Discovery of a New Xanthone against Glioma: Synthesis and Development of (Pro)liposome Formulations
Abstract
:1. Introduction
2. Results and Discussion
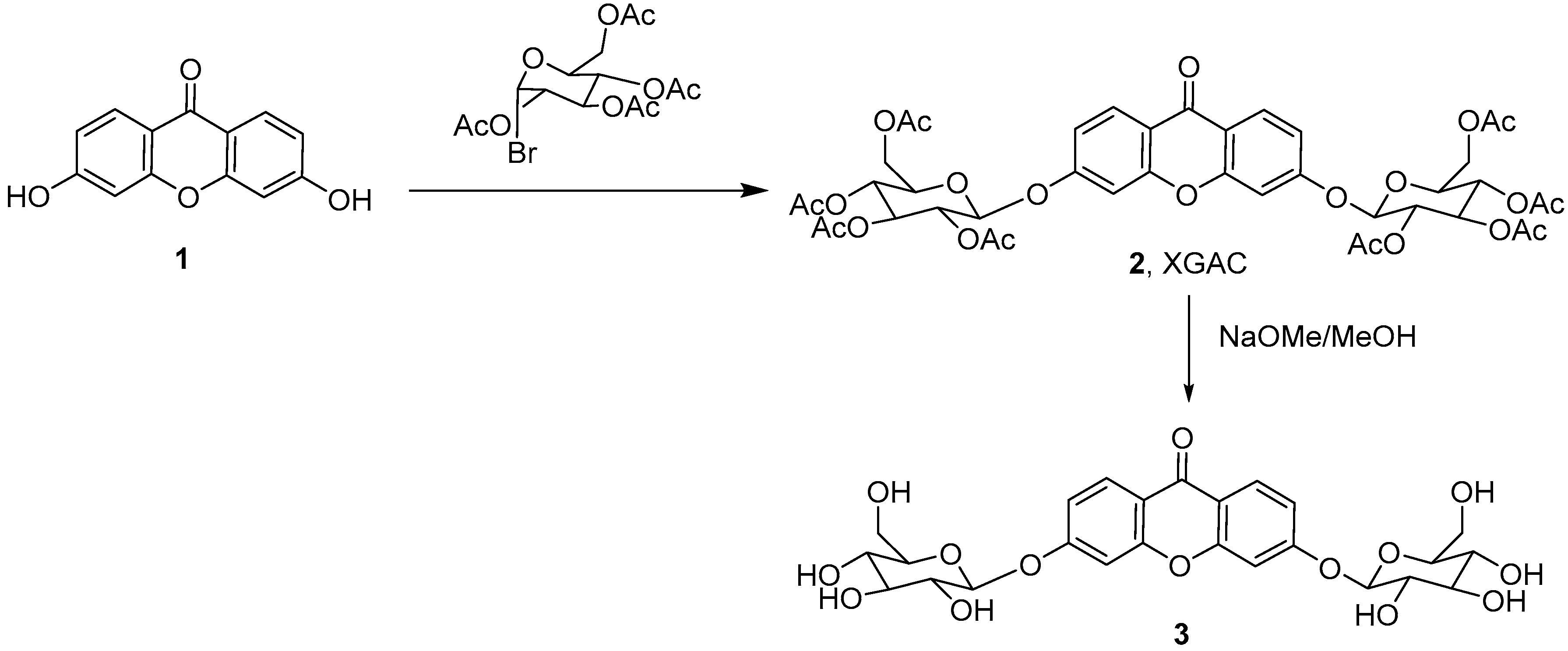
2.1. Synthesis and Structure Elucidation
2.2. Human Tumor Cell Lines Growth Inhibitory Activity of Xanthone (2)
2.3. Preparation of Proliposomes and Liposomes for Drug Delivery of Xanthone (2)
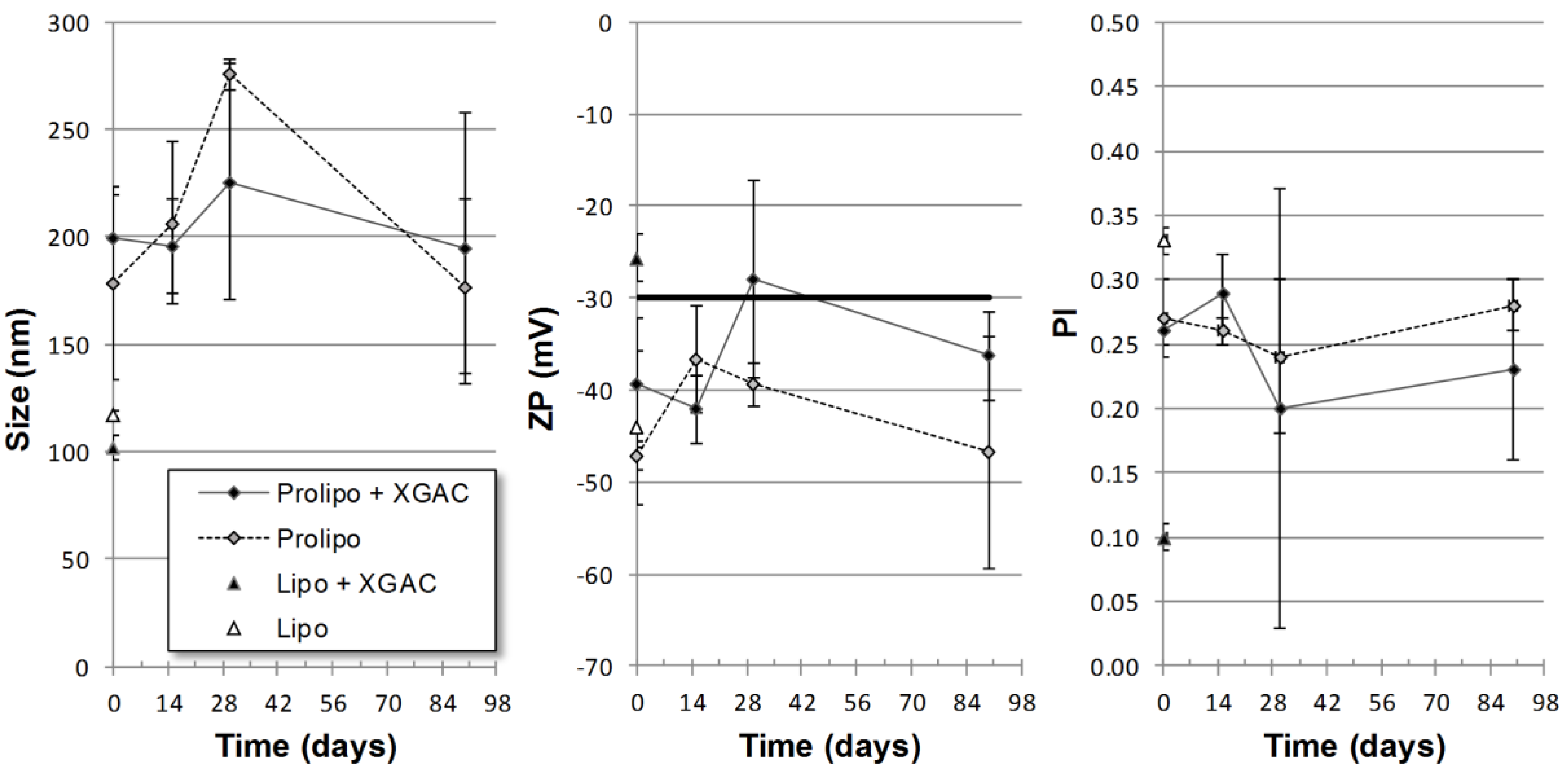
2.3.1. Particle Size, Polidispersity and Zeta Potential
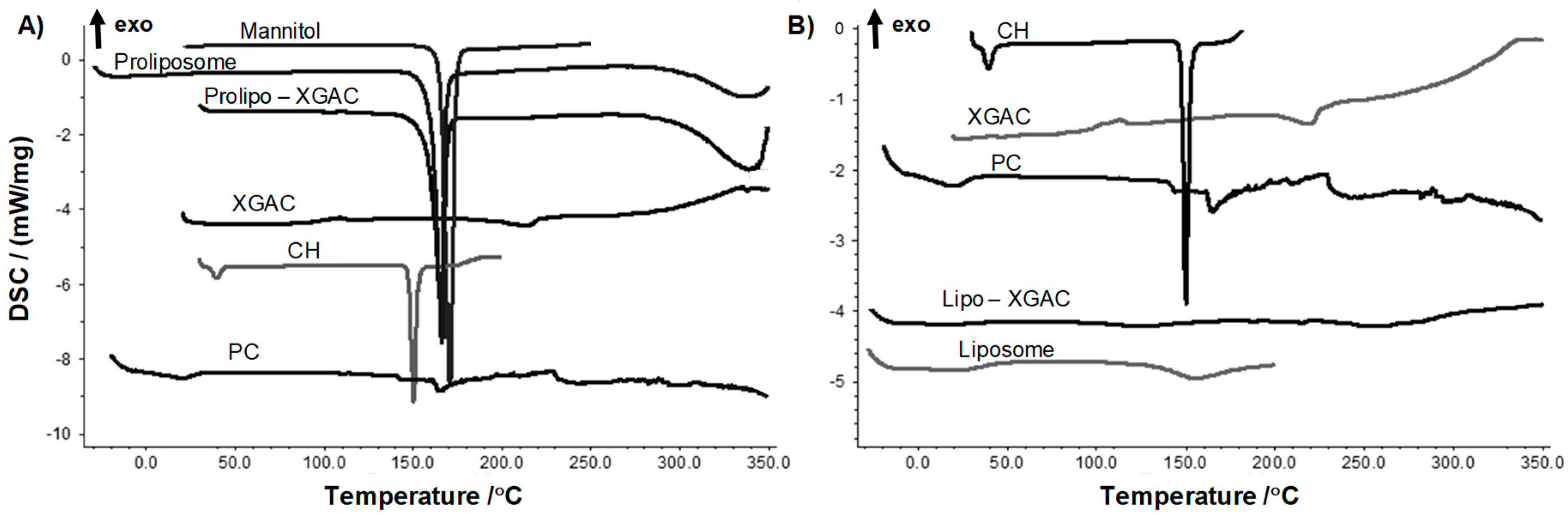
2.3.2. Thermal Behavior
2.3.3. Scanning Electron Microscopic Study
2.3.4. Entrapment Efficiency
2.3.5. Stability Study
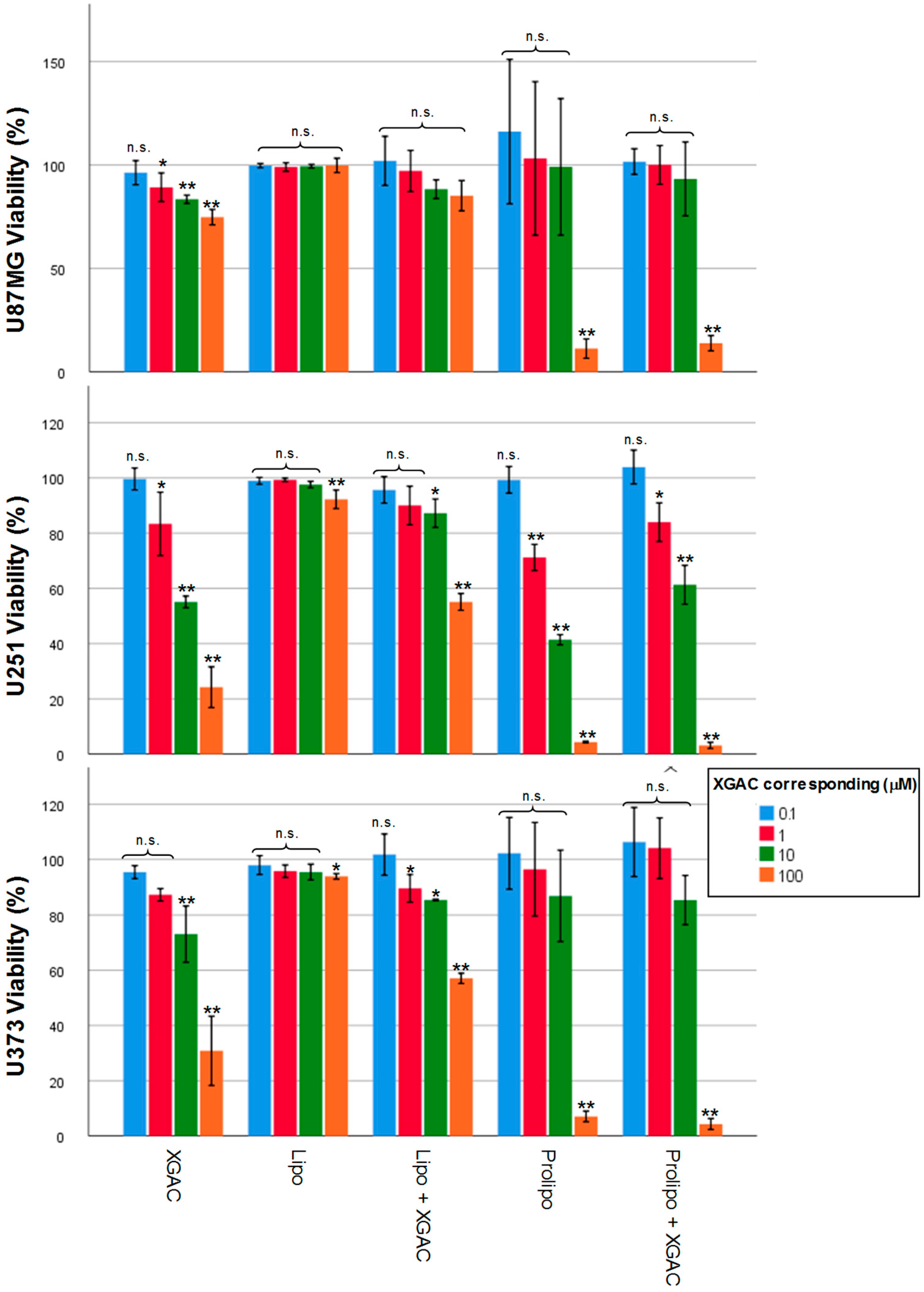
2.4. Tumor Cell Viability Effect with Xanthone 2 Formulations
3. Materials and Methods
3.1. General Information
3.2. Synthesis of 3,6-Bis(2,3,4,6-tetra-O-acetyl-β-glucopyranosyl)xanthone (2, XGAC)
3.3. Cell Lines and Culture Conditions
3.4. Tumor Cell Growth Assay
3.5. Preparation of Proliposomes
3.6. Preparation of Liposomes
3.7. Characterization of Proliposomes and Liposomes
3.7.1. Hydration Study
3.7.2. Thermal Behavior
3.7.3. Scanning Electron Microscopic Study
3.7.4. Entrapment Efficiency
3.7.5. Stability Study
3.7.6. MTT Cell Viability Assay
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, X.-Y.; Zhao, Y.; Sun, M.-G.; Shi, J.-F.; Ju, R.-J.; Zhang, C.-X.; Li, X.-T.; Zhao, W.-Y.; Mu, L.-M.; Zeng, F. Multifunctional liposomes loaded with paclitaxel and artemether for treatment of invasive brain glioma. Biomaterials 2014, 35, 5591–5604. [Google Scholar] [CrossRef] [PubMed]
- Peiris, P.M.; Abramowski, A.; McGinnity, J.; Doolittle, E.; Toy, R.; Gopalakrishnan, R.; Shah, S.; Bauer, L.; Ghaghada, K.B.; Hoimes, C.; et al. Treatment of Invasive Brain Tumors Using a Chain-like Nanoparticle. Cancer Res. 2015, 75, 1356–1365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wrensch, M.; Minn, Y.; Chew, T.; Bondy, M.; Berger, M.S. Epidemiology of primary brain tumors: Current concepts and review of the literature. Neuro-oncology 2002, 4, 278–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, H.; Wang, L.; Wang, J.; Weng, X.; Lei, H.; Wang, X.; Jiang, L.; Zhu, J.; Lu, W.; Wei, X.; et