Soy Isoflavones Improve the Spermatogenic Defects in Diet-Induced Obesity Rats through Nrf2/HO-1 Pathway
Abstract
1. Introduction
2. Results
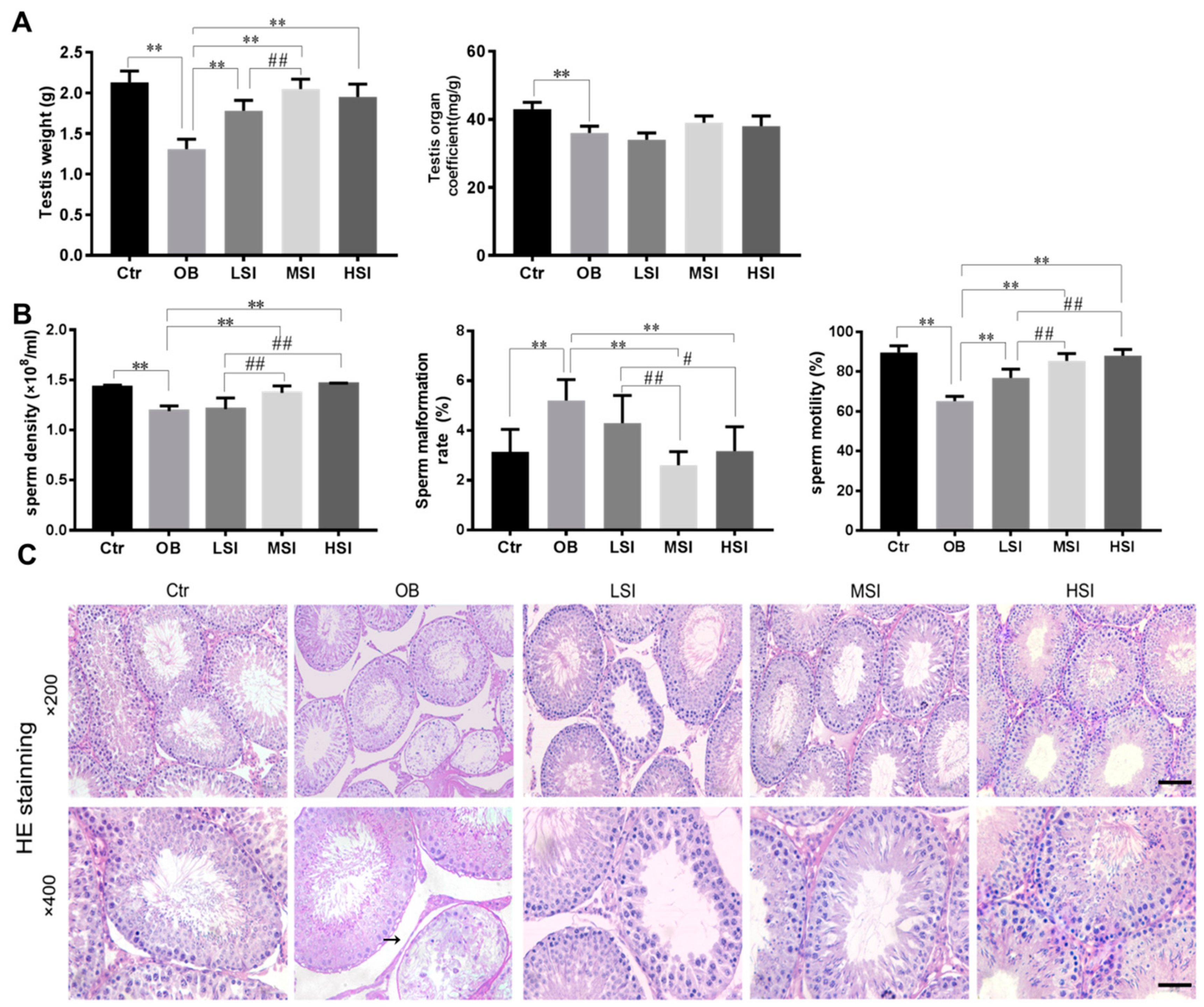
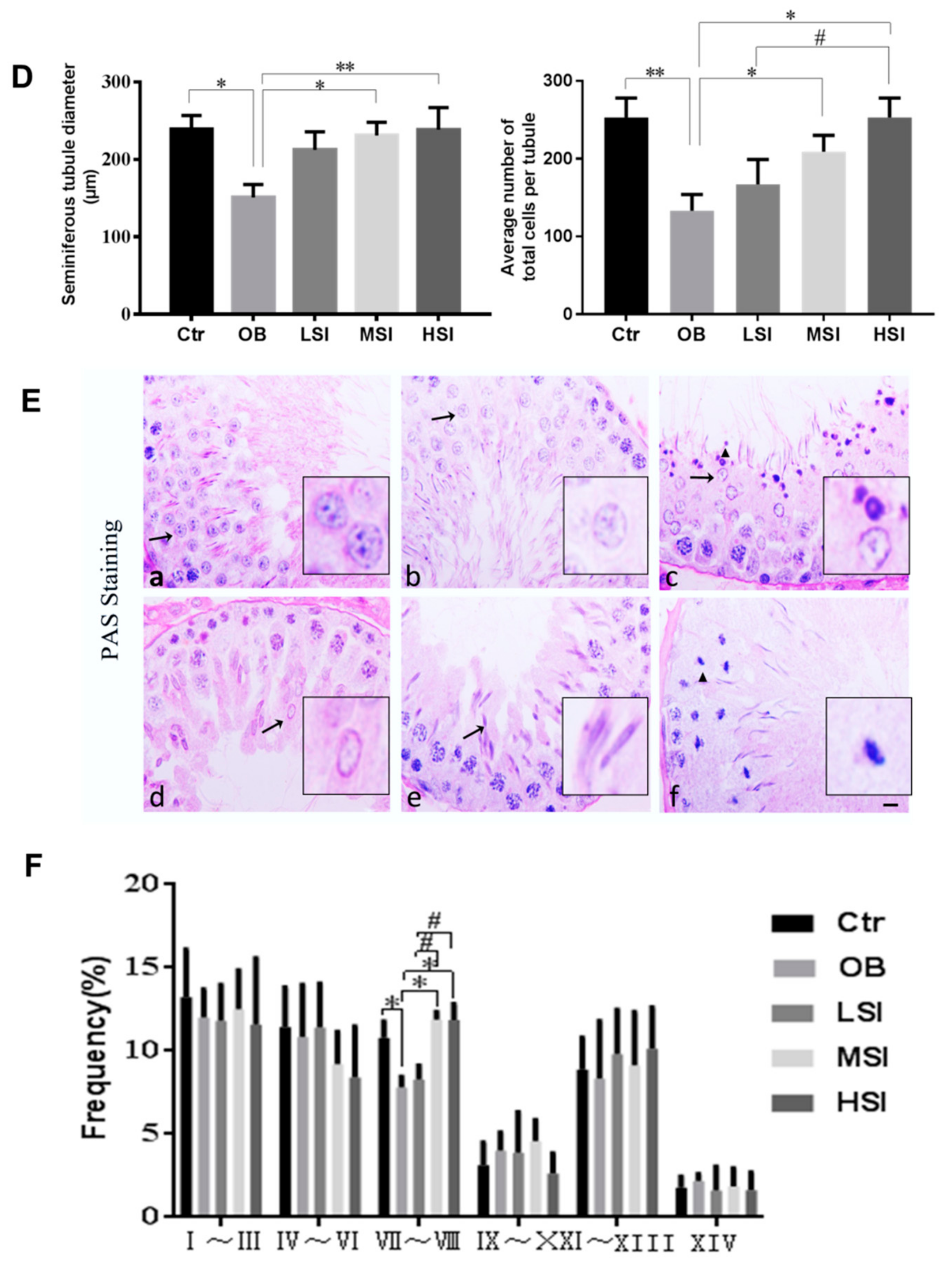
2.1. SIF Promote Spermatogenesis in DIO Male Rats
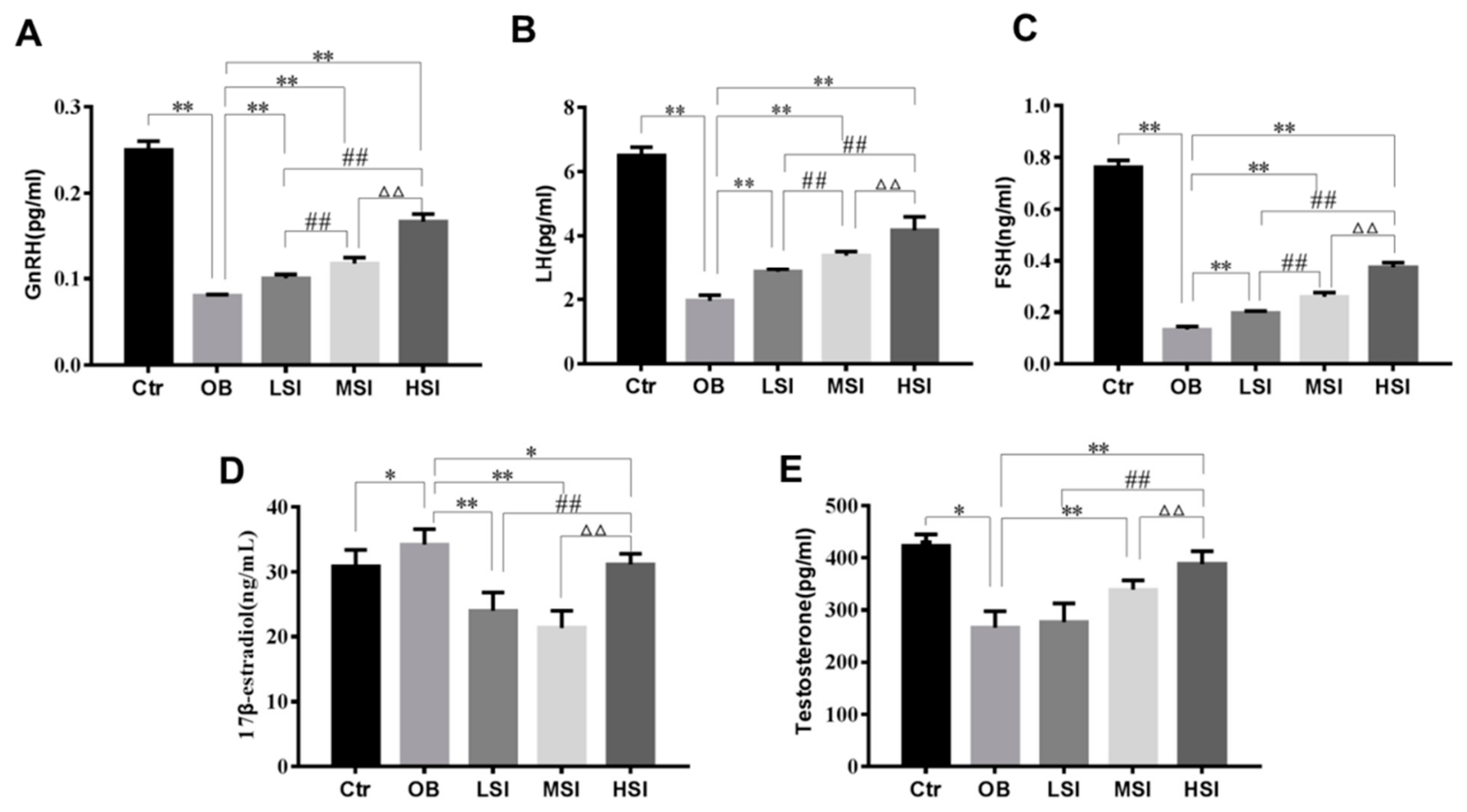
2.2. Effects of SIF on Plasma Reproductive Hormone in DIO Male Rats
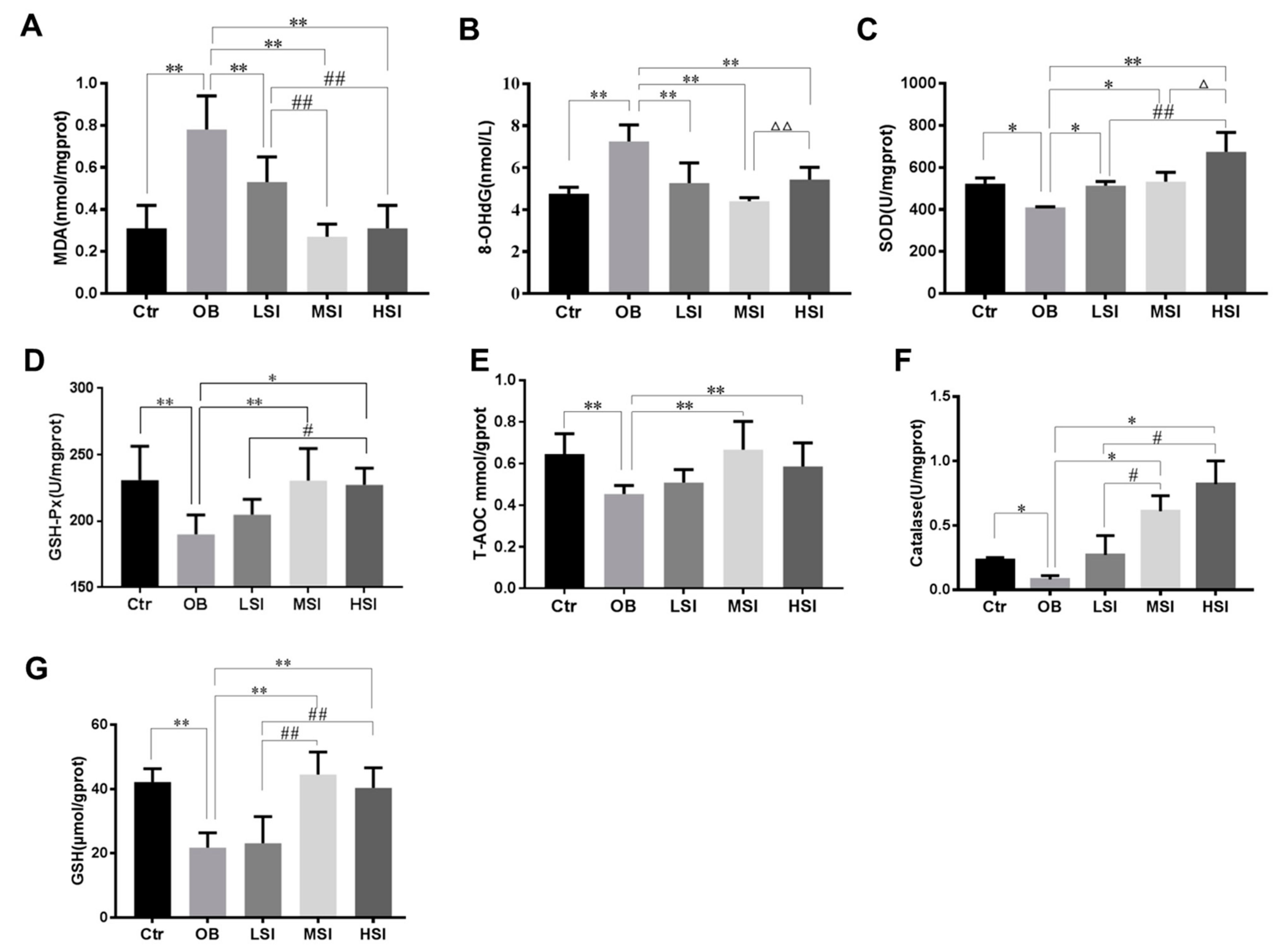
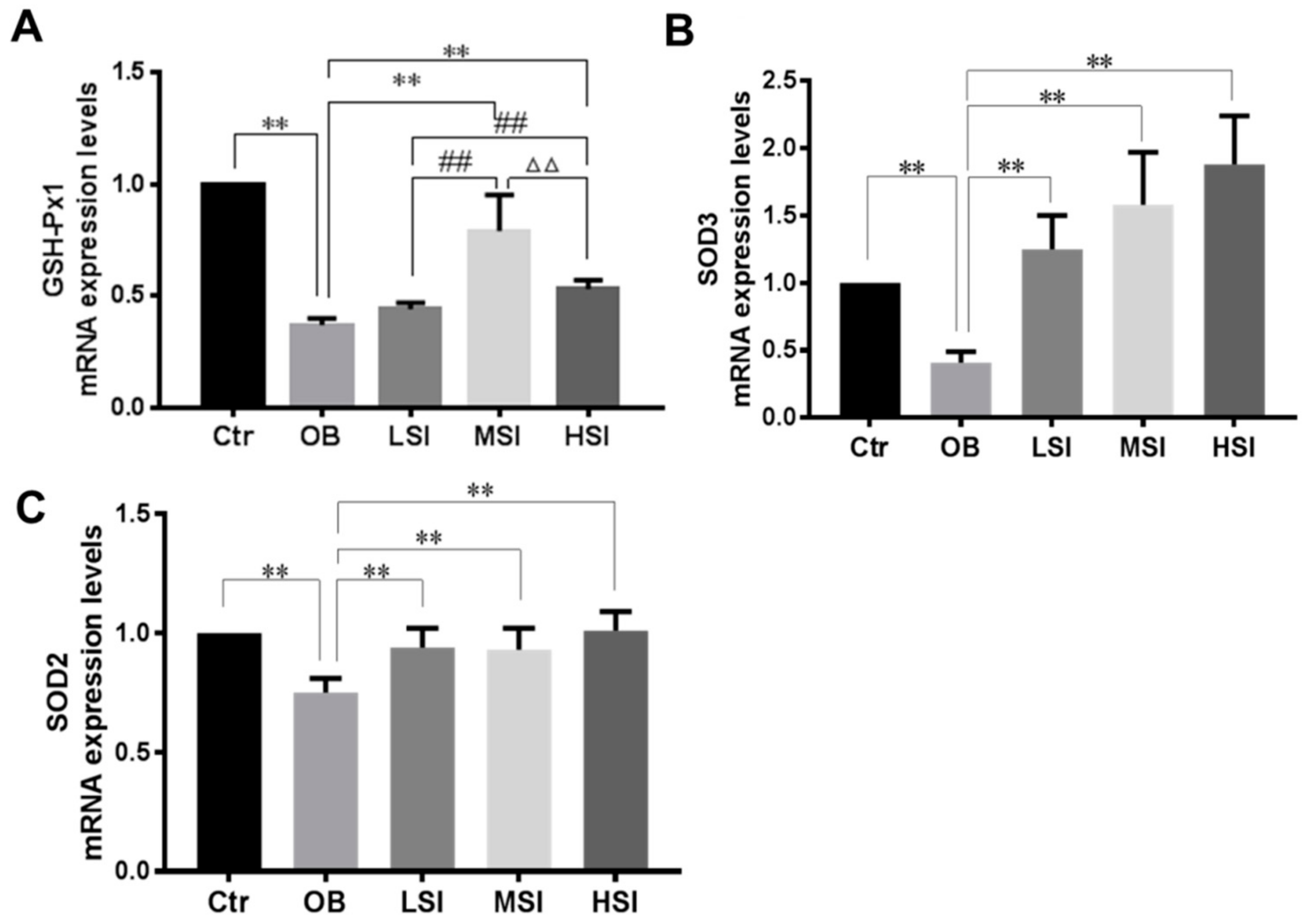
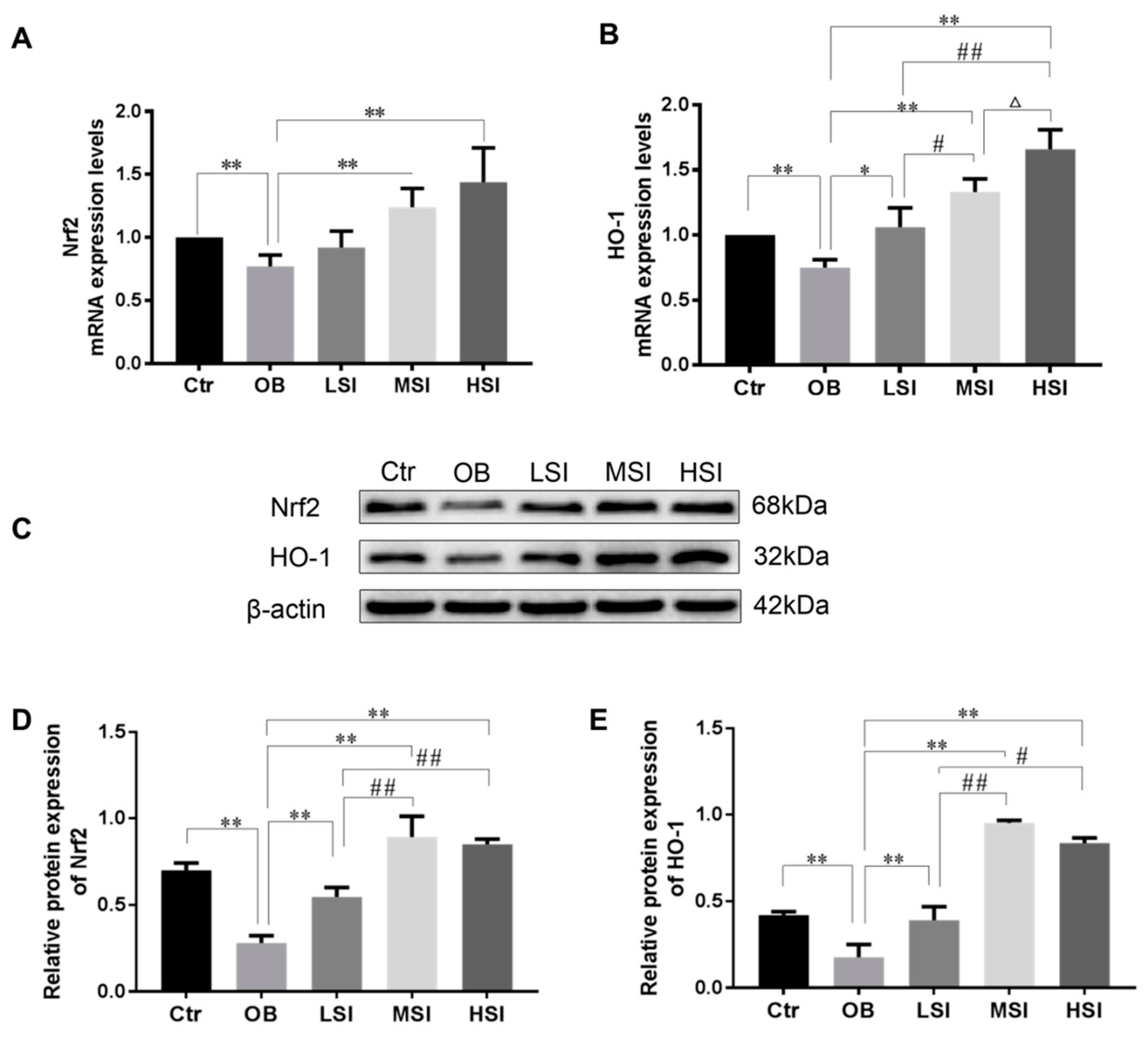
2.3. Effects of SIF on Testis Oxidative Stress in DIO Male Rats
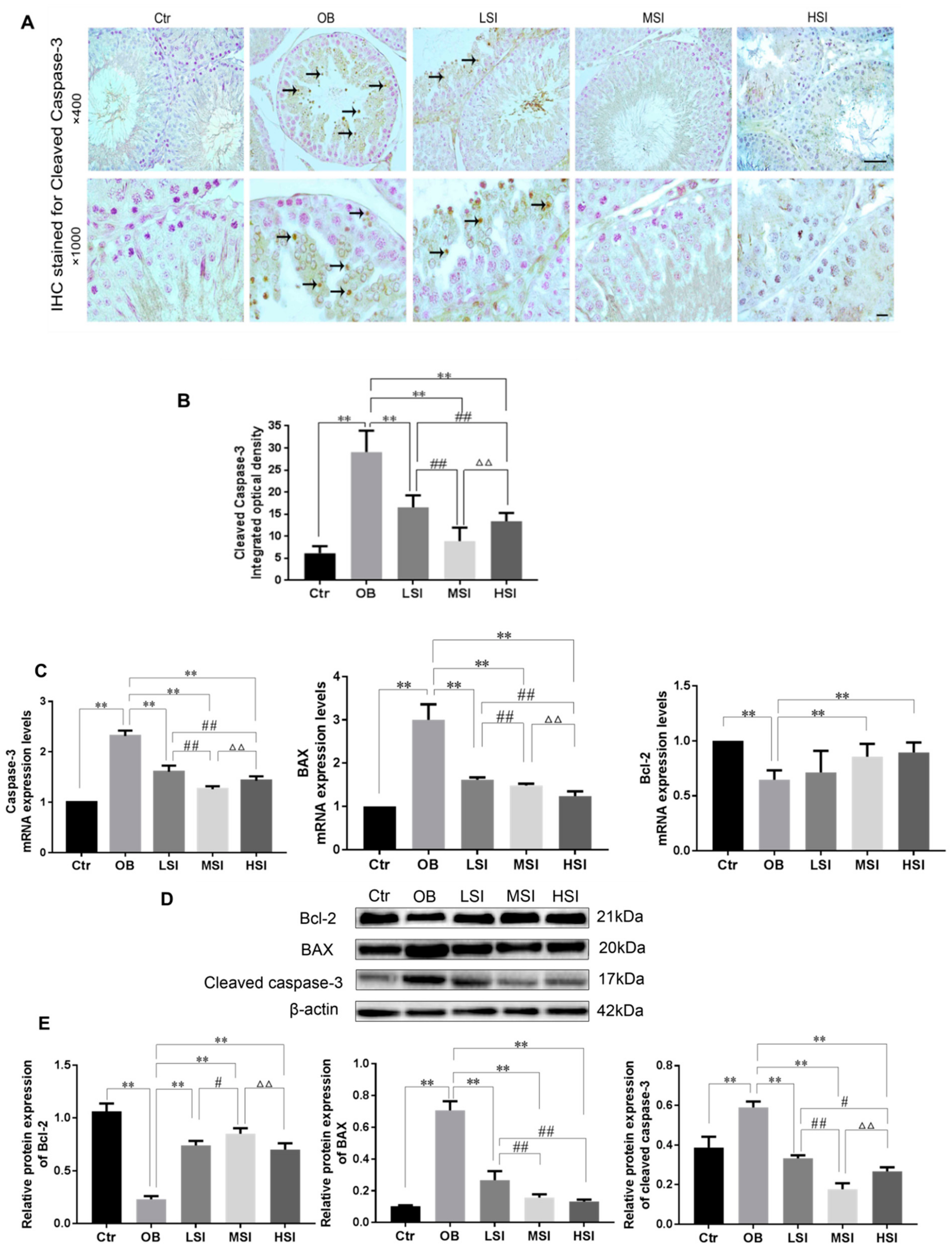
2.4. Effects of SIF on the Testis Cell Apoptosis in DIO Male Rats
3. Discussion
4. Materials and Methods
4.1. Animals Care, Diets and Experimental Design
4.2. Collection of Biological Samples
4.3. Sperm Count, Motility, and Morphology
4.4. Histological Analysis and Staging of Seminiferous Epithelium
4.5. Hormone Assays
4.6. Oxidative Stress Markers in the Testis
4.7. Immunohistochemistry Stainning
4.8. Detection of mRNA Expression in the Testis by Quantitative Realtime PCR
4.9. Western Blotting
4.10. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Younossi, Z.M. The epidemiology of nonalcoholic steatohepatitis. Clin. Liver Dis. 2018, 11, 92–94. [Google Scholar] [CrossRef] [PubMed]
- Kelly, T.; Yang, W.; Chen, C.-S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Bouter, L.M.; McQuillan, G.M.; Wener, M.H.; Harris, T.B. Elevated c-reactive protein levels in overweight and obese adults. JAMA 1999, 282, 2131–2135. [Google Scholar] [CrossRef] [PubMed]
- Bellentani, S.; Scaglioni, F.; Marino, M.; Bedogni, G. Epidemiology of non-alcoholic fatty liver disease. Dig. Dis. 2010, 28, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, W.A.; Buchanan, T.A. Obesity and hypertension. Endocrinol. Metab. Clin. North Am. 1994, 23, 405–427. [Google Scholar] [CrossRef]
- Cazettes, F.; Cohen, J.I.; Yau, P.L.; Talbot, H.; Convit, A. Obesity-mediated inflammation may damage the brain circuit that regulates food intake. Brain Res. 2011, 1373, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Thun, M.J. Obesity and cancer. Oncogene 2004, 23, 6365–6378. [Google Scholar] [CrossRef] [PubMed]
- Lamm, S.; Chidakel, A.; Bansal, R. Obesity and hypogonadism. Urol. Clin. 2016, 43, 239–245. [Google Scholar] [CrossRef]
