Physicochemical and Microstructural Properties of Polymerized Whey Protein Encapsulated 3,3′-Diindolylmethane Nanoparticles
Abstract
1. Introduction
2. Results and Discussion
2.1. ParticleSize and Zeta Potential of PWP–DIMNanoparticles
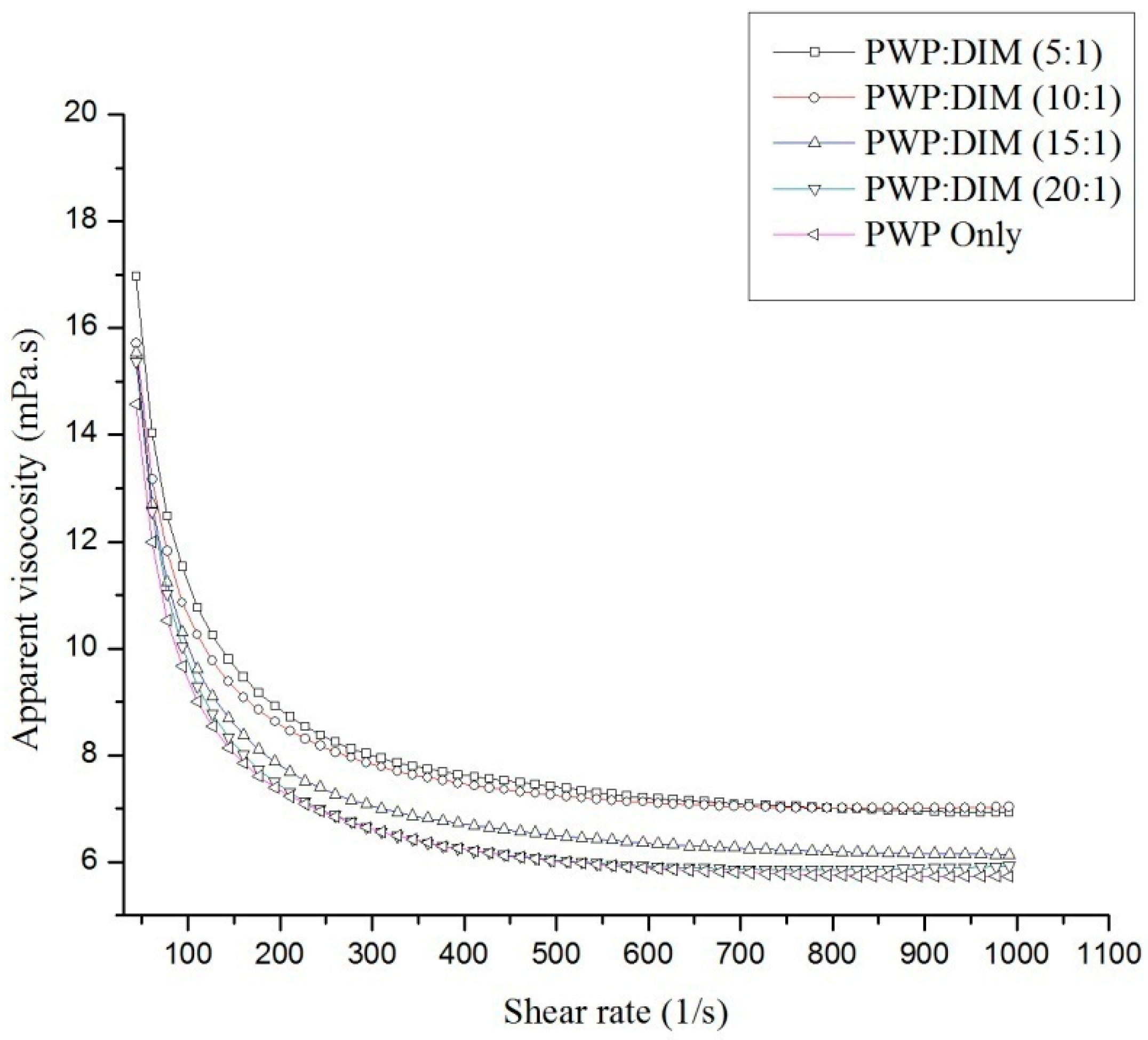
2.2. Rheological Properties of PWP–DIM Nanoparticles
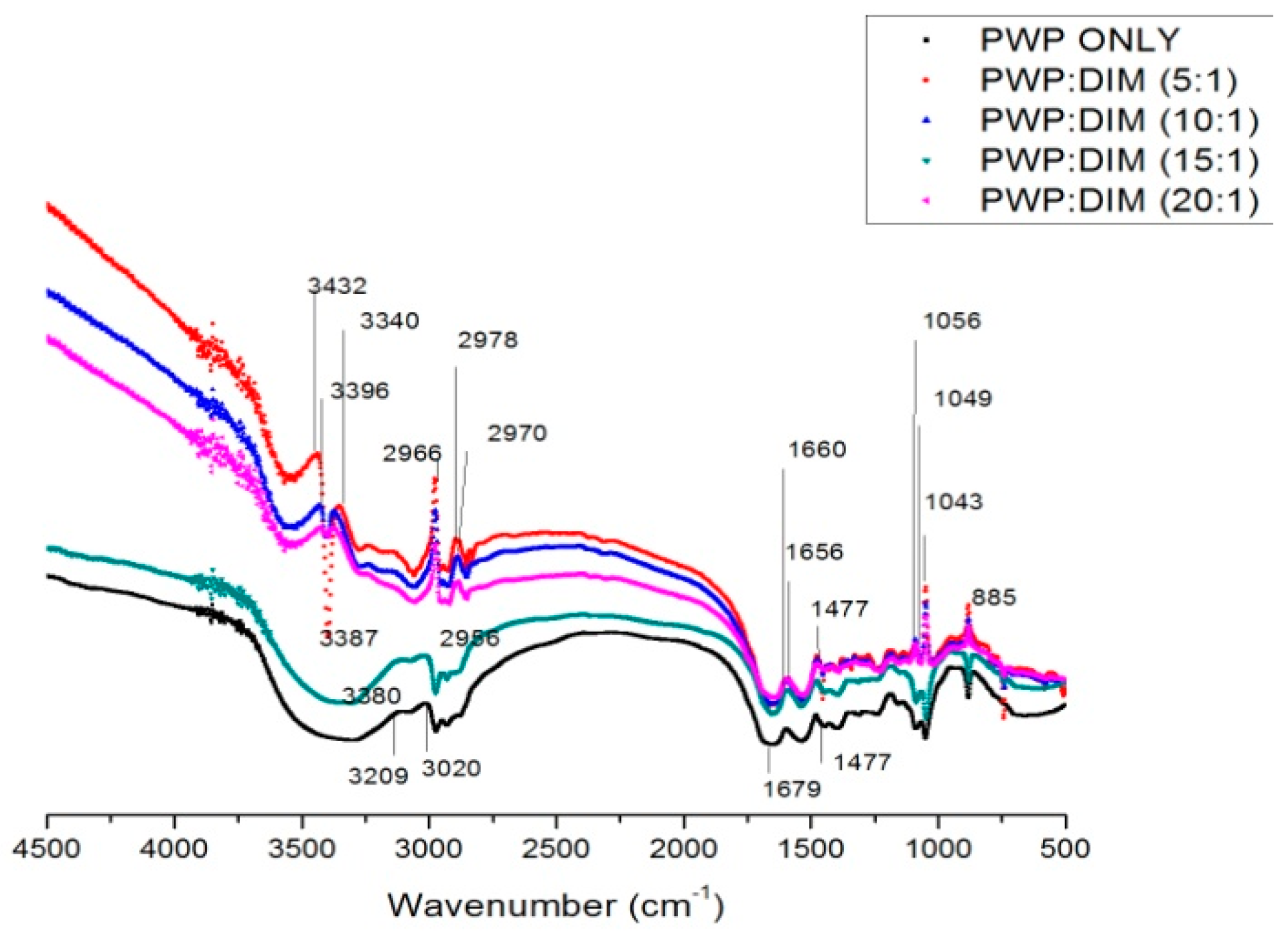
2.3. FTIRSpectra of PWP–DIM Nanoparticles
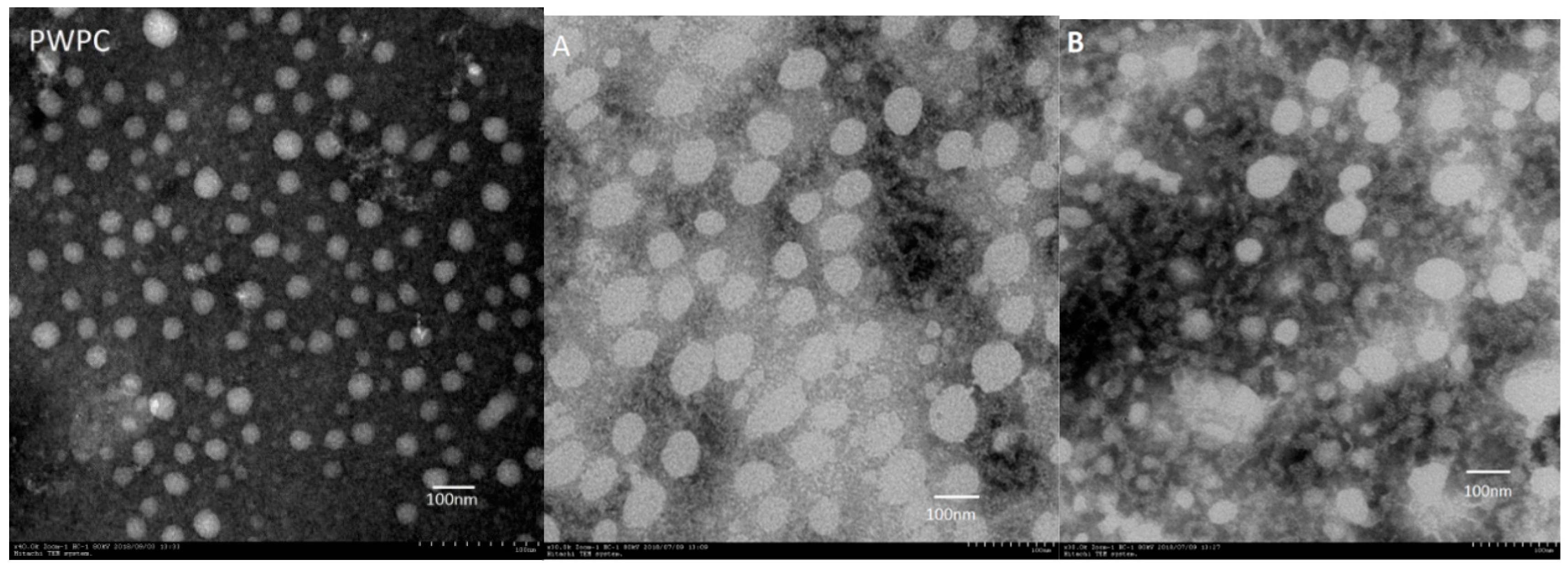
2.4. Microstructure of PWP–DIM Nanoparticles
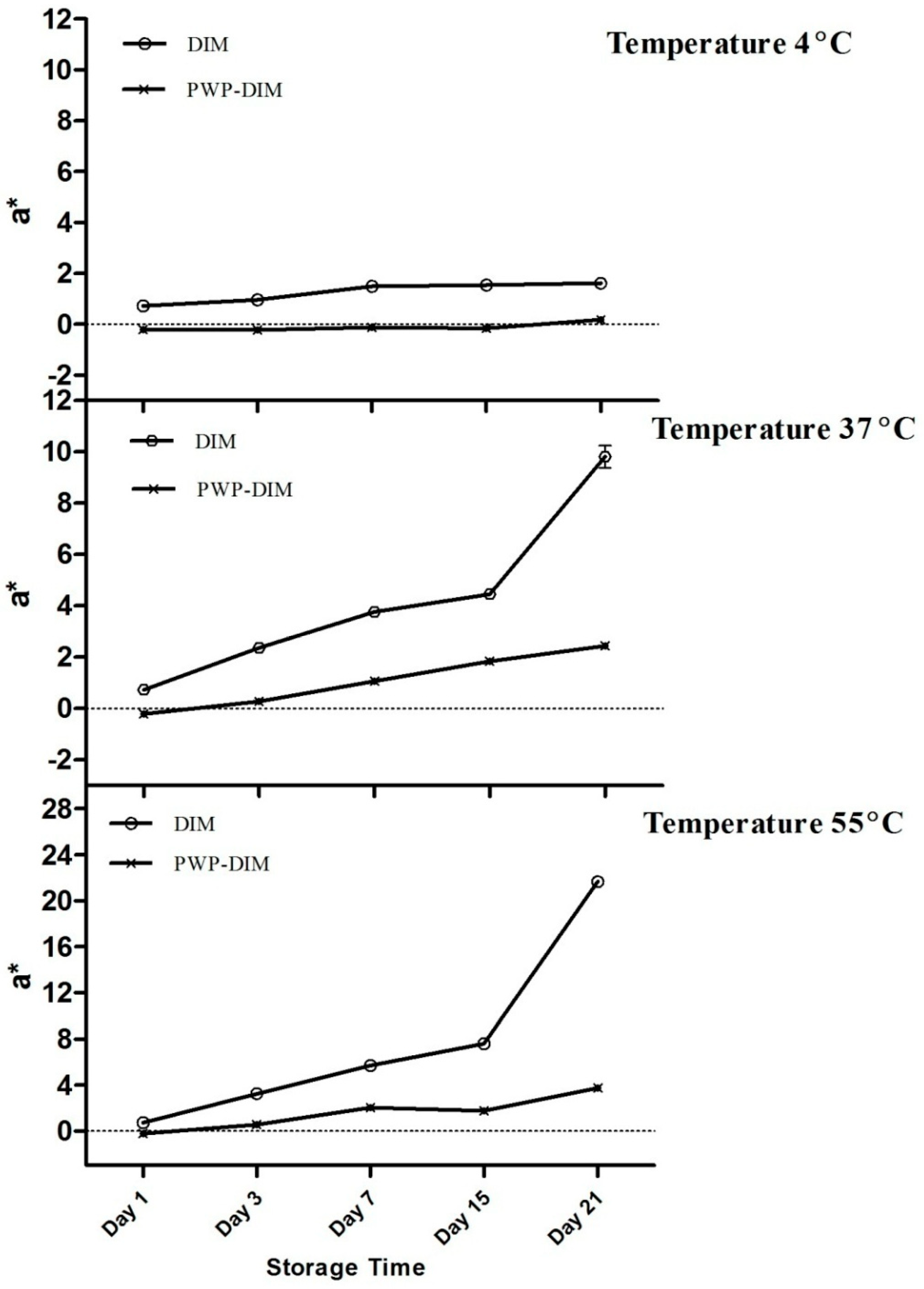
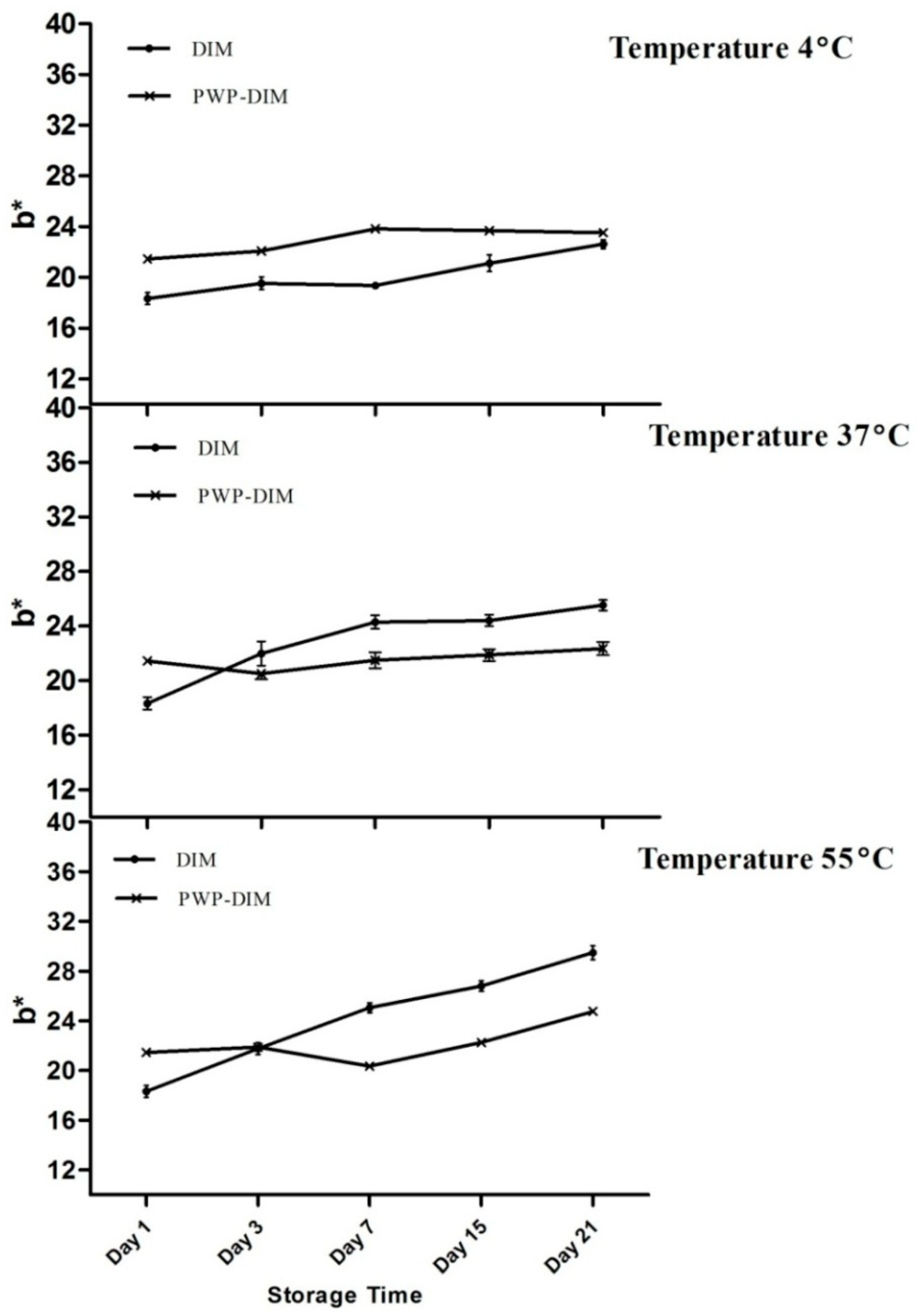
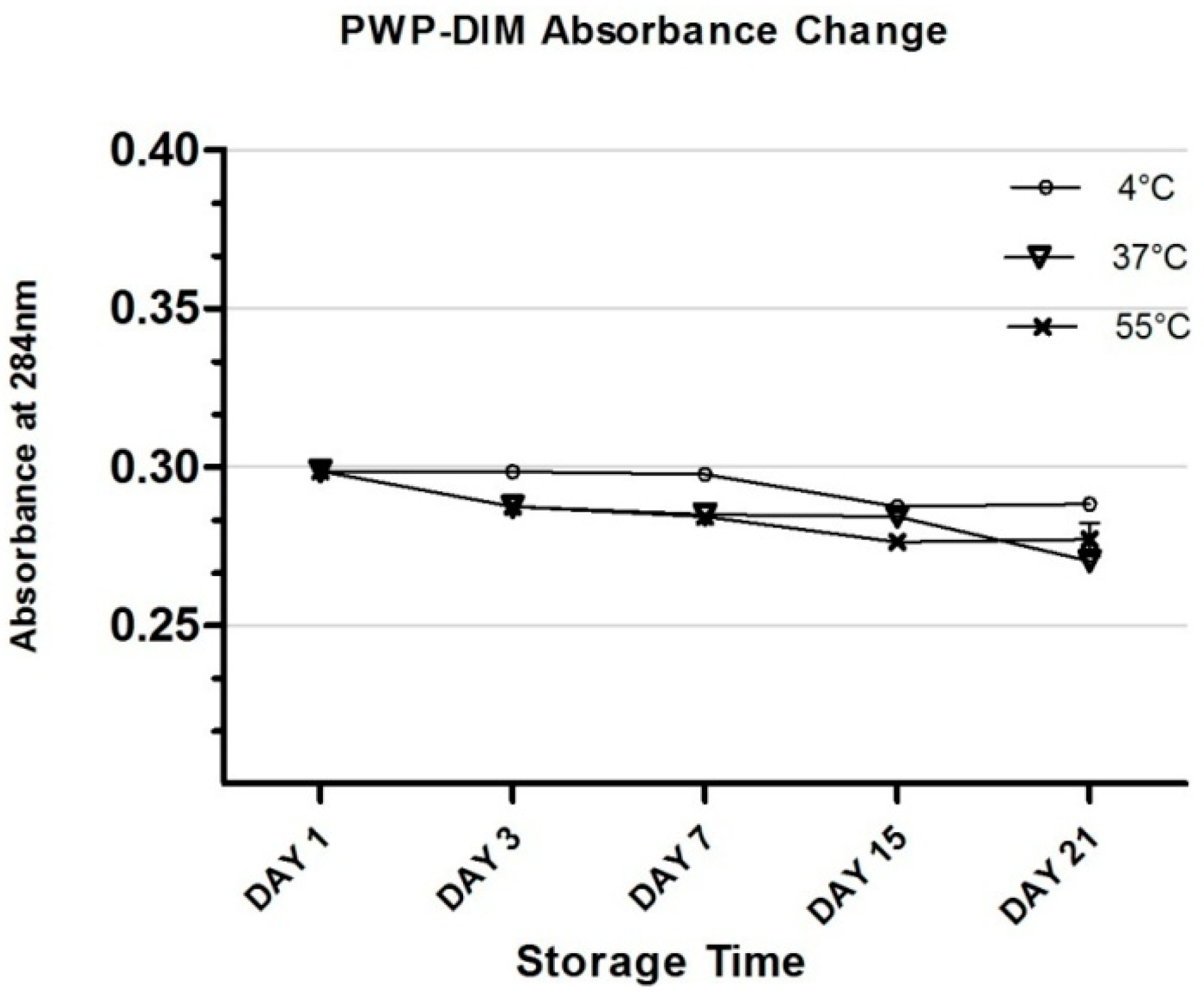
2.5. Changes in Color and Absorbance of PWP–DIM nanoparticles during Storage
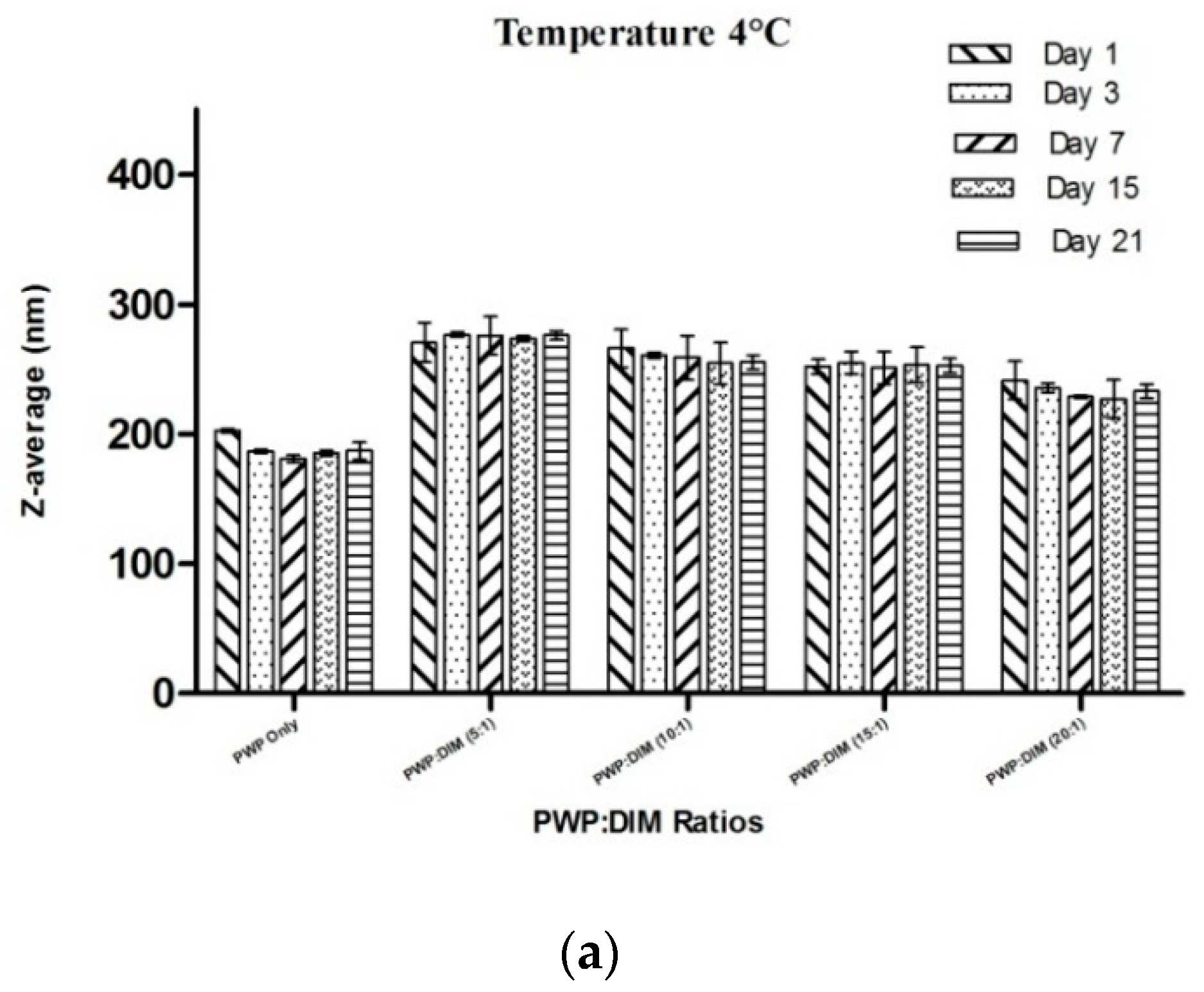
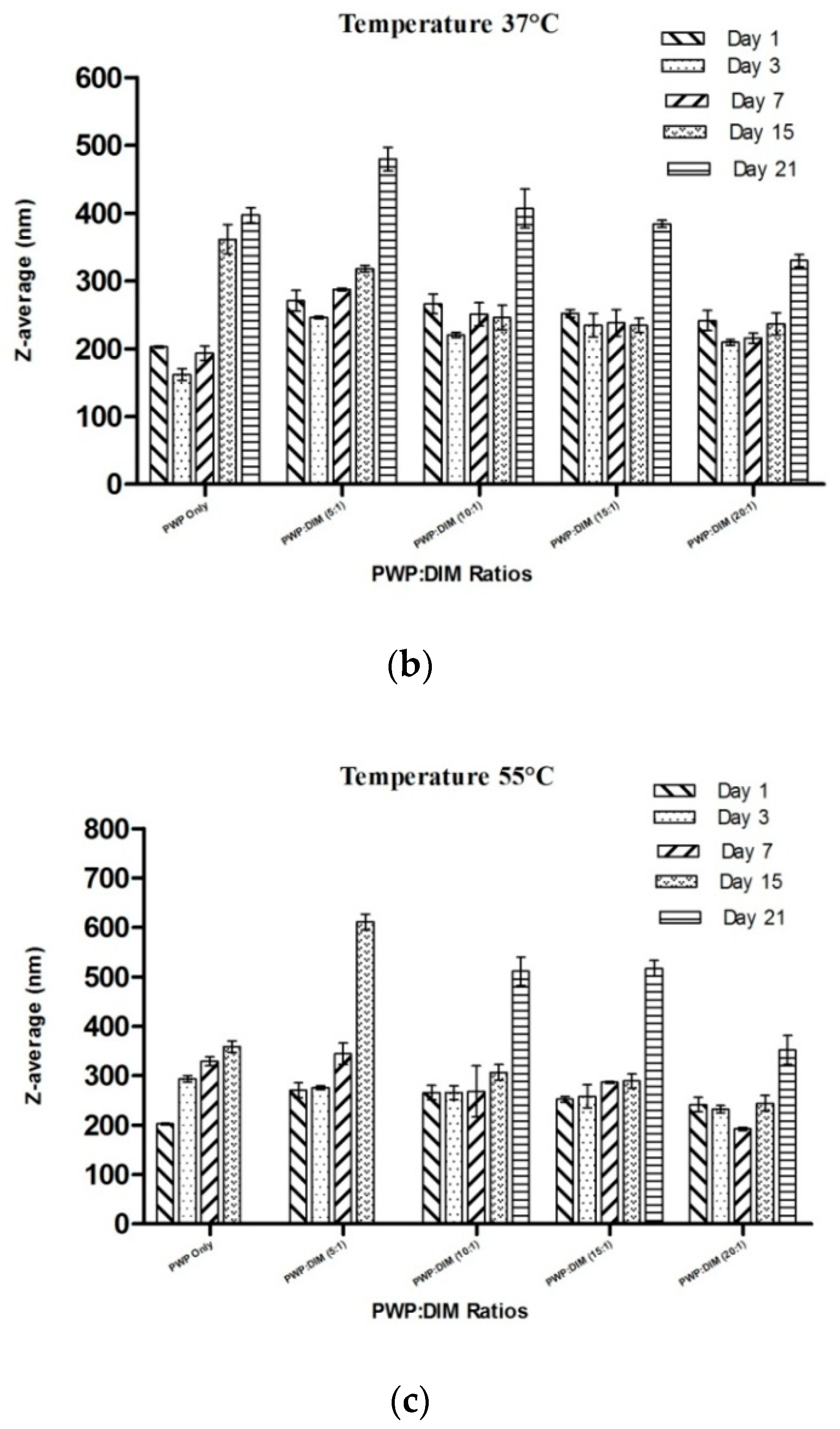
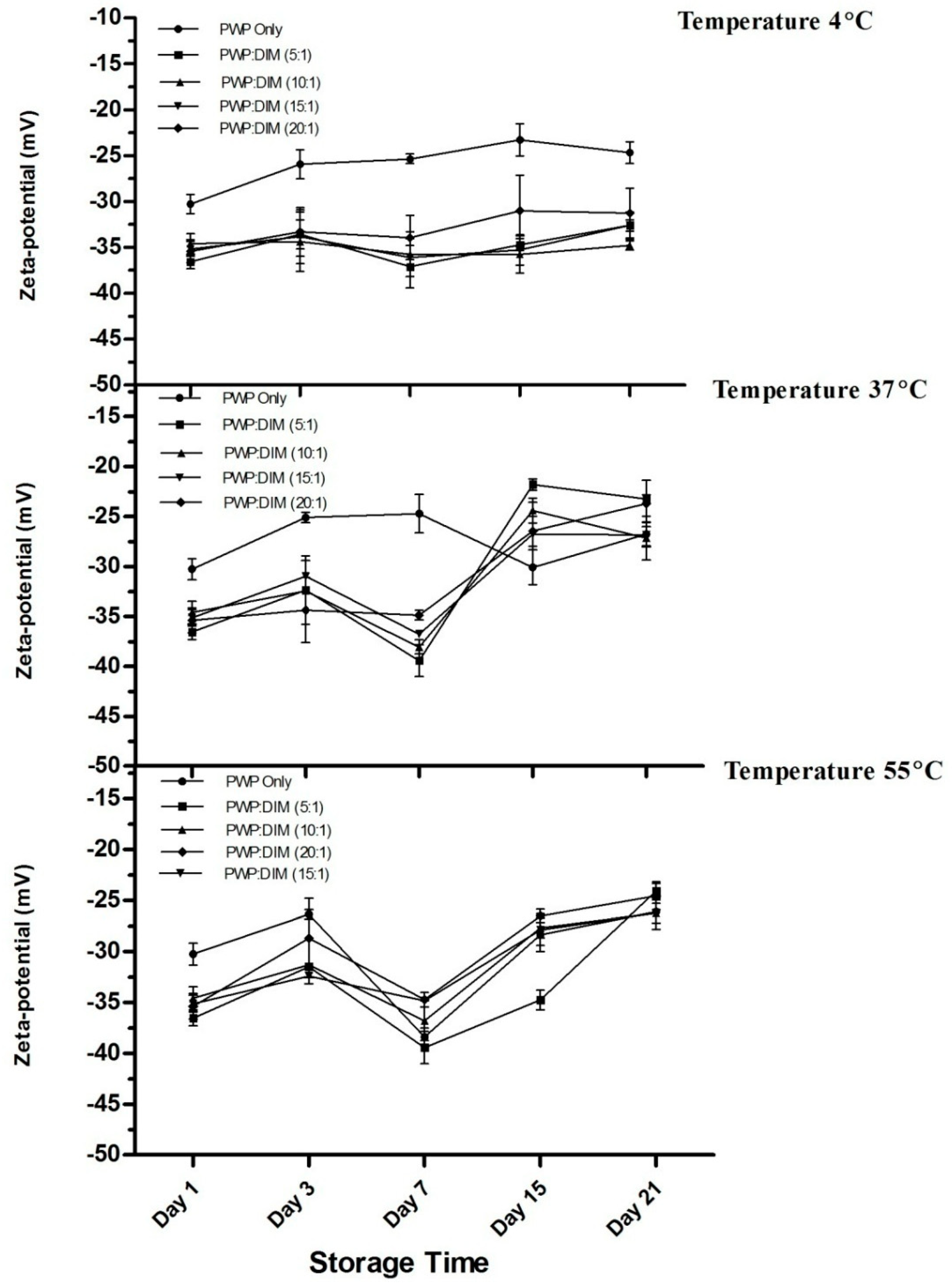
2.6. Changes in Particle Size and Zeta Potential of PWP–DIM Nanoparticles During Storage
3. Materials and Methods
3.1. Materials
3.2. Sample Preparation
3.3. Particle Size and Zeta Potential Measurement
3.4. Rheological Determinations
3.5. Fourier Transform Infrared (FTIR) Spectra Analysis
3.6. Transmission Electron Microscopy (TEM) Analysis
3.7. Storage Stability Analysis
3.7.1. Color and Absorbance Measurement
3.7.2. Mean Particle Size and Zeta Potential Determination
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Staub, R.E.; Onisko, B.; Bjeldanes, L.F. Fate of 3,3‘-Diindolylmethane in Cultured MCF-7 Human Breast Cancer Cells. Chem. Res. Toxicol. 2006, 19, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Chinni, S.R.; Li, Y.; Upadhyay, S.; Koppolu, P.K.; Sarkar, F.H. Indole-3-carbinol (I3C) induced cell growth inhibition, G1 cell cycle arrest and apoptosis in prostate cancer cells. Oncogene 2001, 20, 2927–2936. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.N.; Kucuk, O.; Djuric, Z.; Sarkar, F.H. Soy isoflavone supplementation in healthy men prevents NF-κB activation by TNF-α in blood lymphocytes. Free Radic. Biol. Med. 2001, 30, 1293–1302. [Google Scholar] [CrossRef]
