The Synthesis and Properties of Liquid Crystalline Polyurethanes, Chemically Modified by Polyhedral Oligomericsilsesquioxanes
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of 4,4′-Bis(hydroxyhexyloxy)bisphenyl (BHHBP)
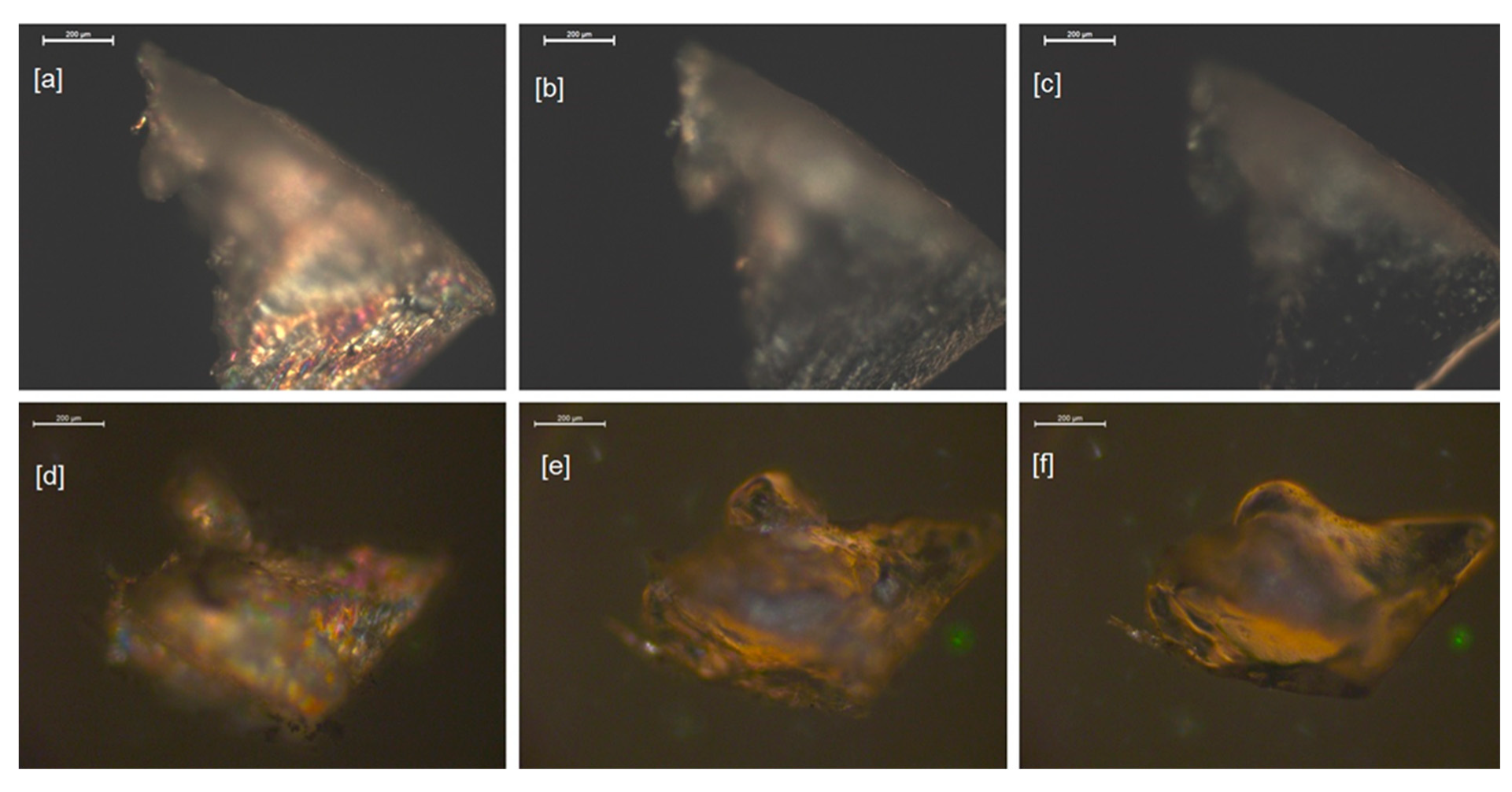
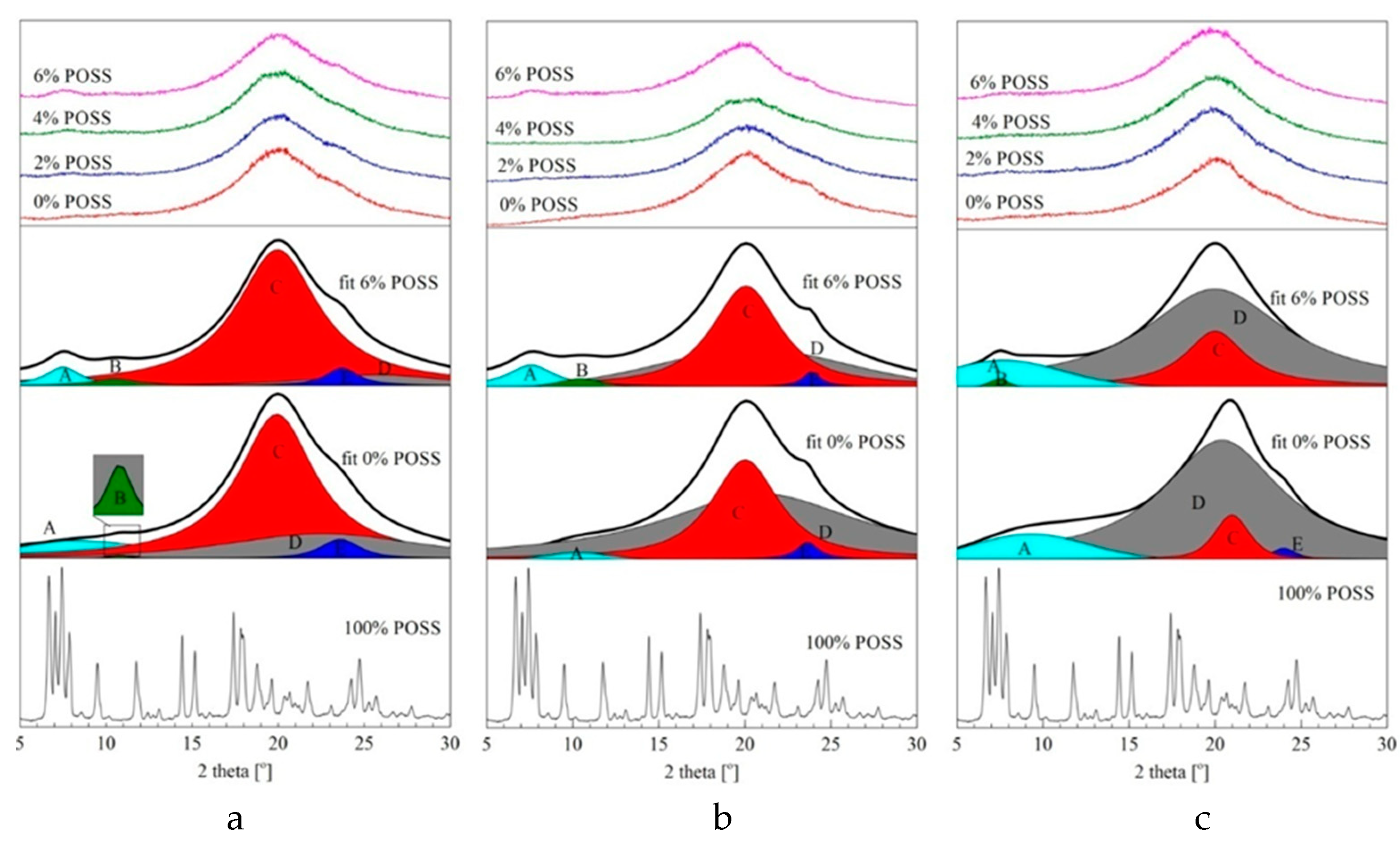
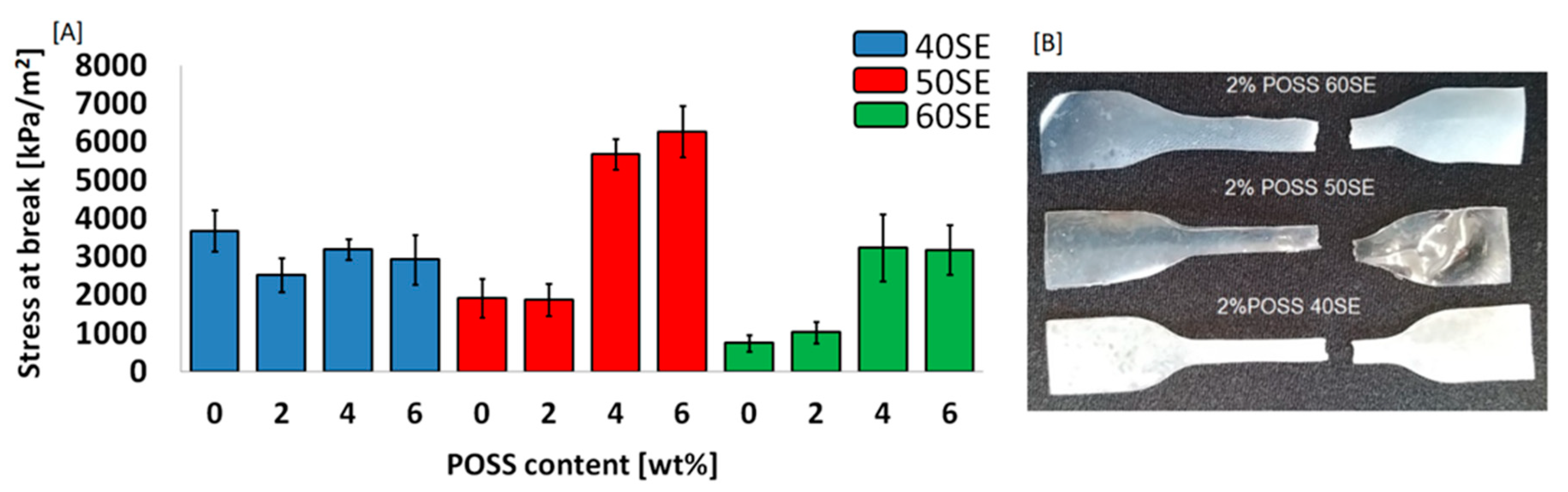
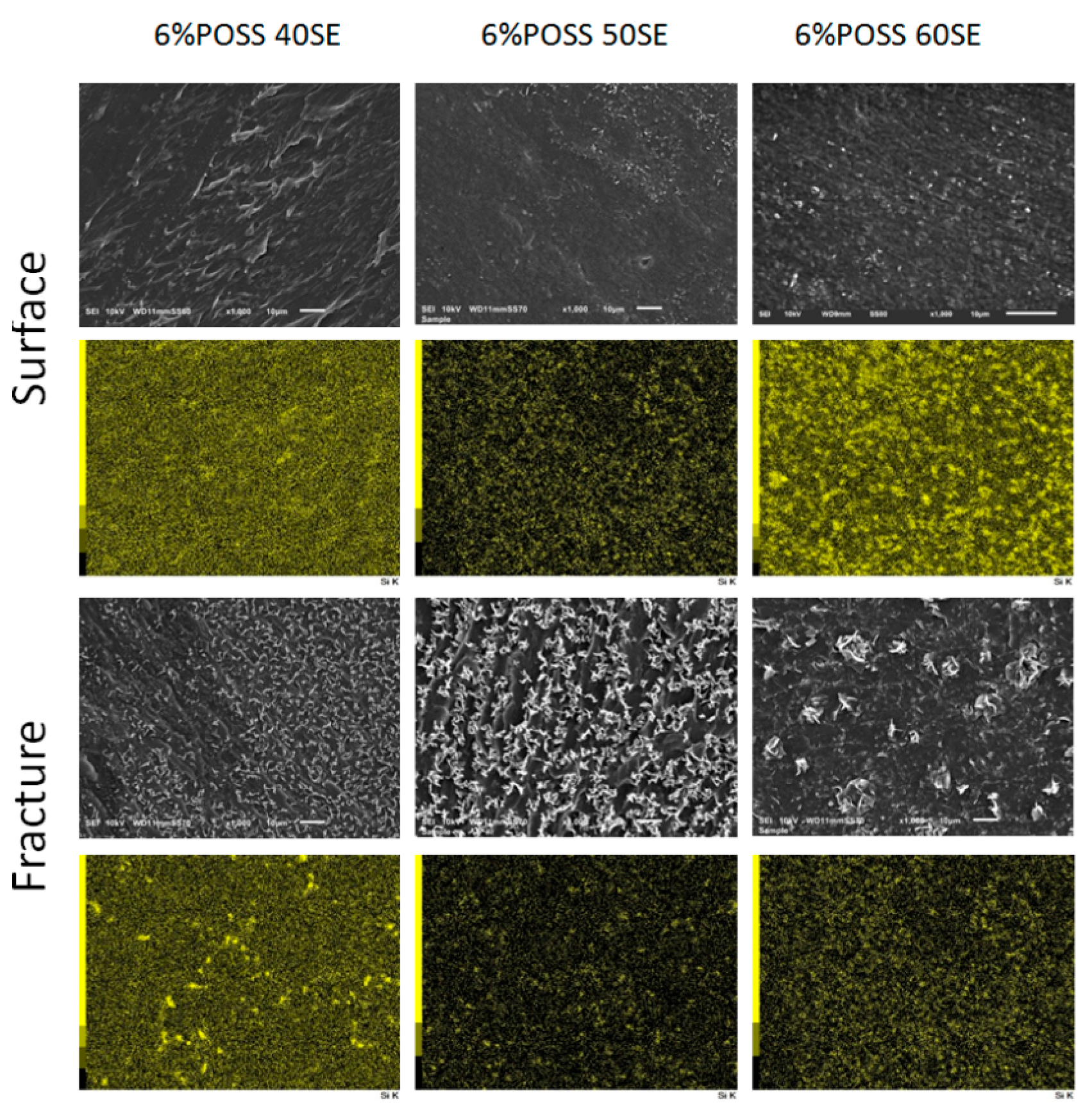
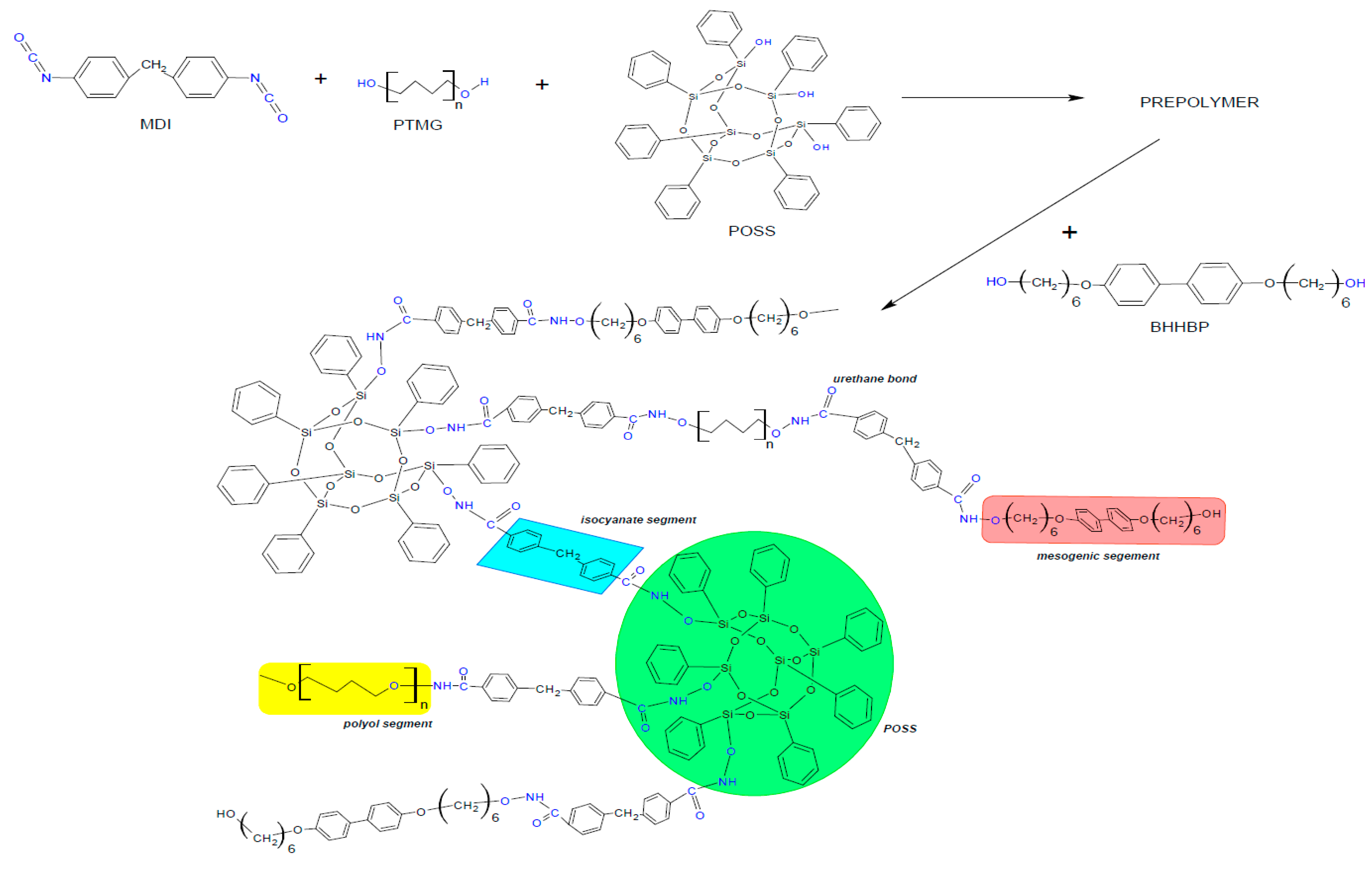
2.2. Characterisation of LCPU/POSS Nanohybrids
3. Materials and Methods
3.1. Synthesis of Mesogenic Diol
3.2. Synthesis of the Liquid Crystalline PU/POSS Hybrids
3.3. Infrared Spectroscopy with Fourier Transformations (FTIR)
3.4. Differential Scanning Calorimetry (DSC)
3.5. Polarized Optical Microscopy (POM)
3.6. Wide-Angle X-Ray Diffraction (WAXD)
3.7. Thermogravimetric Analysis (TGA)
3.8. Mechanical Properties
3.9. Scanning Electron Microscopy (SEM)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Padmavathy, T.; Srinivasan, K.S.V. Liquid Crystalline Polyurethanes — A Review. J. Macromol. Sci. Part C 2007, 43, 45–85. [Google Scholar] [CrossRef]
- Krol, P. Synthesis methods, chemical structures and phase structures of linear polyurethanes. Properties and applications of linear polyurethanes in polyurethane elastomers, copolymers and ionomers. Prog. Mater. Sci. 2007, 52, 915–1015. [Google Scholar] [CrossRef]
- Li, Y.; Lian, H.; Hu, Y.; Chang, W.; Cui, X.; Liu, Y. Enhancement in Mechanical and Shape Memory Properties for Liquid Crystalline Polyurethane Strengthened by Graphene Oxide. Polymers 2016, 8, 236. [Google Scholar] [CrossRef] [PubMed]
