Bioactive Constituents from the Roots of Eurycoma longifolia
Abstract
:1. Introduction
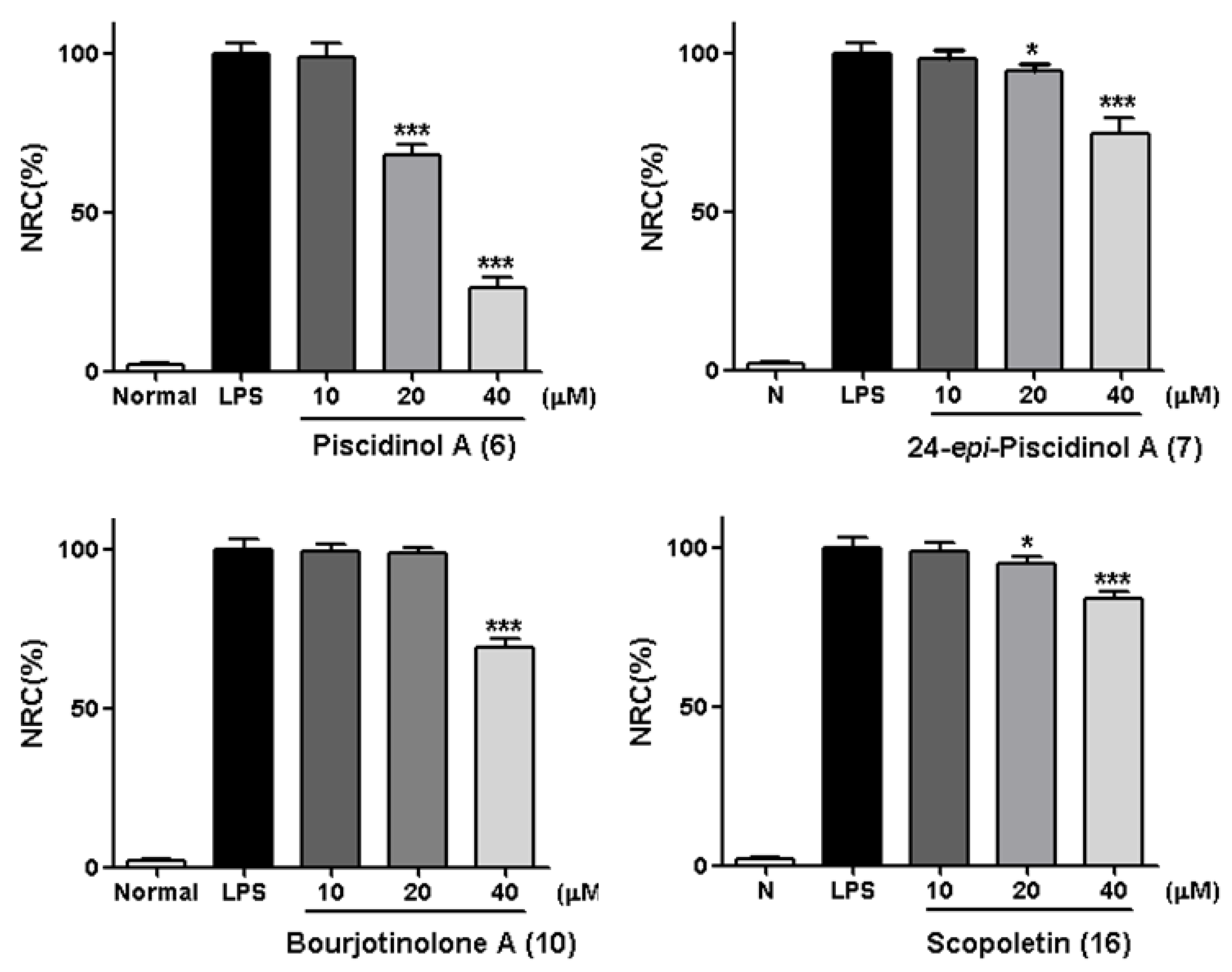
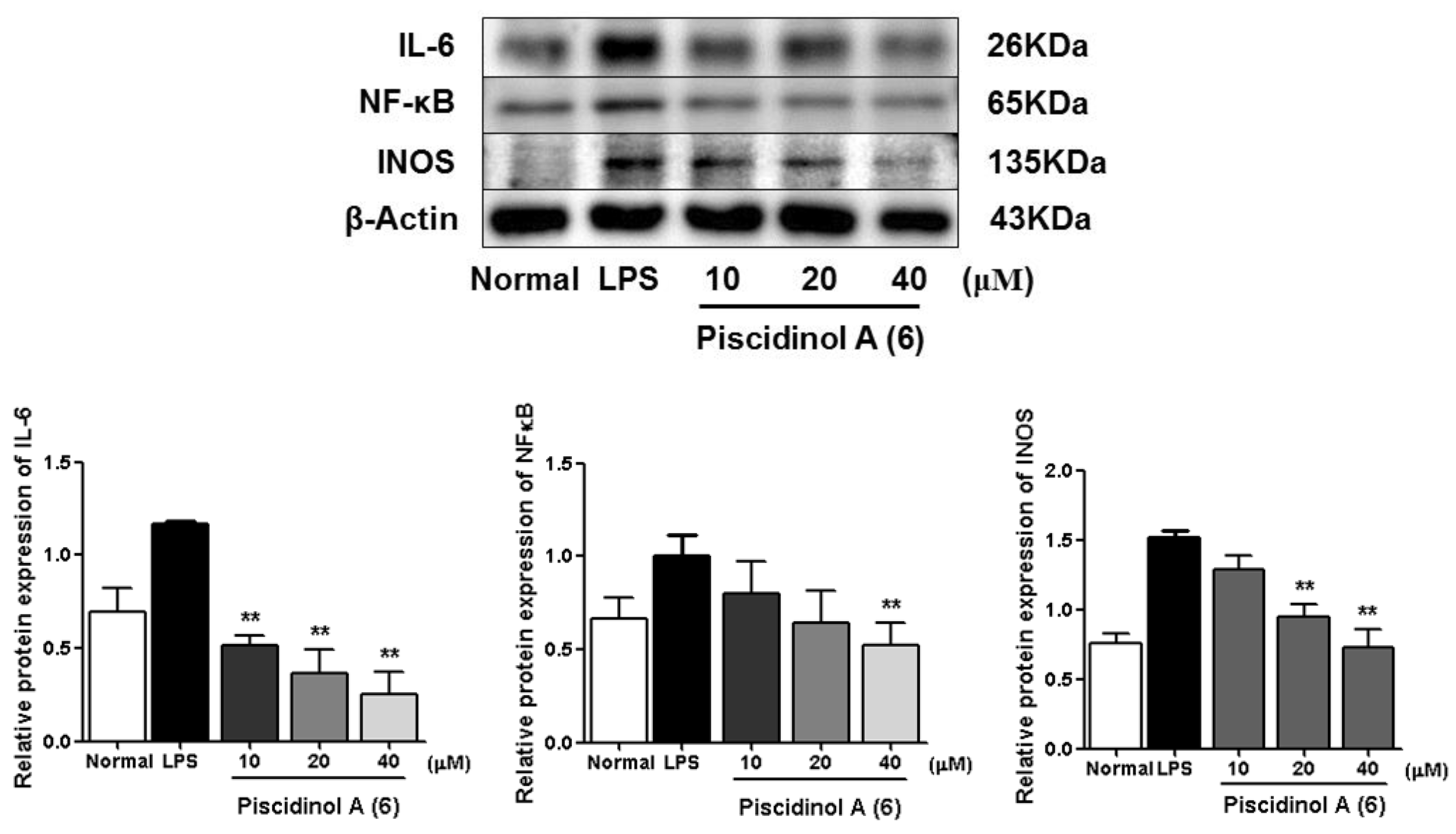
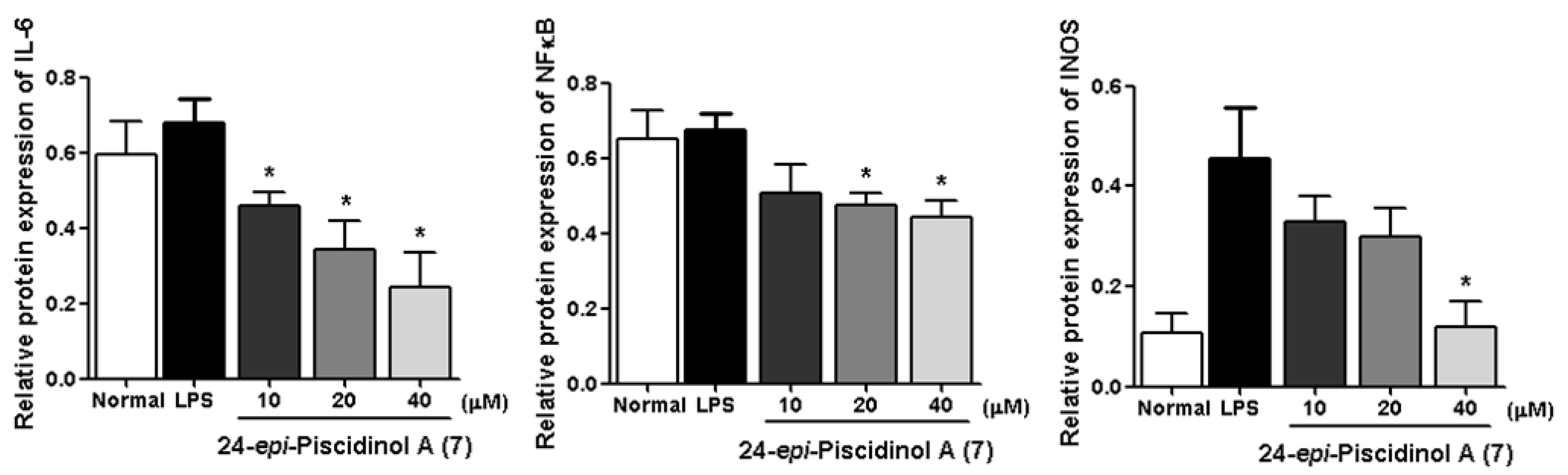
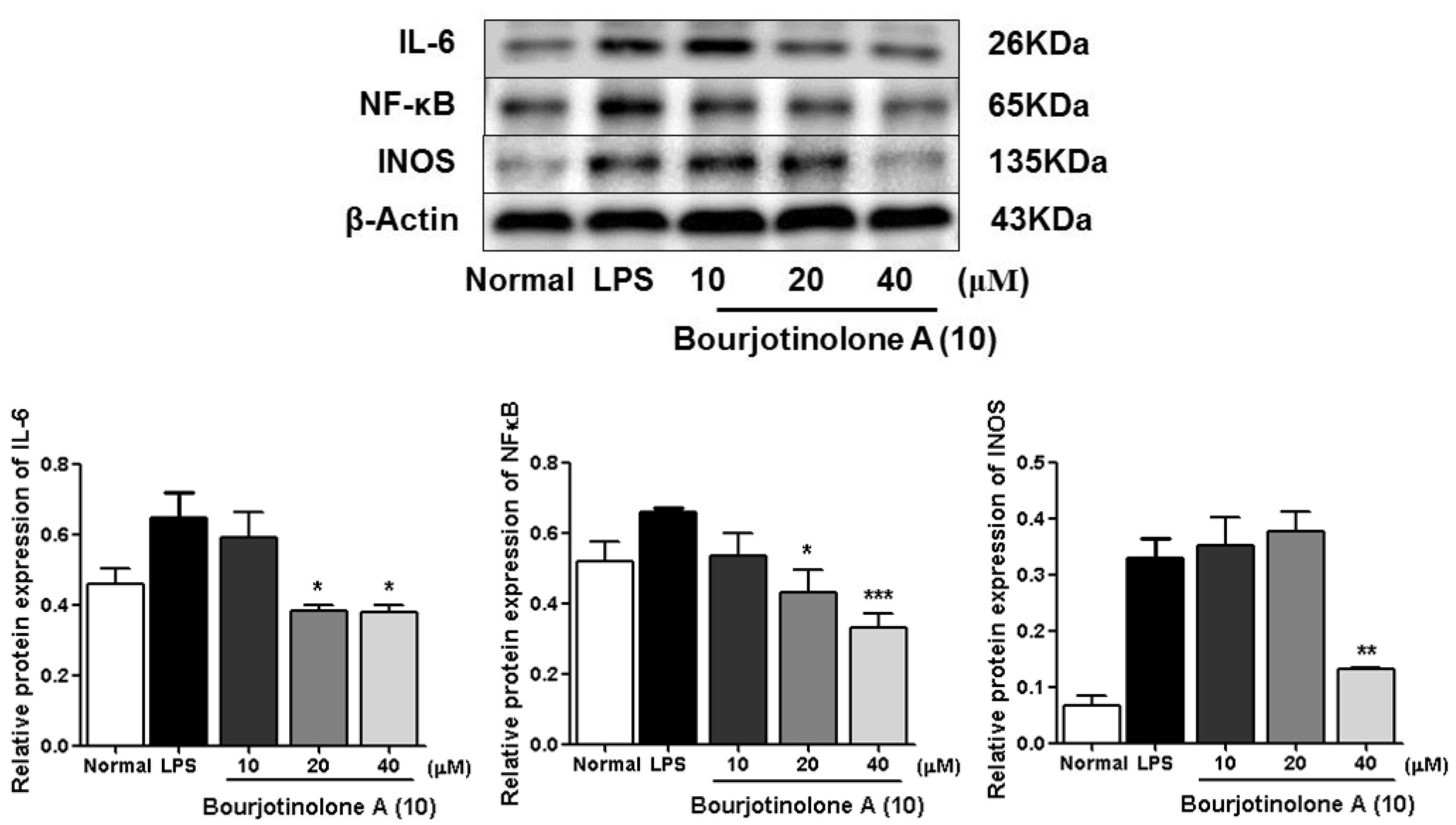
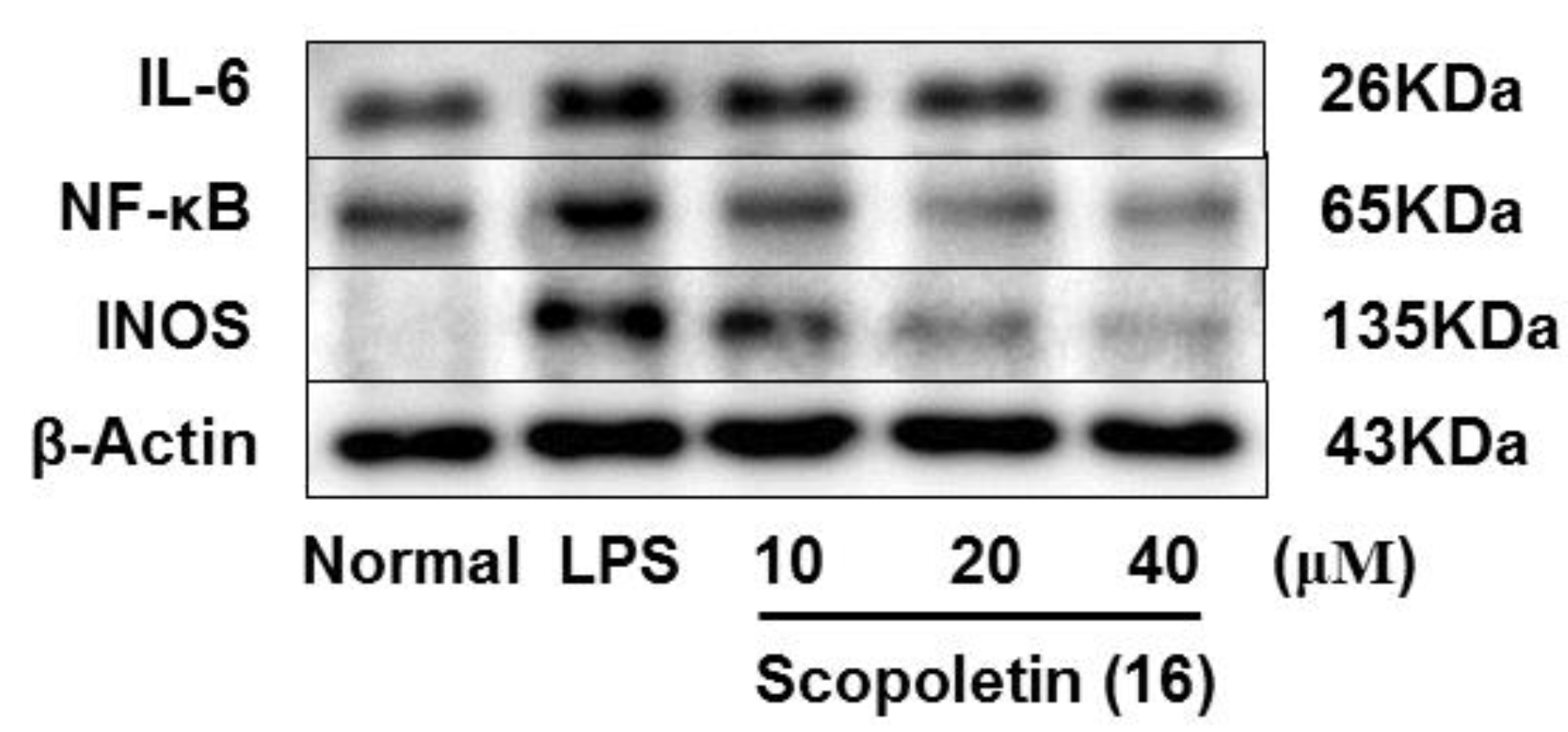
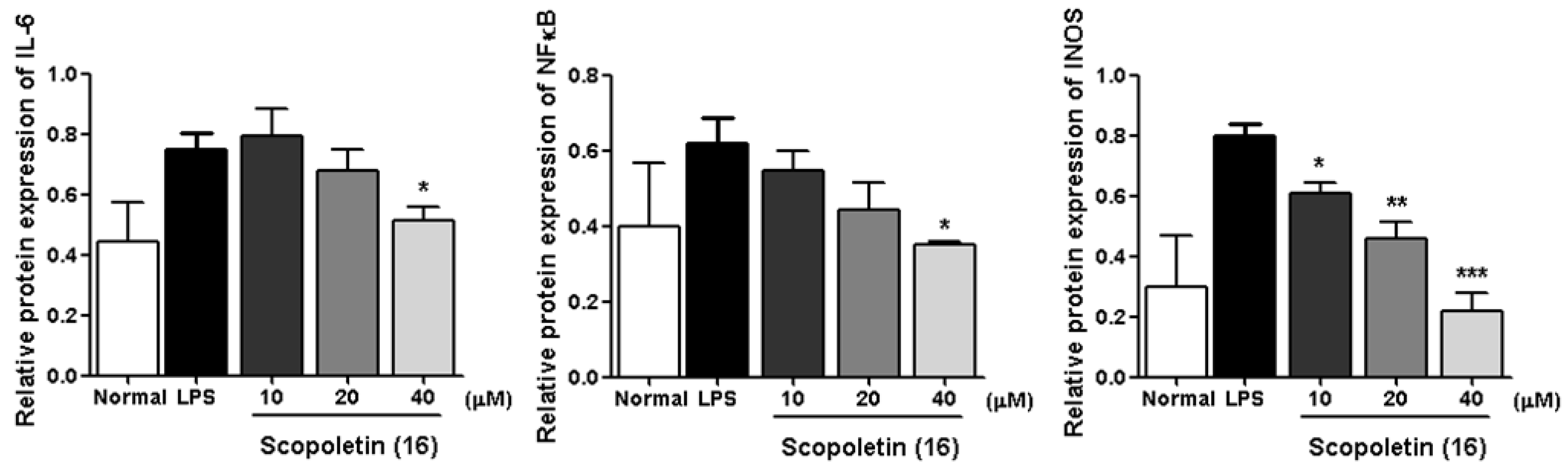
2. Results and Discussion
3. Experimental
3.1. Experimental Procedures for Phytochmistry Study
3.1.1. General Experimental Procedures
3.1.2. Plant Material
3.1.3. Extraction and Isolation
3.2. Experimental Procedures for Bioassay
3.2.1. General Experimental Procedures
3.2.2. Cell Culture
3.2.3. Cell Viability Assay
3.2.4. Measurement of NO levels
3.2.5. Western Blot Analysis
3.2.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ahujaa, A.; Kim, M.Y.; Cho, J.Y. Protium javanicum Burm. methanol extract attenuates LPS-induced inflammatory activities in macrophage-like RAW264.7 cells. Evid. Based Complement Alternat. Med. 2019, 2019, 2910278. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Mao, X. Culture skill and experience of RAW264.7*. Xiandai Shengwu Yixue Jinzhan 2012, 12, 4358–4359. [Google Scholar]
- Liu, H.; Yao, Y. Advances in cross-talk of cellular signalling pathways associated with inflammatory response. Zhongguo Bingli Shengli Zazhi 2005, 21, 1607–1613, 1627. [Google Scholar]
- Ling, M.; He, C.; Gao, J.; Wang, H. Research progresses on Eurycoma longifolia. Guangdong Linye Keji 2013, 29, 66–73. [Google Scholar]
