Synthesis and Properties of a Bay-Annulated-Indigo Tetramer Based on Low-Cost Spiro[Fluorene-9,9′-Xanthene] Core
Abstract
:1. Introduction
2. Results and Discussion
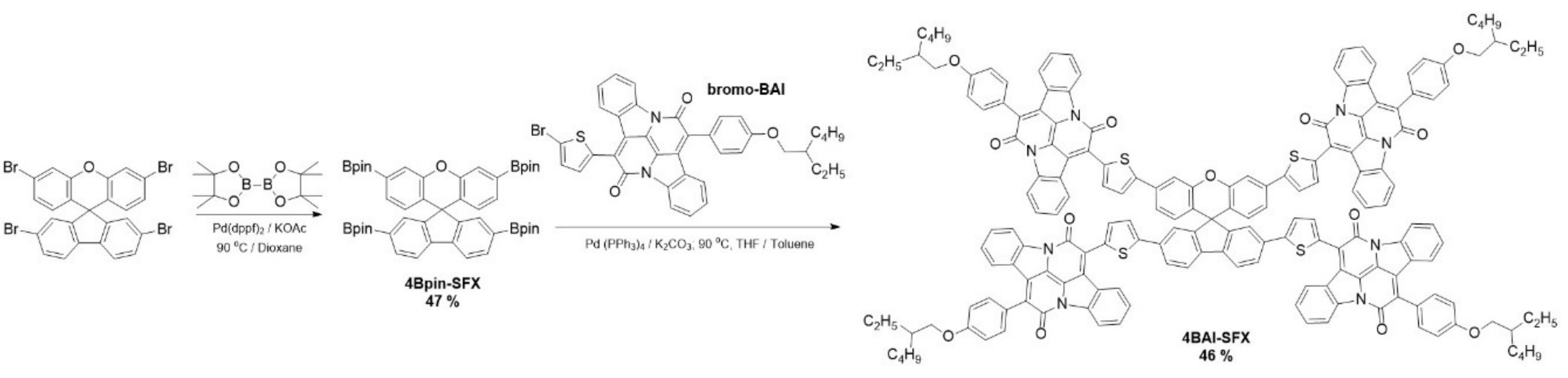
2.1. Synthesis and Cost Analysis
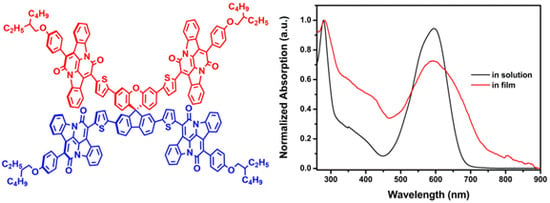
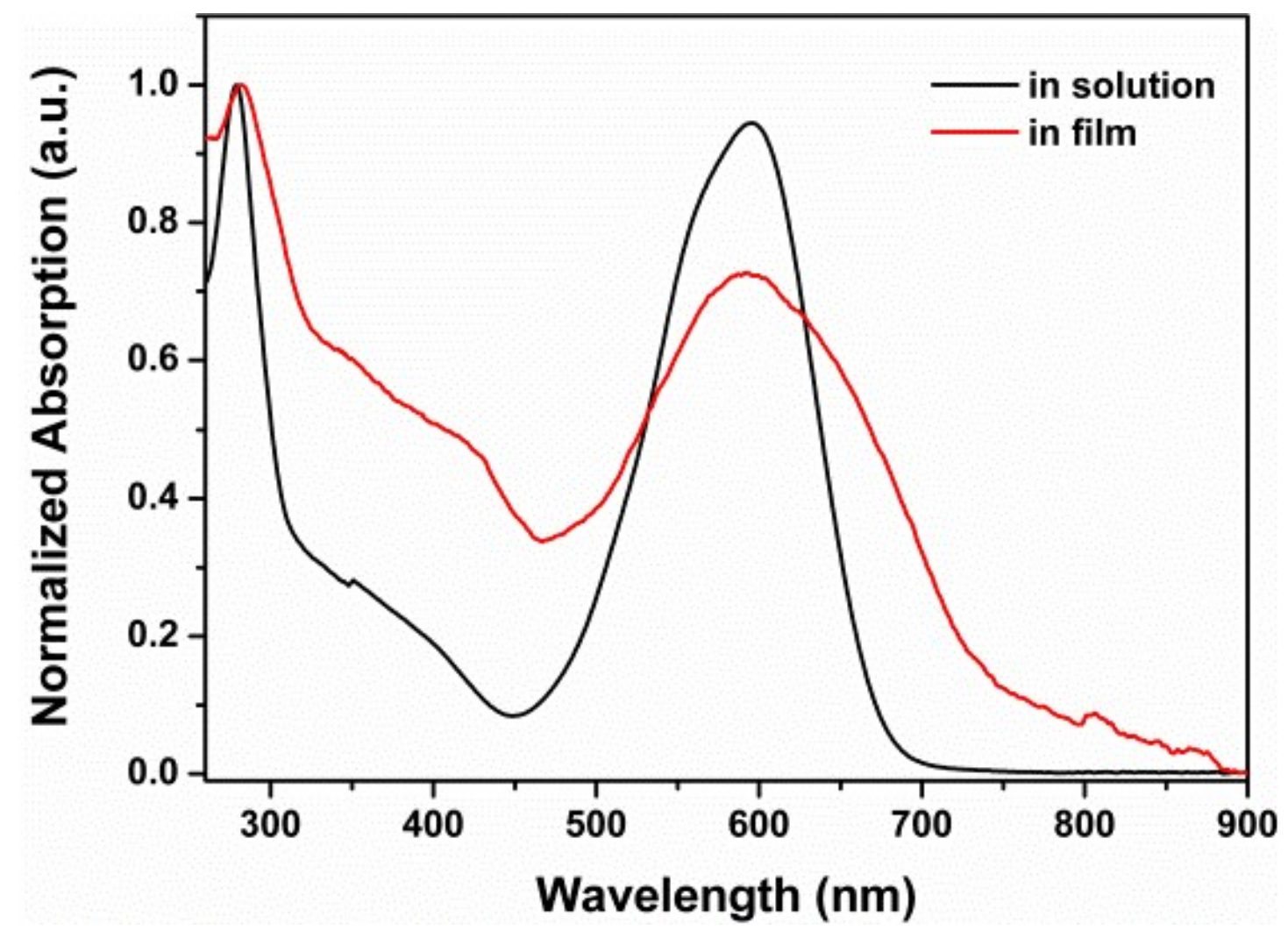
2.2. Photophysical Properties
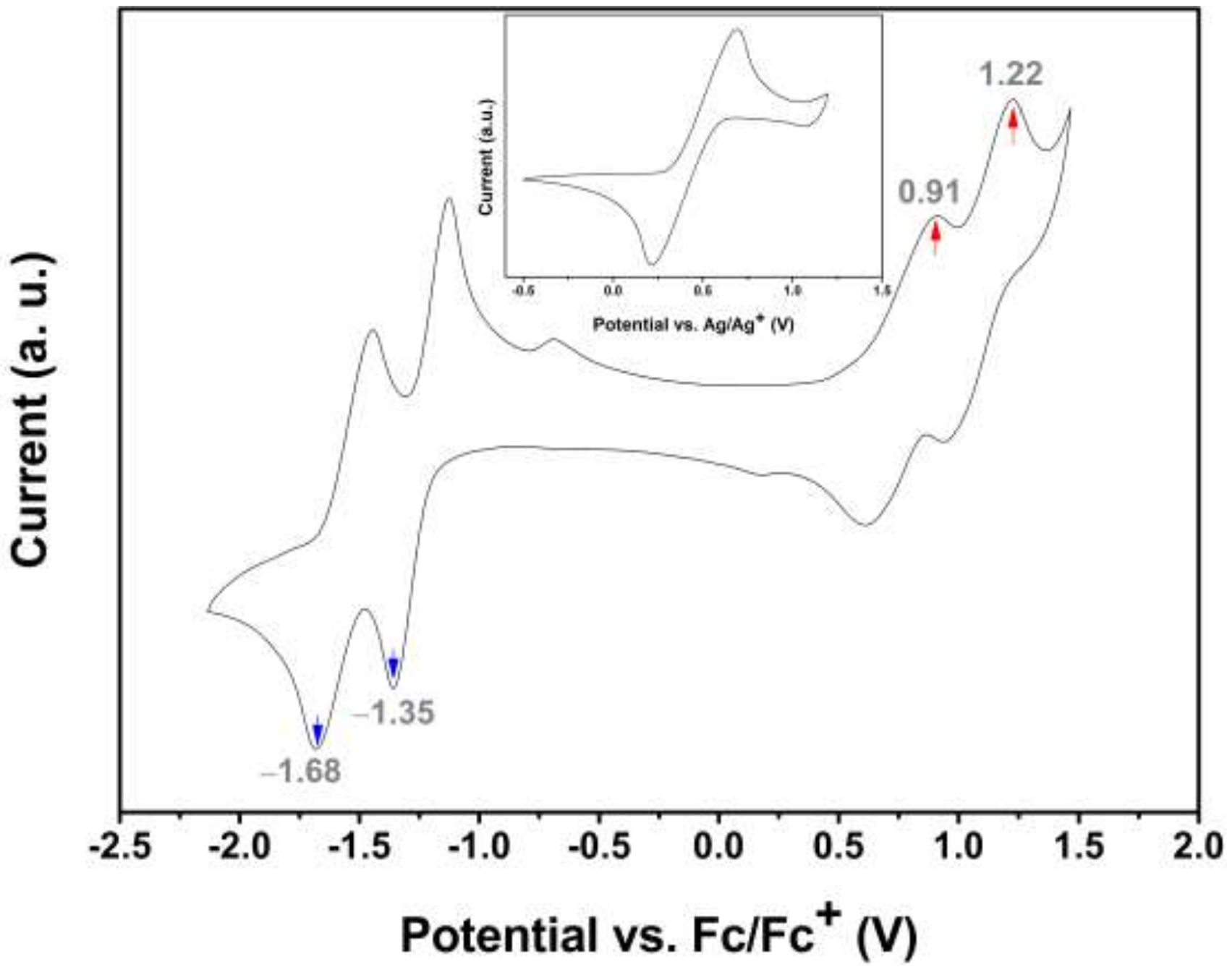
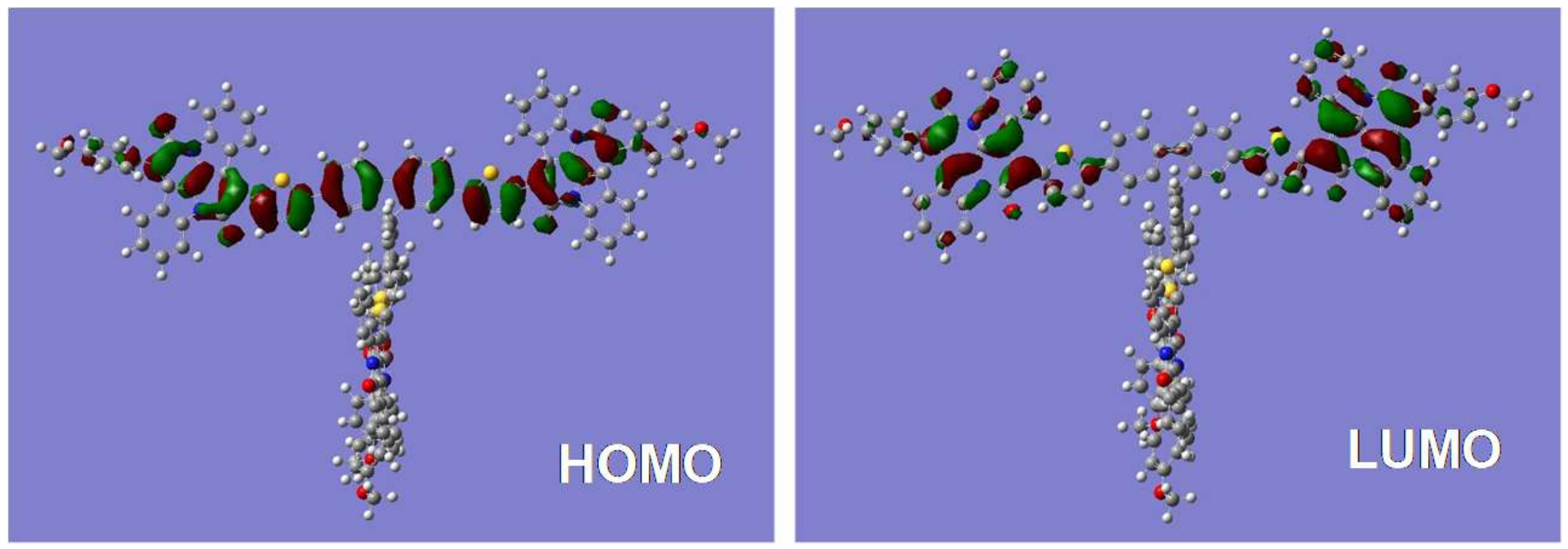
2.3. Electrochemical and Computational Studies
3. Materials and Methods
3.1. General Information
3.2. Synthetic Procedure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, J.; Zhang, H.; Dong, H.; Meng, L.; Jiang, L.; Jiang, L.; Wang, Y.; Yu, J.; Sun, Y.; Hu, W.; et al. High mobility emissive organic semiconductor. Nat. Commun. 2015, 6, 10032. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Zhang, X.; Xie, L.; Qi, D.; Chandran, B.K.; Chen, X.; Huang, W. Stretchable organic semiconductor devices. Adv. Mater. 2016, 28, 9243–9265. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Song, X.; Xie, J.; Sun, P.; Gu, P.; Liu, C.; Chen, F.; Zhang, Q.; Chen, Z.-K.; Huang, W. PDI derivative through fine-tuning the molecular structure for fullerene-free organic solar cells. Acs Appl. Mater. Interfaces 2017, 9, 29924–29931. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Sun, P.; Zhang, C.; Yang, Y.; Gao, X.; Chen, F.; Xu, Z.; Chen, Z.-K.; Huang, W. High-performance organic solar cells based on a non-fullerene acceptor with a spiro core. Chem. Asian. J. 2017, 12, 721–725. [Google Scholar] [CrossRef] [PubMed]
