Application of Chitosan in Bone and Dental Engineering
Abstract
1. Introduction
2. Results
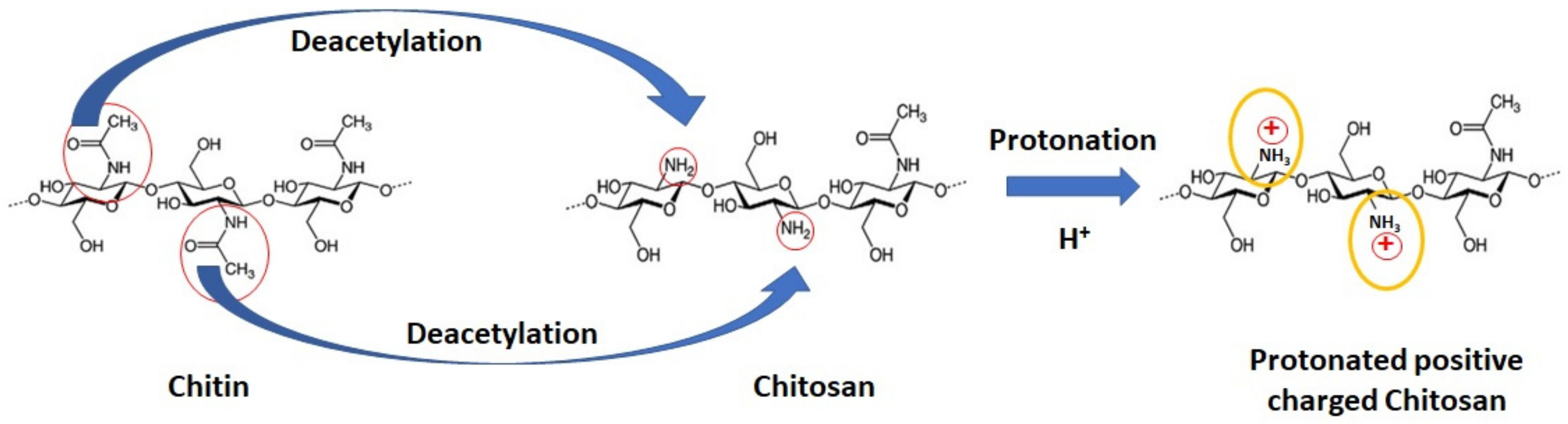
2.1. Chitosan
2.2. Medical and Pharmaceutical Properties of Chitosan
2.2.1. Drug Delivery
2.2.2. Wound Dressings
2.3. Chitosan-Based Scaffold Preparation
2.4. Chitosan-Based Scaffolds for Bone Regeneration
2.5. Chitosan-Based Application for Dental Engineering: The Case of Periodontal Regeneration
2.6. Chitosan-Based Scaffold in Dental-Pulp Regeneration
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vacanti, J.P.; Morse, M.A.; Saltzman, W.M.; Domb, A.J.; Perez-Atayde, A.; Langer, R. Selective cell transplantation using bioabsorbable artificial polymers as matrices. J. Pediatr. Surg. 1988, 23, 3–9. [Google Scholar] [CrossRef]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine. J. 2008, 17, 467–479. [Google Scholar] [CrossRef] [PubMed]
- Loh, Q.L.; Choong, C. Three-Dimensional Scaffolds for Tissue Engineering Applications: Role of Porosity and Pore Size. Tissue Eng. Part B Rev. 2013, 19, 485–502. [Google Scholar] [CrossRef] [PubMed]
- Bellich, B.; D’Agostino, I.; Semeraro, S.; Gamini, A.; Cesàro, A. “The Good, the Bad and the Ugly” of Chitosans. Mar. Drugs 2016, 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Deepthi, S.; Venkatesan, J.; Kim, S.-K.; Bumgardner, J.D.; Jayakumar, R. An overview of chitin or chitosan/nano ceramic composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2016, 93, 1338–1353. [Google Scholar] [CrossRef] [PubMed]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef]
- Aranaz, I.; Harris, R.; Heras, A. Chitosan Amphiphilic Derivatives. Chem. Appl. Curr. Org. Chem. 2010, 308–330. [Google Scholar] [CrossRef]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Madihally, S.V.; Matthew, H.W. Porous chitosan scaffolds for tissue engineering. Biomaterials 1999, 20, 1133–1142. [Google Scholar] [CrossRef]
- Acosta, N.; Jiménez, C.; Borau, V.; Heras, A. Extraction and characterization of chitin from crustaceans. Biomass Bioenergy 1993, 5, 145–153. [Google Scholar] [CrossRef]
- Synowiecki, J.; Al-Khateeb, N.A. Production, properties, and some new applications of chitin and its derivatives. Crit. Rev. Food Sci. Nutr. 2003, 43, 145–171. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.; Wu, L.-Q.; Bentley, W.E.; Ghodssi, R.; Rubloff, G.W.; Culver, J.N.; Payne, G.F. Biofabrication with chitosan. Biomacromolecules 2005, 6, 2881–2894. [Google Scholar] [CrossRef] [PubMed]
- Pavinatto, F.J.; Caseli, L.; Oliveira, O.N. Chitosan in nanostructured thin films. Biomacromolecules 2010, 11, 1897–1908. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Takayama, K.; Machida, Y.; Nagai, T. Characteristics of polyion complexes of chitosan with sodium alginate and sodium polyacrylate. Int. J. Pharm. 1990, 61, 35–41. [Google Scholar] [CrossRef]
- Kean, T.; Thanou, M. Biodegradation, biodistribution and toxicity of chitosan. Adv. Drug Deliv. Rev. 2010, 62, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Baldrick, P. The safety of chitosan as a pharmaceutical excipient. Regul. Toxicol. Pharm. 2010, 56, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Huet, J.; Rucktooa, P.; Clantin, B.; Azarkan, M.; Looze, Y.; Villeret, V.; Wintjens, R. X-ray structure of papaya chitinase reveals the substrate binding mode of glycosyl hydrolase family 19 chitinases. Biochemistry 2008, 47, 8283–8291. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.M.; Hu, W.; Wang, X.D.; Gu, X.S. The controlling biodegradation of chitosan fibers by N-acetylation in vitro and in vivo. J. Mater. Sci Mater. Med. 2007, 18, 2117–2121. [Google Scholar] [CrossRef]
- Xu, J.; McCarthy, S.P.; Gross, R.A.; Kaplan, D.L. Chitosan Film Acylation and Effects on Biodegradability. Macromolecules 1996, 29, 3436–3440. [Google Scholar] [CrossRef]
- Chatelet, C.; Damour, O.; Domard, A. Influence of the degree of acetylation on some biological properties of chitosan films. Biomaterials 2001, 22, 261–268. [Google Scholar] [CrossRef]
- Kadouche, S.; Farhat, M.; Lounici, H.; Fiallo, M.; Sharrock, P.; Mecherri, M.; Hadioui, M. Low Cost Chitosan Biopolymer for Environmental Use Made from Abundant Shrimp Wastes. Waste Biomass Valor. 2017, 8, 401–406. [Google Scholar] [CrossRef]
- Pichyangkura, R. Application of chitin-chitosan from marine by-products in Thailand. In The FFTC-KU Joint Seminar on “Improved Utilization of Fishery By-product as Potential Nutraceuticals and Functional Foods”; Kasetsart University: Bangkok, Thailand, 2014; pp. 190–194. [Google Scholar]
- Roberts, G.A.F. Thirty years of progress in chitin and chitosan. Prog. Chem. Appl. Chitin. Its Deriv. 2018, 13, 7–15. [Google Scholar]
- Tapola, N.S.; Lyyra, M.L.; Kolehmainen, R.M.; Sarkkinen, E.S.; Schauss, A.G. Safety aspects and cholesterol-lowering efficacy of chitosan tablets. J. Am. Coll. Nutr. 2008, 27, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.L.; Tercius, A.; Anglade, M.; White, C.M.; Coleman, C.I. A meta-analysis evaluating the impact of chitosan on serum lipids in hypercholesterolemic patients. Ann. Nutr. Metab. 2009, 55, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Rytwo, G.; Lavi, R.; Rytwo, Y.; Monchase, H.; Dultz, S.; König, T.N. Clarification of olive mill and winery wastewater by means of clay-polymer nanocomposites. Sci. Total Env. 2013, 442, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Rytwo, G. The use of clay-polymer nanocomposites in wastewater pretreatment. Scientific World J. 2012, 2012, 498503. [Google Scholar] [CrossRef]
- He, P.; Davis, S.S.; Illum, L. In vitro evaluation of the mucoadhesive properties of chitosan microspheres. Int. J. Pharm. 1998, 166, 75–88. [Google Scholar] [CrossRef]
