Bunias erucago L.: Glucosinolate Profile and In Vitro Biological Potential
Abstract
1. Introduction
2. Results and Discussion
2.1. Glucosinolates and Volatile Constituents
2.2. Antioxidative and Anticholinesterase Activity
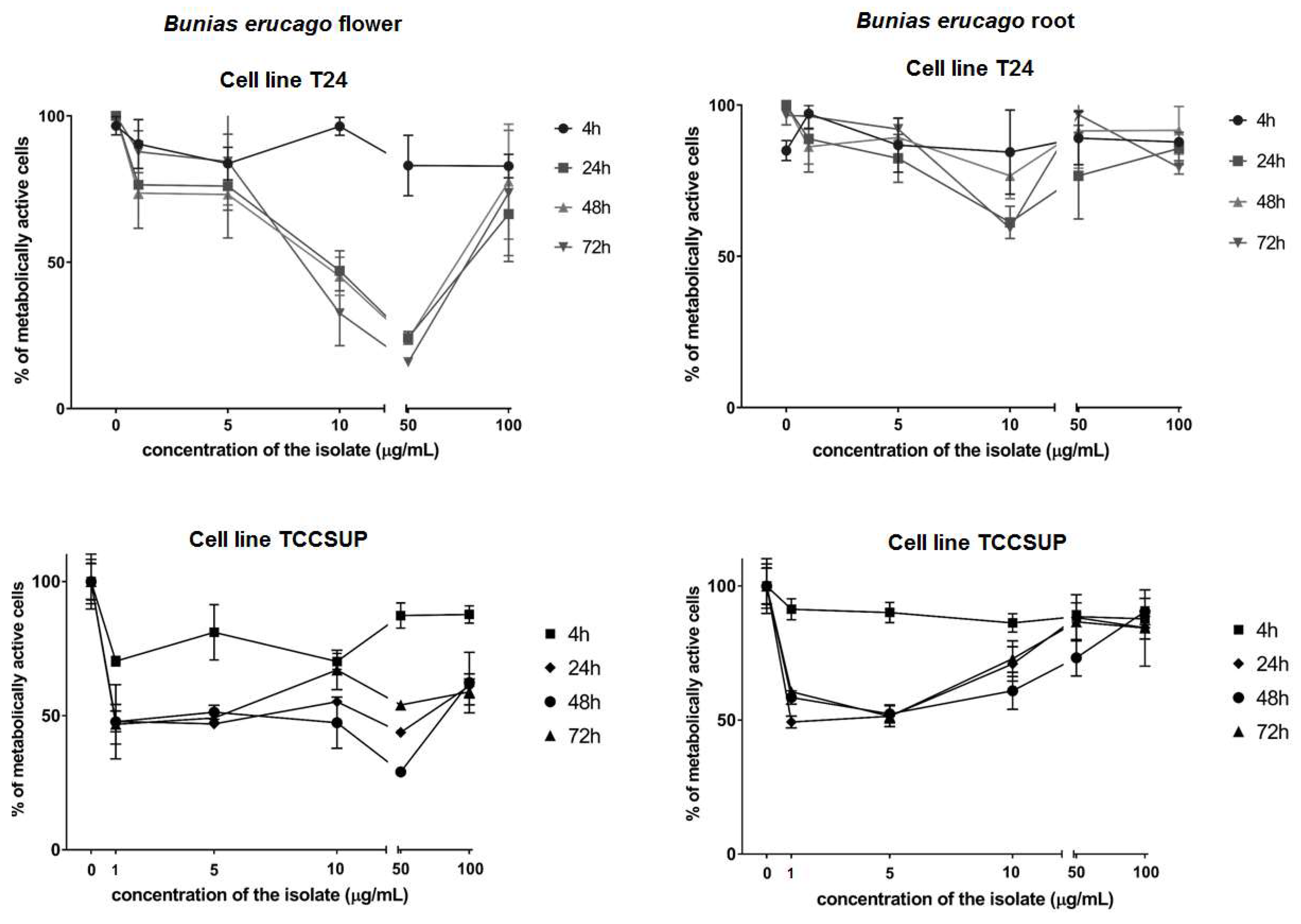
2.3. Cytotoxic Activity
3. Material and Methods
3.1. Plant Material
3.2. Reagents
3.3. Isolation and Chemical Analysis
3.3.1. Isolation
3.3.2. HPLC-PDA-MS/MS Analysis and Quantification of Desulfoglucosinolates
3.3.3. Isolation and GC-MS Analysis of Volatiles
3.3.4. Methods of Identification
3.4. Biological Activities Assays
3.4.1. Antioxidant Activity
3.4.2. Acetylcholinesterase/Butyrylcholinesterase Inhibitory Activity
3.4.3. Cell Proliferation Assay (MTT)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ardenghi, N.M.G.; Ballerini, C.; Bodino, S.; Cauzzi, P.; Guzzon, F. “Lándar”, “Lándra”, “Barlánd” (Bunias erucago L.): A neglected crop from the Po Plain (Northern Italy). Econ. Bot. 2017, 71, 288–295. [Google Scholar] [CrossRef]
- Ančev, M. Catalogue of the family Brassicaceae (Cruciferae) in the flora of Bulgaria. Phytol. Balc. 2007, 13, 153–178. [Google Scholar]
- Di Novella, R.; Di Novella, N.; De Martino, L.; Mancini, E.; De Feo, V. Traditional plant use in the National Park of Cilento and Vallo di Diano, Campania, Southern, Italy. J. Ethnopharmacol. 2013, 145, 328–342. [Google Scholar] [CrossRef] [PubMed]
- Romano, D.; Tribulato, A.; Toscano, S.; Scuderi, D. Ethnobotanical uses of Brassicaceae in Sicily. Acta Hortic. 2013, 1005, 197–204. [Google Scholar] [CrossRef]
- Dolina, K.; Łuczaj, Ł. Wild food plants used on the Dubrovnik coast (south-eastern Croatia). Acta Soc. Bot. Pol. 2014, 83, 175–181. [Google Scholar] [CrossRef]
- Łuczaj, Ł.; Dolina, K. A hundred years of change in wild vegetable use in southern Herzegovina. J. Ethnopharmacol. 2015, 166, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Kovačić, S.; Nikolić, T.; Ruščić, M.; Milović, M.; Stamenković, V.; Mihelj, D.; Jasprica, N.; Bogdanović, S.; Topić, J. Flora jadranske obale i otoka—250 najčešćih vrsta; Školska knjiga: Zagreb, Croatia, 2008; pp. 232–233. [Google Scholar]