- Andey, T.; Patel, A.; Jackson, T.; Safe, S.; Singh, M. 1,1-Bis (3′-indolyl)-1-(p-substitutedphenyl)methane compounds inhibit lung cancer cell and tumor growth in a metastasis model. Eur. J. Pharm. Sci. 2013, 50, 227–241. [Google Scholar] [CrossRef] [PubMed]
- York, M.; Abdelrahim, M.; Chintharlapalli, S.; Lucero, S.D.; Safe, S. 1,1-Bis(3′-Indolyl)-1-(p-Substitutedphenyl)methanes Induce Apoptosis and Inhibit Renal Cell Carcinoma Growth. Clin. Cancer Res. 2007, 13, 6743–6752. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Zou, X.; Feng, X. 3,3′-Diindolylmethane negatively regulates Cdc25A and induces a G2/M arrest by modulation of microRNA 21 in human breast cancer cells. Anti-Cancer Drugs 2010, 21, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Rajoria, S.; Suriano, R.; Parmar, P.S.; Wilson, Y.L.; Megwalu, U.; Moscatello, A.; Tiwari, R.K. 3,3′-Diindolylmethane Modulates Estrogen Metabolism in Patients with Thyroid Proliferative Disease: A Pilot Study. Thyroid 2011, 21, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Gajbhiye, R.; Mandal, M.; Pal, C.; Meyyapan, A.; Mukherjee, J.; Jaisankar, P. Synthesis and antibacterial evaluation of 3,3′-diindolylmethane derivatives. Med. Chem. Res. 2013, 23, 1371–1377. [Google Scholar] [CrossRef]
- Jellinck, P.H.; Makin, H.L.J.; Sepkovic, D.W.; Bradlow, H.L. Influence of indole carbinols and growth hormone on the metabolism of 4-androstenedione by rat liver microsomes. J. Steroid Biochem. Mol. Biol. 1993, 46, 791–798. [Google Scholar] [CrossRef]
- Yang, Z.; Peng, Z.; Li, J.; Li, S.; Kong, L.; Li, P.; Wang, Q. Development and evaluation of novel flavour microcapsules containing vanilla oil using complex coacervation approach. Food Chem. 2014, 145, 272–277. [Google Scholar] [CrossRef]
- Patel, A.; Hu, Y.; Tiwari, J.K.; Velikov, K.P. Synthesis and characterisation of zein–curcumin colloidal particles. Soft Matter 2010, 6, 6192. [Google Scholar] [CrossRef]
- Coimbra, M.; Isacchi, B.; van Bloois, L.; Torano, J.S.; Ket, A.; Wu, X.; Schiffelers, R.M. Improving solubility and chemical stability of natural compounds for medicinal use by incorporation into liposomes. Int. J. Pharm. 2011, 416, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Guenier, A.S.; Salmieri, S.; Lacroix, M. Alginate and Chitosan Functionalization for Micronutrient Encapsulation. J. Agric. Food Chem. 2008, 56, 2528–2535. [Google Scholar] [CrossRef]