- Ban, J.; Zhu, L.; Chen, S.; Wang, Y. The impact of liquid crystal fillers on structure and properties of liquid-crystalline shape-memory polyurethane composites I: 4-dodecyloxybenzoic acid. J. Mater. Sci. 2016, 51, 10229–10244. [Google Scholar] [CrossRef]
- Min, U.; Chang, J. Thermotropic liquid crystalline polyazomethine nanocomposites via in situ interlayer polymerization. Mater. Chem. Phys. 2011, 129, 517–522. [Google Scholar] [CrossRef]
- Tang, Y.; Gao, P.; Ye, L. Organoclay/thermotropic liquid crystalline polymer nanocomposites. Part V: Morphological and rheological studies. J. Mater. Sci. 2010, 45, 2874–2883. [Google Scholar] [CrossRef]
- Riou, O.; Lonetti, B.; Davidson, P.; Tan, R.P.; Cormary, B.; Mingotaud, A.F.; Di Cola, E.; Respaud, M.; Chaudret, B.; Soulantica, K.; et al. Liquid crystalline polymer-co nanorod hybrids: Structural analysis and response to a magnetic field. J. Phys. Chem. B. 2014, 118, 3218–3225. [Google Scholar] [CrossRef]
- Wang, M.; Sayed, S.M.; Guo, L.; Lin, B.; Zhang, X.; Sun, Y.; Yang, H. Multi-Stimuli Responsive Carbon Nanotube Incorporated Polysiloxane Azobenzene Liquid Crystalline Elastomer Composites. Macromolecules 2016, 49, 663–671. [Google Scholar] [CrossRef]
- Talmon, Y. Direct Imaging of Carbon Nanotube Liquid-Crystalline Phase Development in True Solutions. Langmuir 2017, 34, 4011–4018. [Google Scholar]
- Zhang, D.; Zhou, X.; Chen, S.; Luo, H.; Bowen, C.R.; Zhou, K. High Performance Capacitors Using BaTiO 3 Nanowires Engineered by Rigid Liquid-crystalline Polymers. J. Phys. Chem. C. 2017, 121, 20075–20083. [Google Scholar] [CrossRef]
