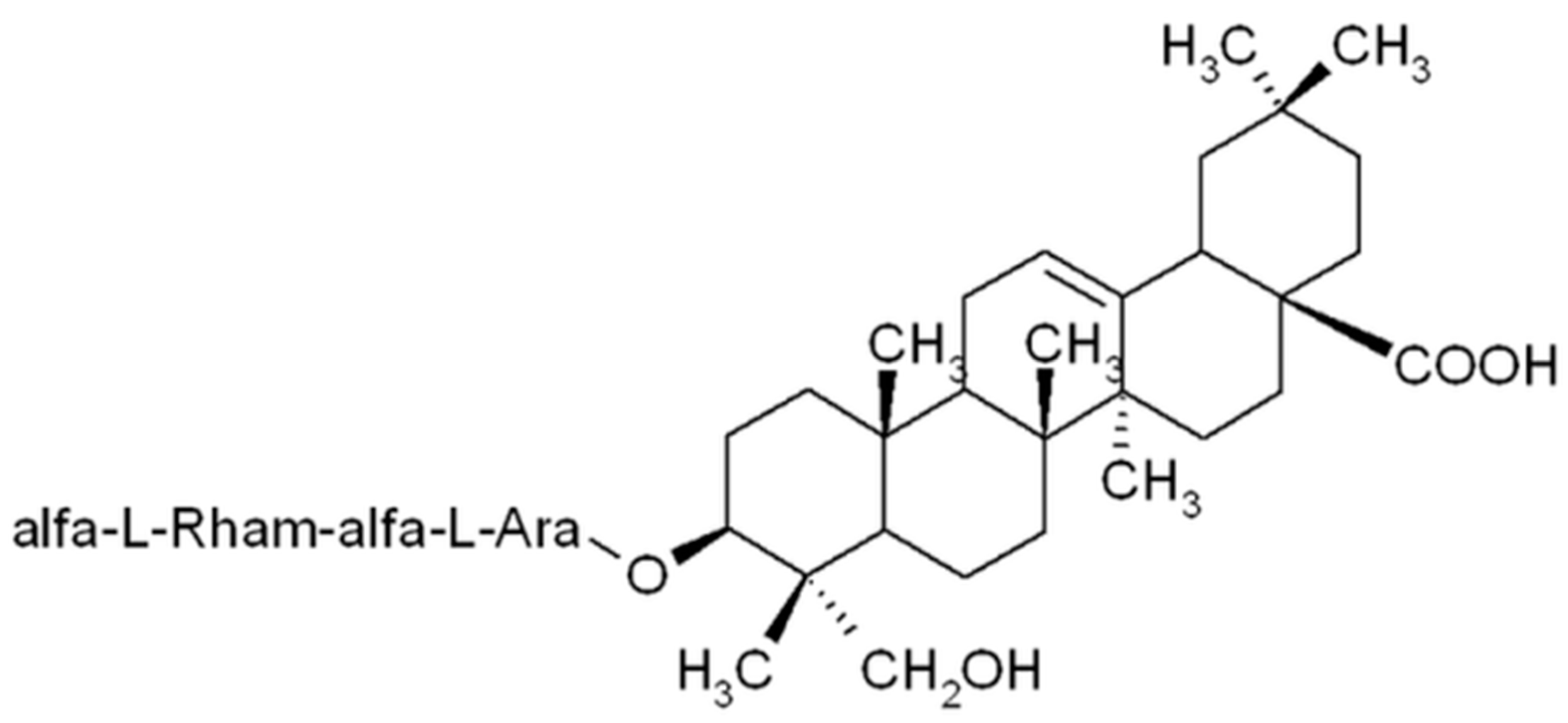
Alpha-Hederin, the Active Saponin of Nigella sativa, as an Anticancer Agent Inducing Apoptosis in the SKOV-3 Cell Line
Abstract
1. Introduction
2. Results
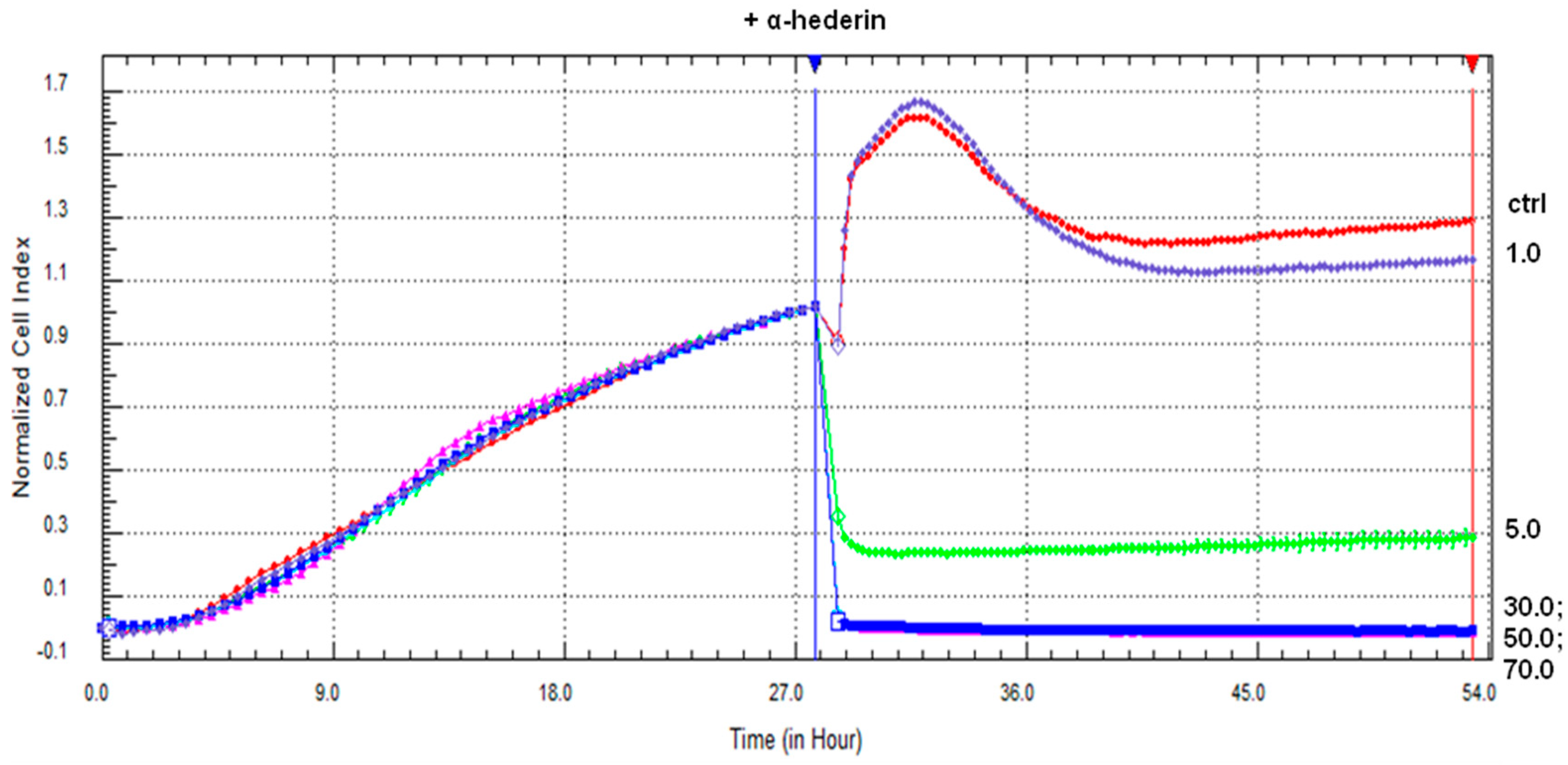
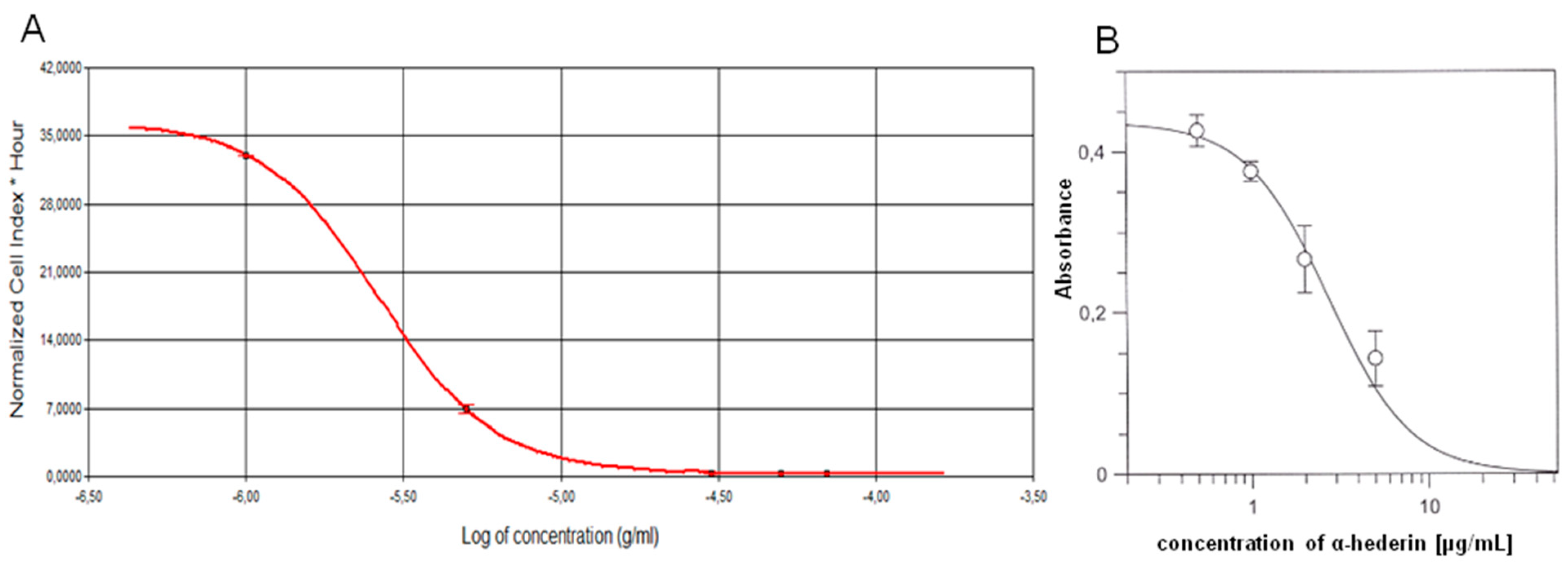
2.1. α-HN Decreased SKOV-3 Cells’ Viability
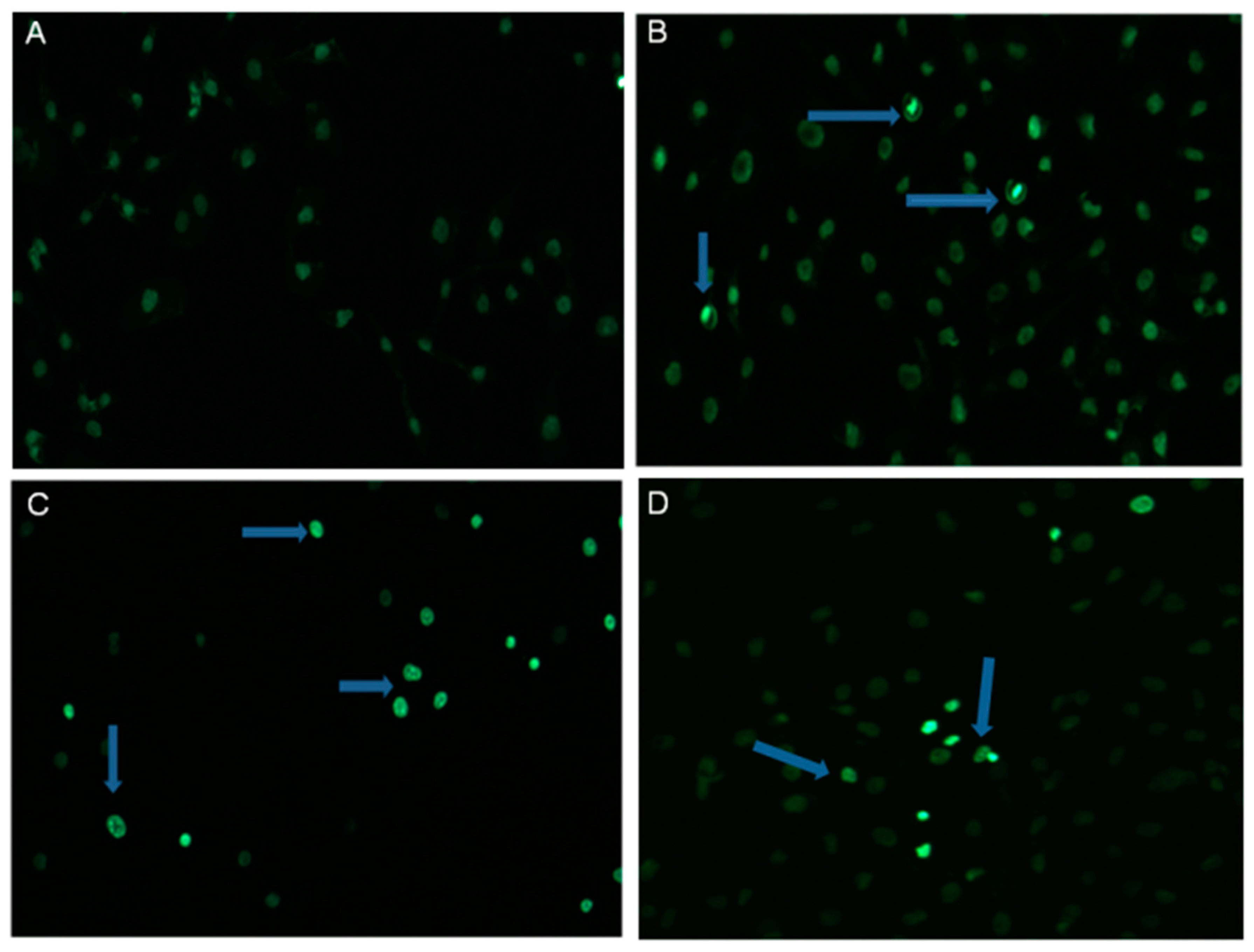
2.2. α-HN Triggered Changes in SKOV-3 Nuclei
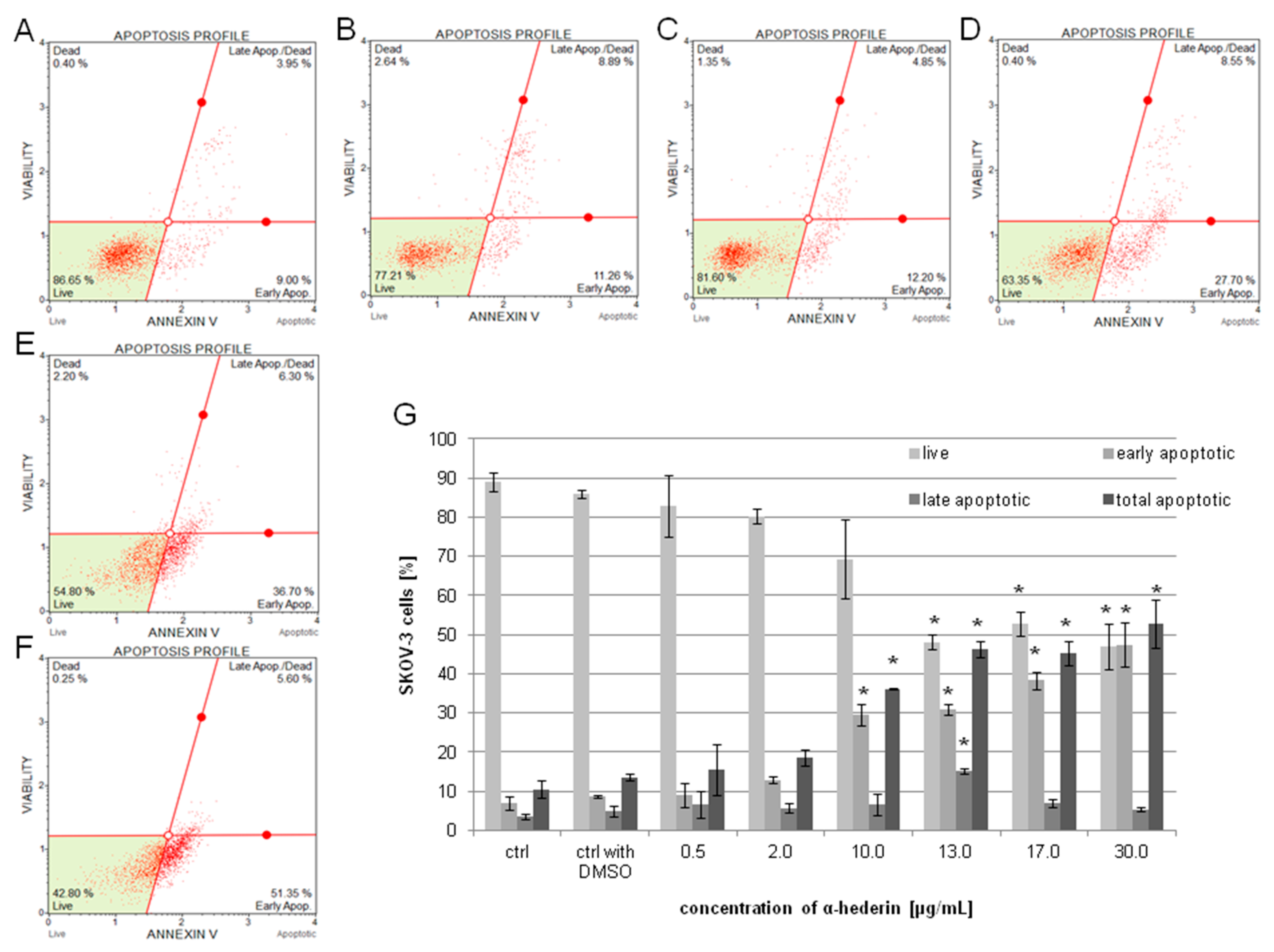
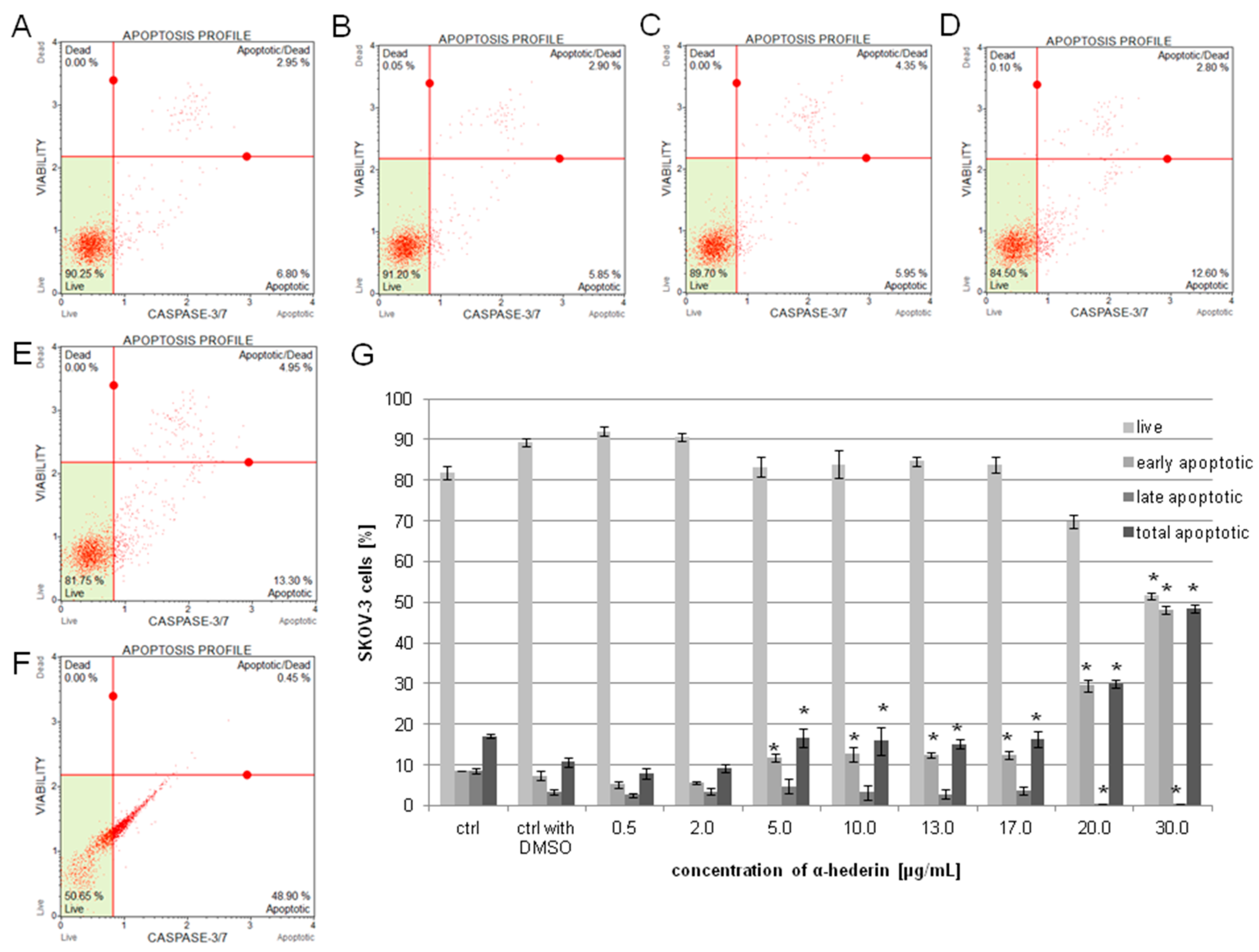
2.3. α-HN Induced Apoptosis in the SKOV-3 Cell Line
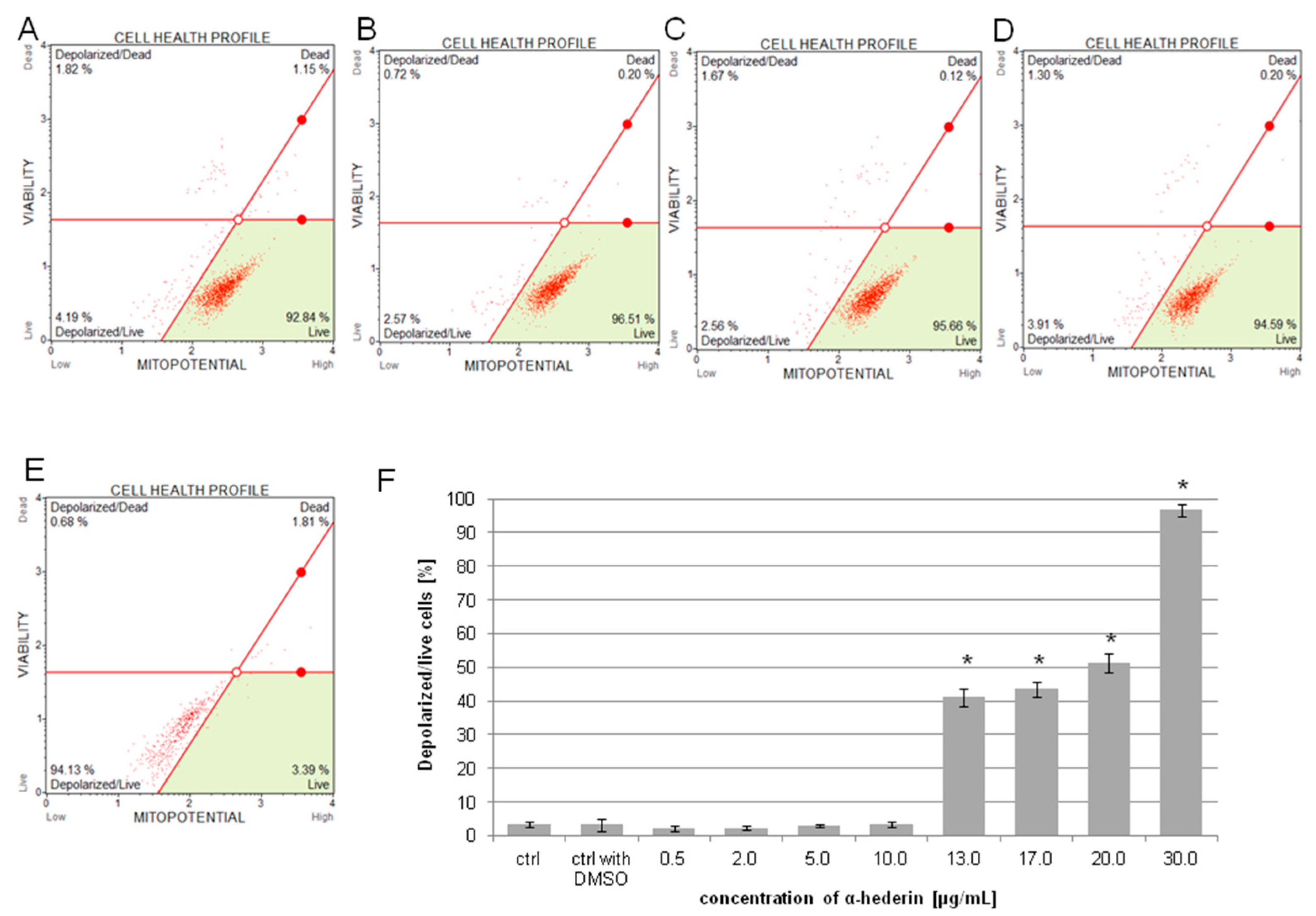
2.4. α-HN Decreased Mitochondrial Membrane Potential (ΔΨm) in SKOV-3 Cells
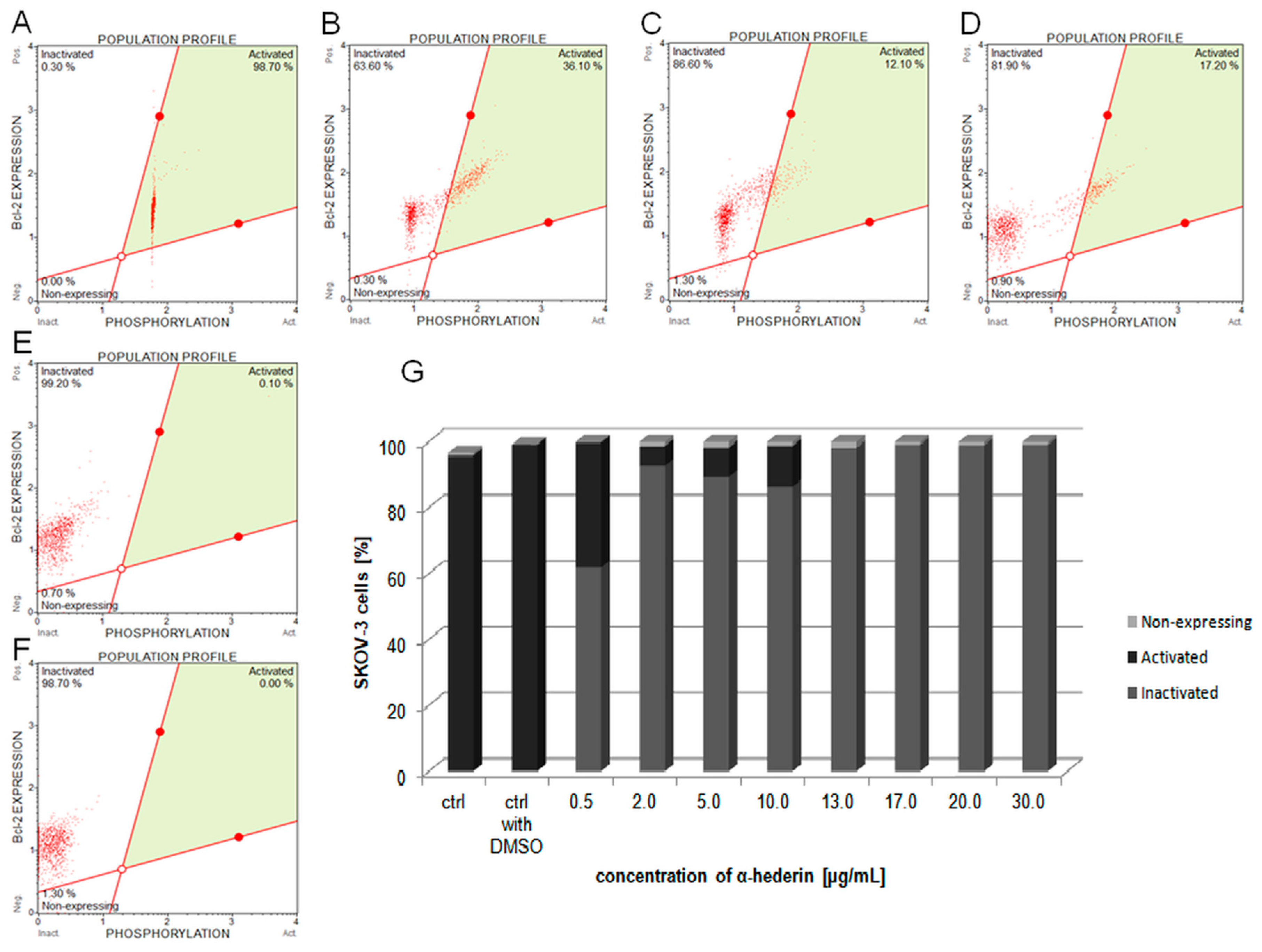
2.5. α-HN Inactivated Bcl-2 Protein
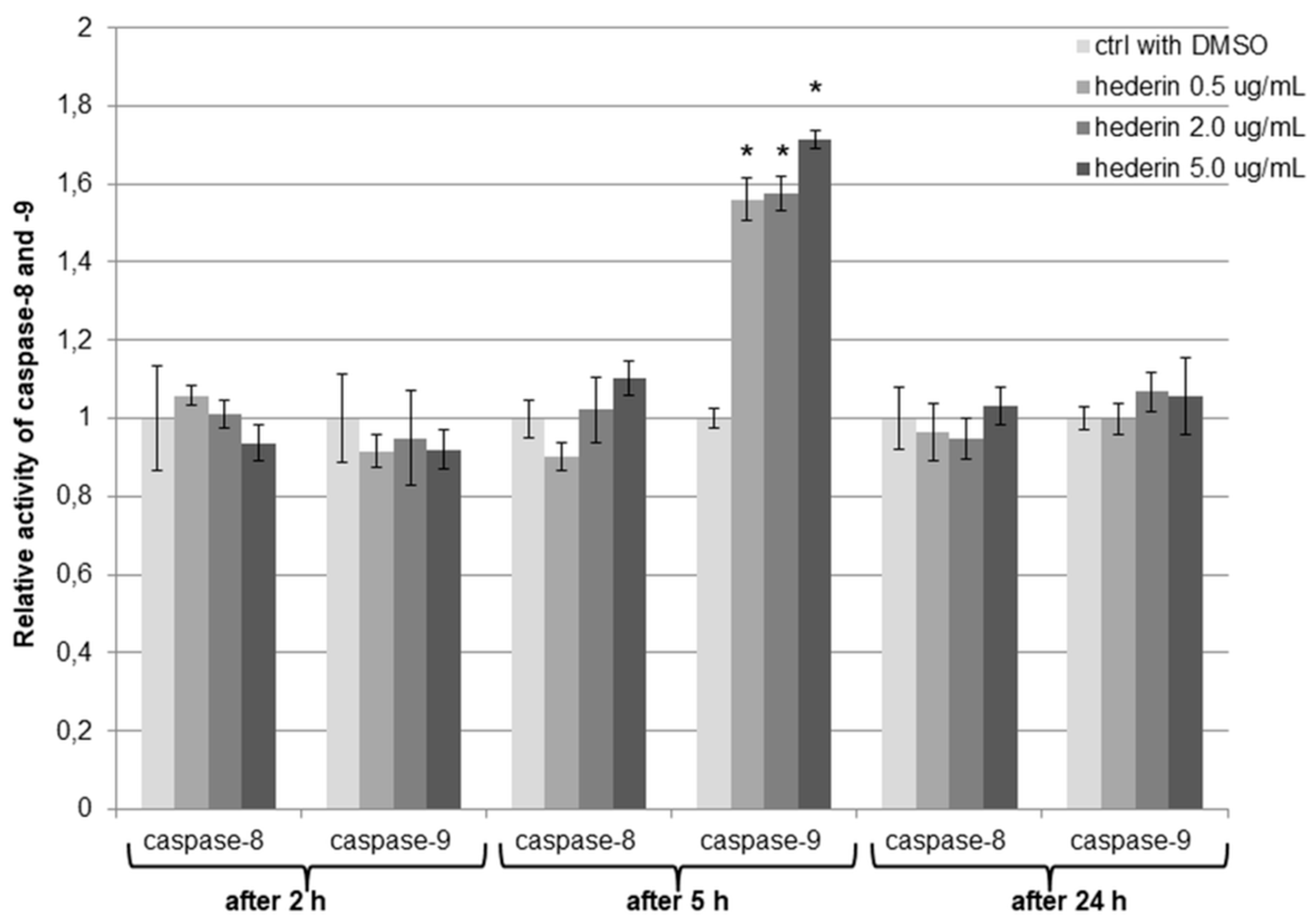
2.6. α-HN Induced Changes in the Caspases’ Activity
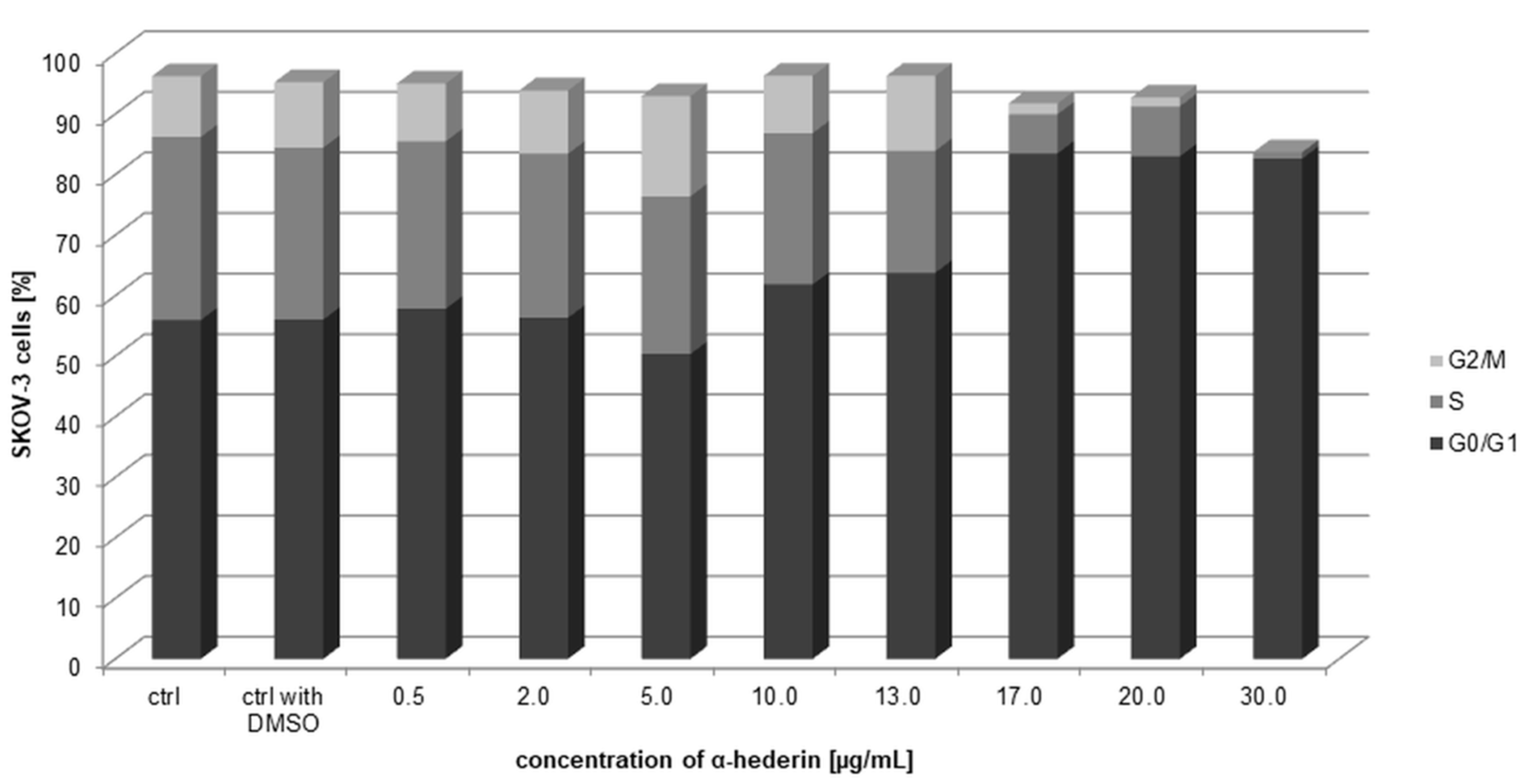
2.7. Arresting the Cell Cycle
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Line Culture
4.3. Cell Viability Assays
4.3.1. Real-Time xCELLigence Cell Proliferation Assay
4.3.2. MTT Assay
4.4. Hoechst Staining
4.5. Apoptosis Assay
4.6. Assessment of Mitochondrial Membrane Potential
4.7. Bcl-2 Activation Dual Detection
4.8. Assessment of Caspase-3/7/8/9 Activity
4.9. Arresting the Cell Cycle
Author Contributions
Funding
Conflicts of Interest
References
- Muhammad, T.I. A pharmacological emphasized revision on alpha-hederin. J. Complement. Med. Alt. Healthc. 2017, 1, 555564. [Google Scholar]
- Gulcin, I.; Mshvildadze, V.; Gepdiremen, A.; Elias, R. Antioxidant activity of saponins isolated from ivy: Alpha-hederin, hederasaponin-C, hederacolchiside-E and hederacolchicide F. Planta Med. 2004, 70, 561–563. [Google Scholar] [CrossRef]
- Keyhanmanesh, R.; Saadat, S.; Mohammadi, M.; Shahbazfar, A.; Fallahi, M. The Protective Effect of α-hederin, the Active Constituent of Nigella sativa, on Lung Inflammation and Blood Cytokines in Ovalbumin Sensitized Guinea Pigs. Phytother. Res. 2015, 29, 1761–1767. [Google Scholar] [CrossRef]