- Maciejczyk, M.; Ivaturi, A.; Robertson, N. SFX as a low-cost “spiro” hole-transport material for efficient perovskite solar cells. J. Mater. Chem. A 2016, 4, 4855–4863. [Google Scholar] [CrossRef]
- Xu, B.; Bi, D.; Hua, Y.; Liu, P.; Cheng, M.; Grätzel, M.; Kloo, L.; Hagfeldt, A.; Sun, L. A low-cost spiro [fluorene-9, 9′-xanthene]-based hole transport material for highly efficient solid-state dye-sensitized solar cells and perovskite solar cells. Energ. Environ. Sci. 2016, 9, 873–877. [Google Scholar] [CrossRef]
- Anthony, J.E.; Facchetti, A.; Heeney, M.; Marder, S.R.; Zhan, X. n-type organic semiconductors in organic electronics. Adv. Mater. 2010, 22, 3876–3892. [Google Scholar] [CrossRef]
- Kolaczkowski, M.A.; He, B.; Liu, Y. Stepwise bay annulation of indigo for the synthesis of desymmetrized electron acceptors and donor-acceptor constructs. Org. Lett. 2016, 18, 5224–5227. [Google Scholar] [CrossRef]
- He, B.; Pun, A.B.; Zherebetskyy, D.; Liu, Y.; Liu, F.; Klivansky, L.M.; McGough, A.M.; Zhang, B.A.; Lo, K.; Russell, T.P. New form of an old natural dye: bay-annulated indigo (bai) as an excellent electron accepting unit for high performance organic semiconductors. J. Am. Chem. Soc. 2014, 136, 15093–15101. [Google Scholar] [CrossRef]
