Differentiation of Fresh and Processed Fruit Juices Using Volatile Composition
Abstract
:1. Introduction
2. Results and Discussion
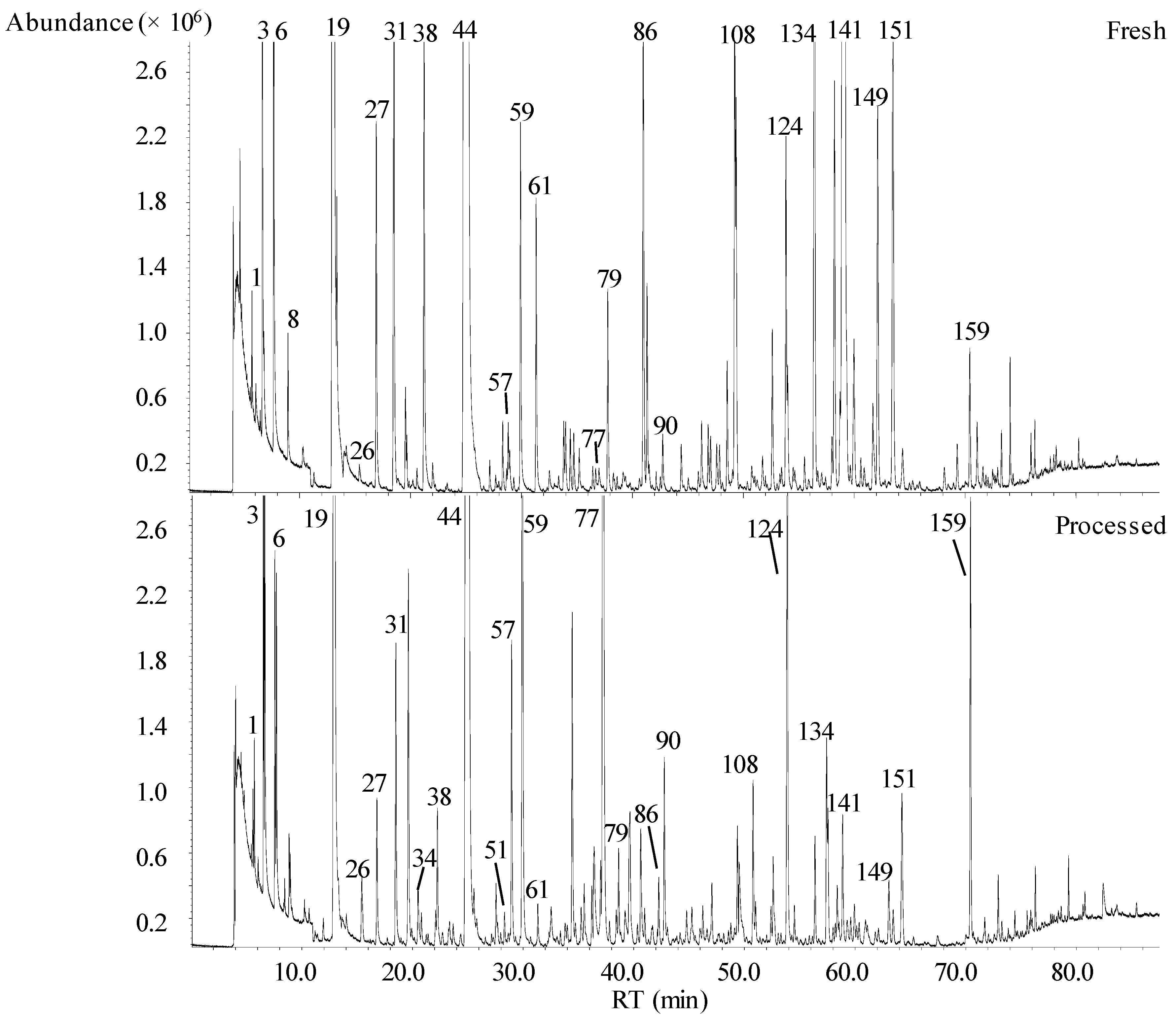
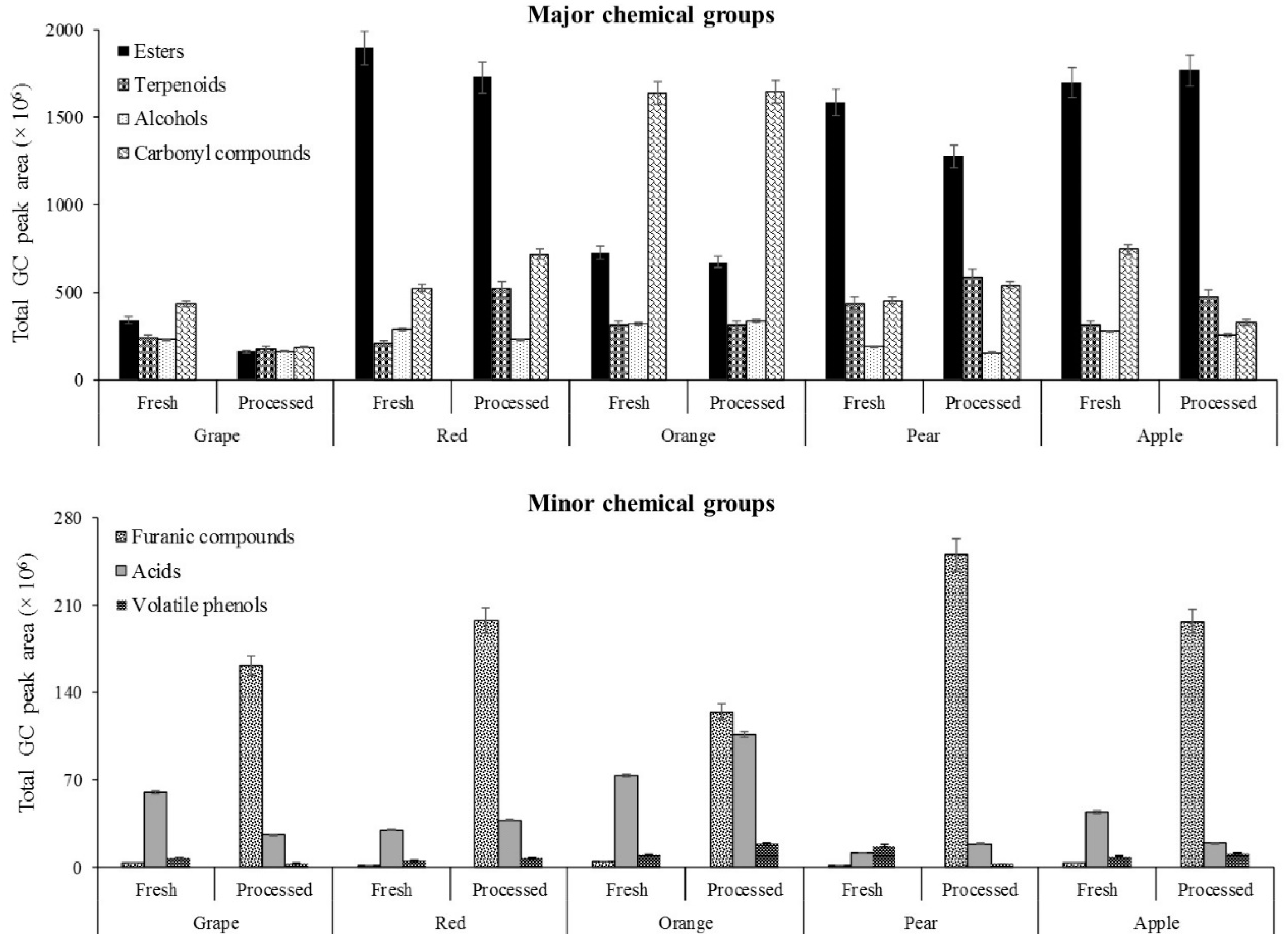
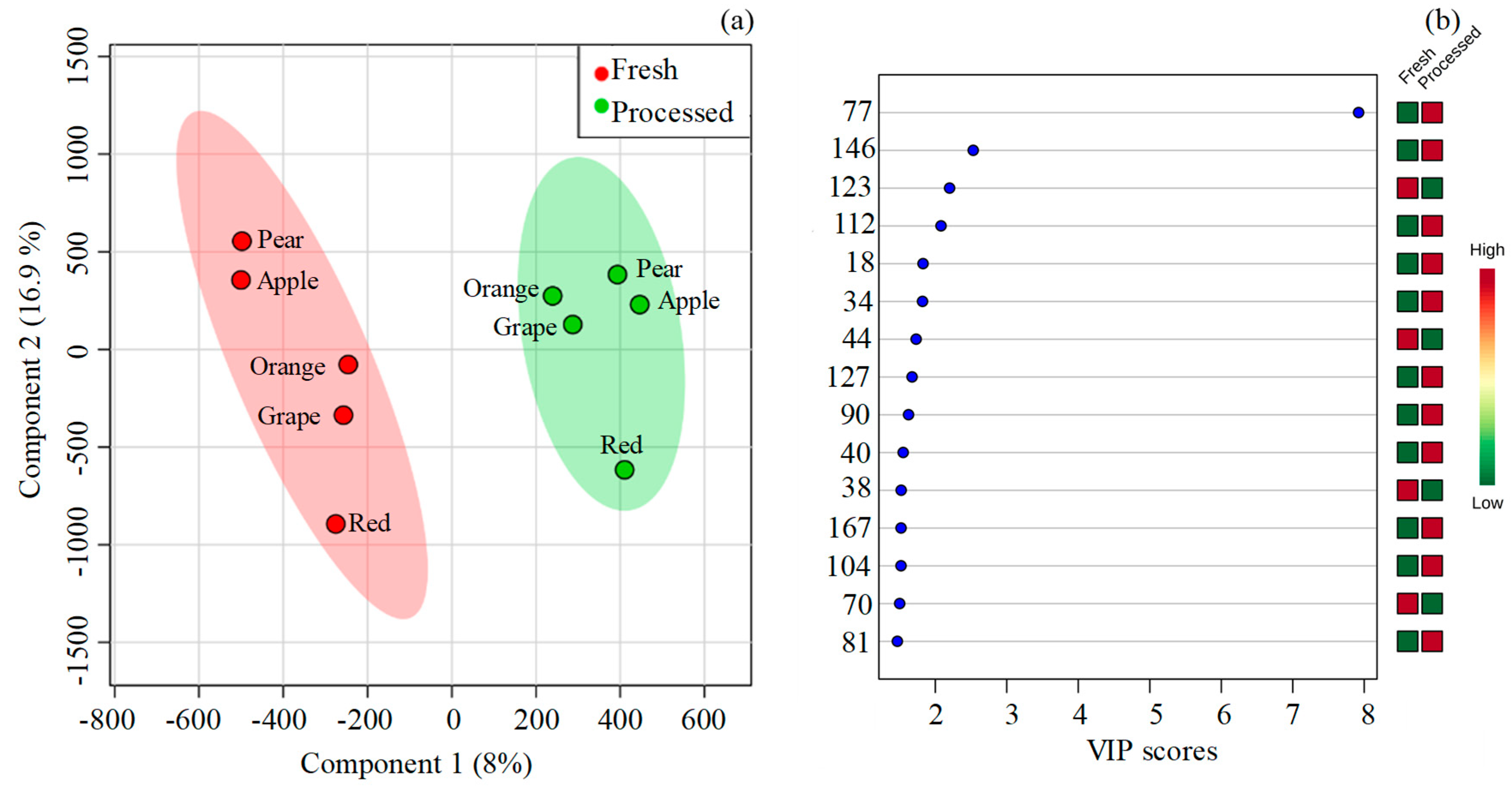
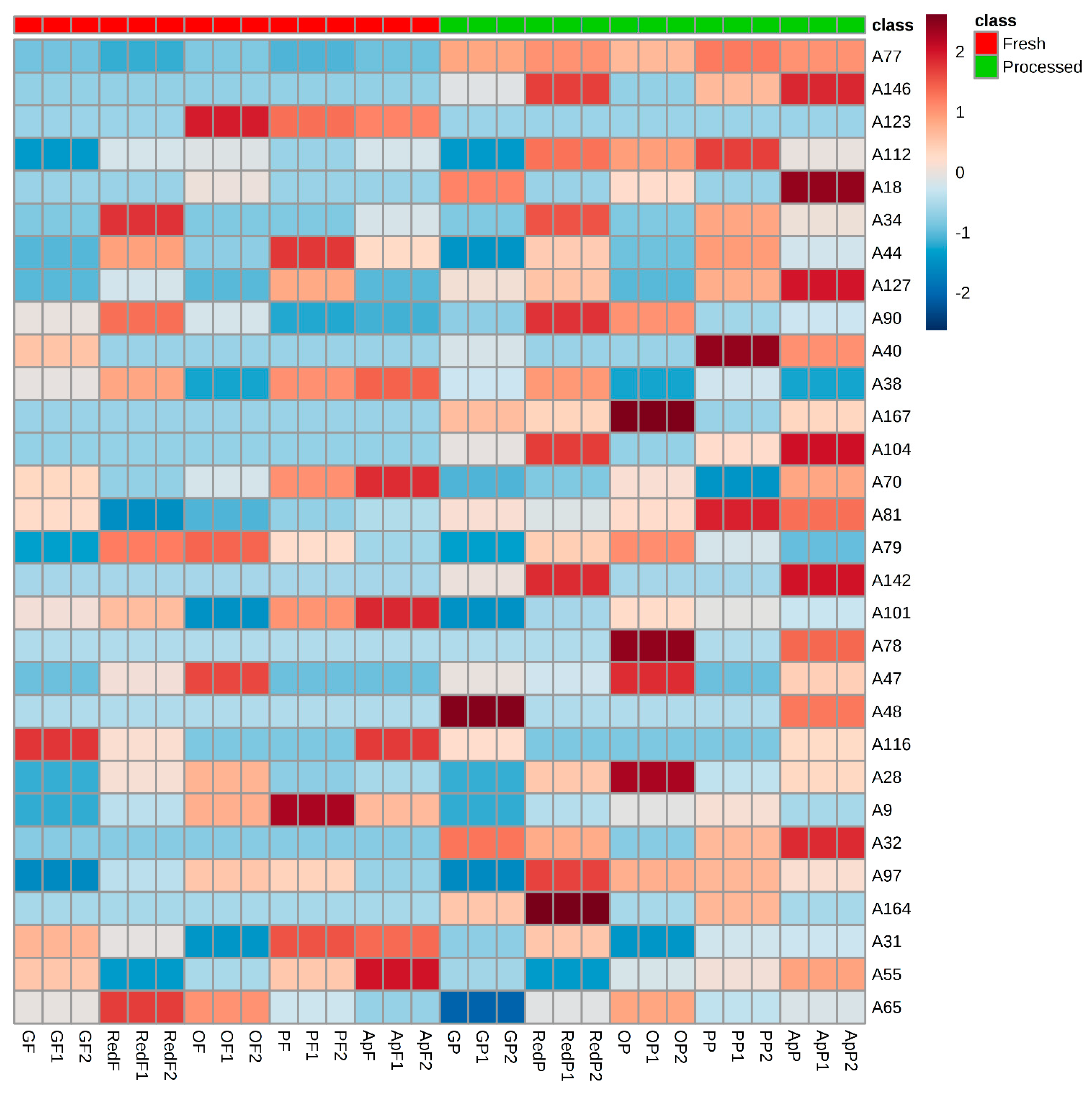
2.1. Volatomic Signature of Fresh and Processed Fruit Juices
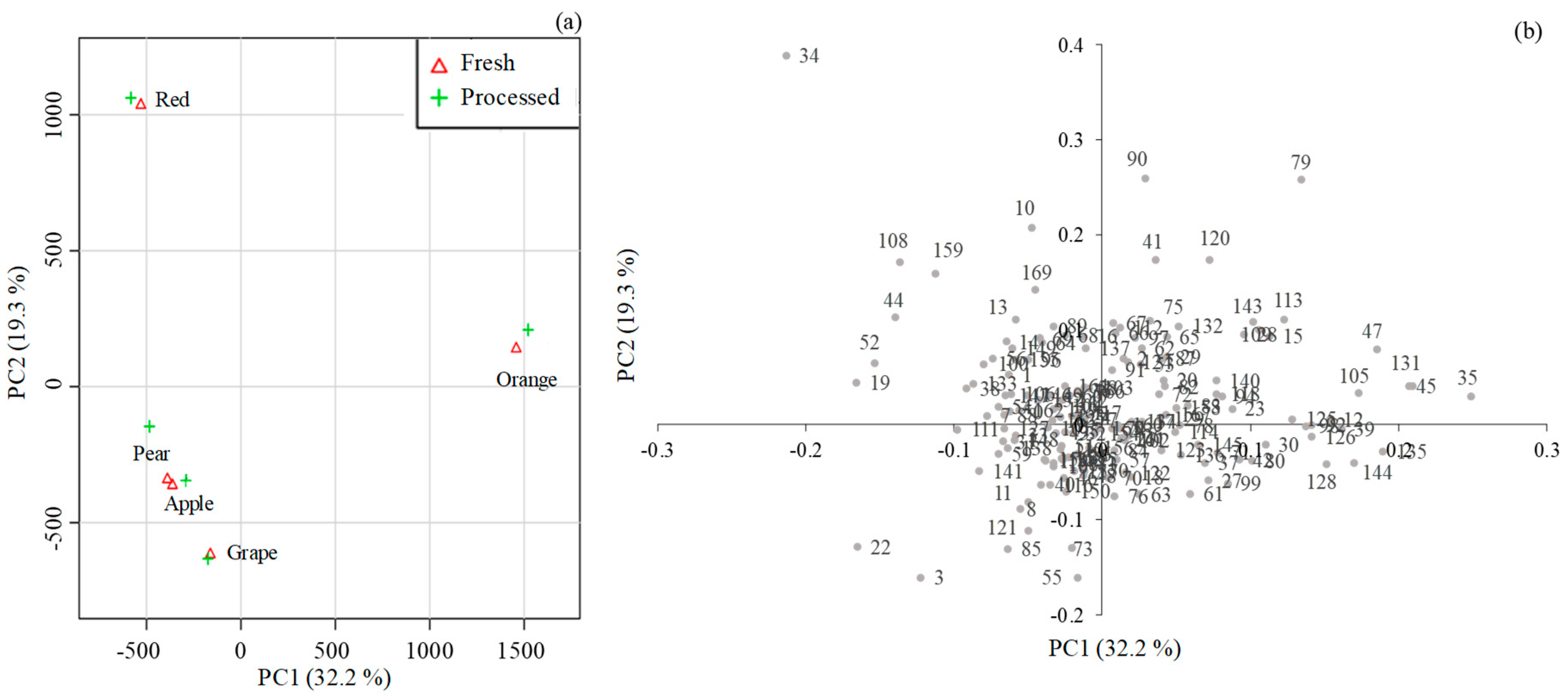
2.2. Multivariate Data Analysis
3. Discussion
4. Materials and Methods
4.1. Material and Chemicals
4.2. Fruit Juice Samples
4.3. Headspace Solid-Phase Microextraction
4.4. Gas Chromatography-Mass Spectrometry Conditions
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Slavin, J.L.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. An Int. Rev. J. 2012, 3, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.; Liu, J.; Zhu, M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ (Clin. Res. Ed.) 2014, 349, g4490. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, M.F.; Sanz, J.; Sanz, M.L.; Giuffrè, A.M.; Sicari, V.; Soria, A.C. Optimization of a solid-phase microextraction method for the gas chromatography–mass spectrometry analysis of blackberry (Rubus ulmifolius Schott) fruit volatiles. Food Chem. 2015, 178, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.N.A.; Fekry, M.I.; Farag, M.A. Metabolome based volatiles profiling in 13 date palm fruit varieties from Egypt via SPME GC–MS and chemometrics. Food Chem. 2017, 217, 171–181. [Google Scholar] [CrossRef] [PubMed]
- El Hadi, M.; Zhang, F.-J.; Wu, F.-F.; Zhou, C.-H.; Tao, J. Advances in fruit aroma volatile research. Molecules 2013, 18, 8200–8229. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.; Perestrelo, R.; Caldeira, M.; Câmara, J.S. Characterization of volatile substances in apples from Rosaceae family by headspace solid-phase microextraction followed by GC-qMS. J. Sep. Sci. 2009, 32, 1875–1888. [Google Scholar] [CrossRef] [PubMed]
- Holland, D.; Larkov, O.; Bar-Ya’akov, I.; Bar, E.; Zax, A.; Brandeis, E.; Ravid, U.; Lewinsohn, E. Developmental and Varietal Differences in Volatile Ester Formation and Acetyl-CoA: Alcohol Acetyl Transferase Activities in Apple (Malus domestica Borkh.). Fruit 2005. [Google Scholar] [CrossRef]
- Rizzolo, A.; Grassi, M.; Eccher Zerbini, P. Influence of harvest date on ripening and volatile compounds in the scab-resistant apple cultivar ‘Golden Orange’. J. Hortic. Sci. Biotechnol. 2006, 81, 681–690. [Google Scholar] [CrossRef]
- Villatoro, C.; Altisent, R.; Echeverría, G.; Graell, J.; López, M.L.; Lara, I. Changes in biosynthesis of aroma volatile compounds during on-tree maturation of ‘Pink Lady®’ apples. Postharvest Biol. Technol. 2008, 47, 286–295. [Google Scholar] [CrossRef]
- Lara, I.; Miró, R.M.; Fuentes, T.; Sayez, G.; Graell, J.; López, M.L. Biosynthesis of volatile aroma compounds in pear fruit stored under long-term controlled-atmosphere conditions. Postharvest Biol. Technol. 2003, 29, 29–39. [Google Scholar] [CrossRef]
- Kahle, K.; Preston, C.; Richling, E.; Heckel, F.; Schreier, P. On-line gas chromatography combustion/pyrolysis isotope ratio mass spectrometry (HRGC-C/P-IRMS) of major volatiles from pear fruit (Pyrus communis) and pear products. Food Chem. 2005, 91, 449–455. [Google Scholar] [CrossRef]
- Rapparini, F.; Predieri, S. Pear Fruit Volatiles. In Horticultural Reviews; John Wiley & Sons, Inc.: Oxford, UK, 2010; pp. 237–324. [Google Scholar]
- Rizzolo, A.; Cambiaghi, P.; Grassi, M.; Zerbini, P.E. Influence of 1-Methylcyclopropene and Storage Atmosphere on Changes in Volatile Compounds and Fruit Quality of Conference Pears. J. Agric. Food Chem. 2005, 53, 9781–9789. [Google Scholar] [CrossRef] [PubMed]
- Kelebek, H.; Selli, S. Determination of volatile, phenolic, organic acid and sugar components in a Turkish cv. Dortyol (Citrus sinensis L. Osbeck) orange juice. J. Sci. Food Agric. 2011, 91, 1855–1862. [Google Scholar] [CrossRef] [PubMed]
- Perestrelo, R.; Barros, A.S.; Rocha, S.M.; Câmara, J.S. Optimisation of solid-phase microextraction combined with gas chromatography–mass spectrometry based methodology to establish the global volatile signature in pulp and skin of Vitis vinifera L. grape varieties. Talanta 2011, 85, 1483–1493. [Google Scholar] [CrossRef] [PubMed]
- Bellincontro, A.; Nicoletti, I.; Valentini, M.; Tomas, A.; De Santis, D.; Corradini, D.; Mencarelli, F. Integration of Nondestructive Techniques with Destructive Analyses to Study Postharvest Water Stress of Winegrapes. Am. J. Enol. Vitic. 2009, 60, 57–65. [Google Scholar]
- Fenoll, J.; Manso, A.; Hellín, P.; Ruiz, L.; Flores, P. Changes in the aromatic composition of the Vitis vinifera grape Muscat Hamburg during ripening. Food Chem. 2009, 114, 420–428. [Google Scholar] [CrossRef]
- Perestrelo, R.; Silva, C.; Silva, P.; Câmara, J.S. Unraveling Vitis vinifera L. grape maturity markers based on integration of terpenic pattern and chemometric methods. Microchem. J. 2018, 142, 367–376. [Google Scholar] [CrossRef]
- Porto-Figueira, P.; Freitas, A.; Cruz, C.J.; Figueira, J.; Câmara, J.S. Profiling of passion fruit volatiles: An effective tool to discriminate between species and varieties. Food Res. Int. 2015, 77, 408–418. [Google Scholar] [CrossRef]
- Riu-Aumatell, M.; Castellari, M.; López-Tamames, E.; Galassi, S.; Buxaderas, S. Characterisation of volatile compounds of fruit juices and nectars by HS/SPME and GC/MS. Food Chem. 2004, 87, 627–637. [Google Scholar] [CrossRef]