- Ridoux, O.; Di Giorgio, C.; Delmas, F.; Elias, R.; Mshvildadze, V.; Dekanosidze, G.; Kemertelidze, E.; Balansard, G.; Timon-David, P. In vitro antileishmanial activity of three saponins isolated from ivy, alpha-hederin, beta-hederin and hederacolchiside A(1), in association with pentamidine and amphotericin B. Phytother. Res. 2001, 15, 298–301. [Google Scholar] [CrossRef]
- Delmas, F.; Di Giorgio, C.; Elias, R.; Gasquet, M.; Azas, N.; Mshvildadze, V.; Dekanosidze, G.; Kemertelidze, E.; Timon-David, P. Antileishmanial activity of three saponins isolated from ivy, alpha-hederin, beta-hederin and hederacolchiside A(1), as compared to their action on mammalian cells cultured in vitro. Planta Med. 2000, 66, 343–347. [Google Scholar] [CrossRef]
- Shi, J.Z.; Liu, G.T. Effect of alpha-hederin and sapindoside B on hepatic microsomal cytochrome P-450 in mice. Acta Pharmacol. Sin. 1996, 17, 264–266. [Google Scholar]
- Jeong, H.G. Suppression of constitutive and inducible cytochrome P450 gene expression by alpha-Hederin in mice. Biochem. Mol. Biol. Int. 1998, 46, 1019–1026. [Google Scholar]
- Liu, J.; Choudhuri, S.; Liu, Y.P.; Kreppel, H.; Andrews, G.K.; Klaassen, C.D. Induction of metallothionein by alpha-hederin. Toxicol. Appl. Pharmacol. 1993, 121, 144–151. [Google Scholar] [CrossRef]
- Kim, J.Y.; Choi, C.Y.; Jeong, H.G. Involvement of cytokines in the hepatic metallothionein expression by alpha-hederin. Planta Med. 2005, 71, 743–747. [Google Scholar] [CrossRef]
- Gepdiremen, A.; Mshvildadze, V.; Suleyman, H.; Elias, R. Acute anti-inflammatory activity of four saponins isolated from ivy: alpha-hederin, hederasaponin-C, hederacolchiside-E and hederacolchiside-F in carrageenan-induced rat paw edema. Phytomedicine 2005, 12, 440–444. [Google Scholar] [CrossRef]
- Danloy, S.; Quetinleclercq, J.; Coucke, P.; Depauwgillet, M.C.; Elias, R.; Balansard, G.; Angenot, L.; Bassleer, R. Effects of alpha-hederin, a saponin extracted from Hedera helix, on cells cultured in vitro. Planta Med. 1994, 60, 45–49. [Google Scholar] [CrossRef]
- Rooney, S.; Ryan, M.F. Effects of alpha-hederin and thymoquinone, constituents of Nigella sativa, on human cancer cell lines. Anticancer Res. 2005, 25, 2199–2204. [Google Scholar]
