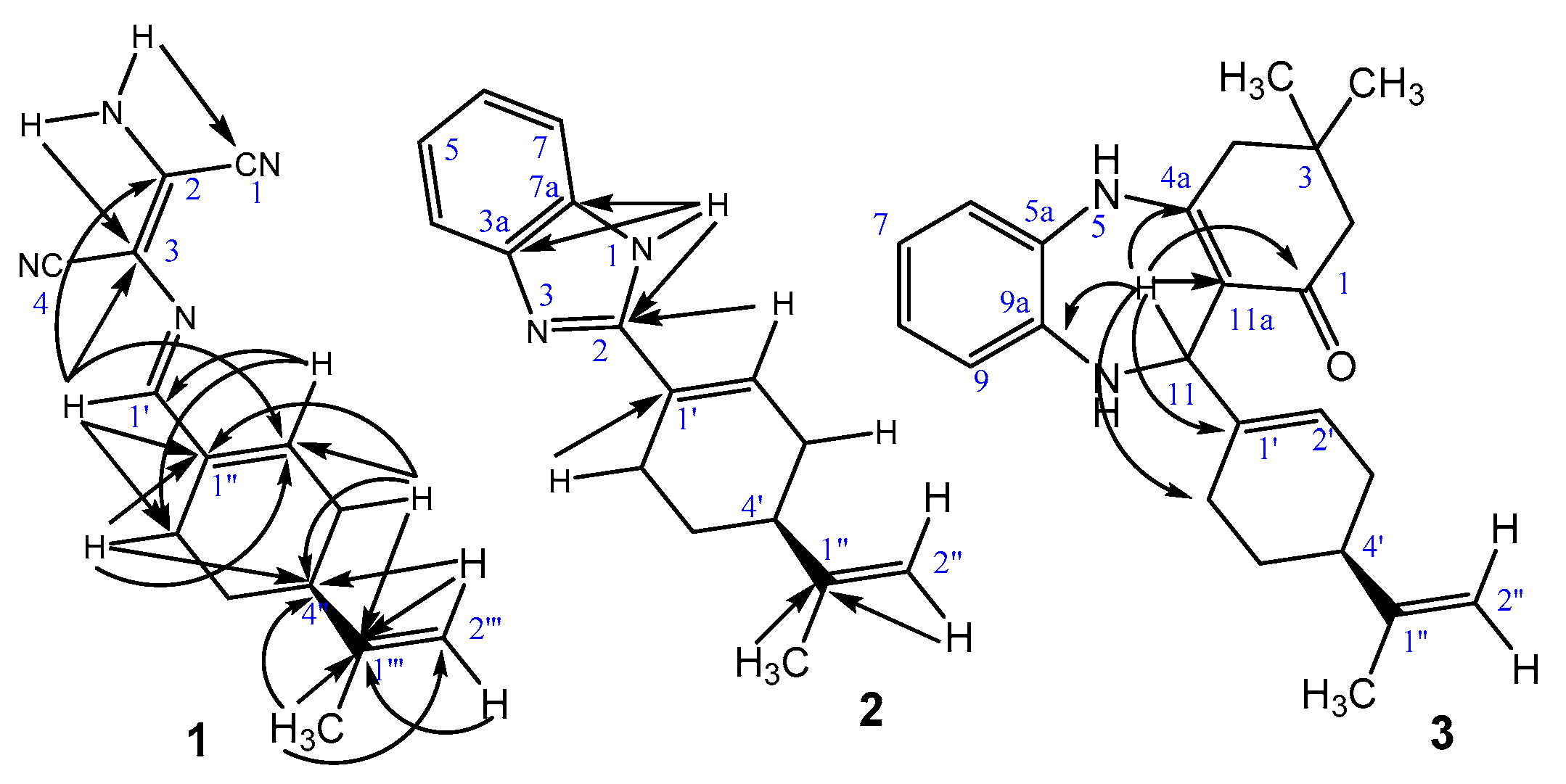
2.3. Nuclear Magnetic Resonance Spectroscopy
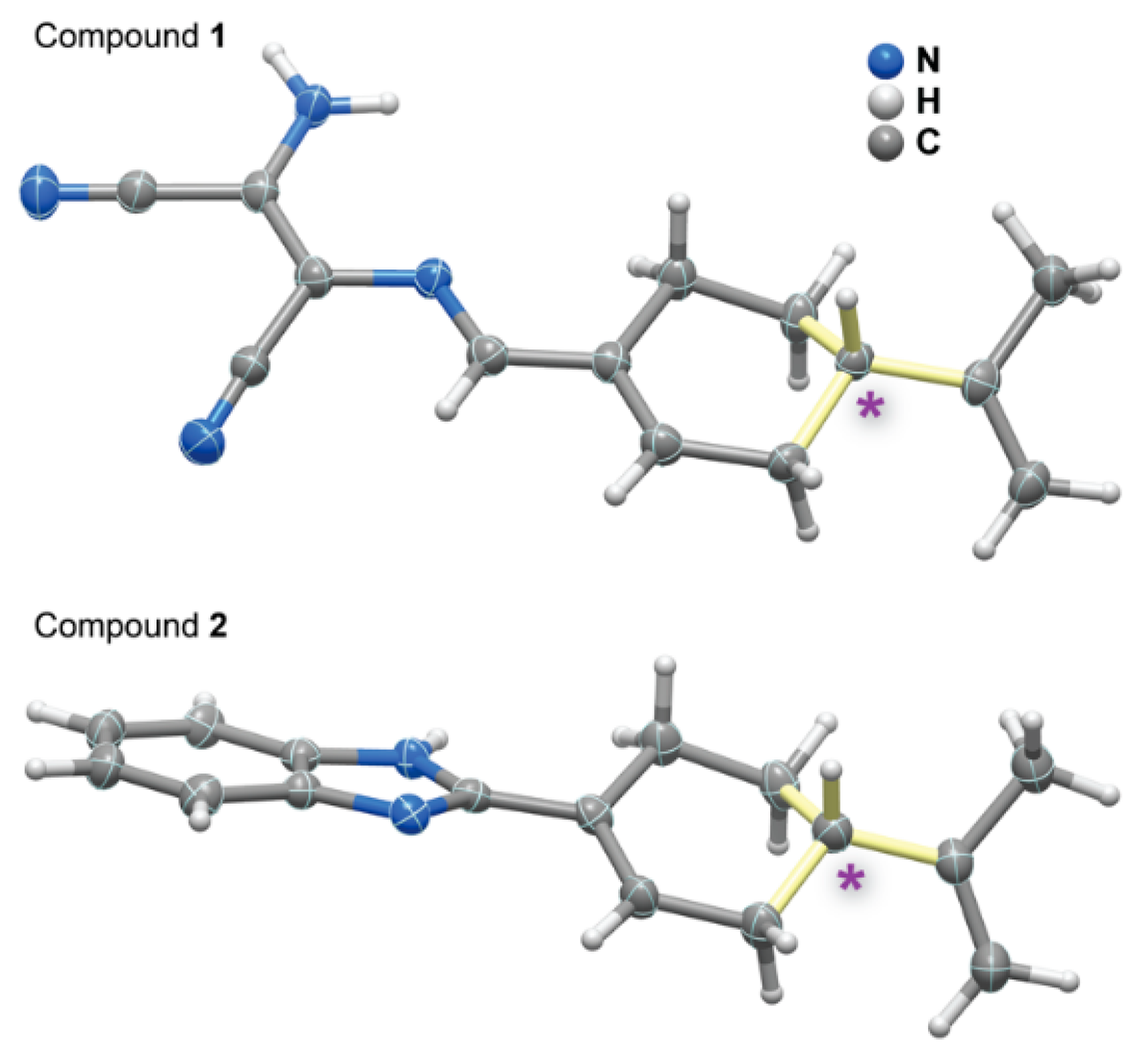
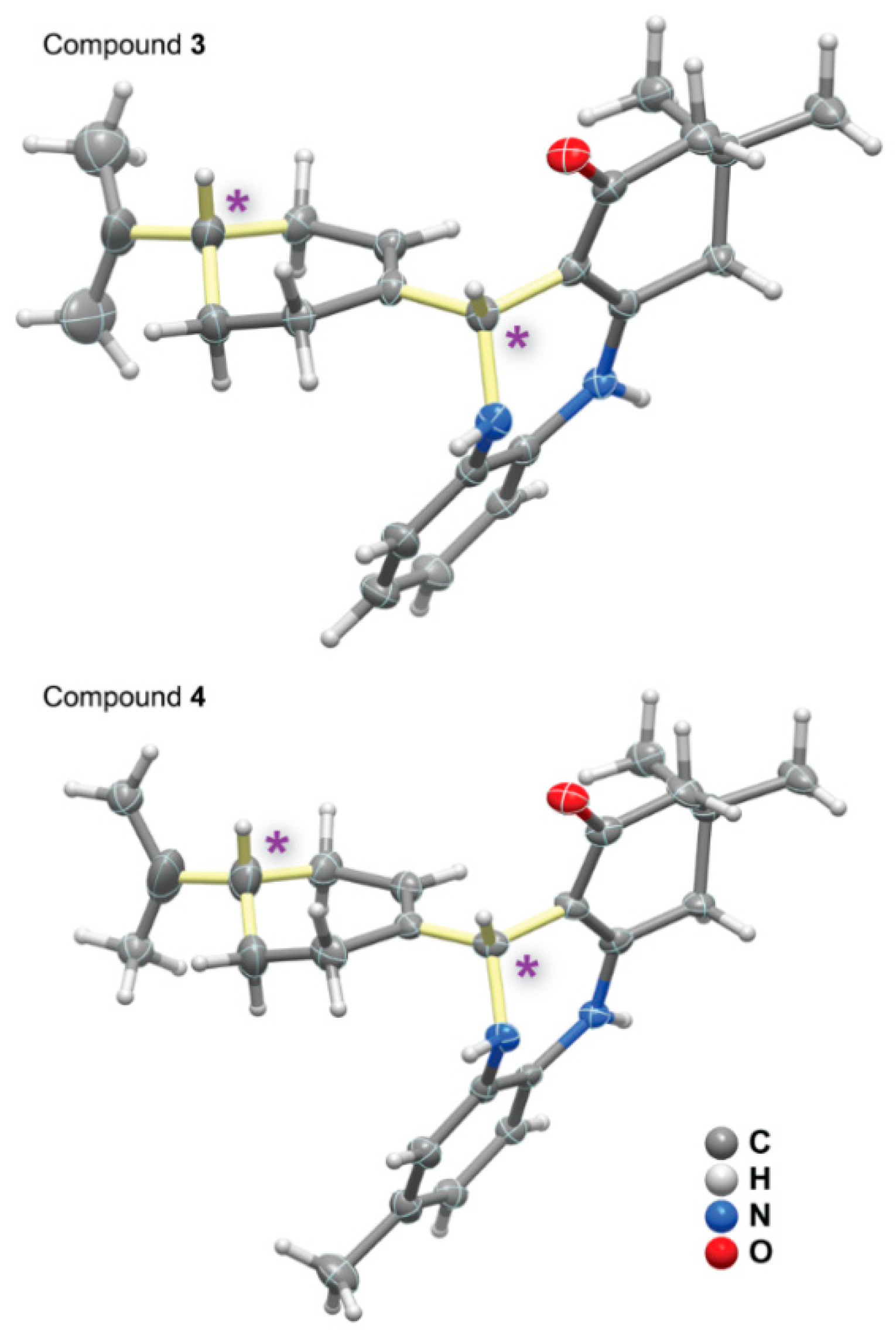
2.4. Single-Crystal X-ray Diffraction
3. Materials and Methods
3.1. General Remarks
3.2. Plant Material and Extraction Procedure
3.3. General Procedure for the Hemi-Synthesis of Compounds 1–4
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Appendino, G.; Minassi, A.; Taglialatela-Scafati, O. Recreational drug discovery: natural products as lead structures for the synthesis of smart drugs. Nat. Prod. Rep. 2014, 31, 880–904. [Google Scholar] [CrossRef] [PubMed]
- Bonikowski, R.; Kula, J.; Bujacz, A.; Wajs-Bonikowska, A.; Zaklos-Szyda, M.; Wysocki, S. Hydroindene-derived chiral synthons from carotol and their cytotoxicity. Tetrahedron Asymm. 2012, 23, 1038–1045. [Google Scholar] [CrossRef]
- Huangyong, L.; Changshui, C.; Cao, X. Essential oils-oriented chiral esters as potential pesticides: Asymmetric syntheses, characterization and bio-evaluation. Ind. Crops Prod. 2015, 76, 432–436. [Google Scholar]
- Sakirigui, A.; Gbaguidi, F.; Kasséhin, U.; JacquesPoupaert; Accrombessi, G.; Kotchoni, S. Structural and antitrypanosomal data of different carbasones of piperitone. Data Brief 2016, 9, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Velasco-Negueruela, A.; Pérez-Alonso, M.J.; Pérez de Paz, P.L.; Palá-Paúl, J.; Sanz, J. Analysis by gas chromatography–mass spectrometry of the volatiles from the fruits of Ammodaucus leucotrichus subsp. leucotrichus and subsp. nanocarpus grown in North Africa and the Canary Islands, respectively. J. Chromatogr. A 2006, 1108, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Anthelme, F.; Waziri Mato, M.; Maley, J. Elevation and local refuges ensure persistence of mountain specific vegetation in the Nigerien Sahara. J. Arid Environ. 