Antioxidant Activities of Vaccinium vitis-idaea L. Leaves within Cultivars and Their Phenolic Compounds
Abstract
:1. Introduction
2. Results
2.1. Antioxidant Activity of Lingonberry Leaves Extracts
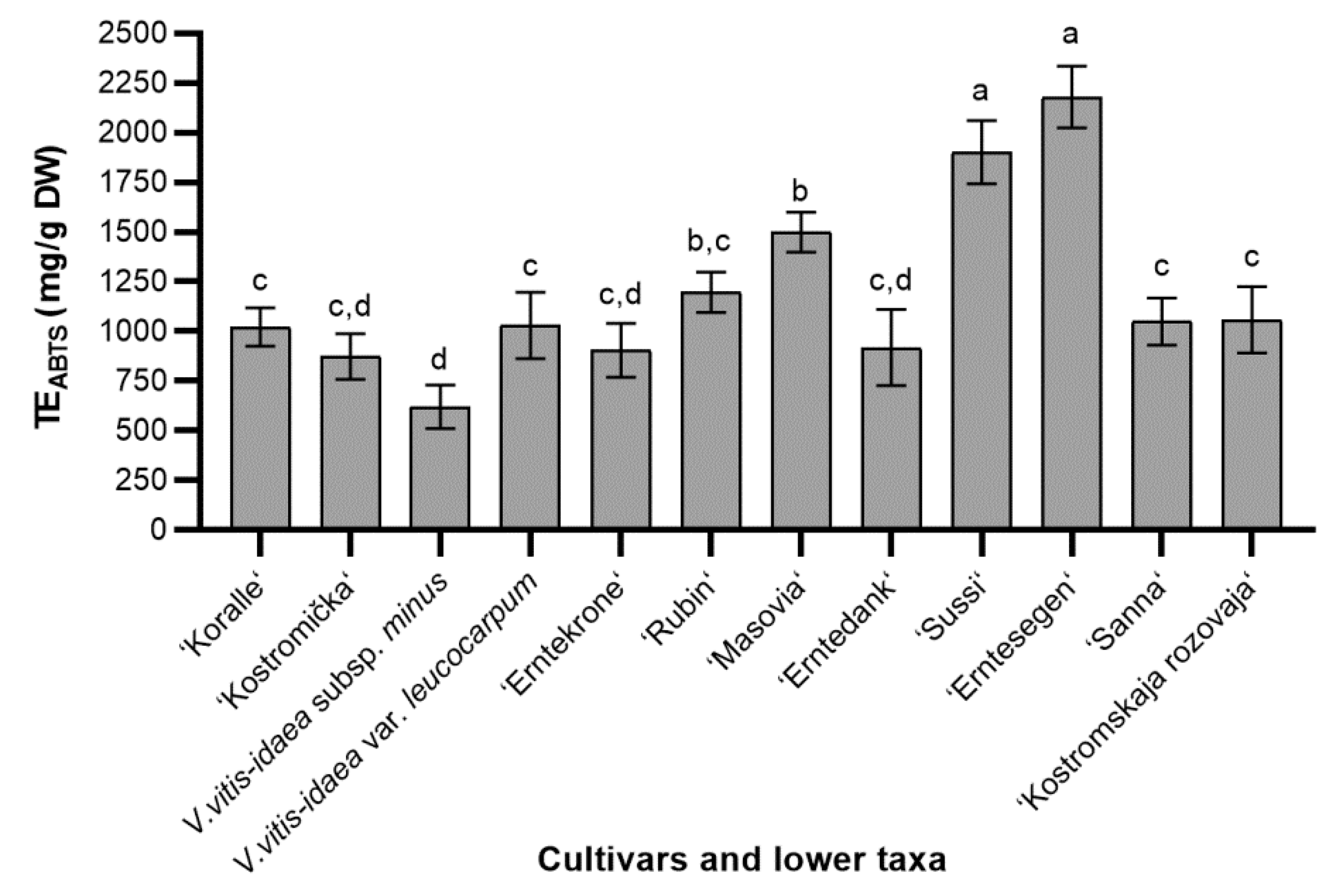
2.1.1. ABTS Radical Scavenging Activity
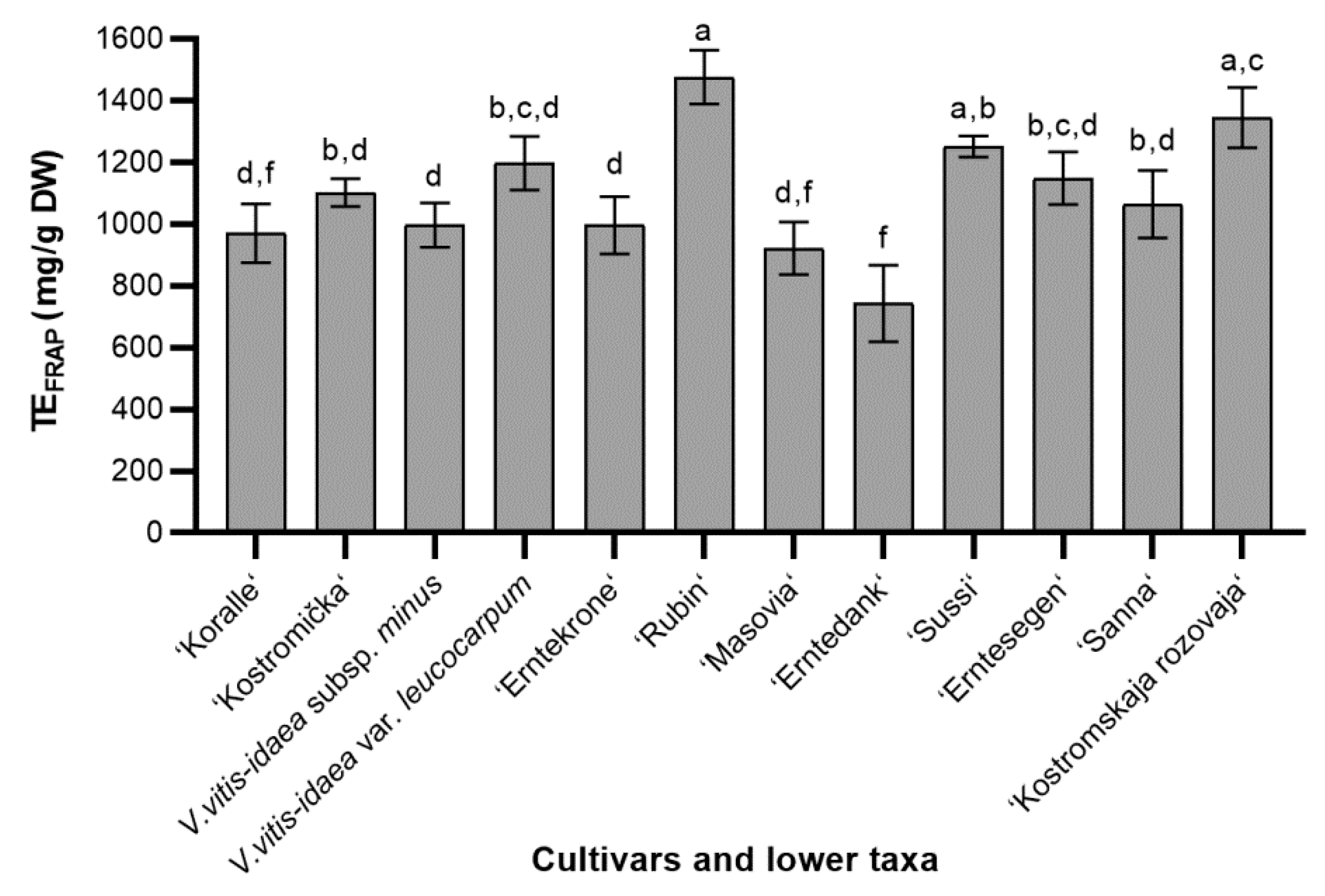
2.1.2. Ferric Reducing Antioxidant Power (FRAP)
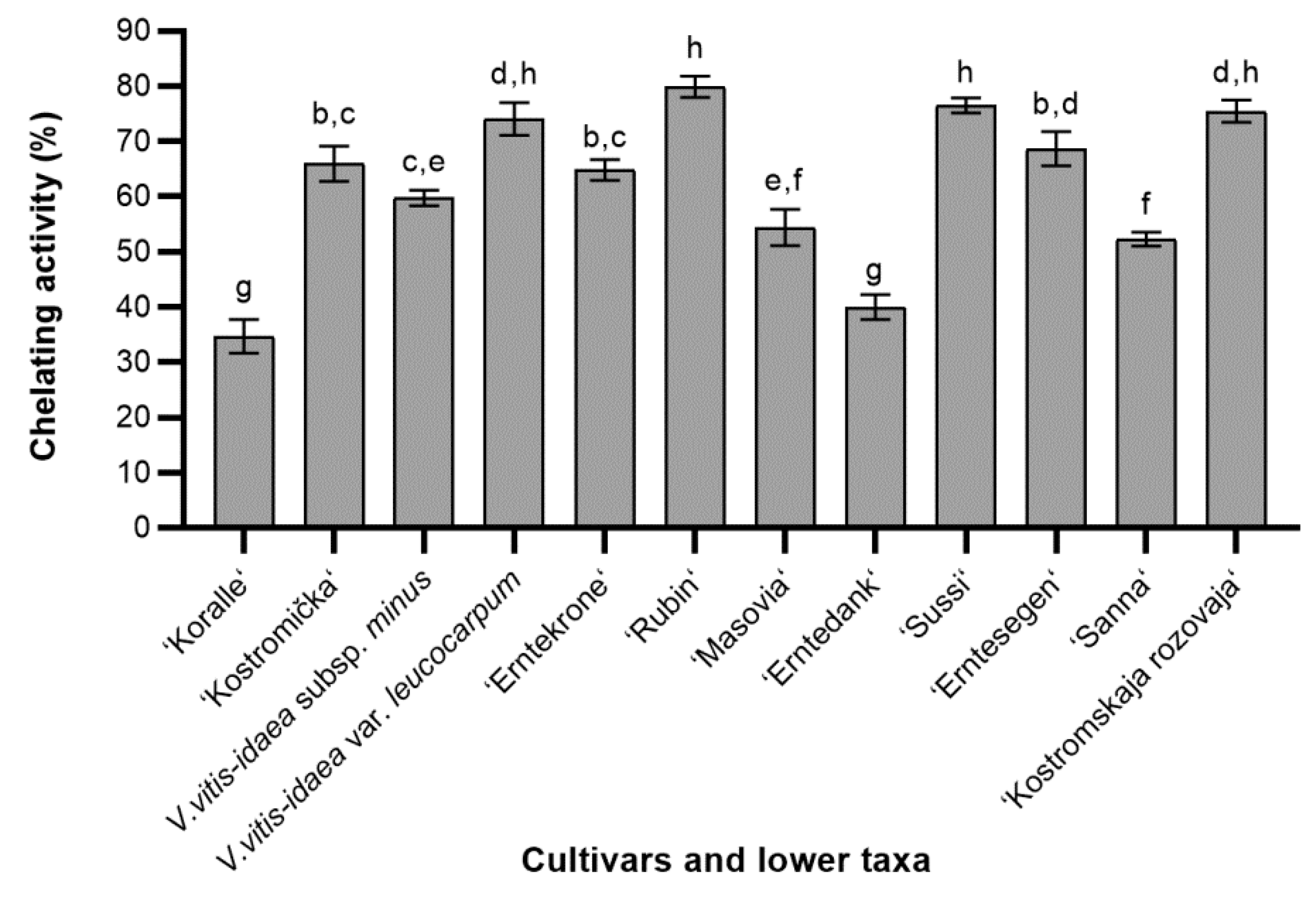
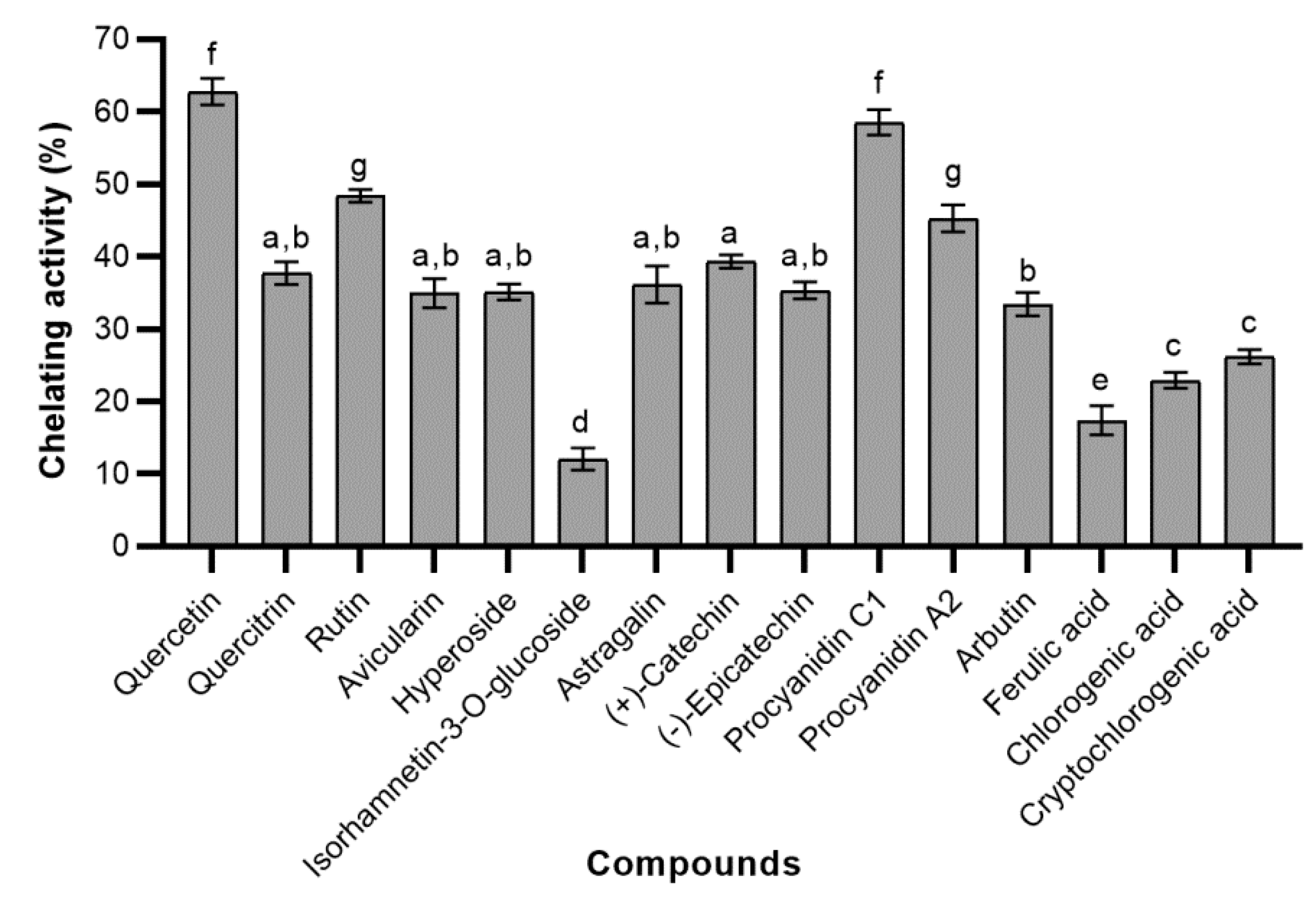
2.1.3. Ferrous Ions Chelating (FIC) Activity
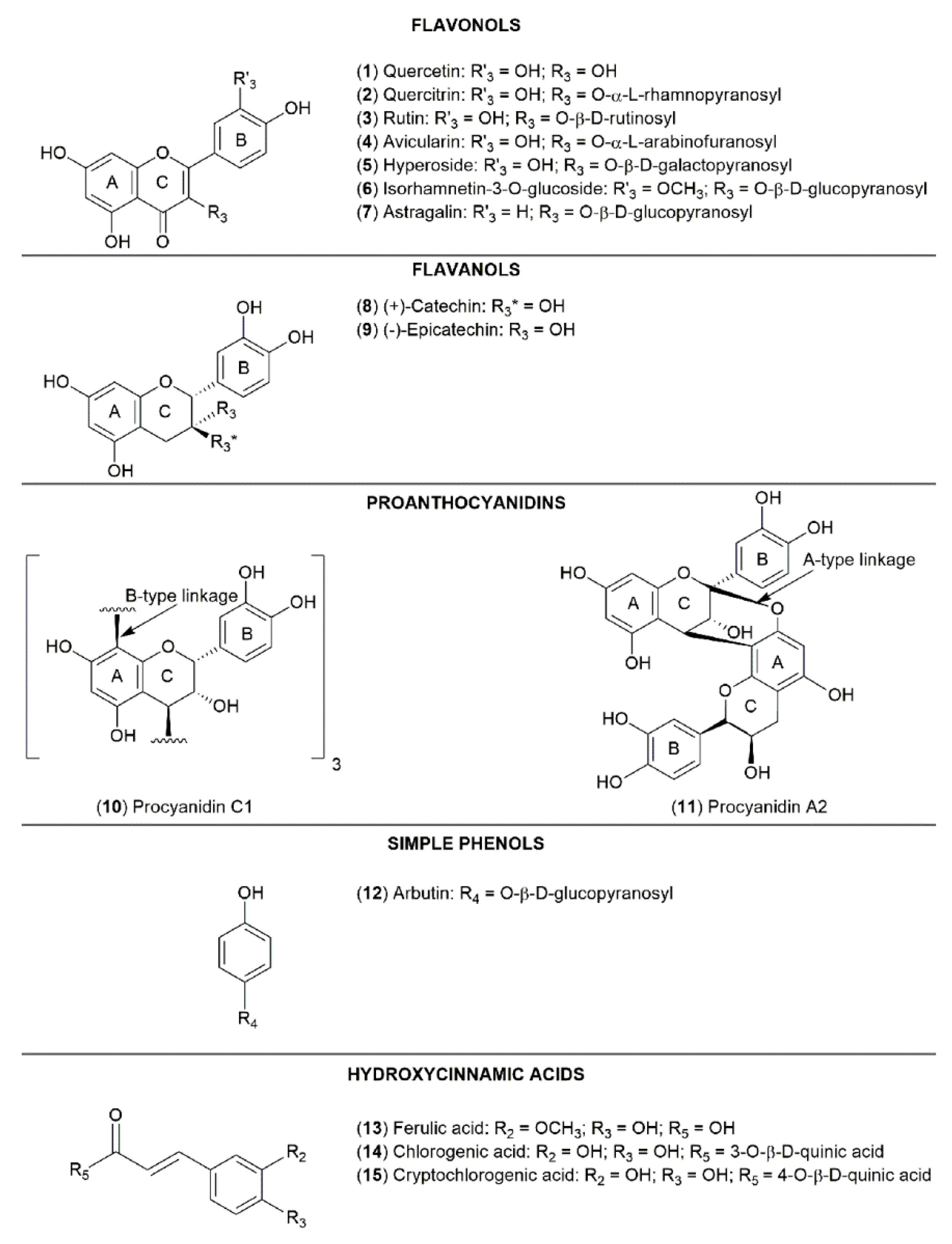
2.2. Qualitative and Quantitative Analysis of Phenolic Compounds in Lingonberry Leaves
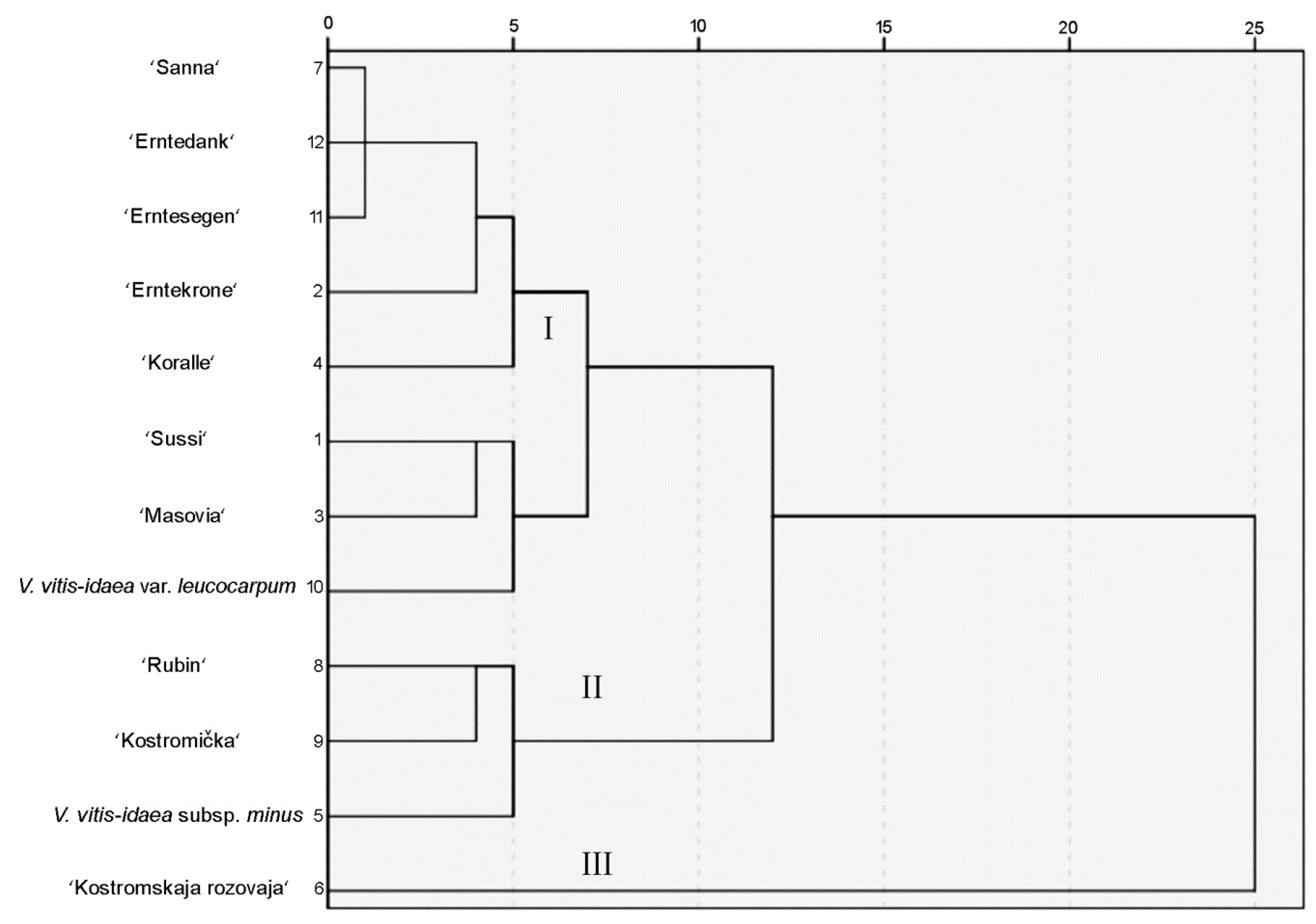
2.3. Hierarchical Cluster Analysis of Phenolic Compounds of Lingonberry Leaves of Different Cultivars and Lower Taxa
2.4. Antioxidant Activity of Lingonberries Phenolic Compounds
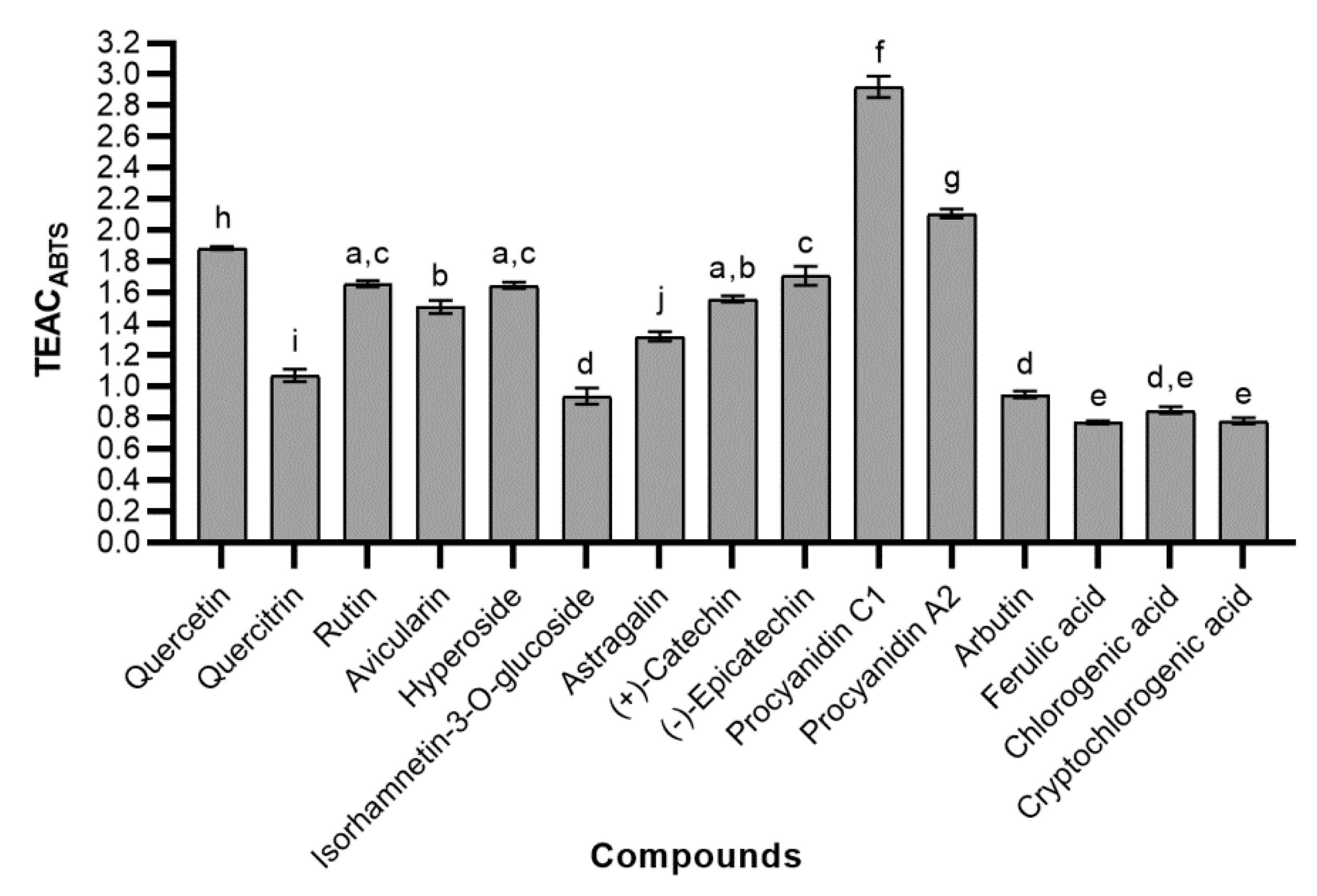
2.4.1. ABTS Radical Scavenging Activity
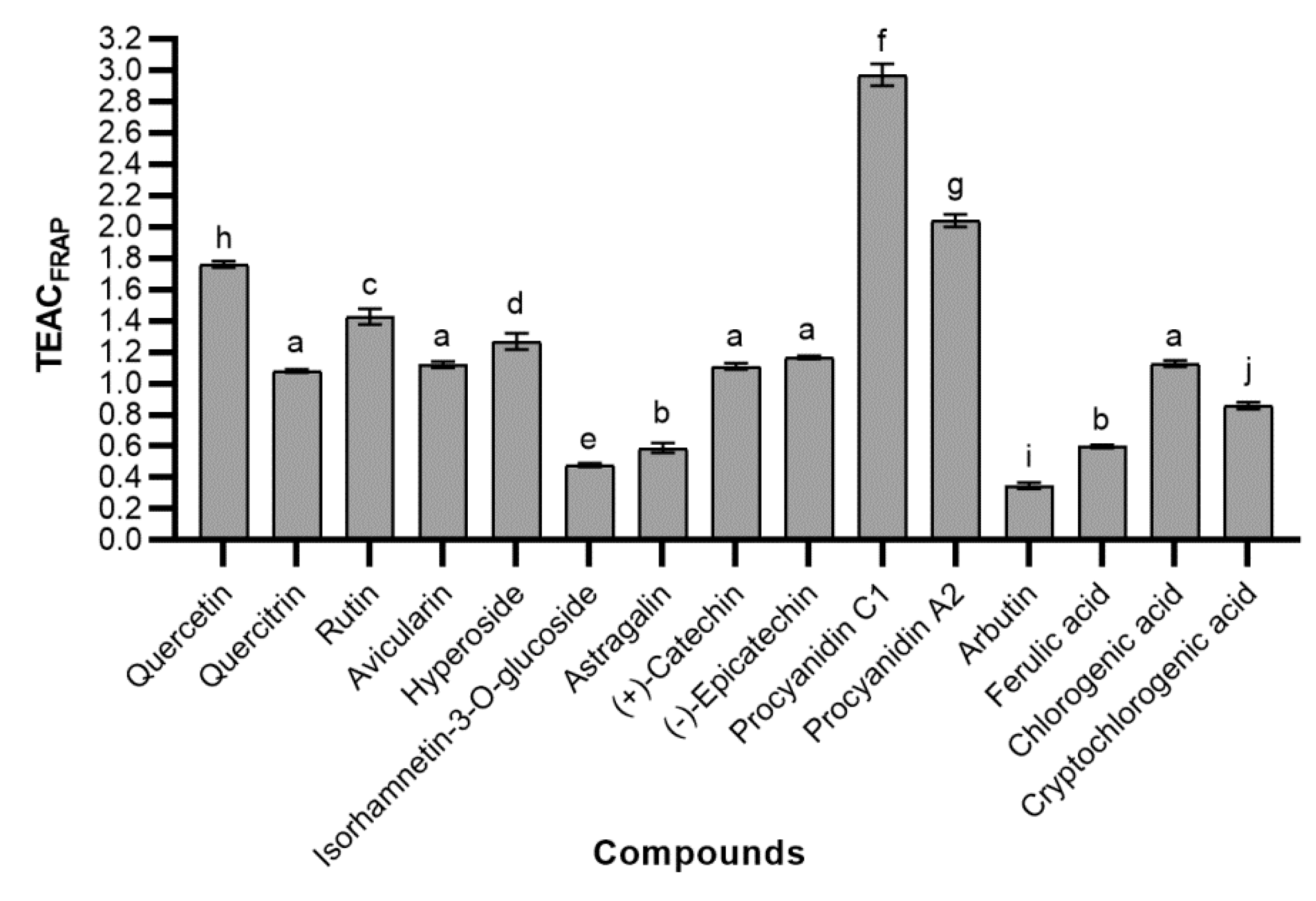
2.4.2. Ferric Reducing Antioxidant Power (FRAP)
2.4.3. Ferrous Ions Chelating (FIC) Activity
2.5. Correlation Analysis
3. Discussion
4. Materials and Methods
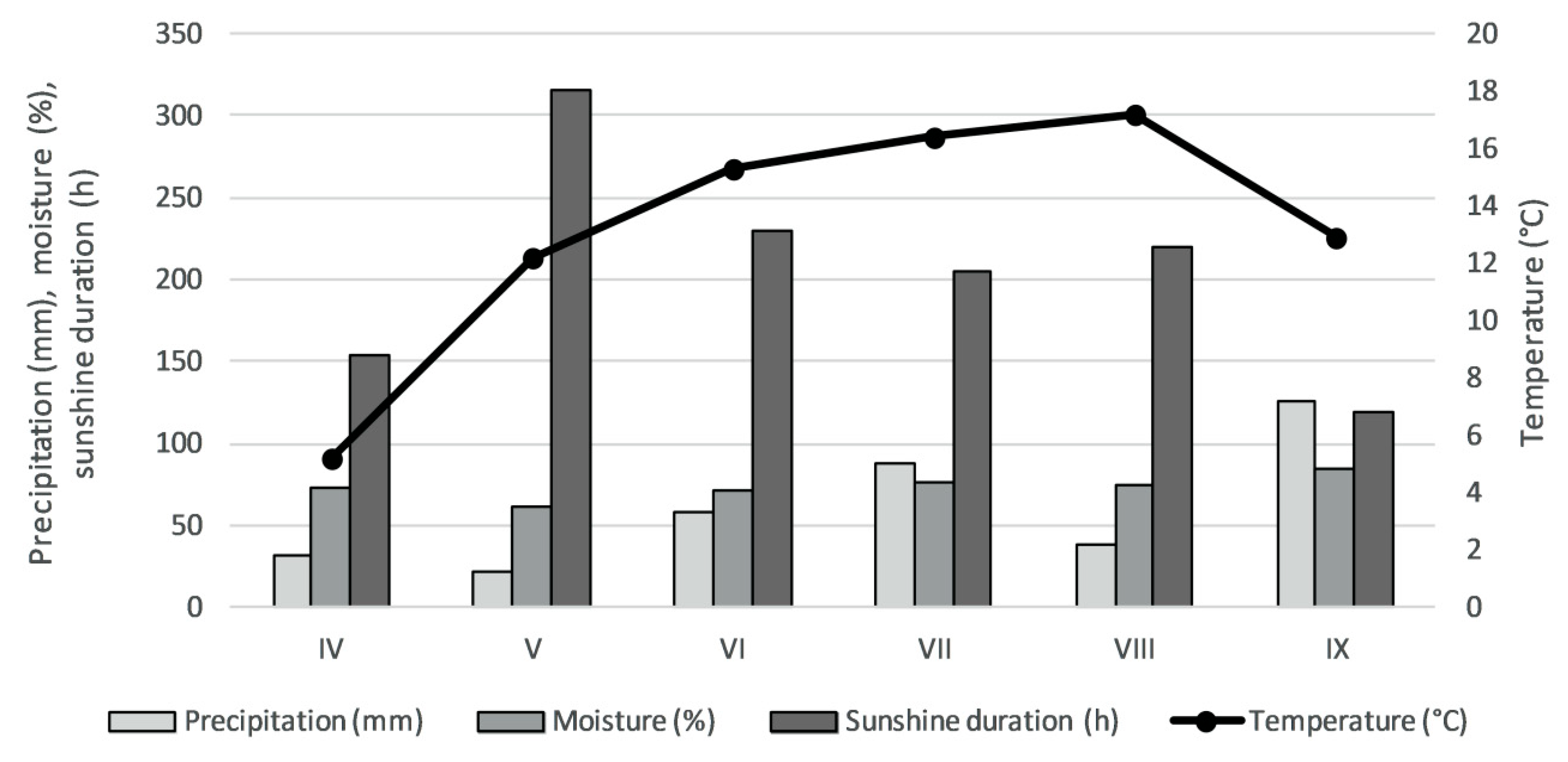
4.1. Plant Materials
4.2. Chemicals and Solvents
4.3. Sample Preparation
4.3.1. Preparation of the Lingonberry Leaves Extracts
4.3.2. Preparation of the Phenolic Compounds
4.4. Determination of Antioxidant Activity
4.4.1. ABTS Radical Cation Decolorization Assay
4.4.2. Ferric Reducing Antioxidant Power (FRAP) Assay
4.4.3. Ferrous Ions Chelating (FIC) Activity Assay
4.5. Qualitative and Quantitative Analysis by HPLC–PDA Method
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Liu, P.; Lindstedt, A.; Markkinen, N.; Sinkkonen, J.; Suomela, J.; Yang, B. Characterization Of Metabolite Profiles Of Leaves Of Bilberry (Vaccinium myrtillus L.) And Lingonberry (Vaccinium vitis-idaea L.). J. Agric. Food Chem. 2014, 62, 12015–12026. [Google Scholar] [CrossRef] [PubMed]
- Szakiel, A.; Pączkowski, C.; Koivuniemi, H.; Huttunen, S. Comparison Of The Triterpenoid Content Of Berries And Leaves Of Lingonberry Vaccinium vitis-idaea From Finland And Poland. J. Agric. Food Chem. 2012, 60, 4994–5002. [Google Scholar] [CrossRef] [PubMed]
- Bandzaitienė, Z.; Daubaras, R.; Labokas, J. Bruknė: Vaccinium vitis-idaea L.; Botanikos Instituto Leidykla: Vilnius, Lithuania, 2007; pp. 1323–1333. [Google Scholar]
