Effects of Portulaca Oleracea Extract on Acute Alcoholic Liver Injury of Rats
Abstract
1. Introduction
2. Results
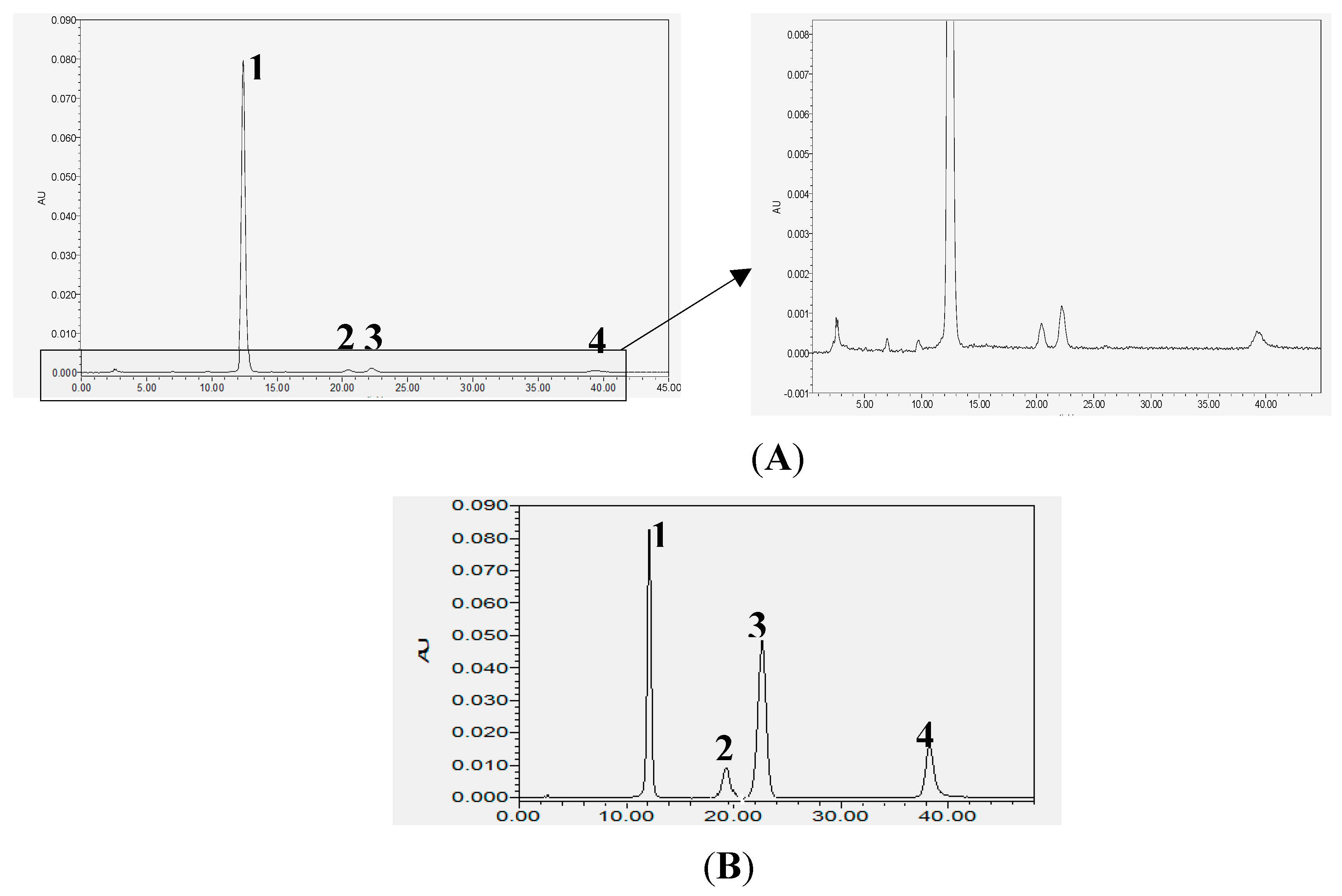
2.1. Analysis of Chemical Constituents of POE
2.2. Effects of POE on Serum Biochemical Indicator in Alcoholic Liver Injury Rats
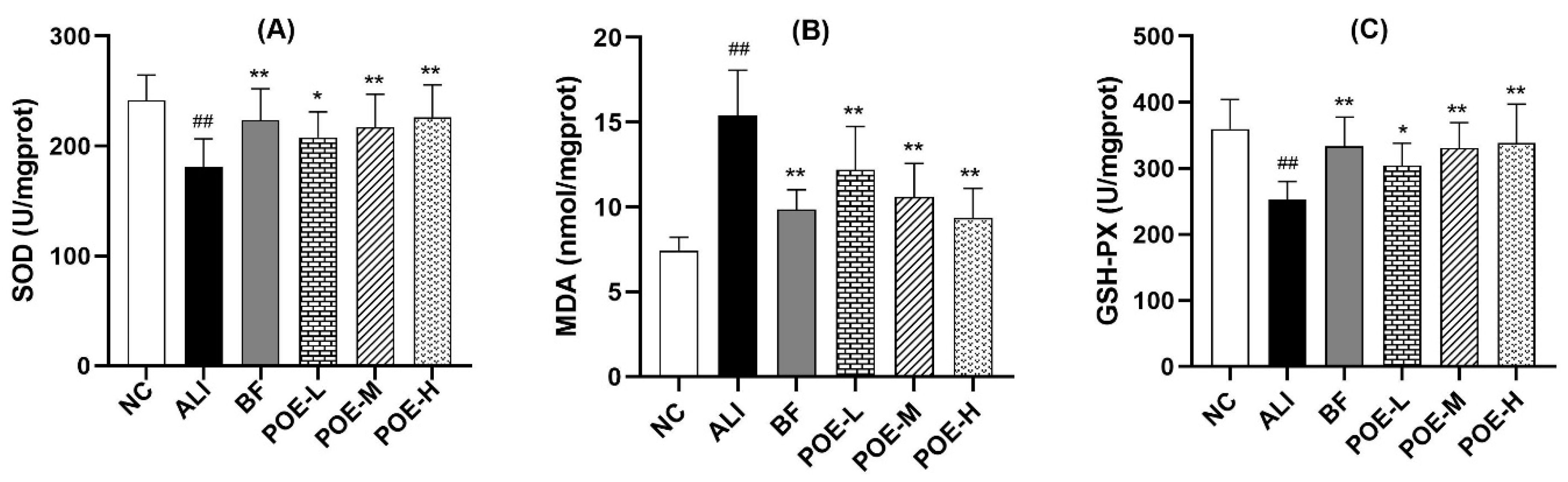
2.3. Effects of POE on Oxidative Damage Index in the Liver
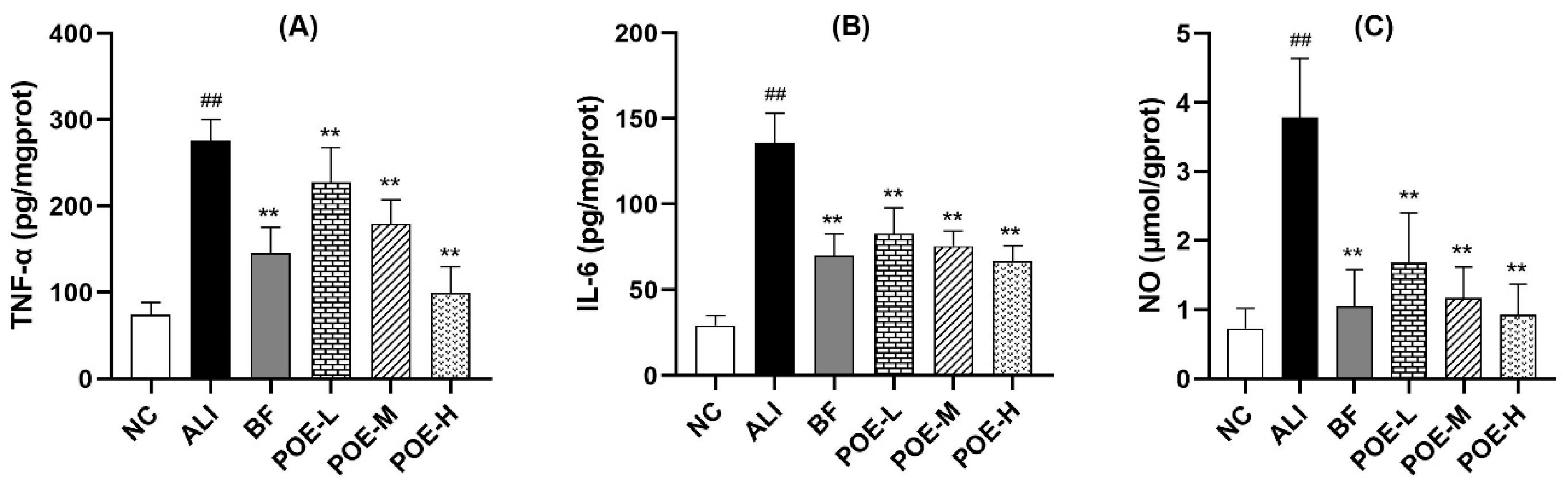
