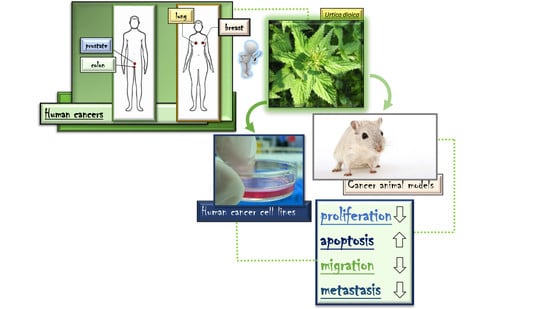
Therapeutic Perspectives of Molecules from Urtica dioica Extracts for Cancer Treatment
Abstract
1. Introduction
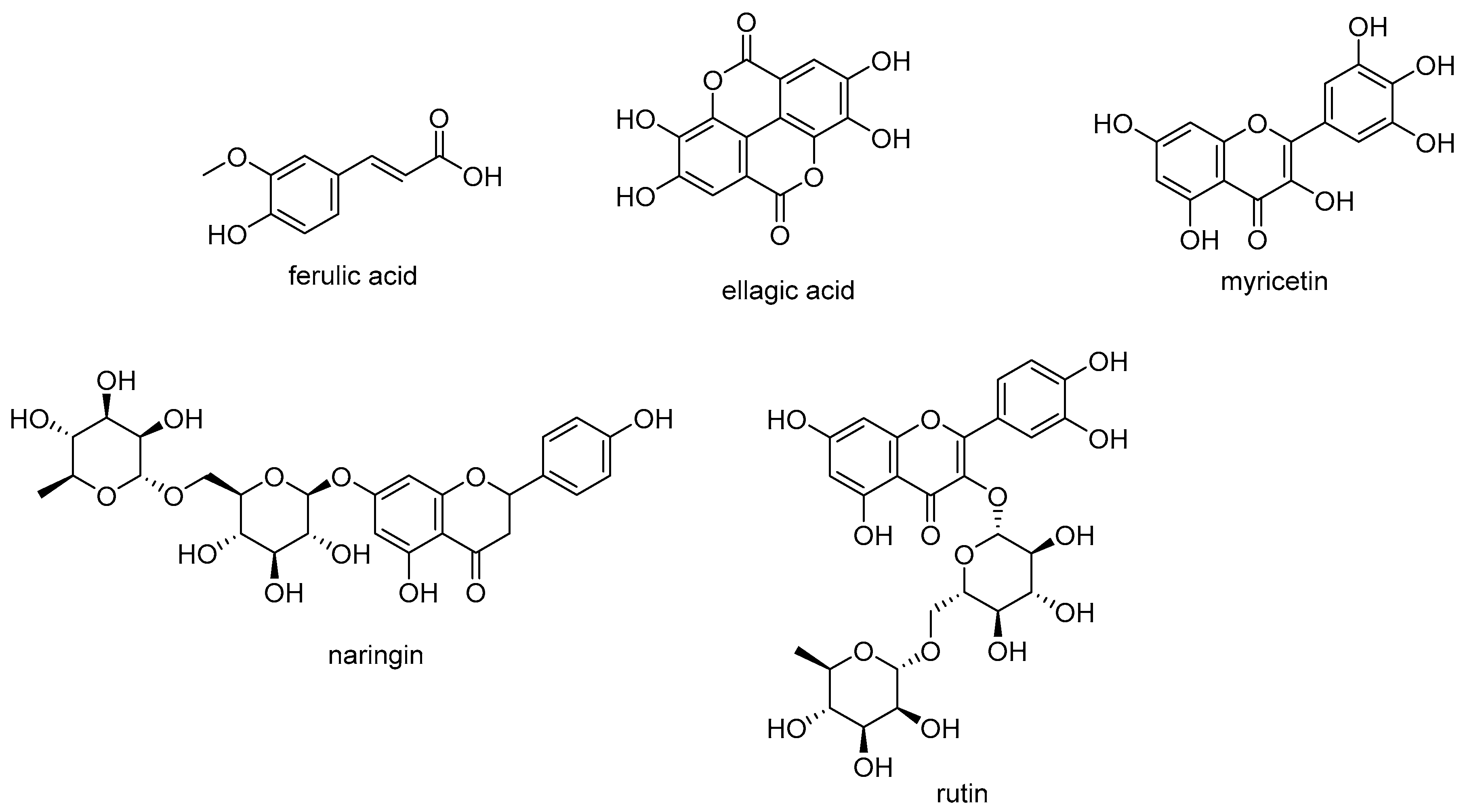
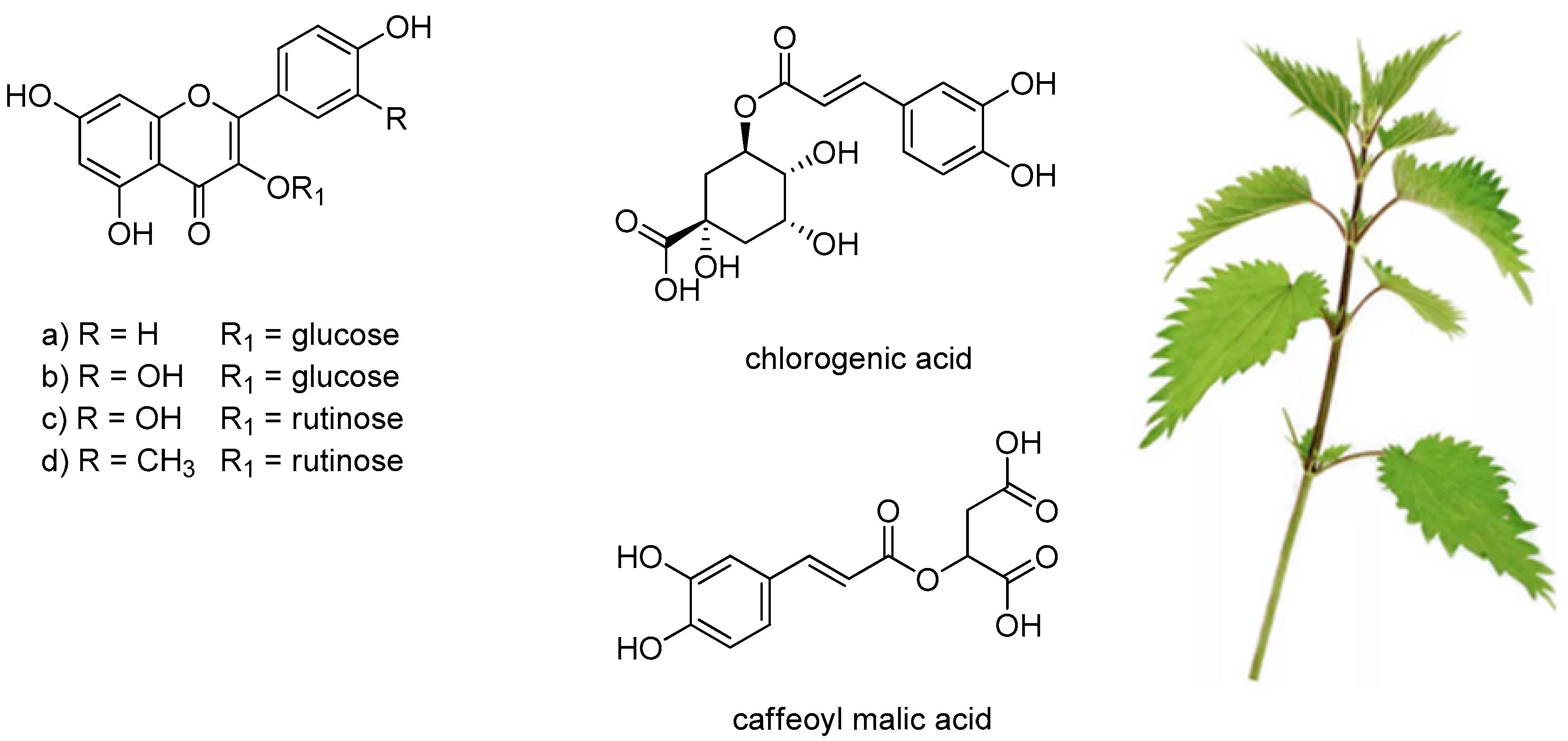
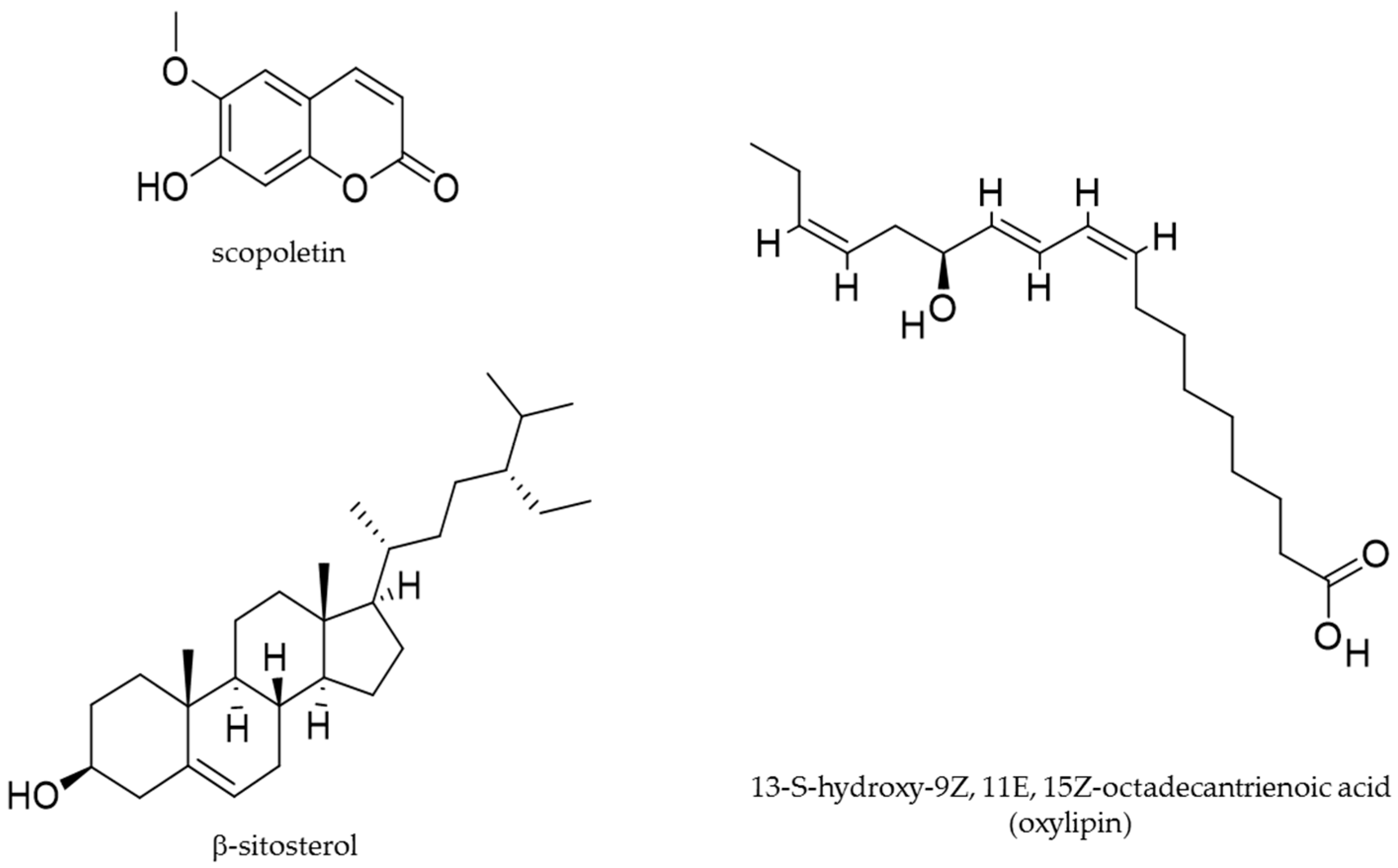
2. Phytochemical Investigations and Biological Activities of Urtica dioica
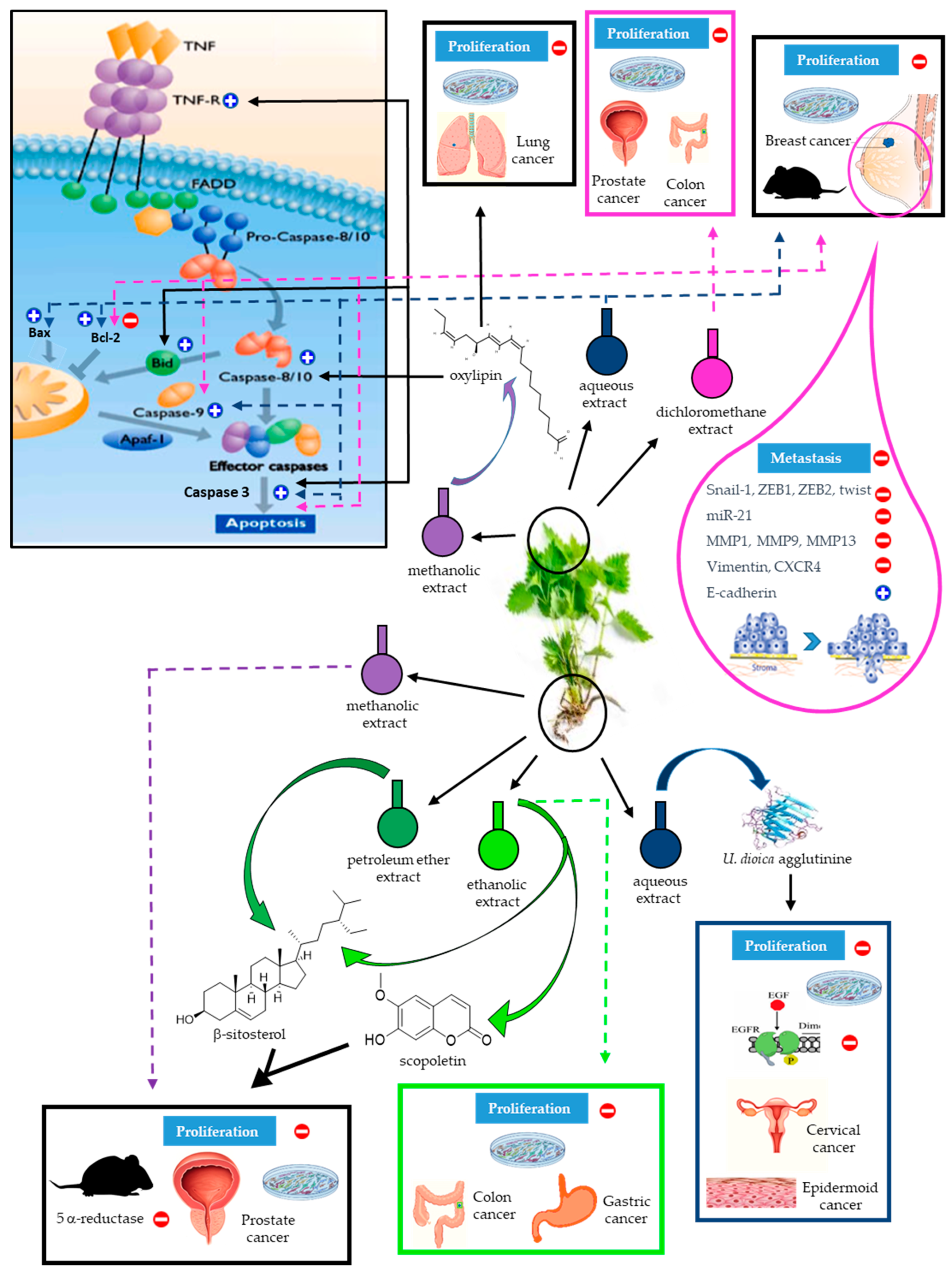
3. Anticancer Activities of Urtica dioica
Urtica dioica and Breast Cancer
4. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wildman, R.E.C.; Wildman, R.; Wallace, T.C. Handbook of Nutraceuticals and Functional Foods; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Angela, M.; Meireles, A. Extracting Bioactive Compounds for Food Products: Theory and Applications; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Kavalali, G.M. Urtica Therapeutic and Nutritional Aspects of Stinging Nettles; Taylor & Francis: London, UK, 2003; Volume 37. [Google Scholar]
- Upton, R. Stinging nettles leaf (Urtica dioica L.): Extraordinary vegetable medicine. J. Herb. Med. 2013, 3, 9–38. [Google Scholar] [CrossRef]
- Ahmed, K.K.M.; Parsuraman, S. Urtica dioica L., (Urticaceae): A Stinging Nettle. Syst. Rev. Pharm. 2014, 5, 6–8. [Google Scholar] [CrossRef]
- Oliver, F.; Amon, E.U.; Breathnach, A.; Francis, D.M.; Sarathchandra, P.; Black, A.K.; Greaves, M.W. Contact urticaria due to the common stinging nettle (Urtica dioica)—Histological, ultrastructural and pharmacological studies. Clin. Exp. Dermatol. 1991, 16, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Di Virgilio, N.; Papazoglou, E.; Jankauskiene, Z.; Lonardo, S.; Praczyk, M.; Wielgusz, K. The potential of stinging nettle (Urtica dioica L.) as a crop with multiple uses. Ind. Crops Prod. 2015, 68, 42–49. [Google Scholar] [CrossRef]
- Law, B.A.; Tamime, A.I. Technology of Cheese Making, 2nd ed.; Wiley-Blackwell: London, UK, 2010; p. 512. [Google Scholar]
