Elucidation of the Interaction between Flavan-3-ols and Bovine Serum Albumin and Its Effect on Their In-Vitro Cytotoxicity
Abstract
1. Introduction
2. Results
2.1. Interaction between FLs and BSA
2.2. Interaction between BSA and (−)-Epicatechin and Procyanidins
2.3. Docking Studies
2.4. Influence of BSA on Cytotoxicity of FLs
2.5. Comparison of Cytotoxicity of Procyanidins
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Analysis of Fluorescence Quenching of BSA
4.3. Molecular Docking Experiment
4.4. Cytotoxicity Test
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Osakabe, N.; Terao, J. Possible mechanisms of postprandial physiological alterations following flavan 3-ol ingestion. Nutr. Rev. 2018, 76, 174–186. [Google Scholar] [CrossRef]
- Osakabe, N.; Yamagishi, M.; Sanbongi, C.; Natsume, M.; Takizawa, T.; Osawa, T. The antioxidative substances in cacao liquor. J. Nutr. Sci Vitaminol. 1998, 44, 313–321. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wollmann, N.; Hofmann, T. Compositional and sensory characterization of red wine polymers. J. Agric. Food Chem. 2013, 61, 2045–2061. [Google Scholar] [CrossRef]
- Ito, C.; Oki, T.; Yoshida, T.; Nanba, F.; Yamada, K.; Toda, T. Characterisation of proanthocyanidins from black soybeans: Isolation and characterisation of proanthocyanidin oligomers from black soybean seed coats. Food Chem. 2013, 141, 2507–2512. [Google Scholar] [CrossRef]
- Schewe, T.; Steffen, Y.; Sies, H. How do dietary flavanols improve vascular function? A position paper. Arch. Biochem. Biophys. 2008, 476, 102–106. [Google Scholar] [CrossRef]