2008, 72, 2232–2242. [Google Scholar] [CrossRef]
- Jouad, H.; Haloui, M.; Rhiouani, H.; El Hilaly, J.; Eddouks, M. Ethnobotanical survey of medicinal plants used for the treatment of diabetes, cardiac and renal diseases in the North centre region of Morocco (Fez–Boulemane). J. Ethnopharmacol. 2001, 77, 175–182. [Google Scholar] [CrossRef]
- Merzouki, A.; Ed-derfoufi, F.; Molero Mesa, J. Contribution to the knowledge of Rifian traditional medicine. II: Folk medicine in Ksar Lakbir district (NW Morocco). Fitoterapia 2000, 71, 278–307. [Google Scholar] [CrossRef]
- Hammiche, V.; Maiza, K. Traditional medicine in Central Sahara: Pharmacopoeia of Tassili N’ajjer. J. Ethnopharmacol. 2006, 105, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Dahmane, D.; Dob, T.; Krimat, S.; Nouasri, A.; Metidji, H.; Ksouri, A. Chemical composition, antioxidant and antibacterial activities of the essential oils of medicinal plant Ammodaucus leucotrichus from Algeria. J. Essent. Oil Res. 2017, 29, 48–55. [Google Scholar] [CrossRef]
- Khaldi, A.; Meddah, B.; Moussaoui, A.; Sonnet, P. Anti-mycotoxin Effect and Antifungal Properties of Essential Oil from Ammodaucus leucotrichus Coss. & Dur. on Aspergillus flavus and Aspergillus ochraceus. J. Essent. Oil Bear. 2017, 20, 36–44. [Google Scholar]
