1. Introduction
Mycobacterium tuberculosis (
Mtb), the etiological agent which causes tuberculosis (TB), is the leading cause of infectious death worldwide.
Mtb is transmitted from person to person via aerosol droplets typically by means of coughing. In 2017, there was an estimated 10.0 million new cases of TB reported, and 1.3 million deaths worldwide among human immunodeficiency virus (HIV)-negative individuals [
1]. Typical treatment of active TB includes an extensive antibiotic regimen of isoniazid, rifampin, ethambutol and pyrazinamide, which have specific mechanisms of action in combating
Mtb infection. However, due to the emergence of antibiotic-resistant strains of
Mtb treatment is becoming more problematic [
2]. Therefore, many new treatment options are being explored as possible alternatives or adjunctive therapies to combat this devastating disease. There is currently an urgency to find new treatment procedures for
Mtb infections due to the emergence of multi-drug resistant strains of
Mtb [
1].
Despite substantial advancements attended in this field, diabetic patients are still considerably more likely to contract TB than nondiabetic individuals [
3]. The number of people with diabetes has risen from 108 million in 1980 to 422 million in 2014. Four out of five adults with diabetes live in low- and middle-income countries where the prevalence of TB and other infectious diseases is high. In 2016, an estimated 1.6 million deaths were directly caused by diabetes [
4]. It is commonly accepted that diabetes decreases the effectiveness of cell mediated immunity thus making these individuals more vulnerable to
Mtb infection, but the exact mechanism for
Mtb susceptibility in individuals with T2DM is not entirely well understood [
3].
Polyphenols are a structural class of organic plant-based compounds, characterized by the presence of multiple units and responsible for many natural food pigmentations. Nearly half of the polyphenols are flavonoids, which can be further divided into flavan-3-ols, flavanones, flavones, isoflavones, flavonols, anthocyanins and proanthocyanidins [
5]. The Phenol-Explorer database on polyphenol content in foods contains values for 500 different polyphenols in over 400 foods in the human diet (
http://phenol-explorer.eu/). Recommendations for dietary polyphenol and flavonoid intake have not yet been established. In Europe, the average dietary polyphenol intake has been estimated at 1.187 g/day with coffee, tea, fruits, and wine as the principal sources [
6]. Nearly all ingested polyphenols pass through the small intestine unabsorbed and reach the colon where bacterial degradation produces smaller phenolics that can be reabsorbed into the circulation after undergoing phase 2 conjugation in the liver [
7]. The gut-derived phenolics circulate throughout the body exerting a variety of bioactive effects. Studies support a robust relationship between high dietary polyphenol intake and reduced risks for overall mortality and several chronic diseases, acute respiratory illness, inflammation, and oxidative stress [
7,
8,
9]. Two human studies suggest an inverse relationship between regular intake of flavonoid-rich tea beverages and risk for TB [
10,
11].
Cell culture studies support a strong anti-pathogenic influence from selected flavonoids including quercetin, anthocyanins, and flavan-3-ols [
12,
13,
14]. For example, serum samples collected from athletes that contained metabolites from blueberry and green tea ingestion protected cells from killing by the vesicular stomatitis virus [
12]. Limited evidence indicates that epigallocatechin-3-gallate (EGCG) and quercetin inhibit
Mtb growth and survival within human macrophages [
13,
14]. We sought to extend these findings by measuring the influence of a flavonoid mixture (quercetin, green tea flavan-3-ols, bilberry anthocyanins) on cell death and resolution of infection in a variety of
Mtb infected cells.
3. Discussion
While TB still plagues roughly one third of the world’s population, flavonoids may offer an exciting and inexpensive alternative therapy due to their direct antimycobacterial and immunomodulating effects. Following food and beverage ingestion, small amounts of flavonoids are absorbed in the small intestine after sulfate, glucuronide, and/or methyl conjugation [
7]. Once in the bloodstream, the conjugated flavonoid metabolites can undergo further phase II metabolism in the liver prior to interactions at the cell level and ultimate urinary excretion. Enzymes at the cell level may deconjugate flavonoid metabolites, allowing the more active aglycones to exert beneficial effects as measured in cell culture studies [
7]. The majority of ingested flavonoids pass into the large intestine where they undergo extensive microbial catabolism followed by fecal excretion or reentry into the circulatory system as biotransformed phenolic metabolites [
7]. Some studies indicate that following flavonoid supplementation, serum samples containing a mixture of conjugated flavonoid metabolites and colon-derived phenolics exert anti-pathogenic effects [
12,
15].
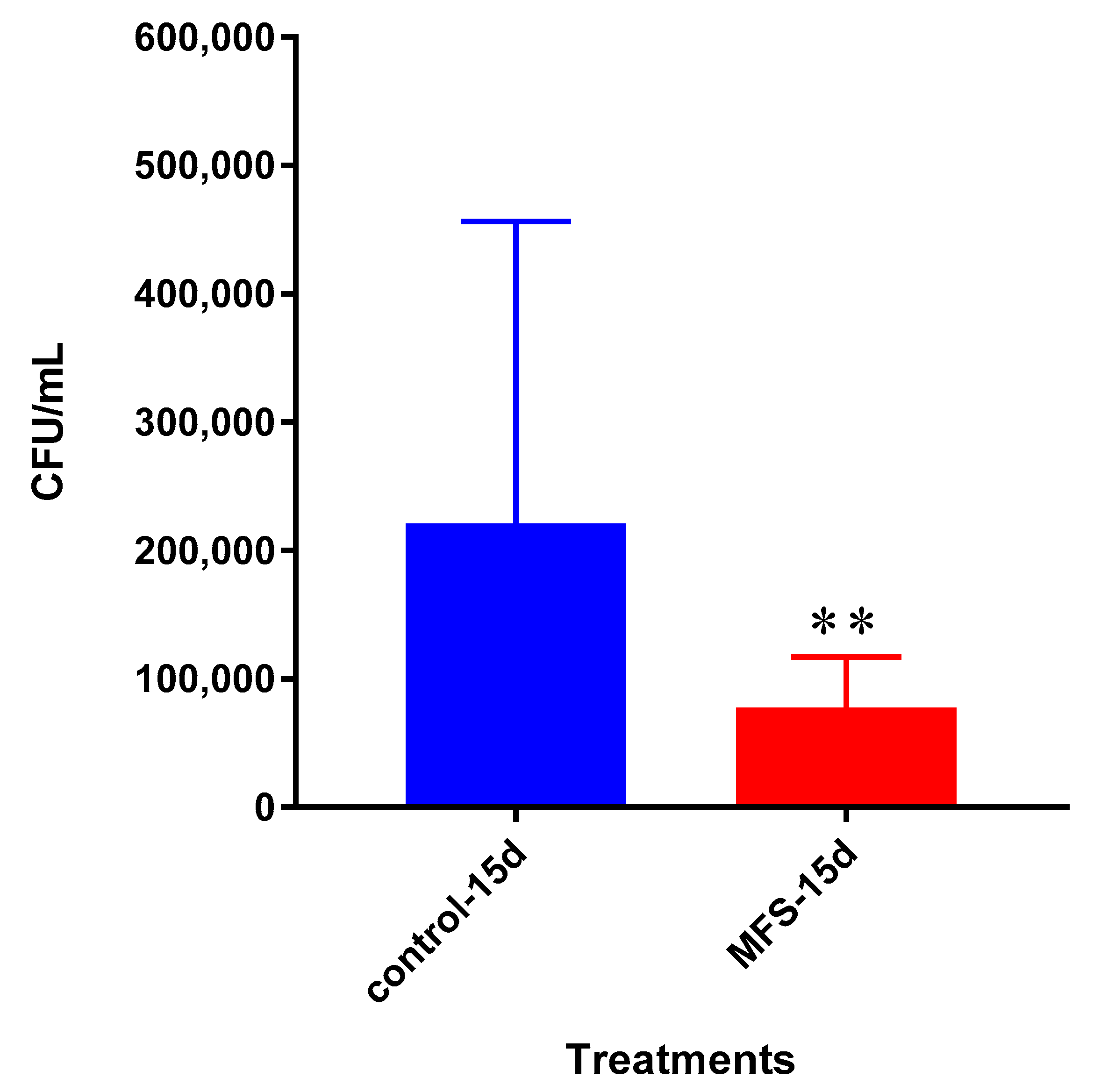
We first measured direct bactericidal influences by adding MFS to
Mtb suspended in 7H9 broth media. After 15 days (
Figure 1), MFS versus control treatment resulted in a reduction in bacterial load (
Figure 1). These results demonstrated that flavonoids exerted direct antibacterial action against
Mtb, although we cannot rule out the potential synergistic effects of the added adjuvants caffeine, vitamin C, and omega 3 fatty acids. This result compares favorably with those from Sasikumar et al. [
13] who showed that quercetin exhibited 99% and 56% inhibition against
Mtb H37Rv at 200 µg/mL and 50 µg/mL, respectively.