- Oidtmann, J.; Schantz, M.; Mäder, K.; Baum, M.; Berg, S.; Betz, M.; Richling, E. Preparation and Comparative Release Characteristics of Three Anthocyanin Encapsulation Systems. J. Agric. Food Chem. 2012, 60, 844–851. [Google Scholar] [CrossRef]
- Desai, K.G.H.; Jin Park, H. Recent Developments in Microencapsulation of Food Ingredients. Dry. Technol. 2005, 23, 1361–1394. [Google Scholar] [CrossRef]
- Borgogna, M.; Bellich, B.; Zorzin, L.; Lapasin, R.; Cesàro, A. Food microencapsulation of bioactive compounds: Rheological and thermal characterisation of non-conventional gelling system. Food Chem. 2010, 122, 416–423. [Google Scholar] [CrossRef]
- Madene, A.; Jacquot, M.; Scher, J.; Desobry, S. Flavour encapsulation and controlled release—A review. Int. J. Food Sci. Technol. 2006, 41, 1–21. [Google Scholar] [CrossRef]
- Jain, K.; Kesharwani, P.; Gupta, U.; Jain, N.K. A review of glycosylated carriers for drug delivery. Biomaterials 2012, 33, 4166–4186. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, W.C.; Chang, C.P.; Gao, Y.L. Controlled release properties of chitosan encapsulated volatile Citronella Oil microcapsules by thermal treatments. Coll. Surf. B Biointerfaces 2006, 53, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Reed, G.A.; Sunega, J.M.; Sullivan, D.K.; Gray, J.C.; Mayo, M.S.; Crowell, J.A.; Hurwitz, A. Single-Dose Pharmacokinetics and Tolerability of Absorption-Enhanced 3,3′-Diindolylmethane in Healthy Subjects. Cancer Epidemiol. Biomark. Prev. 2008, 17, 2619–2624. [Google Scholar] [CrossRef]
- Siddique, M.A.B.; Maresca, P.; Pataro, G.; Ferrari, G. Effect of pulsed light treatment on structural and functional properties of whey protein isolate. Food Res. Int. 2016, 87, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, A.; Emam-Djomeh, Z.; Mousavi, M.A.E.; Davoodi, D. Stability of vitamin D3 encapsulated in nanoparticles of whey protein isolate. Food Chem. 2014, 143, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-R.; Chang, Y.H. Microencapsulation of gallic acid through the complex of whey protein concentrate-pectic polysaccharide extracted from Ulmus Davidiana. Food Hydrocoll. 2018, 85, 222–228. [Google Scholar] [CrossRef]