- Cheng, Z.; Wang, T.; Li, X.; Zhang, Y.; Yu, H. NIR−Vis−UV Light-Responsive Actuator Films of Polymer-Dispersed Liquid Crystal / Graphene Oxide Nanocomposites. ACS. Appl. Mater. Inter. 2015, 7, 27494–27501. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Liu, Q.; Lv, X.; Gao, L. Photoinduced triple shape memory polyurethane enabled by doping with azobenzene and GO. J. Mater. Chem. C. 2016, 4, 9993–9997. [Google Scholar] [CrossRef]
- Zhang, P.; Wu, B.; Huang, S.; Cai, F.; Wang, G.; Yu, H. UV–vis–NIR light-induced bending of shape-memory polyurethane composites doped with azobenzene and upconversion nanoparticles. Polymer 2019, 178, 121644–121649. [Google Scholar] [CrossRef]
- Zhang, S.; Zou, Q.; Wu, L. Preparation and Characterization of Polyurethane Hybrids from Reactive Polyhedral Oligomeric Silsesquioxanes. Macromol. Mater. Eng. 2006, 291, 895–901. [Google Scholar] [CrossRef]
- Zhao, B.; Xu, S.; Adeel, M.; Zheng, S. Formation of POSS-POSS interactions in polyurethanes: From synthesis, morphologies to shape memory properties of materials. Polymer 2019, 160, 82–92. [Google Scholar] [CrossRef]
- Szłapka, M. Reactive & Functional Polymers Novel hybrid polyurethane / POSS materials via bulk polymerization. React. Funct. Polym. 2013, 73, 114–121. [Google Scholar]
- Wang, X.T.; Yang, Y.K.; Yang, Z.F.; Zhou, X.P.; Liao, Y.G.; Lv, C.C.; Chang, F.C.; Xie, X.L. Thermal properties and liquid crystallinity of side-chain azobenzene copolymer containing pendant polyhedral oligomeric silsequioxanes. J. Therm. Anal. Calorim. 2010, 102, 739–744. [Google Scholar] [CrossRef]