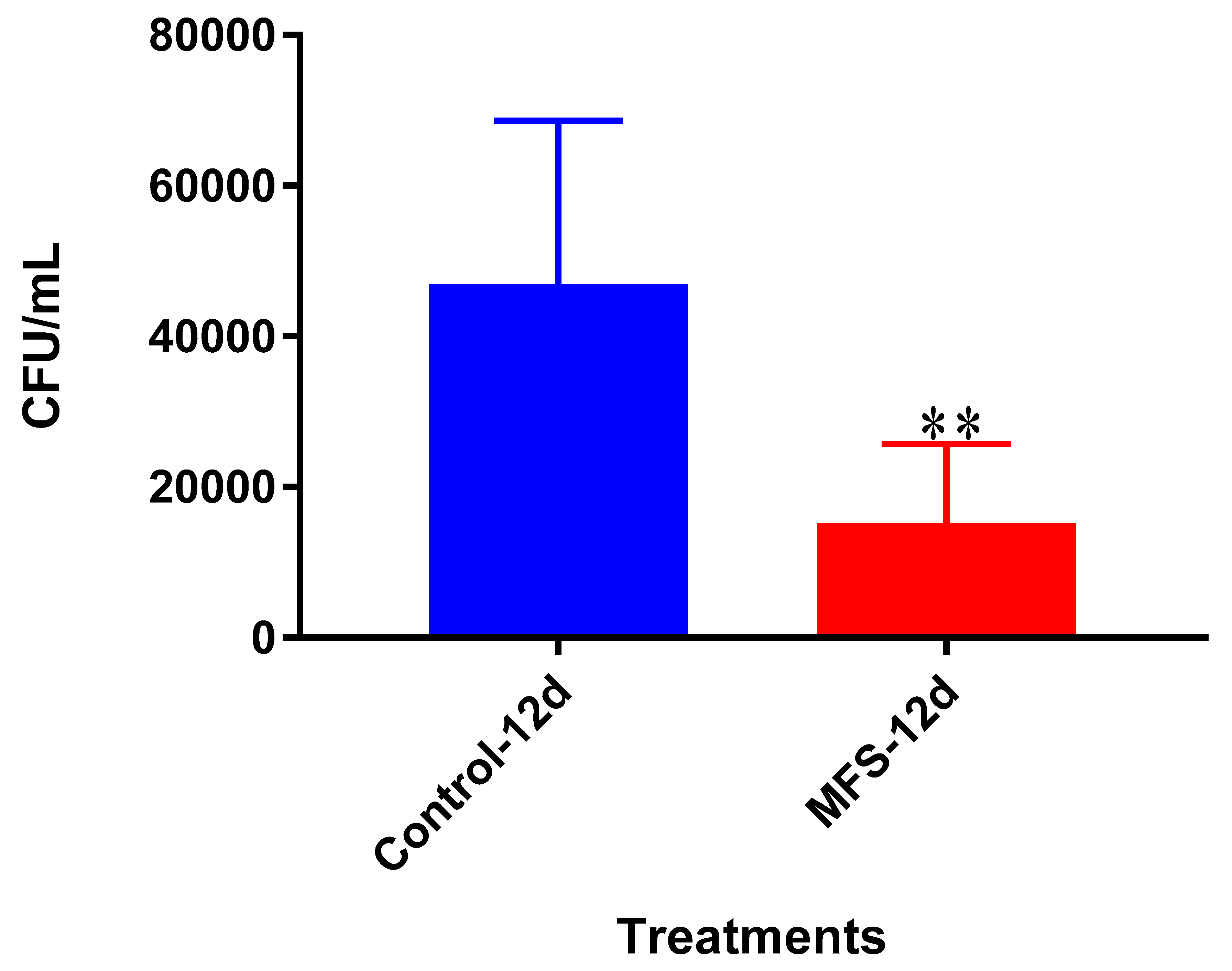
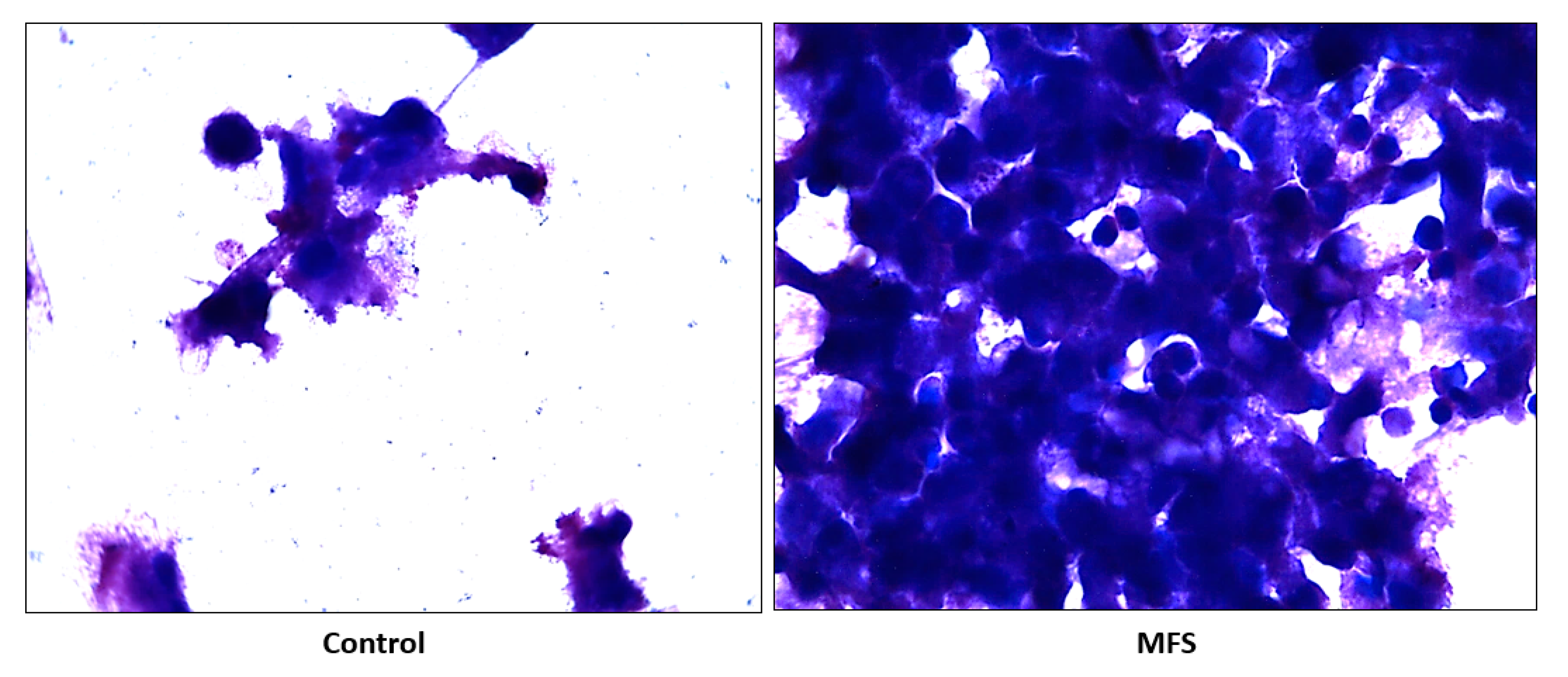
Subsequently, we measured the effects of 12 days of MFS treatment on
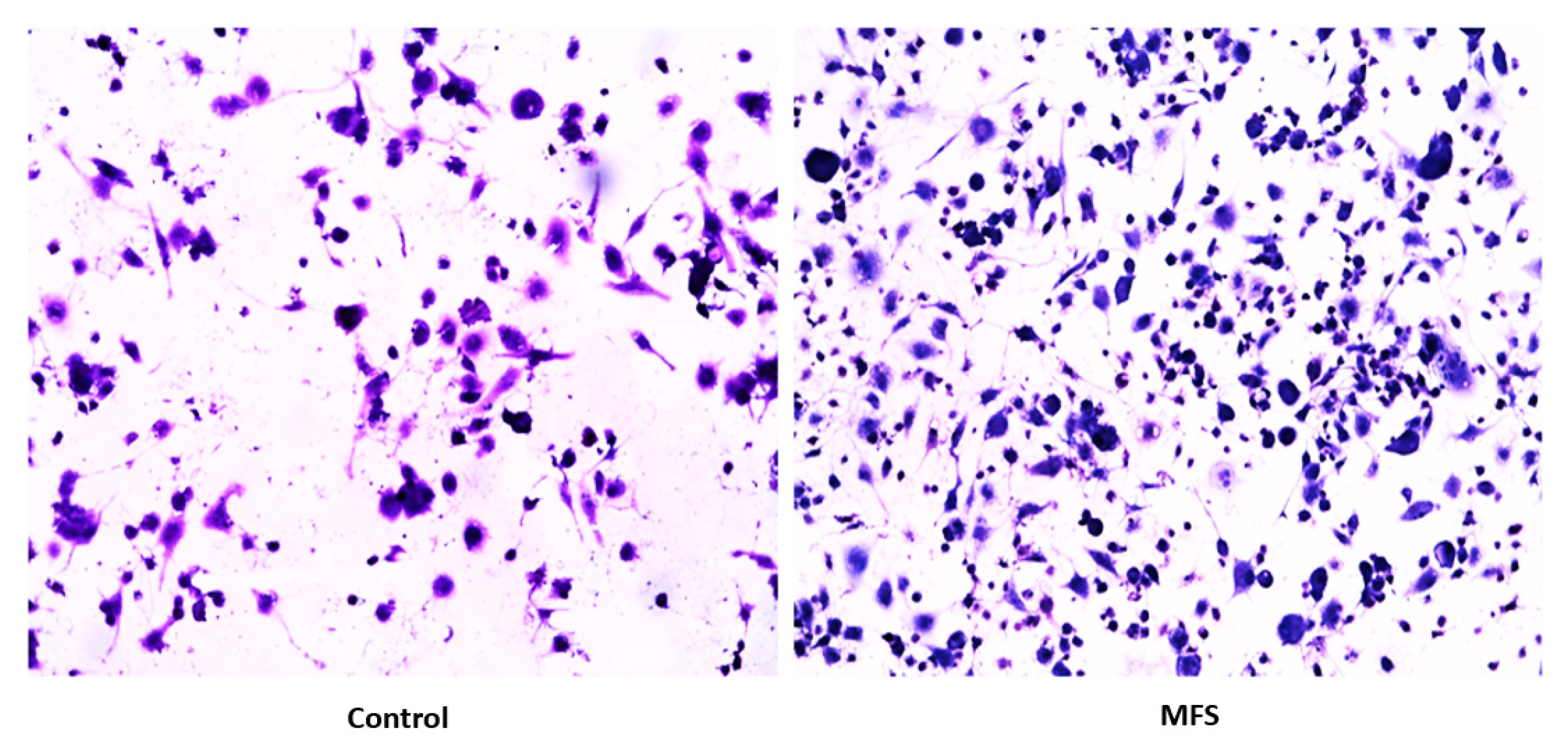
Mtb infected human macrophages derived from THP-1 monocytes. MFS supplementation compared to the control condition over a 12-day period improved the viability of the human THP-1 cells as depicted by the H&E imaging, which portrayed a greater cell magnitude (
Figure 1). This suggested that
Mtb infection became too vigorous for the THP-1 cells to retain and secure in the absence of MFS prophylactic treatment. This conclusion is further supported by the data represented in