- Kolaczkowski, M.A.; Liu, Y. Functional organic semiconductors based on bay-annulated indigo (BAI). Chem. Rec. 2019, 19, 1062–1077. [Google Scholar] [CrossRef]
- He, B.; Neo, W.T.; Chen, T.L.; Klivansky, L.M.; Wang, H.; Tan, T.; Teat, S.J.; Xu, J.; Liu, Y. Low bandgap conjugated polymers based on a nature-inspired bay-annulated indigo (BAI) acceptor as stable electrochromic materials. ACS Sustain. Chem. Eng. 2016, 4, 2797–2805. [Google Scholar] [CrossRef]
- Fallon, K.J.; Wijeyasinghe, N.; Manley, E.F.; Dimitrov, S.D.; Yousaf, S.A.; Ashraf, R.S.; Duffy, W.; Guilbert, A.A.; Freeman, D.M.; Al-Hashimi, M. Indolo-naphthyridine-6, 13-dione thiophene building block for conjugated polymer electronics: molecular origin of ultrahigh n-type mobility. Chem. Mater. 2016, 28, 8366–8378. [Google Scholar] [CrossRef]
- Yang, J.; Jiang, Y.; Tu, Z.; Zhao, Z.; Chen, J.; Yi, Z.; Li, Y.; Wang, S.; Yi, Y.; Guo, Y.; et al. High-performance ambipolar polymers based on electron-withdrawing group substituted bay-annulated indigo. Adv. Funct. Mater. 2019, 29, 1804839. [Google Scholar] [CrossRef]
- Stahl, T.; Bofinger, R.; Lam, I.; Fallon, K.J.; Johnson, P.; Ogunlade, O.; Vassileva, V.; Pedley, R.B.; Beard, P.C.; Hailes, H.C. Tunable semiconducting polymer nanoparticles with indt-based conjugated polymers for photoacoustic molecular imaging. Bioconjugate Chem. 2017, 28, 1734–1740. