Design, Synthesis and Evaluation of New Indolylpyrimidylpiperazines for Gastrointestinal Cancer Therapy
Abstract
1. Introduction
2. Results and Discussion
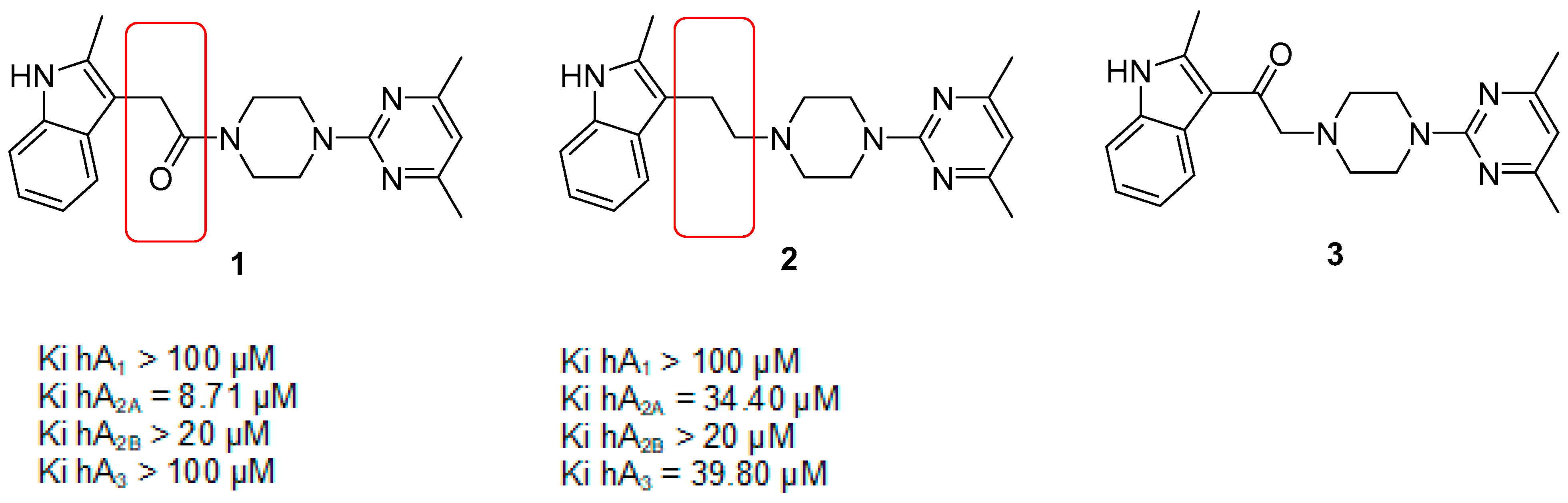