- Rutto, L.K.; Xu, Y.; Ramirez, E.; Brandt, M. Mineral Properties and Dietary Value of Raw and Processed Stinging Nettle (Urtica dioica L.). Int. J. Food Sci. 2013, 2013, 857120. [Google Scholar] [CrossRef] [PubMed]
- Rafajlovska, V.; Rizova, V.; Djarmati, Z.; Tesevic, V.; Cvetkov, L. Contents of fatty acids in stinging nettle extracts (Urtica dioica L.) obtained with supercritical carbon dioxide. Acta Pharm. 2001, 51, 45–51. [Google Scholar]
- Đurović, S.; Zekovic, Z.; Šorgić, S.; Popov, S.; Vujanović, M.; Radojković, M. Fatty acid profile of stinging nettle leaves: Application of modern analytical procedures for sample preparation and analysis. Anal. Methods 2018, 10, 1080–1087. [Google Scholar] [CrossRef]
- D’Abrosca, B.; Ciaramella, V.; Graziani, V.; Papaccio, F.; Della Corte, C.M.; Potenza, N.; Fiorentino, A.; Ciardiello, F.; Morgillo, F. Urtica dioica L. inhibits proliferation and enhances cisplatin cytotoxicity in NSCLC cells via Endoplasmic Reticulum-stress mediated apoptosis. Sci. Rep. 2019, 9, 4986. [Google Scholar] [CrossRef]
- Adhikari, B.M.; Bajracharya, A.; Shrestha, A.K. Comparison of nutritional properties of Stinging nettle (Urtica dioica) flour with wheat and barley flours. Food Sci. Nutr. 2016, 4, 119–124. [Google Scholar] [CrossRef]
- Farag, M.A.; Weigend, M.; Luebert, F.; Brokamp, G.; Wessjohann, L.A. Phytochemical, phylogenetic, and anti-inflammatory evaluation of 43 Urtica accessions (stinging nettle) based on UPLC-Q-TOF-MS metabolomic profiles. Phytochemistry 2013, 96, 170–183. [Google Scholar] [CrossRef]
- Yan, X.G. New chemical constituents of roots of Urtica triangularis HAND-MASS. Chem. Pharm. Bull. 2008, 56, 1463–1465. [Google Scholar] [CrossRef] [PubMed]
- Ðurovic, S.; Pavlic, B.; Šorgic, S.; Popov, S.; Savic, S.; Pertonijevic, M.; Radojkovic, M.; Cvetanovic, A.; Zekovic, Z. Chemical composition of stinging nettle leaves obtained by different analytical approaches. J. Funct. Food. 2017, 32, 18–26. [Google Scholar] [CrossRef]