2.4. Effects of POE on Inflammatory Cytokine and Mediators in Alcoholic Liver Injury Rats
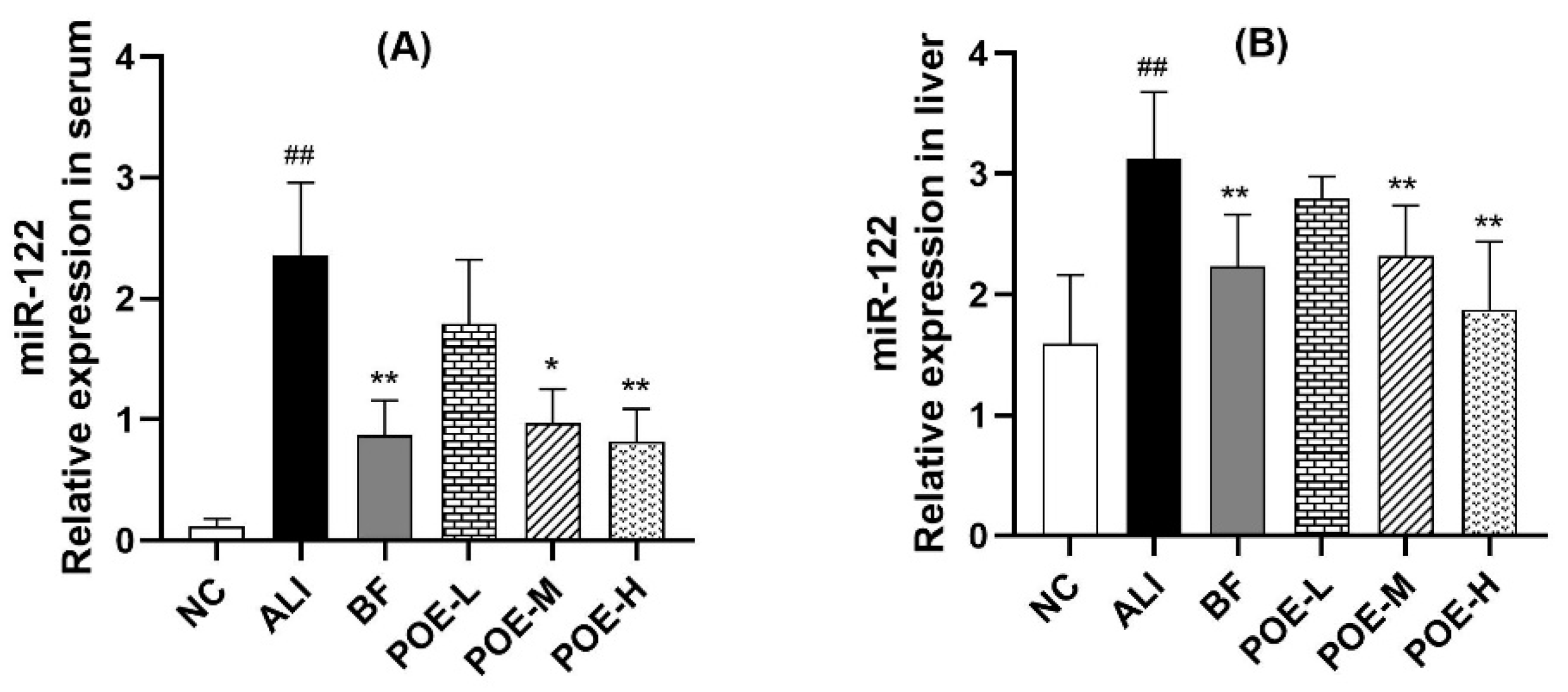
2.5. Effects of POE on Expression of miR-122
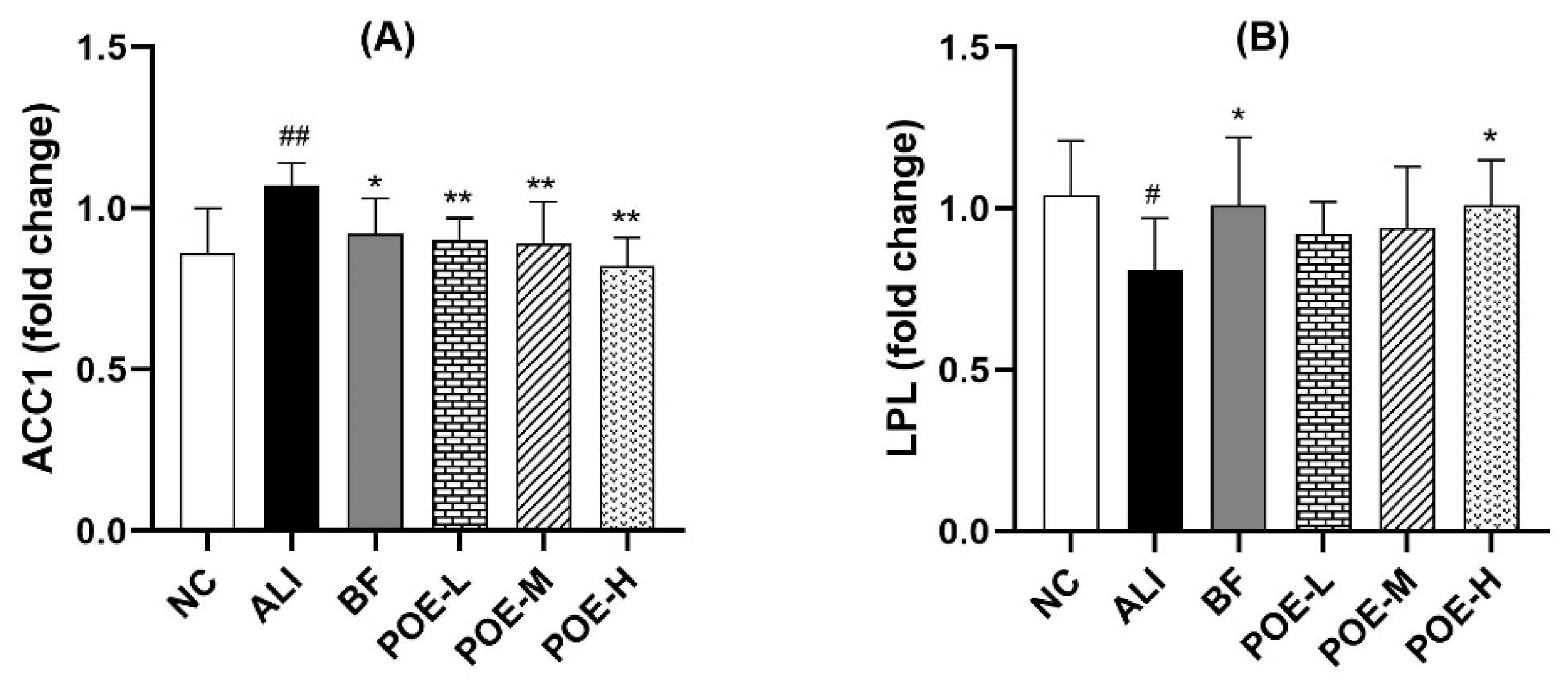
2.6. Effects of POE on the mRNA Expression of ACC1 and LPL
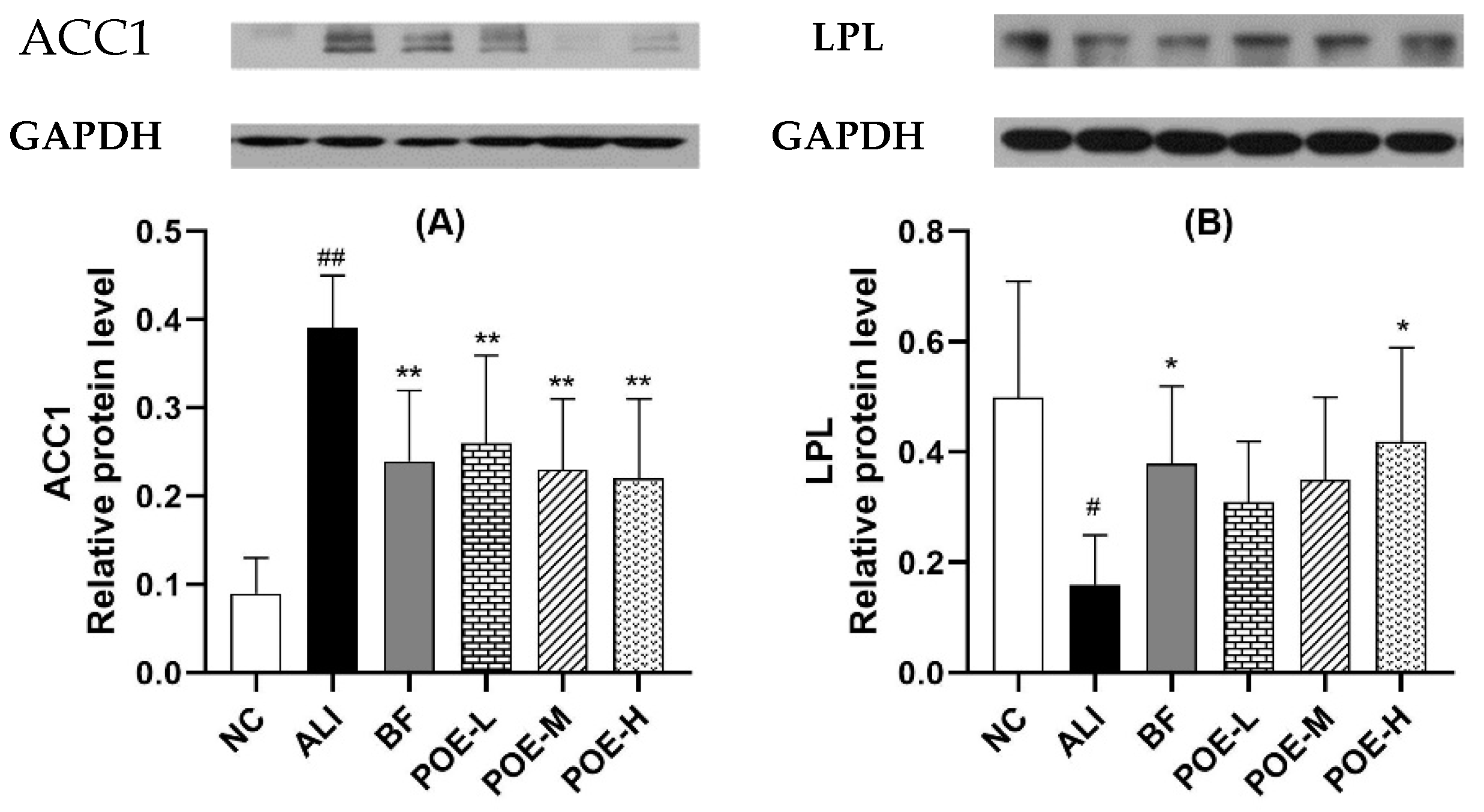
2.7. Effects of POE on the Protein Expression of ACC1 and LPL
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Chemicals and Reagents
4.3. Animals and Treatments
4.4. Blood Biochemical Analysis
4.5. Liver Oxidative Stress and Inflammation Detection
4.6. Real-Time Fluorescence Quantitative polymerase chain reaction (PCR) of MiR-122 in Serum and Liver
4.7. Quantitative Real-Time PCR (qRT-PCR) of ACC and LPL mRNA in Liver
4.8. Western Blot of ACC and LPL protein in Liver
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ellis, J.L.; Yin, C. Histological analyses of acute alcoholic liver injury in zebrafish. J. Vis. Exp. 2017, 25, e55630. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Status Report on Alcohol and Health; World Health Organization: Geneva, Switzerland, 2018; Available online: https://apps.who.int/iris/bitstream/handle/10665/274603/9789241565639-eng.pdf?ua=1: URL (accessed on 7 May 2019).