- Hadjkacem Loukil, L.; Hadjkacem, H.; Bahloul, A.; Ayadi, H. Relation between male obesity and male infertility in a T unisian population. Andrologia 2015, 47, 282–285. [Google Scholar] [CrossRef]
- Adewoyin, M.; Ibrahim, M.; Roszaman, R.; Isa, M.L.M.; Alewi, N.A.M.; Rafa, A.A.A.; Anuar, M.N.N. Male infertility: the effect of natural antioxidants and phytocompounds on seminal oxidative stress. Diseases 2017, 5, 9. [Google Scholar] [CrossRef]
- Hammoud, A.O.; Gibson, M.; Peterson, C.M.; Meikle, A.W.; Carrell, D.T. Impact of male obesity on infertility: a critical review of the current literature. Fertil. Steril. 2008, 90, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest. 2004, 114, 1752–1762. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; De Iuliis, G.N. On the possible origins of DNA damage in human spermatozoa. MHR Basic Sci. Reprod. Med. 2010, 16, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; Smith, T.B.; Jobling, M.S.; Baker, M.A.; De Iuliis, G.N. Oxidative stress and male reproductive health. Asian J. Androl. 2014, 16, 31–38. [Google Scholar] [CrossRef] [PubMed]
- John Aitken, R.; Clarkson, J.S.; Fishel, S. Generation of reactive oxygen species, lipid peroxidation, and human sperm function. Biol. Reprod. 1989, 41, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Alhashem, F.; Alkhateeb, M.; Sakr, H.; Alshahrani, M.; Alsunaidi, M.; Elrefaey, H.; Alessa, R.; Sarhan, M.; Eleawa, S.M.; Khalil, M.A. Exercise protects against obesity induced semen abnormalities via downregulating stem cell factor, upregulating Ghrelin and normalizing oxidative stress. EXCLI J. 2014, 13, 551–572. [Google Scholar]
- Seo, H.; Lee, I.-K. The role of Nrf2: Adipocyte differentiation, obesity, and insulin resistance. Oxid. Med. Cell Longev. 2013. [Google Scholar] [CrossRef]
- Surh, Y.-J.; Kundu, J.K.; Na, H.-K. Nrf2 as a master redox switch in turning on the cellular signaling involved in the induction of cytoprotective genes by some chemopreventive phytochemicals. Planta Med. 2008, 74, 1526–1539. [Google Scholar] [CrossRef]
- Ng, C.P. Effects of habbatus sauda (Nigella sativa) oil on sperm and testis parameters of nicotine treated rats/Ng Cho Ping. Evid. Based Complement. Alternat. Med. 2015. [Google Scholar] [CrossRef]