- Agerbirk, N.; Olsen, C.E. Glucosinolate structures in evolution. Phytochemistry 2012, 77, 16–45. [Google Scholar] [CrossRef]
- Blažević, I.; Montaut, S.; Burčul, F.; Rollin, P. Glucosinolates: Novel sources and biological potential. In Glucosinolates; Mérillon, J.-M., Ramawat, G.K., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 3–60. [Google Scholar]
- Cole, R.A. Isothiocyanates, nitriles and thiocyanates as products of autolysis of glucosinolates in Cruciferae. Phytochemistry 1976, 15, 759–762. [Google Scholar] [CrossRef]
- Daxenbichler, M.E.; Spencer, G.F.; Carlson, D.G.; Rose, G.B.; Brinker, A.M.; Powell, R.G. Glucosinolate composition of seeds from 297 species of wild plants. Phytochemistry 1991, 30, 2623–2638. [Google Scholar] [CrossRef]
- Bennett, R.N.; Mellon, F.A.; Kroon, P.A. Screening crucifer seeds as sources of specific intact glucosinolates using ion-pair high-performance liquid chromatography negative ion electrospray mass spectrometry. J. Agric. Food Chem. 2004, 52, 428–438. [Google Scholar] [CrossRef]
- Bennett, R.N.; Rosa, E.A.S.; Mellon, F.A.; Kroon, P.A. Ontogenic profiling of glucosinolates, flavonoids, and other secondary metabolites in Eruca sativa (salad rocket), Diplotaxis erucoides (wall rocket), Diplotaxis tenuifolia (wild rocket), and Bunias orientalis (turkish rocket). J. Agric. Food Chem. 2006, 54, 4005–4015. [Google Scholar] [CrossRef] [PubMed]
- Maurizi, A.; De Michele, A.; Ranfa, A.; Ricci, A.; Roscini, V.; Coli, R.; Bodesmo, M.; Burini, G. Bioactive compounds and antioxidant characterization of three edible wild plants traditionally consumed in the Umbria Region (Central Italy): Bunias erucago L. (corn rocket), Lactuca perennis L. (mountain lettuce) and Papaver rhoeas L. (poppy). J. Appl. Bot. Food Qual. 2015, 88, 109–114. [Google Scholar]