- Orčić, D.; Francišković, M.; Bekvalac, K.; Svirčev, E.; Beara, I.; Lesjak, M.; Mimica-Dukić, N. Quantitative determination of plant phenolics in Urtica dioica extracts by high-performance liquid chromatography coupled with tandem mass spectrometric detection. Food Chem. 2014, 143, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Otles, S.; Yalcin, B. Phenolic compounds analysis of root, stalk, and leaves of nettle. Sci. World J. 2012, 2012, 564367. [Google Scholar] [CrossRef] [PubMed]
- Pinelli, P.; Ieri, F.; Vignolini, P.; Bacci, L.; Baronti, S.; Romani, A. Extraction and HPLC analysis of phenolic compounds in leaves, stalks, and textile fibers of Urtica dioica L. J. Agric. Food Chem. 2008, 56, 9127–9132. [Google Scholar] [CrossRef] [PubMed]
- Krauss, R.; Spitteler, G. Phenolic compounds from roots of Urtica dioica. Phytochemistry 1990, 29, 1653–1659. [Google Scholar] [CrossRef]
- Grevsen, K.; Frette, X.C.; Christensen, L.P. Concentration and composition of flavonol glycosides and phenolic acids in aerial parts of stinging nettle (Urtica dioica L.) are affected by nitrogen fertilization and by harvest time. Eur. J. Hortic. Sci. 2008, 73, 20–27. [Google Scholar]
- Chaurasia, N.; Wichtl, M. Flavonol Glycosides from Urtica dioica. Planta Med. 1987, 53, 432–434. [Google Scholar] [CrossRef]
- Guil-Guerrero, J.L.; Rebolloso-Fuentes, M.M.; Isasa, M.E.T. Fatty acids and carotenoids from Stinging Nettle (Urtica dioica L.). J. Food Compos. Anal. 2003, 16, 111–119. [Google Scholar] [CrossRef]
- Hirano, T.; Homma, M.; Oka, K. Effects of stinging nettle root extracts and their steroidal components on the Na+, K+-ATPase of the benign prostatic hyperplasia. Planta Med. 1994, 60, 30–33. [Google Scholar] [CrossRef]
- Chaurasia, N.; Wichtl, M. Sterols and Steryl Glycosides from Urtica dioica. J. Nat. Prod. 1987, 50, 881–885. [Google Scholar] [CrossRef]