- Alves Filho, E.G.; Rodrigues, T.H.S.; Fernandes, F.A.N.; de Brito, E.S.; Cullen, P.J.; Frias, J.M.; Bourke, P.; Cavalcante, R.S.; Almeida, F.D.L.; Rodrigues, S. An untargeted chemometric evaluation of plasma and ozone processing effect on volatile compounds in orange juice. Innov. Food Sci. Emerg. Technol. 2017. [Google Scholar] [CrossRef]
- Evelyn; Kim, H.J.; Silva, F.V.M. Modeling the inactivation of Neosartorya fischeri ascospores in apple juice by high pressure, power ultrasound and thermal processing. Food Control 2016, 59, 530–537. [Google Scholar] [CrossRef]
- Yi, J.; Kebede, B.T.; Hai Dang, D.N.; Buvé, C.; Grauwet, T.; Van Loey, A.; Hu, X.; Hendrickx, M. Quality change during high pressure processing and thermal processing of cloudy apple juice. LWT 2017, 75, 85–92. [Google Scholar] [CrossRef]
- Kebede, B.; Lee, P.Y.; Leong, S.Y.; Kethireddy, V.; Ma, Q.; Aganovic, K.; Eyres, G.T.; Hamid, N.; Oey, I. A Chemometrics Approach Comparing Volatile Changes during the Shelf Life of Apple Juice Processed by Pulsed Electric Fields, High Pressure and Thermal Pasteurization. Foods 2018, 7, 169. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Zhao, L.; Ma, Y.; Wang, Y.; Sun, Z.; Liao, X. Microorganisms and Some Quality of Red Grapefruit Juice Affected by High Pressure Processing and High Temperature Short Time. Food Bioprocess Technol. 2015, 8, 2096–2108. [Google Scholar] [CrossRef]
- Evelyn; Silva, F.V.M. High pressure processing pretreatment enhanced the thermosonication inactivation of Alicyclobacillus acidoterrestris spores in orange juice. Food Control 2016, 62, 365–372. [Google Scholar] [CrossRef]
- Mastello, R.B.; Janzantti, N.S.; Bisconsin-Júnior, A.; Monteiro, M. Impact of HHP processing on volatile profile and sensory acceptance of Pêra-Rio orange juice. Innov. Food Sci. Emerg. Technol. 2018, 45, 106–114. [Google Scholar] [CrossRef]
- Yuk, H.G.; Sampedro, F.; Fan, X.; Geveke, D.J. Nonthermal Processing of Orange Juice Using a Pilot-Plant Scale Supercritical Carbon Dioxide System with a Gas-Liquid Metal Contactor. J. Food Process. Preserv. 2014, 38, 630–638. [Google Scholar] [CrossRef]
- Marszałek, K.; Woźniak, Ł.; Barba, F.J.; Skąpska, S.; Lorenzo, J.M.; Zambon, A.; Spilimbergo, S. Enzymatic, physicochemical, nutritional and phytochemical profile changes of apple (Golden Delicious L.) juice under supercritical carbon dioxide and long-term cold storage. Food Chem. 2018, 268, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Amaral, G.V.; Silva, E.K.; Costa, A.L.R.; Alvarenga, V.O.; Cavalcanti, R.N.; Esmerino, E.A.; Guimarães, J.T.; Freitas, M.Q.; Sant’Ana, A.S.; Cunha, R.L.; et al. Whey-grape juice drink processed by supercritical carbon dioxide technology: Physical properties and sensory acceptance. LWT 2018, 92, 80–86. [Google Scholar] [CrossRef]
- Silva, P.; Freitas, J.; Silva, C.L.C.L.; Perestrelo, R.; Nunes, F.M.F.M.; Câmara, J.S.J.S. Establishment of authenticity and typicality of sugarcane honey based on volatile profile and multivariate analysis. Food Control 2016, 73, 1176–1188. [Google Scholar] [CrossRef]
- Ivanova, V.; Stefova, M.; Vojnoski, B.; Stafilov, T.; Bíró, I.; Bufa, A.; Felinger, A.; Kilár, F. Volatile composition of Macedonian and Hungarian wines assessed by GC/MS. Food Bioprocess Technol. 2013, 6, 1609–1617. [Google Scholar] [CrossRef]
- Lee, S.-N.; Kim, N.-S.; Lee, D.-S. Comparative study of extraction techniques for determination of garlic flavor components by gas chromatography-mass spectrometry. Anal. Bioanal. Chem. 2003, 377, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Mendes, B.; Gonçalves, J.; Câmara, J.S. Effectiveness of high-throughput miniaturized sorbent- and solid phase microextraction techniques combined with gas chromatography-mass spectrometry analysis for a rapid screening of volatile and semi-volatile composition of wines—A comparative study. Talanta 2012, 88, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Belandria, V.; Aparecida de Oliveira, P.M.; Chartier, A.; Rabi, J.A.; de Oliveira, A.L.; Bostyn, S. Pressurized-fluid extraction of cafestol and kahweol diterpenes from green coffee. Innov. Food Sci. Emerg. Technol. 2016, 37, 145–152. [Google Scholar] [CrossRef]
- Ochiai, N.; Sasamoto, K.; Takino, M.; Yamashita, S.; Daishima, S.; Heiden, A.C.; Hoffmann, A. Simultaneous determination of preservatives in beverages, vinegar, aqueous sauces, and quasi-drug drinks by stir-bar sorptive extraction (SBSE) and thermal desorption GC–MS. Anal. Bioanal. Chem. 2002, 373, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Uekane, T.M.; Nicolotti, L.; Griglione, A.; Bizzo, H.R.; Rubiolo, P.; Bicchi, C.; Rocha-Leão, M.H.M.; Rezende, C.M. Studies on the volatile fraction composition of three native Amazonian-Brazilian fruits: Murici (Byrsonima crassifolia L., Malpighiaceae), bacuri (Platonia insignis M., Clusiaceae), and sapodilla (Manilkara sapota L., Sapotaceae). Food Chem. 2017, 219, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Sentandreu, E.; Cerdán-Calero, M.; Sendra, J.M. Phenolic profile characterization of pomegranate (Punica granatum) juice by high-performance liquid chromatography with diode array detection coupled to an electrospray ion trap mass analyzer. J. Food Compos. Anal. 2013, 30, 32–40. [Google Scholar] [CrossRef]
- Tempere, S.; Schaaper, M.H.; Cuzange, E.; de Lescar, R.; de Revel, G.; Sicard, G. The olfactory masking effect of ethylphenols: Characterization and elucidation of its origin. Food Q. Prefer. 2016, 50, 135–144. [Google Scholar] [CrossRef]
- Granato, D.; Margraf, T.; Brotzakis, I.; Capuano, E.; van Ruth, S.M. Characterization of conventional, biodynamic, and organic purple grape juices by chemical markers, antioxidant capacity, and instrumental taste profile. J. Food Sci. 2015, 80, C55–C65. [Google Scholar] [CrossRef] [PubMed]