- Marzony, E.T.; Ghanei, M.; Panahi, Y. Relationship of oxidative stress with male infertility in sulfur mustard-exposed injuries. Asian Pac. J. Reprod. 2016, 5, 1–9. [Google Scholar] [CrossRef]
- Do Nascimento, A.; De Lima, E.; Boëchat, G.; Meyrelles, S.; Bissoli, N.; Lenz, D.; Endringer, D.; de Andrade, T. Testosterone induces apoptosis in cardiomyocytes by increasing proapoptotic signaling involving tumor necrosis factor-α and renin angiotensin system. Hum. Exp. Toxicol. 2015, 34, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Katib, A. Mechanisms linking obesity to male infertility. Cent. European J. Urol. 2015, 68, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Adlercreutz, H.; Mazur, W. Phyto-oestrogens and Western diseases. Ann. Med. 1997, 29, 95–120. [Google Scholar] [CrossRef] [PubMed]
- Kurrat, A.; Blei, T.; Kluxen, F.M.; Mueller, D.R.; Piechotta, M.; Soukup, S.T.; Kulling, S.E.; Diel, P. Lifelong exposure to dietary isoflavones reduces risk of obesity in ovariectomized Wistar rats. Mol. Nutr. Food Res. 2015, 59, 2407–2418. [Google Scholar] [CrossRef]
- Messina, M.; Messina, V. Soyfoods, soybean isoflavones, and bone health: A brief overview. J. Renal Nutr. 2000, 10, 63–68. [Google Scholar] [CrossRef]
- Mizushina, Y.; Shiomi, K.; Kuriyama, I.; Takahashi, Y.; Yoshida, H. Inhibitory effects of a major soy isoflavone, genistein, on human DNA topoisomerase II activity and cancer cell proliferation. Int. J. Oncol. 2013, 43, 1117–1124. [Google Scholar] [CrossRef]
- Wagner, J.D.; Anthony, M.S.; Cline, J.M. Soy phytoestrogens: Research on benefits and risks. Clin. Obstet. Gynecol. 2001, 44, 843–852. [Google Scholar] [CrossRef]
- Barbosa, A.C.; Lajolo, F.M.; Genovese, M.I. Effect of free or protein-associated soy isoflavones on the antioxidant status in rats. J. Sci. Food Agric. 2011, 91, 721–731. [Google Scholar] [CrossRef]
- Setchell, K. Phytoestrogens: the biochemistry, physiology, and implications for human health of soy isoflavones. Am. J. Clin. Nutr. 1998. [Google Scholar] [CrossRef]
