Microscopic Characteristic and Chemical Composition Analysis of Three Medicinal Plants and Surface Frosts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characteristics of Three Medicinal Plants and Their Frosts
2.1.1. Characteristics of P. ostii and Its Frosts
2.1.2. Characteristics of H. officinalis and Its Frosts
2.1.3. Characteristics of A. lancea and Its Frosts
2.2. Chemical Analysis of P. ostii
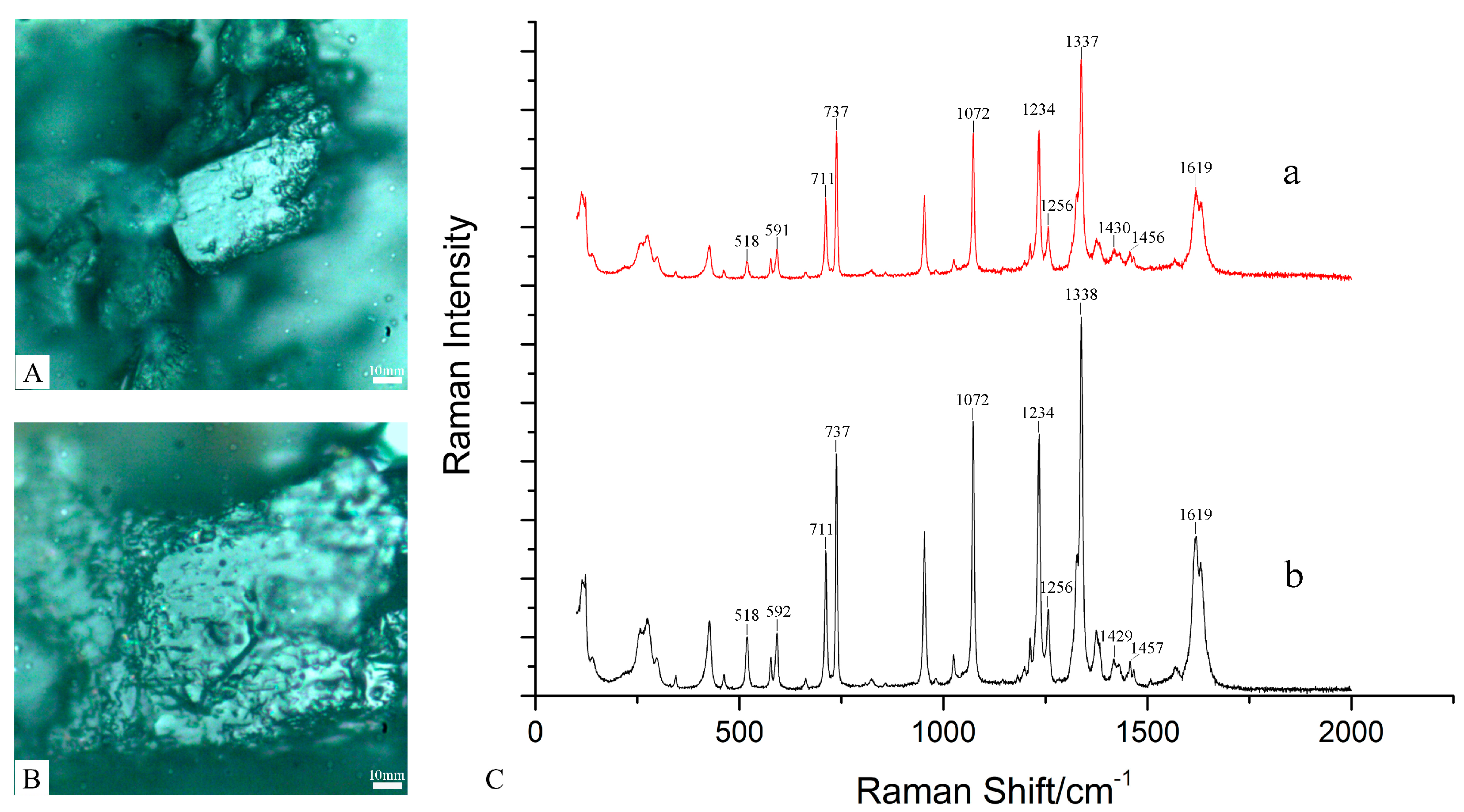
2.2.1. Raman Results of P. ostii
2.2.2. Metabolite Profiling by UPLC-Q/TOF-MS
2.3. Chemical Analysis of H. officinalis
2.3.1. Raman Results of H. officinalis
2.3.2. Metabolite Profiling by UPLC-Q Orbitrap
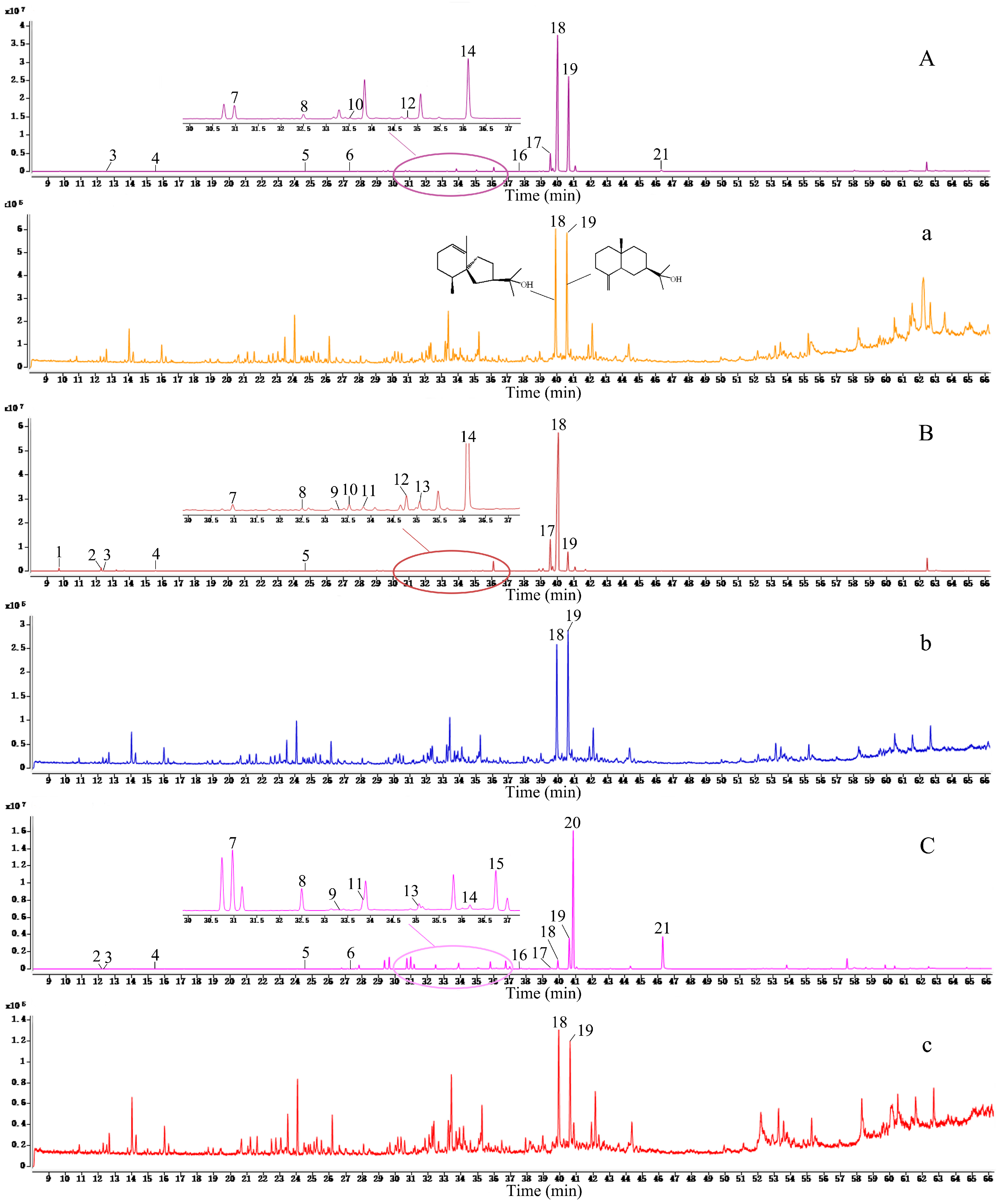
2.4. Chemical Profiling of A. lancea and Its Frosts
3. Materials and Methods
3.1. Sample Information
3.2. Chemicals and Reagents
3.3. Microscopic Analysis
3.4. Macroscopic Raman Spectroscopy
3.5. UPLC-Q/TOF-MS Analysis of P. ostii
3.5.1. Sample Preparation
3.5.2. UPLC-Q/TOF-MS Method
3.6. UPLC-Q Orbitrap Analysis
3.6.1. Sample Preparation
3.6.2. UPLC-Q Orbitrap Method
3.7. Gas Chromatography-Mass Spectrometry Analysis
3.7.1. Sample Preparation
3.7.2. Gas Chromatography-Mass Spectrometry Analysis and Metabolite Identification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hu, Z.H. Anatomy of Plant Secretory Structure; Shanghai Scientific & Technical Publishers: Shanghai, China, 2012. [Google Scholar]
- Taban, A.; Saharkhiz, M.J.; Niakousari, M. Sweet bay (Laurus nobilis L.) essential oil and its chemical composition, antioxidant activity and leaf micromorphology under different extraction methods. Sustain. Chem. Pharm. 2018, 9, 12–18. [Google Scholar] [CrossRef]
- Francoz, E.; Ranocha, P.; Burlat, V.; Dunand, C. Arabidopsis seed mucilage secretory cells: regulation and dynamics. Trends Plant Sci. 2015, 20, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Chu, Q.G.; Hu, Z.H. Comparative anatomy of oil cells and mucilage cells in the leaves of the Lauraceae in China. Acta Phytotaxon. Sin. 1999, 37, 529–540. [Google Scholar]
- Hu, Z.H. Studies on the structure and the ontogeny of the laticiferous canals of Decaisnea fargesii Franch. Acta Bot. Sin. 1963, 11, 129–135. [Google Scholar]
- Renner, T.; Specht, C.D. Inside the trap: Gland morphologies, digestive enzymes, and the evolution of plant carnivory in the Caryophyllales. Curr. Opin. Plant Biol. 2013, 16, 436–442. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.C.; Srividya, N.; Parrish, A.N.; Yue, W.; Shan, M.Q.; Wu, Q.A.; Lange, B.M. Morphology of glandular trichomes of Japanese catnip (Schizonepeta tenuifolia Briquet) and developmental dynamics of their secretory activity. Phytochemistry 2018, 150, 23–30. [Google Scholar] [CrossRef]