- Sajfrtová, M.; Sovova, H.; Opletal, L.; Bártlová, M. Near-critical extraction of b-sitosterol and scopoletin from stinging nettle roots. J. Supercrit. Fluid 2005, 35, 111–118. [Google Scholar] [CrossRef]
- Carvalho, A.R.; Costab, G.; Figueirinha, A.; Liberal, J.; Prior, J.A.V.; Lopes, M.C.; Cruz, M.T.; Batista, M.T. Urtica spp.: Phenolic composition, safety, antioxidant and anti-inflammatory activities. Food Res. Int. 2017, 99, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Lapinskaya, E.S.; Kopytko, Y. Composition of the lipophilic fraction of stinging nettle (Urtica dioica L. and U. urens L.) homeopathic matrix tinctures. Pharm. Chem. J. 2008, 42, 699–702. [Google Scholar] [CrossRef]
- Di Maro, A.; Pacifico, S.; Fiorentino, A.; Galasso, S.; Gallicchio, M.; Guida, V.; Severino, V.; Monaco, P.; Parente, A. Raviscanina wild asparagus (Asparagus acutifolius L.): A nutritionally valuable crop with antioxidant and antiproliferative properties. Food Res. Int. 2013, 53, 180–188. [Google Scholar] [CrossRef]
- Pacifico, S.; Galasso, S.; Piccolella, S.; Kretschmer, N.; Pan, S.; Marciano, S.; Bauer, R.; Monaco, P. Seasonal variation in phenolic composition and antioxidant and anti-inflammatory activities of Calamintha nepeta (L.) Savi. Food Res. Int. 2015, 69, 121–132. [Google Scholar] [CrossRef]
- Pacifico, S.; Piccolella, S.; Nocera, P.; Tranquillo, E.; Dal Poggetto, F.; Catauro, M. New insights into phenol and polyphenol composition of Stevia rebaudiana leaves. J. Pharm. Biomed. Anal. 2019, 163, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, S.; Piccolella, S. Plant-Derived Polyphenols: A Chemopreventive and Chemoprotectant Worth-Exploring Resource in Toxicology. In Advances in Molecular Toxicology; Fishbein, J.C., Heilman, J.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 161–214. [Google Scholar]