- Yeh, T.-H.; Hsu, L.-W.; Tseng, M.T.; Lee, P.-L.; Sonjae, K.; Ho, Y.-C.; Sung, H.-W. Mechanism and consequence of chitosan-mediated reversible epithelial tight junction opening. Biomaterials 2011, 32, 6164–6173. [Google Scholar] [CrossRef]
- Smith, J.; Wood, E.; Dornish, M. Effect of chitosan on epithelial cell tight junctions. Pharm. Res. 2004, 21, 43–49. [Google Scholar] [CrossRef]
- Van der Lubben, I.M.; Kersten, G.; Fretz, M.M.; Beuvery, C.; Coos Verhoef, J.; Junginger, H.E. Chitosan microparticles for mucosal vaccination against diphtheria: Oral and nasal efficacy studies in mice. Vaccine 2003, 21, 1400–1408. [Google Scholar] [CrossRef]
- Slütter, B.; Bal, S.M.; Que, I.; Kaijzel, E.; Löwik, C.; Bouwstra, J.; Jiskoot, W. Antigen-adjuvant nanoconjugates for nasal vaccination: An improvement over the use of nanoparticles? Mol. Pharm. 2010, 7, 2207–2215. [Google Scholar] [CrossRef]
- Bal, S.M.; Slütter, B.; Verheul, R.; Bouwstra, J.A.; Jiskoot, W. Adjuvanted, antigen loaded N-trimethyl chitosan nanoparticles for nasal and intradermal vaccination: Adjuvant-and site-dependent immunogenicity in mice. Eur. J. Pharm. Sci. 2012, 45, 475–481. [Google Scholar] [CrossRef]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.E.; Eccleston, G.M. Wound healing dressings and drug delivery systems: A review. J. Pharm. Sci. 2008, 97, 2892–2923. [Google Scholar] [CrossRef]
- Hurler, J.; Skalko-Basnet, N. Potentials of chitosan-based delivery systems in wound therapy: Bioadhesion study. J. Funct. Biomater. 2012, 3, 37–48. [Google Scholar] [CrossRef]
- Okamoto, Y.; Yano, R.; Miyatake, K.; Tomohiro, I.; Shigemasa, Y.; Minami, S. Effects of chitin and chitosan on blood coagulation. Carbohydr. Polym. 2003, 53, 337–342. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, D.-Y.; Lu, S.-T.; Li, P.-W.; Li, S.-D. Chitosan-Based Composite Materials for Prospective Hemostatic Applications. Mar. Drugs 2018, 16, 273. [Google Scholar] [CrossRef]
- Huang, Y.; Feng, L.; Zhang, Y.; He, L.; Wang, C.; Xu, J.; Wu, J.; Kirk, T.B.; Guo, R.; Xue, W. Hemostasis mechanism and applications of N-alkylated chitosan sponge. Polym. Adv. Technol. 2017, 28, 1107–1114. [Google Scholar] [CrossRef]
- Chou, T.-C.; Fu, E.; Wu, C.-J.; Yeh, J.-H. Chitosan enhances platelet adhesion and aggregation. Biochem. Biophys. Res. Commun. 2003, 302, 480–483. [Google Scholar] [CrossRef]
- Sagnella, S.; Mai-Ngam, K. Chitosan based surfactant polymers designed to improve blood compatibility on biomaterials. Colloids Surf. B Biointerfaces 2005, 42, 147–155. [Google Scholar] [CrossRef]
- Ahmed, S.; Ikram, S. Chitosan Based Scaffolds and Their Applications in Wound Healing. Achiev. Life Sci. 2016, 10, 27–37. [Google Scholar] [CrossRef]
- Choi, B.K.; Kim, K.Y.; Yoo, Y.J.; Oh, S.J.; Choi, J.H.; Kim, C.Y. In vitro antimicrobial activity of a chitooligosaccharide mixture against Actinobacillus actinomycetemcomitans and Streptococcus mutans. Int. J. Antimicrob. Agents 2001, 18, 553–557. [Google Scholar] [CrossRef]
- Ikinci, G.; Senel, S.; Akincibay, H.; Kaş, S.; Erciş, S.; Wilson, C.G.; Hincal, A.A. Effect of chitosan on a periodontal pathogen Porphyromonas gingivalis. Int. J. Pharm. 2002, 235, 121–127. [Google Scholar] [CrossRef]
- Savard, T.; Beaulieu, C.; Boucher, I.; Champagne, C.P. Antimicrobial action of hydrolyzed chitosan against spoilage yeasts and lactic acid bacteria of fermented vegetables. J. Food Prot. 2002, 65, 828–833. [Google Scholar] [CrossRef]
- Martínez-Camacho, A.P.; Cortez-Rocha, M.O.; Ezquerra-Brauer, J.M.; Graciano-Verdugo, A.Z.; Rodriguez-Félix, F.; Castillo-Ortega, M.M.; Yépiz-Gómez, M.S.; Plascencia-Jatomea, M. Chitosan composite films: Thermal, structural, mechanical and antifungal properties. Carbohydr. Polym. 2010, 82, 305–315. [Google Scholar] [CrossRef]
- Ruiz-Caro, R.; Veiga-Ochoa, M.D. Characterization and dissolution study of chitosan freeze-dried systems for drug controlled release. Molecules 2009, 14, 4370–4386. [Google Scholar] [CrossRef]
- Keller, L.; Regiel-Futyra, A.; Gimeno, M.; Eap, S.; Mendoza, G.; Andreu, V.; Wagner, Q.; Kyzioł, A.; Sebastian, V.; Stochel, G.; et al. Chitosan-based nanocomposites for the repair of bone defects. Nanomedicine 2017, 13, 2231–2240. [Google Scholar] [CrossRef]
- Lim, J.I.; Lee, Y.-K.; Shin, J.-S.; Lim, K.-J. Preparation of interconnected porous chitosan scaffolds by sodium acetate particulate leaching. J. Biomater. Sci. Polym. Ed. 2011, 22, 1319–1329. [Google Scholar] [CrossRef]
- Pezeshki-Modaress, M.; Rajabi-Zeleti, S.; Zandi, M.; Mirzadeh, H.; Sodeifi, N.; Nekookar, A.; Aghdami, N. Cell-loaded gelatin/chitosan scaffolds fabricated by salt-leaching/lyophilization for skin tissue engineering: In vitro and in vivo study. J. Biomed. Mater. Res. A 2014, 102, 3908–3917. [Google Scholar] [CrossRef]
- Geng, X.; Kwon, O.-H.; Jang, J. Electrospinning of chitosan dissolved in concentrated acetic acid solution. Biomaterials 2005, 26, 5427–5432. [Google Scholar] [CrossRef]
- Dahlin, R.L.; Kasper, F.K.; Mikos, A.G. Polymeric nanofibers in tissue engineering. Tissue Eng. Part. B Rev. 2011, 17, 349–364. [Google Scholar] [CrossRef]
- Ahmadi, F.; Oveisi, Z.; Samani, S.M.; Amoozgar, Z. Chitosan based hydrogels: Characteristics and pharmaceutical applications. Res. Pharm. Sci. 2015, 10, 1–16. [Google Scholar]
- Bhattarai, N.; Gunn, J.; Zhang, M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv. Drug Deliv. Rev. 2010, 62, 83–99. [Google Scholar] [CrossRef]
- Chen, R.; Chen, Q.; Huo, D.; Ding, Y.; Hu, Y.; Jiang, X. In situ formation of chitosan-gold hybrid hydrogel and its application for drug delivery. Colloids Surf B Biointerfaces 2012, 97, 132–137. [Google Scholar] [CrossRef]
- Walsh, J.S. Normal bone physiology, remodelling and its hormonal regulation. Surg. (Oxf) 2015, 33, 1–6. [Google Scholar] [CrossRef]
