UV Photoprotection, Cytotoxicity and Immunology Capacity of Red Algae Extracts
Abstract
1. Introduction
2. Results
2.1. Mycosporine-Like Amino Acids (MAAs)
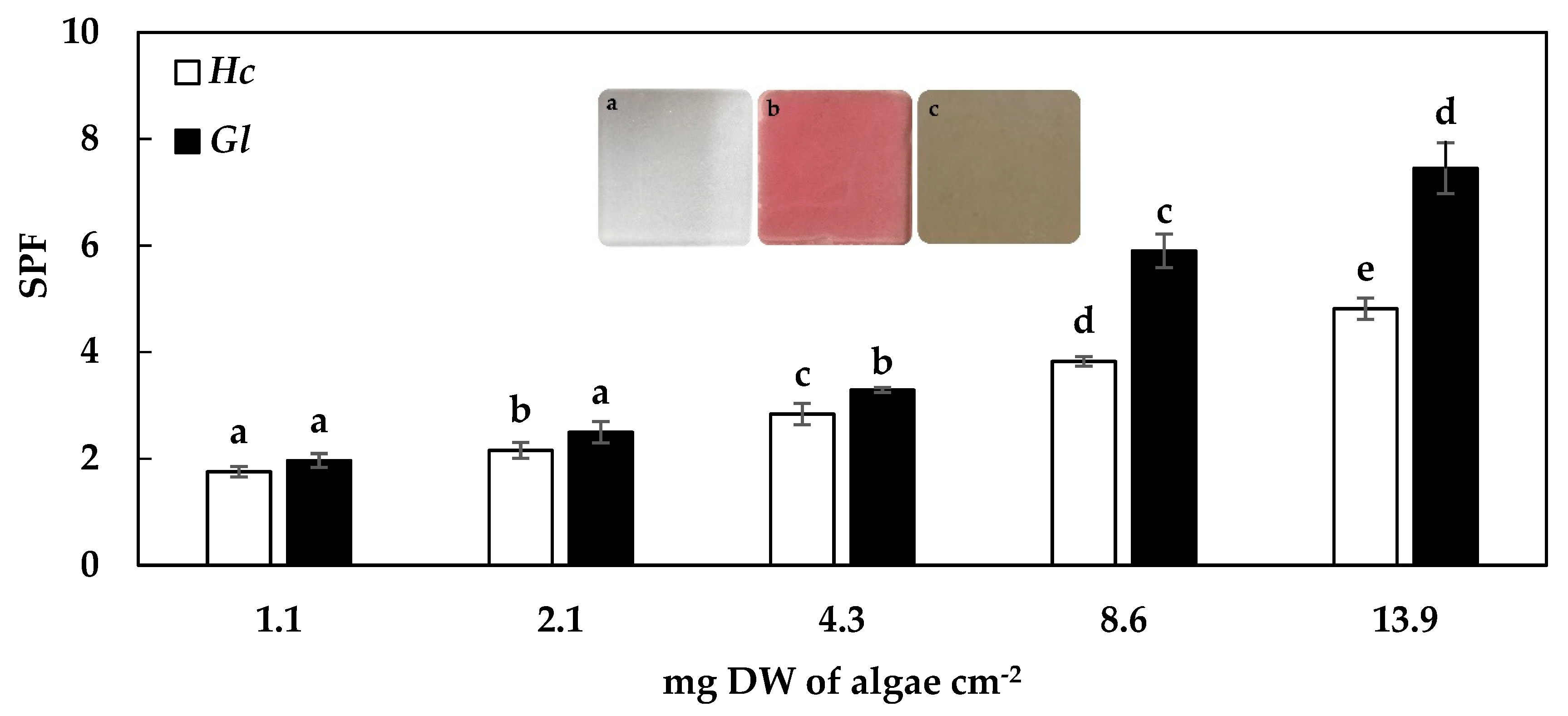
2.2. Sun Protection Factor (SPF)
2.3. Cytotoxicity Assays
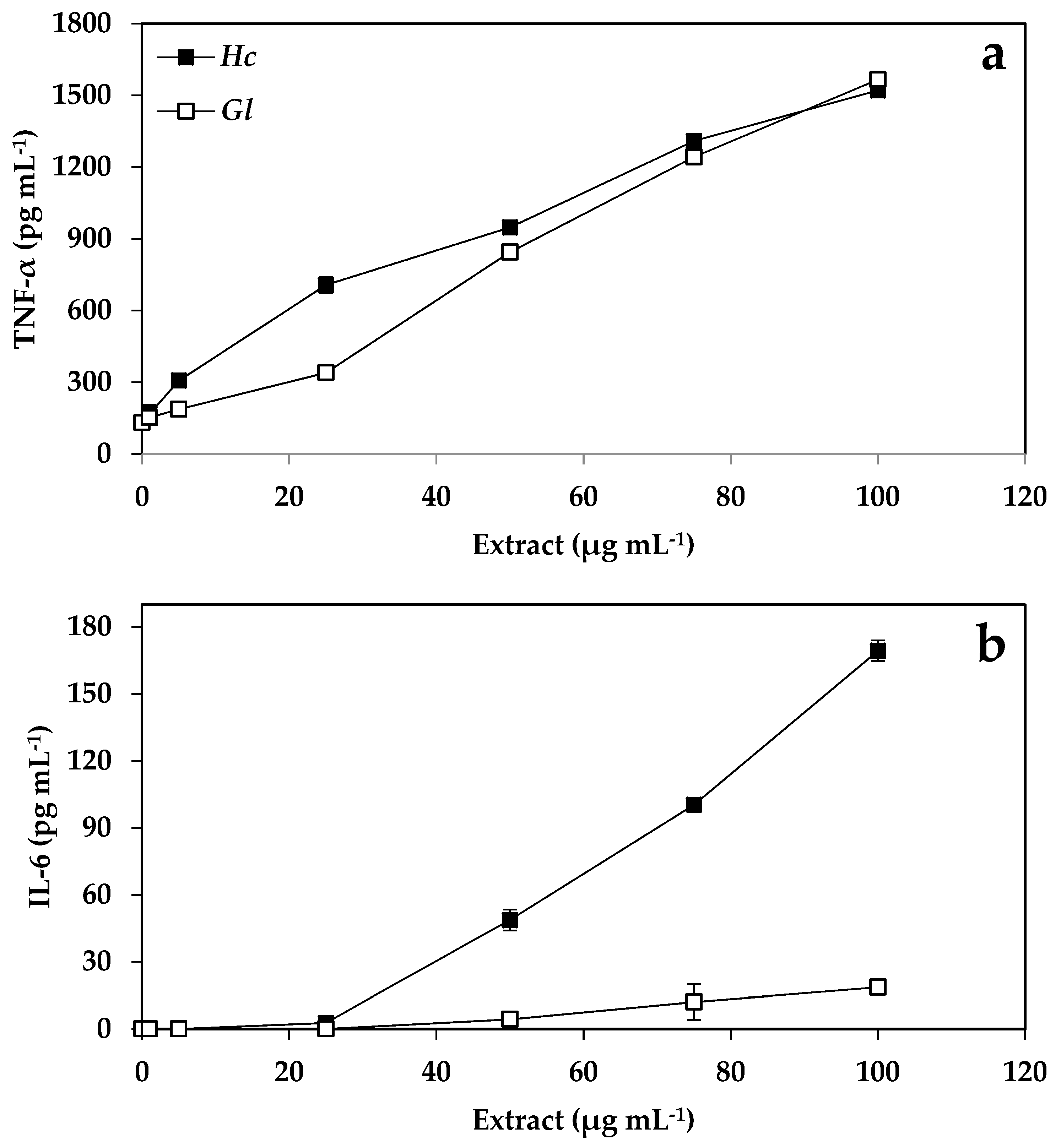
2.4. Immunology Results
3. Discussion
3.1. Mycosporine-Like Amino Acids (MAAs)
3.2. Sun Protection Factor (SPF)
3.3. Cell Viability
3.4. Cytokine Production
4. Materials and Methods
4.1. Mycosporine-Like Amino Acid Analyses
4.1.1. High Pressure Liquid Chromatography-Photodiode Array Detector (HPLC-PDA)
4.1.2. Electrospray Ionization-Mass Spectrometry (ESI-MS)
4.2. Evaluation of the Sun Protection Factor (SPF)
4.3. Cytotoxicity Assays
4.4. Immunological Assays
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Isnansetyo, A.; Lutfia, F.N.L.; Nursid, M.; Susidarti, R.A. Cytotoxicity of Fucoidan from Three Tropical Brown Algae Against Breast and Colon Cancer Cell Lines. Pharmacogn. J. 2017, 9. [Google Scholar] [CrossRef]
- Tannoury, M.Y.; Saab, A.M.; Elia, J.M.; Harb, N.N.; Makhlouf, H.Y.; Diab-Assaf, M. In Vitro Cytotoxic Activity of Laurencia papillosa, Marine Red Algae from the Lebanese Coast. J. Appl. Pharm. Sci. Vol. 2017, 7, 175–179. [Google Scholar]
- Khalifa, K.S.; Hamouda, R.A.; Hanafy, D.; Hamza, A. In vitro antitumor activity of silver nanoparticles biosynthesized by marine algae. Dig. J. Nanomater. Biostructures 2016, 11, 213–221. [Google Scholar]
- Anastyuk, S.D.; Shevchenko, N.M.; Usoltseva, R.V.; Silchenko, A.S.; Zadorozhny, P.A.; Dmitrenok, P.S.; Ermakova, S.P. Structural features and anticancer activity in vitro of fucoidan derivatives from brown alga Saccharina cichorioides. Carbohydr. Polym. 2017, 157, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Irhimeh, M.R. Melanoma and brown seaweed: An integrative hypothesis. J. Appl. Phycol. 2016, 29, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Hewage, S.R.K.M.; Piao, M.J.; Kang, K.A.; Han, X.; Kang, H.K.; Yoo, E.S.; Koh, Y.S.; Lee, N.H.; Ko, C.S. Photoprotective effect of Carpomitra costata extract against ultraviolet B-induced oxidative damage in human keratinocytes. J. Environ. Pathol. Toxicol. Oncol. 2016, 35. [Google Scholar] [CrossRef] [PubMed]
- Abdala-Díaz, R.T.; Chabrillón, M.; Cabello-Pasini, A.; López-Soler, B.; Figueroa, F.L. Effect of Porphyridium cruentum polysaccharides on the activity of murine macrophage cell line RAW264.7. Ciencias Mar. 2010, 36, 345–353. [Google Scholar] [CrossRef]
- Abdala, R.T.; Chabrillón, M.; Cabello-Pasini, A.; Gómez-Pinchetti, J.L.; Figueroa, F.L. Characterization of polysaccharides from Hypnea spinella (Gigartinales) and Halopithys incurva (Ceramiales) and their effect on RAW264.7 macrophage activity. J. Appl. Phycol. 2011, 23, 523–528. [Google Scholar]