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ding, D.; Tao, Y.; Wei, Y.; Chen, R.; Xie, L.; Huang, W.; Xu, H. A significantly twisted spirocyclic phosphine oxide as a universal host for high-efficiency full-color thermally activated delayed fluorescence diodes. Adv. Mater. 2016, 28, 3122–3130. [Google Scholar] [CrossRef] [PubMed]
- Furuyama, T.; Tamura, D.; Maeda, H.; Segi, M. Synthesis and peripheral substituent effects of bay-annulated indigo derivatives. Tetrahedron Lett. 2018, 59, 2913–2916. [Google Scholar] [CrossRef]
- Yan, Q.; Zhou, Y.; Zheng, Y.-Q.; Pei, J.; Zhao, D. Towards rational design of organic electron acceptors for photovoltaics: A study based on perylenediimide derivatives. Chem. Sci. 2013, 4, 4389–4394. [Google Scholar] [CrossRef]
- Xie, L.-H.; Liu, F.; Tang, C.; Hou, X.-Y.; Hua, Y.-R.; Fan, Q.-L.; Huang, W. Unexpected one-pot method to synthesize spiro[fluorene-9,9′-xanthene] building blocks for blue-light-emitting materials. Org. Lett. 2006, 8, 2787–2790. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Xu, R.; Xie, L.; Wei, Y.; Huang, W. Toward eco-friendly green organic semiconductors: Recent advances in spiro[fluorene-9,9′-xanthene](sfx)-based optoelectronic materials and devices. Chin. J. Chem. 2015, 33, 815–827. [Google Scholar] [CrossRef]
- Ren, B.-Y.; Xu, Q.; Kolaczkowski, M.; Sun, C.-L.; Ou, C.-J.; Sun, Y.-G.; Xie, L.-H.; Huang, W. Bay-annulated indigo derivatives based on a core of spiro[fluorene-9,9′-xanthene]: Synthesis, photophysical, and electrochemical properties. Dye. Pigment. 2019, 160, 25–27. [Google Scholar] [CrossRef]
