Neuroprotective Norsesquiterpenoids and Triterpenoids from Populus euphratica Resins
Abstract
:1. Introduction
2. Results and Discussion
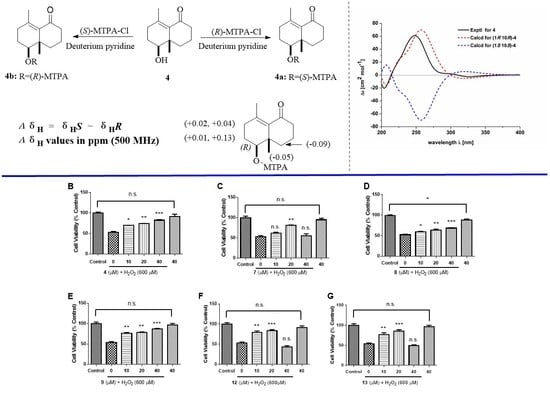
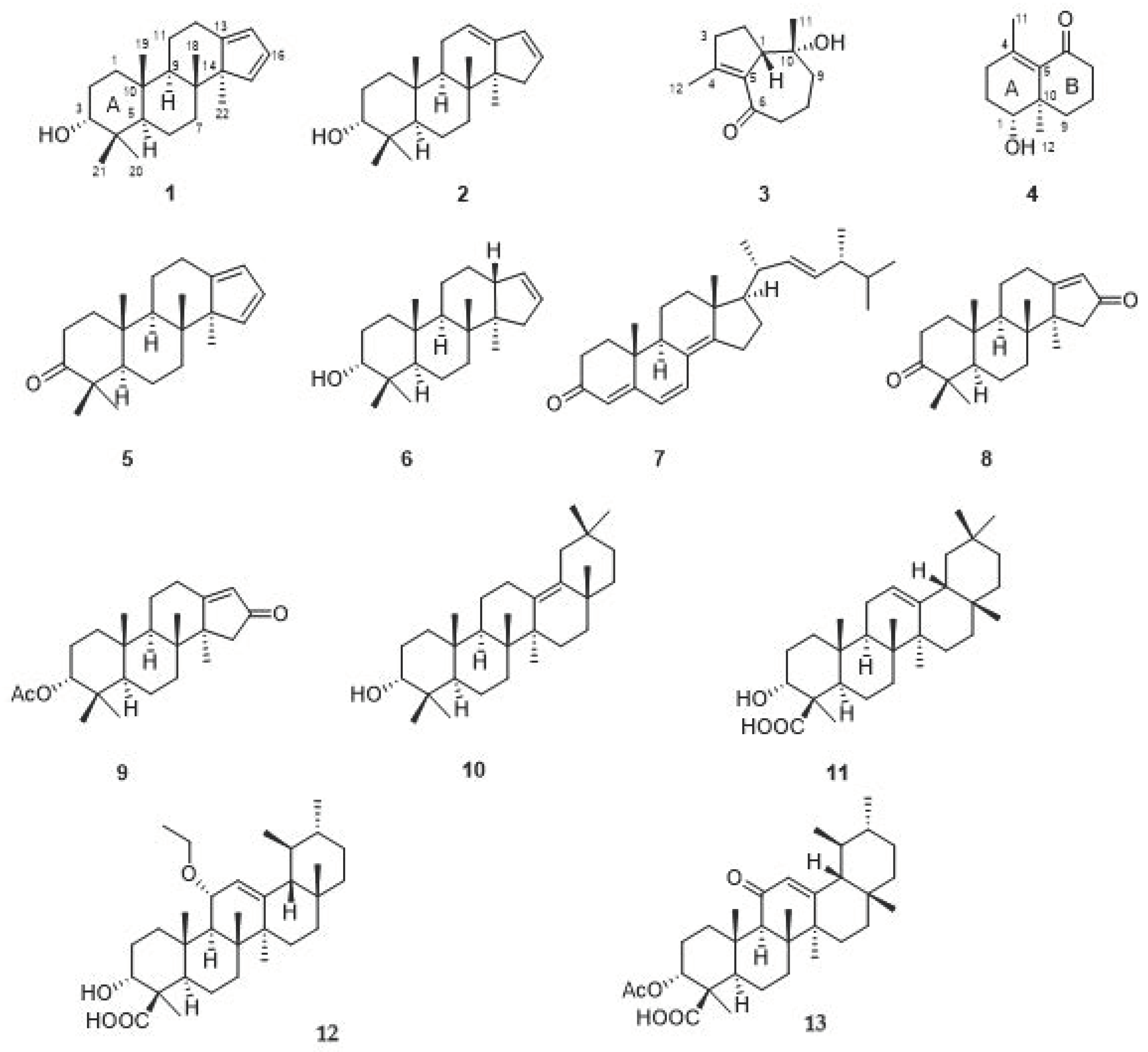
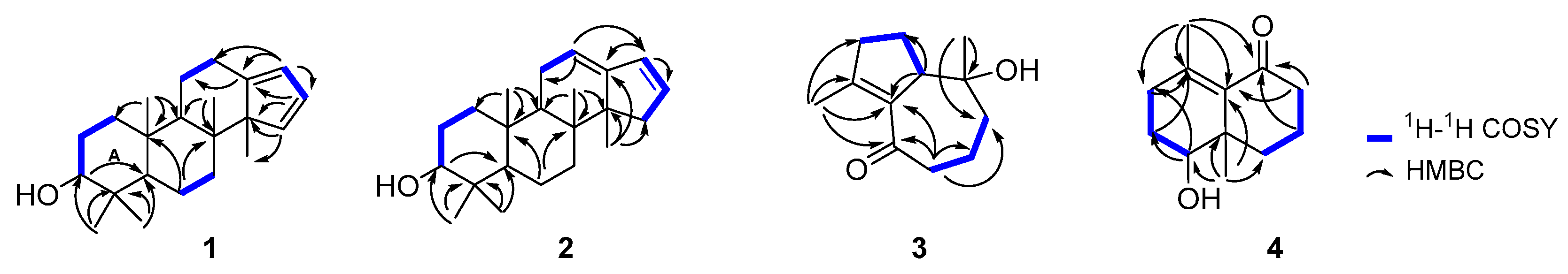
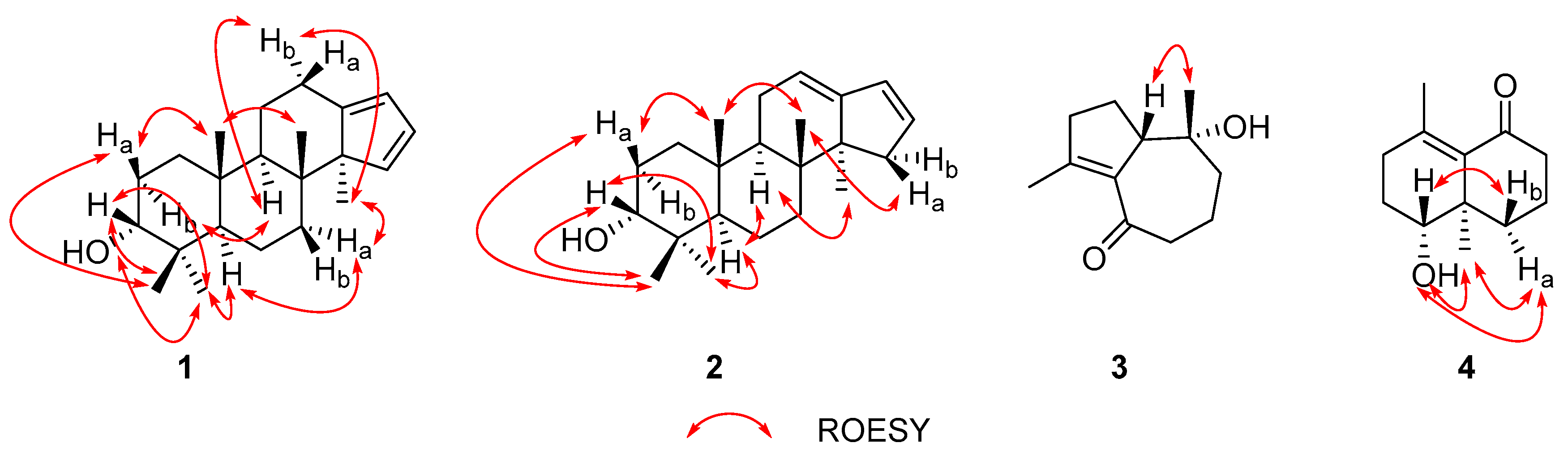
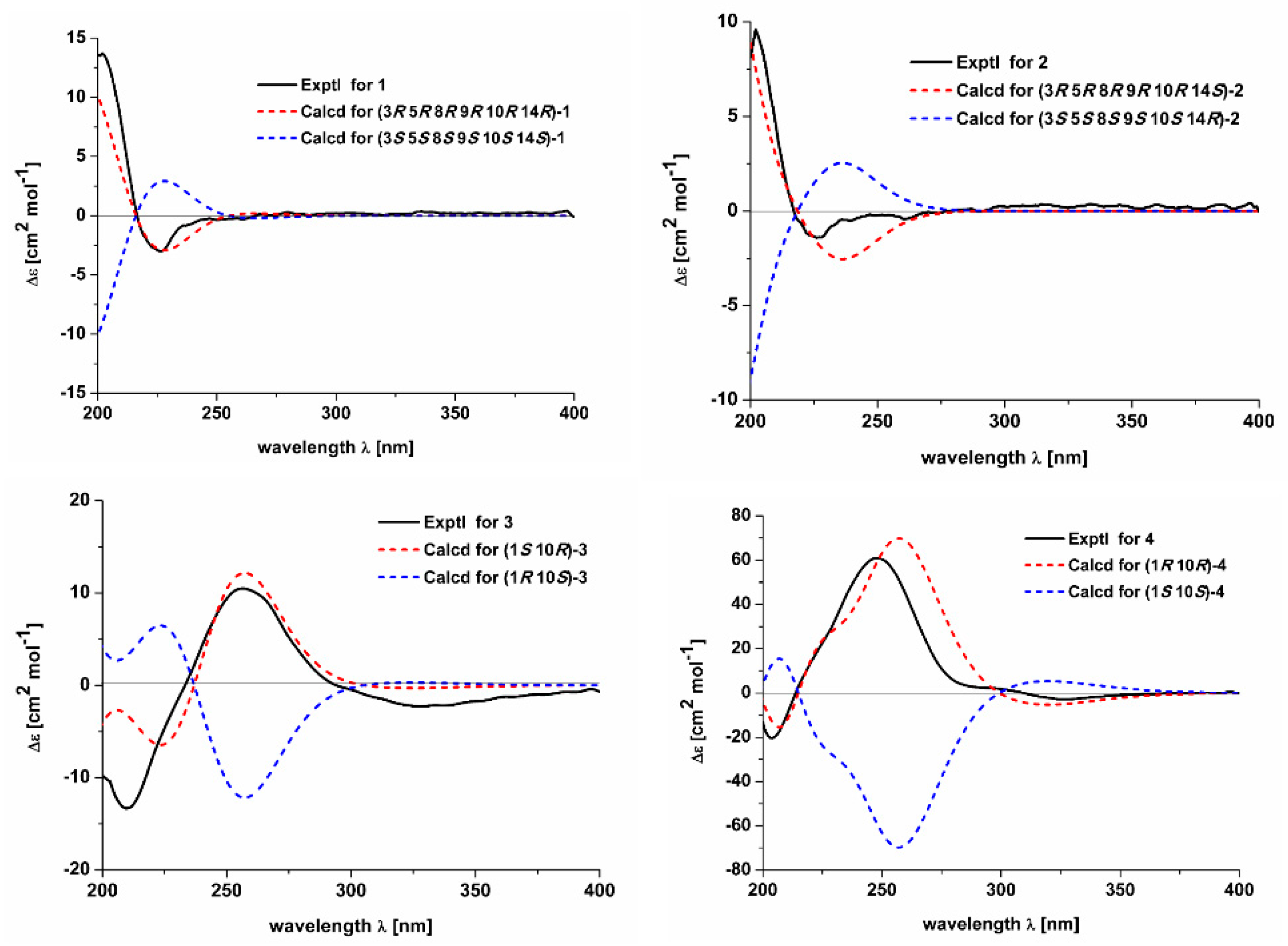
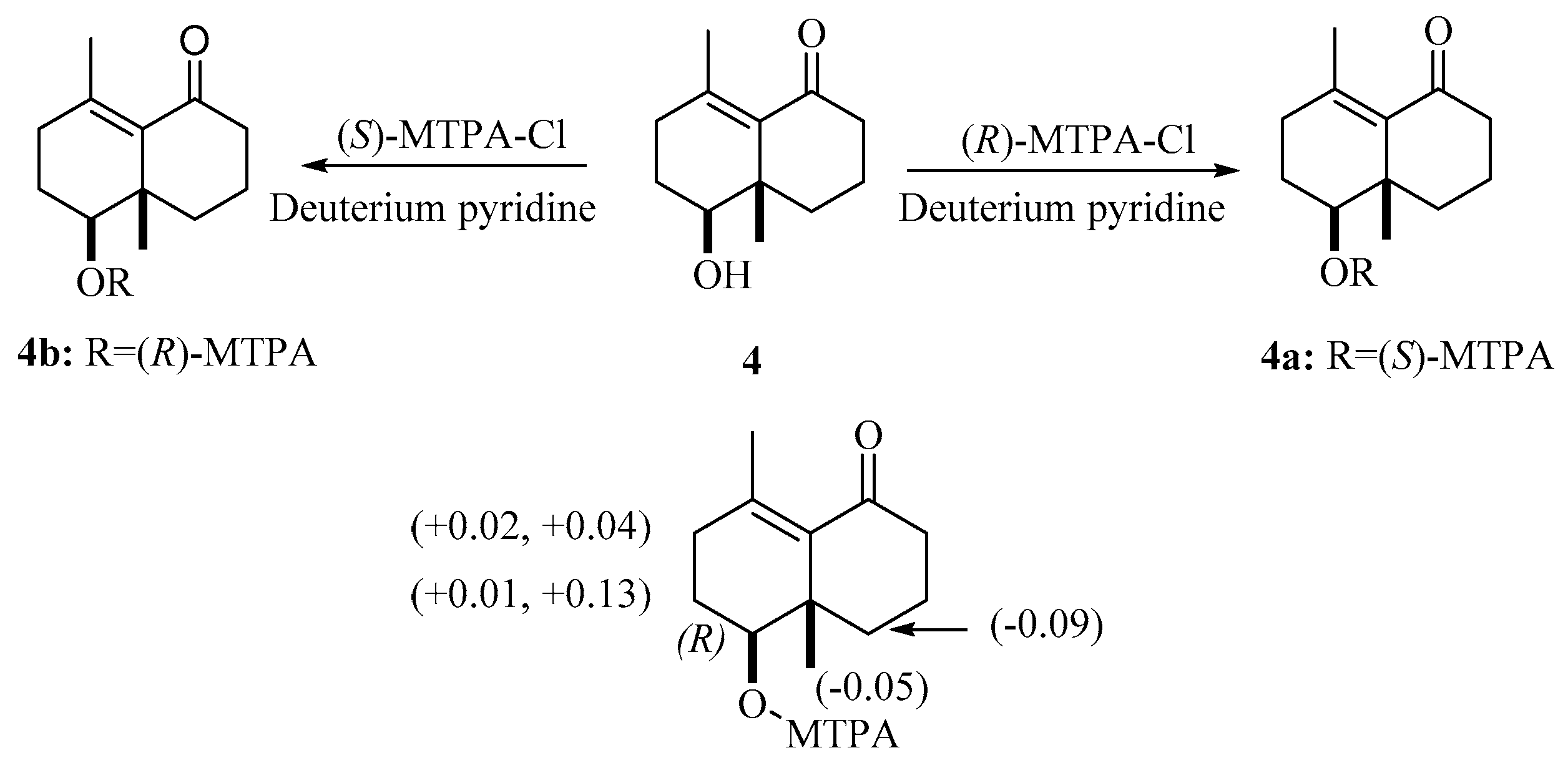
2.1. Structure Elucidation of the Compounds
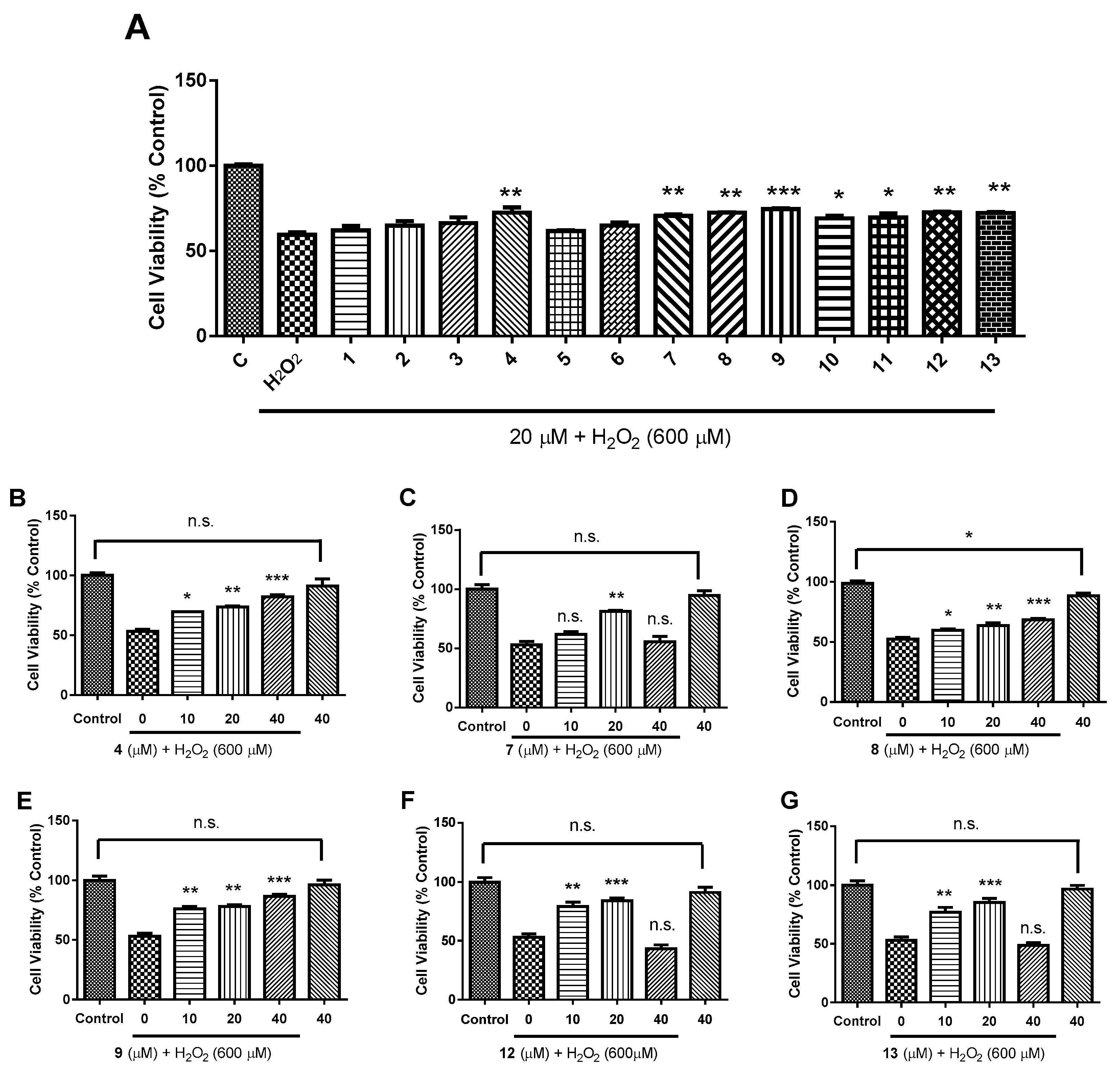
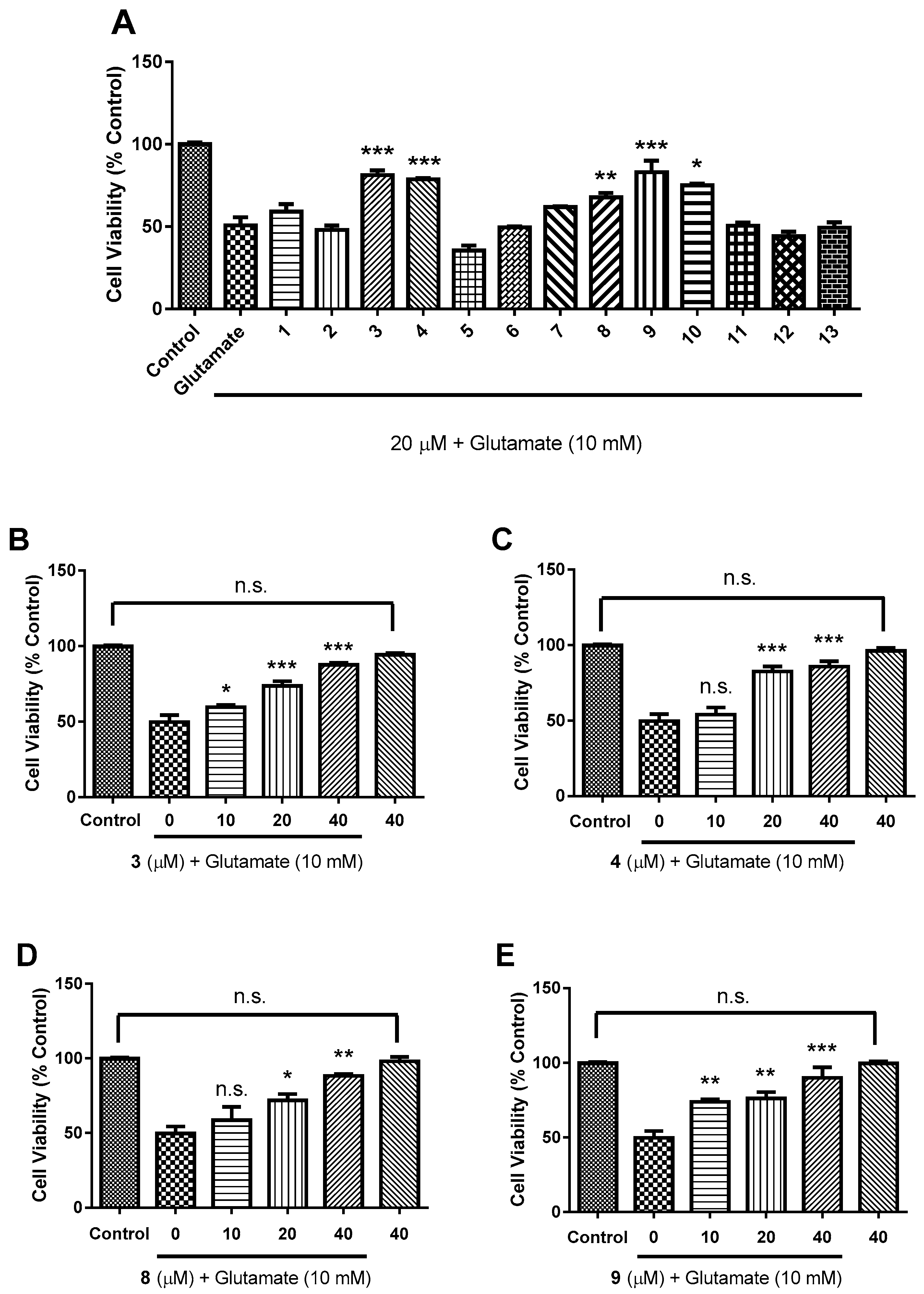
2.2. Biological Evaluation
3. Experimental Section
3.1. General Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Compound Characterization Data
3.5. ECD Calculation for Compounds 1–4
3.6. MTPA Esterification of 4
3.7. Bioactivity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gong, X.; Li, Z.; Zhang, N.; Wang, J.; Han, J.H.; Ren, K.; Zhang, C.H.; Li, M.H. Anti-inflammatory activity and chemical composition of resin extracts from Populus diversifolia Schrenk. Mod. Chin. Med. 2018, 12, 1185–1189. [Google Scholar]
- Wei, W.; Rena, K.; Yang, X.W. New salicin derivatives from the leaves of Populus euphratica. Asian Nat. Prod. Res. 2015, 17, 491–496. [Google Scholar]
- Luo, J.R.; Jiang, H.E.; Zhao, Y.X.; Liu, Y.Q.; Qian, J.F. Volatiles from the heartwood of Populus euphratica in ancient tomb. Nat. Prod. Res. Dev. 2007, 19, 819–821. [Google Scholar]