- Yoon, G.; Park, S. Antioxidant action of soy isoflavones on oxidative stress and antioxidant enzyme activities in exercised rats. Nutr. Res. Pract. 2014, 8, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Mahn, K.; Borrás, C.; Knock, G.A.; Taylor, P.; Khan, I.Y.; Sugden, D.; Poston, L.; Ward, J.P.; Sharpe, R.M.; Viña, J. Dietary soy isoflavone induced increases in antioxidant and eNOS gene expression lead to improved endothelial function and reduced blood pressure in vivo. FASEB J. 2005, 19, 1755–1757. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, X.; Luo, Q.; Huang, C.; Liu, W.; Chen, Z. Effects of soy isoflavones on testosterone synthetase in diet-induced obesity male rats. Int. J. Clin. Exp. Pathol. 2017, 10, 9202–9212. [Google Scholar]
- Shiraishi, K.; Matsuyama, H. Gonadotoropin actions on spermatogenesis and hormonal therapies for spermatogenic disorders. Endocr. J. 2017. [CrossRef] [PubMed]
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Kodama, H.; Yamaguchi, R.; Fukuda, J.; Kasai, H.; Tanaka, T. Increased oxidative deoxyribonucleic acid damage in the spermatozoa of infertile male patients. Fertil. Steril. 1997, 68, 519–524. [Google Scholar] [CrossRef]
- Pourhassanali, N.; Roshan-Milani, S.; Kheradmand, F.; Motazakker, M.; Bagheri, M.; Saboory, E. Zinc attenuates ethanol-induced Sertoli cell toxicity and apoptosis through caspase-3 mediated pathways. Reprod. Toxicol. 2016, 61, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-H.; Lee, I.-C.; Ko, J.-W.; Shin, I.-S.; Moon, C.; Kim, S.-H.; Heo, J.-D.; Kim, J.-C. Mechanism of protection by diallyl disulfide against cyclophosphamide-induced spermatotoxicity and oxidative stress in rats. Mol. Cell. Toxicol. 2016, 12, 301–312. [Google Scholar] [CrossRef]
- Yamori, Y. Worldwide epidemic of obesity: Hope for Japanese diets. Clin. Exp. Pharmacol. Physiol. 2004. [Google Scholar] [CrossRef]
- Tsao, C.-W.; Liu, C.-Y.; Chou, Y.-C.; Cha, T.-L.; Chen, S.-C.; Hsu, C.-Y. Exploration of the association between obesity and semen quality in a 7630 male population. PLoS ONE 2015. [Google Scholar] [CrossRef]
- Ramaraju, G.; Teppala, S.; Prathigudupu, K.; Kalagara, M.; Thota, S.; Kota, M.; Cheemakurthi, R. Association between obesity and sperm quality. Andrologia 2018. [Google Scholar] [CrossRef] [PubMed]
- Navya, H.; Yajurvedi, H.N. Obesity causes weight increases in prepubertal and pubertal male offspring and is related to changes in spermatogenesis and sperm production in rats. Reprod. Fertil. Develop. 2017, 29, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Pang, D.; Luo, Q.; Chen, X.; Gao, Q.; Shi, L.; Liu, W.; Zou, Y.; Li, L.; Chen, Z. Soy Isoflavones regulate lipid metabolism through an AKT/mTORC1 pathway in diet-induced obesity (DIO) male rats. Molecules 2016. [Google Scholar] [CrossRef] [PubMed]