- Bujor, O.; Ginies, C.; Popa, V.; Dufour, C. Phenolic Compounds And Antioxidant Activity Of Lingonberry (Vaccinium vitis-idaea L.) Leaf, Stem And Fruit At Different Harvest Periods. Food Chem. 2018, 252, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Debnath, S.; McRae, K. In Vitro Culture Of Lingonberry (Vaccinium vitis-idaea L.). Small Fruits Rev. 2001, 1, 3–19. [Google Scholar] [CrossRef]
- Ferlemi, A.; Lamari, F. Berry Leaves: An Alternative Source Of Bioactive Natural Products Of Nutritional And Medicinal Value. Antioxidants 2016, 5, 17. [Google Scholar] [CrossRef] [PubMed]
- Vyas, P.; Curran, N.; Igamberdiev, A.; Debnath, S. Antioxidant Properties Of Lingonberry (Vaccinium vitis-idaea L.) Leaves Within A Set Of Wild Clones And Cultivars. Can. J. Plant. Sci. 2015, 95, 663–669. [Google Scholar] [CrossRef]
- Ali, H.; Abo-Shady, A.; Sharaf Eldeen, H.; Soror, H.; Shousha, W.; Abdel-Barry, O.; Saleh, A. Structural Features, Kinetics And SAR Study Of Radical Scavenging And Antioxidant Activities Of Phenolic And Anilinic Compounds. Chem. Cent. J. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Csepregi, K.; Neugart, S.; Schreiner, M.; Hideg, É. Comparative Evaluation Of Total Antioxidant Capacities Of Plant Polyphenols. Molecules 2016, 21, 208. [Google Scholar] [CrossRef] [PubMed]
- Mladěnka, P.; Macáková, K.; Filipský, T.; Zatloukalová, L.; Jahodář, L.; Bovicelli, P.; Silvestri, I.; Hrdina, R.; Saso, L. In Vitro Analysis Of Iron Chelating Activity Of Flavonoids. J. Inorg. Biochem. 2011, 105, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Li, Q.; Bi, K. Bioactive Flavonoids In Medicinal Plants: Structure, Activity And Biological Fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef]
- Lago, J.; Toledo-Arruda, A.; Mernak, M.; Barrosa, K.; Martins, M.; Tibério, I.; Prado, C. Structure-Activity Association Of Flavonoids In Lung Diseases. Molecules 2014, 19, 3570–3595. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Schaich, K. Effects of Molecular Structure on Kinetics And Dynamics of the Trolox Equivalent Antioxidant Capacity Assay with ABTS+•. J. Agric. Food Chem. 2013, 61, 5511–5519. [Google Scholar] [CrossRef] [PubMed]
- Cheynier, V.; Comte, G.; Davies, K.; Lattanzio, V.; Martens, S. Plant Phenolics: Recent Advances On Their Biosynthesis, Genetics, And Ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Lee, J. Establishing A Case For Improved Food Phenolic Analysis. Food Sci. Nutr. 2013, 2, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Enkhtuya, E.; Kashiwagi, T.; Shimamura, T.; Ukeda, H.; Tseye-Oidov, O. Screening Study On Antioxidant Activity Of Plants Grown Wildly In Mongolia. Food Sci. Technol. Res. 2014, 20, 891–897. [Google Scholar] [CrossRef]