- Arvidson, K.; Abdallah, B.M.; Applegate, L.A.; Baldini, N.; Cenni, E.; Gomez-Barrena, E.; Granchi, D.; Kassem, M.; Konttinen, Y.T.; Mustafa, K.; et al. Bone regeneration and stem cells. J. Cell Mol. Med. 2011, 15, 718–746. [Google Scholar] [CrossRef]
- Loi, F.; Córdova, L.A.; Pajarinen, J.; Lin, T.; Yao, Z.; Goodman, S.B. Inflammation, fracture and bone repair. Bone 2016, 86, 119–130. [Google Scholar] [CrossRef]
- Gómez-Barrena, E.; Rosset, P.; Lozano, D.; Stanovici, J.; Ermthaller, C.; Gerbhard, F. Bone fracture healing: Cell therapy in delayed unions and nonunions. Bone 2015, 70, 93–101. [Google Scholar] [CrossRef]
- Rogers, G.F.; Greene, A.K. Autogenous bone graft: basic science and clinical implications. J. Craniofac. Surg. 2012, 23, 323–327. [Google Scholar] [CrossRef]
- Waibel, K.H.; Haney, B.; Moore, M.; Whisman, B.; Gomez, R. Safety of chitosan bandages in shellfish allergic patients. Mil. Med. 2011, 176, 1153–1156. [Google Scholar] [CrossRef]
- Jiang, T.; Ji, H.; Zhang, L.; Wang, Y.; Zhou, H. Chitosan Oligosaccharide Exerts Anti-Allergic Effect against Shrimp Tropomyosin-Induced Food Allergy by Affecting Th1 and Th2 Cytokines. Int. Arch. Allergy Immunol. 2019, 1–7. [Google Scholar] [CrossRef]
- Azuma, K.; Osaki, T.; Minami, S.; Okamoto, Y. Anticancer and anti-inflammatory properties of chitin and chitosan oligosaccharides. J. Funct. Biomater. 2015, 6, 33–49. [Google Scholar] [CrossRef]
- Bhattarai, G.; Lee, Y.-H.; Lee, M.-H.; Park, I.-S.; Yi, H.-K. Insulin-like growth factor binding protein-3 affects osteogenic efficacy on dental implants in rat mandible. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 55, 490–496. [Google Scholar] [CrossRef]
- Bhattarai, G.; Lee, Y.H.; Lee, M.H.; Yi, H.K. Gene delivery of c-myb increases bone formation surrounding oral implants. J. Dent. Res. 2013, 92, 840–845. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Kim, J.-S.; Kim, J.-E.; Lee, M.-H.; Jeon, J.-G.; Park, I.-S.; Yi, H.-K. Nanoparticle mediated PPARγ gene delivery on dental implants improves osseointegration via mitochondrial biogenesis in diabetes mellitus rat model. Nanomedicine 2017, 13, 1821–1832. [Google Scholar] [CrossRef]
- Li, H.; Ji, Q.; Chen, X.; Sun, Y.; Xu, Q.; Deng, P.; Hu, F.; Yang, J. Accelerated bony defect healing based on chitosan thermosensitive hydrogel scaffolds embedded with chitosan nanoparticles for the delivery of BMP2 plasmid DNA. J. Biomed. Mater. Res. A 2017, 105, 265–273. [Google Scholar] [CrossRef]
- Bleuming, S.A.; He, X.C.; Kodach, L.L.; Hardwick, J.C.; Koopman, F.A.; ten Kate, F.J.; van Deventer, S.J.H.; Hommes, D.W.; Peppelenbosch, M.P.; Offerhaus, G.J.; et al. Bone Morphogenetic Protein Signaling Suppresses Tumorigenesis at Gastric Epithelial Transition Zones in Mice. Cancer Res. 2007, 67, 8149–8155. [Google Scholar] [CrossRef]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Primers 2017, 3, 17038. [Google Scholar] [CrossRef]
- Cortellini, P.; Tonetti, M.S. Clinical concepts for regenerative therapy in intrabony defects. Periodontol. 2000 2015, 68, 282–307. [Google Scholar] [CrossRef]
- Cortellini, P.; Tonetti, M.S. Focus on intrabony defects: Guided tissue regeneration. Periodontol. 2000 2000, 22, 104–132. [Google Scholar] [CrossRef]
- Harmouche, L.; Courval, A.; Mathieu, A.; Petit, C.; Huck, O.; Severac, F.; Davideau, J.-L. Impact of tooth-related factors on photodynamic therapy effectiveness during active periodontal therapy: A 6-months split-mouth randomized clinical trial. Photodiagnosis Photodyn. 2019, 190–194. [Google Scholar] [CrossRef]
- Abdul Rahman, N.; Nickles, K.; Gallenbach, K.; Dannewitz, B.; Ramich, T.; Scharf, S.; Röllke, L.; Schacher, B.; Eickholz, P. Five-year stability of clinical attachment after regenerative treatment of infrabony defects compared to controls. J. Clin. Periodontol. 2019, 46, 650–658. [Google Scholar] [CrossRef]
- Da Rocha, H.A.J.; Silva, C.F.; Santiago, F.L.; Martins, L.G.; Dias, P.C.; De Magalhães, D. Local Drug Delivery Systems in the Treatment of Periodontitis: A Literature Review. J. Int. Acad. Periodontol. 2015, 17, 82–90. [Google Scholar]
- Morand, D.N.; Davideau, J.-L.; Clauss, F.; Jessel, N.; Tenenbaum, H.; Huck, O. Cytokines during periodontal wound healing: Potential application for new therapeutic approach. Oral Dis. 2017, 23, 300–311. [Google Scholar] [CrossRef]
- Batool, F.; Strub, M.; Petit, C.; Bugueno, I.M.; Bornert, F.; Clauss, F.; Huck, O.; Kuchler-Bopp, S.; Benkirane-Jessel, N. Periodontal Tissues, Maxillary Jaw Bone, and Tooth Regeneration Approaches: From Animal Models Analyses to Clinical Applications. Nanomaterials 2018, 8, 337. [Google Scholar] [CrossRef]
- Petit, C.; Batool, F.; Bugueno, I.M.; Schwinté, P.; Benkirane-Jessel, N.; Huck, O. Contribution of Statins towards Periodontal Treatment: A Review. Mediat. Inflamm. 2019, 2019, 6367402. [Google Scholar] [CrossRef]
- Sah, A.K.; Dewangan, M.; Suresh, P.K. Potential of chitosan-based carrier for periodontal drug delivery. Colloids Surf. B: Biointerfaces 2019, 178, 185–198. [Google Scholar] [CrossRef]
- Yar, M.; Farooq, A.; Shahzadi, L.; Khan, A.S.; Mahmood, N.; Rauf, A.; Chaudhry, A.A.; Rehman, I.U. Novel meloxicam releasing electrospun polymer/ceramic reinforced biodegradable membranes for periodontal regeneration applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 64, 148–156. [Google Scholar] [CrossRef]
- Kamal, T.; Khan, S.B.; Haider, S.; Alghamdi, Y.G.; Asiri, A.M. Thin layer chitosan-coated cellulose filter paper as substrate for immobilization of catalytic cobalt nanoparticles. Int. J. Biol. Macromol. 2017, 104, 56–62. [Google Scholar] [CrossRef]