- Lorent, J.; Lins, L.; Domenech, O.; Quetin-Leclercq, J.; Brasseur, R.; Mingeot-Leclercq, M.P. Domain formation and permeabilization induced by the saponin alpha-hederin and its aglycone hederagenin in a cholesterol-containing bilayer. Langmuir 2014, 30, 4556–4569. [Google Scholar] [CrossRef]
- Lorent, J.; Le Duff, C.S.; Quetin-Leclercq, J.; Mingeot-Leclercq, M.P. Induction of highly curved structures in relation to membrane permeabilization and budding by the triterpenoid saponins, alpha-and delta-hederin. J. Biol. Chem. 2013, 288, 14000–14017. [Google Scholar] [CrossRef]
- Jeong, H.G.; Choi, C.Y. Expression of inducible nitric oxide synthase by alpha-hederin in macrophages. Planta Med. 2002, 68, 392–396. [Google Scholar] [CrossRef]
- Swamy, S.; Huat, B. Intracellular glutathione depletion and reactive oxygen species generation are important in alpha-hederin-induced apoptosis of P388 cells. Mol. Cell. Biochem. 2003, 245, 127–139. [Google Scholar]
- Cheng, L.; Xia, T.S.; Wang, Y.F.; Zhou, W.B.; Liang, X.Q.; Xue, J.Q.; Shi, L.; Wang, Y.; Ding, Q.; Wang, M.H. The anticancer-effect and mechanism of alpha-hederin on breast cancer cells. Int. J. Oncol. 2014, 45, 757–763. [Google Scholar] [CrossRef]
- Kumara, S.S.M.; Huat, B.T.K. Extraction, isolation and characterisation of antitumor principle, alpha-hederin, from the seeds of Nigella sativa. Planta Med. 2001, 67, 29–32. [Google Scholar] [CrossRef]
- Sieben, A.; Prenner, L.; Sorkalla, T.; Wolf, A.; Jakobs, D.; Runkel, F.; Haberlein, H. Alpha-hederin, but not hederacoside C and hederagenin from Hedera helix, affects the binding behavior, dynamics, and regulation of beta 2-adrenergic receptors. Biochemistry 2009, 48, 3477–3482. [Google Scholar] [CrossRef]
- Bang, S.C.; Seo, H.H.; Shin, H.R.; Lee, K.C.; Hoang, L.T.A.; Jung, S.H. A convenient preparation of a disaccharide motif and its role in the cytotoxicity of the triterpenoid saponin, alpha-hederin. Arch. Pharm. Res. 2008, 31, 555–561. [Google Scholar] [CrossRef]
- Bun, S.S.; Elias, R.; Baghdikian, B.; Ciccolini, J.; Ollivier, E.; Balansard, G. alpha-Hederin potentiates 5-fu antitumor activity in human colon adenocarcinoma cells. Phytother. Res. 2008, 22, 1299–1302. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Zhang, C.G.; Liu, Y.; Yuan, Z.Q.; Chen, W.L.; Yang, S.D.; Li, J.Z.; Zhu, W.J.; Zhou, X.F.; You, B.G.; et al. CD147 monoclonal antibody mediated by chitosan nanoparticles loaded with alpha-hederin enhances antineoplastic activity and cellular uptake in liver cancer cells. Sci. Rep. 2015, 5, 13. [Google Scholar] [CrossRef]