- Samoilova, Z.; Muzyka, N.; Lepekhina, E.; Oktyabrsky, O.; Smirnova, G. Medicinal Plant Extracts Can Variously Modify Biofilm Formation In Escherichia Coli. Anton. Leeuw. 2014, 105, 709–722. [Google Scholar] [CrossRef] [PubMed]
- Ieri, F.; Martini, S.; Innocenti, M.; Mulinacci, N. Phenolic Distribution in Liquid Preparations of Vaccinium myrtillus L. and Vaccinium vitis idaea L. Phytochem. Anal. 2013, 24, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Saario, M.; Koivusalo, S.; Laakso, I.; Autio, J. Allelopathic Potential Of Lingonberry (Vaccinium vitis-idaea L.) Litter For Weed Control. Biol. Agric. Hortic. 2002, 20, 11–28. [Google Scholar] [CrossRef]
- Rychlinska, I.; Nowak, S. Quantitative Determination Of Arbutin And Hydroquinone in Different Plant Materials By HPLC. Not. Bot. Horti Agrobot. Cluj-Napoca 2012, 40, 109–113. [Google Scholar] [CrossRef]
- Tian, Y.; Puganen, A.; Alakomi, H.; Uusitupa, A.; Saarela, M.; Yang, B. Antioxidative And Antibacterial Activities Of Aqueous Ethanol Extracts Of Berries, Leaves, And Branches Of Berry Plants. Food Res. Int. 2018, 106, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Hokkanen, J.; Mattila, S.; Jaakola, L.; Pirttilä, A.; Tolonen, A. Identification Of Phenolic Compounds From Lingonberry (Vaccinium vitis-idaea L.), Bilberry (Vaccinium myrtillus L.) And Hybrid Bilberry (Vaccinium X Intermediumruthe L.) Leaves. J. Agric. Food Chem. 2009, 57, 9437–9447. [Google Scholar] [CrossRef] [PubMed]
- Wojnicz, D.; Kucharska, A.; Sokół-Łętowska, A.; Kicia, M.; Tichaczek-Goska, D. Medicinal Plants Extracts Affect Virulence Factors Expression And Biofilm Formation By The Uropathogenic Escherichia coli. Urol. Res. 2012, 40, 683–697. [Google Scholar] [CrossRef] [PubMed]
- Ek, S.; Kartimo, H.; Mattila, S.; Tolonen, A. Characterization Of Phenolic Compounds From Lingonberry (Vaccinium vitis-idaea). J. Agric. Food Chem. 2006, 54, 9834–9842. [Google Scholar] [CrossRef] [PubMed]
- Jungfer, E.; Zimmermann, B.; Ruttkat, A.; Galensa, R. Comparing Procyanidins In Selected Vaccinium Species By UHPLC-MS2 With Regard To Authenticity And Health Effects. J. Agric. Food Chem. 2012, 60, 9688–9696. [Google Scholar] [CrossRef] [PubMed]
- Kylli, P.; Nohynek, L.; Puupponen-Pimiä, R.; Westerlund-Wikström, B.; Leppänen, T.; Welling, J.; Moilanen, E.; Heinonen, M. Lingonberry (Vaccinium vitis-idaea) And European Cranberry (Vaccinium Microcarpon) Proanthocyanidins: Isolation, Identification, And Bioactivities. J. Agric. Food Chem. 2011, 59, 3373–3384. [Google Scholar] [CrossRef] [PubMed]