- Bhat, R.; Karim, A.A. Tongkat Ali (Eurycoma longifolia Jack): A review on its ethnobotany and pharmacological importance. Fitoterapia 2010, 81, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Xiao, X.; Guo, W.; Zhang, T. Advances in studies on chemistry, pharmacological effect, and harmacokinetics of Eurycoma longifolia. Chin. Herb. Med. 2011, 3, 186–195. [Google Scholar]
- Muhammad, I.; Samoylenko, V. Antimalarial quassinoids: Past, present and future. Expert. Opin. Drug Discov. 2007, 2, 1065–1084. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Nhiem, N.X.; Kiem, P.V.; Minh, C.V.; Tai, B.H.; Kim, N.; Yoo, H.H.; Song, J.H.; Ko, H.J.; Kim, S.H. Five new quassinoids and cytotoxic constituents from the roots of Eurycoma longifolia. Bioorg. Med. Chem. Lett. 2014, 24, 3835–3840. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.C.; Shi, L.S.; Damu, A.G.; Su, C.R.; Huang, C.H.; Ke, C.H.; Wu, J.B.; Lin, A.J.; Bastow, K.F.; Lee, K.H.; et al. Cytotoxic and antimalarial β-carboline alkaloids from the roots of Eurycoma longifolia. J. Nat. Prod. 2003, 66, 1324–1327. [Google Scholar] [CrossRef]
- Chan, K.L.; O’Neill, M.J.; Phillipson, J.D.; Warhurst, D.C. Plants as sources of antimalarial drugs. Part 3. Eurycoma longifolia. Planta Med. 1986, 50, 105–107. [Google Scholar] [CrossRef]
- Ngoc, P.B.; Pham, T.B.; Nguyen, D.; Tran, T.T.; Chu, H.H.; Chau, V.M.; Lee, J.H.; Nguyen, T.D. A new anti-inflammatory β-carboline alkaloid from the hairy-root cultures of Eurycoma longifolia. Nat. Prod. Res. 2016, 30, 1360–1365. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.M.; Woo, S.U.; Choi, M.S.; Park, Y.N.; Kim, S.H.; Yim, H.; Yoo, H.H. Antiinflammatory and analgesic effects of Eurycoma longifolia extracts. Arch. Pharm. Res. 2016, 39, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, Z.; Yao, J.; Wang, J.; Zhao, M.; Zhang, S. Chemical constituents of the branches of Ailanthus altissima Swingle. Linchan Huaxue Yu Gongye 2013, 33, 121–127. [Google Scholar]
- Zhang, F.; Cen, J.; Li, Q.; Wang, H. Chemical constituents from Toona ciliata var. henryi and their anti-inflammatory activities. Zhongcaoyao 2014, 45, 755–759. [Google Scholar]
- Puripattanavong, J.; Weber, S.; Brecht, V.; Frahm, A.W. Phytochemical investigation of Aglaia andamanica. Planta Med. 2000, 66, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.N.; Fan, C.Q.; Yin, S.; Lin, L.P.; Ding, J.; Yue, J.M. Cytotoxic terpenoids from Turraea pubescens. Helv. Chim. Acta 2008, 91, 510–518. [Google Scholar] [CrossRef]
- Zheng, R.; Ya, J.; Wang, W.; Yang, H.; Zhang, Q.; Zhang, X.; Ye, W. Chemical studies on roots of Ficus hirta. Zhongguo Yaowu Huaxue Zazhi 2013, 38, 3696–3701. [Google Scholar]
- Xu, W.; Zhou, G.; Dai, Y.; Yao, X. Chemical constituents in stems of Schima superba. Zhongcaoyao 2010, 41, 863–866. [Google Scholar]
- Kuo, P.C.; Damu, A.G.; Lee, K.H.; Wu, T.S. Cytotoxic and antimalarial constituents from the roots of Eurycoma longifolia. Bioorg. Med. Chem. 2004, 12, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Mei, W.; Dai, H.; Wu, D. Phenolic constituents from Ailanthus fordii Nooteboom. Redai Yaredai Zhiwu Xuebao 2006, 14, 413–416. [Google Scholar]