- Leblond, C.; Clermont, Y. Definition of the stages of the cycle of the seminiferous epithelium in the rat. Ann. N. Y. Acad. Sci. 1952, 55, 548–573. [Google Scholar] [CrossRef] [PubMed]
- Clermont, Y.; Perey, B. The stages of the cycle of the seminiferous epithelium of the rat: practical definitions in PA-Schiff-hematoxylin and hematoxylin-eosin stained sections. Rev. Can. Biol. 1957, 16, 451–462. [Google Scholar] [PubMed]
- Russell, L.D.; Ettlin, R.A.; Hikim, A.P.S.; Clegg, E.D. Histological and histopathological evaluation of the testis. Int. J. Androl. 1993, 16, 83. [Google Scholar] [CrossRef]
- Turner, T.T.; Lysiak, J.J. Oxidative stress: a common factor in testicular dysfunction. Int. J. Androl. 2008, 29, 488–498. [Google Scholar] [CrossRef]
- Jensen, B. Rat testicular lipids and dietary isomeric fatty acids in essential fatty acid deficiency. Lipids 1976, 11, 179–188. [Google Scholar] [CrossRef]
- Yin, J.; Ren, W.; Yang, G.; Duan, J.; Huang, X.; Fang, R.; Li, C.; Li, T.; Yin, Y.; Hou, Y. L-Cysteine metabolism and its nutritional implications. Mol. Nutr. Food Res. 2016, 60, 134–146. [Google Scholar] [CrossRef]
- Yin, J.; Ren, W.; Liu, G.; Duan, J.; Yang, G.; Wu, L.; Li, T.; Yin, Y. Birth oxidative stress and the development of an antioxidant system in newborn piglets. Free Radical Res. 2013, 47, 1027–1035. [Google Scholar] [CrossRef]
- Gong, Y.; Wu, J.; Huang, Y.; Shen, S.; Han, X. Nonylphenol induces apoptosis in rat testicular Sertoli cells via endoplasmic reticulum stress. Toxicol. Lett. 2009, 186, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.-Y.; Liu, Y.-Q.; Shen, H.-M.; Chia, S.E.; Ong, C.N. Does the increase of 8-hydroxydeoxyguanosine lead to poor sperm quality? Mutat. Res. 1997, 381, 77–82. [Google Scholar] [CrossRef]
- Duan, P.; Hu, C.; Butler, H.J.; Quan, C.; Chen, W.; Huang, W.; Tang, S.; Zhou, W.; Yuan, M.; Shi, Y. Effects of 4-nonylphenol on spermatogenesis and induction of testicular apoptosis through oxidative stress-related pathways. Reprod. Toxicol. 