- Okhrimenko, L.P.; Kalinkina, G.I.; Luksha, Y.I.; Kolomiytec, N.E. Study Of Phenolic Compounds Of Blueberry, Lingonberry, Bearberry, Bilberry And Wintergreen Leaves Growing In The Republic Of Sakha (Yakutia). Khimiia Rastitel’nogo Syr’ia 2009, 3, 109–115. (In Russian) [Google Scholar]
- Vysochina, G.I. Phenolic compounds in systematics and phylogeny of the family Polygonaceae Juss. Turczaninowia 2014, 17, 33–41. (In Russian) [Google Scholar] [CrossRef]
- Karpova, E.; Lapteva, N. Phenolic Compounds in Taxonomy of The Genus Spiraea L. Turczaninowia 2014, 17, 42–56. [Google Scholar] [CrossRef]
- Kostikova, V.; Shaldaeva, T. The Antioxidant Activity of the Russian Far East Representatives of the Spiraea L. (Rosaceae Juss.) Genus. Russ. J. Bioorg. Chem. 2017, 43, 790–794. [Google Scholar] [CrossRef]
- Muselík, J.; García-Alonso, M.; Martín-López, M.; Žemlička, M.; Rivas-Gonzalo, J. Measurement Of Antioxidant Activity Of Wine Catechins, Procyanidins, Anthocyanins And Pyranoanthocyanins. Int. J. Mol. Sci. 2007, 8, 797–809. [Google Scholar] [CrossRef]
- Spranger, I.; Sun, B.; Mateus, A.; Freitas, V.; Ricardo-da-Silva, J. Chemical Characterization And Antioxidant Activities Of Oligomeric And Polymeric Procyanidin Fractions From Grape Seeds. Food Chem. 2008, 108, 519–532. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Zhao, F.; Li, B.; Wei, T.; Guan, H.; Ren, S. Antioxidant And Fluorescence Properties Of Hydrogenolyzised Polymeric Proanthocyanidins Prepared Using SO42−/Zro2 Solid Superacids Catalyst. Molecules 2018, 23, 2445. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Zhu, C.; Hu, M.; Wu, A.; Zerendawa, B.; Suolangqimei, K. Structure–Activity Relationships Of Antioxidant Activity In Vitro About Flavonoids Isolated From Pyrethrum tatsienense. J Intercult Ethnopharmacol. 2014, 3, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Fan, H.; Yin, P.; Yang, L.; Xue, Q.; Li, X.; Sun, L.; Liu, Y. Structure–Activity Relationship Of Eight High Content Flavonoids Analyzed With A Preliminary Assign–Score Method And Their Contribution To Antioxidant Ability Of Flavonoids−Rich Extract From Scutellaria baicalensis Shoots. Arab. J. Chem. 2018, 11, 159–170. [Google Scholar] [CrossRef]
- Cai, Y.; Sun, M.; Xing, J.; Luo, Q.; Corke, H. Structure–Radical Scavenging Activity Relationships Of Phenolic Compounds From Traditional Chinese Medicinal Plants. Life Sci. 2006, 78, 2872–2888. [Google Scholar] [CrossRef] [PubMed]
- Natella, F.; Nardini, M.; Di Felice, M.; Scaccini, C. Benzoic And Cinnamic Acid Derivatives As Antioxidants: Structure-Activity Relation. J. Agric. Food Chem. 1999, 47, 1453–1459. [Google Scholar] [CrossRef] [PubMed]