- Latham, L.S.; Hensen, Z.K.; Minor, D.S. Chocolate—guilty pleasure or healthy supplement? J. Clin. Hypertens. 2014, 16, 101–106. [Google Scholar] [CrossRef]
- Desch, S.; Schmidt, J.; Kobler, D.; Sonnabend, M.; Eitel, I.; Sareban, M.; Rahimi, K.; Schuler, G.; Thiele, H. Effect of cocoa products on blood pressure: Systematic review and meta-analysis. Am. J. Hypertens. 2010, 23, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Baba, S.; Osakabe, N.; Kato, Y.; Natsume, M.; Yasuda, A.; Kido, T.; Fukuda, K.; Muto, Y.; Kondo, K. Continuous intake of polyphenolic compounds containing cocoa powder reduces LDL oxidative susceptibility and has beneficial effects on plasma HDL-cholesterol concentrations in humans. Am. J. Clin. Nutr. 2007, 85, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Knekt, P.; Kumpulainen, J.; Jarvinen, R.; Rissanen, H.; Heliovaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Caton, P.W.; Pothecary, M.R.; Lees, D.M.; Khan, N.Q.; Wood, E.G.; Shoji, T.; Kanda, T.; Rull, G.; Corder, R. Regulation of vascular endothelial function by procyanidin-rich foods and beverages. J. Agric. Food Chem. 2010, 58, 4008–4013. [Google Scholar] [CrossRef]
- Kumar, R.; Deep, G.; Wempe, M.F.; Agarwal, R.; Agarwal, C. Procyanidin B2 3,3’’-di-O-gallate inhibits endothelial cells growth and motility by targeting VEGFR2 and integrin signaling pathways. Curr. Cancer Drug Targets 2015, 15, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, Y.; Shao, H.; Bi, Q.; Chen, J.; Ye, Z. Grape seed procyanidin B2 inhibits adipogenesis of 3T3-L1 cells by targeting peroxisome proliferator-activated receptor gamma with miR-483-5p involved mechanism. Biomed. Pharmacother. 