- Luo, J.R.; Jiang, H.E.; Zhao, Y.X.; Liu, Y.Q.; Qian, J.F. Components of the heartwood of Populus euphratica from an ancient tomb. Chem. Nat. Compd. 2008, 44, 6–9. [Google Scholar] [CrossRef]
- Liu, J.W.; Liu, Y.; Yan, Y.M.; Yang, J.; Lu, X.F.; Cheng, Y.X. Commiphoratones A and B, Two sesquiterpenedimers from Resina Commiphora. Org. Lett. 2018, 20, 2220–2223. [Google Scholar] [CrossRef]
- Dong, L.; Cheng, L.Z.; Yan, Y.M.; Wang, S.M.; Cheng, Y.X. Commiphoranes A−D, carbon skeletal terpenoids rom Resina Commiphora. Org. Lett. 2017, 19, 286–289. [Google Scholar] [CrossRef]
- Zhu, C.Z.; Hu, B.Y.; Liu, J.W.; Cai, Y.; Chen, X.C.; Qin, D.P.; Cheng, Y.X.; Zhang, Z.D. Anti-mycobacterium tuberculosis terpenoids from Resina Commiphora. Molecules 2019, 24, 1475. [Google Scholar] [CrossRef]
- Liu, K.X.; Zhu, Y.X.; Yan, Y.M.; Zeng, Y.; Jiao, Y.B.; Qin, F.Y.; Liu, J.W.; Zhang, Y.Y.; Cheng, Y.X. Discovery of populusone, a skeletal stimulator of umbilical cord mesenchymal stem cells from Populus euphratica exudates. Org. Lett. 2019, 21, 1837–1840. [Google Scholar] [CrossRef]
- Cao, Q.; Wang, S.X.; Cheng, Y.X. Abietane diterpenoids with potent cytotoxic activities from the resins of Populus euphratica. Nat. Prod. Commun. 2019. [Google Scholar] [CrossRef]