- Spínola, V.; Perestrelo, R.; Câmara, J.S.; Castilho, P.C. Establishment of monstera deliciosa fruit volatile metabolomic profile at different ripening stages using solid-phase microextraction combined with gas chromatography-mass spectrometry. Food Res. Int. 2015, 67, 409–417. [Google Scholar] [CrossRef]
- Ferreira, V.; Aznar, M.; López, R.; Cacho, J. Quantitative gas chromatography-olfactometry carried out at different dilutions of an extract. Key differences in the odor profiles of four high-quality Spanish aged red wines. J. Agric. Food Chem. 2001, 49, 4818–4824. [Google Scholar] [CrossRef] [PubMed]
- Varming, C.; Petersen, M.A.; Poll, L. Comparison of isolation methods for the determination of important aroma compounds in black currant (Ribes nigrum L.) juice, using nasal impact frequency profiling. J. Agric. Food Chem. 2004, 52, 1647–1652. [Google Scholar] [CrossRef] [PubMed]
- Paduch, R.; Kandefer-Szerszeń, M.; Trytek, M.; Fiedurek, J. Terpenes: Substances useful in human healthcare. Arch. Immunol. Ther. Exp. 2007, 55, 315–327. [Google Scholar] [CrossRef]
- Miller, J.A.; Thompson, P.A.; Hakim, I.A.; Chow, H.-H.S.; Thomson, C.A. d-Limonene: A bioactive food component from citrus and evidence for a potential role in breast cancer prevention and treatment. Oncol. Rev. 2011, 5, 31–42. [Google Scholar] [CrossRef]
- Murali, R.; Karthikeyan, A.; Saravanan, R. Protective effects of D-limonene on lipid peroxidation and antioxidant enzymes in streptozotocin-induced diabetic rats. Basic Clin. Pharmacol. Toxicol. 2013, 112, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-N.; Lim, Y.K.; Freire, M.O.; Cho, E.; Jin, D.; Kook, J.-K. Antimicrobial Effect of Linalool and α-Terpineol Against Periodontopathic and Cariogenic Bacteria. Anaerobe 2012, 18, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lv, O.; Zhou, F.; Li, Q.; Wu, Z.; Zheng, Y. Linalool inhibits LPS-induced inflammation in BV2 microglia cells by activating Nrf2. Neurochem. Res. 2015, 40, 1520–1525. [Google Scholar] [CrossRef] [PubMed]
- Schmutzer, G.R.; Magdas, A.D.; David, L.I.; Moldovan, Z. Determination of the volatile components of apple juice using solid phase microextraction and gas chromatography–mass spectrometry. Anal. Lett. 2014, 47, 1683–1696. [Google Scholar] [CrossRef]
- Perez-Cacho, P.R.; Rouseff, R.L. Fresh Squeezed Orange Juice Odor: A Review. Crit. Rev. Food Sci. Nutr. 2008, 48, 681–695. [Google Scholar] [CrossRef] [PubMed]
- Perez-Cacho, P.R.; Rouseff, R. Processing and Storage Effects on Orange Juice Aroma: A Review. J. Agric. Food Chem. 2008, 56, 9785–9796. [Google Scholar] [CrossRef] [PubMed]
- Arena, E.; Guarrera, N.; Campisi, S.; Nicolosi Asmundo, C. Comparison of odour active compounds detected by gas-chromatography–olfactometry between hand-squeezed juices from different orange varieties. Food Chem. 2006, 98, 59–63. [Google Scholar] [CrossRef]
- Beaulieu, J.C.; Stein-Chisholm, R.E. HS-GC–MS volatile compounds recovered in freshly pressed ‘Wonderful’ cultivar and commercial pomegranate juices. Food Chem. 2016, 190, 643–656. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.C.; Stein-Chisholm, R.E.; Lloyd, S.W.; Bett-Garber, K.L.; Grimm, C.C.; Watson, M.A.; Lea, J.M. Volatile, anthocyanidin, quality and sensory changes in rabbiteye blueberry from whole fruit through pilot plant juice processing. J. Sci. Food Agric. 2017, 97, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Baldwin, E.; McCollum, G.; Plotto, A.; Manthey, J.; Widmer, W.; Luzio, G.; Cameron, R. Changes in volatile and non-volatile flavor chemicals of “Valencia” orange juice over the harvest seasons. Foods 2016, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Araújo, L.; Koppel, K.; Chambers, E., IV; Adhikari, K.; Carbonell-Barrachina, A.A. Instrumental and sensory aroma profile of pomegranate juices from the USA: Differences between fresh and commercial juice. Flavour Fragrance J. 2011, 26, 129–138. [Google Scholar] [CrossRef]
- Qiao, Y.; Xie, B.J.; Zhang, Y.; Zhang, Y.; Fan, G.; Yao, X.L.; Pan, S.Y. Characterization of aroma active compounds in fruit juice and peel oil of Jinchen sweet orange fruit (Citrus sinensis (L.) Osbeck) by GC-MS and GC-O. Molecules (Basel Switzerland) 2008, 13, 1333–1344. [Google Scholar] [CrossRef]
- Edlin, D.A.N.; Narbad, A.; Dickinson, J.R.; Lloyd, D. The biotransformation of simple phenolic compounds by Brettanomyces anomalus. FEMS Microbiol. Lett. 1995, 125, 311–315. [Google Scholar] [CrossRef]
- Daud, M.N.H.; Ahamad, N.; Jabit, M.L.; Kormin, S. A new approach for reduction of chemical compounds causing undesirable odour in Morinda citrifolia fruit juice. J. Trop. Agric. Food Sci. 2013, 41, 257–264. [Google Scholar]
- Perestrelo, R.; Barros, A.S.A.S.; Rocha, S.M.; Câmara, J.S. Establishment of the varietal profile of Vitis vinifera L. grape varieties from different geographical regions based on HS-SPME/GC-qMS combined with chemometric tools. Microchem. J. 2014, 116, 107–117. [Google Scholar] [CrossRef]
- van Den Dool, H. A generalization of the retention index system including linear temperature programmed gas—Liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Acree, T.; Arn, H. Flavornet Home Page. Available online: http://www.flavornet.org/ (accessed on 14 February 2019).
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Peak n° | RT (min) 1 | KICal 2 | KILit 3 | Chemical Groups | GC Peak Area (×106) (RSD) | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grape | Red Fruits | Orange | Pear | Apple | ||||||||||||||||||||
| Fresh | Processed | Fresh | Processed | Fresh | Processed | Fresh | Processed | Fresh | Processed | |||||||||||||||