- Olsson, M.E.; Olofsson, L.M.; Lindahl, A.; Lundgren, A.; Brodelius, M.; Brodelius, P.E. Localization of enzymes of artemisinin biosynthesis to the apical cells of glandular secretory trichomes of Artemisia annua L. Phytochemistry 2009, 70, 1123–1128. [Google Scholar] [CrossRef]
- Cristina, R.; Caroline, M.M.; Taisa, C.O.; Fernanda, M.S.; Lilian, A.O.; Gaspar, H.K.; Regina, M.Q.L.; Monica, L.R.; Neusa, L.N.; Xavier, S.; et al. Si-accumulation in Artemisia annua glandular trichomes increases artemisinin concentration, but does not interfere in the impairment of Toxoplasma gondii growth. Front. Plant Sci. 2016, 7, 1–14. [Google Scholar]
- Sanadhya, P.; Agarwal, P.; Agarwal, P.L. Ion homeostasis in a salt-secreting halophytic grass. AoB Plants 2015, 7, plv055. [Google Scholar] [CrossRef]
- Wightman, R.; Wallis, S.; Aston, P. Hydathode pit development in the alpine plant Saxifraga cochlearis. Flora 2017, 233, 99–108. [Google Scholar] [CrossRef]
- Wightman, R.; Wallis, S.; Aston, P. Leaf margin organization and the existence of vaterite-producing hydathodes in the alpine plant Saxifraga scardica. Flora 2018, 241, 27–34. [Google Scholar] [CrossRef]
- Zhou, C.H.; Sheng, Y.L.; Zhao, D.Q.; Wang, Z.Q.; Tao, J. Variation of oleanolic and ursolic acid in the flesh of persimmon fruit among different cultivars. Molecules 2010, 15, 6580–6587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.Z. Compendium of Meteria Medica; People’s Medical Publishing House: Beijing, China, 1982; Volume II. [Google Scholar]
- Jin, S.Y. Traditional identification experience of traditional Chinese medicine in Jin Shiyuan, 2nd ed.; China Press of Traditional Chinese Medicine: Beijing, China, 2012. [Google Scholar]
- Lin, L.; Jin, L.; Gao, S.F.; Chen, H.G.; Shi, X.L. Studies on the internal relationship between traditional identification term in Chinese medicine and pharmaceutical botany. China Pharm. 2015, 26, 3870–3874. [Google Scholar]
- Zhao, Z.Z.; Liang, Z.T.; Guo, P. Macroscopic identification of Chinese medicinal materials: Traditional experiences and modern understanding. J. Ethnopharmacol. 2011, 134, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Collective Edition of Teaching and Research Group of Pharmacology, Nanjing Pharmaceutical College. Pharmacology; People’s Medical Publishing House: Beijing, China, 1960. [Google Scholar]
- Shanghai Branch of Chinese Pharmaceutical Association, Shanghai Pharmaceutical Company. Compilation of Medicinal Materials; Shanghai Scientific & Technical Publishers: Shanghai, China, 1959; Volume I. [Google Scholar]
