Characterization and Quantification of Major Flavonol Glycosides in Ramps (Allium tricoccum)
Abstract
1. Introduction
2. Results
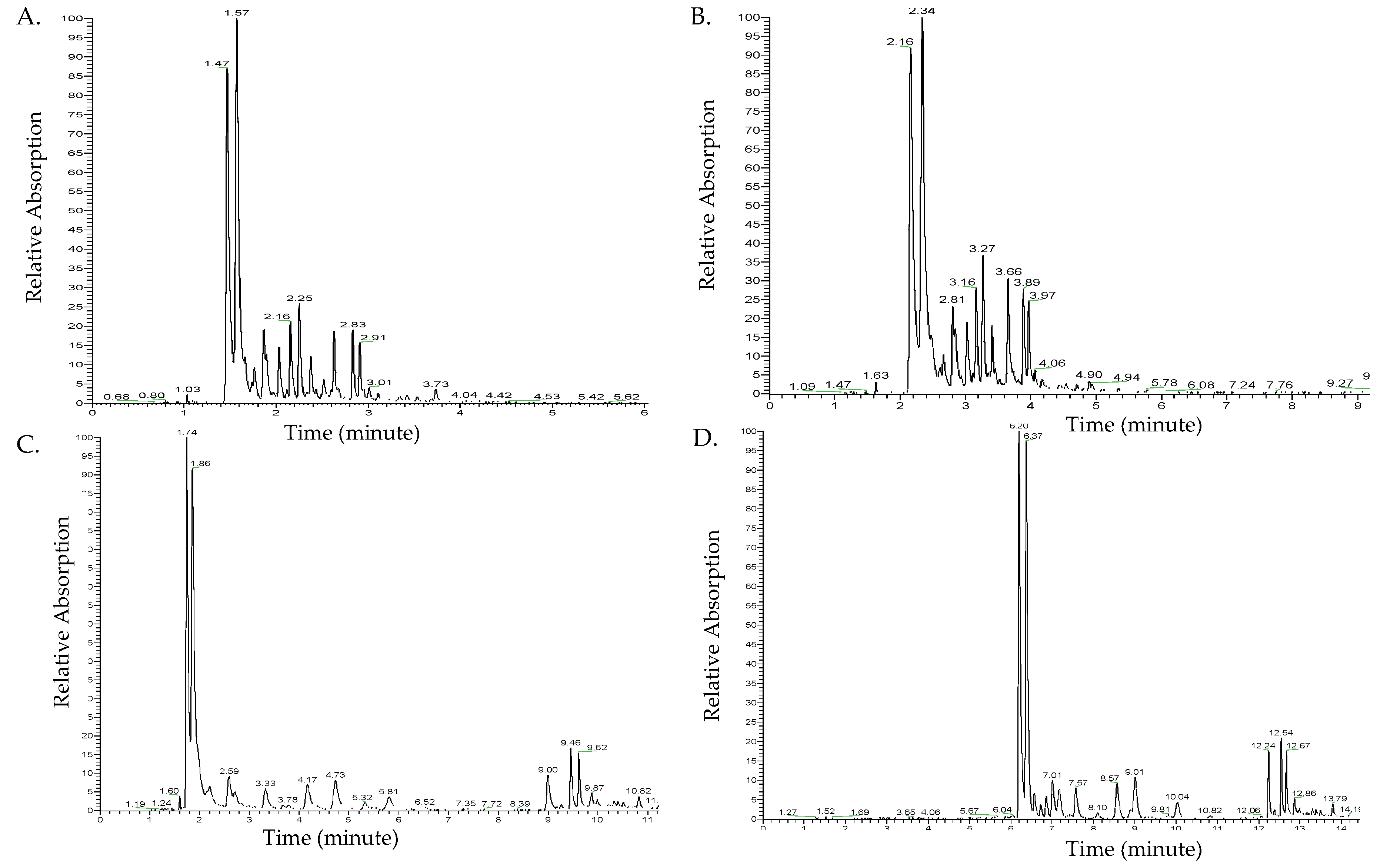
2.1. Optimization of UHPLC Separation of Methanolic Extracts
2.2. Characterization of Flavonol Glycosides
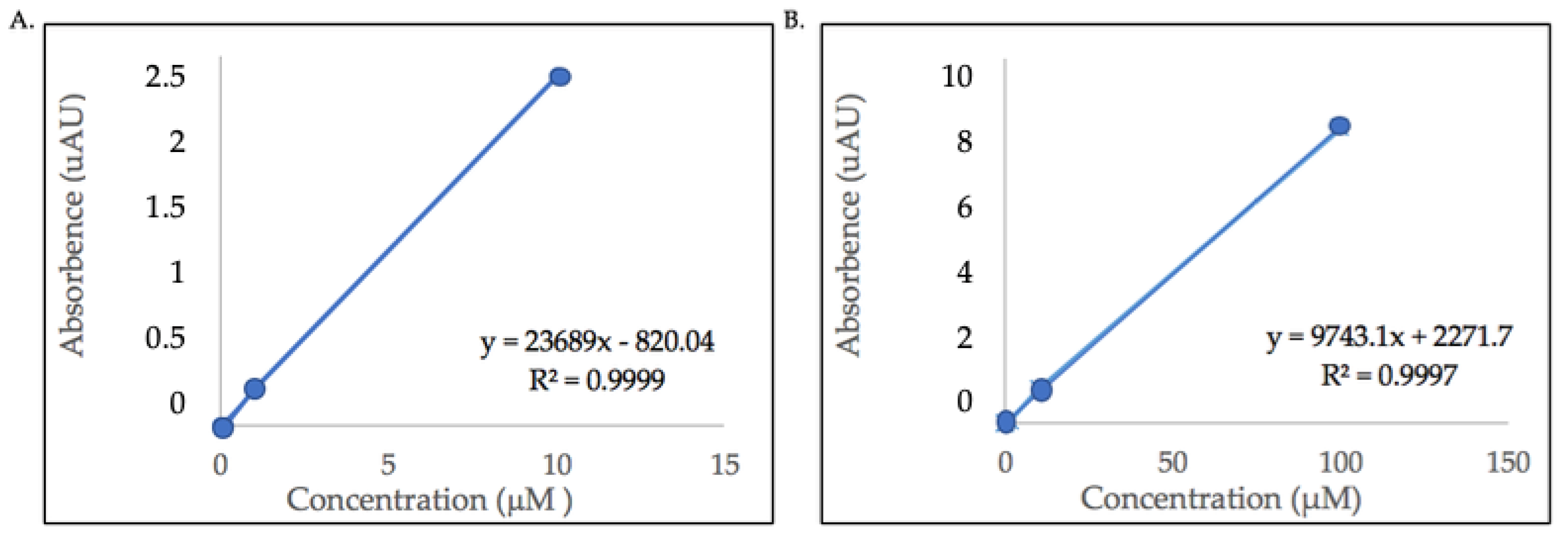
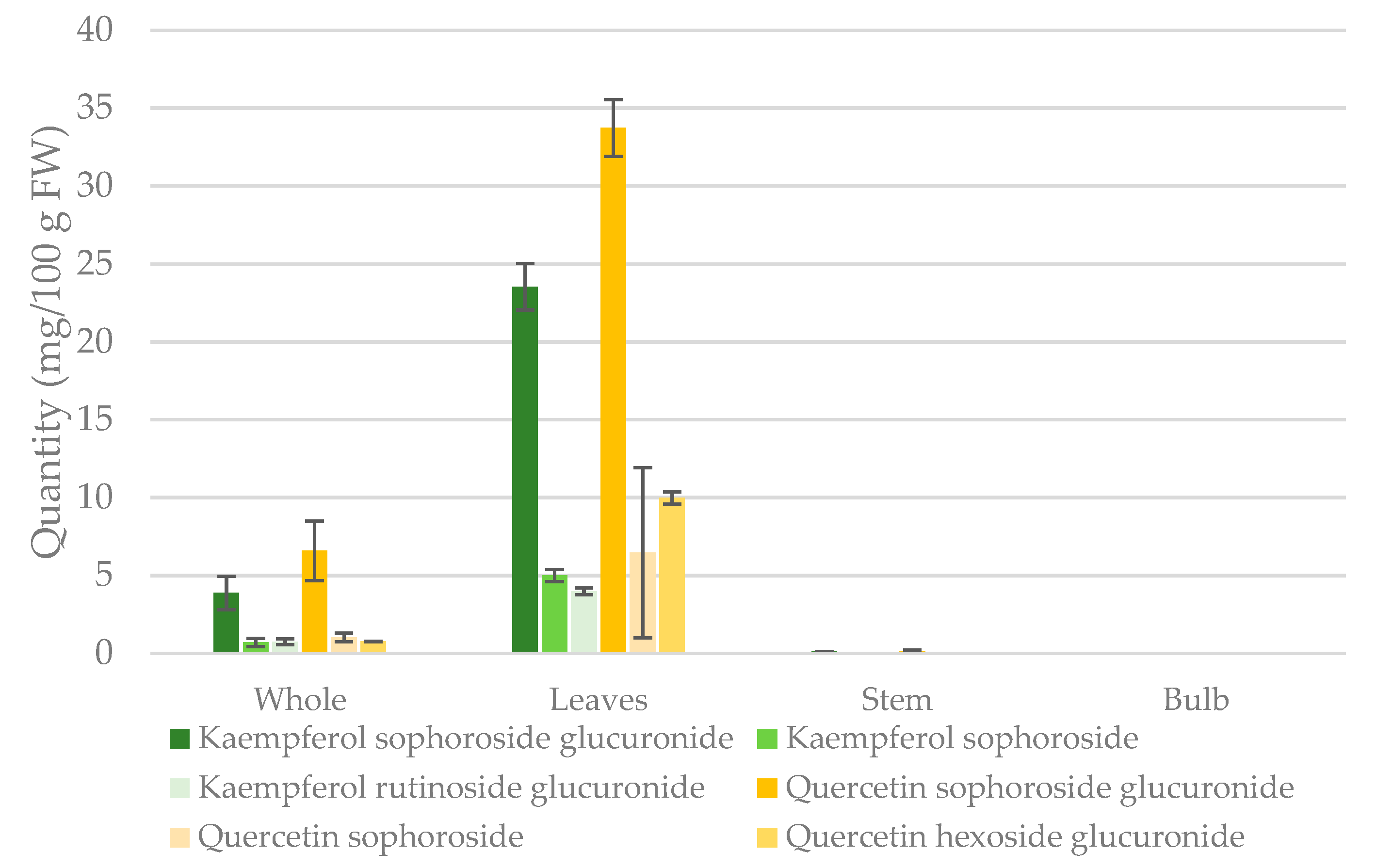
2.3. Quantification of Major Flavonol Glycosides
3. Discussion
3.1. Optimization of UHPLC Separation of Methanolic Extracts
3.2. Characterization of Major Glycosides in Ramps
3.3. Quantification of Major Flavonol Glycosides
4. Conclusions
5. Materials and Methods
5.1. Chemicals
5.2. Plant Material