- Grzesik, M.; Naparło, K.; Bartosz, G.; Sadowska-Bartosz, I. Antioxidant Properties Of Catechins: Comparison With Other Antioxidants. Food Chem. 2018, 241, 480–492. [Google Scholar] [CrossRef] [PubMed]
- Yordi, E.; Pérez, E.; Villares, E.; Matos, M. Antioxidant And Pro–Oxidant Effects Of Polyphenolic Compounds And Structure-Activity Relationship Evidence. In Nutrition, Well-Being and Health, 1st ed.; Bouayed, J., Ed.; InTech Open Access Publisher: Rijeka, Croatia, 2012; pp. 23–48. [Google Scholar]
- Končić, M.; Barbarić, M.; Perković, I.; Zorc, B. Antiradical, Chelating And Antioxidant Activities Of Hydroxamic Acids And Hydroxyureas. Molecules 2011, 16, 6232–6242. [Google Scholar] [CrossRef] [PubMed]
- Sova, M. Antioxidant And Antimicrobial Activities Of Cinnamic Acid Derivatives. Mini Rev. Med. Chem. 2012, 12, 749–767. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xiao, H.; Zheng, J.; Liang, G. Structure–Thermodynamics–Antioxidant Activity Relationships Of Selected Natural Phenolic Acids And Derivatives: An Experimental And Theoretical Evaluation. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Di Majo, D.; La Neve, L.; La Guardia, M.; Casuccio, A.; Giammanco, M. The Influence Of Two Different Ph Levels On The Antioxidant Properties Of Flavonols, Flavan-3-Ols, Phenolic Acids And Aldehyde Compounds Analysed In Synthetic Wine And In A Phosphate Buffer. J. Food Compost Anal. 2011, 24, 265–269. [Google Scholar] [CrossRef]
- Szakiel, A.; Morczek, A. Distribution Of Triterpene Acids And Their Derivatives In Organs Of Cowberry (Vaccinium vitis-idaea L.) Plant. Acta Biochim. Pol. 2007, 54, 733–740. [Google Scholar] [PubMed]
- Bandzaitienė, Z. Bruknės (Vaccinium vitis-idaea var. leucocarpum Asch. et Magnus) baltauogis varietetas Lietuvoje. In Lietuvos Bioįvairovė (Būklė, Struktūra, Apsauga); Lietuvos Edukologijos Universiteto Leidykla: Vilnius, Lithuania, 1997; pp. 28–29. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant Activity Applying An Improved ABTS Radical Cation Decolorization Assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Koleva, I.; Niederländer, H.; van Beek, T. Application Of ABTS Radical Cation For Selective On–Line Detection Of Radical Scavengers In HPLC Eluates. Anal. Chem. 2001, 73, 3373–3381. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.; Strain, J. The Ferric Reducing Ability Of Plasma (FRAP) As A Measure Of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Zhou, C.; Sun, Y.; Zhang, X.; Liu, J.; Hu, Q.; Zeng, X. Antioxidant Activities In Vitro Of Ethanol Extract From Brown Seaweed Sargassum pallidum. Eur. Food Res. Technol. 2009, 230, 101–109. [Google Scholar] [CrossRef]