- Liu, K.X.; Qin, D.P.; Zhu, Y.X.; Wang, S.X.; Jiao, Y.B.; Ge, P.L.; Cheng, Y.X. Populeuphrines A and B, two new cembrane diterpenoids from the resins of Populus euphratica. Nat. Prod. Res. 2019, 1–9. [Google Scholar] [CrossRef]
- Dong, L.; Luo, Q.; Cheng, L.Z.; Yan, Y.M.; Cheng, Y.X.; Wang, S.M. New terpenoids from Resina Commiphora. Fitoterapia 2017, 117, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Morikawa, T.; Ando, S.; Oominami, H.; Murakami, T.; Kimura, I.; Yoshikawa, M. Absolute stereostructures of polypodane- and octanordammarane-type triterpenes with nitric oxide production inhibitory activity from guggul-gum resins. Bioorgan. Med. Chem. 2004, 12, 3037–3046. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Iwami, J.; Matsuda, H.; Mizuno, S.; Yoshikawa, M. Absolute stereostructures of inoterpenes A–F from sclerotia of Inonotus obliquus. Tetrahedron 2009, 65, 2443–2450. [Google Scholar] [CrossRef]
- Ohtani, I.; Kusumi, T.; Kashman, Y.; Kakisawa, H. High-field FT NMR application of Mosher’s method. The absolute configurations of marine terpenoids. J. Am. Chem. Soc. 1991, 113, 4092–4096. [Google Scholar] [CrossRef]