2017, 86, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, Y.; Okabe, M.; Natsume, M.; Ashida, H. Cacao liquor procyanidins prevent postprandial hyperglycaemia by increasing glucagon-like peptide-1 activity and AMP-activated protein kinase in mice. J. Nutr. Sci. 2019, 8, e2. [Google Scholar] [CrossRef] [PubMed]
- Pinent, M.; Blay, M.; Blade, M.C.; Salvado, M.J.; Arola, L.; Ardevol, A. Grape seed-derived procyanidins have an antihyperglycemic effect in streptozotocin-induced diabetic rats and insulinomimetic activity in insulin-sensitive cell lines. Endocrinology 2004, 145, 4985–4990. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Bao, L.; Ren, J.; Li, Y.; Zhang, Z. Grape seed procyanidin B2 protects podocytes from high glucose-induced mitochondrial dysfunction and apoptosis via the AMPK-SIRT1-PGC-1alpha axis in vitro. Food Funct. 2016, 7, 805–815. [Google Scholar] [CrossRef]
- Li, X.; Chen, D.; Wang, G.; Lu, Y. Study of interaction between human serum albumin and three antioxidants: Ascorbic acid, alpha-tocopherol, and proanthocyanidins. Eur. J. Med. Chem. 2013, 70, 22–36. [Google Scholar] [CrossRef]
- Ropiak, H.M.; Lachmann, P.; Ramsay, A.; Green, R.J.; Mueller-Harvey, I. Identification of Structural Features of Condensed Tannins That Affect Protein Aggregation. PLoS ONE 2017, 12, e0170768. [Google Scholar] [CrossRef]
- Wu, Q.; Li, S.; Fu, X.; Yang, T.; Chen, H.; Guan, Y.; Xie, B.; Sun, Z. Spectroscopic Studies on Binding of Lotus Seedpod Oligomeric Procyanidins to Bovine Serum Albumin. J. Appl. Spectrosc. 2014, 80, 884–892. [Google Scholar] [CrossRef]