- Wang, M.; Gao, F.; Zheng, H.; Zhang, T.; Guo, M. Microencapsulation of ginsenosides using polymerised whey protein (PWP) as wall material and its application in probiotic fermented milk. Int. J. Food Sci. Technol. 2017, 52, 1009–1017. [Google Scholar] [CrossRef]
- Chen, L.; Subirade, M. Alginate–whey protein granular microspheres as oral delivery vehicles for bioactive compounds. Biomaterials 2006, 27, 4646–4654. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gao, F.; Zhang, T.; Wang, Y.; Guo, M. Physiochemical, textural, sensory properties and probiotic survivability of Chinese LaosuanNai (protein-fortified set yoghurt) using polymerised whey protein as a co-thickening agent. Int. J. Dairy Technol. 2015, 68, 261–269. [Google Scholar] [CrossRef]
- Hoffmann, M.A.M.; van Mil, P.J.J.M. Heat-Induced Aggregation of β-Lactoglobulin: Role of the Free Thiol Group and Disulfide Bonds. J. Agric Food Chem. 1997, 45, 2942–2948. [Google Scholar] [CrossRef]
- Mensi, A.; Choiset, Y.; Haertlé, T.; Reboul, E.; Borel, P.; Guyon, C.; Chobert, J.M. Interlocking of β-carotene in beta-lactoglobulin aggregates produced under high pressure. Food Chem. 2013, 139, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Rosenberg, M. Microencapsulation of theophylline in whey proteins: Effects of core-to-wall ratio. Int. J. Pharm. 2000, 205, 147–158. [Google Scholar] [CrossRef]
- Sari, T.P.; Mann, B.; Sharma, R.; Kumar, R.; Vikrant, M. Process Optimization for the production of nanoencapsulated curcumin and analysis for physicochemical characteristics and antioxidant mechanism. Int. J. Biotechnol. Bioeng. Res. 2013, 4, 581–586. [Google Scholar]