Figure 2, which illustrates that after 12 days MFS treatment resulted in a statistically significant bacterial reduction compared to the sham-treated control. These results support the findings of Anand et al. [
14] who showed that the green tea flavonoid EGCG inhibited
Mtb survival within macrophages. This research group showed that EGCG down-regulated host molecule tryptophan-aspartate containing coat protein (TACO) gene transcription, thus influencing phagosome maturation and the capacity to contain
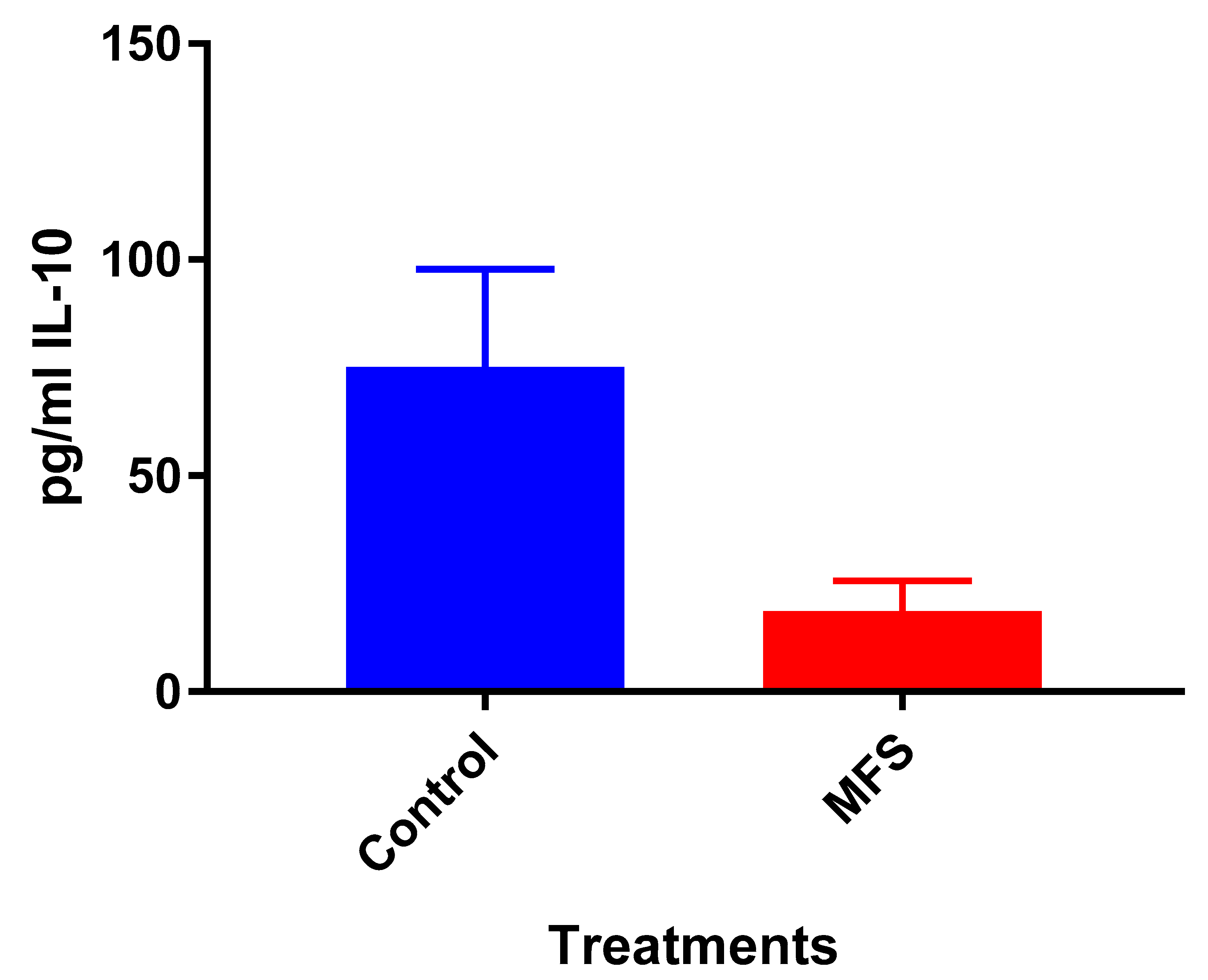
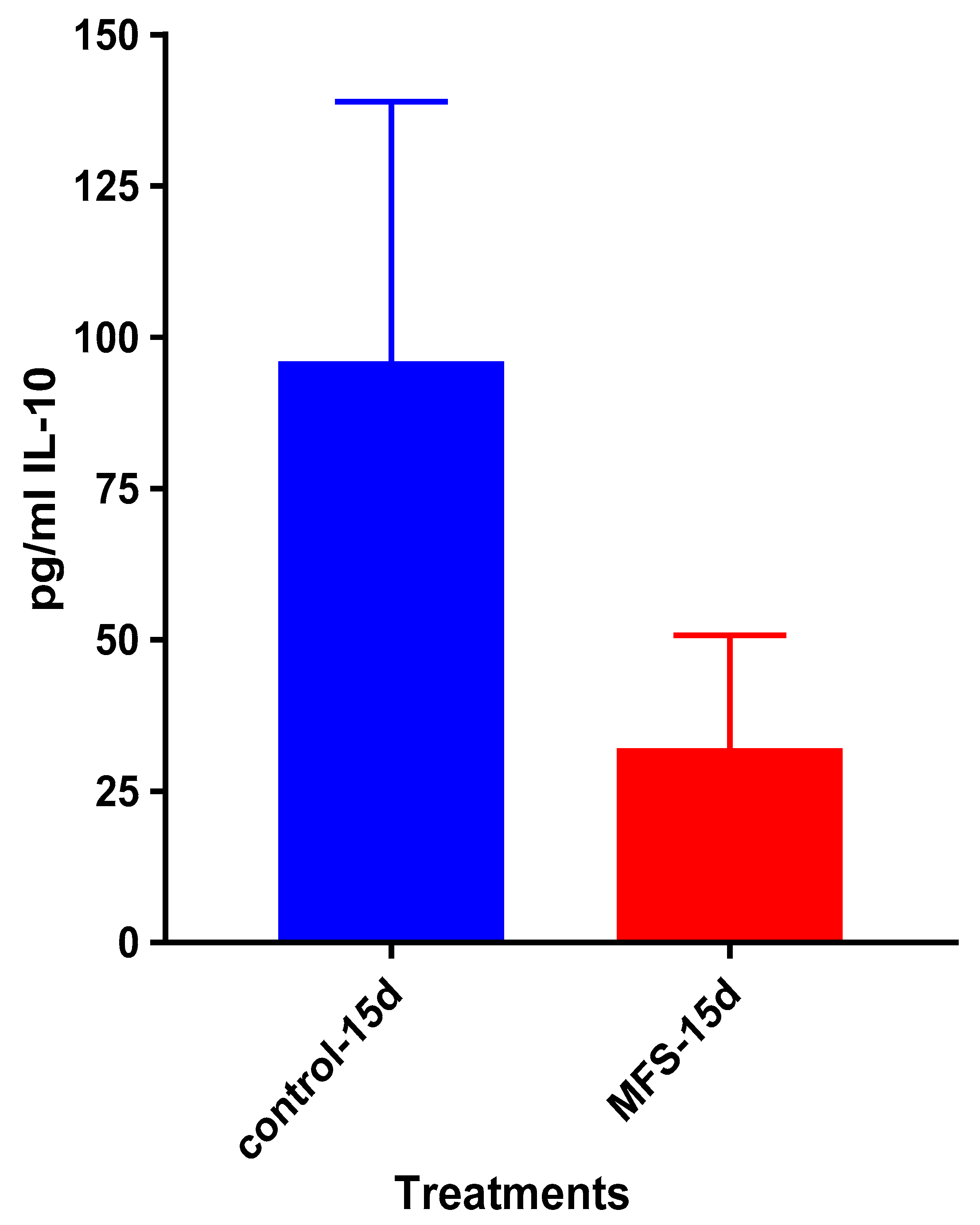
Mtb.Of the three cytokines measured in this study, IL-10 was most effected by MFS treatment with levels half that of the control condition (