- Crouse, H.F.; Potoma, J.; Nejrabi, F.; Snyder, D.L.; Chohan, B.S.; Basu, S. Quenching of tryptophan fluorescence in various proteins by a series of small nickel complexes. Dalton Trans. 2012, 41, 2720–2731. [Google Scholar] [CrossRef]
- Acharya, D.P.; Sanguansri, L.; Augustin, M.A. Binding of resveratrol with sodium caseinate in aqueous solutions. Food Chem. 2013, 141, 1050–1054. [Google Scholar] [CrossRef]
- Maier, G.P.; Bernt, C.M.; Butler, A. Catechol oxidation: Considerations in the design of wet adhesive materials. Biomater. Sci. 2018, 6, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Huang, C.R.; Jiang, M.; Zhu, Y.Y.; Wang, J.; Chen, J.; Shi, J.H. Binding interaction of atorvastatin with bovine serum albumin: Spectroscopic methods and molecular docking. Spectrochim. Acta Part A 2016, 156, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.F.; Zhou, K.L.; Lou, Y.Y.; Pan, D.Q.; Shi, J.H. Investigation of the binding interaction between estazolam and bovine serum albumin: Multi-spectroscopic methods and molecular docking technique. J. Biomol. Struct. Dyn. 2017, 35, 3605–3614. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Ueda-Wakagi, M.; Hayashibara, K.; Kitano, R.; Kawase, M.; Kaihatsu, K.; Kato, N.; Suhara, Y.; Osakabe, N.; Ashida, H. Substitution at the C-3 Position of Catechins Has an Influence on the Binding Affinities against Serum Albumin. Molecules 2017, 22, 314. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zou, W.; Zhong, Y.; An, J.; Zhang, X.; Wu, M.; Yu, Z. The hormesis effect of BDE-47 in HepG2 cells and the potential molecular mechanism. Toxicol. Lett. 2012, 209, 193–201. [Google Scholar] [CrossRef]