5.3. Extraction of Flavonol Glycosides
5.4. Flavonol Glycoside Separation
5.5. Characterization of Flavonol Glycosides by UHPLC-PDA-MS
5.6. Quantification of Flavonol Glycosides
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- How to Grow Ramps, the Wild Leek. Available online: https://www.thespruce.com/growing-ramps-in-the-vegetable-garden-1403463 (accessed on 12 March 2019).
- Wild Leek (Allium tricoccum). Available online: http://www.illinoiswildflowers.info/woodland/plants/wild_leek.htm (accessed on 18 August 2019).
- Davis, J.; Greenfield, J. Cultivation of Ramps (Allium tricoccum and A. burdickii)/NC State Extension Publications. Available online: https://content.ces.ncsu.edu/cultivation-of-ramps-allium-tricoccum-and-a-burdickii (accessed on 21 February 2019).
- RAMPS & WILD LEEKS/Wild Harvest. Available online: http://wild-harvest.com/ramps-wild-leeks/ (accessed on 6 September 2019).
- Foster, S.; Duke, J.A.; National Audubon Society (USA); National Wildlife Federation (USA); Roger Tory Peterson Institute. A Field Guide to Medicinal Plants: Eastern and Central North America. Sci. News 1990. Available online: http://agris.fao.org/agris-search/search.do?recordID=US9132808 (accessed on 6 September 2019).
- WV Ramp Dinners & Festivals. W. Va. Explor. Available online: https://wvexplorer.com (accessed on 12 May 2019).