- Ebrahimi, H.; Fallahi, M.; Khamaneh, A.M.; Ebrahimi, S.M.A.; Saadat, S.; Keyhanmanesh, R. Effect of α-Hederin on IL-2 and IL-17 mRNA and miRNA-133a Levels in Lungs of Ovalbumin-Sensitized Male Rats. Drug Dev. Res. 2016, 77, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, H.; Yonehara, S.; Bonavida, B. Overcoming tumor necrosis factor and drug resistance of human tumor cell lines by combination treatment with anti-Fas antibody and drugs or toxins. Cancer Res. 1993, 53, 2591–2596. [Google Scholar] [PubMed]
- Available online: http://www.lgcstandards-atcc.org/products/all/htb-77.aspx (accessed on 10 September 2018).
- Hernandez, L.; Kim, M.K.; Lyle, L.T.; Bunch, K.P.; House, C.D.; Ning, F.; Noonan, A.M.; Annunziata, C.M. Characterization of ovarian cancer cell lines as in vivo models for preclinical studies. Gynecol. Oncol. 2016, 142, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Kustermann, S.; Boess, F.; Buness, A.; Schmitz, M.; Watzele, M.; Weiser, T.; Singer, T.; Suter, L.; Roth, A. A label-free, impedance-based real time assay to identify drug-induced toxicities and differentiate cytostatic from cytotoxic effects. Toxicol. In Vitro 2013, 27, 1589–1595. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.H.; Huang, B.; Zhang, W.P.; Gabos, S.; Huang, D.Y.; Devendran, V. Cytotoxicity assessment based on the AUC(50) using multi-concentration time-dependent cellular response curves. Anal. Chim. Acta 2013, 764, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Limame, R.; Wouters, A.; Pauwels, B.; Fransen, E.; Peeters, M.; Lardon, F.; De Wever, O.; Pauwels, P. Comparative analysis of dynamic cell viability, migration and invasion assessments by novel real-time technology and classic endpoint assays. PLoS ONE 2012, 7, e46536. [Google Scholar] [CrossRef]
- Dajani, E.Z.; Shahwan, T.G.; Dajani, N.E. Overview of the preclinical pharmacological properties of Nigella sativa (black seeds): A complementary drug with historical and clinical significance. J. Physiol. Pharmacol. 2016, 67, 801–817. [Google Scholar]