- Lou, Z.Q.; Qin, B. Species Systematization and Quality Evaluation of Commonly Used Chinese Traditional Drugs, North-China ed.; Peking University Medical Press: Beijing, China, 1995; Volume III. [Google Scholar]
- Shang, Z.J. The Collation and Annotation of Sheng Nong’s Herbal Classic; Academy Press: Beijing, China, 2008. [Google Scholar]
- Hong, D.Y.; Pan, K.Y. Taxonomical history and revision of Paeonia sect. Moutan (Paeoniaceae). Acta Phytotaxon. Sin. 1999, 37, 351–368. [Google Scholar]
- Han, C.J.; Wang, Q.; Zhang, H.B.; Wang, S.H.; Song, H.D.; Hao, J.M.; Dong, H.Z. Light shading improves the yield and quality of seed in oil-seed peony (Paeonia ostii Feng Dan). J. Integr. Agr. 2018, 17, 1631–1640. [Google Scholar] [CrossRef]
- Wei, X.B.; Xue, J.Q.; Wang, S.L.; Xue, Y.Q.; Lin, H.; Shao, X.F.; Xu, D.H.; Zhang, X.X. Fatty acid analysis in the seeds of 50 Paeonia ostii individuals from the same population. J. Integr. Agr. 2018, 17, 1758–1767. [Google Scholar] [CrossRef] [Green Version]
- Tian, X.; Guo, S.; Zhang, S.S.; Li, P.S.; Wang, T.Y.; Ho, C.T.; Pan, M.H.; Bai, N.S. Chemical characterization of main bioactive constituents in Paeonia ostii seed meal and GC-MS analysis of seed oil. J. Food Biochem. 2019, e13088. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, L.N.; Wang, X.S.; Gao, J.Y.; Yi, J.P.; Deng, R.X. Characterization of Paeonia ostii seed and oil sourced from different cultivation areas in China. Ind. Crops Prod. 2019, 133, 63–71. [Google Scholar] [CrossRef]
- Wang, G.C.; Liu, Z.J. Cultivation, Appreciation and Cultural Origin Culture of Chinese Peony; Forestry Press: Beijing, China, 2001. [Google Scholar]
- Zhang, L.; Li, D.C.; Liu, L.F. Peaonol: Pharmacological effects and mechanisms of action. Int. Immunopharmacol. 2019, 72, 413–421. [Google Scholar] [CrossRef]
- Khalid, S.; Khan, A.; Shal, B.; Ali, H.; Kim, Y.S.; Khan, S. Suppression of TRPV1 and P2Y nociceptors by honokiol isolated from Magnolia officinalis in 3rd degree burn mice by inhibiting inflammatory mediators. Biomed. Pharmacother. 2019, 114, 108777. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Shen, Y.F.; Tu, X.; Wu, X.H.; Wang, G.X.; Ling, F. Isolation of anti-Saprolegnia lignans from Magnolia officinalis and SAR evaluation of honokiol/magnolol analogs. Bioorg. Med. Chem. Lett. 2019, 29, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Lu, M.H.; Guo, D.S.; Zhai, Y.Y.; Miao, D.; Yue, J.Y.; Yuan, C.H.; Zhao, M.M.; An, D.R. Antifungal effect of magnolol and honokiol from Magnolia officinalis on Alternaria alternate causing tobacco brown spot. Molecules 2019, 24, 2140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, H.L.; Wang, Z.G.; Hua, W.; You, Y.; Zou, L. Effect of chemical profiling change of processed Magnolia officinalis on the pharmacokinetic profiling of honokiol and magnolol in rats. J. Chromatogr. Sci. 2016, 54, 1201–1212. [Google Scholar] [CrossRef] [Green Version]