- Tsang, S.W.; Chen, S.; So, F. Energy level alignment and sub-bandgap charge generation in polymer: Fullerene bulk heterojunction solar cells. Adv. Mater. 2013, 25, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 4Br-SFX and 4Bpin-SFX are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, B.-Y.; Tan, Y.-M.; Sun, C.-L.; Sheng, Y.-G. Synthesis and Properties of a Bay-Annulated-Indigo Tetramer Based on Low-Cost Spiro[Fluorene-9,9′-Xanthene] Core. Molecules 2019, 24, 3623. https://doi.org/10.3390/molecules24193623
Ren B-Y, Tan Y-M, Sun C-L, Sheng Y-G. Synthesis and Properties of a Bay-Annulated-Indigo Tetramer Based on Low-Cost Spiro[Fluorene-9,9′-Xanthene] Core. Molecules. 2019; 24(19):3623. https://doi.org/10.3390/molecules24193623
Chicago/Turabian StyleRen, Bao-Yi, Yan-Mei Tan, Chang-Liang Sun, and Yong-Gang Sheng. 2019. "Synthesis and Properties of a Bay-Annulated-Indigo Tetramer Based on Low-Cost Spiro[Fluorene-9,9′-Xanthene] Core" Molecules 24, no. 19: 3623. https://doi.org/10.3390/molecules24193623
APA StyleRen, B.-Y., Tan, Y.-M., Sun, C.-L., & Sheng, Y.-G. (2019). Synthesis and Properties of a Bay-Annulated-Indigo Tetramer Based on Low-Cost Spiro[Fluorene-9,9′-Xanthene] Core. Molecules, 24(19), 3623. https://doi.org/10.3390/molecules24193623