Figure 4). IL-10 is an immunosuppressive cytokine which down regulates the expression of Th1 cytokines, NF-kB activity, and macrophage costimulatory molecules [
16]. Thus, in the context of a
Mtb infection, IL-10′s inhibition subsequent to MFS administration is considered to be favorable.
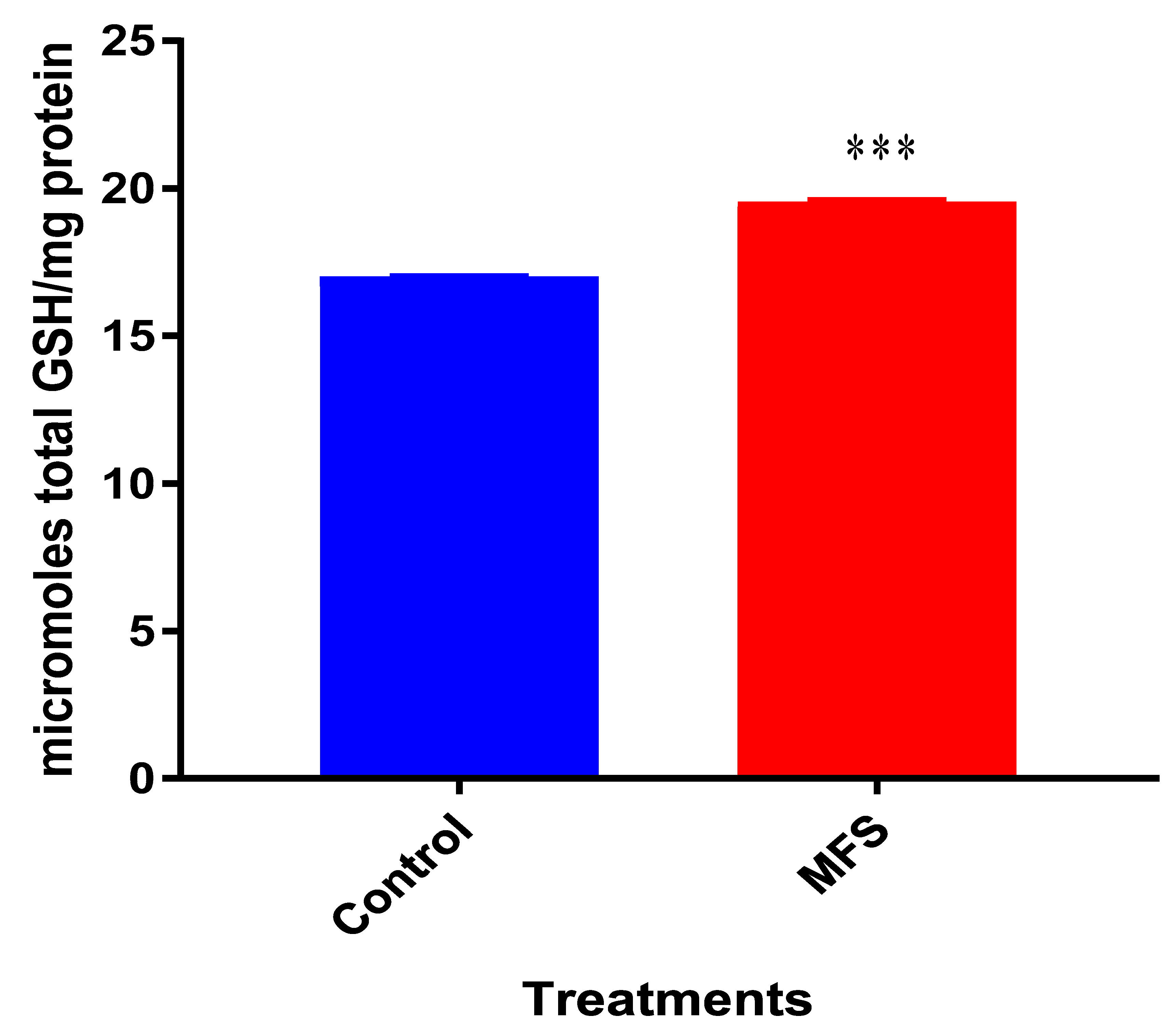
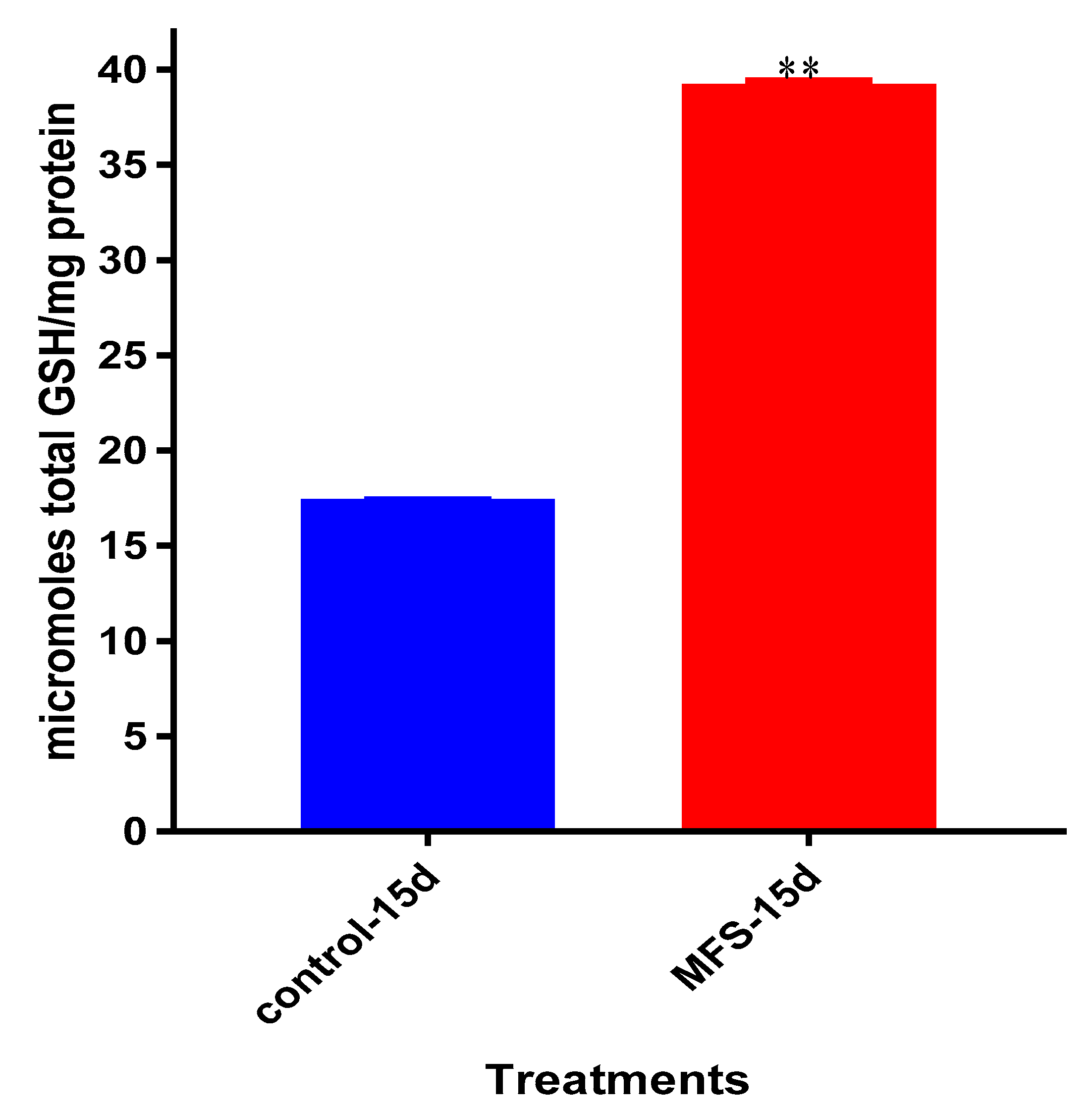
Interestingly, the levels of detectable intracellular GSH were elevated following MFS treatment. Our lab has previously demonstrated that among
Mtb infected cells, the augmentation of GSH will cause mycobactericidal effects in both a direct and immunomodulating indirect manner through downstream mediators [