- Işılay Özdoğan, A.; Akca, G.; Şenel, S. Development and in vitro evaluation of chitosan based system for local delivery of atorvastatin for treatment of periodontitis. Eur. J. Pharm. Sci. 2018, 124, 208–216. [Google Scholar] [CrossRef]
- Ganguly, A.; Ian, C.K.; Sheshala, R.; Sahu, P.S.; Al-Waeli, H.; Meka, V.S. Application of diverse natural polymers in the design of oral gels for the treatment of periodontal diseases. J. Mater. Sci. Mater. Med. 2017, 28, 39. [Google Scholar] [CrossRef]
- Özdoğan, A.I.; İlarslan, Y.D.; Kösemehmetoğlu, K.; Akca, G.; Kutlu, H.B.; Comerdov, E.; Iskit, A.B.; Şenel, S. In vivo evaluation of chitosan based local delivery systems for atorvastatin in treatment of periodontitis. Int. J. Pharm. 2018, 550, 470–476. [Google Scholar] [CrossRef]
- Madi, M.; Pavlic, V.; Samy, W.; Alagl, A. The anti-inflammatory effect of locally delivered nano-doxycycline gel in therapy of chronic periodontitis. Acta Odontol. Scand. 2018, 76, 71–76. [Google Scholar] [CrossRef]
- Khan, G.; Yadav, S.K.; Patel, R.R.; Nath, G.; Bansal, M.; Mishra, B. Development and Evaluation of Biodegradable Chitosan Films of Metronidazole and Levofloxacin for the Management of Periodontitis. Aaps Pharmscitech 2016, 17, 1312–1325. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Tseng, K.-S.; Liu, J.-M.; Chuang, H.-C.; Lien, C.-H.; Chen, Y.-C.; Lai, C.-Y.; Yu, C.-P.; Hsu, R.-J. Increased Risk of Ulcerative Colitis in Patients with Periodontal Disease: A Nationwide Population-Based Cohort Study. Int. J. Env. Res. Public Health 2018, 15, 2602. [Google Scholar] [CrossRef]
- Kilicarslan, M.; Gumustas, M.; Yildiz, S.; Baykara, T. Preparation and characterization of chitosan-based spray-dried microparticles for the delivery of clindamycin phosphate to periodontal pockets. Curr. Drug Deliv. 2014, 11, 98–111. [Google Scholar] [CrossRef]
- Chang, P.-C.; Chao, Y.-C.; Hsiao, M.-H.; Chou, H.-S.; Jheng, Y.-H.; Yu, X.-H.; Lee, N.; Yang, C.; Liu, D.-M. Inhibition of Periodontitis Induction Using a Stimuli-Responsive Hydrogel Carrying Naringin. J. Periodontol. 2017, 88, 190–196. [Google Scholar] [CrossRef]
- Barreras, U.S.; Méndez, F.T.; Martínez, R.E.M.; Valencia, C.S.; Rodríguez, P.R.M.; Rodríguez, J.P.L. Chitosan nanoparticles enhance the antibacterial activity of chlorhexidine in collagen membranes used for periapical guided tissue regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 1182–1187. [Google Scholar] [CrossRef]
- Ferrand, A.; Eap, S.; Richert, L.; Lemoine, S.; Kalaskar, D.; Demoustier-Champagne, S.; Atmani, H.; Mély, Y.; Fioretti, F.; Schlatter, G.; et al. Osteogenetic properties of electrospun nanofibrous PCL scaffolds equipped with chitosan-based nanoreservoirs of growth factors. Macromol. Biosci. 2014, 14, 45–55. [Google Scholar] [CrossRef]
- Issa, J.P.M.; do Nascimento, C.; Bentley, M.V.L.B.; Del Bel, E.A.; Iyomasa, M.M.; Sebald, W.; de Albuquerque, R.F. Bone repair in rat mandible by rhBMP-2 associated with two carriers. Micron 2008, 39, 373–379. [Google Scholar] [CrossRef]
- Seol, Y.-J.; Lee, J.-Y.; Park, Y.-J.; Lee, Y.-M.; Young-Ku, N.; Rhyu, I.-C.; Lee, S.-J.; Han, S.-B.; Chung, C.-P. Chitosan sponges as tissue engineering scaffolds for bone formation. Biotechnol. Lett. 2004, 26, 1037–1041. [Google Scholar] [CrossRef]
- Mohammadi, R.; Amini, K. Guided bone regeneration of mandibles using chitosan scaffold seeded with characterized uncultured omental adipose-derived stromal vascular fraction: An animal study. Int. J. Oral Maxillofac Implant. 2015, 30, 216–222. [Google Scholar] [CrossRef]
- Xu, F.; Wu, Y.; Zhang, Y.; Yin, P.; Fang, C.; Wang, J. Influence of in vitro differentiation status on the in vivo bone regeneration of cell/chitosan microspheres using a rat cranial defect model. J. Biomater. Sci. Polym. Ed. 2019, 30, 1008–1025. [Google Scholar] [CrossRef]
- Danilchenko, S.N.; Kalinkevich, O.V.; Pogorelov, M.V.; Kalinkevich, A.N.; Sklyar, A.M.; Kalinichenko, T.G.; Ilyashenko, V.Y.; Starikov, V.V.; Bumeyster, V.I.; Sikora, V.Z.; et al. Characterization and in vivo evaluation of chitosan-hydroxyapatite bone scaffolds made by one step coprecipitation method. J. Biomed. Mater. Res. Part. A 2011, 96A, 639–647. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, L.; Lv, H.; Cao, Y.; Liu, Y.; Xu, Y.; Ye, W.; Wang, J. Repair of rabbit femoral condyle bone defects with injectable nanohydroxyapatite/chitosan composites. J. Mater. Sci. Mater. Med. 2012, 23, 1941–1949. [Google Scholar] [CrossRef]
- Gaihre, B.; Jayasuriya, A.C. Comparative investigation of porous nano-hydroxyapaptite/chitosan, nano-zirconia/chitosan and novel nano-calcium zirconate/chitosan composite scaffolds for their potential applications in bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 91, 330–339. [Google Scholar] [CrossRef]
- Cho, B.C.; Chung, H.Y.; Lee, D.G.; Yang, J.D.; Park, J.W.; Roh, K.H.; Kim, G.U.; Lee, D.S.; Kwon, I.C.; Bae, E.H.; et al. The effect of chitosan bead encapsulating calcium sulfate as an injectable bone substitute on consolidation in the mandibular distraction osteogenesis of a dog model. J. Oral Maxillofac. Surg. 2005, 63, 1753–1764. [Google Scholar] [CrossRef]
- Pattnaik, S.; Nethala, S.; Tripathi, A.; Saravanan, S.; Moorthi, A.; Selvamurugan, N. Chitosan scaffolds containing silicon dioxide and zirconia nano particles for bone tissue engineering. Int. J. Biol. Macromol. 2011, 49, 1167–1172. [Google Scholar] [CrossRef]
- Shokri, S.; Movahedi, B.; Rafieinia, M.; Salehi, H. A new approach to fabrication of Cs/BG/CNT nanocomposite scaffold towards bone tissue engineering and evaluation of its properties. Appl. Surf. Sci. 2015, 357, 1758–1764. [Google Scholar] [CrossRef]
- Kucharska, M.; Butruk, B.; Walenko, K.; Brynk, T.; Ciach, T. Fabrication of in-situ foamed chitosan/β-TCP scaffolds for bone tissue engineering application. Mater. Lett. 2012, 85, 124–127. [Google Scholar] [CrossRef]