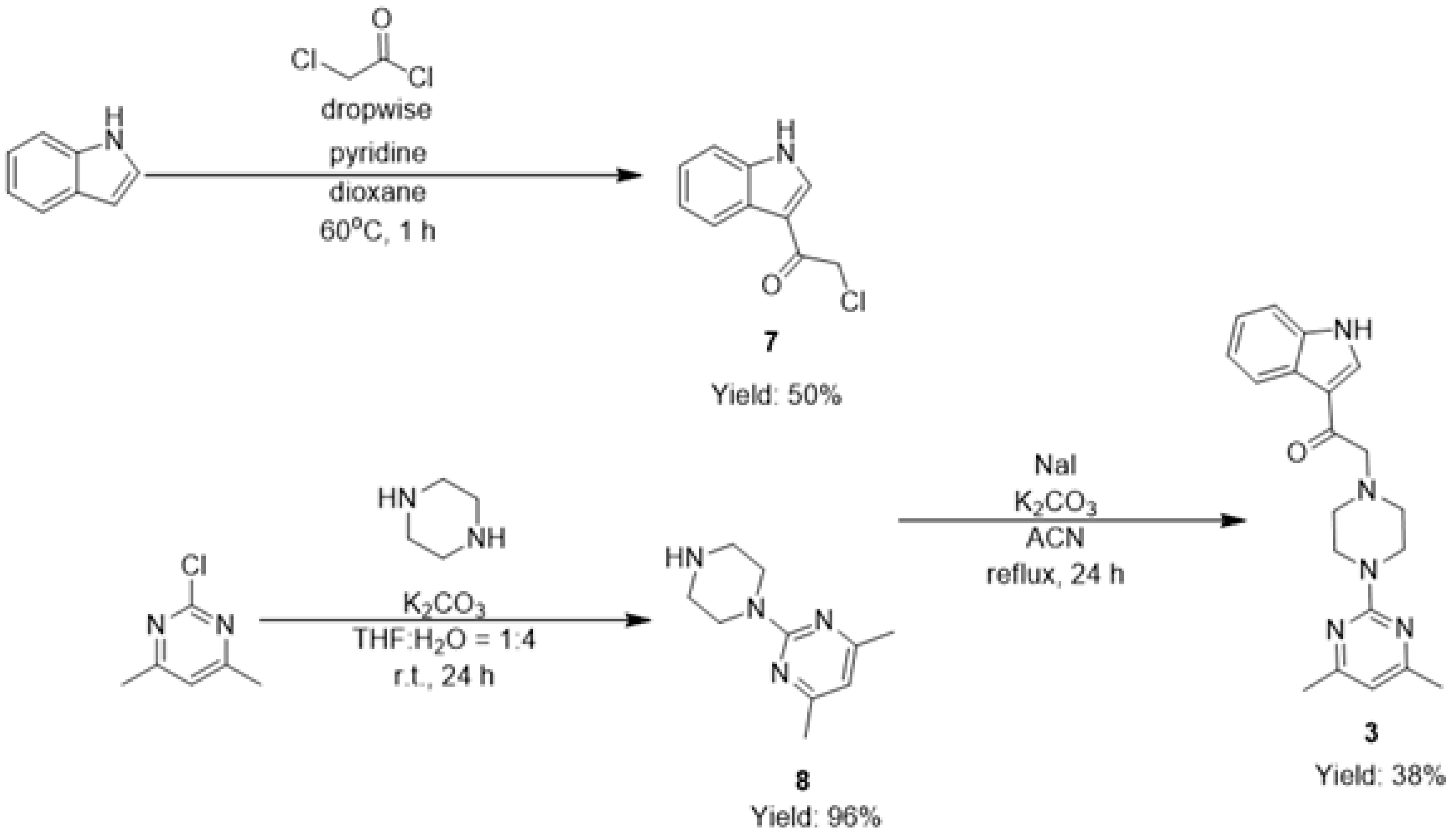
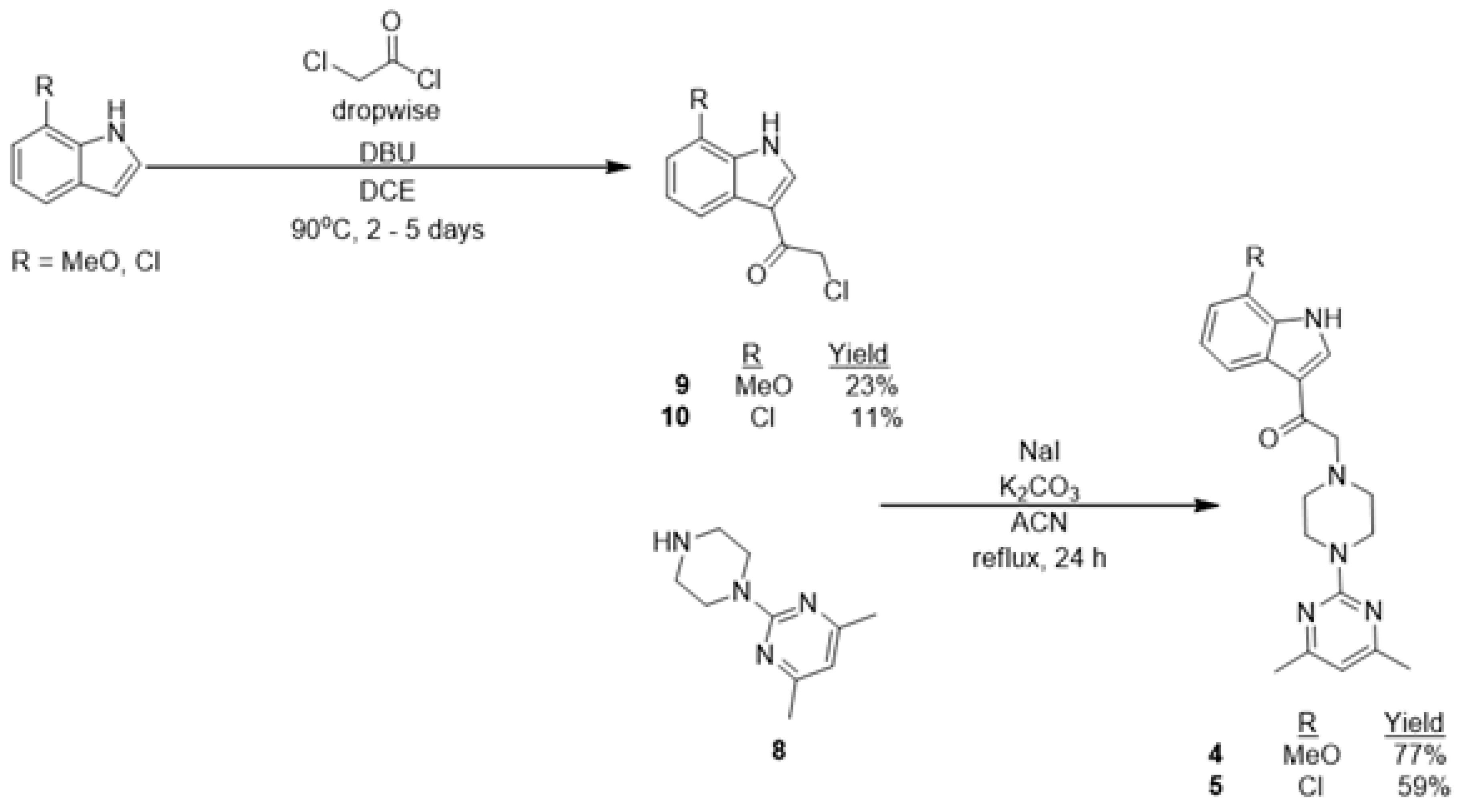
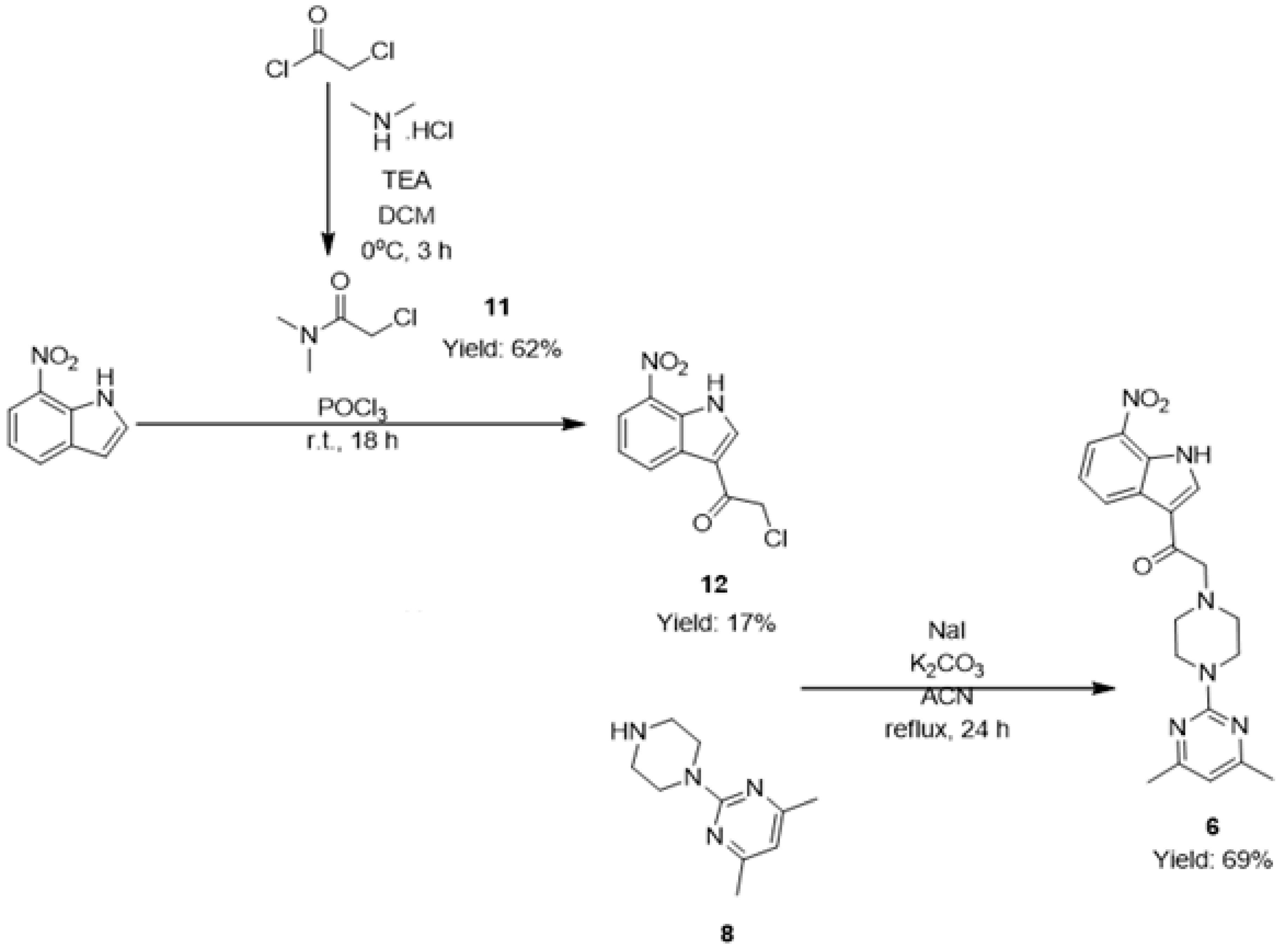
2.1. Chemistry
2.2. Biological Evaluation
2.2.1. Functional Activity Studies at hA2AAR and hA3AR