- Abu Zarga, M.H.; Al-Jaber, H.I.; Baba Amer, Z.Y.; Sakhrib, L.; Al-Qudah, M.A.; Al-humaidi, J.Y.G.; Abaza, I.F.; Afifi, F.U. Chemical Composition, Antimicrobial and Antitumor Activities of Essential Oil of Ammodaucus leucotrichus Growing in Algeria. JAPN 2013, 3, 224–231. [Google Scholar]
- Mayeku, W.P.; Omollo, N.I.; Odalo, O.J.; Hassanali, A. Chemical composition and mosquito repellency of essential oil of Conyza newii propagated in different geographical locations of Kenya. Med. Vet. Entomol. 2014, 28, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Wang, S.Y.; Chen, C. Increasing Antioxidant Activity and Reducing Decay of Blueberries by Essential Oils. J. Agric. Food Chem. 2008, 56, 3587–3592. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Zeng, X.; Lü, A.; Zhu, A.; Peng, X.; Wang, Y. Perillaldehyde, a potential preservative agent in foods: Assessment of antifungal activity against microbial spoilage of cherry tomatoes. Food Sci. Technol. 2015, 60, 63–70. [Google Scholar] [CrossRef]
- Appelt, H.R.; Oliveira, J.S.; Santos, R.C.V.; Rodrigues, O.E.D.; Santos, M.Z.; Heck, E.F.; Rosa, L.C.R. Synthesis and Antimicrobial Activity of Carbohydrate Based Schiff Bases: Importance of Sugar Moiety. Int. J. Carbohydr. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Kundu, A.; Shakil, N.A.; Saxena, D.B.; Pankaj; Kumar, J.; Walia, S. Microwave assisted solvent-free synthesis and biological activities of novel imines (Schiff bases). J. Environ. Sci. Health 2009, 44, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Sonnekar, V.S.; Jadhav, W.N.; Dake, D.S.; Pawar, R. Synthesis and antimicrobial and antifungal activities of novel bis-imine derivatives. Res. J. Pharm. Biol. Chem. Sci. 2013, 4, 1411–1418. [Google Scholar]