- Osna, N.A.; Donohue, T.M., Jr.; Kharbanda, K.K. Alcoholic liver disease: Pathogenesis and current management. Alcohol Res. 2017, 38, 147–161. [Google Scholar] [PubMed]
- Louvet, A.; Mathurin, P. Alcoholic liver disease: Mechanisms of injury and targeted treatment. Nat. Rev. Gastroenterol Hepatol. 2015, 12, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Hou, R.; Yan, J.; Xu, K.; Wu, X.; Lin, W.; Zheng, M.; Fu, J. Purification and characterization of Inonotus hispidus exopolysaccharide and its protective effect on acute alcoholic liver injury in mice. Int. J. Biol. Macromol. 2019, 129, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, Y.; Bai, R.; Wang, M.; Du, S. Baicalin attenuates alcoholic liver injury through modulation of hepatic oxidative stress, inflammation and sonic hedgehog pathway in rats. Cell. Physiol. Biochem. 2016, 39, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; Park, Y.N.; Kwon, O.Y.; Lee, S.H. Orally administration of Neolentinus lepideus extracts attenuated ethanol induced accumulation of hepatic lipid in mice. Food Sci. Biotechnol. 2018, 28, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Bai, T.; Yangm, Y.; Yao, Y.L.; Sun, P.; Lian, L.H.; Wu, Y.L.; Nan, J.X. Betulin alleviated ethanol-induced alcoholic liver injury via SIRT1/AMPK signaling pathway. Pharmacol. Res. 2016, 105, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yu, Y.; Sang, R.; Li, J.; Ge, B.; Zhang, X. Protective effects of taraxasterol against ethanol-induced liver injury by regulating CYP2E1/Nrf2/HO-1 and NF-κB signaling pathways in Mice. Oxid. Med. Cell Longev. 2018, 2018, 8284107. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Jia, Z.; Misra, H.; Li, Y.R. Oxidative stress and redox signaling mechanisms of alcoholic liver disease: Updatedexperimental and clinical evidence. J. Dig. Dis. 2012, 13, 133–142. [Google Scholar] [CrossRef]
- Maiers, J.L.; Malhi, H. Endoplasmic reticulum stress in metabolic liver diseases and hepatic fibrosis. Semin. Liver Dis. 2019, 39, 235–248. [Google Scholar] [CrossRef]
- Jiang, Z.; Wang, J.; Xue, H.; Wang, M.; Jiang, H.; Liang, Y.; Dias, A.C.; Gregory, M.; Chen, C.; Zhang, X. Protective effect of wild Corni fructus methanolic extract against acute alcoholic liverinjury in mice. Redox Rep. 2017, 22, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Sun, J.; Jiang, Z.; Xie, W.; Zhang, X. Hepatoprotective effect of kaempferol against alcoholic liver injury in mice. Am. J. Chin. Med. 2015, 43, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.G.; Dou, X.B.; Zhou, Z.X.; Song, Z.Y. Adipose tissue-liver axis in alcoholic liver disease. World J. Gastrointest. Pathophysiol. 2016, 7, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Shi, X.; Zhong, W.; Zhao, Y.; Tang, Y.; Sun, W.; Yin, X.; Bogdanov, B.; Kim, S.; McClain, C.; et al. Chronic alcohol exposure disturbs lipid homeostasis at the adipose tissue-liver axis in mice: Analysis of triacylglycerols using high-resolution mass spectrometry in combination with in vivo metabolite deuterium labeling. PLoS ONE 2013, 8, e55382. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Petrasek, J.; Mundkur, S.; Catalano, D.; Levin, I.; Ward, J.; Alao, H.; Kodys, K.; Szabo, G. Circulating microRNAs in exosomes indicate hepatocyte injury and inflammation in alcoholic, drug-induced, and inflammatory liver diseases. Hepatology 2012, 56, 1946–1957. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jia, Y.; Zheng, R.; Guo, Y.; Wang, Y.; Guo, H.; Fei, M.; Sun, S. Plasma microRNA-122 as a biomarker for viral-, alcohol-, and chemical-related hepatic diseases. Clin. Chem. 2010, 56, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Esau, C.; Davis, S.; Murray, S.F.; Yu, X.X.; Pandey, S.K.; Pear, M.; Watts, L.; Booten, S.L.; Graham, M.; McKay, R.; et al. MiR-122 regulation of lipid metabolism revealed by in vivo antisense targeting. Cell Metab. 2006, 3, 87–98. [Google Scholar] [CrossRef]