- Plauth, A.; Geikowski, A.; Cichon, S.; Wowro, S.J.; Liedgens, L.; Rousseau, M.; Weidner, C.; Fuhr, L.; Kliem, M.; Jenkins, G.; et al. Hormetic shifting of redox environment by pro-oxidative resveratrol protects cells against stress. Free Radical Biol. Med. 2016, 99, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.S. Binding of the volatile anesthetic chloroform to albumin demonstrated using tryptophan fluorescence quenching. J. Biol. Chem. 1997, 272, 17961–17965. [Google Scholar] [CrossRef]
- Sekula, B.; Zielinski, K.; Bujacz, A. Crystallographic studies of the complexes of bovine and equine serum albumin with 3,5-diiodosalicylic acid. Int. J. Biol. Macromol. 2013, 60, 316–324. [Google Scholar] [CrossRef]
- Corbeil, C.R.; Williams, C.I.; Labute, P. Variability in docking success rates due to dataset preparation. J. Comput. Aided Mol. Des. 2012, 26, 775–786. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, M.; Yang, L.; Huang, F.; Lan, Y.; Li, H.; Wu, H.; Zhang, B.; Shi, H.; Wu, X. P-glycoprotein Inhibitor Tariquidar Potentiates Efficacy of Astragaloside IV in Experimental Autoimmune Encephalomyelitis Mice. Molecules 2019, 24, 561. [Google Scholar] [CrossRef]
- Neri, S.; Mariani, E.; Meneghetti, A.; Cattini, L.; Facchini, A. Calcein-acetyoxymethyl cytotoxicity assay: Standardization of a method allowing additional analyses on recovered effector cells and supernatants. Clin. Diagn. Lab. Immunol. 2001, 8, 1131–1135. [Google Scholar] [CrossRef] [PubMed]
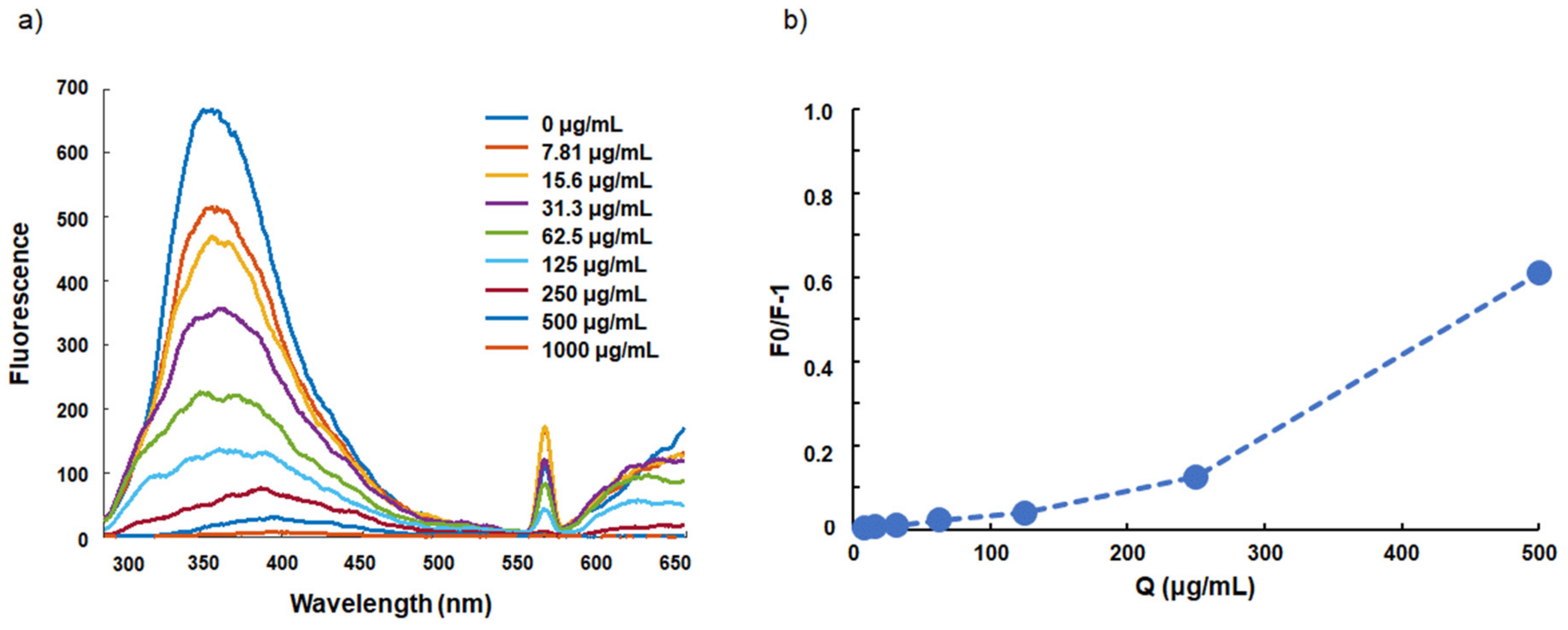
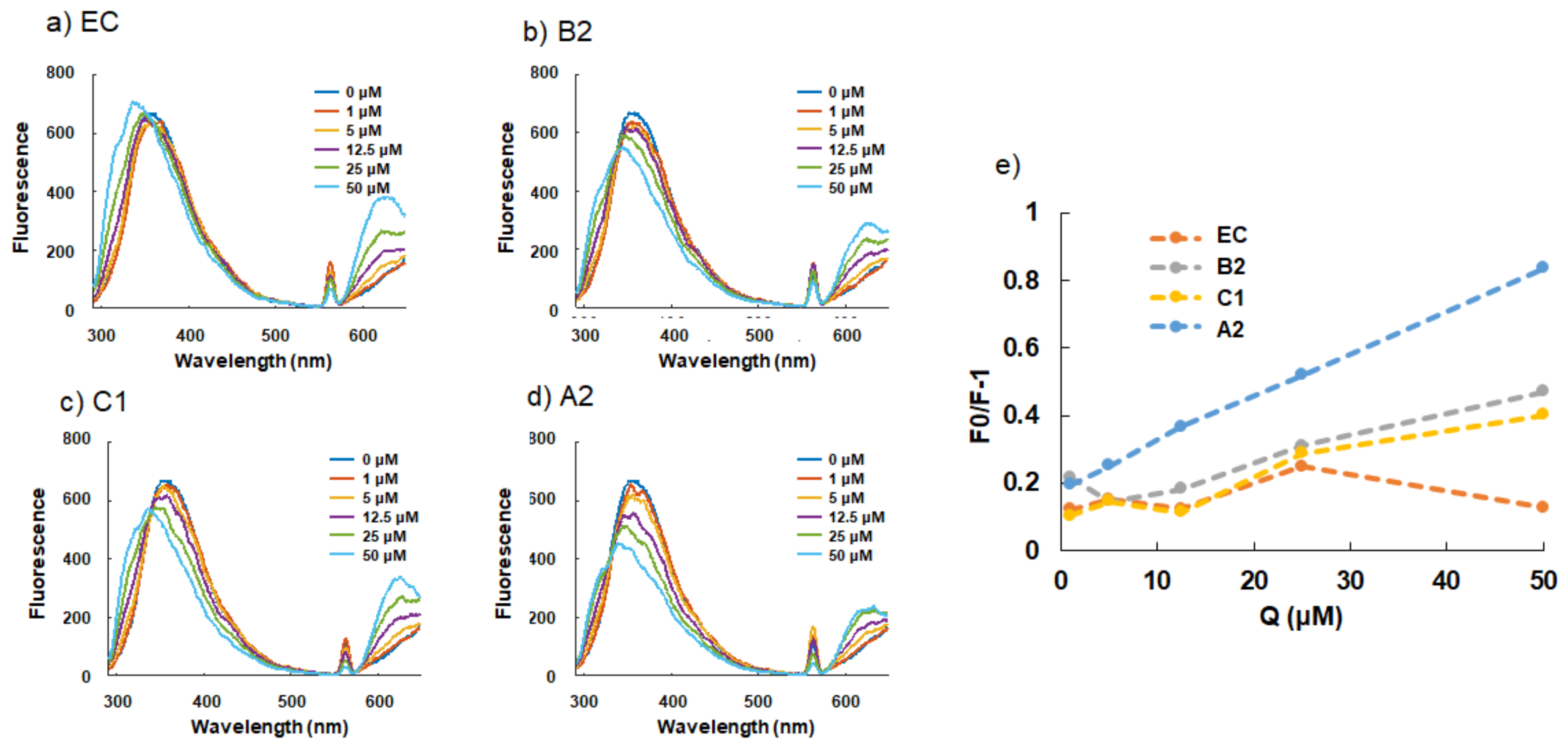