al. Two-order targeted brain tumor imaging by using an optical/paramagnetic nanoprobe across the blood brain barrier. Acs Nano 2012, 6, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Svec, R.L.; Furiassi, L.; Skibinski, C.G.; Fan, T.M.; Riggins, G.J.; Hergenrother, P.J. Tunable Stability of Imidazotetrazines Leads to a Potent Compound for Glioblastoma. Acs Chem. Biol. 2018, 13, 3206–3216. [Google Scholar] [CrossRef] [PubMed]
- Nordling-David, M.M.; Yaffe, R.; Guez, D.; Meirow, H.; Last, D.; Grad, E.; Salomon, S.; Sharabi, S.; Levi-Kalisman, Y.; Golomb, G.; et al. Liposomal temozolomide drug delivery using convection enhanced delivery. J. Control. Release 2017, 261, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.M.; Sousa, M.E.; Nascimento, M.S. Xanthone derivatives: New insights in biological activities. Curr. Med. Chem. 2005, 12, 2517–2538. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef]
- Neves, A.; Correia-da-Silva, M.; Silva, P.; Ribeiro, D.; Sousa, E.; Bousbaa, H.; Pinto, M. Synthesis of New Glycosylated Flavonoids with Inhibitory Activity on Cell Growth. Molecules 2018, 23, 1093. [Google Scholar] [CrossRef]
- Desai, V.; Bhushan, A. Natural Bioactive Compounds: Alternative Approach to the Treatment of Glioblastoma Multiforme. Biomed Res. Int. 2017, 2017, 9363040. [Google Scholar] [CrossRef]
- Suda, I.; Oki, T.; Masuda, M.; Nishiba, Y.; Furuta, S.; Matsugano, K.; Sugita, K.; Terahara, N. Direct absorption of acylated anthocyanin in purple-fleshed sweet potato into rats. J. Agric. Food Chem. 2002, 50, 1672–1676. [Google Scholar] [CrossRef] [PubMed]
- Wezeman, T.; Brase, S.; Masters, K.S. Xanthone dimers: A compound family which is both common and privileged. Nat. Prod. Rep. 2015, 32, 6–28. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.S.; Brandao, P.; Fernandes, C.S.G.; da Silva, M.; de Sousa, M.; Pinto, M.M.M. Drug-like Properties and ADME of Xanthone Derivatives: The Antechamber of Clinical Trials. Curr. Med. Chem. 2016, 23, 3654–3686. [Google Scholar] [CrossRef] [PubMed]