- Cavender, A. Folk medical uses of plant foods in southern Appalachia, United States. J. Ethnopharmacol. 2006, 108, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Preuss, H.G.; Clouatre, D.; Mohamadi, A.; Jarrell, S.T. Wild garlic has a greater effect than regular garlic on blood pressure and blood chemistries of rats. Int. Urol. Nephrol. 2001, 32, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Badary, O.A.; Yassin, N.A.Z.; El-Shenawy, S.M.A.; EL-Moneem, M.A.; AL-Shafeiy, H.M. Study of the effect of Allium porrum on hypertension induced in rats. 2013, 41, 12. Available online: http://www.scielo.org.mx/scielo.php?pid=S0370-59432013000300002&script=sci_arttext&tlng=en (accessed on 12 May 2019).
- Zeng, Y.; Li, Y.; Yang, J.; Pu, X.; Du, J.; Yang, X.; Yang, T.; Yang, S. Therapeutic Role of Functional Components in Alliums for Preventive Chronic Disease in Human Being. Evid.-Based Complement. Altern. Med. ECAM 2017, 2017, 9402849. [Google Scholar] [CrossRef] [PubMed]
- Falcone Ferreyra, M.L.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, biological functions, and biotechnological applications. Front. Plant Sci. 2012, 3, 222. [Google Scholar] [CrossRef] [PubMed]
- Cheynier, V. Polyphenols in foods are more complex than often thought. Am. J. Clin. Nutr. 2005, 81, 223S–229S. [Google Scholar] [CrossRef]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Phenols, polyphenols and tannins: An overview. In Plant Secondary Metabolites: Occurrence, Structure and Role in the Human Diet; Crozier, A., Clifford, M.N., Ashihara, H., Eds.; Blackwell Publishing: Oxford, UK, 2006; pp. 1–24. ISBN 978-1-4051-2509-3. [Google Scholar]
- Bhagwat, S.; Haytowitz, D.B.; Holden, J.M. USDA Database for the Flavonoid Content of Selected Foods Release 3. Available online: https://www.ars.usda.gov/ARSUserFiles/80400525/Data/Flav/Flav_R03.pdf (accessed on 12 May 2019).
- Nuutila, A.M.; Kammiovirta, K.; Oksman-Caldentey, K.-M. Comparison of methods for the hydrolysis of flavonoids and phenolic acids from onion and spinach for HPLC analysis. Food Chem. 2002, 76, 519–525. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 16. [Google Scholar] [CrossRef]
- Hollman, P.C.H. Absorption, Bioavailability, and Metabolism of Flavonoids. Pharm. Biol. 2004, 42, 74–83. [Google Scholar] [CrossRef]
- Xiao, J. Dietary flavonoid aglycones and their glycosides: Which show better biological significance? Crit. Rev. Food Sci. Nutr. 2017, 57, 1874–1905. [Google Scholar] [CrossRef] [PubMed]
- Ramps, Allium tricoccum. Available online: https://wimastergardener.org/article/ramps-allium-tricoccum (accessed on 18 August 2019).
- Calvey, E.M.; White, K.D.; Matusik, J.E.; Sha, D.; Block, E. Allium chemistry: Identification of organosulfur compounds in ramp (Allium tricoccum) homogenates. Phytochemistry 1998, 49, 359–364. [Google Scholar] [CrossRef]
- Zennie, T.; Ogzewalla, D. Ascorbic acid and Vitamin A content of edible wild plants of Ohio and Kentucky. Econ. Bot. 1977, 31, 76–79. [Google Scholar] [CrossRef]
- Li, Z.-H.; Guo, H.; Xu, W.-B.; Ge, J.; Li, X.; Alimu, M.; He, D.-J. Rapid Identification of Flavonoid Constituents Directly from PTP1B Inhibitive Extract of Raspberry (Rubus idaeus L.) Leaves by HPLC–ESI–QTOF–MS-MS. J. Chromatogr. Sci. 2016, 54, 805–810. [Google Scholar] [CrossRef] [PubMed]
- PubChem Kaempferol 3-rutinoside-7-glucuronide. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/44258841 (accessed on 13 August 2019).