- Jun, X.; Fu, P.; Lei, Y.; Cheng, P. Pharmacological effects of medicinal components of Atractylodes lancea (Thunb.) DC. Chin. Med. 2018, 13, 59. [Google Scholar] [CrossRef] [Green Version]
- Guo, W.Q.; Liu, S.B.; Ju, X.; Du, J.H.; Xu, B.; Yuan, H.X.; Qin, F.J.; Li, L.Z. The antitumor effect of hinesol, extract from Atractylodes lancea (Thunb.) DC. by proliferation, inhibition, and apoptosis induction via MEK/ERK and NF-χB pathway in non-small cell lung cancer cell lines A549 and NCI-H1299. J. Cell. Biochem. 2019, 120, 18600–18607. [Google Scholar] [CrossRef]
- Kesara, N.B.; Tullayakorn, P.; Juntra, K. Research and development of Atractylodes lancea (Thunb) DC. as a promising candidate for cholangiocarcinoma chemotherapeutics. Evid-Based Compl. Alt. 2017, 2017, 1–16. [Google Scholar]
- Chen, Q.H.; Li, P.; Yang, H.D.; Li, X.L.; Zhu, J.; Chen, F.C. Identification of volatile compounds of Atractylode lancea Rhizoma using supercritical fluid extraction and GC-MS. J. Sep. Sci. 2009, 32, 3152–3156. [Google Scholar] [CrossRef]
- Ouyang, Z.; Zhang, L.; Zhao, M.; Wang, P.X.; Wei, Y.; Fang, J. Identification and quantification of sesquiterpenes and polyacetylenes in Atractylodes lancea from various geographical origins using GC-MS analysis. Rev. Bras. Farmacogn. 2012, 22, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Zhan, Z.L.; Deng, A.; Kang, L.P.; Tang, J.F.; Nan, T.F.; Chen, T.; He, Y.L.; Guo, L.P.; Huang, L.Q. Chemical profiling in Moutan Cortex after sulfuring and desulfuring processes reveals further insights into the quality control of TCMs by nontargeted metabolomic analysis. J. Pharm. Biomed. Anal. 2018, 156, 340–348. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Hu, L.P.; Liu, G.M.; Zhang, D.S.; He, H.J. Evaluation of the nutritional quality of Chinese kale (Brassica alboglabra Bailey) using UHPLC-Quadrupole-Orbitrap MS/MS-based metabolomics. Molecules 2017, 22, 1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.G.; Li, X.R.; Jiang, Q.J.; Sun, H.N.; Jiang, J.F.; Chen, S.M.; Guan, Z.Y.; Fang, W.M.; Chen, F.D. GC-MS analysis of the volatile constituents in the leaves of 14 Compositae plants. Molecules 2018, 23, 166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chinese Pharmacopoeia Commission, Institute of Medicinal Plant Development Chinese Academy of Medical Sciences. A Colored Identification Atlas of Chinese Materia Medica and Plant as Specified in the Pharmacopoeia of the People’s Republic of China; People’s Medical Publishing House: Beijing, China, 2010. [Google Scholar]