17,
18]. Therefore, MFS treatment of THP-1 cells can diminish
Mtb infectivity in direct and indirect pathways.
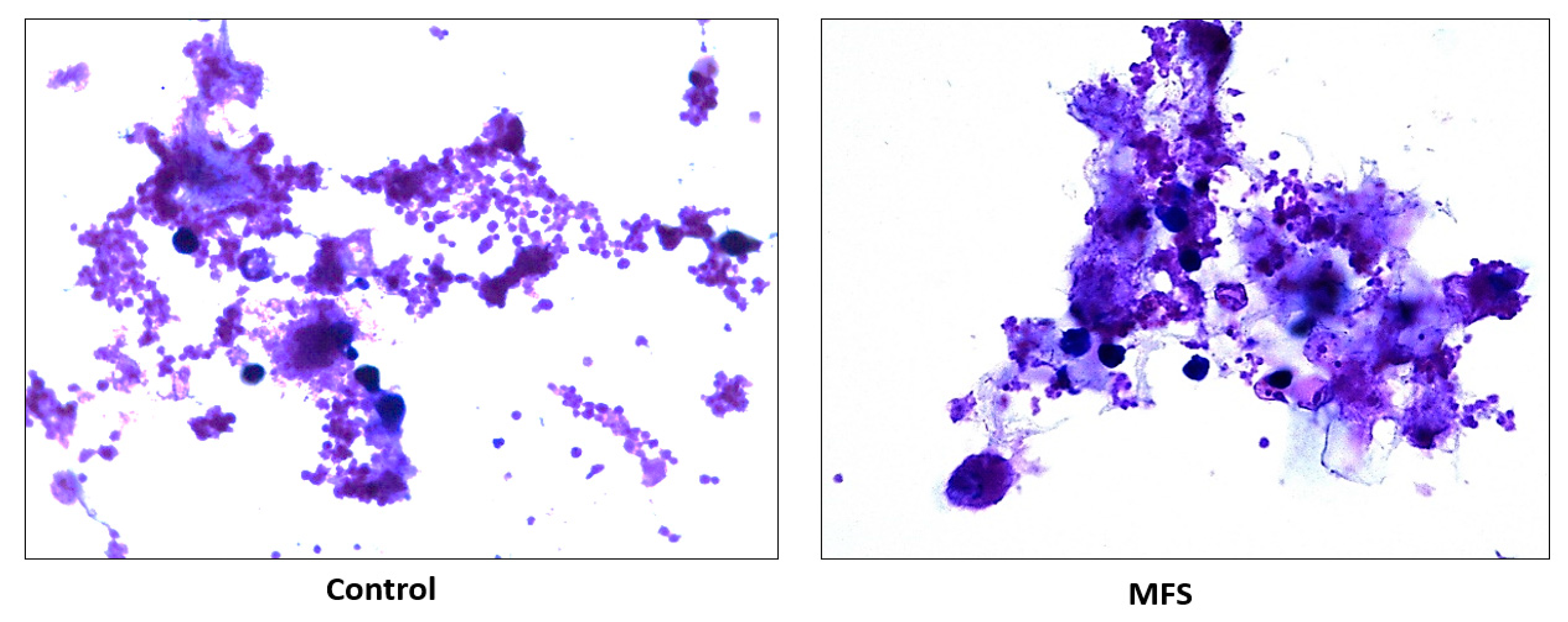
The influence of MFS on
Mtb infected PBMCs collected from eight healthy individuals was similar to the findings from the THP-1 macrophage assays. The H&E data showed enhanced PBMC cell viability, cell aggregation/density, and enriched granuloma formation after MFS treatment (
Figure 6). This is significant because augmented granuloma development is linked to increased bacterial containment and improved host protection [