- Hsu, C.; Huang, M.; Lee, S.H.; Jeong, K. Asymmetric Organic − Inorganic Hybrid Giant Molecule: Cyanobiphenyl Monosubstituted Polyhedral Oligomeric Silsesquioxane Nanoparticles for Vertical Alignment of Liquid Crystals. J. Phys. Chem. C. 2014, 118, 6300–6306. [Google Scholar]
- Kim, K.; Chujo, Y. Liquid-crystalline organic–inorganic hybrid polymers with functionalized silsesquioxanes. J. Polym. Sci. Part A Polym. Chem. 2001, 39, 4035–4043. [Google Scholar] [CrossRef]
- Tanaka, K.; Ishiguro, F.; Jeon, J.; Hiraoka, T.; Chujo, Y. POSS ionic liquid crystals. NPG. Asia. Mater. 2015, 7, 174. [Google Scholar] [CrossRef]
- Szczepaniak, B.; Frisch Kurt, C.; Penczek, P.; Mejsner, J.; Leszczyńska, I.; Rudnik, E. Effect of the poly(oxytetramethylene)diol spacer length on some properties of liquid crystalline polyurethanes. J. Polym. Sci. Part A Polym. Chem. 1993, 31, 3223–3230. [Google Scholar] [CrossRef]
- Wypych, G. Fillers: Chemical Composition, Morphology, Performance, Applications; Elsevier: Toronto, ON, Canada, 2018. [Google Scholar]
- Dierking, I. Textures of Liquid Crystals; Wiley-VCH Verlag: Weinheim, Germany, 2003. [Google Scholar]
- Wolinska-Grabczy, A.; Kaczmarczyk, B.; Jankowski, A. Investigations of hydrogen bonding in the poly(urethane-urea)-based membrane materials by using FTIR spectroscopy. Polish. J. Chem. Technol. 2008, 10, 53–56. [Google Scholar]
- Skrovanek, D.J.; Painter, P.C.; Coleman, M.M. Hydrogen Bonding in Polymers. 2. Infrared Temperature Studies of Nylon 11. Macromolecules 1986, 19, 699–705. [Google Scholar] [CrossRef]