- Zhao, Z.Z.; Chen, H.B. Illustrated Chinese Materia Medica, 2nd ed.; Fujian Science & Technology Publishing House: Fuzhou, China, 2018. [Google Scholar]
- Cai, P.Y. Raman Study on Drug Molecules and Organic Molecules; Southwest University: Chongqing, China, 2011. [Google Scholar]
- Zhu, Z.Y.; Gu, R.N.; Lu, T.H. Application of Raman Spectroscopy in Chemistry; Northeast University Press: Shenyang, China, 1998. [Google Scholar]
- Li, S.S.; Wu, Q.; Yin, D.D.; Peng, C.Y.; Liu, Z.A.; Wang, L.S. Phytochemical variation among the traditional Chinese medicine Mu Dan Pi from Paeonia suffruticosa (tree peony). Phytochemistry 2018, 146, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.F.; Pei, Y.M.; Wu, H.; Xu, Q.; Xu, G.B.; Jiang, L.; Zhou, J.L. Difference analysis of chemical compositions in moutan cortex from different origins by UPLC-Q/TOF-MS. Chin. Tradit. Herb. Drugs 2016, 47, 2984–2992. [Google Scholar]
- Han, C.M.; Zhu, J.B.; Ding, Y.; Kou, Z.N.; Wang, Z.Z.; Xiao, W. Analysis of chemical components in methanol extract of Moutan Cortex by UPLC/Q-TOF-MS. J. Dalian Polytech. Univ. 2014, 33, 166–170. [Google Scholar]
- Xiao, C.N.; Wu, M.; Chen, Y.Y.; Zhang, Y.J.; Zhao, X.F.; Zheng, X.H. Revealing metabolomic variations in Cortex Moutan from different root parts using HPLC-MS method. Phytochem. Analysis. 2015, 26, 86–93. [Google Scholar] [CrossRef]
- Li, J.H.; Zeng, R.; Qu, Y.; Huang, L.F. Rapid identification on chemical constituents in roots of Paeonia delavayi var. lutea by UPLC-Q-TOF-MSE combined with UNIFI informatics platform. Chin. Tradit. Herb. Drugs 2017, 48, 1529–1536. [Google Scholar]
- Zhou, G.; Lv, Q.H. Studies on content determination and fingerprint of active constituents in different parts of Cortex Moutan. J. Chin. Mater. Med. 2008, 33, 2070–2073. [Google Scholar]
- Guo, J.Y.; Li, S.B.; Hou, Y.Z.; Shi, M.K.; Pei, Y.F.; Zhang, J.Y.; Ji, R.L. Study on polyphenol contents in different parts of peony. Food Sci. Technol. 2008, 29, 348–350. [Google Scholar]
- Fan, X.H.; Ma, T.C.; Shen, X.; Zhang, W.T.; Bi, K.S.; Jia, Y. UPLC determination of six active components in different parts of Moutan Cortex from different habitats. Chin. Tradit. Pat. Med. 2012, 34, 317–320. [Google Scholar]
- Wang, L. The Study on Modern Processing Method and UPLC-DAD-TOF-MS Fingerprint of Wen-Hou-Po; Henan University of Chinese Medicine: Zhengzhou, China, 2010. [Google Scholar]
- Zhao, H.; Yan, Y.; Zou, L.S.; Liu, X.H.; Chai, C.; Hua, Y.J.; Wang, S.N. Identification of chemical constituents in Magnoliae officinalis cortex by UFLC-Triple TOF-MS/MS. Chin Pharm. J. 2017, 52, 1824–1831. [Google Scholar]
- Zhao, H.; Yan, Y.; Zou, L.S.; Liu, X.H.; Chai, C.; Hui, Y.J.; Wang, S.N. Difference of chemical compositions in “Chuanpu” and “Wenpu” based on LC-MS/MS and GC-MS/MS. Nat. Prod. Res. Dev. 2018, 30, 1–9. [Google Scholar]