19]. As with the THP-1 macrophages, the increased immune cell viability observed subsequent to MFS treatment coincided with a strong reduction in bacterial density among the infected PBMCs (
Figure 2 and
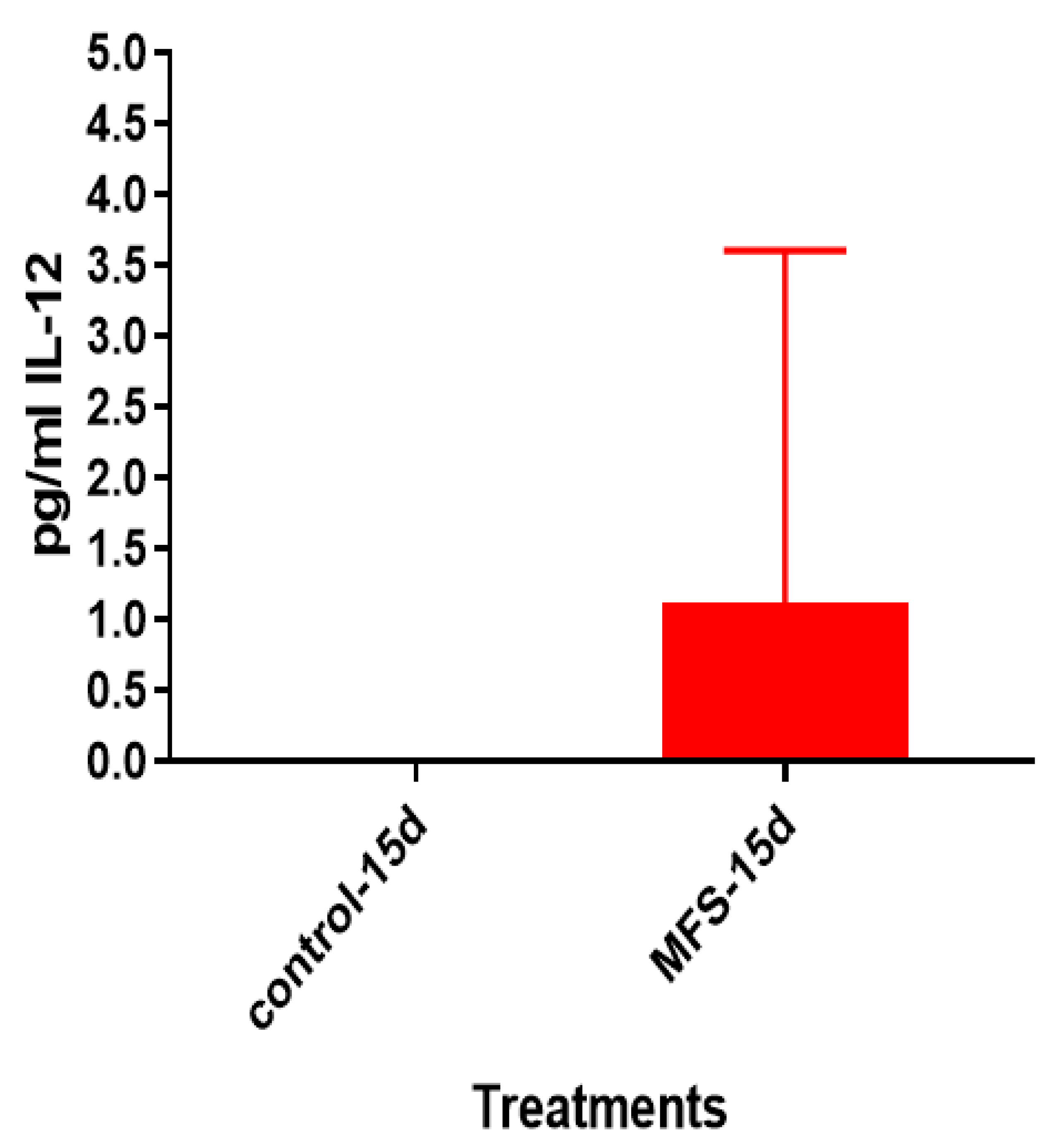
Figure 6). Taken together with the GSH and cytokine data, these results support direct MFS-related mycobactericidal effects and immunomodulation from granuloma formation and the trend for increased IL-12 and IFN-γ cytokine production (
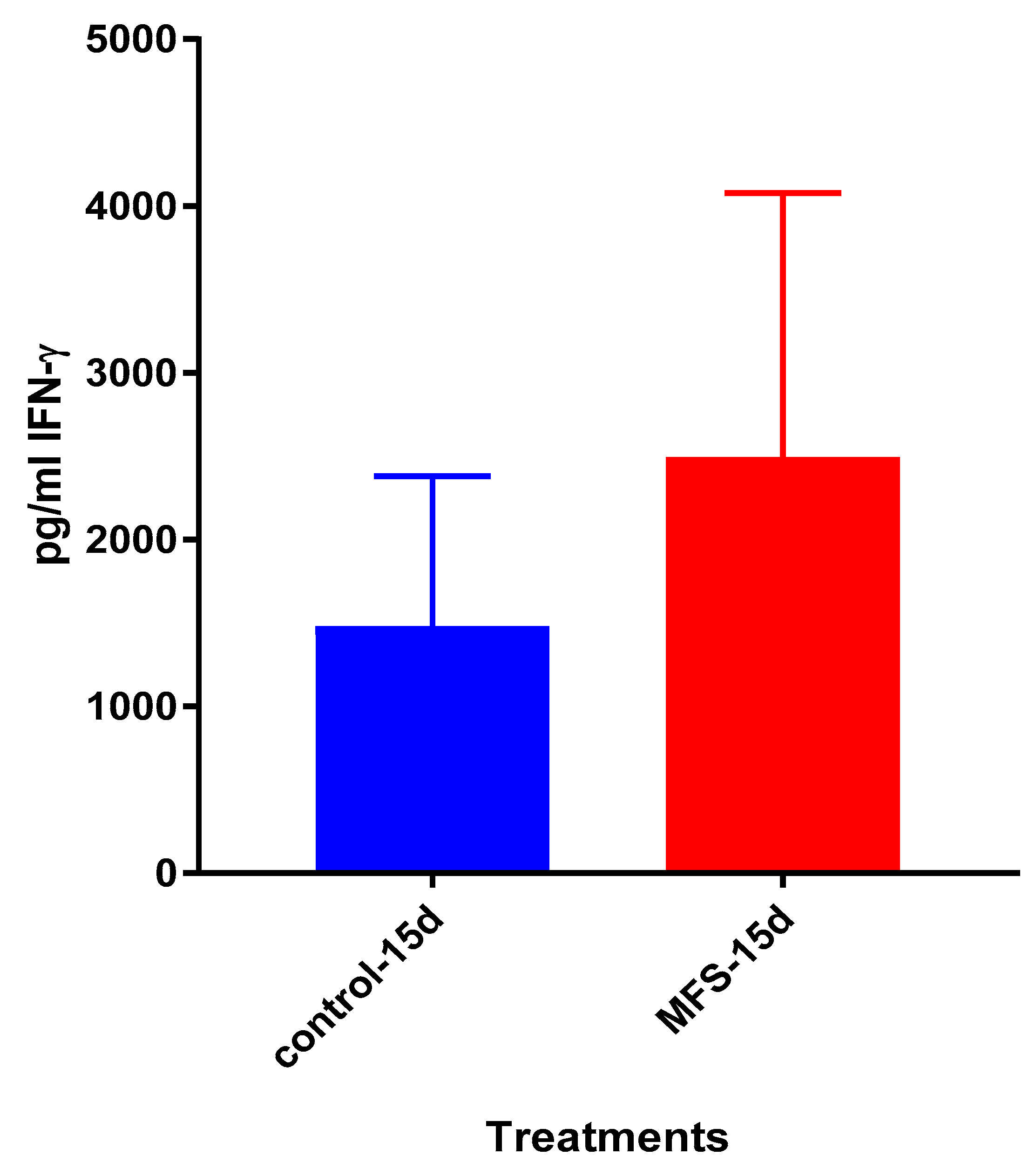
Figure 8,
Figure 9,
Figure 10 and
Figure 11). Both IL-12 and IFN-γ are prominently involved in immune cell differentiation and activation in response to combating
Mtb infection, a process linked to granuloma formation [
20].