- Raftopoulos, K.N.; Koutsoumpis, S.; Jancia, M.; Lewicki, J.P.; Kyriakos, K.; Mason, H.E.; Harley, S.J.; Hebda, E.; Papadakis, C.M.; Pielichowski, K.; et al. Reduced phase separation and slowing of dynamics in polyurethanes with three-dimensional POSS-based cross-linking moieties. Macromolecules 2015, 48, 1429–1441. [Google Scholar] [CrossRef]
- Lewicki, J.P.; Harley, S.J.; Loui, A.; Pielichowski, K.; Mayer, B.P.; Janowski, B.; Maxwell, R.S. The Influence of Polyhedral Oligomeric Silsequioxanes on Domain Microstructure in Polyurethane Elastomers. Silicon 2013, 5, 205–212. [Google Scholar] [CrossRef]
- Lewicki, J.; Pielichowski, K.; De la Croix, P.; Janowski, B.; Todd, D.; Liggat, J. Thermal degradation studies of polyurethane/POSS nanohybrid elastomers. Polym. Degrad. Stabil. 2010, 95, 1099–1105. [Google Scholar] [CrossRef]
- Janowski, B.; Pielichowski, K.; Kwiatkowski, R. Uklady nanohybrydowe poliuretan (PUR)/funkcjonalizowany silseskwioksan (PHIPOSS). Cz. II. Rentgenowskie badania strukturalne metodamiWAXD i SAXS. Polimery 2014, 59, 147–159. [Google Scholar] [CrossRef]
- Lee, J.B.; Kato, T.; Yoshida, T. Synthesis and Liquid Crystalline Properties of Thermotropic Polyurethanes Prepared from 1,4-Diisocyanates and 4,4′-Bis(a-hydroxyalkoxy)biphenyls. Macromolecules 1993, 26, 1–6. [Google Scholar] [CrossRef]
Sample Availability: Samples are available from the authors for limited time. |

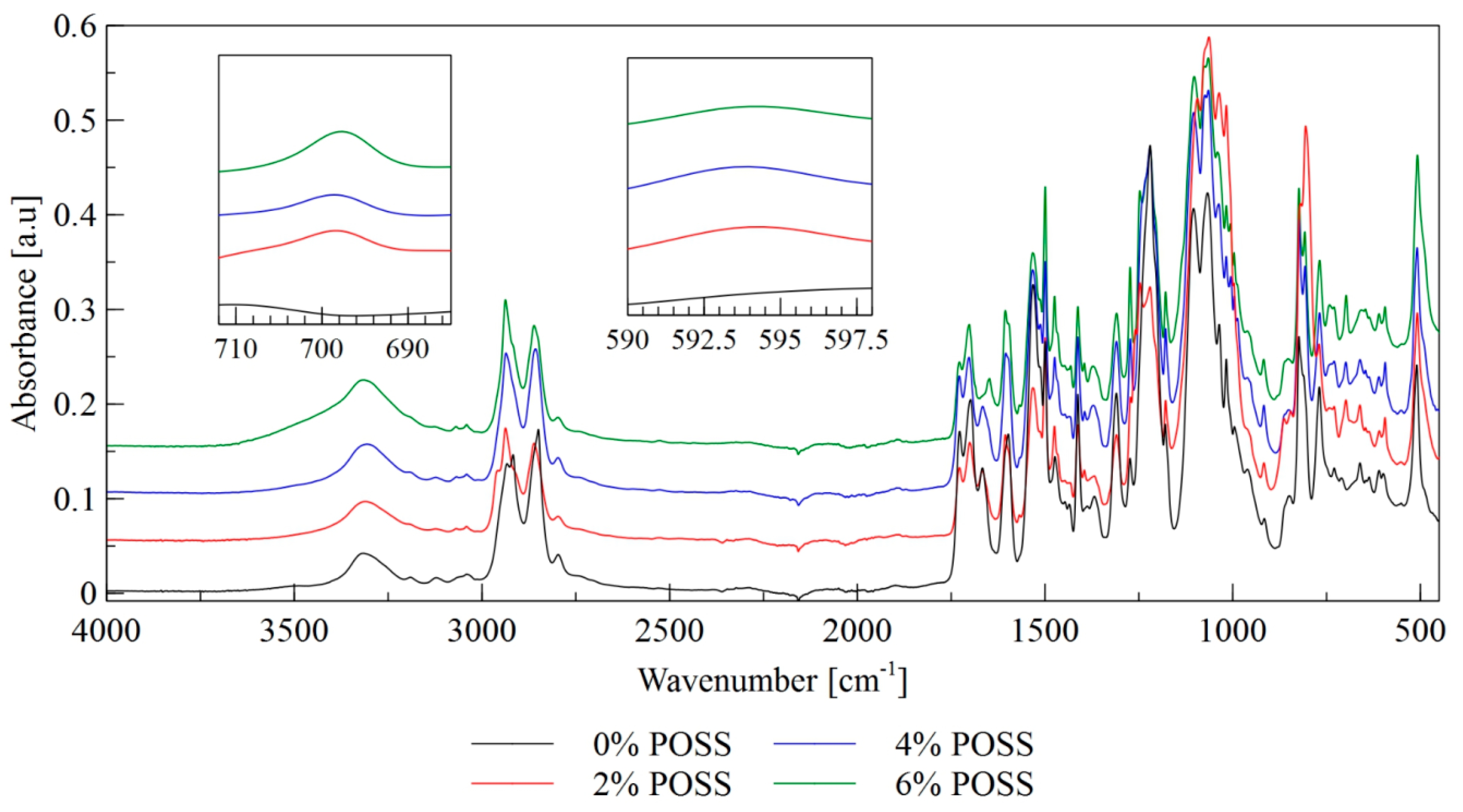

