Rapid Characterization and Discovery of Chemical Markers for Discrimination of Xanthii Fructus by Gas Chromatography Coupled to Mass Spectrometry
Abstract
1. Introduction
2. Results and Discussion
2.1. Analysis of VOCs by HS-SPME/GC-TOF MS
2.1.1. Chemical Profiling of Volatile Organic Compounds (VOCs)
2.1.2. Statistical Analysis of VOC Profiles
2.2. Analysis of Polar Metabolites by GC-TOF MS
2.2.1. Chemical Profiling of Polar Metabolites
2.2.2. Statistical Analysis of Polar Metabolites
2.3. Quantitative Determination of the Chemical Markers
2.4. Fatty Acid Compositions of Xanthii Fructus by GC-MS Analysis
3. Discussion
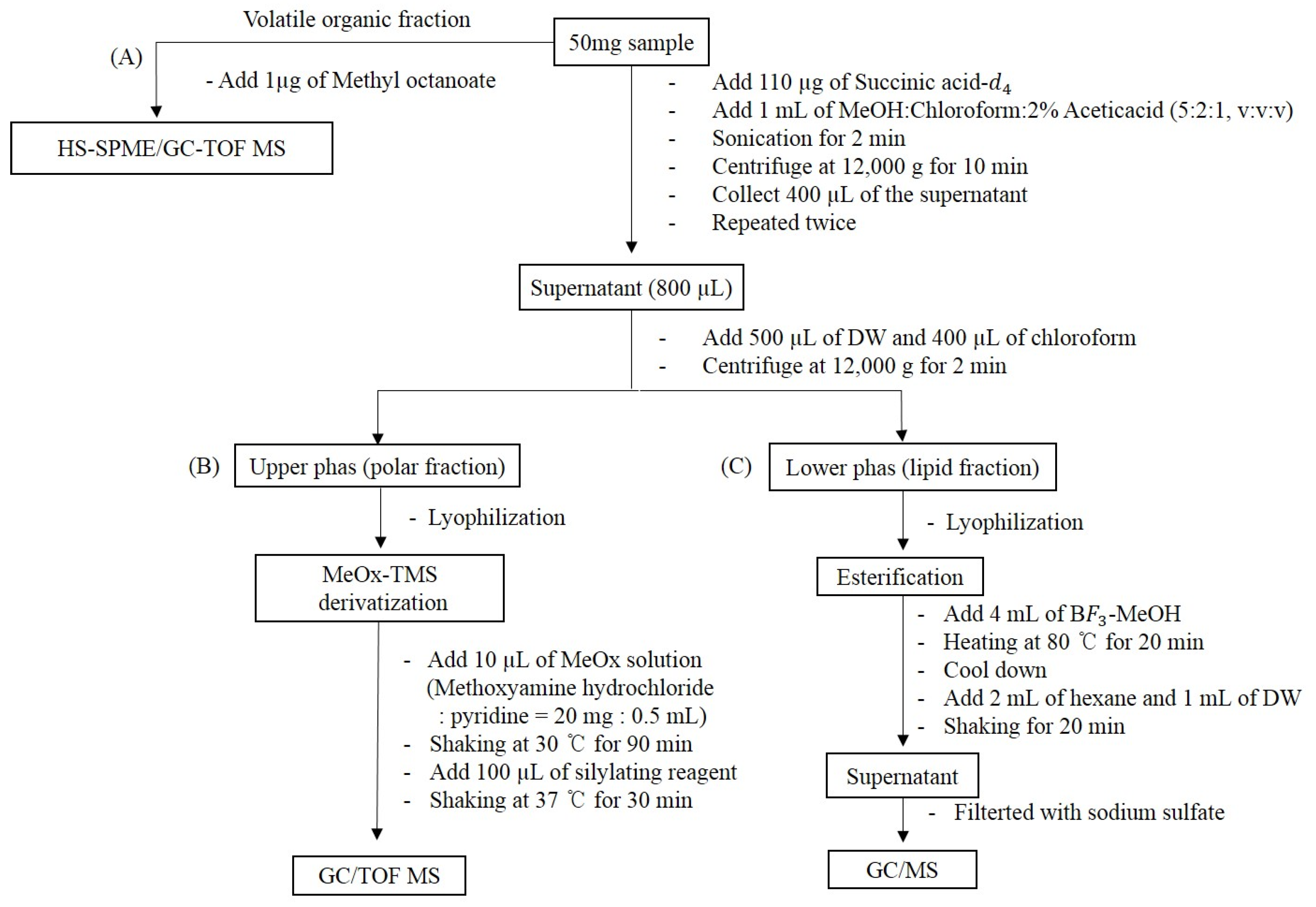
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Preparation of Samples
4.3. Preparation of Standards
4.4. GC-TOF MS Analysis
4.5. Statistical Analysis
4.6. Analysis of Fatty Acid Composition by GC-MS Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Amin, S.; Barkatullah, H.K. Pharmacology of Xanthium species. A review. J. Phytopharm. 2016, 5, 126–127. [Google Scholar]
- Goodarzi, M.; Russell, P.J.; Vander Heyden, Y. Similarity analyses of chromatographic herbal fingerprints: A review. Analytica Chimica Acta 2013, 804, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Kamboj, A.; Saluja, A.K. Phytopharmacological review of Xanthium strumarium L. (Cocklebur). Int. J. Green Pharm. 2010, 4. [Google Scholar] [CrossRef]
- Kan, S.; Chen, G.; Han, C.; Chen, Z.; Song, X.; Ren, M.; Jiang, H. Chemical constituents from the roots of Xanthium sibiricum. Nat. Prod. Res. 2011, 25, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Z.; LI, N. Chemical constituents and bioactivity of Xanthium sibiricum Patrin. ex Widder. J. Sci. Teachers’College Univer. 2016, 10. [Google Scholar]
- Sakuda, Y.; Tahara, T. The Constituents of Essential Oil from Xanthium canadense Mill. J. Japan Oil Chem. Soc. 1982, 31, 151–153. [Google Scholar] [CrossRef][Green Version]
- Li, N.; Zhang, W.-Z. Study on chemical constituents of Xanthium sibiricum Patrin ex Widder. J. Qiqihar Univer. 2016, 4, 13. [Google Scholar]
- Tang, J.S.; Jiang, C.Y.; Liu, Y.; Zhang, X.Y.; Shao, H.; Zhang, C. Allelopathic potential of volatile organic compounds released by Xanthium sibiricum Patrin ex Widder. Allelopathy J. 2019, 47, 233–241. [Google Scholar] [CrossRef]
- Henson, I.; Wareing, P. Cytokinins in Xanthium strumarium L.: Distribution in the plant and production in the root system. J. Exp. Botany 1976, 27, 1268–1278. [Google Scholar] [CrossRef]