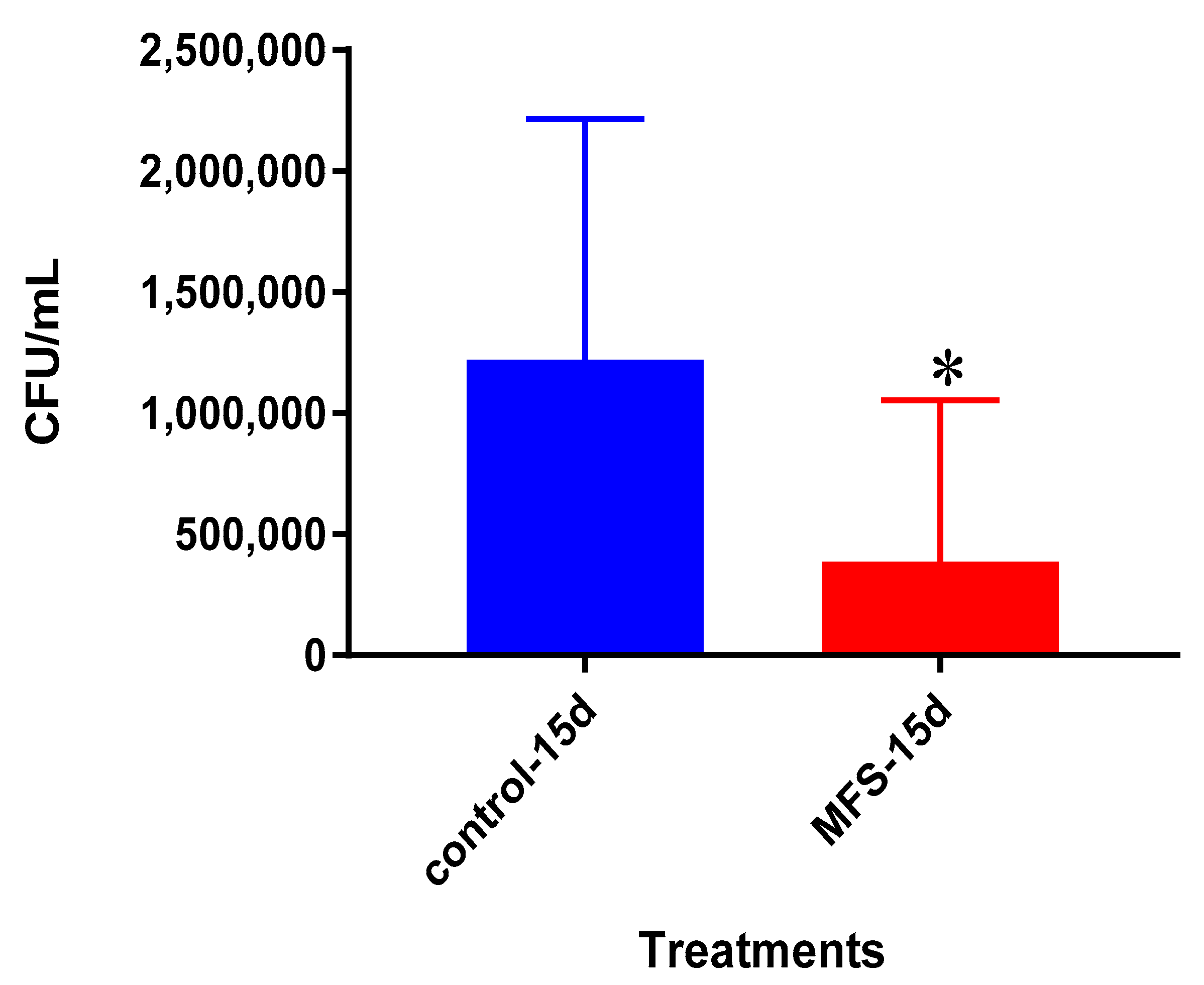
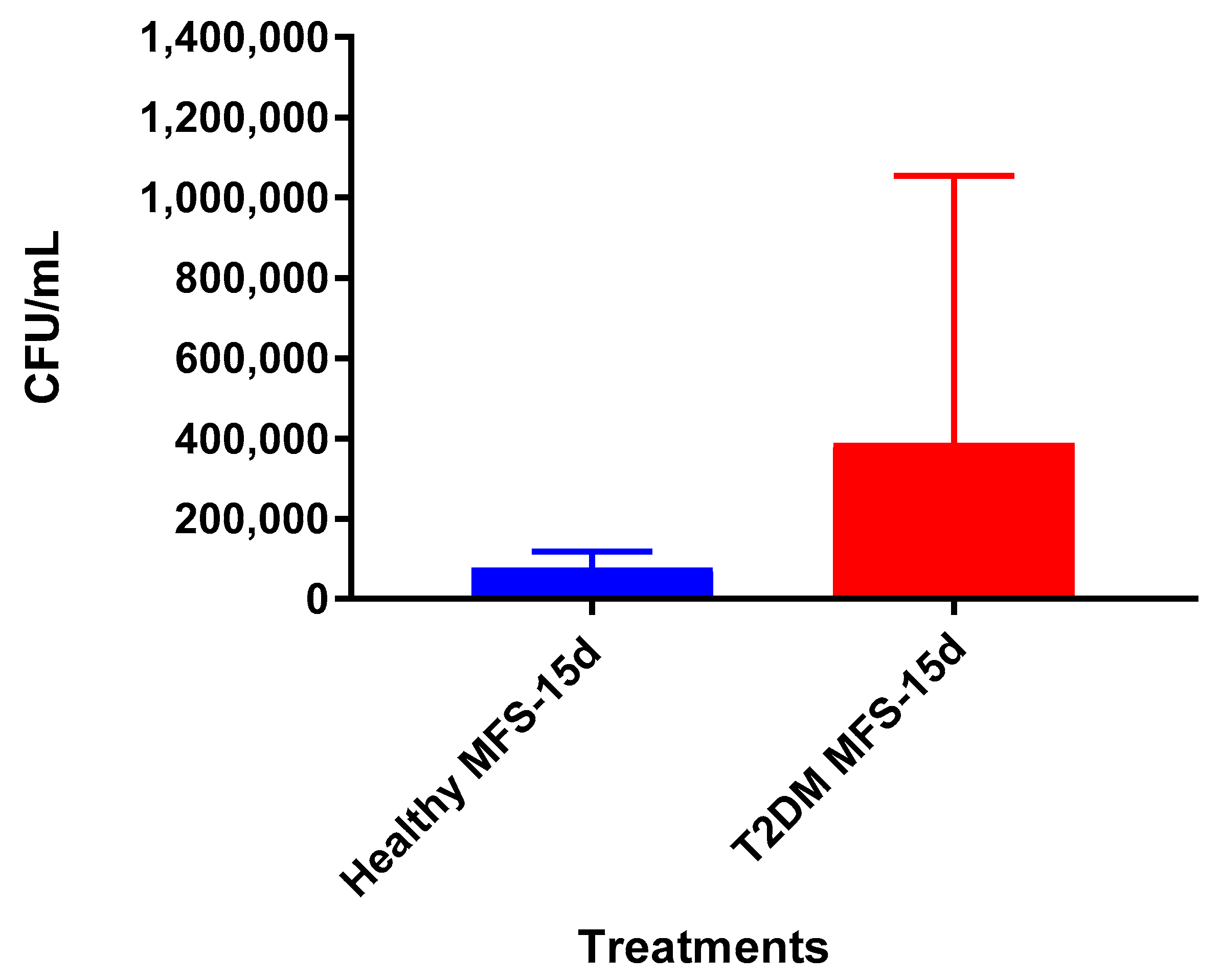
Lastly, we tested the influence of MFS treatment on PBMCs collected from individuals with T2DM, a patient population that is significantly more susceptible to
Mtb infections [
1]. Consistent with findings from the healthy individuals, PBMCs of T2DM patients treated with MFS exhibited more robust cellular aggregations and granuloma formation (
Figure 12). Correspondingly, after MFS administration the PBMC bacterial burden was reduced to roughly one-third that of the T2DM infected control (
Figure 13). Although the abundance of
Mtb was significantly diminished with MFS treatment, the quantity of bacteria remaining was still over double that of levels linked to the same experiments in healthy study participants (
Figure 14). This demonstrates that while MFS supplementation reduced
Mtb infectivity in both healthy individuals and those with T2DM, it is not sufficient to cause complete
Mtb elimination.
4. Materials and Methods
4.1. THP-1 Cell Culture
The THP-1 cell line originated from the American Type Culture Collective (ATCC), was cultured in Roswell Park Institute (RPMI) medium (Sigma, St. Louis, MO, USA) with 10% Fetal bovine serum (FBS-Sigma) and incubated at 37 °C with 5% CO2. After 7–12 days, cells were collected from the flasks, spun at 2000 rpm for 15 min, resuspended in RPMI containing 10% FBS and counted for cell numbers. THP-1 cells (2 × 105/well) were distributed in 0.001% poly-lysine (Sigma, St. Louis, MO, USA) coated 24-well tissue culture plates and incubated overnight. Differentiation of THP-1 cells to macrophages was achieved by adding PMA (Phorbol 12-myristate 13-acetate-Sigma) at a concentration of 10 ng/mL.
4.2. Culture of Mtb Erdmann
Mtb Erdmann expressing green fluorescent protein (GFP) was obtained as a gift from Dr. Selvakumar Subbian at Rutgers New Jersey Medical School, Biomedical and Health Sciences.
Mtb Erdmann is similar to H37Rv (standard laboratory strain of
Mtb) however it is considered to be more virulent due to its faster doubling time [
21].
Mtb Erdmann was used for all experiments conducted in this study, handled inside the biosafety level 3 facility (BSL-3) and cultured in Difco Middlebrook 7H9 broth media supplemented with ADC (Albumin Dextrose Catalase) at 37 °C. Mtb Erdman was processed for infection once the static culture was at the peak logarithmic phase of growth (optical density at 600 nm, between 0.5 and 0.8) and subsequently washed and resuspended in sterile 1× phosphate-buffered saline (PBS). The Mtb Erdman was then processed to disaggregate any clumps by vortexing five times with 3-mm sterile glass beads at 3 min intervals.