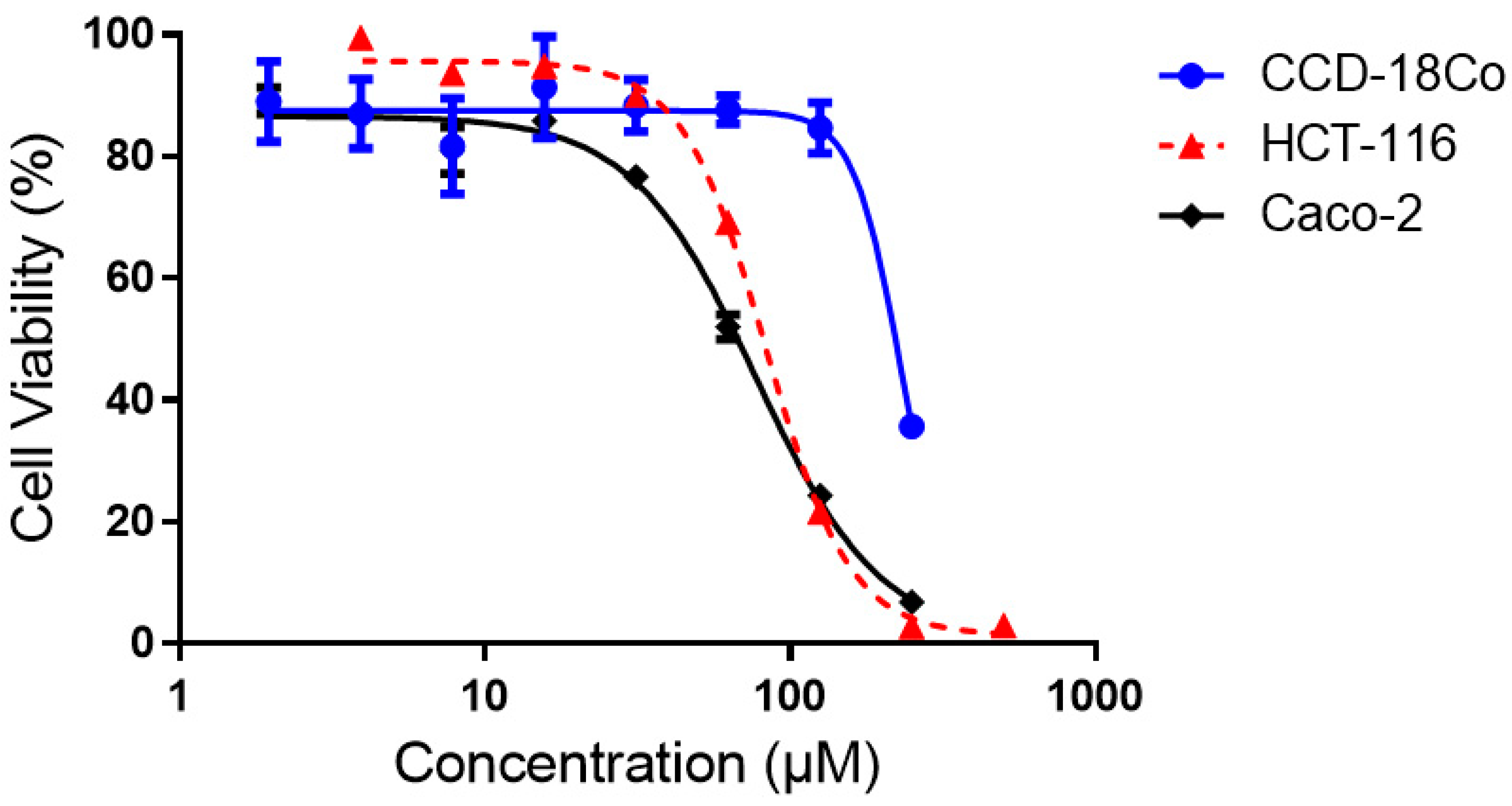
2.2.2. Cytotoxicity Assays of Compound 4 against Human Colorectal Cancer Cell Lines, a Normal Colon Cell Line and a Liver Cancer Cell Line.