- Kim, I.T.; Park, Y.M.; Won, J.H.; Jung, H.J.; Park, H.J.; Choi, J.W.; Lee, K.T. Methanol extract of Xanthium strumarium L. possesses anti-inflammatory and anti-nociceptive activities. Bio. Pharm. Bull. 2005, 28, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Zhao, Y.; Han, P.; Yue, W.; Ma, X.Q.; Rahman, K.; Han, T. Anti-arthritic activity of Xanthium strumarium L. extract on complete Freund’s adjuvant induced arthritis in rats. J. Ethnopharmacol. 2014, 155, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Hoseini-Alfatemi, S.M.; Sharifi-Rad, M.; Sharifi-Rad, M.; Iriti, M.; Sharifi-Rad, M.; Sharifi-Rad, R.; Raeisi, S. Phytochemical compositions and biological activities of essential oil from Xanthium strumarium L. Molecules 2015, 20, 7034–7047. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.; Agarwala, M. Phytochemical analysis of some medicinal plants. J. Phytol. 2011, 3. [Google Scholar]
- Yoon, J.H.; Lim, H.J.; Lee, H.J.; Kim, H.D.; Jeon, R.; Ryu, J.H. Inhibition of lipopolysaccharide-induced inducible nitric oxide synthase and cyclooxygenase-2 expression by xanthanolides isolated from Xanthium strumarium. Bioorg. Med. Chem. Lett. 2008, 18, 2179–2182. [Google Scholar] [CrossRef] [PubMed]
- Iordache, A.; Culea, M.; Gherman, C.; Cozar, O. Characterization of some plant extracts by GC–MS. Nucl. Ins. Methods Phys. Res. 2009, 267, 338–342. [Google Scholar] [CrossRef]
- Taherpour, A.A.; Khaef, S.; Yari, A.; Nikeafshar, S.; Fathi, M.; Ghambari, S. Chemical composition analysis of the essential oil of Mentha piperita L. from Kermanshah, Iran by hydrodistillation and HS/SPME methods. J. Analy. Sci. Technol. 2017, 8, 11. [Google Scholar] [CrossRef]
- Boyacı, E.; Rodriguez-Lafuente, A.; Gorynski, K.; Mirnaghi, F.; Souza-Silva, E.A.; Hein, D.; Pawliszyn, J. Sample preparation with solid phase microextraction and exhaustive extraction approaches: Comparison for challenging cases. Analytica Chimica Acta 2015, 873, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Manzo, A.; Panseri, S.; Vagge, I.; Giorgi, A. Volatile Fingerprint of Italian Populations of Orchids Using Solid Phase Microextraction and Gas Chromatography Coupled with Mass Spectrometry. Molecules 2014, 19, 7913–7936. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.H.; Zhou, L.D.; Chen, H.; Wang, C.Z.; Xia, Z.N.; Yuan, C.S. Solid-phase microextraction technology for in vitro and in vivo metabolite analysis. TrAC Trends Analy. Chem. 2016, 80, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Zhang, Q.Y.; Zhang, H.; Wen, J.; Wang, Y.; Huang, B.K.; Qin, L.P. Authentication and quantitative analysis on the chemical profile of Xanthium fruit (Cang-Er-Zi) by high-performance liquid chromatography-diode-array detection tandem mass spectrometry method. Analytica Chimica Acta 2009, 634, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Burruezo, A.; Kollmannsberger, H.; González-Mas, M.C.; Nitz, S.; Fernando, N. HS-SPME Comparative Analysis of Genotypic Diversity in the Volatile Fraction and Aroma-Contributing Compounds of Capsicum Fruits from the annuum−chinense−frutescens Complex. J. Agric. Food Chem. 2010, 58, 4388–4400. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Nam, P.W.; Lee, S.J.; Lee, K.G. Antioxidant activities of volatile and non-volatile fractions of selected traditionally brewed Korean rice wines. J. Ins. Brewing 2014, 120, 537–542. [Google Scholar] [CrossRef]
- Worley, B.; Powers, R. PCA as a practical indicator of OPLS-DA model reliability. Curr. Metab. 2016, 4, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Chung, I.M.; Kim, J.K.; Han, J.G.; Kong, W.S.; Kim, S.Y.; Yang, Y.J.; Kim, S.H. Potential geo-discriminative tools to trace the origins of the dried slices of shiitake (Lentinula edodes) using stable isotope ratios and OPLS-DA. Food Chem. 2019, 295, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Tambellini, N.; Zaremberg, V.; Turner, R.; Weljie, A. Evaluation of Extraction Protocols for Simultaneous Polar and Non-Polar Yeast Metabolite Analysis Using Multivariate Projection Methods. Metabolites 2013, 3, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Kind, T.; Fiehn, O. Seven Golden Rules for heuristic filtering of molecular formulas obtained by accurate mass spectrometry. BMC Bioinform. 2007, 8, 105. [Google Scholar] [CrossRef] [PubMed]
- Robert, A.; Samwel, P.M.; Limbu, M.; Chacha, J. Mwita Fatty Acid Composition and Levels of Selected Polyunsaturated Fatty Acids in Four Commercial Important Freshwater Fish Species from Lake Victoria, Tanzania. J. Lipids 2014, 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Kirk, J.L.; Beaudette, L.A.; Hart, M.; Moutoglis, P.; Klironomos, J.N.; Lee, H.; Trevors, J.T. Methods of studying soil microbial diversity. J. Microbiol. Methods 2004, 58, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, P.; Fry, F.S.; Curtis, S.K.; Al-Khaldi, S.F.; Mossoba, M.M.; Yurawecz, M.P.; Dunkel, V.C. Use of Fatty Acid Profiles to Identify Food-Borne Bacterial Pathogens and Aerobic Endospore-Forming Bacilli. J. Agric. Food Chem. 2005, 53, 3735–3742. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, H.H.; Sen, N.; Holman, R.T. Characterization of unsaturated fatty acids by gas-liquid chromatography. J. Amer. Oil Chem. Soc. 1965, 42, 537–540. [Google Scholar] [CrossRef]