- Missaoui, B.E.; Ouahrani, M.R.; Kouadri, Y.; Chebrouk, F.; Gherraf, N. Synthesis of Novel Heterocyclic Compounds Containing 1,5-Benzodiazepine. Asian J. Chem. 2015, 27, 2175–2177. [Google Scholar] [CrossRef]
- Kolos, N.N.; Yurchenko, E.N.; Orlov, V.D.; Shishkina, S.V.; Shishkin, O.V. Investigation of the products of interaction of cyclic diketones with nitrogen-containing 1,4-binucleophiles. Chem. Heterocycl. Compd. 2004, 40, 1550–1559. [Google Scholar] [CrossRef]
- Gupta, S.; Gupta, P.; Sachar, A.; Sharma, R.L. Facile and one pot synthetic routes for various novel, differently fused and promising heteropolycycles. J. Heterocycl. Chem. 2010, 47, 334–349. [Google Scholar]
- Cherfaoui, B.; Lakhdari, H.; Bennamane, N.; Ameraoui, R.; Talhi, O.; Almeida Paz, F.A.; Bachari, K.; Kirsch, G.; Nejar-Bellara, K.; Silva, A.M.S. Dibenzo[b,e][1,4]diazepin-1-ones and their Ring-Opened Derivatives: Revisited Synthesis, 2D NMR and Crystal Structure. Synlett 2017, 28, 2247–2252. [Google Scholar]
- Ling, I.; Podanyi, B.; Hamori, T.; Solyom, S. Asymmetric reduction of a carbon-nitrogen double bond: enantioselective synthesis of 4,5-dihydro-3H-2,3-benzodiazepines. J. Chem. Soc. Perkin Trans. 1 1995, 11, 1423–1427. [Google Scholar] [CrossRef]
- Al-Azmi, A.; Elassara, A.A.; Booth, B.L. The chemistry of diaminomaleonitrile and its utility in heterocyclic synthesis. Tetrahedron 2003, 59, 2749–2763. [Google Scholar] [CrossRef]