- Romano, G.; Costantini, M.; Sansone, C.; Lauritano, C.; Ruocco, N.; Ianora, A. Marine microorganisms as a promising and sustainable source of bioactive molecules. Mar. Environ. Res. 2017, 128, 58–69. [Google Scholar] [CrossRef]
- Xu, N.; Fan, X.; Yan, X.; Tseng, C.K. Screening marine algae from China for their antitumor activities. J. Appl. Phycol. 2004, 16, 451–456. [Google Scholar] [CrossRef]
- Choi, S.-Y.; Park, M.-K.; Choi, J.; Koh, E.-J.; Seo, Y.-J.; Song, J.-H.; Chei, S.; Hwang, J.-H.; Lee, Y.-J.; Lee, B.-Y. The synergistic anti-obesity effect of Gelidium elegans extract and orlistat in vivo and in vitro. FASEB J. 2017, 31, 610–646. [Google Scholar]
- Poli, G.; Kinter, A.; Justement, J.S.; Kehrl, J.H.; Bressler, P.; Stanley, S.; Fauci, A.S. Tumor necrosis factor alpha functions in an autocrine manner in the induction of human immunodeficiency virus expression. Proc. Natl. Acad. Sci. USA 1990, 87, 782–785. [Google Scholar] [CrossRef] [PubMed]
- Scheller, J.; Chalaris, A.; Schmidt-Arras, D.; Rose-John, S. The pro-and anti-inflammatory properties of the cytokine interleukin-6. Biochim. Biophys. Acta (BBA)-Molecular Cell Res. 2011, 1813, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Martinez, C.; Delgado, M.; Pozo, D.; Leceta, J.; Calvo, J.R.; Ganea, D.; Gomariz, R.P. Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide modulate endotoxin-induced IL-6 production by murine peritoneal macrophages. J. Leukoc. Biol. 1998, 63, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Rudtanatip, T.; Lynch, S.A.; Wongprasert, K.; Culloty, S.C. Assessment of the effects of sulfated polysaccharides extracted from the red seaweed Irish moss Chondrus crispus on the immune-stimulant activity in mussels Mytilus spp. Fish Shellfish Immunol. 2018, 75, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Tabarsa, M.; You, S.; Dabaghian, E.H.; Surayot, U. Water-soluble polysaccharides from Ulva intestinalis: Molecular properties, structural elucidation and immunomodulatory activities. J. Food Drug Anal. 2018, 26, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Kim, S.M. Characterization and immunomodulatory activities of polysaccharides extracted from green alga Chlorella ellipsoidea. Int. J. Biol. Macromol. 2017, 95, 106–114. [Google Scholar] [CrossRef]
- Alvarez-Gomez, F.; Korbee, N.; Figueroa, F.L.F.L.; Álvarez-Gómez, F.; Korbee, N.; Figueroa, F.L.F.L. Analysis of antioxidant capacity and bioactive compounds in marine macroalgal and lichenic extracts using different solvents and evaluation methods. Ciencias Mar. 2016, 42, 271–288. [Google Scholar] [CrossRef]
- Álvarez-Gómez, F.; Bouzon, Z.L.; Korbee, N.; Celis-Plá, P.; Schmidt, É.C.; Figueroa, F.L. Combined effects of UVR and nutrients on cell ultrastructure, photosynthesis and biochemistry in Gracilariopsis longissima (Gracilariales, Rhodophyta). Algal Res. 2017, 26, 190–202. [Google Scholar] [CrossRef]