- Ji, L.; Ao, P.; Pan, J.G.; Yang, J.Y.; Yang, J.; Hu, S.L. GC-MS analysis of essential oils from rhizomes of Atractylodes lancea (Thunb.) DC. and A. chinensis (DC.) Koidz. China J. Chin. Mater. Med. 2001, 26, 182–185. [Google Scholar]
- Gao, Y.; Wang, Z.B.; Yang, C.J.; Wu, G.S.; Chen, Y.J.; Kuang, H.X. Analysis on the volatile components of Atractylodes lancea from different origins by GC-MS. Acta Chin. Med. Pharmacol. 2017, 45, 35–38. [Google Scholar]
- Zhou, J.Y.; Li, X.; Zheng, J.Y.; Dai, C.C. Volatiles released by endophytic Pesudomonas flurorescens promoting the growth and volatile oil accumulation in Atractylodes lancea. Plant Physiol. Bioch. 2016, 101, 132–140. [Google Scholar] [CrossRef]
- Wang, F.; Ouyang, Z.; Guo, L.P.; Zhao, M.; Peng, H.S.; Liao, J.L.; Liang, Z.P. Comprehensive chemical pattern recognition of Atractylodis Rhizomes. China J. Chin. Mater. Med. 2014, 39, 2536–2541. [Google Scholar]
- Yoichi, N.; Yohya, W.; Takako, S. Studies on the evaluation of crude drugs (I) comparative studies on the components of Atractylodes Rhizomes. Syôyakugaku Zasshi 1975, 29, 139–146. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |


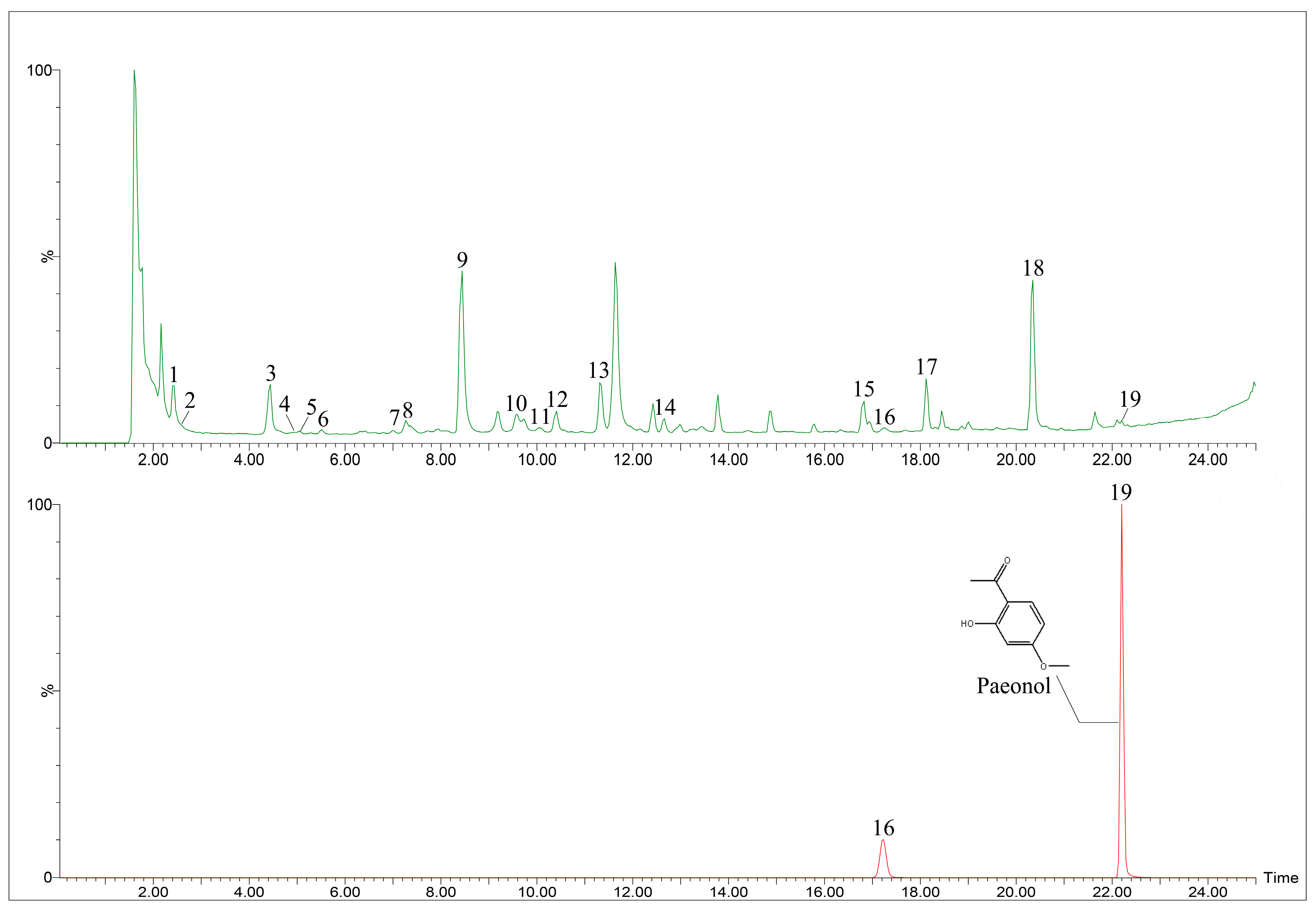


| Peak No. | tR (min) | Molecular Formula | Observed Mass (m/z) | Mass Error (ppm) | MS/MS(m/z) | Identification | Relative Content (%) | |
|---|---|---|---|---|---|---|---|---|
| A | B | |||||||
| 1 | 2.42 | C7H6O5 | 169.0150 | 7.7 | 125.0236 | Gallic acid | 0.14 ± 0.03 | - |
| 2 | 2.54 | C18H24O14 | 463.1086 | −0.4 | 403.0870, 373.0789,343.0667, 301.0577 | Mudanoside B | 0.03 ± 0.01 | - |
| 3 | 4.42 | C23H28O12 | 495.1509 | 1.2 | 465.1407, 137.0242 | Oxypaeoniflorin | 3.16 ± 0.45 | - |
| 4 | 4.95 | C15H14O6 | 289.0727 | 5.2 | 245.0827, 137.0242 | D-Catethin | 0.05 ± 0.03 | - |
| 5 | 5.04 | C24H30O13 | 525.1609 | 0.2 | 495.1509, 167.0336, 165.0535 | Mudanpioside E | 0.24 ± 0.11 | - |
| 6 | 5.53 | C8H8O5 | 183.0285 | −4.4 | 124.0166 | Methyl gallate | 0.15 ± 0.01 | - |
| 7 | 7.01 | C20H28O12 | 459.1496 | −1.5 | 293.0873, 233.0674, 165.0561 | Paeonolide | 5.20 ± 4.92 | - |
| 8 | 7.21 | C26H38O17 | 621.2020 | −1.8 | 455.1421, 293.0418 | Suffruticoside E | 0.38 ± 0.48 | - |
| 9 | 8.40 | C23H28O11 | 479.1548 | −1.0 | 525.1609, 449.1465, 327.1075, 165.0561, 121.0292 | Paeoniflorin | 9.14 ± 1.05 | - |
| 10 | 9.58 | C27H32O16 | 611.1603 | −1.5 | 445.1009, 283.0440, 169.0150 | Suffruticoside A/B/C/D | 1.60 ± 0.24 | - |
| 11 | 10.11 | C27H32O16 | 611.1653 | 6.7 | 445.1009, 283.0475, 169.0150, 121.0292 | Suffruticoside A/B/C/D | 3.07 ± 2.45 | - |