- Mcclements, D.J.; Rao, J. Food-grade nanoemulsions: Formulation, fabrication, properties, performance, biological fate, and potential toxicity. Crit. Rev. Food Sci. Nutr. 2011, 51, 285–330. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Gao, Y.; Li, J.; Subirade, M.; Song, Y.; Liang, L. Effect of resveratrol or ascorbic acid on the stability of α-tocopherol in O/W emulsions stabilized by whey protein isolate: Simultaneous encapsulation of the vitamin and the protective antioxidant. Food Chem. 2016, 196, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Sezgin-Bayindir, Z.; Antep, M.N.; Yuksel, N. Development and Characterization of Mixed Niosomes for Oral Delivery Using Candesartan Cilexetil as a Model Poorly Water-Soluble Drug. AAPS Pharm. SciTech. 2014, 16, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Muller, R.H. Zeta-Potential and Particle Charge in the Laboratory Practice and Introductionto Theory, Practical and Data Interpretation; Wissenschaftliche Verlagsgesellschaft: Stuttgart, Germany, 1996; pp. 104–108. [Google Scholar]
- Freitas, C.; Müller, R.H. Effect of light and temperature on zeta potential and physical stability in solid lipid nanoparticle (SLN™) dispersions. Int. J. Pharm. 1998, 168, 221–229. [Google Scholar] [CrossRef]
- Castel, V.; Rubiolo, A.C.; Carrara, C.R. Droplet size distribution, rheological behavior and stability of corn oil emulsions stabilized by a novel hydrocolloid (Brea gum) compared with gum arabic. Food Hydrocoll. 2017, 63, 170–177. [Google Scholar] [CrossRef]
- Dokić, L.; Krstonošić, V.; Nikolić, I. Physicochemical characteristics andstability of oil-in-water emulsions stabilized by OSA starch. Food Hydrocoll. 2012, 29, 185–192. [Google Scholar] [CrossRef]
- Domian, E.; Brynda-Kopytowska, A.; Oleksza, K. Rheological properties andphysical stability of o/w emulsions stabilized by OSA starch with trehalose. Food Hydrocoll. 2015, 44, 49–58. [Google Scholar]
- Jafari, S.M.; Beheshti, P.; Assadpoor, E. Rheological behavior and stability of D limonene emulsions made by a novel hydrocolloid (Angum gum) comparedwith Arabic gum. J. Food Eng. 2012, 109, 1–8. [Google Scholar]
- Sisko, A.W. The Flow of Lubricating Greases. A relatively simple flowequation is used to fit experimental flow data, and can be applied to other materials as well as greases. Ind. Eng. Chem. 1958, 50, 1789–1792. [Google Scholar] [CrossRef]