- Zhang, D.; Li, Y.; Yu, S. Glycosides from the stems of Photinia parvifolia. Tianran Chanwu Yanjiu Yu Kaifa 2004, 16, 496–499. [Google Scholar]
- Jiang, C.; Mu, S.; Deng, B.; Ge, Y.; Zhang, J.; Hao, X. Isolation and identification of chemical constituents from aerial part of Euphorbia chrysocoma Lévi. et Vant (II). Shenyang Yaoke Daxue Xuebao 2010, 27, 354–356. [Google Scholar]
- Qu, L.; Wang, J.; Ruan, J.; Yao, X.; Huang, P.; Wang, Y.; Yu, H.; Han, L.; Zhang, Y.; Wang, T. Spirostane-type saponins obtained from Yucca schidigera. Molecules 2018, 23, 167. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Yanagisawa, M. Systhesis, separation and NMR spectral analysis of methyl apiofuranosides. Carbohyd. Res. 1998, 313, 189–192. [Google Scholar] [CrossRef]
- Zhang, Y.; Chao, L.; Ruan, J.; Zheng, C.; Yu, H.; Han, L.; Wang, T. Bioactive constituents from the rhizomes of Dioscorea septemloba Thunb. Fitoterapia 2016, 115, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, J.X.; Wang, Y.H.; Su, X.L.; Mei, R.Q.; Yang, J.; Kong, Y.; Long, C.L. Neolignans from Selaginella moellendorffii. Nat. Prod. Bioprospect. 2016, 6, 161–166. [Google Scholar] [CrossRef]
- He, W.; Li, Y.; Liu, M.; Yu, H.; Chen, Q.; Chen, Y.; Ruan, J.; Ding, Z.; Zhang, Y.; Wang, T. Citrus aurantium L. and its flavonoids regulate TNBS-induced inflammatory bowel disease through anti-inflammation and suppressing isolated jejunum contraction. Inter. Int. J. Mol. Sci. 2018, 19, 3057. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Abdallah, H.M.; El-Halawany, A.M.; Esmat, A.; Mohamed, G.A. Thiotagetin B and tagetannins A and B, new acetylenic thiophene and digalloyl glucose derivatives from Tagetes minuta and evaluation of their in vitro antioxidative and anti-inflammatory activity. Fitoterapia 2018, 125, 78–88. [Google Scholar] [CrossRef]
- Sundaram, C.S.; Rao, U.S.M.; Simbak, N. Regulatory efficacy of scopoletin, a biocoumarin on aortic oxido lipidemic stress through antioxidant potency as well as suppression of mRNA expression of inos gene in hypercholesterolemic rats. Pharm. Lett. 2015, 7, 57–67. [Google Scholar]
- Chen, H.; Ma, S.G.; Fang, Z.F.; Bai, J.; Yu, S.S.; Chen, X.G.; Hou, Q.; Yuan, S.P.; Chen, X. Tirucallane triterpenoids from the stems of Brucea mollis. Chem. Biodiver. 2013, 10, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Raju, R.; Singh, A.; Reddell, P.; Munch, G. Anti-inflammatory activity of prenyl and geranyloxy furanocoumarins from Citrus garrawayi (Rutaceae). Phytochem. Lett. 2018, 27, 197–202. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–16 are available from the authors. |
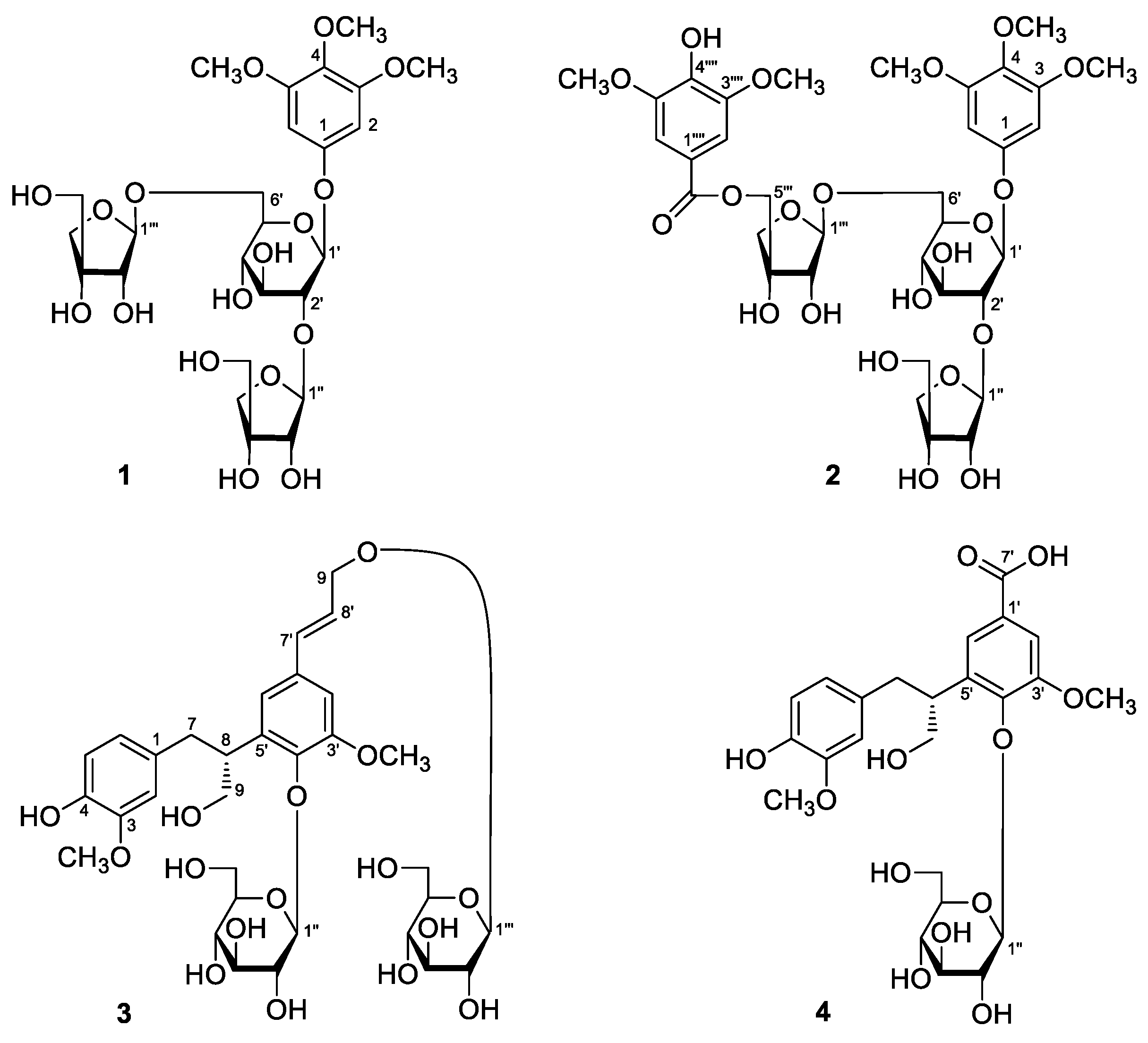
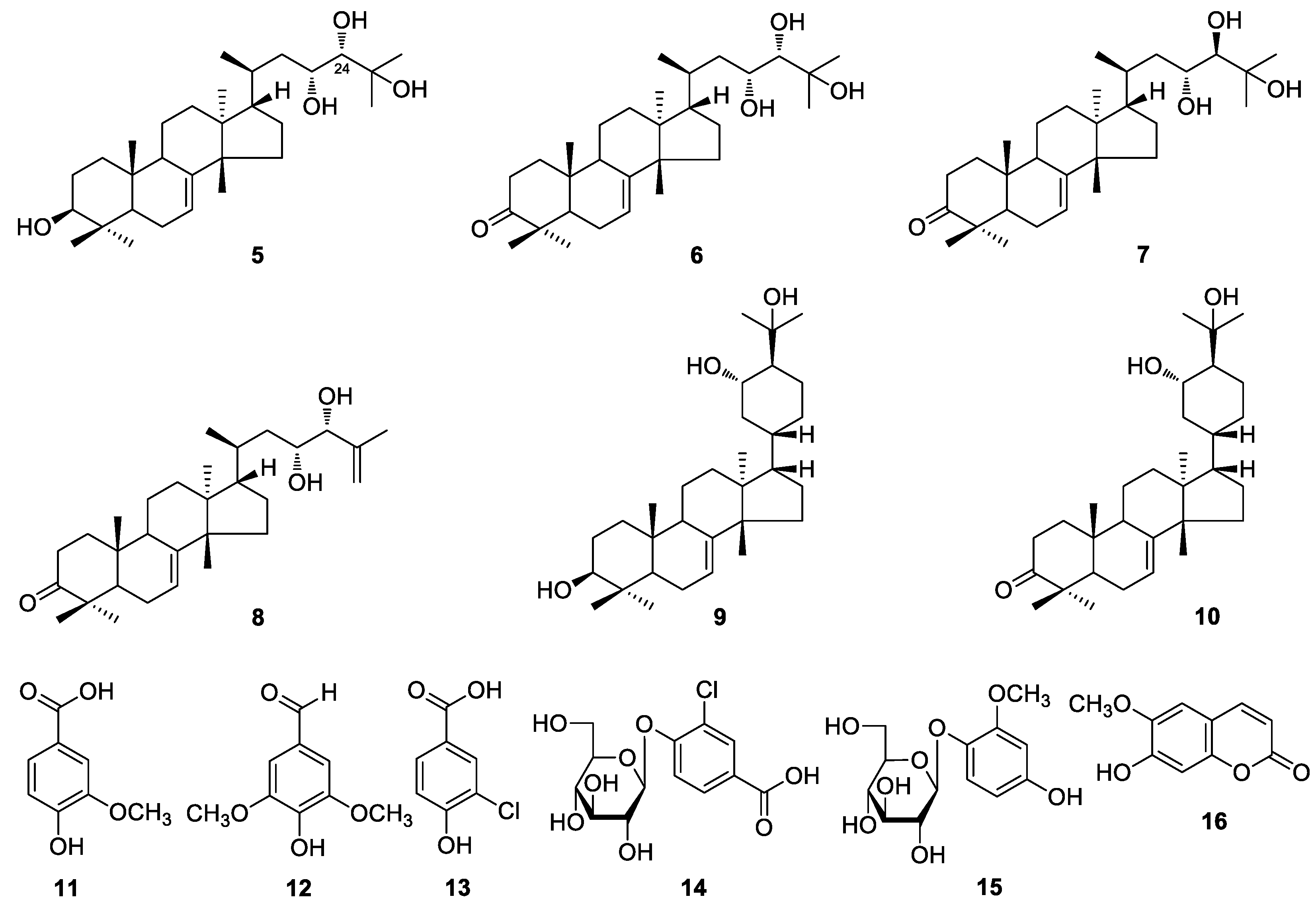
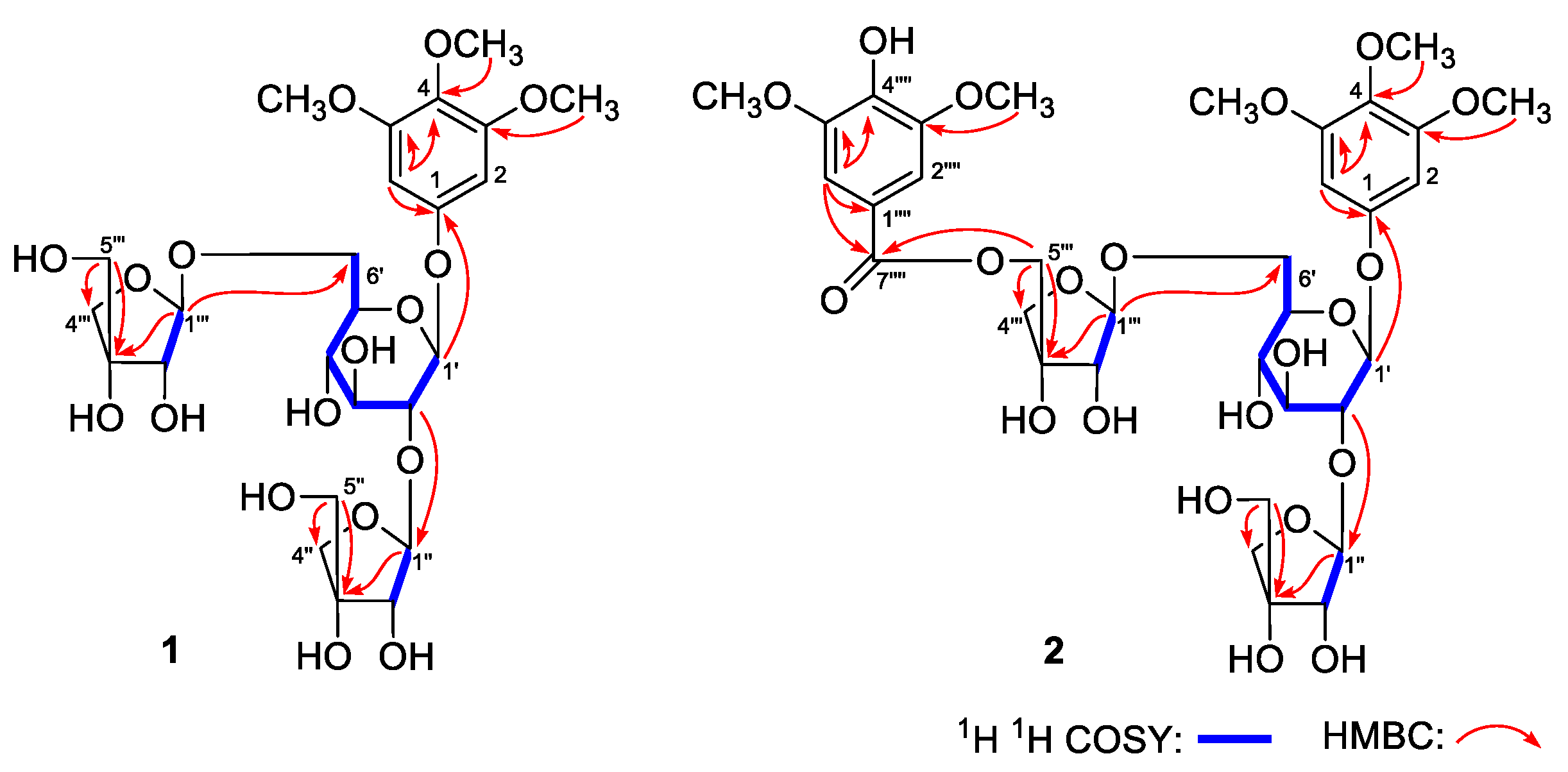
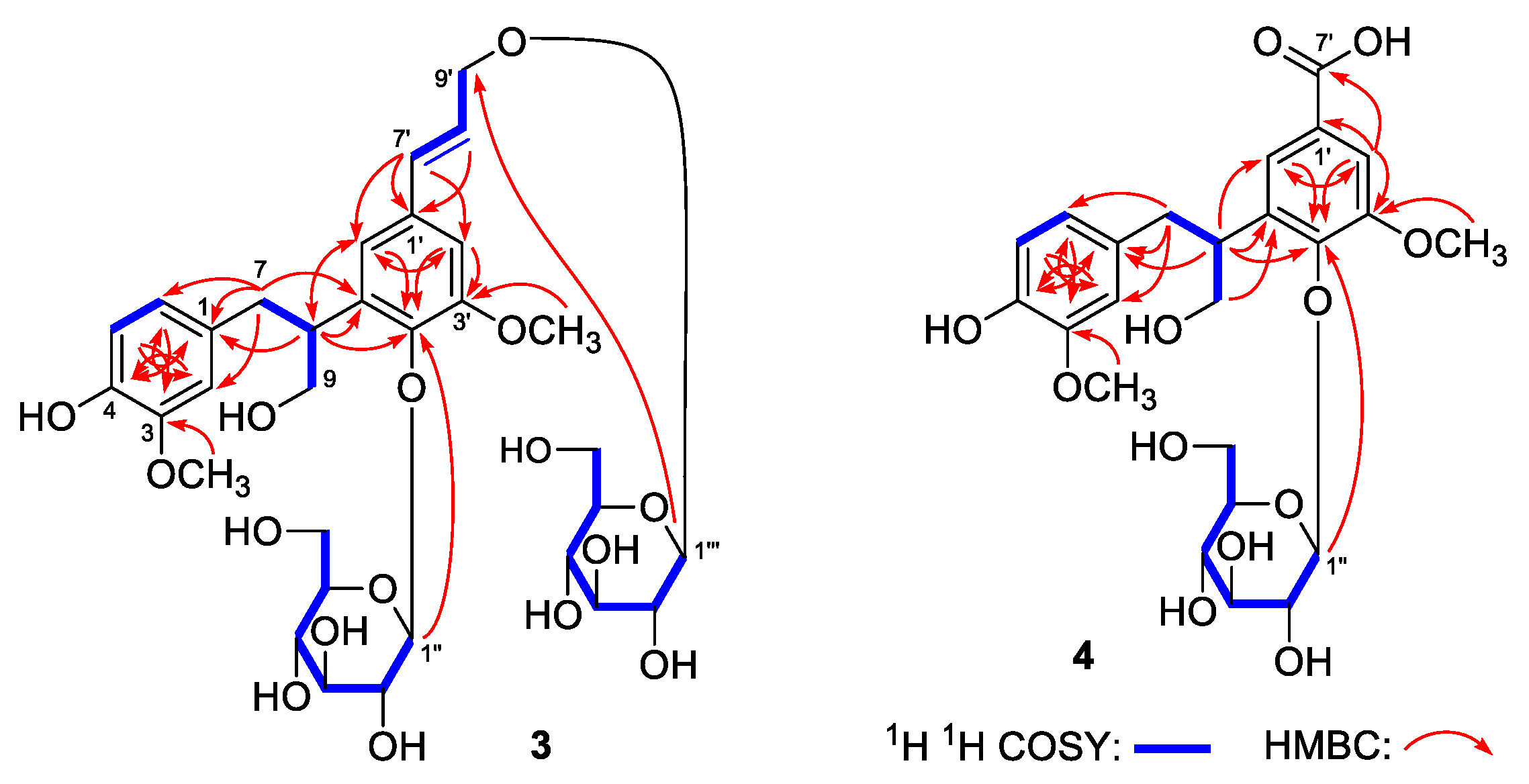
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 155.5 | - | 4’’ | 75.9 | 4.47 (d, 9.6) |
| 2,6 | 95.9 | 6.94 (s) | 4.93 (d, 9.6) | ||
| 3,5 | 154.4 | - | 5’’ | 66.6 | 4.25 (d, 11.4) |
| 4 | 134.3 | - | 4.28 (d, 11.4) | ||
| 1’ | 101.9 | 5.38 (d, 7.8) | 1’’’ | 111.0 | 5.66 (d, 2.4) |
| 2’ | 77.1 | 4.50 (dd, 8.4, 7.8) | 2’’’ | 77.6 | 4.69 (d, 2.4) |