- Lee, W.Y.; Park, Y.K.; Ahn, J.K.; Park, S.Y.; Lee, H. Cytotoxic activity of ergosta-4,6,8(14),22-tetraen-3-one from the sclerotia of Polyporus umbellatus. Bull. Korean Chem. Soc. 2005, 26, 1464–1466. [Google Scholar]
- Provan, G.J.; Gray, A.I.; Waterman, P.G. Mansumbinane derivatives from stem bark of Commiphora kua. Phytochemistry 1992, 31, 2065–2068. [Google Scholar] [CrossRef]
- Wang, Y.G.; Ma, Q.G.; Tian, J.; Ren, J.; Wang, A.G.; Ji, T.F.; Wang, J.B.; Su, Y.L. Hepatoprotective triterpenes from the gum resin of Boswellia carterii. Fitoterapia 2016, 109, 266–273. [Google Scholar] [CrossRef]
- He, A.; Wang, M.; Hao, H.; Zhuang, D.C.; Li, G.H. Hepatoprotective triterpenes from Sedum sarmentosum. Phytochemistry 1998, 49, 2607–2610. [Google Scholar] [CrossRef]
- Culioli, G.; Mathe, C.; Archier, P.; Vieillescazes, C. A lupane triterpene from frankincense (Boswellia sp., Burseraceae). Phytochemistry 2003, 62, 537–541. [Google Scholar] [CrossRef]
- Al-Harrasi, A.; Ali, L.; Rehman, N.U.; Hussain, J.; Hussain, H.; Al-Rawahi, A.; Rizvi, T.S. 11 α-ethoxy-β-boswellic acid and nizwanone, a new boswellic acid derivative and a new triterpene, respectively, from Boswellia sacra. Chem. Biodivers. 2013, 10, 1501–1506. [Google Scholar] [CrossRef]
- Li, F.S.; Yan, D.L.; Liu, R.R.; Xu, K.P.; Tan, G.S. Chemical constituents of Boswellia carterii (Frankincense). Chin. J. Nat. Med. 2010, 115, 25–27. [Google Scholar] [CrossRef]