- Zengin, G.; Aktumsek, A. Investigation Of Antioxidant Potentials Of Solvent Extracts From Different Anatomical Parts Of Asphodeline Anatolica E. Tuzlaci: An Endemic Plant To Turkey. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Raudone, L.; Zymone, K.; Raudonis, R.; Vainoriene, R.; Motiekaityte, V.; Janulis, V. Phenological changes in triterpenic and phenolic composition of Thymus L. species. Ind. Crops Prod. 2017, 109, 445–451. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| Cultivars | Compounds | ||||
|---|---|---|---|---|---|
| Astragalin | Avicularin | Hyperoside | Quercitrin | Arbutin | |
| ‘Sussi’ | 53.22 ± 2.17 d 1 | 1681.92 ± 68.66 a | 1744.85 ± 71.23 a | 2686.48 ± 109.68 a,b | 16,445.51 ± 671.39 a,b |
| ‘Erntekrone’ | 5.19 ± 0.21 a | 3479.62 ± 142.06 e | 4380.64 ± 178.84 e | 1692.27 ± 69.09 d | 12,441.17 ± 507.91 a,c |
| ‘Masovia’ | 3.07 ± 0.13 a | 2313.59 ± 94.45 b | 2522.88 ± 103.00 b | 1676.31 ± 68.43 d | 20,997.03 ± 857.20 b,d |
| ‘Koralle’ | 6.53 ± 0.27 e | 2261.69 ± 92.33 b,c | 2939.20 ± 119.99 c | 7826.68 ± 319.52 f | 13,755.55 ± 561.57 a |
| V. vitis-idaea subsp. minus | 8.89 ± 0.36 a | 2419.27 ± 98.77 b,d | 5771.34 ± 235.61 d | 5782.61 ± 236.07 g | 28,708.70 ± 1172.03 e |
| ‘Kostromskaja rozovaja’ | 114.67 ± 4.68 b,c | 2158.17 ± 88.11 b,c | 2342.03 ± 95.61 b | 1871.89 ± 76.42 d,e | 56,968.56 ± 2325.73 g |
| ‘Sanna’ | 4.38 ± 0.18 a | 2179.45 ± 88.98 f | 1926.56 ± 78.65 a | 2632.32 ± 107.46 a,b | 7070.25 ± 288.64 c |
| ‘Rubin’ | 120.60 ± 4.92 b | 3113.46 ± 127.11 g | 3084.12 ± 125.91 c | 2628.62 ± 107.31 a,b | 36,059.47 ± 1472.12 f |
| ‘Kostromička’ | 108.79 ± 4.44 c | 2715.82 ± 110.87 d | 3520.41 ± 143.72 f | 2471.29 ± 100.89 a,c | 29,877.25 ± 1219.73 e,f |
| V. vitis-idaea var. leucocarpum | 82.02 ± 3.35 f | 5503.44 ± 224.68 g | 5682.61 ± 231.99 d | 2139.10 ± 87.33 c,e | 23,356.78 ± 953.54 d,e |
| ‘Erntesegen’ | 23.15 ± 0.95 g | 1941.79 ± 79.27 c | 1764.92 ± 72.05 a | 1723.17 ± 70.35 d,e | 8963.10 ± 365.92 c |
| ‘Erntedank’ | 111.77 ± 4.56 c | 1946.56 ± 79.47 c | 1561.43 ± 63.74 a | 2953.06 ± 120.56 b | 7443.27 ± 303.87 c |
| Cultivars | Compounds | |||||
|---|---|---|---|---|---|---|
| (+)-Catechin | (−)-Epicatechin | Procyanidin C1 | Procyanidin A2 | Chlorogenic Acid | Cryptochlorogenic Acid | |
| ‘Sussi’ | 897.82 ± 36.65 a 1 | 252.14 ± 10.29 c | 1332.85 ± 54.41 a,b | 1040.11 ± 42.46 a | 346.25 ± 14.14 a | 417.74 ± 17.05 b |