2016, 62, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Eleawa, S.M.; Alkhateeb, M.A.; Alhashem, F.H.; Bin-Jaliah, I.; Sakr, H.F.; Elrefaey, H.M.; Elkarib, A.O.; Alessa, R.M.; Haidara, M.A.; Shatoor, A.S. Resveratrol reverses cadmium chloride-induced testicular damage and subfertility by downregulating p53 and Bax and upregulating gonadotropins and Bcl-2 gene expression. J. Reprod. Develop. 2014, 60, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Li, N.C.; Wei, X.X.; Hu, Y.L.; Hou, X.; Xu, H. Aerobic exercise blocks interleukin-6 levels and germ cell apoptosis in obese rats. Andrologia 2018. [Google Scholar] [CrossRef]
- Hammami, I.; Amara, S.; Benahmed, M.; El May, M.V.; Mauduit, C. Chronic crude garlic-feeding modified adult male rat testicular markers: mechanisms of action. Reprod. Biol. Endocrinol. 2009. [Google Scholar] [CrossRef]
- Jung, E.B.; Lee, C.S. Baicalein attenuates proteasome inhibition-induced apoptosis by suppressing the activation of the mitochondrial pathway and the caspase-8-and Bid-dependent pathways. Eur. J. Pharmacol. 2014, 730, 116–124. [Google Scholar] [CrossRef]
- Jia, Y.-F.; Feng, Q.; Ge, Z.-Y.; Guo, Y.; Zhou, F.; Zhang, K.-S.; Wang, X.-W.; Lu, W.-H.; Liang, X.-W.; Gu, Y.-Q. Obesity impairs male fertility through long-term effects on spermatogenesis. BMC Urol. 2018. [Google Scholar] [CrossRef]
- Rato, L.; Alves, M.; Cavaco, J.; Oliveira, P. High-energy diets: a threat for male fertility? Obes. Rev. 2014, 15, 996–1007. [Google Scholar] [CrossRef]
- Doerge, D.R.; Sheehan, D.M. Goitrogenic and estrogenic activity of soy isoflavones. Environ. Health Perspect. 2002, 110, 349–353. [Google Scholar] [CrossRef]
- Vigueras-Villaseñor, R.M.; Rojas-Castañeda, J.C.; Chavez-Saldana, M.; Gutierrez-Perez, O.; García-Cruz, M.E.; Cuevas-Alpuche, O.; Reyes-Romero, M.M.; Zambrano, E. Alterations in the spermatic function generated by obesity in rats. Acta Histochem. 2011, 113, 214–220. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, L.; McLachlan, R.L.; Wreford, N.G.; de Kretser, D.M. Testosterone withdrawal promotes stage-specific detachment of round spermatids from the rat seminiferous epithelium. Biol. Reprod. 1996, 55, 895–901. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, L.; Meachem, S.; Stanton, P.; McLachlan, R. Endocrine regulation of spermatogenesis. In Physiology of Reproduction, 3rd ed.; Neill, J.D., Ed.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 1, pp. 1017–1069. [Google Scholar]