| Frequency Range [cm−1] | Functional Groups | Vibration Mode |
|---|---|---|
| 3300 | N-H | stretching vibrations of hydrogen bonded NH groups |
| 2950 | C-H | asymmetric stretching vibrations |
| 2850 | C-H | symmetric stretching vibrations |
| 1720–1660 | C=O | stretching vibrations of urethane carbonyl groups |
| 1725 | C=O | stretching vibrations of the free carbonyl groups |
| 1700 | C=O | stretching vibrations of the ordered hydrogen bonded carbonyl groups present in the urethane linkages |
| 1650 | C=O | stretching vibration of the disordered carbonyl groups that are hydrogen bonded in the soft phase |
| 1260–1230 | C=N | stretching vibrations |
| 1250 | CH2OH | stretching vibrations |
| 1150–1030 | C-O-C Si-O-Si | asymmetric stretching vibration of ether groups in soft segments stretching vibrations of groups in POSS cage |
| 1100 | C-O-C | stretching vibrations of free ether groups in the soft segments |
| 1040 | C-O-C | stretching vibrations of hydrogen bonded ether groups |
| 700 | Si-C | stretching vibrations |
| 610–590 | Si-O | stretching vibrations |
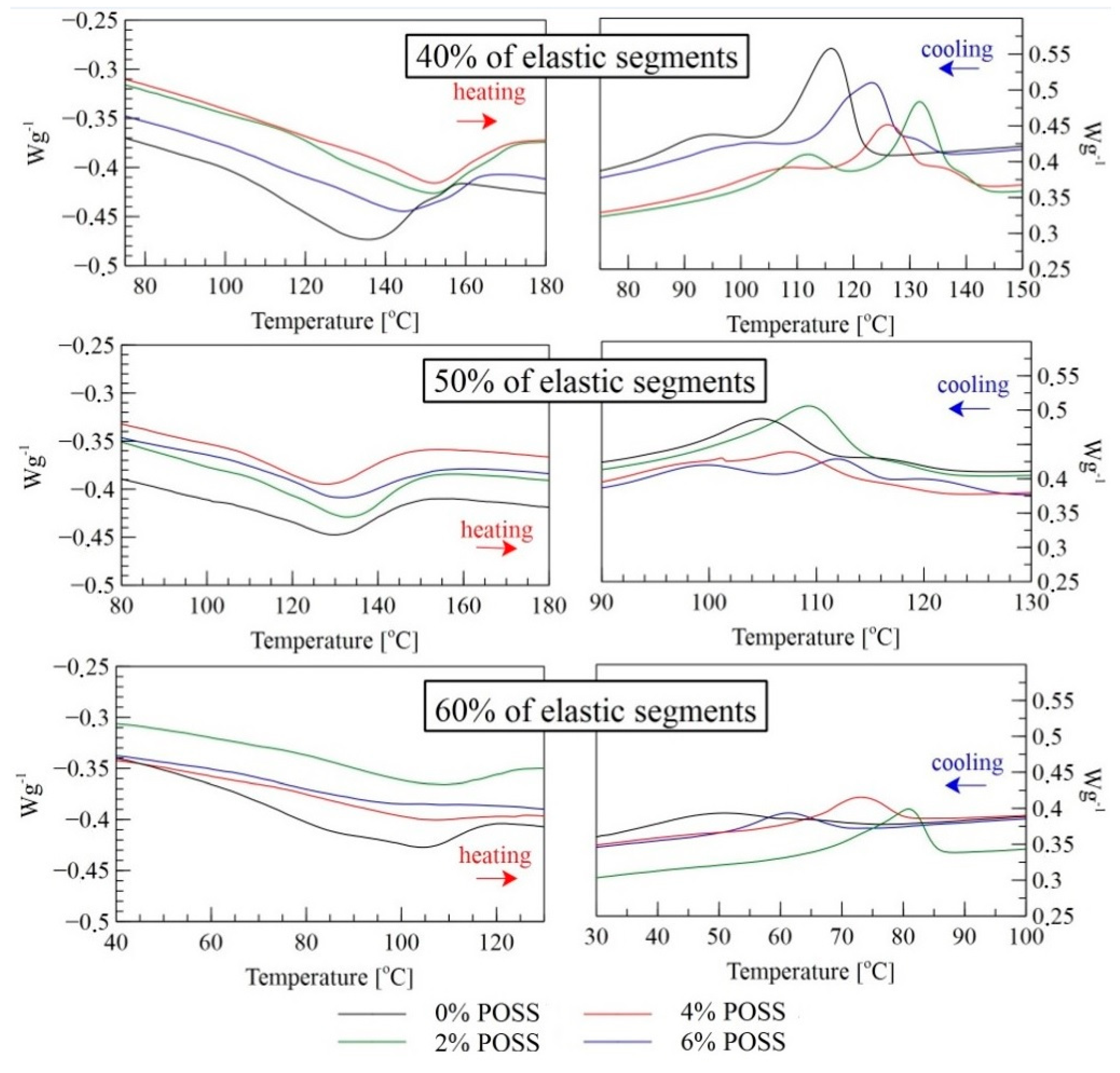
| Sample | Heating | Cooling | ||
|---|---|---|---|---|
| Tm [°C] | Ti [°C] | Ti–lc [°C] | Tlc–k [°C] | |
| 0%POSS 40SE | 105 | 150 | 125 | 72 |
| 2%POSS 40SE | 120 | 170 | 143 | 109 |
| 4%POSS 40SE | 125 | 170 | 141 | 96 |
| 6%POSS 40SE | 115 | 160 | 136 | 92 |
| 0%POSS 50SE | 110 | 145 | 122 | 94 |
| 2%POSS 50SE | 109 | 153 | 124 | 96 |
| 4%POSS 50SE | 110 | 135 | 122 | 90 |
| 6%POSS 50SE | 115 | 147 | 128 | 90 |
| 0%POSS 60SE | 60 | 115 | 66 | 33 |