- Grell, J.; Bernstein, J.; Tinhofer, G. Graph-set analysis of hydrogen-bond patterns: some mathematical conceptsWork supported by grant No. I-0333-263.06/93 from the GIF, the German-Israeli Foundation for Scientific Research and Development. Acta Crystallogr. Sect. B Struct. Sci. 1999, 55, 1030–1043. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–4 are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chebrouk, F.; Madani, K.; Cherfaoui, B.; Boukenna, L.; Válega, M.; Mendes, R.F.; Paz, F.A.A.; Bachari, K.; Talhi, O.; Silva, A.M.S. Hemi-Synthesis of Chiral Imine, Benzimidazole and Benzodiazepines from Essential Oil of Ammodaucus leucotrichus subsp. leucotrichus. Molecules 2019, 24, 975. https://doi.org/10.3390/molecules24050975
Chebrouk F, Madani K, Cherfaoui B, Boukenna L, Válega M, Mendes RF, Paz FAA, Bachari K, Talhi O, Silva AMS. Hemi-Synthesis of Chiral Imine, Benzimidazole and Benzodiazepines from Essential Oil of Ammodaucus leucotrichus subsp. leucotrichus. Molecules. 2019; 24(5):975. https://doi.org/10.3390/molecules24050975
Chicago/Turabian StyleChebrouk, Farid, Khodir Madani, Brahim Cherfaoui, Leila Boukenna, Mónica Válega, Ricardo F. Mendes, Filipe A. A. Paz, Khaldoun Bachari, Oualid Talhi, and Artur M. S. Silva. 2019. "Hemi-Synthesis of Chiral Imine, Benzimidazole and Benzodiazepines from Essential Oil of Ammodaucus leucotrichus subsp. leucotrichus" Molecules 24, no. 5: 975. https://doi.org/10.3390/molecules24050975
APA StyleChebrouk, F., Madani, K., Cherfaoui, B., Boukenna, L., Válega, M., Mendes, R. F., Paz, F. A. A., Bachari, K., Talhi, O., & Silva, A. M. S. (2019). Hemi-Synthesis of Chiral Imine, Benzimidazole and Benzodiazepines from Essential Oil of Ammodaucus leucotrichus subsp. leucotrichus. Molecules, 24(5), 975. https://doi.org/10.3390/molecules24050975