| 3’ | 79.0 | 4.31 (dd, 9.0, 8.4) | 3’’’ | 80.3 | - |
| 4’ | 71.8 | 3.95 (dd, 9.0, 9.0) | 4’’’ | 74.9 | 4.31 (d, 9.6) |
| 5’ | 77.2 | 4.18 (m) | 4.55 (d, 9.6) | ||
| 6’ | 69.0 | 4.03 (dd, 10.8, 7.2) | 5’’’ | 65.2 | 4.09 (d, 11.4) |
| 4.78 (br. d, ca. 11) | 4.14 (d, 11.4) | ||||
| 1’’ | 110.5 | 6.60 (br. s) | 3,5-OCH3 | 56.2 | 3.88 (s) |
| 2’’ | 78.0 | 4.77 (br. s) | 4-OCH3 | 60.6 | 3.79 (s) |
| 3’’ | 81.0 | - |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 155.4 | - | 1’’’ | 110.6 | 5.69 (d, 3.0) |
| 2,6 | 96.0 | 6.92 (s) | 2’’’ | 78.6 | 4.62 (d, 3.0) |
| 3,5 | 154.3 | - | 3’’’ | 78.7 | - |
| 4 | 134.1 | - | 4’’’ | 74.8 | 4.39 (d, 9.5) |
| 1’ | 101.9 | 5.40 (d, 8.0) | 4.49 (d, 9.5) | ||
| 2’ | 77.2 | 4.49 (dd, 9.0, 8.0) | 5’’’ | 67.8 | 4.86 (d, 11.5) |
| 3’ | 78.9 | 4.32 (dd, 9.0, 9.0) | 4.90 (d, 11.5) | ||
| 4’ | 71.7 | 3.98 (dd, 9.5, 9.0) | 1’’’’ | 120.1 | - |
| 5’ | 77.1 | 4.17 (m) | 2’’’’ | 108.3 | 7.68 (s) |
| 6’ | 68.9 | 4.06 (dd, 11.5, 7.5) | 3’’’’ | 148.7 | - |
| 4.76 (br. d, ca. 12) | 4’’’’ | 143.0 | - | ||
| 1’’ | 110.5 | 6.57 (br. s) | 5’’’’ | 148.7 | - |
| 2’’ | 78.1 | 4.78 (br. s) | 6’’’’ | 108.3 | 7.68 (s) |
| 3’’ | 81.0 | - | 7’’’’ | 166.7 | - |
| 4’’ | 75.8 | 4.46 (d, 9.5) | 3,5-OCH3 | 56.2 | 3.87 (s) |
| 4.91 (d, 9.5) | 4-OCH3 | 60.6 | 3.79 (s) | ||
| 5’’ | 66.5 | 4.24 (d, 11.0) | 3’’’’,5’’’’-OCH3 | 56.3 | 3.77 (s) |
| 4.27 (d, 11.0) |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 133.2 | - | 9’ | 70.8 | 4.33 (ddd, 13.0, 6.0, 1.0) |
| 2 | 113.8 | 6.57 (d, 1.5) | 4.52 (ddd, 13.0, 6.0, 1.0) | ||