- Elmén, J.; Lindow, M.; Schütz, S.; Lawrence, M.; Petri, A.; Obad, S.; Lindholm, M.; Hedtjärn, M.; Hansen, H.F.; Berger, U.; et al. LNA-mediated microRNA silencing in non-human primates. Nature 2008, 452, 896–899. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Erbay, E. Nutrient sensing and inflammation in metabolic diseases. Nat. Rev. Immunol. 2008, 8, 923–934. [Google Scholar] [CrossRef]
- Zhou, Y.X.; Xin, H.L.; Rahman, K.; Wangm, S.J.; Pengm, C.; Zhang, H. Portulaca oleracea L.: A review of phytochemistry and pharmacological effects. Biomed Res. Int. 2015, 2015, 925631. [Google Scholar] [CrossRef]
- Zhu, H.B.; Wang, Y.Z.; Liu, Y.X.; Xia, Y.L.; Tang, T. Analysis of flavonoids in Portulaca oleracea L. by UV-Vis spectrophotometry with comparative study on different extraction technologies. Food Anal. Methods 2010, 3, 90–97. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, W.; Ying, X.; Stien, D. New flavonoids from Portulaca oleracea L. and their activities. Fitoterapia 2018, 127, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Ahn, E.Y.; Lee, Y.J.; Park, J.; Chun, P.; Park, Y. Antioxidant potential of Artemisia capillaris, Portulaca oleracea, and Prunella vulgaris extracts for biofabrication of gold nanoparticles and cytotoxicity assessment. Nanoscale Res. Lett. 2018, 13, 348. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Islam, M.W.; Kamil, M.; Radhakrishnan, R.; Zakaria, M.N.; Habibullah, M.; Attas, A. The analgesic and anti-inflammatory effects of Portulaca oleracea L. subsp. Sativa (Haw.) Celak. J. Ethnopharmacol. 2000, 73, 445–451. [Google Scholar] [CrossRef]
- Miao, L.; Tao, H.; Peng, Y.; Wang, S.; Zhong, Z.; El-Seedi, H.; Dragan, S.; Zengin, G.; Cheang, W.S.; Wang, Y.; et al. The anti-inflammatory potential of Portulaca oleracea L. (purslane) extract by partial suppression on NF-κB and MAPK activation. Food Chem. 2019, 290, 239–245. [Google Scholar] [CrossRef] [PubMed]
- E. Abdel Moneim, A. The neuroprotective effects of purslane (Portulaca oleracea) on rotenone-induced biochemical changes and apoptosis in brain of rat. CNS Neurol. Disord. Drug Targets 2013, 12, 830–841. [Google Scholar] [CrossRef]
- Liu, X.F.; Zheng, C.G.; Shi, H.G.; Tang, G.S.; Wang, W.Y.; Zhou, J.; Dong, L.W. Ethanol extract from portulaca oleracea L. attenuated acetaminophen-induced mice liver injury. Am. J. Transl. Res. 2015, 7, 309–318. [Google Scholar] [PubMed]
- Guoyin, Z.; Hao, P.; Min, L.; Wei, G.; Zhe, C.; Changquan, L. Antihepatocarcinoma effect of Portulaca oleracea L. in mice by PI3K/Akt/mTOR and Nrf2/HO-1/NF-κB pathway. Evid. Based Complement. Alternat. Med. 2017, 2017, 8231358. [Google Scholar] [CrossRef]
- Kaveh, M.; Eidi, A.; Nemati, A.; Boskabady, M.H. Modulation of lung inflammation and immune markers in asthmatic rats treated by Portulaca oleracea. Avicenna J. Phytomed. 2017, 7, 409–416. [Google Scholar]
- Iranshahy, M.; Javadi, B.; Iranshahi, M.; Jahanbakhsh, S.P.; Mahyari, S.; Hassani, F.V.; Karimi, G. A review of traditional uses, phytochemistry and pharmacology of Portulaca oleracea L. J. Ethnopharmacol. 2017, 205, 158–172. [Google Scholar] [CrossRef]
- Zheng, G.; Mo, F.; Ling, C.; Peng, H.; Gu, W.; Li, M.; Chen, Z. Portulaca oleracea L. alleviates liver injury in streptozotocin-induced diabetic mice. Drug Des. Devel. Ther. 2017, 12, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Zang, X.; Ma, J.; Xu, G. Anti-diabetic effect of Portulaca oleracea L. Polysaccharideandits mechanism in diabetic rats. Int. J. Mol. Sci. 2016, 17, 1201. [Google Scholar] [CrossRef] [PubMed]