- Cheng, G.; Li, Z.; Wan, Q.; Lv, K.; Li, D.; Xing, X.; Li, Z. A novel animal model treated with tooth extraction to repair the full-thickness defects in the mandible of rabbits. J. Surg. Res. 2015, 194, 706–716. [Google Scholar] [CrossRef]
- Chang, H.-H.; Wang, Y.-L.; Chiang, Y.-C.; Chen, Y.-L.; Chuang, Y.-H.; Tsai, S.-J.; Heish, K.-H.; Lin, F.-H.; Lin, C.-P. A Novel Chitosan-γPGA Polyelectrolyte Complex Hydrogel Promotes Early New Bone Formation in the Alveolar Socket Following Tooth Extraction. PloS ONE 2014, 9, e92362. [Google Scholar] [CrossRef]
- Fan, J.; Park, H.; Lee, M.K.; Bezouglaia, O.; Fartash, A.; Kim, J.; Aghaloo, T.; Lee, M. Adipose-derived stem cells and BMP-2 delivery in chitosan-based 3D constructs to enhance bone regeneration in a rat mandibular defect model. Tissue Eng. Part. A 2014, 20, 2169–2179. [Google Scholar] [CrossRef]
- Pourhaghgouy, M.; Zamanian, A.; Shahrezaee, M.; Masouleh, M.P. Physicochemical properties and bioactivity of freeze-cast chitosan nanocomposite scaffolds reinforced with bioactive glass. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 180–186. [Google Scholar] [CrossRef]
- Nazemi, K.; Azadpour, P.; Moztarzadeh, F.; Urbanska, A.M.; Mozafari, M. Tissue-engineered chitosan/bioactive glass bone scaffolds integrated with PLGA nanoparticles: A therapeutic design for on-demand drug delivery. Mater. Lett. 2015, 138, 16–20. [Google Scholar] [CrossRef]
- Venkatesan, J.; Ryu, B.; Sudha, P.N.; Kim, S.-K. Preparation and characterization of chitosan–carbon nanotube scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2012, 50, 393–402. [Google Scholar] [CrossRef]
- Saravanan, S.; Sameera, D.K.; Moorthi, A.; Selvamurugan, N. Chitosan scaffolds containing chicken feather keratin nanoparticles for bone tissue engineering. Int. J. Biol. Macromol. 2013, 62, 481–486. [Google Scholar] [CrossRef]
- Maglione, M.; Spano, S.; Ruaro, M.E.; Salvador, E.; Zanconati, F.; Tromba, G.; Turco, G. In vivo evaluation of chitosan-glycerol gel scaffolds seeded with stem cells for full-thickness mandibular bone regeneration. J. Oral Sci. 2017, 59, 225–232. [Google Scholar] [CrossRef][Green Version]
- Saravanan, S.; Vimalraj, S.; Anuradha, D. Chitosan based thermoresponsive hydrogel containing graphene oxide for bone tissue repair. Biomed. Pharm. 2018, 107, 908–917. [Google Scholar] [CrossRef]
- Keller, L.; Pijnenburg, L.; Idoux-Gillet, Y.; Bornert, F.; Benameur, L.; Tabrizian, M.; Auvray, P.; Rosset, P.; Gonzalo-Daganzo, R.M.; Barrena, E.G.; et al. Preclinical safety study of a combined therapeutic bone wound dressing for osteoarticular regeneration. Nat. Commun. 2019, 10, 2156. [Google Scholar] [CrossRef]
- Jayakumar, R.; Ramachandran, R.; Sudheesh Kumar, P.T.; Divyarani, V.V.; Srinivasan, S.; Chennazhi, K.P.; Tamura, H.; Nair, S.V. Fabrication of chitin–chitosan/nano ZrO2 composite scaffolds for tissue engineering applications. Int. J. Biol. Macromol. 2011, 49, 274–280. [Google Scholar] [CrossRef]
- Park, J.-S.; Choi, S.-H.; Moon, I.-S.; Cho, K.-S.; Chai, J.-K.; Kim, C.-K. Eight-week histological analysis on the effect of chitosan on surgically created one-wall intrabony defects in beagle dogs. J. Clin. Periodontol. 2003, 30, 443–453. [Google Scholar] [CrossRef]
- Eap, S.; Ferrand, A.; Schiavi, J.; Keller, L.; Kokten, T.; Fioretti, F.; Mainard, D.; Ladam, G.; Benkirane-Jessel, N. Collagen implants equipped with ’fish scale’-like nanoreservoirs of growth factors for bone regeneration. Nanomed. (Lond) 2014, 9, 1253–1261. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, B.; Li, C.; Wang, Y.; Chen, Y.; Zhang, W.; Luo, T.; Cheng, X. The synergetic bone-forming effects of combinations of growth factors expressed by adenovirus vectors on chitosan/collagen scaffolds. J. Control. Release 2009, 136, 172–178. [Google Scholar] [CrossRef]
- Zhang, Q.; Hubenak, J.; Iyyanki, T.; Alred, E.; Turza, K.C.; Davis, G.; Chang, E.I.; Branch-Brooks, C.D.; Beahm, E.K.; Butler, C.E. Engineering vascularized soft tissue flaps in an animal model using human adipose-derived stem cells and VEGF+PLGA/PEG microspheres on a collagen-chitosan scaffold with a flow-through vascular pedicle. Biomaterials 2015, 73, 198–213. [Google Scholar] [CrossRef]
- Shi, S.; Cheng, X.; Wang, J.; Zhang, W.; Peng, L.; Zhang, Y. RhBMP-2 microspheres-loaded chitosan/collagen scaffold enhanced osseointegration: An experiment in dog. J. Biomater. Appl. 2009, 23, 331–346. [Google Scholar]
- Elango, J.; Saravanakumar, K.; Rahman, S.U.; Henrotin, Y.; Regenstein, J.M.; Wu, W.; Bao, B. Chitosan-Collagen 3D Matrix Mimics Trabecular Bone and Regulates RANKL-Mediated Paracrine Cues of Differentiated Osteoblast and Mesenchymal Stem Cells for Bone Marrow Macrophage-Derived Osteoclastogenesis. Biomolecules 2019, 9, 173. [Google Scholar] [CrossRef]
- Ansarizadeh, M.; Mashayekhan, S.; Saadatmand, M. Fabrication, modeling and optimization of lyophilized advanced platelet rich fibrin in combination with collagen-chitosan as a guided bone regeneration membrane. Int. J. Biol. Macromol. 2019, 125, 383–391. [Google Scholar] [CrossRef]
- Sowjanya, J.A.; Singh, J.; Mohita, T.; Sarvanan, S.; Moorthi, A.; Srinivasan, N.; Selvamurugan, N. Biocomposite scaffolds containing chitosan/alginate/nano-silica for bone tissue engineering. Colloids Surf. B Biointerfaces 2013, 109, 294–300. [Google Scholar] [CrossRef]
- Kim, H.-L.; Jung, G.-Y.; Yoon, J.-H.; Han, J.-S.; Park, Y.-J.; Kim, D.-G.; Zhang, M.; Kim, D.-J. Preparation and characterization of nano-sized hydroxyapatite/alginate/chitosan composite scaffolds for bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 54, 20–25. [Google Scholar] [CrossRef]