| ‘Erntekrone’ | 595.09 ± 24.29 b | 26.27 ± 1.07 a | 1071.21 ± 43.73 a,c | 3609.57 ± 147.36 b | 318.35 ± 13.00 a | 368.86 ± 15.06 b,c |
| ‘Masovia’ | 2954.64 ± 120.62 e | 571.32 ± 23.32 d | 1090.83 ± 44.53 a,c | 3554.68 ± 145.12 b | 532.09 ± 21.72 b | 334.89 ± 13.67 c |
| ‘Koralle’ | 605.81 ± 24.73 b,c | 71.80 ± 2.93 b | 1248.83 ± 50.98 a,b,d | 3003.60 ± 122.62 c,d | 189.75 ± 7.75 d | 217.17 ± 8.87 a |
| V. vitis-idaea subsp. minus | 247.92 ± 10.12 d | 34.29 ± 1.40 a,b | 1310.87 ± 53.52 a,d | 7074.60 ± 288.82 g | 182.31 ± 7.44 d | 673.22 ± 27.48 d |
| ‘Kostromskaja rozovaja’ | 2273.92 ± 92.83 f | 743.67 ± 30.36 e | 2481.71 ± 101.32 e | 3027.88 ± 123.61 c,f | 486.65 ± 19.87 b,c | 942.07 ± 38.46 f |
| ‘Sanna’ | 141.29 ± 5.77 d | 60.48 ± 2.47 a,b | 1431.05 ± 58.42 b | 1364.89 ± 55.72 a | 442.12 ± 18.05 c | 184.24 ± 7.52 a |
| ‘Rubin’ | 2093.02 ± 85.45 g | 669.11 ± 27.32 f | 2525.23 ± 103.09 e | 3109.55 ± 126.95 d,e,f | 175.47 ± 7.16 d | 760.37 ± 31.04 e |
| ‘Kostromička’ | 767.38 ± 31.33 a,c | 33.11 ± 1.35 a,b | 3894.92 ± 159.01 f | 2684.38 ± 109.59 c | 444.92 ± 18.16 c | 704.39 ± 28.76 d,e |
| V. vitis-idaea var. leucocarpum | 1705.80 ± 69.64 h | 131.83 ± 5.38 g | 4645.97 ± 189.67 g | 3448.43 ± 140.78 b,e | 954.03 ± 38.95 e | 1025.34 ± 41.86 g |
| ‘Erntesegen’ | 168.93 ± 6.90 d | n.d. 2 | 1055.40 ± 43.09 c,d | 1365.75 ± 55.76 a | 243.70 ± 9.95 f | 218.81 ± 8.93 a |
| ‘Erntedank’ | 171.29 ± 6.99 d | n.d. | 936.48 ± 38.23 c | 476.93 ± 19.47 h | 352.68 ± 14.40 a | 231.42 ± 9.45 a |
| Compound | Concentration Range (mM) | Slope (a) | Intercept (b) | Correlation (R) | Coefficient of Determination (R2) |
|---|---|---|---|---|---|
| Trolox | 0.13–2.00 | 0.3559 | +0.0580 | 0.9983 | 0.9966 |
| Quercetin | 0.10–1.66 | 0.6709 | −0,0028 | 0.9968 | 0.9935 |
| Quercitrin | 0.06–0.89 | 0.3810 | +0.0538 | 0.9986 | 0.9973 |
| Rutin | 0.05–0.82 | 0.5378 | +0.0627 | 0.9952 | 0.9976 |
| Avicularin | 0.07–1.15 | 0.5378 | +0.1791 | 0.9988 | 0.9977 |
| Hyperoside | 0.05–0.86 | 0.5861 | +0.0224 | 0.9975 | 0.9981 |
| Isorhamnetin-3-O-glucoside | 0.07–1.05 | 0.3332 | +0.0642 | 0.9986 | 0.9993 |
| Astragalin | 0.07–1.12 | 0.4714 | +0.0989 | 0.9911 | 0.9955 |
| (+)-Catechin | 0.09–1.38 | 0.5544 | +0.0632 | 0.9964 | 0.9929 |
| (−)-Epicatechin | 0.09–1.38 | 0.6069 | +0.0649 | 0.9969 | 0.9938 |
| Procyanidin C1 | 0.04–0.58 | 1.0404 | +0.9958 | 0.9958 | 0.9917 |
| Procyanidin A2 | 0.05–0.87 | 0.7511 | +0.1408 | 0.9995 | 0.9990 |
| Arbutin | 0.11–1.84 | 0.3392 | +0.0512 | 0.9960 | 0.9920 |
| Ferulic acid | 0.16–2.58 | 0.2730 | +0.1183 | 0.9966 | 0.9932 |