- Wong, C.-H.; Xia, W.; Lee, N.P.; Mruk, D.D.; Lee, W.M.; Cheng, C.Y. Regulation of ectoplasmic specialization dynamics in the seminiferous epithelium by focal adhesion-associated proteins in testosterone-suppressed rat testes. Endocrinology 2005, 146, 1192–1204. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, H. Soybean isoflavones ameliorate ischemic cardiomyopathy by activating Nrf2-mediated antioxidant responses. Food Funct. 2017, 8, 2935–2944. [Google Scholar] [CrossRef] [PubMed]
- Froyen, E.B.; Steinberg, F.M. Soy isoflavones increase quinone reductase in hepa-1c1c7 cells via estrogen receptor beta and nuclear factor erythroid 2-related factor 2 binding to the antioxidant response element. J. Nutr. Biochem. 2011, 22, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Yu, H.L.; Ma, W.W.; Xi, Y.D.; Zhao, X.; Yuan, L.H.; Feng, J.F.; Xiao, R. Soy isoflavone attenuates brain mitochondrial oxidative stress induced by beta-amyloid peptides 1–42 injection in lateral cerebral ventricle. J. Neurosci. Res. 2013, 91, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; An, Y.; Lv, C.; Ma, W.; Xi, Y.; Xiao, R. Dietary soybean isoflavones in Alzheimer’s disease prevention. Asia Pac. J. Clin. Nutr. 2018, 27, 946. [Google Scholar] [PubMed]
- Singh, S.; Singh, S.K. Prepubertal exposure to perfluorononanoic acid interferes with spermatogenesis and steroidogenesis in male mice. Ecotox. Environ. Safe. 2019, 170, 590–599. [Google Scholar] [CrossRef]
- Niture, S.K.; Jaiswal, A.K. Nrf2-induced antiapoptotic Bcl-xL protein enhances cell survival and drug resistance. Free Radical Biol. Med. 2013, 57, 119–131. [Google Scholar] [CrossRef]
- Niture, S.K.; Jaiswal, A.K. Nrf2 protein up-regulates antiapoptotic protein Bcl-2 and prevents cellular apoptosis. J. Biol. Chem. 2012, 287, 9873–9886. [Google Scholar] [CrossRef]
- Atanassova, N.; McKinnell, C.; Fisher, J.; Sharpe, R.M. Neonatal treatment of rats with diethylstilboestrol (DES) induces stromal-epithelial abnormalities of the vas deferens and cauda epididymis in adulthood following delayed basal cell development. Reproduvtion 2005, 129, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, B.; Thorstensen, E.; Ponnampalam, A.; Mitchell, M. Transplacental transfer and biotransformation of genistein in human placenta. Placenta 2010, 31, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Lehraiki, A.; Chamaillard, C.; Krust, A.; Habert, R.; Levacher, C. Genistein impairs early testosterone production in fetal mouse testis via estrogen receptor alpha. Toxicol. in Vitro 2011, 25, 1542–1547. [Google Scholar] [CrossRef] [PubMed]