- Urushibara, S.; Kitayama, Y.; Watanabe, T.; Okuno, T.; Watarai, A.; Matsumoto, T. New flavonol glycosides, major determinants inducing the green fluorescence in the guard cells of allium cepa. Tetrahedron Lett. 1992, 33, 1213–1216. [Google Scholar] [CrossRef]
- Rommel, A.; Wrolstad, R.E. Composition of flavonols in red raspberry juice as influenced by cultivar, processing, and environmental factors. J. Agric. Food Chem. 1993, 41, 1941–1950. [Google Scholar] [CrossRef]
- Lin, L.-Z.; Sun, J.; Chen, P.; Harnly, J. UHPLC-PDA-ESI/HRMS/MS(n) analysis of anthocyanins, flavonol glycosides, and hydroxycinnamic acid derivatives in red mustard greens (Brassica juncea Coss variety). J. Agric. Food Chem. 2011, 59, 12059–12072. [Google Scholar] [CrossRef] [PubMed]
- Olsen, H.; Aaby, K.; Borge, G.I.A. Characterization and Quantification of Flavonoids and Hydroxycinnamic Acids in Curly Kale (Brassica oleracea L. Convar. acephala Var. sabellica) by HPLC-DAD-ESI-MSn. J. Agric. Food Chem. 2009, 57, 2816–2825. [Google Scholar] [CrossRef]
- Sasidharan, S.; Chen, Y.; Saravanan, D.; Sundram, K.M.; Yoga Latha, L. Extraction, Isolation and Characterization of Bioactive Compounds from Plants’ Extracts. Afr. J. Tradit. Complement. Altern. Med. 2010, 8, 1–10. [Google Scholar] [CrossRef]
- Coskun, O. Separation techniques: Chromatography. North. Clin. Istanb. 2016, 3, 156–160. [Google Scholar]
- Kovinich, N.; Kayanja, G.; Chanoca, A.; Riedl, K.; Otegui, M.S.; Grotewold, E. Not all anthocyanins are born equal: Distinct patterns induced by stress in Arabidopsis. Planta 2014, 240, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Liquid Chromatography. Available online: https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Supplemental_Modules_(Analytical_Chemistry)/Instrumental_Analysis/Chromatography/Liquid_Chromatography (accessed on 7 July 2019).
- Lachman, J.; Proněk, D.; Hejtmánková, A.; Dudjak, J.; Pivec, V.; Faitová, K. Total polyphenol and main flavonoid antioxidants in different onion (Allium cepa L.) varieties. Hortic. Sci. 2011, 30, 142–147. [Google Scholar] [CrossRef]
- Carotenuto, A.; De Feo, V.; Fattorusso, E.; Lanzotti, V.; Magno, S.; Cicala, C. The flavonoids of Allium ursinum. Phytochemistry 1996, 41, 531–536. [Google Scholar] [CrossRef]
- Vallejo, F.; Tomás-Barberán, F.A.; Ferreres, F. Characterisation of flavonols in broccoli (Brassica oleracea L. var. italica) by liquid chromatography–UV diode-array detection–electrospray ionisation mass spectrometry. J. Chromatogr. A 2004, 1054, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef]