- Suh, S.-S.; Hwang, J.; Park, M.; Seo, H.H.; Kim, H.-S.; Lee, J.H.; Moh, S.H.; Lee, T.-K. Anti-inflammation activities of mycosporine-like amino acids (MAAs) in response to UV radiation suggest potential anti-skin aging activity. Mar. Drugs 2014, 12, 5174–5187. [Google Scholar] [CrossRef]
- Becker, K.; Hartmann, A.; Ganzera, M.; Fuchs, D.; Gostner, J.M. Immunomodulatory Effects of the Mycosporine-Like Amino Acids Shinorine and Porphyra-334. Mar. Drugs 2016, 14, 119. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, S.; Ghosal, P.K.; Pujol, C.A.; Carlucci, M.J.; Damonte, E.B.; Ray, B. Isolation, chemical investigation and antiviral activity of polysaccharides from Gracilaria corticata (Gracilariaceae, Rhodophyta). Int. J. Biol. Macromol. 2002, 31, 87–95. [Google Scholar] [CrossRef]
- Moo-Puc, R.; Robledo, D.; Freile-Pelegrin, Y. Evaluation of selected tropical seaweeds for in vitro anti-trichomonal activity. J. Ethnopharmacol. 2008, 120, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Stabili, L.; Acquaviva, M.I.; Biandolino, F.; Cavallo, R.A.; Cecere, E.; Lo Noce, R.; Narracci, M.; Petrocelli, A. Antibacterial activity of Gracilariopsis longissima (Rhodophyta, Gracilariales) lipidic extract. Rapp. Comm. int. Mer. Méd. 2010, 39. [Google Scholar]
- Stabili, L.; Acquaviva, M.I.; Biandolino, F.; Cavallo, R.A.; de Pascali, S.A.; Fanizzi, F.P.; Narracci, M.; Petrocelli, A.; Cecere, E. The lipidic extract of the seaweed Gracilariopsis longissima (Rhodophyta, Gracilariales): A potential resource for biotechnological purposes? New Biotechnol. 2012, 29, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Saeidnia, S.; Permeh, P.; Gohari, A.R.; Mashinchian-Moradi, A. Gracilariopsis persica, from Persian Gulf, Contains Bioactive Sterols. Iran. J. Pharm. Res. 2012, 11, 845–849. [Google Scholar] [PubMed]
- Whitehead, K.; Hedges, J.I. Electrospray ionization tandem mass spectrometric and electron impact mass spectrometric characterization of mycosporine-like amino acids. Rapid Commun. Mass Spectrom. 2003, 17, 2133–2138. [Google Scholar] [CrossRef]
- Banaszak, A.T. Photoprotective physiological and biochemical responses of aquatic organisms. UV Eff. Aquat. Org. Ecosyst. 2003, 1, 329–356. [Google Scholar]
- Korbee, N.; Abdala Díaz, R.T.; Figueroa, F.L.; Helbling, E.W.; Peinado, N.K.; Abdala Díaz, R.T.; Figueroa, F.L.; Helbling, E.W. Ammonium and UV radiation stimulate the accumulation of mycosporine-like amino acids in Porphyra columbina (Rhodophyta) from Patagonia, Argentina. J. Phycol. 2004, 40, 248–259. [Google Scholar] [CrossRef]
- Figueroa, F.L.; Korbee, N. Interactive effects of UV radiation and nutrients on ecophysiology: Vulnerability and adaptation to climate change. In Seaweeds and Their Role in Globally Changing Environments; Springer: Dordrecht, The Netherlands, 2010; pp. 157–182. [Google Scholar]
- Cabello-Pasini, A.; Macías-Carranza, V.; Abdala, R.; Korbee, N.; Figueroa, F.L. Effect of nitrate concentration and UVR on photosynthesis, respiration, nitrate reductase activity, and phenolic compounds in Ulva rigida (Chlorophyta). J. Appl. Phycol. 2011, 23, 363–369. [Google Scholar] [CrossRef]