Mtb Erdman suspension was then filtered using a 5-μm syringe filter to remove any remaining bacterial aggregations. The single cell suspension of now processed
Mtb was then serially diluted and plated on 7H11 agar (Hi Media, Santa Maria, CA, USA) to determine the bacterial concentration of the processed stock. Aliquots of processed bacterial stocks were then frozen and stored in the cryogenic freezer at −80 °C. At the time of the experimental trial, the frozen-processed stocks of
Mtb were thawed and used for the infection.
4.3. Subject Recruitment
Study subjects were recruited after obtaining signed informed consent. The protocols for all the studies pertaining to in vitro Mtb infection were approved by the Institutional Biosafety committee of Western University of Health Sciences. The protocols for the study involving healthy subjects and participants with T2DM were approved by the Institutional Review Board of Western University of Health Sciences. Healthy adults (N = 8, ages 21 to 28 years, no known diseases or medication use) and individuals with T2DM (N = 5, ages 51 to 74 years, hemoglobin A1C > 7%) were recruited after obtaining signed informed consent. Exclusion criteria included the use of MFS within the last 6 months, or history of chemotherapy treatment within the last year. Other exclusion criteria included women who were currently pregnant, lactating, or had been pregnant within the last 6 months; pregnancy was considered a reason for study termination. After signing the informed consent form, 40 mL of blood was drawn from each participant.
4.4. Flavonoids
The mixed flavonoid supplement (MFS) was prepared by Reoxcyn LLC (Pleasant Grove, UT, USA). Supplement ingredients (US Patent 9,839,624) provided (in one tablet) 82.3 mg total monomeric flavonoids and included 25 mg vitamin C (as ascorbyl palmitate) (Green Wave Ingredients, La Mirada, CA, USA), wild bilberry fruit extract with 16 mg anthocyanins
(FutureCeuticals, Momence, IL, USA), green tea leaf extract with 46 mg total flavan-3-ols (Watson Industries, Inc., Pomona, CA, USA), 26 mg quercetin aglycone (Novel Ingredients, East Hanover, NJ, USA), 26.8 mg caffeine (Creative Compounds, Scott City, MO, USA), and 15 mg omega 3 fatty acids (Novotech Nutraceuticals, Ventura, CA, USA). As previously reported, the tablet contents were analyzed prior to the study for flavonoid content using high-performance liquid chromatography (HPLC) [
22]. Thirteen anthocyanins were identified in the bilberry extract including delphinidin, cyanidin, petunidin, peonidin, and malvidin galactosides, glucosides, and arabinsosides. The supplement tablets were analyzed again after 12 weeks and 12 months of storage at room temperature, and the data indicate that all chemical components were stable. As previously described, vitamin C, caffeine, and omega 3 fatty acids were included as adjuvants to improve flavonoid bioactivity [
22]. For in vitro administration, the flavonoid tablet (138 mg) was dissolved in 1 mL of 100% DMSO. Once dissolved, the solution was diluted with 9 mL of RPMI. This method brought the stock concentration to 13.8 mg of dissolved flavonoid tablet per mL. We used 25 uL of this solution per each 500 uL well, making the final concentration 0.69 mg of flavonoids per mL.
4.5. Determination of the Direct Antimycobacterial Effects of MFS
To determine the direct effects of MFS in altering the survival of Mtb Erdman, bacteria (6 × 104 /well) were grown in 7H9 media in 24 well tissue culture plates (Corning, NY, USA) in the presence and absence of MFS up to 15 days. Mtb Erdman cultures were then either sham-treated (control) or treated with the MFS at a concentration of 0.69 mg/per mL. The MFS treatment groups received their MFS supplementation every 4 days until termination. Mtb cultures were maintained at 37 °C, with 5% CO2 until they were terminated at 8 and 15 days post-infection to determine the viability of Mtb. Mtb viability was ascertained by plating the diluted samples on 7H11 agar medium (Hi Media, Santa Maria, CA, USA) enriched with albumin dextrose complex (ADC) (Gemini, West Sacramento, CA, USA).
4.6. Isolation of Plasma and Monocytes
Plasma and peripheral blood mononuclear cells (PBMCs) were isolated from the whole blood of T2DM subjects and healthy individuals by Ficoll-Paque (Sigma) density centrifugation. This procedure involves centrifugation of blood layered on Ficoll-Paque at a 1:1 ratio at 1800 rpm for 30 min. Plasma (the top layer) was collected and stored at −80 °C, while PBMCs (the second layer from the top) were further washed three times with 1× phosphate-buffered saline (PBS) from Fisher Scientific International Inc. (pH 7.4 ± 0.1) and then resuspended in RPMI containing L-glutamine and 5% human AB serum.
4.7. Induction of Granulomas
PBMCs were plated on a 24-well tissue culture plate, precoated with 0.001% poly-l-lysine, and infected with Mtb at a multiplicity of infection (MOI) of 0.1:1 bacteria-to-cell ratio (approximately 6 × 104 bacteria were added to 6 × 105 PBMCs). Infected cells were maintained at 37 °C with 5% CO2 for up to 15 days. Infected PBMCs were then either sham-treated or treated with 0.69 mg of MFS every 4 days until termination. Granulomas were terminated at 8 days and 15 days post-infection to determine the effects of the MFS in altering the granulomatous responses against Mtb infection.
4.8. Cell Termination and Determination of Mtb Erdman Survival
Cell free supernatants from each well were first collected and stored, the cells remaining were then harvested by adding 250 μL of ice-cold, sterile 1× PBS (Sigma, St. Louis, MO, USA) followed by gentle scraping to achieve maximum recovery of cell lysate from the wells. Lysates were then efficaciously vortexed followed by a freeze/thaw cycle in order to ensure sufficient rupture of cells and release of intracellular Mtb. The collected lysates and supernatants were then diluted as necessary in sterile 1× PBS (Sigma, St. Louis, MO, USA) and plated on 7H11 agar medium (Hi Media, Santa Maria, CA, USA) enriched with ADC (GEMINI, West Sacramento, CA, USA) and incubated at 37 °C for 3 weeks in order to evaluate the mycobacterial growth or survival subsequent to MFS treatment by counting the colony forming units (CFUs).
4.9. Hematoxylin and Eosin Staining
Granulomas on cover-glasses terminated at 15 days post-infection were fixed with 4% Paraformaldehyde (PFA) at room temperature for an hour. Fixed granulomas were washed once with 1x PBS and stained with Rapid H&E (Scientific R&D Corp, NY, USA) for 2 min and the excess stain was washed away with tap water. The cover glasses were inverted and mounted onto slides with mounting media (HistoChoice, Solon, OH, USA).
4.10. Quantification of GSH Levels
Measurement of total and oxidized glutathione was performed using the GSH colorimetric assay kit from Arbor Assays (Ann Arbor, MI, USA). Granuloma lysates were first comprehensively mixed with an equal volume of cold 5% sulfosalicylic acid (SSA), followed by incubation for 10 min at 4 °C, and subsequently centrifuged at 14,000 rpm for 10 min. The GSH was thereupon measured in the supernatants following the manufacturer’s instructions. Reduced GSH (rGSH) was calculated by subtracting the oxidized glutathione disulfide (GSSG) from the total GSH. All measurements were corrected for total protein levels.
4.11. Quantification of Total Protein Levels
Total protein was measured with a BCA Protein Assay Kit and performed per user instructions from Thermo Scientific (Rockford, IL, USA).
4.12. Assay of Cytokines
Levels of IL-10, IL-12, and IFN-γ, from the supernatants of THP-1 cells, and granulomas derived from PBMCs of healthy subjects and individuals with T2DM were measured by sandwich ELISA. The assay kits were purchased from eBioscience (San Diego, CA, USA) and performed as per the manufacturers’ protocol.
4.13. Statistical Analysis
Statistical data analyses were performed using GraphPad Prism version 7 (San Diego, CA, USA). Baseline levels of GSH, rGSH, MDA, IL-6, IL-10, IL-12, IFN-γ, and TNF-α were compared between healthy individuals and T2DM group using the unpaired t-test with Welch correction. Reported values are in means. Statistical significance was determined at * p < 0.05, ** p < 0.005, *** p < 0.0005.