- Pradip, K.K.; Anuradha, K.; Shivika, R.; Supriya, S.; Santoshkumar, T.; Chandishwar, N.; Neetu, T. Mechanism of oxidative stress and synapse dysfunction in the pathogenesis of Alzheimer’s disease: Understanding the therapeutics strategies. Mol. Neurobiol. 2016, 53, 648–661. [Google Scholar]
- Erika, B.; George, G. Oxidative stress differentially induces tau dissociation from neuronal microtubules in neurites of neurons cultured from different regions of the embryonic Gallus domesticus brain. J. Neurosci. Res. 2019. [Google Scholar] [CrossRef]
- Zhao, H.P.; Han, Z.P.; Ji, X.M.; Luo, Y.M. Epigenetic regulation of oxidative stress in ischemic stroke. Aging Dis. 2016, 7, 295–306. [Google Scholar] [CrossRef]
- Vera, D.; Eunsung, J.; Mouradian, M.M. The role of oxidative stress in Parkinson’s disease. J. Parkinsons. Dis. 2013, 3, 461–491. [Google Scholar]
- Pamela, J.S. Excitatory amino acid receptors, excitotoxicity, and the human nervous system. Curr. Opin. Neurol. Neurosurg. 1993, 6, 414–422. [Google Scholar]
- Wu, B.; He, S.; Wu, X.D.; Wu, D.K.; Pan, Y.J. Cadinane and eudesmane sesquiterpenoids from Chloranthas henryi. Helv. Chim. Acta 2007, 90, 1586–1592. [Google Scholar] [CrossRef]