- Garcia-Pichel, F.; Wingard, C.E.; Castenholz, R.W. Evidence regarding the UV sunscreen role of a mycosporine-like compound in the cyanobacterium Gloeocapsa sp. Appl. Environ. Microbiol. 1993, 59, 170–176. [Google Scholar] [PubMed]
- Torres, P.; Santos, J.P.; Chow, F.; Ferreira, M.J.P.; dos Santos, D.Y.A.C. Comparative analysis of in vitro antioxidant capacities of mycosporine-like amino acids (MAAs). Algal Res. 2018, 34, 57–67. [Google Scholar] [CrossRef]
- Kageyama, H.; Waditee-Sirisattha, R. Mycosporine-Like Amino Acids as Multifunctional Secondary Metabolites in Cyanobacteria: From Biochemical to Application Aspects. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; Volume 59, pp. 153–194. ISBN 1572-5995. [Google Scholar]
- Korbee, N.; Figueroa, F.L.; Aguilera, J. Accumulation of mycosporine-like amino acids (MAAs): Biosynthesis, photocontrol and ecophysiological functions. Rev. Chil. Hist. Nat. 2006, 79, 119–132. [Google Scholar]
- Figueroa, F.L.; Korbee, N.; Abdala, R.; Jerez, C.G.; López-de la Torre, M.; Güenaga, L.; Larrubia, M.A.; Gómez-Pinchetti, J.L. Biofiltration of fishpond effluents and accumulation of N-compounds (phycobiliproteins and mycosporine-like amino acids) versus C-compounds (polysaccharides) in Hydropuntia cornea (Rhodophyta). Mar. Pollut. Bull. 2012, 64, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Renner, G.; Audebert, F.; Burfeindt, J.; Calvet, B.; Caratas-Perifan, M.; Leal, M.E.; Gorni, R.; Long, A.; Meredith, E.; O’Sullivan, Ú. Cosmetics Europe guidelines on the management of undesirable effects and reporting of serious undesirable effects from cosmetics in the European Union. Cosmetics 2017, 4, 1. [Google Scholar] [CrossRef]
- Baran, R.; Maibach, H.I. Textbook of cosmetic dermatology; CRC Press: Oxford, NY, USA, 2017; ISBN 1482257351. [Google Scholar]
- Jahan, A.; Ahmad, I.Z.; Fatima, N.; Ansari, V.A.; Akhtar, J. Algal bioactive compounds in the cosmeceutical industry: A review. Phycologia 2017, 56, 410–422. [Google Scholar] [CrossRef]
- Álvarez-Gómez, F. Producción de Compuestos Bioactivos a Partir de Biomasa Algal Basada en la Biofiltración y la Biorrefinería. Ph.D. Thesis, Universidad de Málaga, Málaga, Spain, 18 December 2017. [Google Scholar]
- Bhatia, S.; Sharma, K.; Namdeo, A.G.; Chaugule, B.B.; Kavale, M.; Nanda, S. Broad-spectrum sun-protective action of Porphyra-334 derived from Porphyra vietnamensis. Pharmacognosy Res. 2010, 2, 45. [Google Scholar] [CrossRef] [PubMed]
- Wada, N.; Sakamoto, T.; Matsugo, S. Mycosporine-Like Amino Acids and Their Derivatives as Natural Antioxidants. Antioxidants 2015, 4, 603–646. [Google Scholar] [CrossRef]
- Schmid, D.; Schürch, C.; Zülli, F. Mycosporine-like amino acids from red algae protect against premature skin-aging. Euro Cosmet. 2006, 9, 1–4. [Google Scholar]