- De Nicola, G.R.; Montaut, S.; Rollin, P.; Nyegue, M.; Menut, C.; Iori, R.; Tatibouët, A. Stability of Benzylic-Type Isothiocyanates in Hydrodistillation-Mimicking Conditions. J. Agric. Food Chem. 2013, 61, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Borek, V.; Morra, M.J. Ionic Thiocyanate (SCN-) Production from 4-Hydroxybenzyl Glucosinolate Contained in Sinapis alba Seed Meal. J. Agric. Food Chem. 2005, 53, 8650–8654. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wang, M.; Rosen, R.T.; Ho, C.-T. Thermal degradation of sulforaphane in aqueous solution. J. Agric. Food Chem. 1999, 47, 3121–3123. [Google Scholar] [CrossRef]
- Blažević, I.; Mastelić, J. Glucosinolate degradation products and other bound and free volatiles in the leaves and roots of radish (Raphanus sativus L.). Food Chem. 2009, 113, 96–102. [Google Scholar] [CrossRef]
- Spencer, G.F.; Daxenbichler, M.E. Gas chromatography-mass spectrometry of nitriles, isothiocyanates and oxazolidinethiones derived from cruciferous glucosinolates. J. Sci. Food Agric. 1980, 31, 359–367. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Berhow, M.A. Glucosinolate hydrolysis products from various plant sources: pH effects, isolation, and purification. Ind. Crops Prod. 2005, 21, 193–202. [Google Scholar] [CrossRef]
- Kjær, A.; Ohashi, M.; Wilson, J.M.; Djerassi, C. Mass spectra of isothiocyanates. Acta Chem. Scand. 1963, 17, 2143–2154. [Google Scholar] [CrossRef]
- Burčul, F.; Radan, M.; Politeo, O.; Blažević, I. Cholinesterase-inhibitory activity of essential oils. In Advances in Chemistry Research; Taylor, J.C., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2017; Volume 37, pp. 15–86. [Google Scholar]
- Burčul, F.; Generalić Mekinić, I.; Radan, M.; Rollin, P.; Blažević, I. Isothiocyanates: cholinesterase inhibiting, antioxidant, and anti-inflammatory activity. J. Enzym. Inhib. Med. Chem. 2018, 33, 577–582. [Google Scholar] [CrossRef]
- Blazevic, I.; Burcul, F.; Ruscic, M.; Mastelic, J. Glucosinolates, volatile constituents, and acetylcholinesterase inhibitory activity of Alyssoides utriculata. Chem. Nat. Compd. 2013, 49, 374–378. [Google Scholar] [CrossRef]