- Spina, M.; Cuccioloni, M.; Sparapani, L.; Acciarri, S.; Eleuteri, A.M.; Fioretti, E.; Angeletti, M. Comparative evaluation of flavonoid content in assessing quality of wild and cultivated vegetables for human consumption. J. Sci. Food Agric. 2008, 88, 294–304. [Google Scholar] [CrossRef]
- Akbay, P.; Basaran, A.A.; Undeger, U.; Basaran, N. In vitro immunomodulatory activity of flavonoid glycosides from Urtica dioica L. Phytother. Res. 2003, 17, 34–37. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, I.; Küfrevioglu, O.I.; Oktay, M.; Büyükokuroglu, M.E. Antioxidant, antimicrobial, antiulcer and analgesic activities of nettle (Urtica dioica L.). J. Ethnopharmacol. 2004, 90, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, J.E.; Roufogalis, B.D.; Wagner, H.; Chrubasik, S.A. A comprehensive review on nettle effect and efficacy profiles, Part I: Herba urticae. Phytomedicine 2007, 14, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, J.E.; Roufogalis, B.D.; Wagner, H.; Chrubasik, S. A comprehensive review on the stinging nettle effect and efficacy profiles. Part II: Urticae radix. Phytomedicine 2007, 14, 568–579. [Google Scholar] [CrossRef] [PubMed]
- El Haouari, M.; Rosado, J.A. Phytochemical, Anti-diabetic and Cardiovascular Properties of Urtica dioica L. (Urticaceae): A Review. Mini Rev. Med. Chem. 2019, 19, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Zemmouri, H.; Sekiou, O.; Ammar, S.; El Feki, A.; Bouaziz, M.; Messarah, M.; Boumendjel, A. Urtica dioica attenuates ovalbumin-induced inflammation and lipid peroxidation of lung tissues in rat asthma model. Pharm. Biol. 2017, 55, 1561–1568. [Google Scholar] [CrossRef]
- Schulze-Tanzil, G. Effects of the antirheumatic remedy hox alpha-a new stinging nettle leaf extract–on matrix metalloproteinases in human chondrocytes in vitro. Histol. Histopathol. 2002, 17, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Riehemann, K.; Behnke, B.; Schulze-Osthoff, K. Plant extracts from stinging nettle (Urtica dioica), an antirheumatic remedy, inhibit the proinflammatory transcription factor NF-kappaB. FEBS Lett. 1999, 442, 89–94. [Google Scholar] [CrossRef]