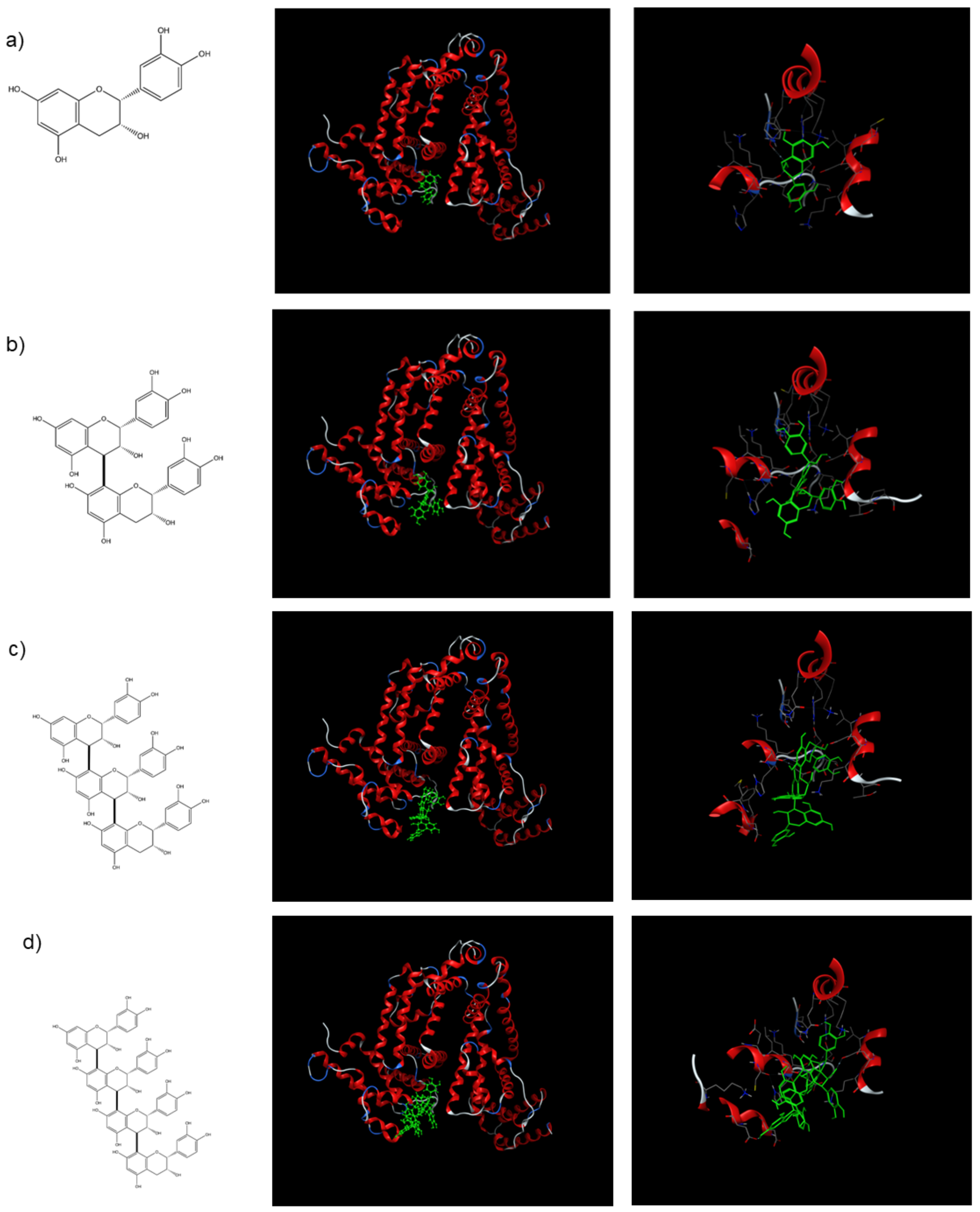
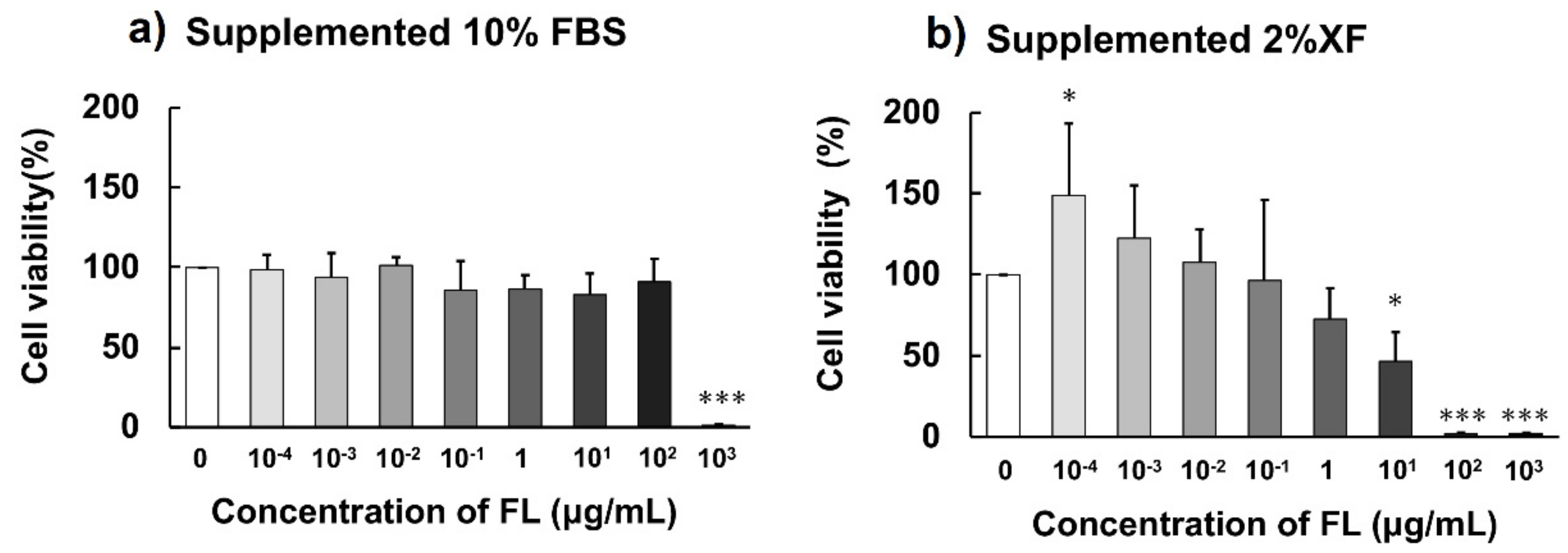

| Kq (×10−3, L·mol−1·s−1) | Coefficient of Determination (R2) | |
|---|---|---|
| (−)-epicatechin (EC) | 7.44 ± 2.13 1 | 0.0157 |
| procyanidin B2 (B2) | 16.35 ± 4.37 | 0.9201 |
| procyandin C1 (C1) | 14.33 ± 4.98 | 0.8957 |
| cinnamtannin A2 (A2) | 29.28 ± 7.47 | 0.9987 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujii, Y.; Suhara, Y.; Sukikara, Y.; Teshima, T.; Hirota, Y.; Yoshimura, K.; Osakabe, N. Elucidation of the Interaction between Flavan-3-ols and Bovine Serum Albumin and Its Effect on Their In-Vitro Cytotoxicity. Molecules 2019, 24, 3667. https://doi.org/10.3390/molecules24203667
Fujii Y, Suhara Y, Sukikara Y, Teshima T, Hirota Y, Yoshimura K, Osakabe N. Elucidation of the Interaction between Flavan-3-ols and Bovine Serum Albumin and Its Effect on Their In-Vitro Cytotoxicity. Molecules. 2019; 24(20):3667. https://doi.org/10.3390/molecules24203667
Chicago/Turabian StyleFujii, Yasuyuki, Yoshitomo Suhara, Yusuke Sukikara, Tomohiro Teshima, Yoshihisa Hirota, Kenjiro Yoshimura, and Naomi Osakabe. 2019. "Elucidation of the Interaction between Flavan-3-ols and Bovine Serum Albumin and Its Effect on Their In-Vitro Cytotoxicity" Molecules 24, no. 20: 3667. https://doi.org/10.3390/molecules24203667
APA StyleFujii, Y., Suhara, Y., Sukikara, Y., Teshima, T., Hirota, Y., Yoshimura, K., & Osakabe, N. (2019). Elucidation of the Interaction between Flavan-3-ols and Bovine Serum Albumin and Its Effect on Their In-Vitro Cytotoxicity. Molecules, 24(20), 3667. https://doi.org/10.3390/molecules24203667