- Al-Gendy, A.; Nematallah, K.; S Zaghloul, S.; Ayoub, N. Glucosinolates profile, volatile constituents, antimicrobial, and cytotoxic activities of Lobularia libyca. Pharm. Biol. 2016, 54, 3257–3263. [Google Scholar] [CrossRef]
- Grosser, K.; van Dam, N.M. A straightforward method for glucosinolate extraction and analysis with high-pressure liquid chromatography (HPLC). JoVE 2017, 121, e55425. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.D.; Tokuhisa, J.G.; Reichelt, M.; Gershenzon, J. Variation of glucosinolate accumulation among different organs and developmental stages of Arabidopsis thaliana. Phytochemistry 2003, 62, 471–481. [Google Scholar] [CrossRef]
- Wathelet, J.-P.; Iori, R.; Leoni, O.; Quinsac, A.; Palmieri, S.; Rollin, P. Guidelines for glucosinolate analysis in green tissues used for biofumigation. Agroindustria 2004, 3, 257–266. [Google Scholar]
- Blažević, I.; Radonić, A.; Mastelić, J.; Zekić, M.; Skočibušić, M.; Maravić, A. Hedge mustard (Sisymbrium officinale): Chemical diversity of volatiles and their antimicrobial activity. Chem. Biodivers. 2010, 7, 2023–2034. [Google Scholar] [CrossRef]
- Bezić, N.; Vuko, E.; Dunkić, V.; Ruščić, M.; Blažević, I.; Burčul, F. Antiphytoviral activity of sesquiterpene-rich essential oils from four Croatian Teucrium species. Molecules 2011, 16, 8119–8129. [Google Scholar] [CrossRef] [PubMed]
- Olsen, C.E.; Huang, X.-C.; Hansen, C.I.C.; Cipollini, D.; Ørgaard, M.; Matthes, A.; Geu-Flores, F.; Koch, M.A.; Agerbirk, N. Glucosinolate diversity within a phylogenetic framework of the tribe Cardamineae (Brassicaceae) unraveled with HPLC-MS/MS and NMR-based analytical distinction of 70 desulfoglucosinolates. Phytochemistry 2016, 132, 33–56. [Google Scholar] [CrossRef] [PubMed]
- Fredotović, Ž.; Šprung, M.; Soldo, B.; Ljubenkov, I.; Budić-Leto, I.; Bilušić, T.; Čikeš-Čulić, V.; Puizina, J. Chemical composition and biological activity of Allium cepa L. and Allium × cornutum (Clementi ex Visiani 1842) methanolic extracts. Molecules 2017, 22, 448. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