2.3. UV-Vis Stability Study of Compound 4
2.4. Induced-Fit Docking of Compound 1 and Compounds 3–6 in hA3AR Homology Model
3. Materials and Methods
3.1. Chemistry
3.2. Biological Evaluation
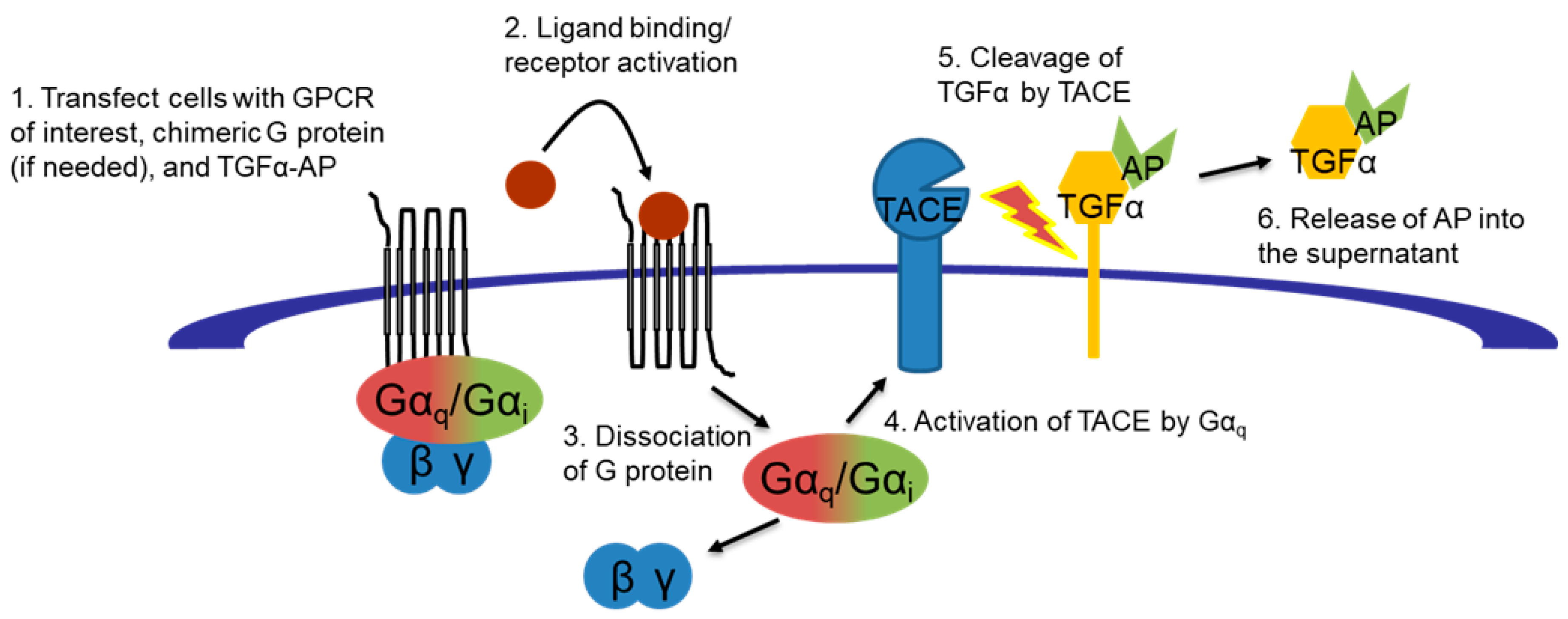
3.2.1. TGFα Shedding Assays for hA2AAR and hA3AR
3.2.2. Cytotoxicity Assays
3.3. UV-Vis Stability Assay
3.4. Molecular Modeling
3.4.1. Homology Modelling and Protein Preparation
3.4.2. Ligand Docking Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Stewart, B.W.; Wild, C.P. World Cancer Report 2014; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Valery, P.C.; Laversanne, M.; Clark, P.J.; Petrick, J.L.; McGlynn, K.A.; Bray, F. Projections of primary liver cancer to 2030 in 30 countries worldwide. Hepatology 2018, 67, 600–611. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef]
- Sakowicz-Burkiewicz, M.; Kitowska, A.; Grden, M.; Maciejewska, I.; Szutowicz, A.; Pawelczyk, T. Differential effect of adenosine receptors on growth of human colon cancer HCT 116 and HT-29 cell lines. Arch. Biochem. Biophys. 2013, 533, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Stemmer, S.M.; Shani, A.; Klein, B.; Silverman, M.H.; Lorber, I.; Farbstein, M.; Shmueli, E.; Figer, A. A phase II, multi-center study of a new non-cytotoxic A3 adenosine receptor agonist CF101, dose-finding (randomized blinded) in patients (pts)with refractory metastatic colorectal cancer. J. Clin. Oncol. 2004, 22 (Suppl. 14), 3153. [Google Scholar] [CrossRef]
- Bar-Yehuda, S.; Madi, L.; Silberman, D.; Gery, S.; Shkapenuk, M.; Fishman, P. CF101, An Agonist to the A3 Adenosine Receptor, Enhances the Chemotherapeutic Effect of 5-Fluorouracil in a Colon Carcinoma Murine Model. Neoplasia 2005, 7, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Borea, P.A.; Varani, K.; Vincenzi, F.; Baraldi, P.G.; Tabrizi, M.A.; Merighi, S.; Gessi, S. The A3 Adenosine Receptor: History and Perspectives. Pharmacol. Rev. 2015, 67, 74–102. [Google Scholar] [CrossRef] [PubMed]
- Gessi, S.; Cattabriga, E.; Avitabile, A.; Gafa, R.; Lanza, G.; Cavazzini, L.; Bianchi, N.; Gambari, R.; Feo, C.; Liboni, A.; et al. Elevated Expression of A3 Adenosine Receptors in Human Colorectal Cancer Is Reflected in Peripheral Blood Cells. Clin. Cancer Res. 2004, 10, 5895–5901. [Google Scholar] [CrossRef]
- Madi, L.; Ochaion, A.; Rath-Wolfson, L.; Bar-Yehuda, S.; Erlanger, A.; Ohana, G.; Harish, A.; Merimski, O.; Barer, F.; Fishman, P. The A3 Adenosine Receptor Is Highly Expressed in Tumor versus Normal Cells. Potential Target for Tumor Growth Inhibition. Clin. Cancer Res. 2004, 10, 4472–4479. [Google Scholar] [CrossRef]
- Panjehpour, M.; Hemati, S.; Forghani, M.A. Expression of A1 and A3 Adenosine Receptors in Human Breast Tumors. Tumori J. 2012, 98, 137–141. [Google Scholar] [CrossRef]
- Trincavelli, M.L.; Tuscano, D.; Marroni, M.; Falleni, A.; Gremigni, V.; Ceruti, S.; Abbracchio, M.P.; Jacobson, K.A.; Cattabeni, F.; Martini, C. A3 Adenosine Receptors in Human Astrocytoma Cells: Agonist-Mediated Desensitization, Internalization, and Down-Regulation. Mol. Pharmacol. 2002, 62, 1373–1384. [Google Scholar] [CrossRef] [PubMed]
- Kohno, Y.; Sei, Y.; Koshiba, M.; Kim, H.O.; Jacobson, K.A. Induction of Apoptosis in HL-60 Human Promyelocytic Leukemia Cells by Adenosine A3Receptor Agonists. Biochem. Biophys. Res. Commun. 1996, 219, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.; Zucman-Rossi, J.; Pikarsky, E. Hepatocellular carcinoma [serial online]. Nat. Rev. Dis. Primers 2016, 2, 16018. [Google Scholar] [CrossRef] [PubMed]