| 12 | 10.38 | C27H32O16 | 611.1603 | −1.5 | 445.1009, 283.0440, 165.1561 | Suffruticoside A/B/C/D | 1.15 ± 0.21 | - |
| 13 | 11.36 | C27H32O16 | 611.1603 | −1.5 | 445.1009, 283.0475, 169.0150, 121.0292 | Suffruticoside A/B/C/D | 2.78 ± 0.63 | - |
| 14 | 12.64 | C30H32O14 | 615.1733 | −2.6 | 447.1279, 431.1346, 281.0681, 137.0242 | Mudanpioside H | 0.59 ± 0.26 | - |
| 15 | 16.79 | C30H32O13 | 599.1752 | −2.2 | 477.1413, 447.1279, 431.1346, 281.0681, 137.0242 | Mudanpioside C | 2.09 ± 0.28 | - |
| 16 | 17.27 | C9H10O3 | 165.0561 | −2.2 | 150.0331, 122.0353 | Unidentified | 0.19 ± 0.04 | 8.73 ± 3.44 |
| 17 | 18.14 | C30H32O13 | 599.1752 | 4.7 | 569.1677, 477.1413, 137.0228, 165.0270, 121.0270 | Benzoyloxpaeoniflorin | 1.37 ± 0.47 | - |
| 18 | 20.32 | C30H32O12 | 583.1804 | −2.1 | 629.1885, 553.1722, 431.1346, 165.0561, 121.0292 | Benzoylpaeoniflorin | 4.89 ± 2.41 | - |
| 19 | 22.20 | C9H10O3 | 165.0561 | 5.5 | 150.0331, 122.0353 | Paeonol | 0.65 ± 0.49 | 47.66 ± 14.17 |
| Peak no. | tR(min) | Molecular Formula | Observed Mass (m/z) | Mass Error (ppm) | MS2 m/z | Proposed Compounds | Relative Content (%) | |
|---|---|---|---|---|---|---|---|---|
| A | B | |||||||
| 1 | 9.64 | C19H22O5 | 329.1354 | −5.01 | 267.0998, 249.0897, 239.1043, 221.0945, 133.0631 | Magnolignan D | 0.04 ± 0.03 | - |
| 2 | 9.77 | C18H20O4 | 299.1255 | −7.64 | 239.1048, 221.0945, 133.0630 | Magnolignan A/C | 0.58 ± 0.48 | - |
| 3 | 10.53 | C18H18O4 | 297.1099 | −7.52 | 253.0841, 249.0904, 239.1047, 225.0890 | Magnolignan E | 0.07 ± 0.05 | - |
| 4 | 10.69 | C15H14O3 | 241.0841 | −7.55 | 223.0736, 197.0940, 133.0642 | Randaiol | 2.38 ± 2.08 | - |
| 5 | 11.71 | C16H14O3 | 253.0841 | −7.19 | 235.0737, 207.0790 | Magnaldehyde D | 2.92 ± 2.60 | - |
| 6 | 12.07 | C18H16O4 | 295.0944 | −7.07 | 251.1049, 233.0942, 231.0789 | Dimethylstrobochrysin | 0.03 ± 0.01 | - |
| 7 | 12.30 | C18H16O3 | 279.0996 | −7.06 | 261.0901, 233.0948 | Randainal | 0.35 ± 0.15 | - |
| 8 | 13.87 | C19H20O3 | 295.1302 | −9.05 | 295.1302, 265.1206, 263.1048, 245.0945 | 3-OMe-magnalol | 0.05 ± 0.03 | - |
| 9 | 14.04 | C18H18O2 | 265.1205 | −6.81 | 224.0813, 223.0740, 197.0574 | Honokiol | 15.95 ± 6.28 | 2.67 ± 1.25 |
| 10 | 14.80 | C18H18O3 | 281.1151 | −7.54 | 164.0451, 136.0377, 133.0633 | Obovatol | 2.90 ± 1.67 | - |
| 11 | 14.94 | C18H18O2 | 265.1205 | −6.81 | 224.0813 | Magnolol | 44.43 ± 3.59 | 25.58±2.24 |
| Peak No. | tR(min) | Molecular Formula | Molecular Weight | Identification | Relative Content (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| A | a | B | b | C | c | |||||
| 1 | 9.73 | C10H16 | 136 | α-Pinene | - | - | 0.41 ± 0.23 | - | - | - |
| 2 | 12.28 | C10H16 | 136 | α-phellandrene | - | - | 0.40 ± 0.29 | - | 0.11 ± 0.09 | - |