- Khan, M.A.; Younus, H. Thymoquinone shows the diverse therapeutic actions by modulating multiple cell signaling pathways: single drug for multiple targets. Curr. Pharm. Biotechnol. 2018, 19, 934–945. [Google Scholar] [CrossRef]
- Salomi, M.J.; Nair, S.C.; Panikkar, K.R. Inhibitory effects of Nigella-sativa and saffron (Crocus-sativus) on chemical carcinogenesis in mice. Nutr. Cancer 1991, 16, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Salomi, N.J.; Nair, S.C.; Jayawardhanan, K.K.; Varghese, C.D.; Panikkar, K.R. Antitumor principles from Nigella-sativa seeds. Cancer Lett. 1992, 63, 41–46. [Google Scholar] [CrossRef]
- Soltani, A.; Pourgheysari, B.; Shirzad, H.; Sourani, Z. Antiproliferative and apoptosis-inducing activities of thymoquinone in lymphoblastic leukemia cell line. Indian J. Hematol. Blood Transfus. 2017, 33, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Shoieb, A.M.; Elgayyar, M.; Dudrick, P.S.; Bell, J.L.; Tithof, P.K. In vitro inhibition of growth and induction of apoptosis in cancer cell lines by thymoquinone. Int. J. Oncol. 2003, 22, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Odeh, L.H.; Talib, W.H.; Basheti, I.A. Synergistic effect of thymoquinone and melatonin against breast cancer implanted in mice. J. Cancer Res. Ther. 2018, 14, S324–S330. [Google Scholar] [CrossRef]
- Ng, W.K.; Yazan, L.S.; Ismail, M. Thymoquinone from Nigella sativa was more potent than cisplatin in eliminating of SiHa cells via apoptosis with down-regulation of Bcl-2 protein. Toxicol. In Vitro 2011, 25, 1392–1398. [Google Scholar] [CrossRef]
- Dastjerdi, M.N.; Mehdiabady, E.M.; Iranpour, F.G.; Bahramian, H. Effect of thymoquinone on p53 gene expression and consequence apoptosis in breast cancer cell line. Int. J. Prev. Med. 2016, 7, 6. [Google Scholar] [CrossRef]
- Bang, S.C.; Kim, Y.; Lee, J.H.; Ahn, B.Z. Triterpenoid saponins from the roots of Pulsatilla koreana. J. Nat. Prod. 2005, 68, 268–272. [Google Scholar] [CrossRef]
- Kim, Y.; Bang, S.C.; Lee, J.H.; Ahn, B.Z. Pulsatilla saponin D: The antitumor principle from Pulsatilla koreana. Arch. Pharm. Res. 2004, 27, 915–918. [Google Scholar] [CrossRef]