- Bangham, A.; Standish, M.M.; Watkins, J. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252, IN26–IN27. [Google Scholar] [CrossRef]
- Frezard, F.; Schettini, D.A.; Rocha, O.G.F.; Demicheli, C. Liposomes: Physicochemical and pharmacological properties, applications in antimony-based chemotherapy. Quim. Nova 2005, 28, 511–518. [Google Scholar]
- Batista, C.M.; de Carvalho, C.M.B.; Magalhães, N.S.S. Lipossomas e suas aplicações terapêuticas: Estado da arte. Rev. Bras. De Ciências Farm. 2007, 43, 167–179. [Google Scholar] [CrossRef]
- Machado, L.C.; Gnoatto, S.A.; Klüppel, M.L.W. Lipossomas aplicados em farmacologia: Uma revisão da literatura. Estud. Biol. 2007, 29, 215–224. [Google Scholar] [CrossRef]
- Allen, T.; Hansen, C.; Martin, F.; Redemann, C.; Yau-Young, A. Liposomes containing synthetic lipid derivatives of poly (ethylene glycol) show prolonged circulation half-lives in vivo. Biochim. Biophys. Acta (Bba)-Biomembr. 1991, 1066, 29–36. [Google Scholar] [CrossRef]
- Kim, S.S.; Harford, J.B.; Pirollo, K.F.; Chang, E.H. Effective treatment of glioblastoma requires crossing the blood-brain barrier and targeting tumors including cancer stem cells: The promise of nanomedicine. Biochem. Biophys. Res. Commun. 2015, 468, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.L.; Huang, M.; Wang, X.R.; Fu, J.; Han, M.; Shen, Y.Q.; Xia, Z.; Gao, J.Q. Transferrin-modified liposome promotes alpha-mangostin to penetrate the blood-brain barrier. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 421–430. [Google Scholar] [CrossRef]
- Payne, N.I.; Ambrose, C.V.; Timmins, P.; Ward, M.D.; Ridgway, F. Proliposomes: A Novel Solution to an Old Problem. J. Pharm. Sci. 1986, 75, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Dua, J.; Rana, A.; Bhandari, A. Liposome: Methods of preparation and applications. Int. J. Pharm. Stud. Res. 2012, 3, 14–20. [Google Scholar]
- Janga, K.Y.; Jukanti, R.; Velpula, A.; Sunkavalli, S.; Bandari, S.; Kandadi, P.; Veerareddy, P.R. Bioavailability enhancement of zaleplon via proliposomes: Role of surface charge. Eur. J. Pharm. Biopharm. 2012, 80, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Falconer, J.R.; Svirskis, D.; Adil, A.A.; Wu, Z. Supercritical Fluid Technologies to Fabricate Proliposomes. J. Pharm. Pharm. Sci. A Publ. Can. Soc. Pharm. Sci. Soc. Can. Des. Sci. Pharm. 2015, 18, 747–764. [Google Scholar] [CrossRef] [Green Version]