| Esters | ||||||||||||||||||||||||
| 1 | 5.67 | 897 | 864 | Methyl acetate | 11.18 | (2) | 2.17 | (5) | 29.89 | (7) | 16.76 | (8) | 1.23 | (2) | 1.98 | (5) | 23.94 | (16) | 21.71 | (13) | 1.98 | (19) | 2.58 | (8) |
| 3 | 6.59 | 925 | 907 | Ethyl acetate 4 | 51.31 | (3) | 12.45 | (5) | 4.58 | (6) | 14.56 | (15) | 7.83 | (11) | 3.08 | (15) | 222.09 | (13) | 257.16 | (10) | 144.12 | (8) | 131.55 | (16) |
| 7 | 8.38 | 969 | 950 | Ethyl propanoate 4 | 7.97 | (6) | 26.03 | (5) | 10.21 | (4) | 23.32 | (7) | - | - | - | - | 28.73 | (6) | 7.32 | (12) | ||||
| 8 | 8.95 | 981 | 969 | Propyl acetate 4 | 5.66 | (6) | 0.70 | (9) | - | - | - | - | 9.82 | (5) | 35.95 | (1) | 0.76 | (18) | 17.32 | (1) | ||||
| 10 | 9.29 | 988 | 982 | Methyl butanoate | - | - | 94.87 | (12) | 46.71 | (8) | 1.27 a | (3) | 1.45 a | (1) | - | - | 6.14 | (4) | 1.51 | (13) | ||||
| 11 | 10.17 | 992 | 1007 | Methyl 2-methylbutanoate | 12.72 | (11) | 1.48 | (13) | 1.06 | (3) | - | - | - | 2.15 | (3) | 4.52 | (16) | 10.32 | (9) | 12.34 | (5) | |||
| 13 | 10.53 | 1002 | 1015 | Methyl 3-methylbutanoate | - | - | 13.65 | (3) | 17.86 | (6) | - | - | 3.20 | (1) | 5.41 | (9) | - | - | ||||||
| 15 | 11.33 | 1024 | 1028 | Ethyl butanoate 4 | 17.95 | (6) | 6.42 | (3) | 30.48 | (8) | 18.24 | (8) | 64.68 | (10) | 62.50 | (5) | - | - | 10.08 | (6) | 1.94 | (8) | ||
| 16 | 12.08 | 1043 | 1050 | Ethyl 2-methylbutanoate | - | - | 1.72 | (18) | 42.60 | (5) | 2.35 | (12) | 1.94 | (9) | - | - | 21.85 | (9) | 2.19 | (15) | ||||
| 18 | 12.75 | 1059 | 1060 | Ethyl 3-methylbutanoate | - | 10.39 | (4) | - | - | 1.25 | (4) | 2.16 | (6) | - | - | - | 40.51 | (2) | ||||||
| 19 | 13.02 | 1065 | 1075 | Butyl acetate 4 | 2.18 | (11) | 2.34 | (17) | 67.76 | (2) | 56.12 | (4) | - | - | 21.32 | (4) | 16.93 | (6) | 132.24 | (13) | 221.32 | (8) | ||
| 22 | 15.40 | 1115 | 1117 | 3-Methylbutanol acetate 4 | 10.03 | (13) | 0.88 | (11) | 19.70 | (7) | 5.20 | (15) | - | - | 6.55 | (12) | 19.26 | (14) | 617.98 | (12) | 745.10 | (8) | ||
| 25 | 16.18 | 1130 | 1120 | Ethyl pentanoate | - | - | - | - | - | - | - | - | 6.03 | (13) | 11.65 | (5) | ||||||||
| 26 | 16.51 | 1136 | 1120 | Butyl propanoate | - | - | 6.05 | (14) | 1.46 | (7) | 11.25 | (3) | 26.43 | (3) | 22.63 | (1) | 40.01 | (9) | 33.36 | (18) | 49.34 | (4) | ||
| 28 | 17.65 | 1156 | 1160 | Propyl propanoate | - | - | 5.51 | (7) | 10.41 | (9) | 14.16 | (4) | 66.78 | (6) | 0.65 | (13) | 1.97 | (6) | 1.18 | (9) | 7.71 | (6) | ||
| 30 | 17.88 | 1159 | 1152 | Ethyl 2-butenoate | 6.73 | (10) | 0.52 | (13) | - | - | 15.49 | (14) | 27.52 | (2) | - | - | 3.26 | (3) | - | |||||
| 31 | 18.52 | 1170 | 1147 | Pentyl acetate | 7.25 | (8) | 0.78 | (11) | 2.78 | (8) | 5.89 | (3) | - | - | 16.30 | (9) | 2.02 | (9) | 13.85 | (14) | 1.90 | (6) | ||
| 34 | 19.28 | 1182 | 1188 | Methyl hexanoate | - | - | 564.99 | (7) | 430.05 | (7) | - | - | - | 158.81 | (3) | 13.23 | (8) | 28.72 | (9) | |||||
| 40 | 22.23 | 1229 | 1228 | Propyl butanoate | 4.59 | (6) | 0.74 | (15) | - | - | - | - | - | 48.35 | (7) | - | 10.56 | (15) | ||||||
| 41 | 22.36 | 1231 | 1220 | Ethyl hexanoate 4 | 27.68 | (11) | 1.19 | (5) | 77.27 | (13) | 82.60 | (15) | 23.42 | (7) | 52.65 | (15) | 1.82 | (16) | 6.00 | (9) | 34.09 | (7) | 2.50 | (1) |
| 44 | 24.85 | 1269 | 1270 | Hexyl acetate 4 | 31.63 | (4) | 12.66 | (12) | 356.35 | (8) | 233.56 | (5) | 53.36 | (9) | 39.23 | (2) | 676.33 | (6) | 368.37 | (1) | 193.62 | (10) | 109.58 | (5) |
| 49 | 26.85 | 1296 | 1292 | Ethyl 3-hexenoate 4 | - | - | 3.06 | (12) | 1.28 | (2) | - | - | 2.09 | (14) | 4.47 | (5) | 1.01 | (14) | - | |||||
| 50 | 27.11 | 1300 | 1305 | Ethyl 2-hexenoate | 2.58 | (9) | 1.14 | (12) | 3.25 | (5) | 8.77 | (4) | - | - | 11.41 | (5) | 3.99 | (8) | 0.98 | (5) | 3.36 | (8) | ||
| 51 | 27.51 | 1306 | 1304 | Ethyl 3-ethoxypropanoate | - | - | - | - | - | - | - | 22.33 | (5) | 4.02 | (15) | - | ||||||||
| 52 | 27.76 | 1310 | 1328 | 3-Hexen-1-ol acetate isomer | 4.27 | (3) | 3.36 | (3) | 36.28 | (2) | 142.79 | (2) | - | - | 5.91 | (14) | 9.81 | (7) | 78.43 | (14) | 56.48 | (5) | ||
| 54 | 28.31 | 1319 | 1327 | 2-Hexen-1-ol acetate isomer | - | 0.25 | (12) | 5.57 | (7) | 4.12 | (1) | - | - | 27.12 | (9) | 11.09 | (8) | 3.32 | (17) | 2.88 | (6) | |||
| 56 | 29.10 | 1332 | 1332 | Hexyl propanoate | - | 0.76 | (11) | 26.11 | (4) | 7.36 | (11) | - | - | 6.93 | (19) | 3.66 | (7) | 4.42 | (12) | 4.52 | (12) | |||
| 57 | 29.37 | 1336 | 1305 | Ethyl 2-hexenoate | 3.12 | (5) | 8.58 | (13) | 0.39 | (6) | 2.53 | (4) | 2.01 | (5) | 5.47 | (8) | 1.52 | (17) | 2.24 | (7) | 6.87 | (16) | 3.75 | (1) |
| 61 | 31.36 | 1366 | 1359 | Heptyl acetate | 5.91 | (12) | 0.33 | (9) | - | - | 12.28 | (8) | 23.95 | (7) | 20.09 | (14) | 6.38 | (4) | 1.26 | (7) | 30.99 | (3) | ||
| 64 | 32.35 | 1380 | 1389 | Methyl octanoate | - | - | 10.17 | (15) | 5.98 | (2) | - | - | 0.93 | (13) | 1.76 | (15) | - | - | ||||||
| 68 | 33.66 | 1398 | 1391 | Butyl methylbutanoate | - | - | 21.27 | (12) | 7.13 | (1) | 2.61 | (3) | 1.33 | (2) | 21.82 | (4) | 9.65 | (11) | - | - | ||||
| 69 | 33.99 | 1404 | 1394 | Ethyl 2-hydroxyisovalerate | - | - | 36.49 | (10) | 10.24 | (2) | 2.66 | (17) | 2.25 | (6) | 29.38 | (12) | 7.58 | (18) | 7.22 | (13) | 3.23 | (6) | ||
| 71 | 35.22 | 1425 | 1436 | Ethyl octanoate 4 | 3.97 | (4) | 2.61 | (4) | 4.11 | (7) | 4.39 | (14) | 72.42 | (12) | 42.25 | (4) | 16.89 | (16) | 16.83 | (17) | 47.25 | (4) | 53.46 | (11) |
| 78 | 37.64 | 1465 | 1441 | Isopentyl hexanoate | - | - | - | - | - | 18.74 | (2) | - | - | - | 6.28 | (9) | ||||||||