- Zhang, X.J.; Huang, L.L.; Cai, X.J.; Li, P.; Wang, Y.T.; Wan, J.B. Fatty acid variability in three medicinal herbs of Panaxspecies. Chem. Central J. 2013, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Vibha, J.B.; Choudhary, K.; Singh, M.; Rathore, M.S.; Shekhawat, N.S. A study on pharmacokinetics and therapeutic efficacy of Glycyrrhiza glabra: A miracle medicinal herb. Botany Res. Int. 2009, 2, 157–163. [Google Scholar]
- Tanaka, K.; Taniguchi, S.; Tamaoki, D.; Yoshitomi, K.; Akimitsu, K.; Gomi, K. Multiple roles of plant volatiles in jasmonate-induced defense response in rice. Plant Signaling Behavior 2014, 9, e29247. [Google Scholar] [CrossRef] [PubMed]
- Ullah, I.; Khan, A.L.; Ali, L.; Khan, A.R.; Waqas, M.; Hussain, J.; Shin, J.H. Benzaldehyde as an insecticidal, antimicrobial, and antioxidant compound produced by Photorhabdus temperata M1021. J. Microbiol. 2015, 53, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Kahriman, N.; Tosun, G.; Terzioglu, S.; Karaoglu, S.A.; Yayli, N. Chemical composition and antimicrobial activity of the essential oils from the flower, leaf, and stem of Senecio pandurifolius. Records Nat. Prod. 2011, 5, 82. [Google Scholar]
- Shebl, M. Coordination behavior of new bis (tridentate ONO, ONS and ONN) donor hydrazones towards some transition metal ions: Synthesis, spectral, thermal, antimicrobial and antitumor studies. J. Mol. Struc. 2017, 1128, 79–93. [Google Scholar] [CrossRef]
- Sharma, K.; Pasricha, V.; Satpathy, G.; Gupta, R.K. Evaluation of phytochemical and antioxidant activity of raw Pyrus communis (l), an underexploited fruit. J. Pharm. Phytochem. 2015, 3. [Google Scholar]
- Goicoechea, E.; Van Twillert, K.; Duits, M.; Brandon, E.D.; Kootstra, P.R.; Blokland, M.H.; Guillén, M.D. Use of an in vitro digestion model to study the bioaccessibility of 4-hydroxy-2-nonenal and related aldehydes present in oxidized oils rich in omega-6 acyl groups. J. Agric. Food Chem. 2008, 56, 8475–8483. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.S.; Du, S.S.; Liu, Q.Z.; Liu, Q.R.; Liu, Z.L. Composition and insecticidal activity of the essential oil of Artemisia igniaria Maxim. Flowering aerial parts against Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae). J. Med. Plants Res. 2012, 6, 3188–3192. [Google Scholar]
- Holt, S.; Miks, M.H.; de Carvalho, B.T.; Foulquié-Moreno, M.R.; Thevelein, J.M. The molecular biology of fruity and floral aromas in beer and other alcoholic beverages. FEMS Microbiol. Rev. 2018, 43, 193–222. [Google Scholar] [CrossRef] [PubMed]
- Ko, K.H.; Nam, S. Antioxidant activities of volatile aroma components from Cudrania tricuspidata (carr.) bureau extracts. J. Korean Soc. Food Sci. Nutr. 2012, 41, 1493–1501. [Google Scholar] [CrossRef]