- Talifu, A.; Saimaiti, R.; Maitinuer, Y.; Liu, G.; Abudureyimu, M.; Xin, X. Multiomics analysis profile acute liver injury module clusters to compare the therapeutic efficacy of bifendate and muaddil sapra. Sci. Rep. 2019, 9, 4335. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wang, Y.; Jiang, X.; Sun, Y.; Zhao, Z.; Li, S. Protective effect of flavonoids from Ziziphus jujuba cv. Jinsixiaozao against acetaminophen-induced liver injury by inhibiting oxidative stress and inflammation in mice. Molecules 2017, 22, 1781. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Y.; Liu, R.; Li, X.; Cui, Y.; Qu, L. Geniposide protects against acute alcohol-induced liver injury in mice via up-regulating the expression of the main antioxidant enzymes. Can. J. Physiol. Pharmacol. 2015, 93, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.C.; Shen, S.R.; Lai, Y.J.; Wu, S.C. Rutin and quercetin, bioactive compounds from tartary buckwheat, prevent liver inflammatory injury. Food Funct. 2013, 4, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Domitrović, R.; Jakovac, H.; Marchesi, V.V.; Vladimir-Knežević, S.; Cvijanović, O.; Tadić, Z.; Romić, Z.; Rahelić, D. Differential hepatoprotective mechanisms of rutin and quercetin in CCl(4)-intoxicated BALB/cN mice. Acta Pharmacol. Sin. 2012, 33, 1260–1270. [Google Scholar]
- Liu, Q.; Pan, R.; Ding, L.; Zhang, F.; Hu, L.; Ding, B.; Zhu, L.; Xia, Y.; Dou, X. Rutin exhibits hepatoprotective effects in a mouse model of non-alcoholic fatty liver disease by reducing hepatic lipid levels and mitigating lipid-induced oxidative injuries. Int. Immunopharmacol. 2017, 49, 132–141. [Google Scholar] [CrossRef]
- Lamas-Paz, A.; Hao, F.; Nelson, L.J.; Vázquez, M.T.; Canals, S.; Del Moral, M.G.; Martínez-Naves, E.; Nevzorova, Y.A.; Cubero, F.J. Alcoholic liver disease: Utility of animal models. World J. Gastroenterol. 2018, 24, 5063–5075. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, J.; Chen, Y.; Xu, C.; Chen, X.; Shao, T.; Zhang, K.; Pan, H. Linderae radix ethanol extract attenuates alcoholic liver injury via attenuating inflammation and regulating gut microbiota in rats. Braz. J. Med. Biol. Res. 2019, 52, e7628. [Google Scholar] [CrossRef]
- Khan, A.; Pan, J.H.; Cho, S.; Lee, S.; Kim, Y.J.; Park, Y.H. Investigation of the hepatoprotective effect of Prunus mume Sieb. et Zucc extract in a mouse model of alcoholic liver injury through high-resolution metabolomics. J. Med. Food 2017, 20, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Church, R.J.; Watkins, P.B. Serum biomarkers of drug-induced liver injury: Current status and futuredirections. J. Dig. Dis. 2019, 20, 2–10. [Google Scholar] [CrossRef]
- Dogan, A.; Anuk, O.O. Investigation of the phytochemical composition and antioxidant properties of chinar (Platanus orientalis L.) leaf infusion against ethanol-induced oxidative stress in rats. Mol. Biol. Rep. 2019, 46, 3049–3061. [Google Scholar] [CrossRef]
- Ambade, A.; Mandrekar, P. Oxidative stress and inflammation: Essential partners in alcoholic liver disease. Int. J. Hepatol. 2012, 2012, 853175. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; Jokelainen, K.; Rahemtulla, A.; Miao, L.; Fogt, F.; Matsumoto, H.; Tahan, S.R.; Su, G.L. Activation of nuclear factor kappa B and cytokine imbalance in experimental alcoholic liverdisease in the rat. Hepatology 1999, 30, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Li, H.D.; Chen, X.; Yang, Y.; Huang, H.M.; Zhang, L.; Zhang, X.; Zhang, L.; Huang, C.; Meng, X.M.; Li, J. Wogonin attenuates inflammation by activating PPAR-γ in alcoholic liver disease. Int. Immunopharmacol. 2017, 50, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Nakatake, R.; Hishikawa, H.; Kotsuka, M.; Ishizaki, M.; Matsui, K.; Nishizawa, M.; Yoshizawa, K.; Kaibori, M.; Okumura, T. The proton pump inhibitor lansoprazole has hepatoprotective effects in in vitro and in vivo rat models of acute liver injury. Dig. Dis. Sci. 2019. [Google Scholar] [CrossRef]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Chen, X.; Ba, Y.; Ma, L.; Cai, X.; Yin, Y.; Wang, K.; Guo, J.; Zhang, Y.; Chen, J.; Guo, X.; et al. Characterization of microRNAs in serum: Novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008, 18, 997–1006. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, S.; Marzolf, B.; Troisch, P.; Brightman, A.; Hu, Z.; Hood, L.E.; Galas, D.J. Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proc. Natl. Acad. Sci. USA 2009, 106, 4402–4407. [Google Scholar] [CrossRef] [PubMed]