| 79 | 37.81 | 1468 | 1483 | Ethyl 3-hydroxybutanoate 4 | - | - | 181.49 | (8) | 67.28 | (15) | 220.28 | (7) | 154.85 | (5) | 46.42 | (9) | 20.54 | (16) | 7.41 | (15) | 1.67 | (16) | ||
| 89 | 41.82 | 1532 | 1533 | Hexyl butanoate | - | - | 12.44 | (14) | 7.26 | (4) | - | - | - | - | - | - | ||||||||
| 100 | 45.96 | 1597 | 1591 | Methyl decanoate | - | 0.38 | (4) | 6.96 | (10) | 25.22 | (5) | - | - | 8.15 | (18) | 5.16 | (10) | 17.67 | (8) | 1.84 | (15) | |||
| 104 | 47.59 | 1624 | 1610 | Butyl octanoate | - | 0.54 | (5) | - | 7.06 | (18) | - | - | - | 0.98 | (7) | - | 9.51 | (14) | ||||||
| 108 | 48.69 | 1643 | 1636 | Ethyl decanoate 4 | - | - | 102.43 | (12) | 84.04 | (8) | - | - | 60.17 | (19) | 6.74 | (15) | 6.79 | (12) | 19.02 | (13) | ||||
| 110 | 49.08 | 1650 | 1648 | Ethyl benzoate 4 | 2.14 | (3) | 1.99 | (2) | - | 1.68 | (2) | - | - | - | 2.04 | (1) | 2.39 | (7) | 0.97 | (1) | ||||
| 112 | 49.54 | 1657 | 1664 | Ethyl 3-hydroxyhexanoate 4 | - | - | 5.45 | (17) | 40.42 | (5) | 6.28 | (4) | 27.82 | (12) | 1.93 | (7) | 58.21 | (9) | 5.41 | (16) | 7.43 | (9) | ||
| 116 | 51.36 | 1687 | 1683 | Benzyl butanoate | 18.43 | (7) | 2.27 | (9) | 2.01 | (14) | - | - | - | - | - | 17.49 | (8) | 2.58 | (13) | |||||
| 117 | 52.22 | 1700 | 1689 | Diethyl succinate 4 | - | 1.04 | (14) | 3.19 | (14) | - | - | - | - | - | 1.48 | (11) | - | |||||||
| 120 | 52.69 | 1708 | 1694 | Ethyl 9-decenoate | 6.83 | (7) | 1.03 | (4) | 66.22 | (18) | 204.61 | (5) | 184.42 | (9) | 93.89 | (8) | 4.33 | (3) | 10.33 | (7) | 190.78 | (7) | 74.42 | (9) |
| 130 | 55.09 | 1747 | 1745 | Methyl salicylate 4 | 53.55 | (4) | 27.22 | (5) | 3.86 | (12) | 1.97 | (3) | - | 4.59 | (10) | - | 1.59 | (5) | - | 0.78 | (9) | |||
| 134 | 56.42 | 1768 | 1729 | 3-Hexenyl hexanoate | - | - | 0.68 | (19) | 2.06 | (5) | 6.97 | (2) | 3.53 | (10) | 20.76 | (16) | 1.23 | (2) | 3.25 | (8) | 4.63 | (14) | ||
| 137 | 57.41 | 1783 | 1803 | 2-Phenylethyl acetate 4 | 27.70 | (7) | 2.12 | (11) | 59.28 | (13) | 16.31 | (7) | 12.34 | (17) | 7.78 | (8) | 4.50 | (8) | 12.12 | (2) | 1.06 | (2) | 10.47 | (8) |
| 141 | 58.86 | 1807 | 1821 | Benzyl propanoate | - | - | 0.54 | (7) | 1.98 | (4) | - | - | 168.13 | (14) | 42.07 | (5) | 10.62b | (18) | 10.74b | (4) | ||||
| 146 | 60.32 | 1839 | 1837 | Ethyl dodecanoate 4 | - | 1.07 | (9) | - | 22.39 | (18) | - | - | - | 5.47 | (6) | - | 27.04 | (12) | ||||||
| 148 | 61.85 | 1872 | 1872 | Geranyl butanoate | 4.23 | (4) | 2.10 | (18) | 0.74 | (17) | 9.42 | (15) | - | - | 9.88 | (4) | 2.37 | (11) | - | 22.58 | (5) | |||
| 151 | 63.49 | 1906 | 1895 | Geranyl isovalerate | - | 1.71 | (13) | 0.50 | (6) | - | - | - | 64.89 | (14) | 2.89 | (4) | - | - | ||||||
| 154 | 65.54 | 1944 | 1960 | Geranyl valerate | - | - | - | - | - | - | - | - | 1.98 | (17) | - | |||||||||
| 155 | 66.22 | 1956 | 1974 | Methyl jasmonate | 11.10 | (4) | 21.96 | (11) | 15.26 | (7) | 29.99 | (9) | - | - | - | - | - | - | ||||||
| 161 | 73.02 | 2126 | 2114 | Bornyl benzoate | - | - | - | - | 7.79 | (11) | - | 4.47 | (14) | 18.77 | (10) | - | - | |||||||
| 163 | 74.72 | 2135 | 2139 | Ethyl cinnamate 4 | - | 2.89 | (9) | 2.15 | (4) | 6.64 | (7) | 1.02 c | (14) | 0.98 c | (2) | 37.80 | (19) | 1.45 | (10) | 3.40 | (17) | 2.97 | (7) | |
| 168 | 77.99 | 2154 | 2171 | Diethyl 2-hydroxyglutarate | 1.28 | (2) | 0.52 | (12) | 0.39 | (5) | 0.15 | (4) | - | - | 4.77 | (10) | 2.15 | (16) | 2.34 | (8) | 2.03 | (10) | ||
| % RPA | 25.98 | 18.45 | 64.16 | 50.29 | 23.58 | 20.90 | 58.84 | 45.15 | 55.00 | 57.83 | ||||||||||||||
| Alcohols | ||||||||||||||||||||||||
| 6 | 7.62 | 952 | 929 | Ethanol 4 | 106.00 | (4) | 82.28 | (9) | 51.19 | (15) | 119.72 | (9) | 67.62 | (12) | 89.99 | (8) | 185.09 | (14) | 111.82 | (12) | 93.32 | (6) | 136.76 | (14) |
| 14 | 11.29 | 1023 | 1037 | 1-Propanol 4 | 40.25 | (3) | 11.05 | (5) | 52.66 | (7) | 84.80 | (4) | 4.26 | (3) | 3.98 | (2) | 4.17 | (7) | 3.63 | (13) | 10.21 | (3) | 24.98 | (2) |
| 27 | 16.86 | 1142 | 1099 | 2-Butanol 4 | 2.80 | (5) | 0.35 | (6) | - | - | 18.87 | (6) | 21.16 | (6) | 11.99 | (17) | 6.14 | (5) | 24.91 | (4) | 1.80 | (9) | ||
| 37 | 20.56 | 1201 | 1206 | 3-Methyl-1-butanol 4 | 1.83 | (2) | 0.99 | (7) | 2.15 | (3) | 4.25 | (2) | 40.21 | (2) | 55.17 | (4) | 8.39 | (18) | 7.23 | (2) | 109.54 | (4) | 99.08 | (2) |
| 59 | 29.87 | 1344 | 1332 | 2-Heptanol 4 | 33.40 | (6) | 30.15 | (8) | 13.20 | (10) | 53.42 | (4) | 36.18 | (17) | 4.01 | (1) | 160.92 | (10) | 296.92 | (8) | 6.78 | (19) | 13.78 | (3) |
| 60 | 30.59 | 1355 | 1360 | 1-Hexanol 4 | - | - | - | 6.89 | (5) | - | - | - | - | - | 7.00 | (10) | ||||||||
| 62 | 31.93 | 1374 | 1379 | 3-Ethoxypropanol | 2.55 | (6) | 0.83 | (10) | 7.36 | (10) | 18.74 | (9) | 6.42 | (17) | 8.26 | (2) | - | - | - | 19.92 | (2) | |||
| 67 | 33.31 | 1394 | 1391 | 3-Hexen-1-ol isomer 4 | 6.95 | (5) | 0.89 | (3) | 23.25 | (8) | 66.75 | (5) | 13.55 | (13) | 25.46 | (5) | 3.37 | (16) | 4.28 | (8) | 17.11 | (10) | 28.55 | (2) |
| 74 | 36.18 | 1441 | 1445 | 1-Octen-3-ol 4 | 3.04 | (14) | 2.20 | (3) | 4.27 | (2) | 3.33 | (4) | 8.90 | (7) | 6.30 | (15) | 8.57 | (5) | 21.73 | (6) | 1.19 | (18) | 15.40 | (3) |
| 75 | 36.37 | 1444 | 1467 | Heptanol 4 | - | - | 27.03 | (3) | 9.14 | (8) | 12.33 | (18) | 9.70 | (3) | - | - | 8.51 | (6) | 13.99 | (15) | ||||
| 81 | 38.61 | 1480 | 1487 | 2-Ethyl-1-hexanol 4 | 10.47 | (9) | 9.44 | (13) | 1.56 | (9) | 7.55 | (9) | 2.82 | (10) | 10.25 | (2) | 4.43 | (13) | 30.86 | (5) | 5.58 | (8) | 22.00 | (7) |
| 91 | 42.69 | 1546 | 1553 | 1-Octanol 4 | 6.62 | (2) | 7.29 | (8) | 20.37 | (4) | 76.80 | (4) | 72.34 | (9) | 22.57 | (3) | 27.20 | (5) | 68.14 | (4) | 24.58 | (11) | 75.42 | (2) |