| Chlorogenic acid | 0.09–1.41 | 0.3038 | +0.0775 | 0.9975 | 0.9950 |
| Cryptochlorogenic acid | 0.02–0.28 | 0.2761 | +0.0151 | 0.9969 | 0.9938 |
| Compound | Concentration Range (mM) | Slope (a) | Intercept (b) | Correlation (R) | Coefficient of Determination (R2) |
|---|---|---|---|---|---|
| Trolox | 0.13–2.00 | 0.5277 | −0.0165 | 0.9998 | 0.9997 |
| Quercetin | 0.10–1.66 | 0.9283 | −0.0671 | 0.9996 | 0.9992 |
| Quercitrin | 0.06–0.89 | 0.5712 | −0.0217 | 0.9975 | 0.9950 |
| Rutin | 0.05–0.82 | 0.7531 | −0.0157 | 0.9969 | 0.9938 |
| Avicularin | 0.07–1.15 | 0.5936 | +0.0268 | 0.9950 | 0.9900 |
| Hyperoside | 0.05–0.86 | 0.6692 | −0.0121 | 0.9995 | 0.9989 |
| Isorhamnetin-3-O-glucoside | 0.07–1.05 | 0.2559 | +0.0292 | 0.9951 | 0.9903 |
| Astragalin | 0.07–1.12 | 0.3119 | −0.0231 | 0.9976 | 0,9951 |
| (+)-Catechin | 0.09–1.38 | 0.5836 | −0.0203 | 0.9980 | 0.9960 |
| (−)-Epicatechin | 0.09–1.38 | 0.6172 | −0.0080 | 0.9991 | 0.9981 |
| Procyanidin C1 | 0.04–0.58 | 1.5679 | +0.0101 | 0.9999 | 0.9998 |
| Procyanidin A2 | 0.05–0.87 | 1.0766 | −0.0248 | 0.9960 | 0.9921 |
| Arbutin | 0.11–1.84 | 0.1826 | −0.0021 | 0.9957 | 0.9957 |
| Ferulic acid | 0.16–2.58 | 0.3173 | +0.0708 | 0.9961 | 0.9914 |
| Chlorogenic acid | 0.09–1.41 | 0.5937 | +0.0218 | 0.9992 | 0.9984 |
| Cryptochlorogenic acid | 0.02–0.28 | 0.4563 | +0.0092 | 0.9972 | 0.9944 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raudone, L.; Vilkickyte, G.; Pitkauskaite, L.; Raudonis, R.; Vainoriene, R.; Motiekaityte, V. Antioxidant Activities of Vaccinium vitis-idaea L. Leaves within Cultivars and Their Phenolic Compounds. Molecules 2019, 24, 844. https://doi.org/10.3390/molecules24050844
Raudone L, Vilkickyte G, Pitkauskaite L, Raudonis R, Vainoriene R, Motiekaityte V. Antioxidant Activities of Vaccinium vitis-idaea L. Leaves within Cultivars and Their Phenolic Compounds. Molecules. 2019; 24(5):844. https://doi.org/10.3390/molecules24050844
Chicago/Turabian StyleRaudone, Lina, Gabriele Vilkickyte, Lina Pitkauskaite, Raimondas Raudonis, Rimanta Vainoriene, and Vida Motiekaityte. 2019. "Antioxidant Activities of Vaccinium vitis-idaea L. Leaves within Cultivars and Their Phenolic Compounds" Molecules 24, no. 5: 844. https://doi.org/10.3390/molecules24050844
APA StyleRaudone, L., Vilkickyte, G., Pitkauskaite, L., Raudonis, R., Vainoriene, R., & Motiekaityte, V. (2019). Antioxidant Activities of Vaccinium vitis-idaea L. Leaves within Cultivars and Their Phenolic Compounds. Molecules, 24(5), 844. https://doi.org/10.3390/molecules24050844