- Andre, G.; Pellegrini, M.; Pellegrini, L. Algal extracts containing amino acid analogs of mycosporine are useful as dermatological protecting agents against ultraviolet radiation. Patent No. FR2,803,201, 6 July 2001. [Google Scholar]
- De La Coba, F.; Aguilera, J.; Figueroa, F.L.; De Gálvez, M.V.; Herrera, E. Antioxidant activity of mycosporine-like amino acids isolated from three red macroalgae and one marine lichen. J. Appl. Phycol. 2009, 21, 161–169. [Google Scholar] [CrossRef]
- De la Coba, F.; Aguilera, J.; De Galvez, M.V.; Alvarez, M.; Gallego, E.; Figueroa, F.L.; Herrera, E. Prevention of the ultraviolet effects on clinical and histopathological changes, as well as the heat shock protein-70 expression in mouse skin by topical application of algal UV-absorbing compounds. J. Dermatol. Sci. 2009, 55, 161–169. [Google Scholar] [CrossRef]
- Berridge, M.V.; Tan, A.S. Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT): Subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Arch. Biochem. Biophys. 1993, 303, 474–482. [Google Scholar] [CrossRef]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar]
- Van Meerloo, J.; Kaspers, G.J.L.; Cloos, J. Cell sensitivity assays: The MTT assay. Cancer cell Cult. methods Protoc. 2011, 237–245. [Google Scholar]
- Kladi, M.; Vagias, C.; Roussis, V. Volatile halogenated metabolites from marine red algae. Phytochem. Rev. 2004, 3, 337–366. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.E.; Lin, X. Bromophenols in marine algae and their bioactivities. Mar. Drugs 2011, 9, 1273–1292. [Google Scholar] [CrossRef]
- Suh, S.-S.; Oh, S.K.; Lee, S.G.; Kim, I.-C.; Kim, S. Porphyra-334, a mycosporine-like amino acid, attenuates UV-induced apoptosis in HaCaT cells. Acta Pharm. 2017, 67, 257–264. [Google Scholar] [CrossRef]
- Garcia-Galaz, A.; Gutiérrez-Millán, L.E.; Acedo-Félix, E.; Burgos-Hernández, A.; LópezTorres, M.; Valdés-Covarrubias, M.; Burboa-Zazueta, M.G. Las algas y otros organismos marinos como fuente de moléculas bioactivas. Biotecnia 2014, 15, 25–32. [Google Scholar] [CrossRef]
- International Organization for Standardization ISO 10993-12. Biological Evaluation of Medical Devices-Part 12, Sample Preparation and Reference Materials. Br. Stand. Institution, UK 2004. [Google Scholar] [CrossRef]
- Fernandes, S.C.M.; Alonso-Varona, A.; Palomares, T.; Zubillaga, V.; Labidi, J.; Bulone, V. Exploiting mycosporines as natural molecular sunscreens for the fabrication of UV-absorbing green materials. ACS Appl. Mater. Interfaces 2015, 7, 16558–16564. [Google Scholar] [CrossRef]
- Oyamada, C.; Kaneniwa, M.; Ebitani, K.; Murata, M.; Ishihara, K. Mycosporine-like amino acids extracted from scallop (Patinopecten yessoensis) ovaries: UV protection and growth stimulation activities on human cells. Mar. Biotechnol. 2008, 10, 141–150. [Google Scholar] [CrossRef]