- Arezoo, R.; Hamid, R.S.; Azar, H.; Seyed, H.M.; Mohammad, T.B. 3-Acetyl-11-keto-β-boswellic acid attenuated oxidative glutamate toxicity in neuron-like cell lines by apoptosis inhibition. J. Cell. Biochem. 2019. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1‒13 are available from the authors. |
| 1 | 2 | ||||
|---|---|---|---|---|---|
| no. | δH | δC | no. | δH | δC |
| 1 | Ha: 1.50 (m) | 34.2, CH2 | 1 | Ha: 1.38 (m) | 33.2, CH2 |
| Hb: 1.40 (m) | Hb: 1.33 (overlap) | ||||
| 2 | Ha: 1.97 (m) | 25.6, CH2 | 2 | Ha: 1.96 (m) | 25.3, CH2 |
| Hb: 1.57 (m) | Hb: 1.56 (m) | ||||
| 3 | 3.42 (t-like, 2.63) | 76.3, CH | 3 | 3.42 (t-like, 2.8) | 76.3, CH |
| 4 | 37.8, C | 4 | 37.6, C | ||
| 5 | 1.36 (m) | 49.7, CH | 5 | 1.33 (overlap) | 49.7, CH |
| 6 | Ha: 1.48 (m) | 18.4, CH2 | 6 | Ha: 1.48 (m) | 18.3, CH2 |
| Hb: 1.38 (m) | Hb: 1.44 (m) | ||||
| 7 | Ha: 1.84 (m) | 37.3, CH2 | 7 | Ha: 1.62 (m) | 34.5, CH2 |
| Hb: 1.51 (overlap) | Hb: 1.31 (m) | ||||
| 8 | 41.4, C | 8 | 37.6, C | ||
| 9 | 1.51 (overlap) | 51.2, CH | 9 | 1.83 (dd, 10.6, 7.1) | 48.2, CH |
| 10 | 37.9, C | 10 | 37.5, C | ||
| 11 | Ha: 1.70 (m) | 23.1, CH2 | 11 | Ha: 2.13 (m) | 23.6, CH2 |
| Hb: 1.34 (m) | Hb: 1.93 (m) | ||||
| 12 | Ha: 2.61 (ddd, 13.6, 4.7, 1.7) | 26.4, CH2 | 12 | 5.40 (t, 3.7) | 114.7, CH |
| Hb: 2.16 (tdd, 13.6, 5.2, 1.7) | |||||