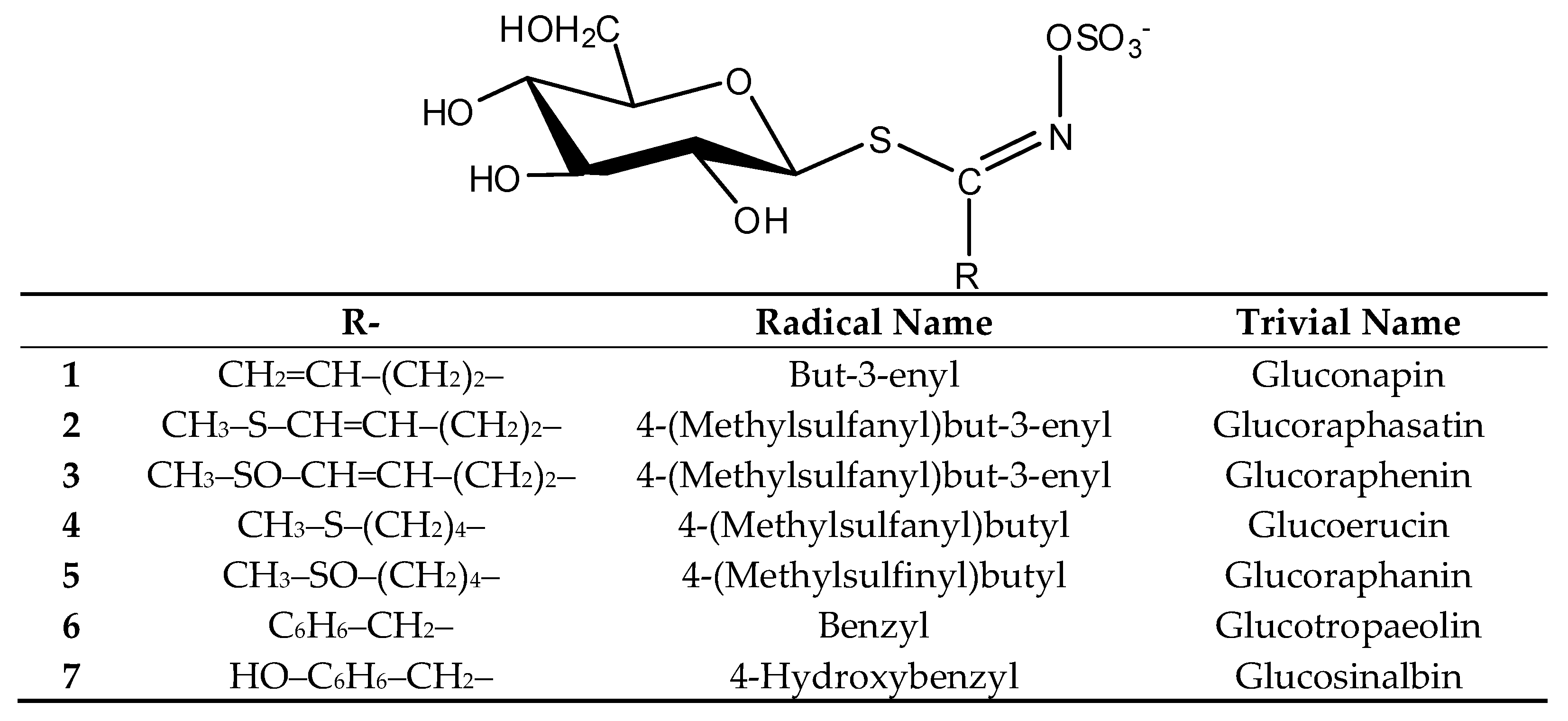
| Glucosinolate | tR (minutes) | Flower | Leaf and Stem | Root |
|---|---|---|---|---|
| Glucoraphanin (5) | 8.1 | 0.3 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| Glucoraphenin (3) | 8.4 | 5.7 ± 0.8 | 1.0 ± 0.2 | 1.6 ± 0.4 |
| Glucosinalbin (7) | 11.3 | 6.8 ± 0.5 | 5.6 ± 0.9 | 8.3 ± 1.0 |
| Gluconapin (1) | 12.1 | 0.6 ± 0.1 | 0.2 ± 0.0 | 0.5 ± 0.1 |
| Glucotropaeolin (6) | 18.2 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| Glucoerucin (4) | 18.9 | - | - | tr |
| Glucoraphasatin (2) | 19.5 | 1.0 ± 0.2 | - | 2.2 ± 0.2 |
| Total (μmol/g dry weight) | 14.6 ± 1.6 | 7.0 ± 1.1 | 12.9 ± 1.7 |
| Hydrodistilate | Extract | |||||
|---|---|---|---|---|---|---|
| Glucosinolate | Identified Breakdown Compound | RI | Flower | Leaf and Stem | Root | Whole Plant Material |
| Gluconapin (1) | But-3-enyl ITC a,b,c | 993 | 0.8 | 2.0 | 10.3 | - |
| Glucotropaeolin (6) | 2-Phenylacetonitrile a,b | 1178 | 1.0 | tr | - | - |
| Benzyl ITC a,b | 1394 | - | - | tr | - | |
| Glucoraphasatin (2) | 5-(Methylsulfanyl)pent-4-enenitrile *,a,b | 1214 | 14.5 | - | 1.4 | - |
| 5-(Methylsulfanyl)pent-4-enenitrile *,a,b | 1251 | 4.7 | - | 1.0 | - | |
| 4-(Methylsulfanyl)but-3-enyl ITC a,b,c | 1440 | 0.1 | - | 2.4 | - | |
| Glucoerucin (4) | 4-(Methylsulfanyl)pentanenitrile a,b,c | 1233 | 2.3 | - | - | - |
| 4-(Methylsulfanyl)butyl ITC (erucin) a,c | 1457 | - | - | 1.1 | - | |
| Glucoraphenin (3) | 4-(Methylsulfinyl)but-3-enyl ITC a,c | 1817 | - | - | - | 88.3 |
| Other Volatiles | ||||||
| 1,2-Dimethylbenzene b | 874 | 3.9 | 0.8 | 4.0 | - | |
| 3-Methylsulfanylpropanal b | 919 | - | - | 0.3 | - | |
| Dimethyltrisulfide a,b | 981 | tr | - | 9.4 | - | |
| Benzeneacetaldehyde a,b | 1063 | 0.5 | 1.1 | 1.3 | - | |
| (E)-Non-2-en-1-ol a,b | 1113 | 1.3 | 0.8 | 0.5 | - | |
| Dimethyltetrasulfide a,b | 1235 | - | - | 0.3 | - | |
| 4-Vinyl-2-methoxyphenol a,b | 1353 | 0.7 | 0.8 | tr | - | |