- Shen, X.; Fang, T.; Gao, F.; Guo, M. Effects of ultrasound treatment on physicochemical and emulsifying properties of whey proteins pre- and post-thermal aggregation. Food Hydrocoll. 2017, 63, 668–676. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, T.T.Y.; Teng, Z.; Chen, P.; Sun, J.; Wang, Q. Encapsulation of indole-3-carbinol and 3,3′-diindolylmethane in zein/carboxymethyl chitosan nanoparticles with controlled release property and improved stability. Food Chem. 2013, 139, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, L.; Madadlou, A.; Yarmand, M.; Mousavi, M.E. Nanoencapsulation of date palm pit extract in whey protein particles generated via desolvation method. Food. Res. Int. 2013, 51, 866–871. [Google Scholar] [CrossRef]
- Teng, Z.; Luo, Y.; Wang, Q. Carboxymethyl chitosan–soy protein complex nanoparticles for the encapsulation and controlled release of vitamin D3. Food Chem. 2013, 141, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.N. Stability testing of nutraceuticals and functional foods. In Handbook of Nutraceuticals and Functional Foods; Wildman, R.E.C., Ed.; CRC Press: New York, NY, USA, 2001; pp. 501–516. [Google Scholar]
- Geara, C. Study of the Gelation of Whey Protein Isolate by FTIR Spectroscopy and Rheological Measurements. Master’s Thesis, McGill University, Montreal, QC, Canada, 1999. [Google Scholar]
- Kretschmer, C.B. Infrared Spectroscopy and Optical Rotatory Dispersion of Zein, Wheat Gluten and Gliadin. J. Phys. Chem. 1957, 61, 1627–1631. [Google Scholar] [CrossRef]
- Sessa, D.J.; Mohamed, A.; Byars, J.A. Chemistry and Physical Properties of Melt-Processed and Solution-Cross-Linked Corn Zein. J. Agri. Food Chem. 2008, 56, 7067–7075. [Google Scholar] [CrossRef] [PubMed]