| 2%POSS 60SE | 90 | 120 | 85 | 64 |
| 4%POSS 60SE | 85 | 122 | 80 | 65 |
| 6%POSS 60SE | 82 | 98 | 69 | 53 |
| %POSS | 40SE | 50SE | 60SE | 40SE | 50SE | 60SE |
|---|---|---|---|---|---|---|
| TI [°C] | TI [°C] | TI [°C] | Tflow [°C] | Tflow [°C] | Tflow [°C] | |
| 0% | 155 ± 10 | 140 ± 10 | 105 ± 10 | 165 | 152 | 120 |
| 2% | 160 ± 10 | 140 ± 10 | 120 ± 10 | 170 | 155 | 125 |
| 4% | 165 ± 10 | 145 ± 10 | 110 ± 10 | 177 | 160 | 145 |
| 6% | 175 ± 10 | 155 ± 10 | 120 ± 10 | 180 | 170 | Wide range 100–150 |
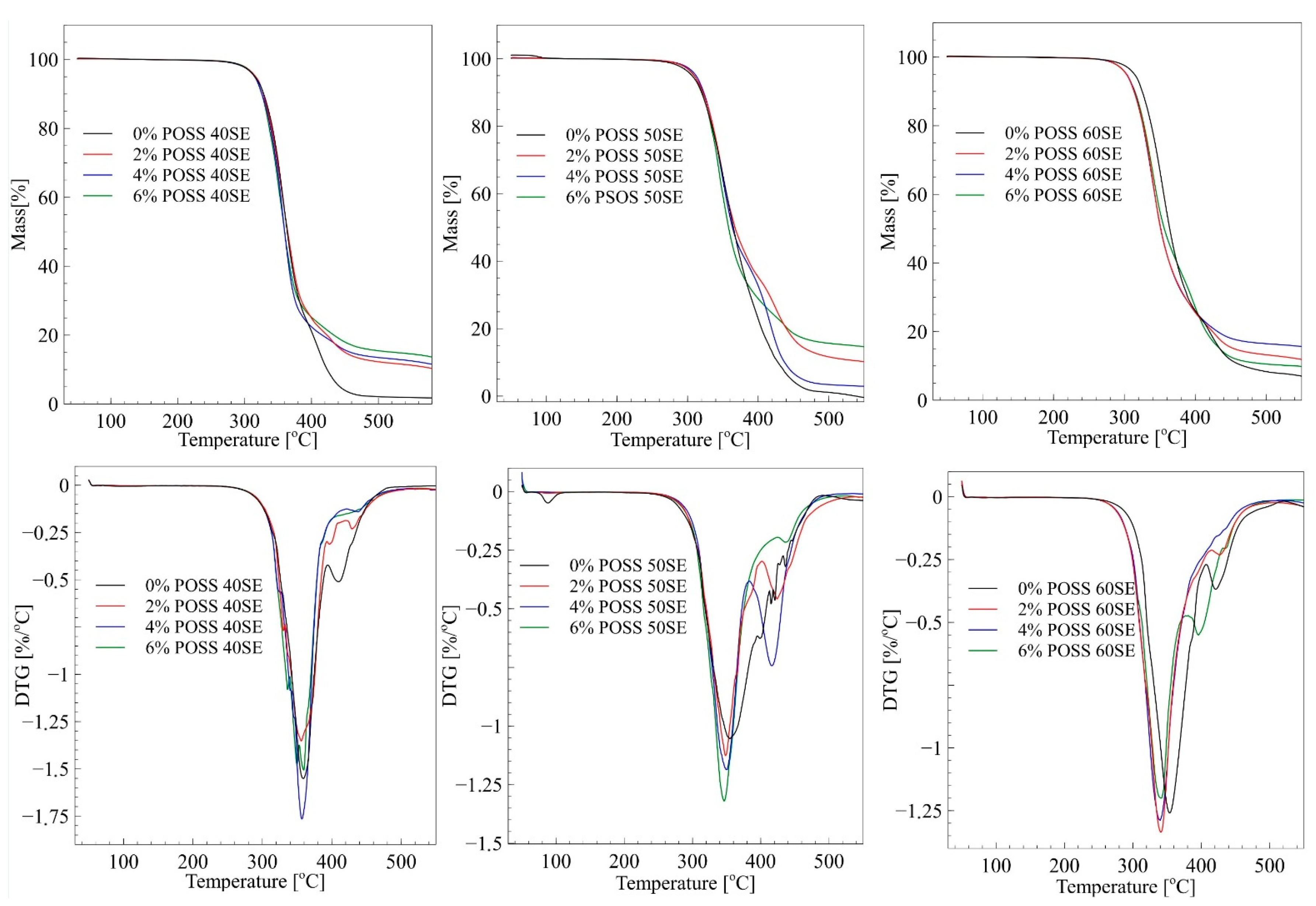
| Sample | T1% [°C] | T3% [°C] | T5% [°C] | T10% [°C] | T50% [°C] | DTG max [°C] | Residue at 500 °C [%] | |
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | |||||||
| 0% POSS 40SE | 282.3 | 305.5 | 315.5 | 328.5 | 363.7 | 359.3 | 409.8 | 2.13 |
| 2% POSS 40SE | 282.1 | 306.4 | 316.7 | 328.0 | 364.4 | 355.9 | 430.6 | 12.25 |
| 4% POSS 40SE | 280.5 | 305.4 | 314.9 | 325.8 | 359.8 | 357.3 | 436.6 | 13.49 |
| 6% POSS 40SE | 284.8 | 306.9 | 315.8 | 325.8 | 359.8 | 357.4 | 405.7 | 15.46 |
| 0% POSS 50SE | 270.4 | 295.3 | 306.4 | 319.9 | 364.9 | 355.5 | - | 1.10 |
| 2% POSS 50SE | 277.9 | 300.1 | 309.2 | 321.9 | 367.2 | 349.0 | 425.1 | 11.63 |
| 4% POSS 50SE | 279.5 | 303.0 | 312.1 | 323.0 | 363.7 | 349.5 | 416.2 | 3.41 |
| 6% POSS 50SE | 277.7 | 300.4 | 309.7 | 320.6 | 358.2 | 346.2 | 436.2 | 15.66 |
| 0% POSS 60SE | 278.3 | 304.5 | 314.2 | 325.1 | 363.9 | 353.8 | 421.0 | 8.34 |
| 2% POSS 60SE | 274.7 | 294.3 | 302.9 | 313.5 | 351.0 | 341.1 | 426.6 | 13.25 |
| 4% POSS 60SE | 273.7 | 294.4 | 303.2 | 314.0 | 351.3 | 339.2 | - | 16.48 |