- Hollman, P.C.; van Trijp, J.M.; Buysman, M.N.; van der Gaag, M.S.; Mengelers, M.J.; de Vries, J.H.; Katan, M.B. Relative bioavailability of the antioxidant flavonoid quercetin from various foods in man. FEBS Lett. 1997, 418, 152–156. [Google Scholar] [CrossRef]
- Lu, X.; Ross, C.F.; Powers, J.R.; Rasco, B.A. Determination of Quercetins in Onion (Allium cepa) Using Infrared Spectroscopy. J. Agric. Food Chem. 2011, 59, 6376–6382. [Google Scholar] [CrossRef] [PubMed]
- De Vries, J.H.; Hollman, P.C.; Van Amersfoort, I.; Olthof, M.R.; Katan, M.B. Red Wine Is a Poor Source of Bioavailable Flavonols in Men. J. Nutr. 2001, 131, 745–748. [Google Scholar] [CrossRef]
- Day, A.J.; DuPont, M.S.; Ridley, S.; Rhodes, M.; Rhodes, M.J.C.; Morgan, M.R.A.; Williamson, G. Deglycosylation of flavonoid and isoflavonoid glycosides by human small intestine and liver β-glucosidase activity. FEBS Lett. 1998, 436, 71–75. [Google Scholar] [CrossRef]
- Thilakarathna, S.H.; Rupasinghe, H.P.V. Flavonoid Bioavailability and Attempts for Bioavailability Enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef]
- Walgren, R.A.; Lin, J.T.; Kinne, R.K.; Walle, T. Cellular uptake of dietary flavonoid quercetin 4′-beta-glucoside by sodium-dependent glucose transporter SGLT1. J. Pharmacol. Exp. Ther. 2000, 294, 837–843. [Google Scholar] [PubMed]
- Hollman, P.C.; de Vries, J.H.; van Leeuwen, S.D.; Mengelers, M.J.; Katan, M.B. Absorption of dietary quercetin glycosides and quercetin in healthy ileostomy volunteers. Am. J. Clin. Nutr. 1995, 62, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.E.; Kall, M.; Justesen, U.; Schou, A.; Dragsted, L.O. Human absorption and excretion of flavonoids after broccoli consumption. Cancer Lett. 1997, 114, 173–174. [Google Scholar] [CrossRef]
- DuPont, M.S.; Day, A.J.; Bennett, R.N.; Mellon, F.A.; Kroon, P.A. Absorption of kaempferol from endive, a source of kaempferol-3-glucuronide, in humans. Eur. J. Clin. Nutr. 2004, 58, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Block, E.; Naganathan, S.; Putman, D.; Zhao, S.H. Allium chemistry: HPLC analysis of thiosulfinates from onion, garlic, wild garlic (ramsoms), leek, scallion, shallot, elephant (great-headed) garlic, chive, and Chinese chive. Uniquely high allyl to methyl ratios in some garlic samples. J. Agric. Food Chem. 1992, 40, 2418–2430. [Google Scholar] [CrossRef]
- Ried, K. Garlic Lowers Blood Pressure in Hypertensive Individuals, Regulates Serum Cholesterol, and Stimulates Immunity: An Updated Meta-analysis and Review. J. Nutr. 2016, 146, 389S–396S. [Google Scholar] [CrossRef] [PubMed]