| (E)-β-Ionone a,b | 1492 | 1.8 | - | - | - | |
| Tetradecanoic acid a,b | 1828 | 3.9 | 18.3 | 6.7 | - | |
| 6,10,14-Trimethylpentadecan-2-one a,b | 1854 | 28.2 | 25.5 | 1.9 | - | |
| Hexadecanoic acid a,b | 2016 | 24.6 | 39.8 | 53.5 | 6.1 | |
| Total sum (%) | 88.3 | 89.1 | 94.1 | 94.4 | ||
| Flower Hydro-Distillate (FH) | Root Hydro-Distillate (RH) | |
|---|---|---|
| Antioxidant Activity | ||
| DPPH (Inhibition %) | 9.6 ± 0.4 | 10.2 ± 1.4 |
| FRAP (µM Fe2+) | 41.7 ± 0.9 | 24.3 ± 0.1 |
| Cholinesterase Activity | ||
| AChE (Inhibition %) | 40.9 ± 0.2 | 13.7 ± 0.2 |
| BChE (Inhibition %) | 25.0 ± 0.1 | 54.3 ± 0.3 |
| Flower Hydro-Distillate (FH) | Root Hydro-Distillate (RH) | |||||||
|---|---|---|---|---|---|---|---|---|
| 4 h | 24 h | 48 h | 72 h | 4 h | 24 h | 48 h | 72 h | |
| T24 | 362.6 | 21.4 | 23.8 | 16.0 | 517.7 | 267.5 | 676.0 | 366.8 |
| TCCSUP | 418.4 | 15.2 | 7.8 | 49.9 | 519.1 | 326.0 | 258.3 | 316.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blažević, I.; Đulović, A.; Čikeš Čulić, V.; Burčul, F.; Ljubenkov, I.; Ruščić, M.; Generalić Mekinić, I. Bunias erucago L.: Glucosinolate Profile and In Vitro Biological Potential. Molecules 2019, 24, 741. https://doi.org/10.3390/molecules24040741
Blažević I, Đulović A, Čikeš Čulić V, Burčul F, Ljubenkov I, Ruščić M, Generalić Mekinić I. Bunias erucago L.: Glucosinolate Profile and In Vitro Biological Potential. Molecules. 2019; 24(4):741. https://doi.org/10.3390/molecules24040741
Chicago/Turabian StyleBlažević, Ivica, Azra Đulović, Vedrana Čikeš Čulić, Franko Burčul, Ivica Ljubenkov, Mirko Ruščić, and Ivana Generalić Mekinić. 2019. "Bunias erucago L.: Glucosinolate Profile and In Vitro Biological Potential" Molecules 24, no. 4: 741. https://doi.org/10.3390/molecules24040741
APA StyleBlažević, I., Đulović, A., Čikeš Čulić, V., Burčul, F., Ljubenkov, I., Ruščić, M., & Generalić Mekinić, I. (2019). Bunias erucago L.: Glucosinolate Profile and In Vitro Biological Potential. Molecules, 24(4), 741. https://doi.org/10.3390/molecules24040741