- Silva Moreira, G. Desenvolvimento de Prolipossomas com Paclitaxel: Uma Nova Abordagem na Terapia do Cancro. Master’s Thesis, University of Porto Faculty of Pharmacy, Porto, Portugal, 2015. [Google Scholar]
- Moreira, A. Development of Proliposomes as a Vehicle to Deliver New Molecules with Antitumor Activity. Master’s Thesis, University of Porto Faculty of Pharmacy, Porto, Portugal, 2015. [Google Scholar]
- Jäger, A.; Stefani, V.; Guterres, S.S.; Pohlmann, A.R. Physico-chemical characterization of nanocapsule polymeric wall using fluorescent benzazole probes. Int. J. Pharm. 2007, 338, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Rojanarat, W.; Changsan, N.; Tawithong, E.; Pinsuwan, S.; Chan, H.K.; Srichana, T. Isoniazid proliposome powders for inhalation-preparation, characterization and cell culture studies. Int. J. Mol. Sci. 2011, 12, 4414–4434. [Google Scholar] [CrossRef] [PubMed]
- Liua, Q.; Sua, Q.; Luoa, G.; Wanga, Y.; Zanga, Q. Pharmacokinetics and tissue distribution of carboplatin liposomes after intravenous administration to mice. Asian J. Pharm. Sci. 2006, 1, 159–167. [Google Scholar]
- Srinath, P.; Chary, M.; Vyas, S.; Diwan, P. Long-circulating liposomes of indomethacin in arthritic rats—A biodisposition study. Pharm. Acta Helv. 2000, 74, 399–404. [Google Scholar] [CrossRef]
- Zhao, L.; Xiong, H.; Peng, H.; Wang, Q.; Han, D.; Bai, C.; Liu, Y.; Shi, S.; Deng, B. PEG-coated lyophilized proliposomes: Preparation, characterizations and in vitro release evaluation of vitamin E. Eur. Food Res. Technol. 2011, 232, 647–654. [Google Scholar] [CrossRef]
- Zhao, L.; Wei, Y.M.; Zhong, X.D.; Liang, Y.; Zhang, X.M.; Li, W.; Li, B.B.; Wang, Y.; Yu, Y. PK and tissue distribution of docetaxel in rabbits after i.v. administration of liposomal and injectable formulations. J. Pharm. Biomed. Anal. 2009, 49, 989–996. [Google Scholar] [CrossRef]