- Chwalek, M.; Lalun, N.; Bobichon, H.; Ple, K.; Voutquenne-Nazabadioko, L. Structure-activity relationships of some hederagenin diglycosides: Haemolysis, cytotoxicity and apoptosis induction. Biochim. Biophys. Acta Gen. Subj. 2006, 9, 1418–1427. [Google Scholar] [CrossRef]
- Hengartner, M.O. The biochemistry of apoptosis. Nature 2000, 407, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Koopman, G.; Reutelingsperger, C.P.M.; Kuijten, G.A.M.; Keehnen, R.M.J.; Pals, S.T.; Vanoers, M.H.J. Annexin-V for flow cytometric detection of phosphatidylserine expression on B-cells undergoing apoptosis. Blood 1994, 84, 1415–1420. [Google Scholar] [PubMed]
- Vermes, I.; Haanen, C.; Steffensnakken, H.; Reutelingsperger, C. A Novel assay for apoptosis—flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein-labeled annexin-V. J. Immunol. Methods 1995, 184, 39–51. [Google Scholar] [CrossRef]
- Van Engeland, M.; Ramaekers, F.C.S.; Schutte, B.; Reutelingsperger, C.P.M. A novel assay to measure loss of plasma membrane asymmetry during apoptosis of adherent cells in culture. Cytometry 1996, 24, 131–139. [Google Scholar] [CrossRef]
- Fulda, S.; Debatin, K.M. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 2006, 25, 4798–4811. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, S.H.; Clark, A.C. Targeting cell death in tumors by activating caspases. Curr. Cancer Drug Targets 2008, 8, 98–109. [Google Scholar] [PubMed]
- Danial, N.N.; Korsmeyer, S.J. Cell death: Critical control points. Cell 2004, 116, 205–219. [Google Scholar] [CrossRef]
- Kroemer, G.; Reed, J. Mitochondrial control of cell death. Nat. Med. 2000, 6, 513–519. [Google Scholar] [CrossRef]
- Wang, X.D. The expanding role of mitochondria in apoptosis. Genes Dev. 2001, 15, 2922–2933. [Google Scholar]
- Ly, J.D.; Grubb, D.R.; Lawen, A. The mitochondrial membrane potential (Delta psi m) in apoptosis; an update. Apoptosis 2003, 8, 115–128. [Google Scholar] [CrossRef]