- Bisht, R.; Joshi, B.C.; Kalia, A.N.; Prakash, A. Antioxidant-Rich Fraction of Urtica dioica Mediated Rescue of Striatal Mito-Oxidative Damage in MPTP-Induced Behavioral, Cellular, and Neurochemical Alterations in Rats. Mol. Neurobiol. 2017, 54, 5632–5645. [Google Scholar] [CrossRef]
- Di Sotto, A.; Mazzanti, G.; Savickiene, N.; Staršelskytė, R.; Baksenskaite, V.; Di Giacomo, S.; Vitalone, A. Antimutagenic and antioxidant activity of a protein fraction from aerial parts of Urtica dioica. Pharm. Biol. 2015, 53, 935–938. [Google Scholar] [CrossRef]
- Fattahi, S.; Ardekani, A.M.; Zabihi, E.; Abedian, Z.; Mostafazadeh, A.; Pourbagher, R.; Akhavan-Niaki, H. Antioxidant and apoptotic effects of an aqueous extract of Urtica dioica on the MCF-7 human breast cancer cell line. Asian Pac. J. Cancer Prev. 2013, 14, 5317–5323. [Google Scholar] [CrossRef]
- Kukric, Z.; Topalić-Trivunović, L.; Kukavica, B.; Matos, S.; Pavicic, S.; Boroja, M.; Savić, A. Characterization of antioxidant and antimicrobial activities of nettle leaves (Urtica dioica L.). Acta Period. Technol. 2012, 43, 257–272. [Google Scholar] [CrossRef]
- Ghaima, K.K.; Hashim, N.M.; Ali, S.A. Antibacterial and antioxidant activities of ethyl acetate extract of nettle (Urtica dioica) and dandelion (Taraxacum officinale). J. Pharm. Sci. 2013, 3, 96–99. [Google Scholar]
- Kregiel, D.; Pawlikowska, E.; Antolak, H. Urtica spp.: Ordinary Plants with Extraordinary Properties. Molecules 2018, 23, 1664. [Google Scholar] [CrossRef] [PubMed]