| 13 | 156.7, C | 13 | 151.9, C | ||
| 14 | 61.4, C | 14 | 49.4, C | ||
| 15 | 6.16 (d, 5.4) | 142.7, CH | 15 | Ha: 2.59 (brd,17.4) | 41.4, CH2 |
| Hb: 1.85 (brdt,17.4, 1.8) | |||||
| 16 | 6.21 (dd, 5.4, 1.7) | 129.6, CH | 16 | 5.83 (dt, 5.7, 2.6) | 133.5, CH |
| 17 | 5.79 (q-like, 1.7) | 120.2, CH | 17 | 6.04 (dt, 5.7, 1.8) | 131.3, CH |
| 18 | 0.64 (s) | 15.5, CH3 | 18 | 0.71 (s) | 17.6, CH3 |
| 19 | 0.84 (s) | 16.3, CH3 | 19 | 0.93 (s) | 15.2, CH3 |
| 20 | 0.98 (s) | 28.5, CH3 | 20 | 0.97 (s) | 28.5, CH3 |
| 21 | 0.84 (s) | 22.4, CH3 | 21 | 0.86 (s) | 22.3, CH3 |
| 22 | 1.05 (s) | 17.3, CH3 | 22 | 1.08 (s) | 24.9, CH3 |
| 3 | 4 | ||||
|---|---|---|---|---|---|
| no. | δH | δC | no. | δH | δC |
| 1 | 3.18 (m) | 56.7, CH | 1 | 3.61 (dd, 11.4, 4.6) | 76.6, CH |
| 2 | Ha: 1.99 (m) | 23.7, CH2 | 2 | 1.72 (m) | 26.7, CH2 |
| Hb: 1.93 (m) | |||||
| 3 | Ha: 2.45 (m) | 39.0, CH2 | 3 | 2.19 (m) | 32.4, CH2 |
| Hb: 2.36 (m) | |||||
| 4 | 158.3, C | 4 | 139.0, C | ||
| 5 | 135.3, C | 5 | 138.2, C | ||
| 6 | 202.1, C | 6 | 205.8, C | ||
| 7 | 2.46 (m) | 45.2, CH2 | 7 | Ha: 2.49 (m) | 43.2, CH2 |
| Hb: 2.29 (m) | |||||
| 8 | Ha: 1.86 (m) | 21.4, CH2 | 8 | Ha: 1.94 (m) | 20.8, CH2 |
| Hb:1.49 (m) | Hb: 1.90 (m) | ||||
| 9 | Ha: 1.95 (m) | 46.8, CH2 | 9 | Ha: 2.09 (ddd, 13.2, 5.2, 3.3) | 36.7, CH2 |