- Rohloff, J. Analysis of phenolic and cyclic compounds in plants using derivatization techniques in combination with GC-MS-based metabolite profiling. Molecules 2015, 20, 3431–3462. [Google Scholar] [CrossRef] [PubMed]
- Brglez Mojzer, E.; Knez Hrnčič, M.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef] [PubMed]
- Ros, E.; Mataix, J. Fatty acid composition of nuts–implications for cardiovascular health. British J. Nutr. 2006, 96, S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Abate, S.; Ahn, Y.G.; Kind, T.; Cataldi, T.R.; Fiehn, O. Determination of elemental compositions by gas chromatography/time-of-flight mass spectrometry using chemical and electron ionization. Rapid Commun. Mass Spectr. 2010, 24, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |





| Class | Compound | X. canadense M | X. sibiricum PW | Linear Range (ng/mg) | Calibration Curve | |||
|---|---|---|---|---|---|---|---|---|
| Concentration (ng/mg) | RSD (%) | Concentration (ng/mg) | RSD (%) | Equation | γ2 | |||
| VOCs (ng/mg) | Benzeneethanol | 3.82 | 20.11 | 0.65 | 10.15 | 0.005~25 | y = 0.0003x − 0.004 | 0.9974 |
| Benzaldehyde | 0.10 | 20.17 | n.d. | y = 0.0013x + 0.0137 | 0.9994 | |||
| 1H-Pyrrole-2-carboxaldehyde | 2.38 | 8.38 | 0.69 | 8.47 | y = 0.0003x − 0.0158 | 0.9984 | ||
| 3-Octen-2-one | 0.57 | 13.37 | n.d. | y = 0.0002x − 0.004 | 0.9978 | |||
| Butyrolactone | 57.12 | 12.76 | 31.54 | 1.20 | y = 0.000002x − 0.0002 | 0.9951 | ||
| γ-Caprolactone | 11.05 | 9.19 | 0.79 | 4.76 | y = 0.00007x − 0.0049 | 0.9974 | ||
| δ-Hexalactone | 0.37 | 1.83 | 0.32 | 0.21 | y = 0.0073x − 0.4665 | 0.9981 | ||
| Pantolactone | 19.10 | 22.31 | 2.03 | 5.02 | y = 0.00001x + 0.0009 | 0.9969 | ||
| γ-Octalactone | 2.15 | 7.79 | 0.31 | 4.93 | y = 0.0001x − 0.0049 | 0.9983 | ||
| Polar Metabolites (ng/mg) | Ethylene glycol | 102.70 | 0.17 | n.d. | 20~2000 | y = 0.0009x − 4.5373 | 0.9953 | |
| l-(−)-Arabitol | 511.18 | 2.15 | 3684.11 | 2.36 | 20~10000 | y = 0.0001x − 2.014 | 0.9955 | |
| d-Mannitol | 424.32 | 5.31 | 4404.07 | 13.64 | 20~2000 | y = 0.00006x − 0.2687 | 0.9921 | |
| Scyllo-inositol | 1080.15 | 1.41 | 650.46 | 5.46 | 20~4000 | y = 0.0001x − 1.4494 | 0.9947 | |
| Succinic acid | 750.50 | 3.09 | 259.24 | 2.73 | 20~4000 | y = 0.00005x − 0.5167 | 0.995 | |
| d-Glyceric acid | 267.90 | 6.19 | 205.64 | 8.27 | 20~2000 | y = 0.0001x − 0.6062 | 0.9939 | |
| Fumaric acid | 185.21 | 10.72 | n.d. | 20~2000 | y = 0.00003x − 0.1361 | 0.995 | ||
| Malic acid | 422.72 | 9.63 | n.d. | 20~4000 | y = 0.00008x − 0.832 | 0.994 | ||
| Azelaic acid | 353.06 | 13.43 | n.d. | 20~2000 | y = 0.000008x − 0.0378 | 0.9931 | ||