| 109 | 48.80 | 1645 | 1624 | 1-Nonanol | 3.45 | (12) | 3.78 | (18) | 4.67 | (20) | 29.29 | (6) | 23.40 | (7) | 46.54 | (9) | - | - | - | - | ||||
| 147 | 60.80 | 1850 | 1865 | Benzyl alcohol 4 | 1.96 | (14) | 3.47 | (12) | 1.51 | (13) | 30.96 | (3) | - | - | 9.62 | (15) | 5.55 | (2) | - | - | ||||
| 150 | 62.97 | 1896 | 1925 | Phenylethyl alcohol 4 | 19.48 | (6) | 22.53 | (7) | 1.92 | (5) | 6.72 | (10) | 3.10 | (8) | 8.23 | (4) | 13.47 | (3) | 31.77 | (17) | 11.77 | (4) | 10.93 | (13) |
| 156 | 67.46 | 1978 | 1952 | 1-Tridecanol | - | 0.77 | (4) | - | - | - | - | - | - | - | 7.71 | (17) | ||||||||
| % RPA | 18.14 | 19.97 | 7.14 | 15.08 | 10.05 | 9.67 | 16.21 | 20.74 | 10.14 | 15.60 | ||||||||||||||
| Carbonyl compounds | ||||||||||||||||||||||||
| 2 | 6.46 | 921 | - | Butanal 4 | - | - | 5.47 | (18) | 9.43 | (17) | 4.25 | (2) | 9.50 | (3) | 3.19 | (5) | 7.42 | (16) | 1.70 | (15) | 5.24 | (11) | ||
| 4 | 7.19 | 941 | 926 | 2-Methylbutanal | 3.34 | (2) | 1.75 | (1) | 2.34 | (7) | 1.97 | (2) | - | - | - | - | 0.26 | (11) | 2.38 | (9) | ||||
| 5 | 7.33 | 944 | 936 | 3-Methylbutanal 4 | 4.63 | (4) | 1.69 | (14) | - | - | - | - | - | - | - | - | ||||||||
| 9 | 8.98 | 982 | 973 | 2-Pentanone | - | - | 1.08 d | (2) | 0.96 d | (3) | 7.34 | (5) | 2.14 | (12) | 30.36 | (5) | 2.87 | (8) | 6.22 | (5) | 0.77 | (4) | ||
| 20 | 13.31 | 1072 | 1084 | Hexanal 4 | 17.26 | (11) | 2.06 | (18) | 18.35 | (7) | 9.78 | (12) | 18.53 | (12) | 14.47 | (7) | - | - | 18.57 | (18) | 7.93 | (6) | ||
| 24 | 15.93 | 1125 | 1131 | 2-Pentenal | - | 2.44 | (9) | - | 3.02 | (13) | - | - | - | - | - | 1.28 | (15) | |||||||
| 32 | 18.95 | 1177 | 1170 | 2-Heptanone | - | 2.12 | (1) | - | 1.27 | (18) | - | - | - | 1.08 | (10) | - | 3.51 | (16) | ||||||
| 33 | 19.13 | 1179 | 1174 | Heptanal 4 | 21.96 | (10) | 24.03 | (7) | 5.36 | (17) | 12.33 | (5) | - | - | - | 0.34 | (8) | 1.66 | (13) | 4.05 | (7) | |||
| 36 | 20.22 | 1196 | 1192 | 2-Hexenal isomer | 22.33 | (1) | 25.43 | (6) | 36.36 | (5) | 27.90 | (1) | 10.24 | (3) | 20.25 | (2) | 3.27 | (11) | 38.82 | (4) | 8.61 | (16) | 20.50 | (13) |
| 38 | 21.17 | 1211 | 1200 | 2-Hexenal isomer | 4.66 | (10) | 2.89 | (10) | 17.01 | (15) | 19.74 | (5) | - | - | 21.20 | (5) | 3.08 | (2) | 31.28 | (13) | - | |||
| 43 | 23.49 | 1248 | 1244 | 3-Octanone 4 | 14.11 | (9) | 1.51 | (9) | 0.52 | (12) | 5.30 | (8) | - | - | 0.41 | (1) | - | - | 1.90 | (12) | ||||
| 46 | 25.58 | 1279 | 1272 | 3-Hydroxy-2-butanone | 9.30 | (9) | 0.97 | (4) | - | - | - | - | - | 13.09 | (10) | - | 1.18 | (17) | ||||||
| 47 | 25.73 | 1281 | 1280 | Octanal 4 | - | 7.10 | (6) | 8.63 | (3) | 3.73 | (10) | 99.19 | (13) | 122.34 | (4) | - | - | - | 18.05 | (5) | ||||
| 48 | 26.58 | 1292 | 1287 | 1-Octen-3-one 4 | - | 18.29 | (7) | - | - | - | - | - | - | - | 4.89 | (9) | ||||||||
| 53 | 27.92 | 1313 | 1299 | 2-Heptenal isomer | - | - | 6.75 | (3) | 4.23 | (6) | 4.53 | (16) | 10.89 | (9) | 1.57 | (15) | 4.26 | (3) | 6.51 | (4) | 1.02 | (6) | ||
| 55 | 28.90 | 1329 | 1319 | 6-Methyl-5-hepten-2-one | 25.18 | (7) | 3.10 | (15) | - | - | 3.57 | (3) | 8.24 | (11) | 24.52 | (10) | 12.91 | (6) | 119.93 | (11) | 40.24 | (8) | ||
| 63 | 32.20 | 1378 | 1388 | 2-Nonanone | 2.24 | (1) | 16.47 | (8) | - | - | 4.74 | (1) | 1.91 | (7) | 0.98 | (2) | 1.63 | (9) | 2.22 | (10) | 2.83 | (1) | ||
| 65 | 32.51 | 1383 | 1385 | Nonanal 4 | 19.70 | (15) | 0.91 | (13) | 72.47 | (9) | 18.60 | (13) | 46.59 | (7) | 41.59 | (13) | 15.56 | (1) | 14.10 | (3) | 10.13 | (13) | 17.48 | (10) |
| 66 | 33.17 | 1392 | 1401 | 2,4-Heptadienal isomer | - | - | 10.93 | (1) | 7.66 | (4) | 2.25 | (1) | 2.86 | (6) | - | - | 1.20 | (4) | 1.94 | (5) | ||||
| 70 | 34.73 | 1417 | 1408 | 2-Octenal isomer | 7.59 | (15) | 0.43 | (5) | 1.32 | (14) | 0.96 | (6) | 3.58 | (19) | 5.86 | (2) | 18.06 | (7) | - | 35.95 | (8) | 14.73 | (9) | |
| 82 | 38.78 | 1483 | 1458 | Methional 4 | 1.41 | (3) | 2.42 | (8) | 3.53 | (5) | 6.54 | (14) | 10.12 | (14) | 15.97 | (3) | 1.13 | (18) | 13.33 | (7) | 0.67 | (7) | 2.14 | (2) |
| 83 | 39.12 | 1488 | 1484 | Decanal 4 | 40.95 | (6) | 14.28 | (15) | 37.57 | (13) | 16.69 | (12) | 84.32 | (7) | 35.24 | (1) | 15.61 | (3) | 11.92 | (15) | 14.23 | (18) | 28.73 | (8) |
| 86 | 40.58 | 1512 | 1495 | Benzaldehyde 4 | 25.94 | (12) | 21.09 | (8) | 5.38 | (17) | 18.16 | (2) | 2.67 | (18) | 14.01 | (12) | 16.05 | (8) | 3.19 | (3) | 6.91 | (11) | 24.45 | (2) |
| 87 | 41.30 | 1524 | 1527 | 2-Nonenal isomer 4 | - | - | 6.10 | (9) | 9.37 | (14) | 10.19 | (11) | 16.22 | (2) | 5.74 | (13) | 1.59 | (8) | 3.01 | (12) | 9.74 | (19) | ||
| 95 | 44.40 | 1573 | 1575 | 2,6-Nonadienal isomer | 1.46 | (1) | 6.51 | (5) | 1.08 e | (15) | 0.98 e | (1) | - | - | - | 2.07 | (16) | - | - | |||||
| 96 | 44.77 | 1579 | 1561 | 6-Methyl-3,5-heptadiene-2-one | - | 2.47 | (12) | 12.57 | (19) | 6.44 | (2) | - | - | - | 3.18 | (15) | - | 4.35 | (9) | |||||
| 106 | 48.08 | 1633 | 1645 | Acetophenone 4 | 9.26 | (1) | 1.05 | (17) | 9.61 | (5) | 7.82 | (9) | - | - | 17.96 | (18) | 3.14 | (2) | - | - | ||||
| 127 | 54.61 | 1739 | 1710 | 2,4-Decadienal 4 | - | 3.17 | (14) | 1.66 | (11) | 6.89 | (17) | - | - | 10.06 | (6) | 9.62 | (15) | - | 34.88 | (9) | ||||
| 129 | 54.96 | 1745 | 1753 | Ethyl benzaldehyde 4 | - | - | - | - | - | 10.48 | (4) | - | 2.75 | (9) | - | - | ||||||||
| 136 | 57.07 | 1778 | 1758 | 2,4-Decadienal | - | 0.23 | (5) | - | - | 11.22 | (6) | 7.15 | (2) | 5.39 | (11) | 3.27 | (3) | 8.91 | (15) | 2.37 | (4) | |||
| 164 | 75.14 | 2138 | 2103 | γ-Decalactone 4 | - | 1.07 | (5) | - | 10.56 | (9) | - | - | - | 1.36 | (11) | - | - | |||||||
| 166 | 76.38 | 2145 | 2112 | Hexadecanone | - | 0.61 | (12) | 0.60 | (10) | 2.75 | (11) | - | - | - | - | - | - | |||||||