| Hb: 1.72(m) | Hb: 1.53 (ddd, 13.2, 11.1, 6.4) | ||||
| 10 | 75.1, C | 10 | 42.7, C | ||
| 11 | 0.99 (s) | 20.8, CH3 | 11 | 1.74 (s) | 21.2, CH3 |
| 12 | 2.07 (brs) | 17.2, CH3 | 12 | 0.81 (s) | 18.8, CH3 |
| 10 -OH | 4.83 (s) a | 1 -OH | 4.67 (d, 4.8) a | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.-Y.; Huang, D.-L.; Dong, Y.; Qin, D.-P.; Yan, Y.-M.; Cheng, Y.-X. Neuroprotective Norsesquiterpenoids and Triterpenoids from Populus euphratica Resins. Molecules 2019, 24, 4379. https://doi.org/10.3390/molecules24234379
Liu Y-Y, Huang D-L, Dong Y, Qin D-P, Yan Y-M, Cheng Y-X. Neuroprotective Norsesquiterpenoids and Triterpenoids from Populus euphratica Resins. Molecules. 2019; 24(23):4379. https://doi.org/10.3390/molecules24234379
Chicago/Turabian StyleLiu, Yun-Yun, Dan-Ling Huang, Yun Dong, Da-Peng Qin, Yong-Ming Yan, and Yong-Xian Cheng. 2019. "Neuroprotective Norsesquiterpenoids and Triterpenoids from Populus euphratica Resins" Molecules 24, no. 23: 4379. https://doi.org/10.3390/molecules24234379
APA StyleLiu, Y.-Y., Huang, D.-L., Dong, Y., Qin, D.-P., Yan, Y.-M., & Cheng, Y.-X. (2019). Neuroprotective Norsesquiterpenoids and Triterpenoids from Populus euphratica Resins. Molecules, 24(23), 4379. https://doi.org/10.3390/molecules24234379