- Batool, R.; Salahuddin, H.; Mahmood, T.; Ismail, M. Study of anticancer and antibacterial activities of Foeniculum vulgare, Justicia adhatoda and Urtica dioica as natural curatives. Cell. Mol. Biol. 2017, 63, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Ranjbari, A.; Azarbayjani, M.A.; Yusof, A.; Halim Mokhtar, A.; Akbarzadeh, S.; Ibrahim, M.Y.; Tarverdizadeh, B.; Farzadinia, P.; Hajiaghaee, R.; Dehghan, F. In vivo and in vitro evaluation of the effects of Urtica dioica and swimming activity on diabetic factors and pancreatic beta cells. BMC Complement. Altern. Med. 2016, 16, 101. [Google Scholar] [CrossRef]
- Domola, M.S.; Vu, V.; Robson-Doucette, C.A.; Sweeney, G.; Wheeler, M.B. Insulin mimetics in Urtica dioica: Structural and computational analyses of Urtica dioica extracts. Phytother. Res. 2010, 24 (Suppl. 2), S175–S182. [Google Scholar] [CrossRef]
- Francišković, M.; Gonzalez-Pérez, R.; Orčić, D.; Sánchez de Medina, F.; Martínez-Augustin, O.; Svirčev, E.; Simin, N.; Mimica-Dukić, N. Chemical Composition and Immuno-Modulatory Effects of Urtica dioica L. (Stinging Nettle) Extracts. Phytother. Res. 2017, 31, 1183–1191. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends--An Update. Cancer Epidemiol. Biomark. Prev. 2016, 25, 16–27. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Gelmann, E.P.; Sawyers, C.L.; Rauscher, F.J.I. Molecular Oncology: Causes of Cancer and Targets for Treatment; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Steward, W.P.; Brown, K. Cancer chemoprevention: A rapidly evolving field. Br. J. Cancer 2013, 109, 1–7. [Google Scholar] [CrossRef]
- Kucuk, O. New opportunities in chemoprevention research. Cancer Investig. 2002, 20, 237–245. [Google Scholar] [CrossRef]
- Kocasli, S.; Demircan, Z. Herbal product use by the cancer patients in both the pre and post surgery periods and during chemotherapy. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 325–333. [Google Scholar] [CrossRef][Green Version]
- Liao, G.S.; Apaya, M.K.; Shyur, L.F. Herbal medicine and acupuncture for breast cancer palliative care and adjuvant therapy. Evid. Based Complement. Altern. Med. 2013, 2013, 437948. [Google Scholar] [CrossRef]