- Oszmiański, J.; Kolniak-Ostek, J.; Wojdyło, A. Characterization and Content of Flavonol Derivatives of Allium ursinum L. Plant. J. Agric. Food Chem. 2013, 61, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Geleijnse, J.M.; Launer, L.J.; Van der Kuip, D.A.M.; Hofman, A.; Witteman, J.C.M. Inverse association of tea and flavonoid intakes with incident myocardial infarction: The Rotterdam Study. Am. J. Clin. Nutr. 2002, 75, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Rexrode, K.M.; Hu, F.; Albert, C.M.; Chae, C.U.; Rimm, E.B.; Stampfer, M.J.; Manson, J.E. Dietary Intakes of Flavonols and Flavones and Coronary Heart Disease in US Women. Am. J. Epidemiol. 2007, 165, 1305–1313. [Google Scholar] [CrossRef] [PubMed]
- Rabeta, M.S.; Lin, S.P. Effects of Different Drying Methods on the Antioxidant Activities of Leaves and Berries of Cayratia trifolia. Sains Malays. 2015, 44, 275–280. [Google Scholar] [CrossRef]
- Kovinich, N.; Saleem, A.; Arnason, J.T.; Miki, B. Combined analysis of transcriptome and metabolite data reveals extensive differences between black and brown nearly-isogenic soybean (Glycine max) seed coats enabling the identification of pigment isogenes. BMC Genomics 2011, 12, 381. [Google Scholar] [CrossRef] [PubMed]
- Saldanha, L.L.; Vilegas, W.; Dokkedal, A.L. Characterization of flavonoids and phenolic acids in Myrcia bella Cambess. using FIA-ESI-IT-MS(n) and HPLC-PAD-ESI-IT-MS combined with NMR. Mol. Basel Switz. 2013, 18, 8402–8416. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
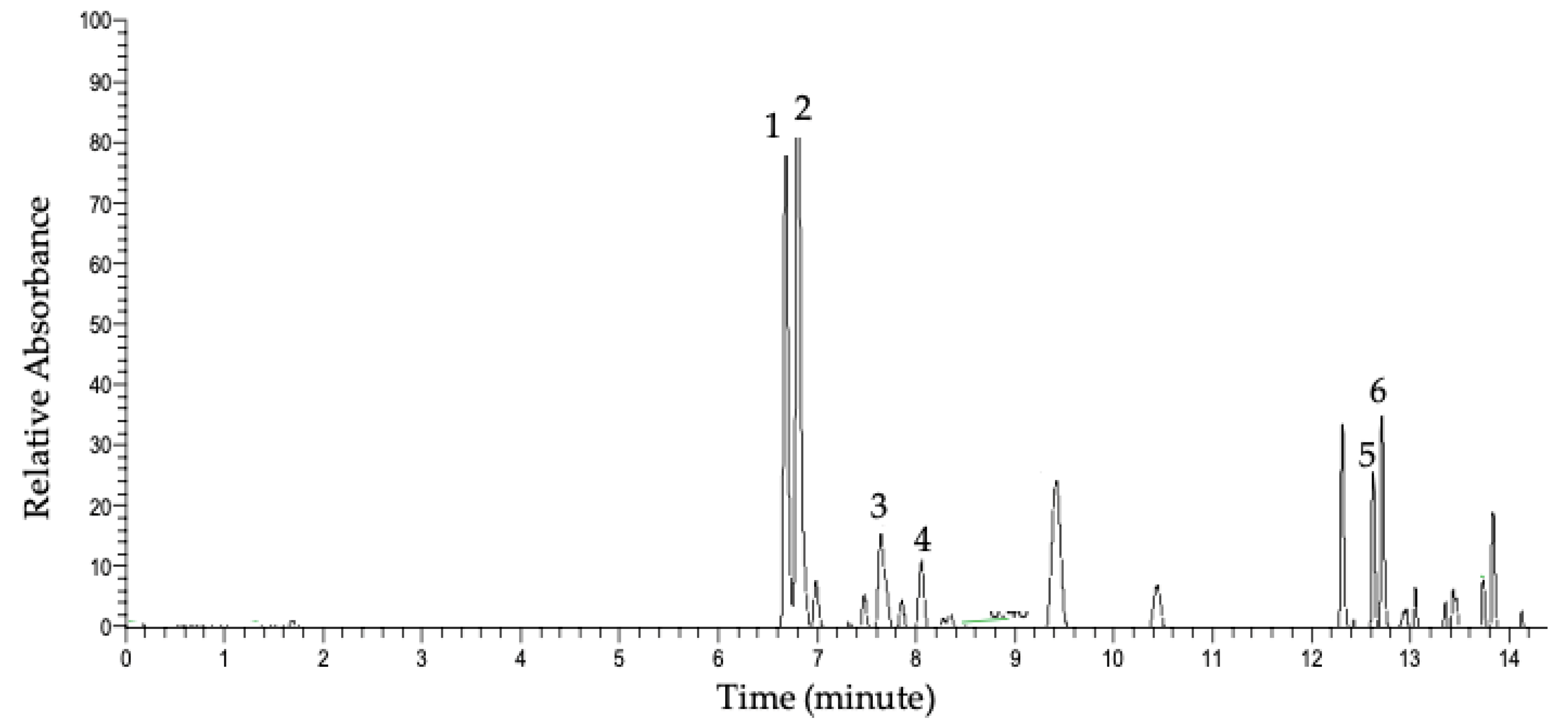
| Peak | Rt (minute) | [M-H]- (m/z) | MS/MS (m/z) | Molecular Weight (g/mol) | Molecular Formula | Compound | Parts | Ref * |
|---|---|---|---|---|---|---|---|---|
| 1 | 6.63 | 801.1719 | 301.0352 625.1405 463.0879 300.0274 343.0454 178.9976 | 802 | C33H38O23 | Quercetin sophoroside glucuronide | Leaf, stem | [22] |
| 2 | 6.77 | 785.1769 | 285.0405 446.0851 609.1459 447.0921 283.0246 255.0296 113.0231 300.0271 489.1038 | 786 | C33H38O22 | Kaempferol sophoroside glucuronide | Leaf, stem | [22] |
| 3 | 7.68 | 769.1817 | 285.0397 284.0324 593.1509 | 770 | C33H38O21 | Kaempferol rutinoside glucuronide | Leaf | [23] |
| 4 | 8.02 | 639.1188 | 301.0344 463.0872 300.0274 271.0247 | 640 | C27H28O18 | Quercetin hexoside glucuronide | Leaf | [24] |
| 5 | 12.65 | 625.1398 | 301.0352 300.0274 463.0890 151.0030 178.9976 276.0706 | 626 | C27H30O17 | Quercetin-sophoroside | Leaf | [25,26] |
| 6 | 12.72 | 609.1443 | 285.0404 283.0246 446.0842 255.0296 489.1040 | 610 | C27H30O16 | Kaempferol-sophoroside | Leaf | [25,27] |