- Stemmer, S.M.; Benjaminov, O.; Medalia, G.; Ciuraru, N.B.; Silverman, M.H.; Bar-Yehuda, S.; Fishman, S.; Harpaz, Z.; Farbstein, M.; Cohen, S.; et al. CF102 for the Treatment of Hepatocellular Carcinoma: A Phase I/II, Open-Label, Dose-Escalation Study. Oncologist 2013, 18, 25–26. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.-M.; Ma, X.; Paira, P.; Tan, A.; Herr, D.R.; Lim, K.L.; Ng, C.H.; Venkatesan, G.; Klotz, K.N.; Federico, S.; et al. Discovery of indolylpiperazinylpyrimidines with dual-target profiles at adenosine A2A and dopamine D2 receptors for Parkinson’s disease treatment. PLoS ONE 2018, 13, e0188212. [Google Scholar] [CrossRef]
- Thompson, M.J.; Louth, J.C.; Ferrara, S.; Sorrell, F.J.; Irving, B.J.; Cochrane, E.J.; Meijer, A.J.; Chen, B. Structure–Activity Relationship Refinement and Further Assessment of Indole-3-glyoxylamides as a Lead Series against Prion Disease. ChemMedChem 2011, 6, 115–130. [Google Scholar] [CrossRef]
- Johansson, H.; Urruticoechea, A.; Larsen, I.; Sejer Pedersen, D. A Scalable Method for Regioselective 3-Acylation of 2-Substituted Indoles under Basic Conditions. J. Org. Chem. 2015, 80, 471–481. [Google Scholar] [CrossRef]
- Gitto, R.; De Luca, L.; Ferro, S.; Russo, E.; De Sarro, G.; Chisari, M.; Ciranna, L.; Alvarez-Builla, J.; Alajarin, R.; Buemi, M.R.; et al. Synthesis, modelling and biological characterization of 3-substituted-1H-indoles as ligands of GluN2B-containing N-methyl-d-aspartate receptors. Bioorg. Med. Chem. 2014, 22, 1040–1048. [Google Scholar] [CrossRef]
- Wang, B.-L.; Shi, Y.-X.; Zhang, S.-J.; Ma, Y.; Wang, H.-X.; Zhang, L.-Y.; Wei, W.; Liu, X.H.; Li, Y.H.; Li, Z.M.; et al. Syntheses, biological activities and SAR studies of novel carboxamide compounds containing piperazine and arylsulfonyl moieties. Eur. J. Med. Chem. 2016, 117, 167–178. [Google Scholar] [CrossRef]
- Gilbert, E.J.; Greenlee, W.J.; Miller, M.W.; Scott, J.D.; Stamford, A.W.; Inventors; USA. Assignee. Substituted Piperazines as CB1 Antagonists and Their Preparation. U.S. Patent US20130072468A1, 21 March 2013. [Google Scholar]
- Inoue, A.; Ishiguro, J.; Kitamura, H.; Arima, N.; Okutani, M.; Shuto, A.; Higashiyama, S.; Ohwada, T.; Arai, H.; Makide, K.; et al. TGFα shedding assay: An accurate and versatile method for detecting GPCR activation. Nat. Methods 2012, 9, 1021. [Google Scholar] [CrossRef]
- Park, K.-S.; Hoffmann, C.; Kim, H.O.; Padgett, W.L.; Daly, J.W.; Brambilla, R.; Motta, C.; Abbracchio, M.P.; Jacobson, K.A. Activation and desensitization of rat A3-adenosine receptors by selective adenosine derivatives and xanthine-7-ribosides. Drug Dev. Res. 1998, 44, 97–105. [Google Scholar] [CrossRef]
- Storme, J.; Cannaert, A.; Van Craenenbroeck, K.; Stove, C.P. Molecular dissection of the human A3 adenosine receptor coupling with β-arrestin2. Biochem. Pharmacol. 2018, 148, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Bar-Yehuda, S.; Stemmer, S.M.; Madi, L.; Castel, D.; Ochaion, A.; Cohen, S.; Barer, F.; Zabutti, A.; Perez-Liz, G.; Del Valle, L.; et al. The A3 adenosine receptor agonist CF102 induces apoptosis of hepatocellular carcinoma via de-regulation of the Wnt and NF-κB signal transduction pathways. Int. J. Oncol. 2008, 33, 287–295. [Google Scholar] [PubMed]