- Hosseinnia, M.; Khaledabad, M.A.; Almasi, H. Optimization of Ziziphoraclinopodiodes essential oil microencapsulation by whey protein isolate and pectin: A comparative study. Int. J. Biol. Macromol. 2017, 101, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Teng, Z.; Wang, Q. Development of Zein Nanoparticles Coated with Carboxymethyl Chitosan for Encapsulation and Controlled Release of Vitamin D3. J. Agric. Food Chem. 2012, 60, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Boon, C.S.; McClements, D.J.; Weiss, J.; Decker, E.A. Factors influencing thechemical stability of carotenoids in foods. Crit. Rev. Food Sci. Nutr. 2010, 50, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Davidov-Pardo, G.; Gumus, C.E.; McClements, D.J. Lutein-enriched emulsion-based delivery systems: Influence of pH and temperature on physical and chemical stability. Food Chem. 2016, 196, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Mehrad, B.; Ravanfar, R.; Licker, J.; Regenstein, J.M.; Abbaspourrad, A. Enhancing the physicochemical stability of β-carotene solid lipid nanoparticle (SLNP) using whey protein isolate. Food Res. Int. 2018, 105, 962–969. [Google Scholar] [CrossRef] [PubMed]
- Gumus, C.E.; Davidov-Pardo, G.; McClements, D.J. Lutein-enriched emulsion-based delivery systems: Impact of Maillard conjugation on physicochemical stability and gastrointestinal fate. Food Hydrocoll. 2016, 60, 38–49. [Google Scholar] [CrossRef]
- Mcclements, D.J. Food emulsions: Principles, practices, and techniques. Int. J. Food Sci Technol. 2005, 36, 223–224. [Google Scholar]
- Zhao, C.; Shen, X.; Guo, M. Stability of lutein encapsulated whey protein nano-emulsion during storage. PLOS ONE 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Decker, E.; McClements, D. Role of postadsorptionconformationchanges of β-lactoglobulin on its ability to stabilize oil droplets against flocculationduring heating at neutral pH. Langmuir 2002, 18, 7577–7583. [Google Scholar] [CrossRef]
- McClements, D.J. Protein-stabilized emulsions. Curr. Opin. Coll. Interface Sci. 2004, 9, 305–313. [Google Scholar] [CrossRef]
- Banks, W.; Muir, D.D. Effect of alcohol content on emulsion stability of cream liqueurs. Food Chem. 1985, 18, 139–152. [Google Scholar] [CrossRef]
- Chen, E.; Cao, L.; McClements, D.J.; Liu, S.; Li, B.; Li, Y. Enhancement of physicochemical properties of whey protein-stabilized nanoemulsions by interfacial cross-linking using cinnamaldehyde. Food Hydrocoll. 2018, 77, 976–985. [Google Scholar] [CrossRef]