| Gluconic acid | 141.72 | 10.74 | n.d. | 20~2000 | y = 0.00003x + 0.1344 | 0.9915 | ||
| d-Psicofuranose | 866.10 | 5.26 | 3748.90 | 1.57 | 20~10000 | y = 0.0001x −2.1083 | 0.9948 | |
| Fatty Acids | X. canadense M | X. sibiricum PW | ||||
|---|---|---|---|---|---|---|
| Common Name | Symbol | GC RT | % | %RSD | % | %RSD |
| Lauric | C12:0 | 27.35 | 0.1 | 0.7 | 0.1 | 2.2 |
| Tridecanoic | C13:0 | 29.27 | 0.3 | 6.9 | 0.3 | 3.9 |
| Palmitic | C16:0 | 34.06 | 0.3 | 3.6 | 0.2 | 5.6 |
| Saturated fatty acids (SFA) | 0.7 | 0.6 | ||||
| Myristoleic | C14:1 | 31.11 | 0.2 | 6.1 | 0.2 | 1.5 |
| Cis-10-pentadecanoic | C15:1 | 32.86 | 19.4 | 0.3 | 20.4 | 1.0 |
| Palmitoleic | C16:1 | 34.50 | 0.3 | 1.1 | 0.3 | 2.5 |
| Cis-10-heptadecenoic | C17:1 | 36.07 | 5.0 | 0.9 | 5.9 | 0.9 |
| Elaidic | C18:1n9t | 37.09 | 8.1 | 0.6 | 20.3 | 0.3 |
| Cis-11-eicosanoic | C20:1n9 | 40.19 | 1.7 | 1.2 | 0.8 | 4.5 |
| Nervonic | C24:1n9 | 45.72 | 0.2 | 12.8 | 1.7 | 2.4 |
| Monounsaturated (MUFA) | 34.9 | 49.6 | ||||
| Linolelaidic | C18:2n6t | 38.58 | 61.5 | 0.2 | 48.6 | 0.3 |
| Linoleic | C18:2n6c | 39.04 | 0.6 | 9.4 | 0.3 | 8.2 |
| Gamma-linolenic | C18:3n3-6 | 39.96 | 0.1 | 15.2 | 0.1 | 2.3 |
| Linolenic | C18:3n3-3 | 40.40 | 0.3 | 28.3 | - | - |
| Cis-11,14-eicosadienoic | C20:2 | 41.81 | 1.2 | 2.1 | 0.6 | 2.9 |
| Cis-11,14,17-eicosatrienoic | C20:3n3 | 43.22 | 0.3 | 7.0 | 0.1 | 20.9 |
| Cis-13,16-docosadienoic | C22:2 | 44.68 | 0.4 | 5.8 | 0.1 | 9.6 |
| Polyunsaturated (PUFA) | 64.4 | 49.8 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Jung, Y.; Jeon, S.H.; Hwang, G.-S.; Ahn, Y.G. Rapid Characterization and Discovery of Chemical Markers for Discrimination of Xanthii Fructus by Gas Chromatography Coupled to Mass Spectrometry. Molecules 2019, 24, 4079. https://doi.org/10.3390/molecules24224079
Kim H, Jung Y, Jeon SH, Hwang G-S, Ahn YG. Rapid Characterization and Discovery of Chemical Markers for Discrimination of Xanthii Fructus by Gas Chromatography Coupled to Mass Spectrometry. Molecules. 2019; 24(22):4079. https://doi.org/10.3390/molecules24224079
Chicago/Turabian StyleKim, Hayoung, Youngae Jung, So Hyeon Jeon, Geum-Sook Hwang, and Yun Gyong Ahn. 2019. "Rapid Characterization and Discovery of Chemical Markers for Discrimination of Xanthii Fructus by Gas Chromatography Coupled to Mass Spectrometry" Molecules 24, no. 22: 4079. https://doi.org/10.3390/molecules24224079
APA StyleKim, H., Jung, Y., Jeon, S. H., Hwang, G.-S., & Ahn, Y. G. (2019). Rapid Characterization and Discovery of Chemical Markers for Discrimination of Xanthii Fructus by Gas Chromatography Coupled to Mass Spectrometry. Molecules, 24(22), 4079. https://doi.org/10.3390/molecules24224079