- Kim, S.; You, D.H.; Han, T.; Choi, E.-M. Modulation of viability and apoptosis of UVB-exposed human keratinocyte HaCaT cells by aqueous methanol extract of laver (Porphyra yezoensis). J. Photochem. Photobiol. B Biol. 2014, 141, 301–307. [Google Scholar] [CrossRef]
- Ryu, J.; Park, S.-J.; Kim, I.-H.; Choi, Y.; Nam, T.-J. Protective effect of porphyra-334 on UVA-induced photoaging in human skin fibroblasts. Int. J. Mol. Med. 2014, 796–803. [Google Scholar] [CrossRef]
- Choi, Y.-H.; Yang, D.J.; Kulkarni, A.; Moh, S.H.; Kim, K.W. Mycosporine-Like Amino Acids Promote Wound Healing through Focal Adhesion Kinase (FAK) and Mitogen-Activated Protein Kinases (MAP Kinases) Signaling Pathway in Keratinocytes. Mar. Drugs 2015, 13, 7055–7066. [Google Scholar] [CrossRef]
- Torres, A.; Hochberg, M.; Pergament, I.; Smoum, R.; Niddam, V.; Dembitsky, V.M.; Temina, M.; Dor, I.; Lev, O.; Srebnik, M. A new UV-B absorbing mycosporine with photo protective activity from the lichenized ascomycete Collema cristatum. Eur. J. Biochem. 2004, 271, 780–784. [Google Scholar] [CrossRef]
- Fernando, I.P.S.; Nah, J.-W.; Jeon, Y.-J. Potential anti-inflammatory natural products from marine algae. Environ. Toxicol. Pharmacol. 2016, 48, 22–30. [Google Scholar] [CrossRef]
- Yoon, W.-J.; Ham, Y.M.; Kim, K.-N.; Park, S.-Y.; Lee, N.H.; Hyun, C.-G.; Lee, W.J. Anti-inflammatory activity of brown alga Dictyota dichotoma in murine macrophage RAW 264.7 cells. J. Med. Plants Res. 2009, 3, 1–8. [Google Scholar]
- Yang, Y.-I.; Shin, H.-C.; Kim, S.H.; Park, W.-Y.; Lee, K.-T.; Choi, J.-H. 6,6′-Bieckol, isolated from marine alga Ecklonia cava, suppressed LPS-induced nitric oxide and PGE 2 production and inflammatory cytokine expression in macrophages: The inhibition of NF-κB. Int. Immunopharmacol. 2012, 12, 510–517. [Google Scholar] [CrossRef]
- Yoshizawa, Y.; Ametani, A.; Tsunehiro, J.; Nomura, K.; Itoh, M.; Fukui, F.; Kaminogawa, S. Macrophage stimulation activity of the polysaccharide fraction from a marine alga (Porphyra yezoensis): Structure-function relationships and improved solubility. Biosci. Biotechnol. Biochem. 1995, 59, 1933–1937. [Google Scholar] [CrossRef]
- Yim, J.H.; Son, E.; Pyo, S.; Lee, H.K. Novel sulfated polysaccharide derived from red-tide microalga Gyrodinium impudicum strain KG03 with immunostimulating activity in vivo. Mar. Biotechnol. 2005, 7, 331–338. [Google Scholar] [CrossRef]
- Steentoft, M.; Irvine, L.M.; Farnham, W.F. Two terete species of Gracilaria and Gracilariopsis (Gracilariales, Rhodophyta) in Britain. Phycologia 1995, 34, 113–127. [Google Scholar] [CrossRef]
- Lee, H.-J.; Dang, H.-T.; Kang, G.-J.; Yang, E.-J.; Park, S.-S.; Yoon, W.-J.; Jung, J.H.; Kang, H.-K.; Yoo, E.-S. Two enone fatty acids isolated from Gracilaria verrucosa suppress the production of inflammatory mediators by down-regulating NF-κB and STAT1 activity in lipopolysaccharide-stimulated Raw264.7 cells. Arch. Pharm. Res. 2009, 32, 453–462. [Google Scholar] [CrossRef]