- Blum, R.H.; Carter, S.K. Adriamycin. A new anticancer drug with significant clinical activity. Ann. Intern. Med. 1974, 80, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Ana, P.; Sousa, M. Development and validation of a HPLC method for the quantification of a cytotoxic dihydropyranoxanthone in biodegradable nanoparticles. Int. J. Drug Deliv. 2013, 5, 224–232. [Google Scholar]
- Paiva, A.M.; Pinto, R.A.; Teixeira, M.; Barbosa, C.M.; Lima, R.T.; Vasconcelos, M.H.; Sousa, E.; Pinto, M. Development of noncytotoxic PLGA nanoparticles to improve the effect of a new inhibitor of p53-MDM2 interaction. Int. J. Pharm. 2013, 454, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Correia-da-Silva, M.; Sousa, E.; Duarte, B.; Marques, F.; Carvalho, F.; Cunha-Ribeiro, L.M.; Pinto, M.M.M. Polysulfated Xanthones: Multipathway Development of a New Generation of Dual Anticoagulant/Antiplatelet Agents. J. Med. Chem. 2011, 54, 5373–5384. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.; Sousa, E.; Nazareth, N.; S.J. Nascimento, M.; M.M. Pinto, M. Synthesis of Xanthones and Benzophenones as Inhibitors of Tumor Cell Growth. Lett. Drug Des. Discov. 2010, 7, 487–493. [Google Scholar] [CrossRef]
- Barba, A.A.; Bochicchio, S.; Lamberti, G.; Dalmoro, A. Ultrasonic energy in liposome production: Process modelling and size calculation. Soft Matter 2014, 10, 2574–2581. [Google Scholar] [CrossRef] [PubMed]
- Stylianopoulos, T. EPR-effect: Utilizing size-dependent nanoparticle delivery to solid tumors. Ther. Deliv. 2013, 4, 421–423. [Google Scholar] [CrossRef]
- Chauhan, V.P.; Stylianopoulos, T.; Martin, J.D.; Popovic, Z.; Chen, O.; Kamoun, W.S.; Bawendi, M.G.; Fukumura, D.; Jain, R.K. Normalization of tumour blood vessels improves the delivery of nanomedicines in a size-dependent manner. Nat. Nanotechnol. 2012, 7, 383–388. [Google Scholar] [CrossRef] [Green Version]
- Chauhan, V.P.; Stylianopoulos, T.; Boucher, Y.; Jain, R.K. Delivery of molecular and nanoscale medicine to tumors: Transport barriers and strategies. Annu. Rev. Chem. Biomol. Eng. 2011, 2, 281–298. [Google Scholar] [CrossRef]