- Jin, H.-H.; Kim, D.-H.; Kim, T.-W.; Shin, K.-K.; Jung, J.S.; Park, H.-C.; Yoon, S.-Y. In vivo evaluation of porous hydroxyapatite/chitosan–alginate composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2012, 51, 1079–1085. [Google Scholar] [CrossRef]
- Florczyk, S.J.; Leung, M.; Li, Z.; Huang, J.I.; Hopper, R.A.; Zhang, M. Evaluation of three-dimensional porous chitosan-alginate scaffolds in rat calvarial defects for bone regeneration applications. J. Biomed. Mater. Res. A 2013, 101, 2974–2983. [Google Scholar] [CrossRef]
- Teimouri, A.; Ebrahimi, R.; Emadi, R.; Beni, B.H.; Chermahini, A.N. Nano-composite of silk fibroin-chitosan/Nano ZrO2 for tissue engineering applications: Fabrication and morphology. Int. J. Biol. Macromol. 2015, 76, 292–302. [Google Scholar] [CrossRef]
- Lima, P.A.L.; Resende, C.X.; de Almeida Soares, G.D.; Anselme, K.; Almeida, L.E. Preparation, characterization and biological test of 3D-scaffolds based on chitosan, fibroin and hydroxyapatite for bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 3389–3395. [Google Scholar] [CrossRef]
- Tong, S.; Xu, D.-P.; Liu, Z.-M.; Du, Y.; Wang, X.-K. Synthesis of and in vitro and in vivo evaluation of a novel TGF-β1-SF-CS three-dimensional scaffold for bone tissue engineering. Int. J. Mol. Med. 2016, 38, 367–380. [Google Scholar] [CrossRef]
- Niu, X.; Fan, Y.; Liu, X.; Li, X.; Li, P.; Wang, J.; Sha, Z.; Feng, Q. Repair of bone defect in femoral condyle using microencapsulated chitosan, nanohydroxyapatite/collagen and poly(L-lactide)-based microsphere-scaffold delivery system. Artif. Organs 2011, 35, E119–E128. [Google Scholar] [CrossRef]
- Sainitya, R.; Sriram, M.; Kalyanaraman, V.; Dhivya, S.; Saravanan, S.; Vairamani, M.; Sastry, T.P.; Selvamurugan, N. Scaffolds containing chitosan/carboxymethyl cellulose/mesoporous wollastonite for bone tissue engineering. Int. J. Biol. Macromol. 2015, 80, 481–488. [Google Scholar] [CrossRef]
- Miranda, S.C.C.C.; Silva, G.A.B.; Mendes, R.M.; Abreu, F.A.M.; Caliari, M.V.; Alves, J.B.; Goes, A.M. Mesenchymal stem cells associated with porous chitosan–gelatin scaffold: A potential strategy for alveolar bone regeneration. J. Biomed. Mater. Res. Part. A 2012, 100A, 2775–2786. [Google Scholar] [CrossRef]
- Olad, A.; Farshi Azhar, F. The synergetic effect of bioactive ceramic and nanoclay on the properties of chitosan–gelatin/nanohydroxyapatite–montmorillonite scaffold for bone tissue engineering. Ceram. Int. 2014, 40, 10061–10072. [Google Scholar] [CrossRef]
- Kavya, K.C.; Jayakumar, R.; Nair, S.; Chennazhi, K.P. Fabrication and characterization of chitosan/gelatin/nSiO2 composite scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2013, 59, 255–263. [Google Scholar] [CrossRef]
- Serra, I.R.; Fradique, R.; Vallejo, M.C.S.; Correia, T.R.; Miguel, S.P.; Correia, I.J. Production and characterization of chitosan/gelatin/β-TCP scaffolds for improved bone tissue regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 55, 592–604. [Google Scholar] [CrossRef]
- Acevedo, C.A.; Olguín, Y.; Briceño, M.; Forero, J.C.; Osses, N.; Díaz-Calderón, P.; Jaques, A.; Ortiz, R. Design of a biodegradable UV-irradiated gelatin-chitosan/nanocomposed membrane with osteogenic ability for application in bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 99, 875–886. [Google Scholar] [CrossRef]
- Maji, K.; Dasgupta, S.; Kundu, B.; Bissoyi, A. Development of gelatin-chitosan-hydroxyapatite based bioactive bone scaffold with controlled pore size and mechanical strength. J. Biomater. Sci. Polym. Ed. 2015, 26, 1190–1209. [Google Scholar] [CrossRef]
- Puvaneswary, S.; Talebian, S.; Raghavendran, H.B.; Murali, M.R.; Mehrali, M.; Afifi, A.M.; Kasim, N.H.B.A.; Kamarul, T. Fabrication and in vitro biological activity of βTCP-Chitosan-Fucoidan composite for bone tissue engineering. Carbohydr. Polym. 2015, 134, 799–807. [Google Scholar] [CrossRef]
- Jing, X.; Mi, H.-Y.; Peng, J.; Peng, X.-F.; Turng, L.-S. Electrospun aligned poly(propylene carbonate) microfibers with chitosan nanofibers as tissue engineering scaffolds. Carbohydr. Polym. 2015, 117, 941–949. [Google Scholar] [CrossRef]
- Zhang, S.; Prabhakaran, M.P.; Qin, X.; Ramakrishna, S. Biocomposite scaffolds for bone regeneration: Role of chitosan and hydroxyapatite within poly-3-hydroxybutyrate-co-3-hydroxyvalerate on mechanical properties and in vitro evaluation. J. Mech. Behav. Biomed. Mater. 2015, 51, 88–98. [Google Scholar] [CrossRef]
- Yao, Q.; Li, W.; Yu, S.; Ma, L.; Jin, D.; Boccaccini, A.R.; Liu, Y. Multifunctional chitosan/polyvinyl pyrrolidone/45S5 Bioglass® scaffolds for MC3T3-E1 cell stimulation and drug release. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 56, 473–480. [Google Scholar] [CrossRef]
- Sajesh, K.M.; Jayakumar, R.; Nair, S.V.; Chennazhi, K.P. Biocompatible conducting chitosan/polypyrrole-alginate composite scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2013, 62, 465–471. [Google Scholar] [CrossRef]
- Pon-On, W.; Charoenphandhu, N.; Teerapornpuntakit, J.; Thongbunchoo, J.; Krishnamra, N.; Tang, I.-M. Mechanical properties, biological activity and protein controlled release by poly(vinyl alcohol)-bioglass/chitosan-collagen composite scaffolds: A bone tissue engineering applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 38, 63–72. [Google Scholar] [CrossRef]
- Peng, L.; Zhou, Y.; Lu, W.; Zhu, W.; Li, Y.; Chen, K.; Zhang, G.; Xu, J.; Deng, Z.; Wang, D. Characterization of a novel polyvinyl alcohol/chitosan porous hydrogel combined with bone marrow mesenchymal stem cells and its application in articular cartilage repair. BMC Musculoskelet. Disord. 2019, 20, 257. [Google Scholar] [CrossRef]