- Rao, P.P.S. Biological investigations of Indian phaeophyceae: 17. Seasonal variation of antibacterial activity of total sterols obtained from frozen samples of Sargassum johnstonii Setchell et Gardner. Seaweed Res. Util. 1998, 20, 91–95. [Google Scholar]
- Kamenarska, Z.G.; Dimitrova-Konaklieva, S.D.; Stefanov, L.K.; Popov, S.S. A comparative study on the sterol composition of some brown algae from the Black Sea. J. Serbian Chem. Soc. 2003, 68, 269–275. [Google Scholar] [CrossRef]
- Lee, Y.S.; Shin, K.H.; Kim, B.-K.; Lee, S. Anti-diabetic activities of fucosterol from Pelvetia siliquosa. Arch. Pharm. Res. 2004, 27, 1120–1122. [Google Scholar] [CrossRef]
- Permeh, P.; Gohari, A.; Saeidnia, S.; Mashinchian-Moradi, A.; Dasian, Z. Bioactivity and sterols from Gracilariopsis persica and Sargassum oligocystum. Planta Med. 2010, 76, 322. [Google Scholar] [CrossRef]
- Lee, S.; Lee, Y.S.; Jung, S.H.; Kang, S.S.; Shin, K.H. Anti-oxidant activities of fucosterol from the marine algae Pelvetia siliquosa. Arch. Pharm. Res. 2003, 26, 719–722. [Google Scholar] [CrossRef]
- Wilt, T.J.; MacDonald, R.; Ishani, A. β-sitosterol for the treatment of benign prostatic hyperplasia: A systematic review. BJU Int. 1999, 83, 976–983. [Google Scholar] [CrossRef]
- Loizou, S.; Lekakis, I.; Chrousos, G.P.; Moutsatsou, P. β-Sitosterol exhibits anti-inflammatory activity in human aortic endothelial cells. Mol. Nutr. Food Res. 2010, 54, 551–558. [Google Scholar] [CrossRef]
- Pathak, J.; Sonker, A.S.; Singh, V.; Sinha, R.P. Potential Applications of Natural Bioactive Cyanobacterial UV-Protective Compounds. Blue Biotechnol. Prod. Use Mar. Mol. 2018, 2, 683–707. [Google Scholar]
- Rastogi, R.P.; Sinha, R.P.; Singh, S.P.; Häder, D.-P. Photoprotective compounds from marine organisms. J. Ind. Microbiol. Biotechnol. 2010, 37, 537–558. [Google Scholar] [CrossRef]
- Pugh, N.; Ross, S.A.; ElSohly, H.N.; ElSohly, M.A.; Pasco, D.S. Isolation of three high molecular weight polysaccharide preparations with potent immunostimulatory activity from Spirulina platensis, Aphanizomenon flos-aquae and Chlorella pyrenoidosa. Planta Med. 2001, 67, 737–742. [Google Scholar] [CrossRef]
- Parages, M.L.; Rico, R.M.; Abdala-Díaz, R.T.; Chabrillón, M.; Sotiroudis, T.G.; Jiménez, C. Acidic polysaccharides of Arthrospira (Spirulina) platensis induce the synthesis of TNF-α in RAW macrophages. J. Appl. Phycol. 2012, 24, 1537–1546. [Google Scholar] [CrossRef]
- Bandaranayake, W.M. Mycosporines: Are they nature’s sunscreens? Nat. Prod. Rep. 1998, 15, 159–172. [Google Scholar] [CrossRef]
- Cosmetics Europe. In Vitro Method for the Determination of the UVA Protection Factor and “Critical Wavelength” Values of Sunscreen Products. 2011. Available online: http://s6ce4e41c97d2da30.jimcontent.com/download/version/1349752658/module/6226369377/name/Colipa%20UVA%20In%20Vitro%20Method%20-%20March%202011-Final.pdf (accessed on 11 September 2017).