- Musameh, N.I.; Zin, S.R.M.; Kassim, N.M. Effects of genistein on male sprague dawley rats reproductive development. Biomed. Res. 2014, 25, 391–400. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
| Gene | Primer Sequences (5′-3′) | Length (bp) | |
|---|---|---|---|
| Forward | Reverse | ||
| Nrf2 | GAGACGGCCATGACTGAT | TGTGGAACATCTGGTAGACGGC | 196 |
| HO-1 | CTGGAATGGAAGGAGATGCC | TCAGAACAGCCGCCTCTACCG | 132 |
| GSH-Px1 | CAGTTCGGACATCAGGAGAAT | AGAGCGGGTGAGCCTTCT | 139 |
| Sod2 | AAAGGAGAGTTGCTGGAGGC | TGATTAGAGCAGGCGGCAAT | 161 |
| Sod3 | TTGTTCTGCAACCTGCTACTGG | AGTGCGTGTCGCCTATCTTCTC | 124 |
| Caspase-3 | CGGACCTGTGGACCTGAAAA | CGGCCTCCACTGGTATCTTC | 182 |
| Bcl-2 | GGCATCTTCTCCTTCCAGCC | CGACGAGAGAAGTCATCCCC | 173 |
| BAX | TGGCGATGAACTGGACAACA | CCCAGTTGAAGTTGCCGTCT | 125 |
| β-actin | ACGGTCAGGTCATCACTATCG | GGCATAGAGGTCTTTACGGATG | 155 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, Q.; Li, Y.; Huang, C.; Cheng, D.; Ma, W.; Xia, Y.; Liu, W.; Chen, Z. Soy Isoflavones Improve the Spermatogenic Defects in Diet-Induced Obesity Rats through Nrf2/HO-1 Pathway. Molecules 2019, 24, 2966. https://doi.org/10.3390/molecules24162966
Luo Q, Li Y, Huang C, Cheng D, Ma W, Xia Y, Liu W, Chen Z. Soy Isoflavones Improve the Spermatogenic Defects in Diet-Induced Obesity Rats through Nrf2/HO-1 Pathway. Molecules. 2019; 24(16):2966. https://doi.org/10.3390/molecules24162966
Chicago/Turabian StyleLuo, Qihui, Yifan Li, Chao Huang, Dongjing Cheng, Wenjing Ma, Yu Xia, Wentao Liu, and Zhengli Chen. 2019. "Soy Isoflavones Improve the Spermatogenic Defects in Diet-Induced Obesity Rats through Nrf2/HO-1 Pathway" Molecules 24, no. 16: 2966. https://doi.org/10.3390/molecules24162966
APA StyleLuo, Q., Li, Y., Huang, C., Cheng, D., Ma, W., Xia, Y., Liu, W., & Chen, Z. (2019). Soy Isoflavones Improve the Spermatogenic Defects in Diet-Induced Obesity Rats through Nrf2/HO-1 Pathway. Molecules, 24(16), 2966. https://doi.org/10.3390/molecules24162966