| 3 | 148.4 | - | 1’’ | 105.4 | 4.68 (d, 7.5) |
| 4 | 145.4 | - | 2’’ | 76.0 | 3.46 (dd, 8.0, 7.5) |
| 5 | 115.7 | 6.56 (d, 8.0) | 3’’ | 78.0 | 3.41 (dd,8.0, 8.0) |
| 6 | 122.6 | 6.48 (dd, 8.0, 1.5) | 4’’ | 71.3 | 3.37 (dd, 8.5, 8.5) |
| 7 | 39.2 | 2.73 (dd, 14.0, 9.5) | 5’’ | 77.9 | 3.12 (m) |
| 2.96 (dd, 14.0, 5.5) | 6’’ | 62.5 | 3.66 (m, overlapped) | ||
| 8 | 42.8 | 3.97 (m) | 3.77 (m, overlapped) | ||
| 9 | 66.8 | 3.69 (m, overlapped) | 1’’’ | 103.3 | 4.37 (d, 8.0) |
| 3.76 (m, overlapped) | 2’’’ | 75.2 | 3.24 (dd, 8.5, 8.0) | ||
| 1’ | 135.2 | - | 3’’’ | 78.2 | 3.37 (dd, 8.5, 8.5) |
| 2’ | 109.2 | 6.95 (d, 2.0) | 4’’’ | 71.7 | 3.29 (m, overlapped) |
| 3’ | 153.5 | - | 5’’’ | 78.1 | 3.29 (m, overlapped) |
| 4’ | 145.2 | - | 6’’’ | 62.9 | 3.68 (m, overlapped) |
| 5’ | 139.0 | - | 3.88 (dd, 12.0, 2.0) | ||
| 6’ | 119.4 | 6.92 (d, 2.0) | 3-OCH3 | 56.3 | 3.69 (s) |
| 7’ | 133.7 | 6.65 (br. d, ca. 16.0) | 3’-OCH3 | 56.4 | 3.83 (s) |
| 8’ | 126.4 | 6.30 (dt, 16.0, 6.0) |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 133.1 | - | 4’ | 148.8 | - |
| 2 | 113.8 | 6.60 (d, 1.5) | 5’ | 139.0 | - |
| 3 | 148.5 | - | 6’ | 122.6 | 7.63 (br. s) |
| 4 | 145.4 | - | 7’ | 170.4 | - |
| 5 | 115.8 | 6.57 (d, 8.0) | 1’’ | 105.0 | 4.80 (d, 8.0) |
| 6 | 122.6 | 6.49 (dd, 8.0, 1.5) | 2’’ | 76.0 | 3.48 (dd, 8.0, 8.0) |
| 7 | 39.3 | 2.73 (dd, 14.0, 9.5) | 3’’ | 77.9 | 3.43 (dd, 9.0, 8.0) |
| 2.99 (dd, 14.0, 6.0) | 4’’ | 71.2 | 3.39 (dd, 8.5, 8.5) | ||
| 8 | 43.0 | 3.99 (m) | 5’’ | 78.1 | 3.13 (m) |
| 9 | 66.7 | 3.71 (m) | 6’’ | 62.4 | 3.66 (dd, 12.0, 5.0) |