- Gao, Z.-G.; Jacobson, K.A. Translocation of arrestin induced by human A3 adenosine receptor ligands in an engineered cell line: Comparison with G protein-dependent pathways. Pharmacol. Res. 2008, 57, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Fishman, P.; Bar-Yehuda, S.; Ardon, E.; Rath-Wolfson, L.; Barrer, F.; Ochaion, A.; Madi, L. Targeting the A3 adenosine receptor for cancer therapy: Inhibition of prostate carcinoma cell growth by A3AR agonist. Anticancer Res. 2003, 23, 2077–2084. [Google Scholar] [PubMed]
- Kim, S.-K.; Riley, L.; Abrol, R.; Jacobson, K.A.; Goddard, W.A., III. Predicted structures of agonist and antagonist bound complexes of adenosine A3 receptor. Proteins 2011, 79, 1878–1897. [Google Scholar] [CrossRef] [PubMed]
- Ciancetta, A.; Jacobson, K.A. Structural Probing and Molecular Modeling of the A3 Adenosine Receptor: A Focus on Agonist Binding. Molecules 2017, 22, 449. [Google Scholar] [CrossRef]
- Dal Ben, D.; Buccioni, M.; Lambertucci, C.; Marucci, G.; Thomas, A.; Volpini, R.; Cristalli, G. Molecular modeling study on potent and selective adenosine A3 receptor agonists. Bioorg. Med. Chem. 2010, 18, 7923–7930. [Google Scholar] [CrossRef]
- Tosh, D.K.; Ciancetta, A.; Warnick, E.; O’Connor, R.; Chen, Z.; Gizewski, E.; Crane, S.; Gao, Z.G.; Auchampach, J.A.; Salvemini, D.; et al. Purine (N)-Methanocarba Nucleoside Derivatives Lacking an Exocyclic Amine as Selective A3 Adenosine Receptor Agonists. J. Med. Chem. 2016, 59, 3249–3263. [Google Scholar] [CrossRef]
- Gao, Z.-G.; Kim, S.-K.; Biadatti, T.; Chen, W.; Lee, K.; Barak, D.; Kim, S.G.; Johnson, C.R.; Jacobson, K.A. Structural Determinants of A3 Adenosine Receptor Activation: Nucleoside Ligands at the Agonist/Antagonist Boundary. J. Med. Chem. 2002, 45, 4471–4484. [Google Scholar] [CrossRef]
- Gao, Z.-G.; Chen, A.; Barak, D.; Kim, S.-K.; Müller, C.E.; Jacobson, K.A. Identification by Site-directed Mutagenesis of Residues Involved in Ligand Recognition and Activation of the Human A3 Adenosine Receptor. J. Biol. Chem. 2002, 277, 19056–19063. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, K.A.; Kim, H.S.; Ravi, G.; Kim, S.-K.; Lee, K.; Chen, A.; Chen, W.; Kim, S.; Barak, D.; Liang, B.T. Engineering of A3 adenosine and P2Y nucleotide receptors and their ligands. Drug Dev. Res. 2003, 58, 330–339. [Google Scholar] [CrossRef]
- Pandy-Szekeres, G.; Munk, C.; Tsonkov, T.M.; Mordalski, S.; Harpsoe, K.; Hauser, A.S.; Bojarski, A.J.; Gloriam, D.E. GPCRdb in 2018: Adding GPCR structure models and ligands. Nucleic Acids Res. 2018, 46, D440–D446. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, M.P.; Pincus, D.L.; Rapp, C.S.; Day, T.J.F.; Honig, B.; Shaw, D.E.; Friesner, R.A. A hierarchical approach to all-atom protein loop prediction. Proteins 2004, 55, 351–367. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhao, L.X.; Park, J.; Lee, H.W.; Sahu, P.K.; Cui, M.; Moss, S.M.; Hammes, E.; Warnick, E.; Gao, Z.G.; et al. N6-Substituted 5′-N-Methylcarbamoyl-4′-selenoadenosines as Potent and Selective A3 Adenosine Receptor Agonists with Unusual Sugar Puckering and Nucleobase Orientation. J. Med. Chem. 2017, 60, 3422–3437. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, R.A.; MacArthur, M.W.; Moss, D.S.; Thornton, J.M. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 1993, 26, 283–291. [Google Scholar] [CrossRef]
- Schrödinger Release 2018-3: LigPrep; Schrödinger, LLC: New York, NY, USA, 2018.
- Schrödinger Release 2018-3: Epik; Schrödinger, LLC: New York, NY, USA, 2018.
- Schrödinger Release 2018-3: Schrödinger Suite 2018-3 Induced Fit Docking Protocol; Glide, Schrödinger, LLC: New York, NY, USA, 2016; Prime, Schrödinger, LLC: New York, NY, USA, 2018.