- Momen-Heravi, F.; Saha, B.; Kodys, K.; Catalano, D.; Satishchandran, A.; Szabo, G. Increased number of circulating exosomes and their microRNA cargos are potential novel biomarkers in alcoholic hepatitis. J. Transl. Med. 2015, 13, 261. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.H. The Multiple Modulation of miR-122 in the Attenuation of Alcoholic Liver Disease. Gastroenterology 2018, 154, 1857. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, K.; Herrera, L.; Zhou, T.; Francis, H.; Han, Y.; Levine, P.; Lin, E.; Glaser, S.; Alpini, G.; Meng, F. The functional role of microRNAs in alcoholic liver injury. J. Cell. Mol. Med. 2014, 18, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Sidorkiewicz, M.; Grek-Kowalinska, M.; Piekarska, A. The Correlation between miR-122 and Lipoprotein Lipase Expression in Chronic Hepatitis C Patients. Can. J. Gastroenterol. Hepatol. 2018, 2018, 6348948. [Google Scholar] [CrossRef] [PubMed]
- Geldenhuys, W.J.; Lin, L.; Darvesh, A.S.; Sadana, P. Emerging strategies of targeting lipoprotein lipase for metabolic and cardiovascular diseases. Drug Discov. Today 2017, 22, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Olivecrona, G. Role of lipoprotein lipase in lipid metabolism. Curr. Opin. Lipidol. 2016, 27, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Seo, T.; Al-Haideri, M.; Treskova, E.; Worgall, T.S.; Kako, Y.; Goldberg, I.J.; Deckelbaum, R.J. Lipoprotein lipase-mediated selective uptake from low density lipoprotein requires cell surface proteoglycans and is independent of scavenger receptor class B type 1. J. Biol. Chem. 2000, 275, 30355–30362. [Google Scholar] [CrossRef] [PubMed]
- Capel, F.; Rolland-Valognes, G.; Dacquet, C.; Brun, M.; Lonchampt, M.; Ktorza, A.; Lockhart, B.; Galizzi, J.P. Analysis of sterol-regulatory element-binding protein 1c target genes in mouseliver during aging and high-fat diet. Lifestyle Genom. 2013, 6, 107–122. [Google Scholar]
- Salie, M.J.; Thelen, J.J. Regulation and structure of the heteromeric acetyl-CoA carboxylase. Biochim. Biophys. Acta. 2016, 1861, 1207–1213. [Google Scholar] [CrossRef]
- Hsu, J.Y.; Lin, H.H.; Hsu, C.C.; Chen, B.C.; Chen, J.H. Aqueous extract of Pepino (Solanum muriactum Ait) leaves ameliorate lipid accumulation and oxidative stress in alcoholic fatty liver disease. Nutrients 2018, 10, 931. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds POE are available from the authors. |
| Groups | Dose (mg/kg) | ALT (U/L) | AST (U/L) | ALP (U/L) | TG (mmol/L) |
|---|---|---|---|---|---|
| NC | - | 25.65 ± 4.80 | 88.60 ± 13.41 | 142.35 ± 37.21 | 0.56 ± 0.22 |
| ALI | - | 42.38 ± 8.01 ## | 137.20 ± 26.91 ## | 254.71 ± 53.55 ## | 1.00 ± 0.31 ## |
| BF | 3.75 | 31.26 ± 6.04 ** | 110.60 ± 18.06 ** | 177.05 ± 43.79 ** | 0.77 ± 0.28 * |
| POE-L | 25 | 34.87 ± 6.10 * | 117.10 ± 13.91 * | 209.00 ± 55.79 * | 0.75 ± 0.23 * |
| POE-M | 50 | 31.37 ± 6.92 ** | 113.80 ± 16.49 ** | 191.91 ± 56.88 ** | 0.70 ± 0.20 * |
| POE-H | 100 | 30.93 ± 6.23 ** | 109.44 ± 14.68 ** | 185.90 ± 47.86 ** | 0.59 ± 0.23 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiao, J.-Y.; Li, H.-W.; Liu, F.-G.; Li, Y.-C.; Tian, S.; Cao, L.-H.; Hu, K.; Wu, X.-X.; Miao, M.-S. Effects of Portulaca Oleracea Extract on Acute Alcoholic Liver Injury of Rats. Molecules 2019, 24, 2887. https://doi.org/10.3390/molecules24162887
Qiao J-Y, Li H-W, Liu F-G, Li Y-C, Tian S, Cao L-H, Hu K, Wu X-X, Miao M-S. Effects of Portulaca Oleracea Extract on Acute Alcoholic Liver Injury of Rats. Molecules. 2019; 24(16):2887. https://doi.org/10.3390/molecules24162887
Chicago/Turabian StyleQiao, Jing-Yi, Han-Wei Li, Fu-Gang Liu, Yu-Cheng Li, Shuo Tian, Li-Hua Cao, Kai Hu, Xiang-Xiang Wu, and Ming-San Miao. 2019. "Effects of Portulaca Oleracea Extract on Acute Alcoholic Liver Injury of Rats" Molecules 24, no. 16: 2887. https://doi.org/10.3390/molecules24162887
APA StyleQiao, J.-Y., Li, H.-W., Liu, F.-G., Li, Y.-C., Tian, S., Cao, L.-H., Hu, K., Wu, X.-X., & Miao, M.-S. (2019). Effects of Portulaca Oleracea Extract on Acute Alcoholic Liver Injury of Rats. Molecules, 24(16), 2887. https://doi.org/10.3390/molecules24162887