| Compound | Whole (mg/g) | Leaves (mg/g) | Stem (mg/g) | Bulb (mg/g) |
|---|---|---|---|---|
| Quercetin | 0.5972 ± 0.235 | 3.582 ± 1.06 | 0.0112 ± 0.003 | - |
| -sophoroside glucuronide | 0.4706 ± 0.137 | 2.408 ± 0.130 | 0.0112 ± 0.003 | - |
| -sophoroside | 0.0729 ± 0.032 | 0.4611 ± 0.046 | - | - |
| - hexoside glucuronide | 0.0536 ± 0.020 | 0.7132 ± 0.390 | - | - |
| Kaempferol | 0.3792 ± 0.130 | 2.323 ± 0.787 | 0.0079 ± 0.001 | - |
| - sophoroside glucuronide | 0.2763 ± 0.077 | 1.682 ± 0.106 | 0.0079 ± 0.001 | - |
| -sophoroside | 0.0503 ± 0.019 | 0.3565 ± 0.028 | - | - |
| - rutinoside glucuronide | 0.0526 ± 0.013 | 0.2840 ± 0.016 | - | - |
| Method | Solvent A | Solvent B |
|---|---|---|
| Flow Rate (µL/min) | ||
| 300 500 | 0 min, 90%; 2 min, 75%; 7 min, 65%; 9 min, 0%; 11 min, 0%; 12 min, 90%; 15 min, 90% | 0 min, 10%; 2 min, 25%; 7 min, 35%; 9 min, 100%; 11 min, 100%; 12 min, 10%; 15 min, 10% |
| Gradient elution time (minutes) | ||
| 15 | 0 min, 90%; 2 min, 75%; 7 min, 65%; 9 min, 0%; 11 min, 0%; 12 min, 90%; 15 min, 90% | 0 min, 10%; 2 min, 25%; 7 min, 35%; 9 min, 100%; 11 min, 100%; 12 min, 10%; 15 min, 10% |
| 21 | 0 min, 90%; 6 min, 90%; 8 min, 75%; 13 min, 65%; 15 min, 0%; 17 min, 0%; 18 min, 90%; 21 min, 90% | 0 min, 10%; 6 min, 10%; 8 min, 25%; 13 min, 35%; 15 min, 100%; 17 min, 100%; 18 min, 10%; 21 min, 10% |
| 24 | 0 min, 98%; 2 min, 98%; 3 min, 90%; 9 min, 90%; 11 min, 75%; 16 min, 65%; 18 min, 0%; 20 min, 0%; 21 min, 98%; 24 min, 98% | 0 min, 2%; 2 min, 2%; 3 min, 10%; 9 min, 10%; 11 min, 25%; 16 min, 35%; 18 min, 100%; 20 min, 100%; 21 min, 2%; 24 min, 2% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dabeek, W.M.; Kovinich, N.; Walsh, C.; Ventura Marra, M. Characterization and Quantification of Major Flavonol Glycosides in Ramps (Allium tricoccum). Molecules 2019, 24, 3281. https://doi.org/10.3390/molecules24183281
Dabeek WM, Kovinich N, Walsh C, Ventura Marra M. Characterization and Quantification of Major Flavonol Glycosides in Ramps (Allium tricoccum). Molecules. 2019; 24(18):3281. https://doi.org/10.3390/molecules24183281
Chicago/Turabian StyleDabeek, Wijdan M., Nik Kovinich, Callee Walsh, and Melissa Ventura Marra. 2019. "Characterization and Quantification of Major Flavonol Glycosides in Ramps (Allium tricoccum)" Molecules 24, no. 18: 3281. https://doi.org/10.3390/molecules24183281
APA StyleDabeek, W. M., Kovinich, N., Walsh, C., & Ventura Marra, M. (2019). Characterization and Quantification of Major Flavonol Glycosides in Ramps (Allium tricoccum). Molecules, 24(18), 3281. https://doi.org/10.3390/molecules24183281