Sample Availability: Not available. |

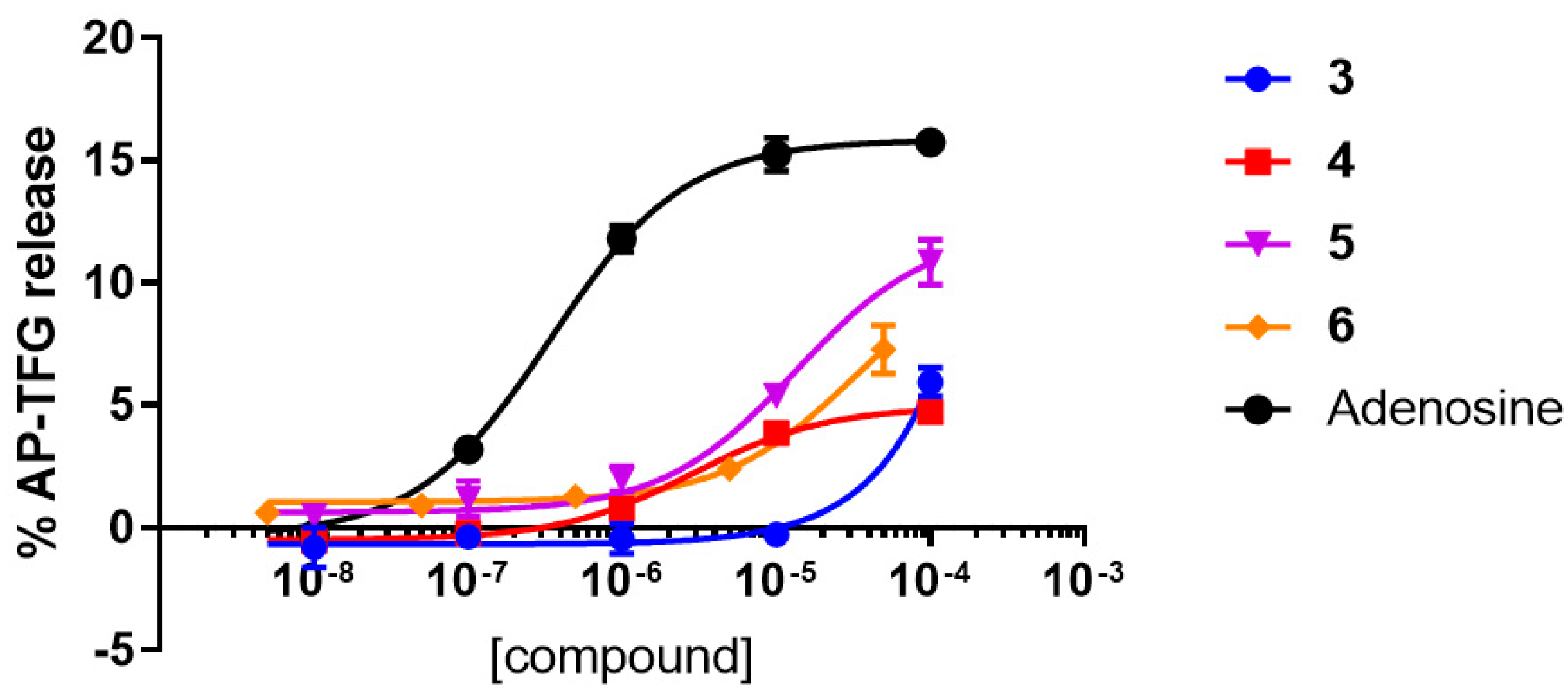
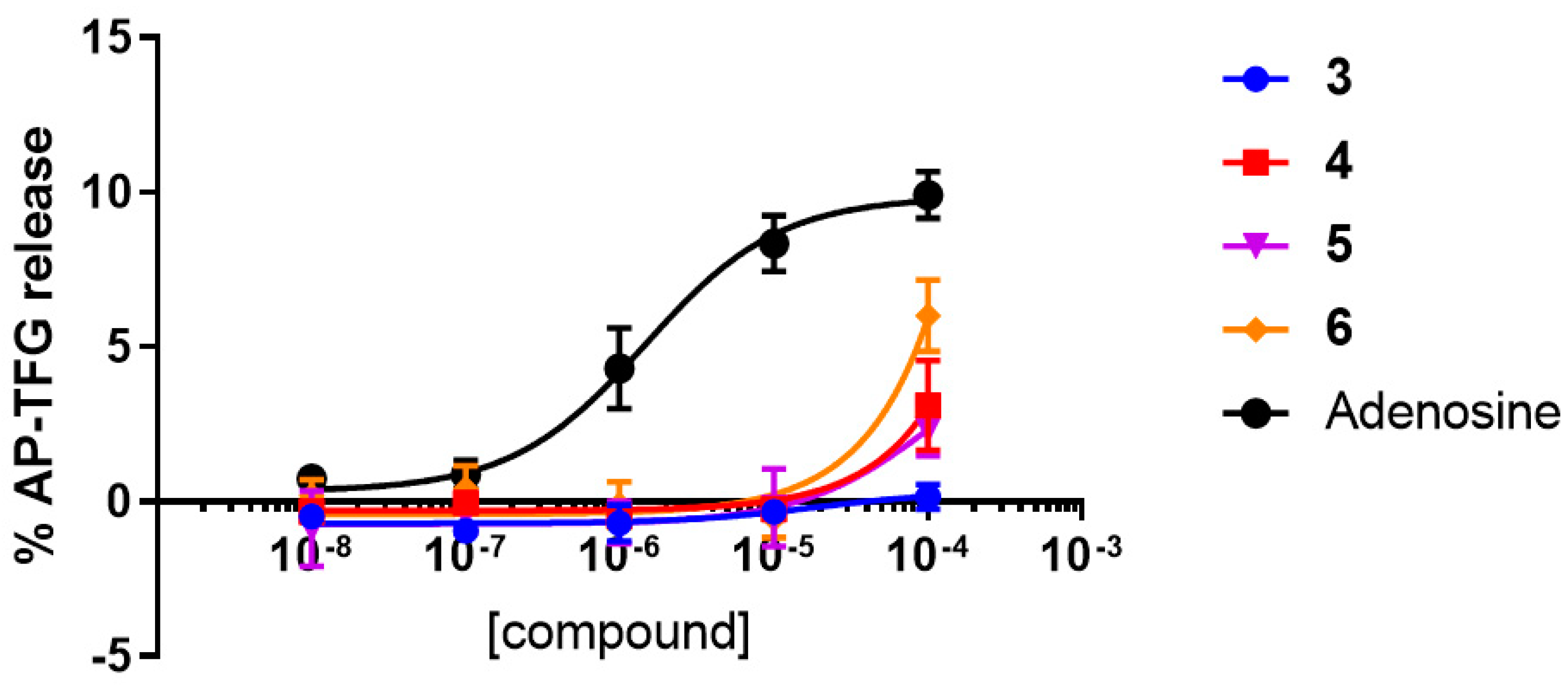
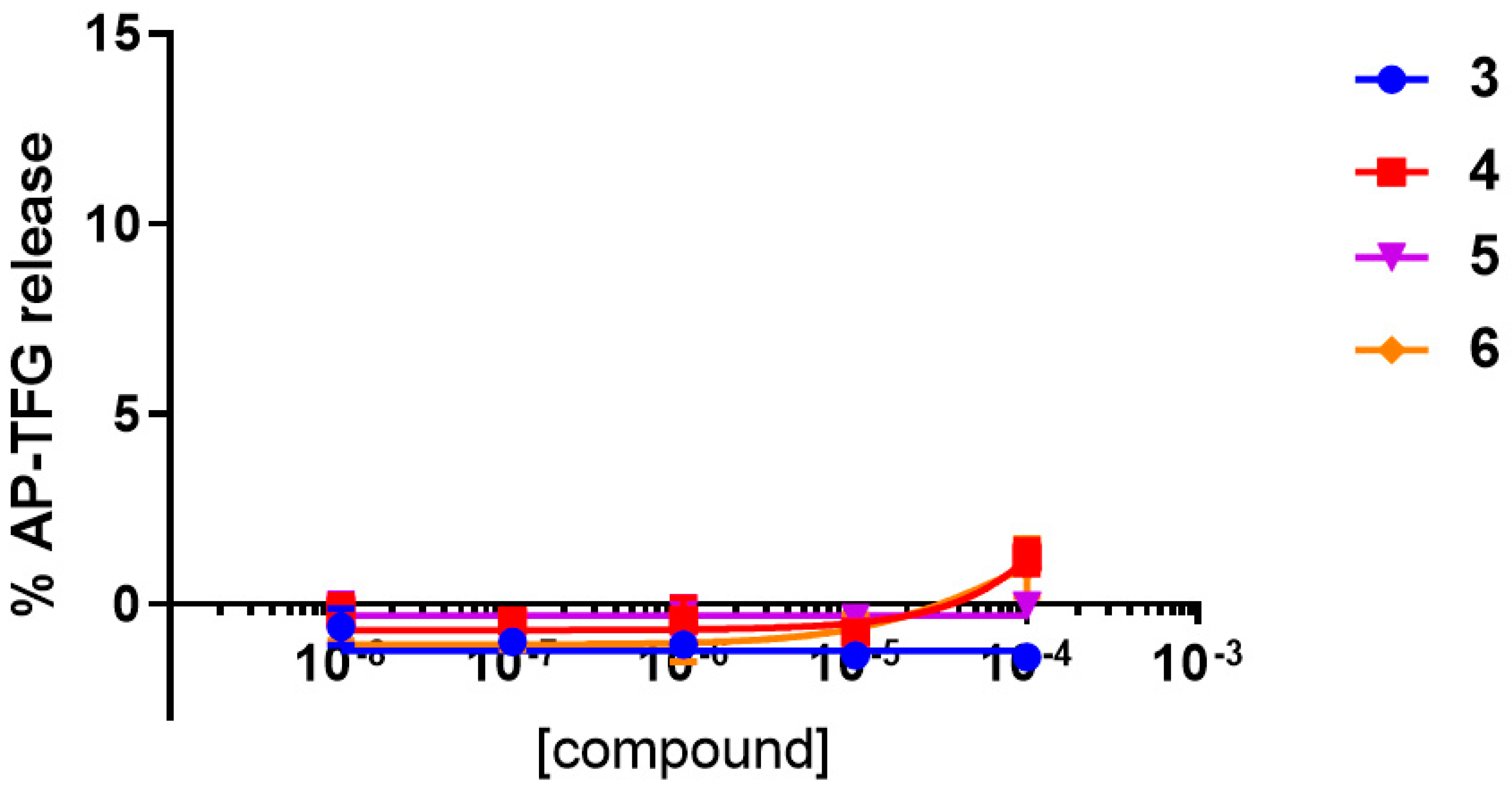


| Compound | EC50 (μM) | Emax (%) |
|---|---|---|
| 3 | Inactive | N.A. |
| 4 | 2.89 ± 0.55 | 31 |
| 5 | 13.4 ± 2.96 | 77 |
| 6 | 33.2 ± 16.50 | 72 |
| Adenosine | 0.35 ± 0.03 | 100 |
| IC50 (μM) | ||
|---|---|---|
| 4 | 2-Cl-IB-MECA | |
| HCT-116 | 84 ± 9 | 21 ± 6 |
| Caco-2 | 77 ± 10 | 31 ± 4 |
| HepG2 | 30 ± 7 | 13 ± 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, A.; Babak, M.V.; Venkatesan, G.; Lim, C.; Klotz, K.-N.; Herr, D.R.; Cheong, S.L.; Federico, S.; Spalluto, G.; Ong, W.-Y.; et al. Design, Synthesis and Evaluation of New Indolylpyrimidylpiperazines for Gastrointestinal Cancer Therapy. Molecules 2019, 24, 3661. https://doi.org/10.3390/molecules24203661