| 169 | 80.45 | 2167 | 2153 | Palmitaldehyde | - | - | 25.00 | (12) | 17.37 | (14) | - | - | - | - | - | - | ||||||||
| % RPA | 17.57 | 18.62 | 9.80 | 6.70 | 10.48 | 10.53 | 7.08 | 5.47 | 8.99 | 8.39 | ||||||||||||||
| Terpenoids | ||||||||||||||||||||||||
| 12 | 10.45 | 1000 | 1032 | α-Pinene 4 | - | - | - | - | 79.43 | (4) | 38.60 | (5) | - | 5.59 | (16) | 6.55 | (18) | 1.47 | (6) | |||||
| 17 | 12.26 | 1047 | 1075 | Camphene | - | - | - | - | 1.17 | (13) | 0.98 | (6) | - | 1.03 | (3) | - | - | |||||||
| 21 | 14.03 | 1087 | 1116 | β-Pinene 4 | - | - | - | - | 4.84 | (3) | - | - | 6.22 | (14) | - | 5.23 | (11) | |||||||
| 23 | 15.90 | 1124 | 1148 | δ-3-Carene | - | - | - | - | 9.57 | (11) | 8.96 | (5) | - | - | - | - | ||||||||
| 29 | 17.73 | 1157 | 1141 | β-Myrcene 4 | - | - | 0.72 | (7) | 7.17 | (5) | 16.13 | (12) | 4.96 | (2) | - | - | - | - | ||||||
| 35 | 19.68 | 1188 | 1198 | Limonene 4 | 5.61 | (5) | 1.16 | (6) | 0.96 | (4) | 7.72 | (10) | 359.17 | (9) | 203.95 | (3) | 14.12 | (3) | 1.81 | (6) | 9.31 | (4) | 11.47 | (6) |
| 39 | 21.92 | 1224 | 1225 | Ocimene | - | - | - | - | 55.58 | (3) | 99.12 | (3) | 8.89 | (14) | 12.83 | (7) | - | 12.73 | (12) | |||||
| 42 | 22.85 | 1239 | 1238 | γ-Terpinene | 20.74 | (7) | 1.75 | (8) | - | - | 20.58 | (16) | 8.16 | (4) | - | - | - | - | ||||||
| 45 | 25.06 | 1271 | 1261 | p-Cymene 4 | - | - | - | - | 76.29 | (9) | 99.40 | (11) | - | - | - | - | ||||||||
| 58 | 29.73 | 1342 | 1337 | Rose oxide isomer | - | - | 1.05 | (5) | 1.36 | (12) | - | - | - | - | - | - | ||||||||
| 72 | 35.47 | 1429 | 1423 | Linalool oxide isomer | - | - | 2.98 | (2) | 3.17 | (15) | 15.28 | (7) | 12.36 | (3) | 2.14 | (5) | 24.11 | (11) | 10.13 | (2) | 5.48 | (1) | ||
| 73 | 35.58 | 1431 | 1449 | Dihydrolinalool | 83.01 | (5) | 48.23 | (8) | - | - | - | - | - | - | - | - | ||||||||
| 80 | 37.90 | 1469 | 1488 | Citronellal 4 | 6.33 | (2) | 4.23 | (5) | - | - | 26.50 | (15) | 12.15 | (6) | - | - | 0.95 | (17) | 1.20 | (8) | ||||
| 84 | 39.77 | 1498 | 1491 | Camphor | 17.77 | (15) | 2.93 | (10) | 3.26 | (10) | 3.21 | (11) | 7.98 | (5) | 8.22 | (4) | 8.05 | (14) | 25.32 | (6) | 1.79 | (7) | 7.35 | (11) |
| 85 | 40.32 | 1507 | 1506 | Isoborneol | 7.60 | (9) | 6.11 | (2) | - | - | - | - | 24.88 | (15) | 7.13 | (2) | 26.74 | (7) | 11.44 | (8) | ||||
| 88 | 41.61 | 1529 | 1516 | Isocitral | 16.34 | (15) | 1.63 | (6) | 10.86 | (15) | 5.51 | (3) | - | - | 3.52 | (3) | 5.51 | (10) | 0.96 | (13) | 3.48 | (13) | ||
| 90 | 42.18 | 1538 | 1537 | Linalool 4 | 60.66 | (2) | 18.25 | (4) | 254.57 | (17) | 375.08 | (4) | 46.40 | (17) | 198.03 | (4) | 5.54 | (9) | 24.57 | (8) | 7.48 | (4) | 41.07 | (6) |
| 92 | 43.37 | 1557 | 1550 | Dihydrocarvone | - | - | - | - | 40.21 | (3) | 58.42 | (5) | 2.76 | (2) | 4.43 | (9) | 1.32 | (13) | 3.73 | (11) | ||||
| 94 | 44.12 | 1569 | 1574 | Fenchyl alcohol | - | - | 4.67 | (12) | 4.88 | (10) | 64.49 | (6) | 24.65 | (6) | 20.45 | (18) | 3.72 | (11) | 15.76 | (4) | 30.35 | (5) | ||
| 97 | 45.21 | 1585 | 1580 | Bornyl acetate | - | - | 3.18 | (13) | 37.19 | (1) | 12.33 | (12) | 16.01 | (6) | 10.25 | (3) | 14.70 | (3) | 1.95 | (15) | 7.62 | (10) | ||
| 98 | 45.58 | 1591 | 1562 | Isodihydrocarveol | - | - | - | - | 25.67 | (6) | 65.65 | (5) | 0.78 | (4) | 1.84 | (7) | 1.64 | (13) | 9.09 | (5) | ||||
| 99 | 45.89 | 1596 | 1596 | Mrytenal | 39.62 | (6) | 6.52 | (3) | - | - | 12.08 | (6) | 11.93 | (14) | - | - | - | - | ||||||
| 101 | 46.15 | 1599 | 1598 | β-Ciclocitral 4 | 13.87 | (7) | 1.25 | (2) | 24.00 | (12) | 6.40 | (4) | 1.26 | (9) | 16.60 | (8) | 32.96 | (4) | 11.92 | (7) | 62.18 | (11) | 9.01 | (7) |
| 102 | 46.53 | 1606 | 1602 | Citral 4 | 11.21 | (14) | 5.50 | (6) | 2.98 | (2) | 3.17 | (5) | 5.01 | (13) | 7.72 | (6) | 4.96 | (3) | 5.21 | (3) | 1.53 | (1) | 2.96 | (2) |
| 103 | 46.99 | 1614 | 1646 | β-Terpineol 4 | 53.04 | (11) | 20.97 | (4) | 34.86 | (15) | 26.29 | (7) | 6.56 | (8) | 14.37 | (10) | 10.01 | (3) | 2.70 | (8) | 1.21 | (2) | 4.88 | (7) |
| 105 | 47.70 | 1626 | 1648 | Safranal | - | - | - | - | 49.88 | (8) | 55.30 | (12) | - | - | - | - | ||||||||
| 107 | 48.44 | 1639 | 1639 | Carvone | - | - | - | - | - | - | 4.21 | (2) | 4.02 | (5) | 9.15 | (8) | 2.16 | (18) | ||||||
| 111 | 49.37 | 1654 | 1655 | Estragole | - | - | 8.27 | (15) | 4.27 | (10) | - | - | 41.13 | (16) | 32.61 | (9) | 31.53 | (14) | 13.11 | (9) | ||||
| 113 | 50.21 | 1668 | 1648 | β-Farnesene isomer | - | - | 14.23 | (5) | 8.96 | (3) | 54.22 | (2) | 90.62 | (8) | 5.01 | (3) | 16.44 | (12) | 1.57 | (17) | 2.33 | (12) | ||
| 114 | 50.80 | 1678 | 1688 | α-Terpineol 4 | 21.09 | (6) | 13.47 | (9) | 9.70 | (1) | 12.44 | (9) | 40.86 | (7) | 29.87 | (7) | 15.12 | (4) | 11.91 | (11) | 10.18 | (7) | 15.52 | (1) |
| 115 | 51.03 | 1681 | 1667 | Neral | - | - | - | - | - | - | 4.06 | (8) | 2.06 | (3) | 5.59 | (5) | 1.34 | (8) | ||||||
| 118 | 52.30 | 1701 | 1705 | Germacrene D | - | - | 2.76 | (9) | 1.96 | (5) | 128.16 | (4) | 18.84 | (5) | 1.77 | (17) | 1.66 | (12) | 9.10 | (8) | 11.36 | (4) | ||
| 119 | 52.57 | 1706 | 1715 | Geranial isomer | - | - | - | - | - | - | 34.53 | (11) | 19.82 | (5) | - | - | ||||||||
| 121 | 53.05 | 1714 | 1718 | Naphthalene | 4.28 | (1) | 8.10 | (13) | - | - | - | - | 3.00 | (6) | 3.80 | (14) | 17.96 | (2) | 19.54 | (5) | ||||
| 122 | 53.26 | 1717 | 1716 | TDN | 12.46 | (6) | 1.79 | (4) | - | - | 3.16 | (11) | - | - | - | - | 0.99 | (9) | ||||||
| 123 | 53.65 | 1724 | 1726 | Valencene | - | - | - | - | 24.14 | (7) | - | 11.69 | (7) | - | 9.51 | (10) | - | |||||||
| 124 | 53.91 | 1728 | 1725 | α-Farnesene | - | - | 13.75 | (19) | 20.96 | (5) | 27.25 | (4) | 21.86 | (7) | 18.58 | (10) | 27.96 | (12) | 419.32 | (3) | 8.16 | (5) | ||