- Krysko, D.V.; Roels, F.; Leybaert, L.; D’Herde, K. Mitochondrial transmembrane potential changes support the concept of mitochondrial heterogeneity during apoptosis. J. Histochem. Cytochem. 2001, 49, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.M.; Cory, S. Life-or-death decisions by the Bcl-2 protein family. Trends Biochem. Sci. 2001, 26, 61–66. [Google Scholar] [CrossRef]
- Verma, S.; Zhao, L.J.; Chinnadurai, G. Phosphorylation of the pro-apoptotic protein BIK—Mapping of phosphorylation sites and effect on apoptosis. J. Biol. Chem. 2001, 276, 4671–4676. [Google Scholar] [CrossRef] [PubMed]
- Lemeshko, V.V. Theoretical evaluation of a possible nature of the outer membrane potential of mitochondria. Eur. Biophys. J. 2006, 36, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Haridas, V.; Li, X.; Mizumachi, T.; Higuchi, M.; Lemeshko, V.V.; Colombini, M.; Gutterman, J.U. Avicins, a novel plant-derived metabolite lowers energy metabolism in tumor cells by targeting the outer mitochondrial membrane. Mitochondrion 2007, 7, 234–240. [Google Scholar] [CrossRef]
- Zou, H.; Henzel, W.J.; Liu, X.S.; Lutschg, A.; Wang, X.D. Apaf-1, a human protein homologous to C-elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 1997, 90, 405–413. [Google Scholar] [CrossRef]
- Bernardi, P.; Scorrano, L.; Colonna, R.; Petronilli, V.; Di Lisa, F. Mitochondria and cell death. Mechanistic aspects and methodological issues. Eur. J. Biochem. 1999, 265, 847. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Hockenbery, D.; Nunez, G.; Milliman, C.; Schreiber, R.D.; Korsmeyer, S.J. Bcl-2 is an inner mitochondrial-membrane protein that blocks programmed cell-death. Nature 1990, 348, 334–336. [Google Scholar] [CrossRef]
- Portt, L.; Norman, G.; Clapp, C.; Greenwood, M.; Greenwood, M.T. Anti-apoptosis and cell survival: A review. Biochim. Biophys. Acta Mol. Cell Res. 2011, 1813, 238–259. [Google Scholar] [CrossRef]
- Khwaja, T.A.; Wajahat, T.; Ahmad, I.; Hoessli, D.C.; Walker-Nasir, E.; Kaleem, A.; Qazi, W.M.; Shakoori, A.R.; Nasir Ud, D. In silico modulation of apoptotic Bcl-2 proteins by mistletoe lectin-1: Functional consequences of protein modifications. J. Cell. Biochem. 2008, 103, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Van Cruchten, S.; Van den Broeck, W. Morphological and biochemical aspects of apoptosis, oncosis and necrosis. Anat. Histol. Embryol. 