- Pluen, A.; Boucher, Y.; Ramanujan, S.; McKee, T.D.; Gohongi, T.; di Tomaso, E.; Brown, E.B.; Izumi, Y.; Campbell, R.B.; Berk, D.A.; et al. Role of tumor-host interactions in interstitial diffusion of macromolecules: Cranial vs. subcutaneous tumors. Proc. Natl. Acad. Sci. USA 2001, 98, 4628–4633. [Google Scholar] [CrossRef] [Green Version]
- Stetefeld, J.; McKenna, S.A.; Patel, T.R. Dynamic light scattering: A practical guide and applications in biomedical sciences. Biophys. Rev. 2016, 8, 409–427. [Google Scholar] [CrossRef] [PubMed]
- Rojanarat, W.; Nakpheng, T.; Thawithong, E.; Yanyium, N.; Srichana, T. Levofloxacin-proliposomes: Opportunities for use in lung tuberculosis. Pharmaceutics 2012, 4, 385–412. [Google Scholar] [CrossRef] [PubMed]
- Davidsen, J.; Rosenkrands, I.; Christensen, D.; Vangala, A.; Kirby, D.; Perrie, Y.; Agger, E.M.; Andersen, P. Characterization of cationic liposomes based on dimethyldioctadecylammonium and synthetic cord factor from M. tuberculosis (trehalose 6,6′-dibehenate)—A novel adjuvant inducing both strong CMI and antibody responses. Biochim. Biophys. Acta (Bba) Biomembr. 2005, 1718, 22–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gill, P.; Moghadam, T.T.; Ranjbar, B. Differential scanning calorimetry techniques: Applications in biology and nanoscience. J. Biomol. Tech. JBT 2010, 21, 167. [Google Scholar]
- Sun, C.; Wang, J.; Liu, J.; Qiu, L.; Zhang, W.; Zhang, L. Liquid proliposomes of nimodipine drug delivery system: Preparation, characterization, and pharmacokinetics. AAPS PharmSciTech 2013, 14, 332–338. [Google Scholar] [CrossRef]
- Gupta, V.; Barupal, A.K.; Ramteke, S. Formulation Development and in vitro Characterization of Proliposomes for Topical Delivery of Aceclofenac. Indian J. Pharm. Sci. 2008, 70, 768–775. [Google Scholar] [PubMed]
- Nekkanti, V.; Rueda, J.; Wang, Z.; Betageri, G.V. Design, Characterization, and In Vivo Pharmacokinetics of Tacrolimus Proliposomes. AAPS PharmSciTech 2016, 17, 1019–1029. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.G.; Ming, L.C.; Lee, K.S.; Yuen, K.H. Influence of the Encapsulation Efficiency and Size of Liposome on the Oral Bioavailability of Griseofulvin-Loaded Liposomes. Pharmaceutics 2016, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Bandari, S.; Gangishetty, S.; Eedara, B.B.; Jukanti, R. Proliposomes of Lisinopril Dihydrate for Transdermal Delivery: Formulation Aspects and Evaluation; Department of Pharmaceutics, St. Peter’s Institute of Pharmaceutical Sciences: Warangal, India, 2013; Volume 30, pp. 1659–1666. [Google Scholar]
- Omer, H.K.; Hussein, N.R.; Ferraz, A.; Najlah, M.; Ahmed, W.; Taylor, K.M.G.; Elhissi, A.M.A. Spray-Dried Proliposome Microparticles for High-Performance Aerosol Delivery Using a Monodose Powder Inhaler. AAPS PharmSciTech 2018, 19, 2434–2448. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wei, Q.; Yang, Q.; Cao, X.; Li, Q.; Shi, F.; Tong, S.S.; Feng, C.; Yu, Q.; Yu, J.; et al. A novel formulation of [6]-gingerol: Proliposomes with enhanced oral bioavailability and antitumor effect. Int. J. Pharm. 2018, 535, 308–315. [Google Scholar] [CrossRef]
- Geng, F.; Chai, H.; Ma, L.; Luo, G.; Li, Y.; Yuan, Z. Simulation of dynamic transport of flexible ribbon particles in a rotary dryer. Powder Technol. 2016, 297, 115–125. [Google Scholar] [CrossRef]
- Lasic, D.D. Liposomes: From Physics to Applications; Elsevier: New York, NY, USA, 1993. [Google Scholar]
- Nunes, C.; Brezesinski, G.; Pereira-Leite, C.; Lima, J.L.; Reis, S.; Lucio, M. NSAIDs interactions with membranes: A biophysical approach. Langmuir 2011, 27, 10847–10858. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Hu, D.; Jin, H.; Zhao, Y.; Liang, J. Preparation of lutein proliposomes by supercritical anti-solvent technique. Food Hydrocoll. 2012, 26, 456–463. [Google Scholar] [CrossRef]
- Berne, B.J.; Pecora, R. Dynamic Light Scattering: With Applications to Chemistry, Biology, and Physics; Courier Corporation; John Wiley & Sons: New York, NY, USA, 1976. [Google Scholar]
- Keller, B.C. Liposomes in nutrition. Trends Food Sci. Technol. 2001, 12, 25–31. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1, 2, and 3 are available from the authors. |


| Compound. | GI50 (μM) | |||||
|---|---|---|---|---|---|---|
| A375-C5 | MCF-7 | NCI-H460 | U251 | U373 | U87-MG | |