- De la Riva, B.; Nowak, C.; Sánchez, E.; Hernández, A.; Schulz-Siegmund, M.; Pec, M.K.; Delgado, A.; Evora, C. VEGF-controlled release within a bone defect from alginate/chitosan/PLA-H scaffolds. Eur. J. Pharm. Biopharm. 2009, 73, 50–58. [Google Scholar] [CrossRef]
- Keller, L.; Offner, D.; Schwinté, P.; Morand, D.; Wagner, Q.; Gros, C.; Bornert, F.; Bahi, S.; Musset, A.-M.; Benkirane-Jessel, N.; et al. Active Nanomaterials to Meet the Challenge of Dental Pulp Regeneration. Mater. (Basel) 2015, 8, 7461–7471. [Google Scholar] [CrossRef]
- Zein, N.; Harmouch, E.; Lutz, J.-C.; Fernandez De Grado, G.; Kuchler-Bopp, S.; Clauss, F.; Offner, D.; Hua, G.; Benkirane-Jessel, N.; Fioretti, F. Polymer-Based Instructive Scaffolds for Endodontic Regeneration. Mater. (Basel) 2019, 12, 2347. [Google Scholar] [CrossRef]
- Liao, F.; Chen, Y.; Li, Z.; Wang, Y.; Shi, B.; Gong, Z.; Cheng, X. A novel bioactive three-dimensional beta-tricalcium phosphate/chitosan scaffold for periodontal tissue engineering. J. Mater. Sci. Mater. Med. 2010, 21, 489–496. [Google Scholar] [CrossRef]
- Albuquerque, M.T.P.; Valera, M.C.; Nakashima, M.; Nör, J.E.; Bottino, M.C. Tissue-engineering-based strategies for regenerative endodontics. J. Dent. Res. 2014, 93, 1222–1231. [Google Scholar] [CrossRef]
- Bordini, E.A.F.; Cassiano, F.B.; Silva, I.S.P.; Usberti, F.R.; Anovazzi, G.; Pacheco, L.E.; Pansani, T.N.; Leite, M.L.; Hebling, J.; de Souza Costa, C.A.; et al. Synergistic potential of 1α,25-dihydroxyvitamin D3 and calcium-aluminate-chitosan scaffolds with dental pulp cells. Clin. Oral Investig. 2019, 1–12. [Google Scholar] [CrossRef]
- Ducret, M.; Montembault, A.; Josse, J.; Pasdeloup, M.; Celle, A.; Benchrih, R.; Mallein-Gerin, F.; Alliot-Licht, B.; David, L.; Farges, J.-C. Design and characterization of a chitosan-enriched fibrin hydrogel for human dental pulp regeneration. Dent. Mater. 2019, 35, 523–533. [Google Scholar] [CrossRef]
- Zhu, N.; Chatzistavrou, X.; Ge, L.; Qin, M.; Papagerakis, P.; Wang, Y. Biological properties of modified bioactive glass on dental pulp cells. J. Dent. 2019, 83, 18–26. [Google Scholar] [CrossRef]
- Loyola-Rodríguez, J.P.; Torres-Méndez, F.; Espinosa-Cristobal, L.F.; García-Cortes, J.O.; Loyola-Leyva, A.; González, F.J.; Soto-Barreras, U.; Nieto-Aguilar, R.; Contreras-Palma, G. Antimicrobial activity of endodontic sealers and medications containing chitosan and silver nanoparticles against Enterococcus faecalis. J. Appl. Biomater. Funct. Mater. 2019, 17, 2280800019851771. [Google Scholar] [CrossRef]
| Chitosan-Based Biocomposite Scaffolds | |||||
|---|---|---|---|---|---|
| Polymers and/or Biomaterials | Bioactive Molecule | Models | Observations as Compared to the Properties of Chitosan Polymer Scaffolds Alone | Reference | |
| BMP-2 | In vitro | No cytotoxicity and increased osteogenesis | [90] | ||
| In vivo | No cytotoxicity, increased biomineralization, and increased osteogenesis | ||||
| Recombinant human BMP-2 | In vivo | Enhanced bone regeneration | [91] | ||
| In vitro | Increased biomineralization and increased osteogenesis | [92] | |||
| In vivo | Improved and earlier bone regeneration | [93] | |||
| In vitro | No cytotoxicity, increased biomineralization | [94] | |||
| In vivo | Generation of a substantial amount of bone in rat cranium | ||||
| Hydroxyapatite | In vivo | New bone tissue formation in rat | [95] | ||
| Nano hydroxyapatite | In vivo | Regeneration of segmental bone defects with cortical bone in rabbit | [96] | ||
| Nano hydroxyapatite/Nano ZrO2/Nano CaZrO3 | In vitro | No cytotoxicity, decreased water retention and increased mechanical properties | [97] | ||
| Calcium sulfate | In vivo | Early bony consolidation | [98] | ||
| SiO2 + ZrO2 | In vitro | No cytotoxicity at low concentration, decreased water retention, increased protein adsorption, biomineralization, and biodegradation | [99] | ||
| Bioactive glass + carbon nanotube | In vitro | No cytotoxicity, increased water retention, biodegradation, and mechanical properties | [100] | ||
| β-tricalcium phosphate | In vitro | No cytotoxicity at low concentration, decreased biodegradation, and increased mechanical properties | [101] | ||
| In vivo | Increased new bone formation | [102] | |||
| γ-polyglutamic acid | In vivo | Increased new bone formation | [103] | ||
| Chondroitine sulfate + apatite | BMP-2 | In vivo | Enhanced bone regeneration | [104] | |
| Bioactive glass | In vitro | Decreased water retention, increased biomineralization, biodegradation, and mechanical properties | [105] | ||
| Bioactive glass + poly lactic-co-glycolic acid (PLGA) nanoparticles | In vitro | Decreased water retention, and increased mechanical properties | [106] | ||
| Carbon nanotube | In vitro | No cytotoxicity, increased biomineralization | [107] | ||
| Keratin nanoparticles | In vitro | No cytotoxicity, increased protein adsorption and biodegradation | [108] | ||
| Glycerophosphate | In vivo | Enhanced bone regeneration | [109] | ||
| Glycerophosphate + graphene oxide | In vitro | No cytotoxicity, increased water retention, protein adsorption, biomineralization, biodegradation, and osteogenesis | [110] | ||
| poly-ɛ-caprolactone | BMP-2 | In vitro | No cytotoxicity | [111] | |
| In vivo | Regeneration of both the subchondral bone and the cartilage in large animal model | ||||
| Chitin + Nano ZrO2 | In vitro | No cytotoxicity, decreased water retention and biodegradation, increased biomineralization and osteogenesis | [112] | ||
| Collagen | In vivo | Enhanced bone regeneration | [113] | ||
| BMP-2 | In vitro | No cytotoxicity, increased biomineralization and osteogenesis | [114] | ||
| In vivo | No cytotoxicity, increased biomineralization and osteogenesis | ||||
| BMP-7 | In vivo | Accelerated regeneration of alveolar bone tissue | [115] | ||