2002, 31, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Feng, Y.; Wang, Y.; Ji, Q.; Cai, G.; Shi, L.; Wang, Y.; Huang, Y.; Zhang, J.; Li, Q. α-hederin induces autophagic cell death in colorectal cancer cells through reactive oxygen species dependent AMPK/mTOR signaling pathway activation. Int. J. Oncol. 2019, 54, 1601–1612. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wu, B.; Wang, H. Alpha-hederin induces the apoptosis of oral cancer SCC-25 cells by regulating PI3K/Akt/mTOR singaling pathway. Electron. J. Biotechnol. 2019, 38, 27–31. [Google Scholar] [CrossRef]
- Sun, D.; Shen, W.; Zhang, F.; Fan, H.; Tan, J.; Li, L.; Xu, C.; Zhang, H.; Yang, Y.; Cheng, H. α-Hederin Arrests Cell Cycle at G2/M Checkpoint and Promotes Mitochondrial Apoptosis by Blocking Nuclear Factor-κB Signaling in Colon Cancer Cells. Biomed. Res. Int. 2018, 2018, 2548378. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Cell Line | SKOV-3 | HaCaT | ||
|---|---|---|---|---|
| Method | RTCA | MTT | RTCA | MTT |
| 2.62 * ± 0.04; 1 a | 2.48 ** ± 0.32 | 2.71 * ± 0.35; 0.99 a | 2.57 ** ± 0.21 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adamska, A.; Stefanowicz-Hajduk, J.; Ochocka, J.R. Alpha-Hederin, the Active Saponin of Nigella sativa, as an Anticancer Agent Inducing Apoptosis in the SKOV-3 Cell Line. Molecules 2019, 24, 2958. https://doi.org/10.3390/molecules24162958
Adamska A, Stefanowicz-Hajduk J, Ochocka JR. Alpha-Hederin, the Active Saponin of Nigella sativa, as an Anticancer Agent Inducing Apoptosis in the SKOV-3 Cell Line. Molecules. 2019; 24(16):2958. https://doi.org/10.3390/molecules24162958
Chicago/Turabian StyleAdamska, Anna, Justyna Stefanowicz-Hajduk, and J. Renata Ochocka. 2019. "Alpha-Hederin, the Active Saponin of Nigella sativa, as an Anticancer Agent Inducing Apoptosis in the SKOV-3 Cell Line" Molecules 24, no. 16: 2958. https://doi.org/10.3390/molecules24162958
APA StyleAdamska, A., Stefanowicz-Hajduk, J., & Ochocka, J. R. (2019). Alpha-Hederin, the Active Saponin of Nigella sativa, as an Anticancer Agent Inducing Apoptosis in the SKOV-3 Cell Line. Molecules, 24(16), 2958. https://doi.org/10.3390/molecules24162958