| 2 (XGAC) | 135 | 0.46 ± 0.19 | 0.19 ± 0.04 | 0.55 ± 0.04 | 0.42 ± 0.02 | 0.42 ± 0.07 |
| 3 | >150 | >150 | >150 | nd | nd | nd |
| Doxorubicin | 0.014 ± 0.002 | 0.009 ± 0.001 | 0.009 ± 0.002 | 0.011 ± 0.004 | 0.009 ± 0.001 | 0.010 ± 0.002 |
| Dispersion Method | Solvent | Effective Diameter (nm) |
|---|---|---|
| Ultrasonication | PBS | 173.8 ± 1.3 |
| Manual agitation | PBS | 243.8 ± 3.7 |
| Ultrasonication | Water | 163.3 ± 0.7 |
| Manual agitation | Water | 199.4 ± 20.6 |
| Formulation | Effective Diameter (nm) | PI | ZP (mV) |
|---|---|---|---|
| Prolipo + XGAC (2) | 199.4 ± 20.6 | 0.26 ± 0.01 | −39 ± 7 |
| Proliposome | 178.6± 45.0 | 0.27 ± 0.03 | −47 ± 2 |
| Lipo + XGAC (2) | 102.4 ± 2.1 | 0.10 ± 0.01 | −26 ± 3 |
| Liposome | 117.1 ± 5.8 | 0.33 ± 0.01 | −44 ± 9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alves, A.; Correia-da-Silva, M.; Nunes, C.; Campos, J.; Sousa, E.; Silva, P.M.A.; Bousbaa, H.; Rodrigues, F.; Ferreira, D.; Costa, P.C.; et al. Discovery of a New Xanthone against Glioma: Synthesis and Development of (Pro)liposome Formulations. Molecules 2019, 24, 409. https://doi.org/10.3390/molecules24030409
Alves A, Correia-da-Silva M, Nunes C, Campos J, Sousa E, Silva PMA, Bousbaa H, Rodrigues F, Ferreira D, Costa PC, et al. Discovery of a New Xanthone against Glioma: Synthesis and Development of (Pro)liposome Formulations. Molecules. 2019; 24(3):409. https://doi.org/10.3390/molecules24030409
Chicago/Turabian StyleAlves, Ana, Marta Correia-da-Silva, Claúdia Nunes, João Campos, Emília Sousa, Patrícia M.A. Silva, Hassan Bousbaa, Francisca Rodrigues, Domingos Ferreira, Paulo C. Costa, and et al. 2019. "Discovery of a New Xanthone against Glioma: Synthesis and Development of (Pro)liposome Formulations" Molecules 24, no. 3: 409. https://doi.org/10.3390/molecules24030409
APA StyleAlves, A., Correia-da-Silva, M., Nunes, C., Campos, J., Sousa, E., Silva, P. M. A., Bousbaa, H., Rodrigues, F., Ferreira, D., Costa, P. C., & Pinto, M. (2019). Discovery of a New Xanthone against Glioma: Synthesis and Development of (Pro)liposome Formulations. Molecules, 24(3), 409. https://doi.org/10.3390/molecules24030409