| 6% POSS 60SE | 273.4 | 293.9 | 302.9 | 314.9 | 356.3 | 340.7 | 433.9 | 10.54 |
| Sample | χ | Sample | χ | Sample | χ |
|---|---|---|---|---|---|
| [%] | [%] | [%] | |||
| 0%POSS 40SE | 69 | 0%POSS 50SE | 34 | 0%POSS 60SE | 7 |
| 2%POSS 40SE | 77 | 2%POSS 50SE | 32 | 2%POSS 60SE | 10 |
| 4%POSS 40SE | 69 | 4%POSS 50SE | 32 | 4%POSS 60SE | 12 |
| 6%POSS 40SE | 68 | 6%POSS 50SE | 34 | 6%POSS 60SE | 12 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bukowczan, A.; Hebda, E.; Czajkowski, M.; Pielichowski, K. The Synthesis and Properties of Liquid Crystalline Polyurethanes, Chemically Modified by Polyhedral Oligomericsilsesquioxanes. Molecules 2019, 24, 4013. https://doi.org/10.3390/molecules24224013
Bukowczan A, Hebda E, Czajkowski M, Pielichowski K. The Synthesis and Properties of Liquid Crystalline Polyurethanes, Chemically Modified by Polyhedral Oligomericsilsesquioxanes. Molecules. 2019; 24(22):4013. https://doi.org/10.3390/molecules24224013
Chicago/Turabian StyleBukowczan, Artur, Edyta Hebda, Maciej Czajkowski, and Krzysztof Pielichowski. 2019. "The Synthesis and Properties of Liquid Crystalline Polyurethanes, Chemically Modified by Polyhedral Oligomericsilsesquioxanes" Molecules 24, no. 22: 4013. https://doi.org/10.3390/molecules24224013
APA StyleBukowczan, A., Hebda, E., Czajkowski, M., & Pielichowski, K. (2019). The Synthesis and Properties of Liquid Crystalline Polyurethanes, Chemically Modified by Polyhedral Oligomericsilsesquioxanes. Molecules, 24(22), 4013. https://doi.org/10.3390/molecules24224013