Tan A, Babak MV, Venkatesan G, Lim C, Klotz K-N, Herr DR, Cheong SL, Federico S, Spalluto G, Ong W-Y, et al. Design, Synthesis and Evaluation of New Indolylpyrimidylpiperazines for Gastrointestinal Cancer Therapy. Molecules. 2019; 24(20):3661. https://doi.org/10.3390/molecules24203661
Chicago/Turabian StyleTan, Aaron, Maria V. Babak, Gopalakrishnan Venkatesan, Clarissa Lim, Karl-Norbert Klotz, Deron Raymond Herr, Siew Lee Cheong, Stephanie Federico, Giampiero Spalluto, Wei-Yi Ong, and et al. 2019. "Design, Synthesis and Evaluation of New Indolylpyrimidylpiperazines for Gastrointestinal Cancer Therapy" Molecules 24, no. 20: 3661. https://doi.org/10.3390/molecules24203661
APA StyleTan, A., Babak, M. V., Venkatesan, G., Lim, C., Klotz, K.-N., Herr, D. R., Cheong, S. L., Federico, S., Spalluto, G., Ong, W.-Y., Chen, Y. Z., Loo, J. S. E., & Pastorin, G. (2019). Design, Synthesis and Evaluation of New Indolylpyrimidylpiperazines for Gastrointestinal Cancer Therapy. Molecules, 24(20), 3661. https://doi.org/10.3390/molecules24203661