| 125 | 54.10 | 1731 | 1714 | α-Muurolene | - | - | - | - | 38.90 | (1) | 31.01 | (8) | 12.77 | (9) | 1.80 | (1) | - | - | ||||||
| 126 | 54.27 | 1734 | 1711 | Geranyl acetate | - | - | - | - | 31.54 | (4) | 66.51 | (10) | - | - | 23.65 | (12) | 13.37 | (13) | ||||||
| 128 | 54.74 | 1741 | 1715 | Geranial isomer | 14.43 | (13) | 0.50 | (3) | - | - | 50.82 | (13) | 72.06 | (10) | 0.98 | (2) | 1.97 | (2) | 3.61 | (5) | 5.64 | (12) | ||
| 131 | 55.43 | 1752 | 1751 | Carvone | - | - | - | - | 66.83 | (3) | 103.53 | (8) | - | - | - | - | ||||||||
| 132 | 55.82 | 1758 | 1759 | Cumin aldehyde | - | - | 3.88 | (4) | 9.76 | (7) | 2.48 | (16) | 15.89 | (12) | - | - | - | - | ||||||
| 133 | 55.95 | 1760 | 1762 | β-Citronellol 4 | - | - | 10.91 | (16) | 8.67 | (10) | - | - | 7.43 | (5) | 34.98 | (5) | 23.08 | (15) | 1.84 | (11) | ||||
| 135 | 56.53 | 1769 | 1770 | Nerol | 7.34 | (4) | 4.67 | (7) | - | - | 82.71 | (11) | 87.95 | (9) | - | - | 2.87 | (1) | 1.59 | (3) | ||||
| 138 | 57.50 | 1784 | 1765 | Perillaldehyde | 17.32 | (10) | 10.26 | (9) | 8.62 | (4) | 12.92 | (5) | 1.36 | (3) | 2.15 | (4) | 1.83 | (9) | 45.48 | (10) | 19.86 | (6) | 22.03 | (10) |
| 139 | 58.01 | 1792 | 1813 | β-Damascenone 4 | 6.98 | (3) | 10.03 | (13) | 2.41 | (14) | 20.16 | (2) | 1.34 | (9) | 1.21 | (2) | 5.65 | (11) | 4.37 | (4) | 1.10 | (8) | 0.92 | (5) |
| 140 | 58.38 | 1798 | 1786 | Cadinadiene | - | - | 1.74 | (15) | 2.76 | (1) | 16.07 | (18) | 33.65 | (12) | 22.32 | (17) | 17.16 | (6) | - | - | ||||
| 142 | 59.08 | 1812 | 1809 | α-Ionone 4 | - | 0.53 | (6) | - | 8.66 | (6) | - | - | - | - | - | 10.22 | (9) | |||||||
| 144 | 59.60 | 1824 | 1840 | Geranyl acetone isomer | 1.76 | (1) | 3.27 | (4) | - | - | 109.91 | (15) | 73.38 | (7) | 7.53 | (1) | 6.58 | (6) | - | 27.62 | (15) | |||
| 145 | 60.10 | 1834 | 1839 | trans-carveol | - | 2.29 | (7) | - | - | 8.23 | (16) | 11.22 | (9) | 9.89 | (7) | 4.08 | (9) | - | - | |||||
| 149 | 62.13 | 1878 | 1847 | Geraniol 4 | - | - | 9.17 | (1) | 11.90 | (10) | - | - | 16.53 | (8) | 6.74 | (14) | - | - | ||||||
| 152 | 64.72 | 1929 | 1912 | β-Ionone 4 | - | 1.19 | (16) | 1.01 | (8) | 15.77 | (7) | 8.42 | (15) | - | 29.36 | (16) | 84.02 | (2) | 1.27 | (14) | - | |||
| 153 | 64.98 | 1933 | 1927 | Calamenene | - | - | - | - | - | - | 4.86 | (4) | 3.73 | (4) | 2.14 | (16) | 1.98 | (2) | ||||||
| 157 | 68.40 | 1994 | 2009 | Nerolidol 4 | 3.09 | (6) | 2.47 | (14) | - | - | - | - | 2.81 | (12) | 1.96 | (3) | - | - | ||||||
| 158 | 69.19 | 2103 | 2089 | Elemol | - | - | 0.61 | (2) | 0.87 | (2) | 3.05 | (11) | 22.75 | (12) | 18.99 | (7) | 12.15 | (3) | - | - | ||||
| 159 | 71.74 | 2118 | 2113 | Cedrenol | 4.21 | (3) | 6.03 | (4) | 91.13 | (11) | 75.19 | (11) | - | - | 3.69 | (2) | 35.73 | (6) | 2.73 | (8) | 1.19 | (3) | ||
| 162 | 73.21 | 2127 | 2129 | Spathulenol | 3.98 | (1) | 5.33 | (7) | 0.73 | (1) | 20.72 | (7) | - | - | 19.20 | (14) | 1.13 | (13) | - | - | ||||
| % RPA | 32.87 | 21.38 | 17.69 | 20.86 | 53.03 | 51.16 | 16.77 | 19.07 | 24.06 | 10.77 | ||||||||||||||
| Volatile phenols | ||||||||||||||||||||||||
| 165 | 75.82 | 2142 | 2141 | Eugenol 4 | 7.97 | (7) | 1.12 | (13) | 5.01 | (5) | 6.20 | (8) | 10.10 | (1) | 2.98 | (1) | 16.87 | (9) | 2.65 | (12) | 8.54 | (4) | 9.63 | (10) |
| 167 | 76.56 | 2146 | 2170 | 4-Ethyl phenol 4 | - | 1.88 | (14) | - | 1.21 | (2) | - | 15.45 | (16) | - | - | - | 1.16 | (12) | ||||||
| % RPA | 0.61 | 0.34 | 0.17 | 0.22 | 0.33 | 0.57 | 0.63 | 0.09 | 0.28 | 0.35 | ||||||||||||||
| Fatty acids | ||||||||||||||||||||||||
| 76 | 36.53 | 1447 | 1450 | Acetic acid 4 | 34.60 | (5) | 20.77 | (6) | 1.37 | (15) | 7.08 | (8) | 2.45 | (12) | 32.50 | (5) | 7.74 | (11) | 13.49 | (8) | 24.06 | (3) | 12.43 | (6) |
| 143 | 59.21 | 1815 | 1820 | Hexanoic acid 4 | 9.88 | (15) | 2.40 | (16) | 26.48 | (13) | 18.33 | (9) | 68.43 | (13) | 58.96 | (2) | 1.26 | (5) | 2.98 | (3) | 8.16 | (11) | 1.03 | (13) |
| 160 | 72.60 | 2123 | 2083 | Octanoic acid 4 | 15.88 | (18) | 2.65 | (8) | 2.14 | (4) | 12.49 | (13) | 2.73 | (14) | 14.74 | (8) | 2.60 | (10) | 2.07 | (10) | 11.71 | (4) | 5.36 | (7) |
| % RPA | 4.58 | 2.93 | 1.01 | 1.10 | 2.39 | 3.30 | 0.43 | 0.65 | 1.42 | 0.62 | ||||||||||||||
| Furanic compounds | ||||||||||||||||||||||||
| 77 | 37.12 | 1457 | 1455 | 2-Furfural 4 | 3.39 | (1) | 159.04 | (4) | 0.79 | (7) | 194.85 | (7) | 4.67 | (6) | 124.55 | (10) | 1.25 | (1) | 247.37 | (4) | 3.26 | (5) | 197.05 | (4) |
| 93 | 43.71 | 1562 | 1560 | 5-Methyl-2-furfural 4 | - | 2.40 | (6) | - | 2.68 | (6) | - | - | - | 2.73 | (13) | - | - | |||||||
| % RPA | 0.26 | 18.32 | 0.03 | 5.75 | 0.15 | 3.87 | 0.05 | 8.82 | 0.11 | 6.44 | ||||||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perestrelo, R.; Silva, C.; Silva, P.; Medina, S.; Câmara, J.S. Differentiation of Fresh and Processed Fruit Juices Using Volatile Composition. Molecules 2019, 24, 974. https://doi.org/10.3390/molecules24050974
Perestrelo R, Silva C, Silva P, Medina S, Câmara JS. Differentiation of Fresh and Processed Fruit Juices Using Volatile Composition. Molecules. 2019; 24(5):974. https://doi.org/10.3390/molecules24050974
Chicago/Turabian StylePerestrelo, Rosa, Catarina Silva, Pedro Silva, Sonia Medina, and José S. Câmara. 2019. "Differentiation of Fresh and Processed Fruit Juices Using Volatile Composition" Molecules 24, no. 5: 974. https://doi.org/10.3390/molecules24050974
APA StylePerestrelo, R., Silva, C., Silva, P., Medina, S., & Câmara, J. S. (2019). Differentiation of Fresh and Processed Fruit Juices Using Volatile Composition. Molecules, 24(5), 974. https://doi.org/10.3390/molecules24050974