- Schepetkin, I.A.; Quinn, M.T. Botanical polysaccharides: Macrophage immunomodulation and therapeutic potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef]
Sample Availability: Samples of the lyophilized algal biomass are available from the authors. |
| Species | MAA Type | % | Mol. Formula | λmax (nm) | Exact (ppm) | Calculated (m/z [M + H]+) | Observed (m/z [M + H]+) |
|---|---|---|---|---|---|---|---|
| Hc | Palythine | 29.9 ± 1.5 | C10H16N2O5 | 320 | 1.2 | 245.11320 | 245.11290 |
| Asterina-330 | 12.9 ± 1.8 | C12H20N2O6 | 330 | 1.8 | 289.13941 | 289.13889 | |
| Shinorine | 5 ± 1.5 | C13H20N2O8 | 334 | 1.4 | 333.12924 | 333.12878 | |
| Porphyra-334 | 3 ± 0.8 | C14H22N2O8 | 334 | 0.8 | 347.14489 | 347.14462 | |
| Palythinol | 49.2 ± 3.6 | C13H22N2O6 | 332 | 0.9 | 303.15506 | 303.15479 | |
| Total MAAs | 0.8 ± 0.1 mg g−1 DW | ||||||
| Gl | Palythine | 0.3 ± 0.1 | C10H16N2O5 | 320 | 1.2 | 245.1132 | 245.11290 |
| Asterina-330 | 42.9 ± 1.1 | C12H20N2O6 | 330 | 1.6 | 289.13941 | 289.13895 | |
| Shinorine | 41.2 ± 2 | C13H20N2O8 | 334 | 1.8 | 333.12924 | 333.12863 | |
| Porphyra-334 | 1.7 ± 0.1 | C14H22N2O8 | 334 | 1.6 | 347.14489 | 347.14435 | |
| Palythinol | 13.9 ± 0.5 | C13H22N2O6 | 332 | 1.5 | 303.15506 | 303.15460 | |
| Total MAAs | 1.6 ± 0.1 mg g−1 DW | ||||||
| Species | RAW264.7 | HGF | HaCaT |
|---|---|---|---|
| Hydropuntia cornea | 0.12 | 250.7 | 259.5 |
| Gracilariopsis longissima | 0.41 | 4.2 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Álvarez-Gómez, F.; Korbee, N.; Casas-Arrojo, V.; Abdala-Díaz, R.T.; Figueroa, F.L. UV Photoprotection, Cytotoxicity and Immunology Capacity of Red Algae Extracts. Molecules 2019, 24, 341. https://doi.org/10.3390/molecules24020341
Álvarez-Gómez F, Korbee N, Casas-Arrojo V, Abdala-Díaz RT, Figueroa FL. UV Photoprotection, Cytotoxicity and Immunology Capacity of Red Algae Extracts. Molecules. 2019; 24(2):341. https://doi.org/10.3390/molecules24020341
Chicago/Turabian StyleÁlvarez-Gómez, Félix, Nathalie Korbee, Virginia Casas-Arrojo, Roberto T. Abdala-Díaz, and Félix L. Figueroa. 2019. "UV Photoprotection, Cytotoxicity and Immunology Capacity of Red Algae Extracts" Molecules 24, no. 2: 341. https://doi.org/10.3390/molecules24020341
APA StyleÁlvarez-Gómez, F., Korbee, N., Casas-Arrojo, V., Abdala-Díaz, R. T., & Figueroa, F. L. (2019). UV Photoprotection, Cytotoxicity and Immunology Capacity of Red Algae Extracts. Molecules, 24(2), 341. https://doi.org/10.3390/molecules24020341