| Sample | Dz (nm) | PDI | Zeta Potential (mV) |
|---|---|---|---|
| PWP Only | 202.50 ± 1.56 a | 0.37 ± 0.0 a | −30.26 ± 1.05 a |
| PWP:DIM (5:1) | 270.57 ± 15.28 b | 0.61 ± 0.05 b | −36.56 ± 0.73 b |
| PWP:DIM (10:1) | 265.97 ± 14.51 b | 0.42 ± 0.01 ca | −34.60 ± 1.12 b |
| PWP:DIM (15:1) | 252.00 ± 5.63 b | 0.44 ± 0.01 c | −35.13 ± 0.83 b |
| PWP:DIM (20:1) | 241.33 ± 14.82 b | 0.44 ± 0.05 c | −35.40 ± 1.24 b |
| Samples | η∞0 (mPa∙s) Infinite-Shear-Rate Viscosity | k0 Consistency Index | n0 Flow Index |
|---|---|---|---|
| PWP Only | 5.205 ± 0.014 a | 386.701 ± 10.141 ab | 0.016 ± 0.006 a |
| PWP:DIM (5:1) | 6.344 ± 0.011 b | 390.108 ± 6.4156 a | 0.046 ± 0.004 b |
| PWP:DIM (10:1) | 6.470 ± 0.023 c | 392.158 ± 16.327 ab | 0.010 ± 0.010 c |
| PWP:DIM (15:1) | 5.656 ± 0.011 d | 441.536 ± 8.677 c | −0.004± 0.005 d |
| PWP:DIM (20:1) | 5.340 ± 0.034 e | 537.473 ± 34.462 d | −0.049 ± 0.016 e |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, A.; Wang, C.; Sun, X.; Killpartrick, A.; Guo, M. Physicochemical and Microstructural Properties of Polymerized Whey Protein Encapsulated 3,3′-Diindolylmethane Nanoparticles. Molecules 2019, 24, 702. https://doi.org/10.3390/molecules24040702
Khan A, Wang C, Sun X, Killpartrick A, Guo M. Physicochemical and Microstructural Properties of Polymerized Whey Protein Encapsulated 3,3′-Diindolylmethane Nanoparticles. Molecules. 2019; 24(4):702. https://doi.org/10.3390/molecules24040702
Chicago/Turabian StyleKhan, Abbas, Cuina Wang, Xiaomeng Sun, Adam Killpartrick, and Mingruo Guo. 2019. "Physicochemical and Microstructural Properties of Polymerized Whey Protein Encapsulated 3,3′-Diindolylmethane Nanoparticles" Molecules 24, no. 4: 702. https://doi.org/10.3390/molecules24040702
APA StyleKhan, A., Wang, C., Sun, X., Killpartrick, A., & Guo, M. (2019). Physicochemical and Microstructural Properties of Polymerized Whey Protein Encapsulated 3,3′-Diindolylmethane Nanoparticles. Molecules, 24(4), 702. https://doi.org/10.3390/molecules24040702