| 3 | 12.41 | C10H16 | 136 | 3-Carene | 0.02 ± 0.01 | - | 0.14 ± 0.08 | - | 0.01 ± 0.01 | - |
| 4 | 15.60 | C10H16 | 136 | Terpinolene | 0.02 ± 0.01 | - | 0.12 ± 0.06 | - | 0.01 ± 0.01 | - |
| 5 | 24.73 | C12H20O2 | 196 | Bornyl acetate | 0.02 ± 0.02 | - | 0.04 ± 0.01 | - | 0.02 ± 0.02 | - |
| 6 | 27.36 | C12H22O2 | 198 | Citronellyl acetate | 0.01 ± 0.01 | - | - | - | 0.05 ± 0.02 | - |
| 7 | 30.97 | C15H24 | 204 | β-Caryophyllene | 0.22 ± 0.16 | - | 0.08 ± 0.05 | - | 2.66 ± 0.53 | - |
| 8 | 32.50 | C15H24 | 204 | Humulene | 0.08 ± 0.05 | - | 0.03 ± 0.02 | - | 0.99 ± 0.18 | - |
| 9 | 33.28 | C15H22 | 202 | α-Curcumene | - | - | 0.01 ± 0.00 | - | 0.07 ± 0.04 | - |
| 10 | 33.52 | C15H24 | 204 | β-Cubebene | 0.01 ± 0.00 | - | 0.05 ± 0.01 | - | - | - |
| 11 | 33.83 | C15H24 | 204 | Zingiberene | - | - | 0.13 ± 0.09 | - | 1.60 ± 0.26 | - |
| 12 | 34.78 | C15H24 | 204 | γ-Cadinene | 0.03 ± 0.01 | - | 0.13 ± 0.03 | - | - | - |
| 13 | 35.07 | C15H24 | 204 | β-sesquiphellandrene | - | - | 0.09 ± 0.02 | - | 0.17 ± 0.11 | - |
| 14 | 36.11 | C15H26O | 222 | Elemol | 1.19 ± 0.47 | - | 2.96 ± 0.94 | - | 0.18 ± 0.04 | - |
| 15 | 36.75 | C15H24 | 204 | γ-elemene | - | - | - | - | 1.39 ± 0.47 | - |
| 16 | 37.69 | C15H24O | 220 | Caryophyllene Oxide | 0.04 ± 0.03 | - | - | - | 0.30 ± 0.12 | - |
| 17 | 39.56 | C15H26O | 222 | Agarospirol | 4.37 ± 1.22 | - | 7.80 ± 2.11 | - | 0.08 ± 0.08 | - |
| 18 | 40.00 | C15H26O | 222 | Hinesol | 42.77 ± 10.23 | 5.10 ± 3.13 | 63.85 ± 10.87 | 7.56 ± 2.45 | 1.21 ± 0.99 | 3.61 ± 1.94 |
| 19 | 40.66 | C15H26O | 222 | β - Eudesmol | 40.81 ± 11.75 | 4.83 ± 3.02 | 12.45 ± 13.18 | 6.67 ± 5.26 | 5.47 ± 3.37 | 4.38 ± 1.10 |
| 20 | 40.86 | C15H20O | 216 | Atractylon | - | - | - | - | 33.89 ± 9.78 | - |
| 21 | 46.30 | C13H10O | 182 | Atractylodin | 0.36 ± 0.23 | - | - | 13.53 ± 5.07 | - | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, D.Q.; Han, X.J.; Shan, T.Y.; Xu, R.; Hu, J.; Cheng, W.X.; Zha, L.P.; Peng, H.S. Microscopic Characteristic and Chemical Composition Analysis of Three Medicinal Plants and Surface Frosts. Molecules 2019, 24, 4548. https://doi.org/10.3390/molecules24244548
Yu DQ, Han XJ, Shan TY, Xu R, Hu J, Cheng WX, Zha LP, Peng HS. Microscopic Characteristic and Chemical Composition Analysis of Three Medicinal Plants and Surface Frosts. Molecules. 2019; 24(24):4548. https://doi.org/10.3390/molecules24244548
Chicago/Turabian StyleYu, Da Qing, Xiao Jing Han, Ting Yu Shan, Rui Xu, Jin Hu, Wang Xing Cheng, Liang Ping Zha, and Hua Sheng Peng. 2019. "Microscopic Characteristic and Chemical Composition Analysis of Three Medicinal Plants and Surface Frosts" Molecules 24, no. 24: 4548. https://doi.org/10.3390/molecules24244548