| 3.79 (m) | 3.75 (dd, 12.0, 2.0) | ||||
| 1’ | 148.5 | - | 3-OCH3 | 56.3 | 3.71 (s) |
| 2’ | 112.5 | 7.49 (br. s) | 3’-OCH3 | 56.4 | 3.85 (s) |
| 3’ | 153.2 | - |
| NO. | NRC (%) | NO. | NRC (%) |
|---|---|---|---|
| Normal | 2.2 ± 0.4 | 8 | 101.3 ± 6.1 |
| Control | 100 ± 3.5 | 9 | 90.3 ± 5.3 * |
| DEX | 82.7 ± 3.1 *** | 10 | 69.0 ± 2.7 *** |
| 1 | 93.6 ± 3.7 * | 11 | 98.5 ± 2.8 |
| 2 | 101.1 ± 3.6 | 12 | 96.3 ± 2.4 |
| 3 | 102.2 ± 4.0 | 13 | 95.1 ± 0.9 |
| 4 | 96.0 ± 1.1 | 14 | 98.2 ± 5.2 |
| 5 | 87.0 ± 3.3 * | 15 | 97.1 ± 1.0 |
| 6 | 26.5 ± 3.0 *** | 16 | 83.9 ± 2.2 *** |
| 7 | 74.9 ± 4.4 *** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruan, J.; Li, Z.; Zhang, Y.; Chen, Y.; Liu, M.; Han, L.; Zhang, Y.; Wang, T. Bioactive Constituents from the Roots of Eurycoma longifolia. Molecules 2019, 24, 3157. https://doi.org/10.3390/molecules24173157
Ruan J, Li Z, Zhang Y, Chen Y, Liu M, Han L, Zhang Y, Wang T. Bioactive Constituents from the Roots of Eurycoma longifolia. Molecules. 2019; 24(17):3157. https://doi.org/10.3390/molecules24173157
Chicago/Turabian StyleRuan, Jingya, Zheng Li, Ying Zhang, Yue Chen, Mengyang Liu, Lifeng Han, Yi Zhang, and Tao Wang. 2019. "Bioactive Constituents from the Roots of Eurycoma longifolia" Molecules 24, no. 17: 3157. https://doi.org/10.3390/molecules24173157
APA StyleRuan, J., Li, Z., Zhang, Y., Chen, Y., Liu, M., Han, L., Zhang, Y., & Wang, T. (2019). Bioactive Constituents from the Roots of Eurycoma longifolia. Molecules, 24(17), 3157. https://doi.org/10.3390/molecules24173157