| PLGA/Polyethylene glycol (PEG) | VEGF | In vitro | Induced angiogenesis | [116] | |
| In vivo | Induced angiogenesis and vascularization in rat | ||||
| PLGA | rhBMP-2 | In vitro | Controlled growth factor release rate | [117] | |
| In vivo | Enhanced bone formation and fast bone regeneration in dog | ||||
| Chondroitine sulfate + hydroxyapatite | In vitro | Secretion of higher level of receptor activator of nuclear factor kappa-B ligand (RANKL) to mediate osteoclastogenesis | [118] | ||
| Advanced platelet rich fibrin (A-PRF) | In vitro | No cytotoxicity, increased biomineralization and mechanical properties, decreased biodegradation | [119] | ||
| Alginate | Nano SiO2 | In vitro | No cytotoxicity, decreased water retention and mechanical properties, increased protein adsorption, biomineralization, biodegradation, and osteogenesis | [120] | |
| Nano-sized hydroxyapatite | In vitro | No cytotoxicity, increased biomineralization, osteogenesis, and mechanical properties | [121] | ||
| Hydroxyapatite | In vitro | No cytotoxicity | [122] | ||
| In vivo | Strong positive effect on bone formation in mice | ||||
| BMP-2 | In vitro | No cytotoxicity | [123] | ||
| In vivo | Great osteogenesis and reconstruction of critical size bone defects | ||||
| Silk fibroin | Nano ZrO2 | In vitro | No cytotoxicity, increased water retention, biomineralization, biodegradation, and mechanical properties | [124] | |
| Hydroxyapatite | In vitro | Increased biomineralization and osteogenesis | [125] | ||
| TGF-β1 | In vivo | Biocompatibility and extensive osteoconductivity and osteogenesis | [126] | ||
| Collagen + Poly(L-Lactide) | Nanohydroxyapatite | BMP-2 | In vitro | Controlled growth factor release rate and more favorable cytocompatibility | [127] |
| In vivo | Accelerated regeneration of cancellous bone defect in rabbit | ||||
| Carboxyme-thylcellulose | mesoporous wollastonite | In vitro | No cytotoxicity, decreased water retention and biodegradation, increased protein adsorption, biomineralization, and osteogenesis | [128] | |
| Gelatin | In vivo | Increased amount of new bone formation | [129] | ||
| Hydroxyapatite –montmorillonite | In vitro | Decreased biodegradation, increased biomineralization and mechanical properties | [130] | ||
| Nano SiO2 | In vitro | No cytotoxicity, decreased water retention, increased protein adsorption, biomineralization, biodegradation, and mechanical properties | [131] | ||
| β-tricalcium phosphate | In vitro | No cytotoxicity, increased water retention, biomineralization, osteogenesis, and mechanical properties | [132] | ||
| Hydroxyapatite+ titania | In vitro | No cytotoxicity, increased biomineralization and mechanical properties, decreased biodegradation | [133] | ||
| Hydroxyapatite | In vitro | No cytotoxicity, increased osteogenesis and mechanical properties | [134] | ||
| Fucoidan | β-tricalcium phosphate | In vitro | No cytotoxicity, increased protein adsorption, biomineralization, osteogenesis, and mechanical properties | [135] | |
| poly(propylene carbonate) | In vitro | No cytotoxicity, increased mechanical properties | [136] | ||
| Poly-3-hydro xybutyrate-co3-hydroxyvalerate (PHBV) | Hydroxyapatite | In vitro | No cytotoxicity, increased biomineralization, osteogenesis, and mechanical properties | [137] | |
| Polyvinyl pyrrolidone | Bioactive glass | In vitro | No cytotoxicity, decreased biodegradation | [138] | |
| Polypyrrole-alginate | In vitro | No cytotoxicity, increased biomineralization, decreased water retention, protein adsorption, and biodegradation | [139] | ||
| Polyvinyl alcohol or collagen | Bioactive glass | In vitro | No cytotoxicity, increased biomineralization and mechanical properties, decreased water retention and biodegradation | [140] | |
| Polyvinyl alcohol | In vitro | No cytotoxicity, increased water retention, osteogenesis, and mechanical properties | [141] | ||
| In vivo | Good cartilage healing in rabbit | ||||
| Polylactide + Alginate | VEGF | In vitro | Good VEGF release rate, enhanced neovascularization in bone healing and maintenance of bioactivity | [142] | |
| In vivo | |||||
| Chitosan-Based Biocomposite Scaffolds | ||||
|---|---|---|---|---|
| Polymers and/or Biomaterials | Bioactive Molecule | Model | Observations | Reference |
| β-tricalcium phosphate | In vitro |
| [145] | |
| Collagen | BMP-7 | In vivo |
| [146] |
| Calcium-aluminate | 1α,25-dihydroxyvitamin D3 (1α,25VD) | In vitro |
| [147] |
| Fibrin | In vitro |
| [148] | |
| Silver-doped bioactive glass | In vitro |
| [149] | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguilar, A.; Zein, N.; Harmouch, E.; Hafdi, B.; Bornert, F.; Offner, D.; Clauss, F.; Fioretti, F.; Huck, O.; Benkirane-Jessel, N.; et al. Application of Chitosan in Bone and Dental Engineering. Molecules 2019, 24, 3009. https://doi.org/10.3390/molecules24163009
Aguilar A, Zein N, Harmouch E, Hafdi B, Bornert F, Offner D, Clauss F, Fioretti F, Huck O, Benkirane-Jessel N, et al. Application of Chitosan in Bone and Dental Engineering. Molecules. 2019; 24(16):3009. https://doi.org/10.3390/molecules24163009
Chicago/Turabian StyleAguilar, Alicia, Naimah Zein, Ezeddine Harmouch, Brahim Hafdi, Fabien Bornert, Damien Offner, François Clauss, Florence Fioretti, Olivier Huck, Nadia Benkirane-Jessel, and et al. 2019. "Application of Chitosan in Bone and Dental Engineering" Molecules 24, no. 16: 3009. https://doi.org/10.3390/molecules24163009
APA StyleAguilar, A., Zein, N., Harmouch, E., Hafdi, B., Bornert, F., Offner, D., Clauss, F., Fioretti, F., Huck, O., Benkirane-Jessel, N., & Hua, G. (2019). Application of Chitosan in Bone and Dental Engineering. Molecules, 24(16), 3009. https://doi.org/10.3390/molecules24163009