- Nahata, A.; Saxena, A.; Suri, N.; Saxena, A.K.; Dixit, V.K. Sphaeranthus indicus induces apoptosis through mitochondrial-dependent pathway in HL-60 cells and exerts cytotoxic potential on several human cancer cell lines. Integr. Cancer Ther. 2013, 12, 236–247. [Google Scholar] [CrossRef]
- Olaku, O.; White, J.D. Herbal therapy use by cancer patients: A literature review on case reports. Eur. J. Cancer 2011, 47, 508–514. [Google Scholar] [CrossRef]
- Balunas, M.J.; Kinghorn, A.D. Drug discovery from medicinal plants. Life Sci. 2005, 78, 431–441. [Google Scholar] [CrossRef]
- D’Incalci, M.; Steward, W.P.; Gescher, A.J. Use of cancer chemopreventive phytochemicals as antineoplastic agents. Lancet Oncol. 2005, 6, 899–904. [Google Scholar] [CrossRef]
- Paterson, I.; Anderson, E.A. The renaissance of natural products as drug candidates. Science 2005, 310. [Google Scholar] [CrossRef]
- Smit, A.J. Medicinal and pharmaceutical uses of seaweed natural products: A review. J. Appl. Phycol. 2004, 16. [Google Scholar] [CrossRef]
- Jung Park, E.; Pezzuto, J.M. Botanicals in cancer chemoprevention. Cancer Metastasis Rev. 2002, 21. [Google Scholar] [CrossRef]
- Mann, J. Natural products in cancer chemotherapy: Past, present and future. Nat. Rev. Cancer 2002, 2. [Google Scholar] [CrossRef]
- Kandaswami, C.; Kanadaswami, C.; Lee, L.T.; Lee, P.P.; Hwang, J.J.; Ke, F.C.; Huang, Y.T.; Lee, M.T. The antitumor activities of flavonoids. In Vivo 2005, 19, 895–909. [Google Scholar]
- Kawaii, S.; Tomono, Y.; Katase, E.; Ogawa, K.; Yano, M. Antiproliferative activity of flavonoids on several cancer cell lines. Biosci. Biotechnol. Biochem. 1999, 63. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Plants as a source of anti-cancer agents. J. Ethnopharmacol. 2005, 100, 72–79. [Google Scholar] [CrossRef]
- Jin, Z.; El-Deiry, W.S. Overview of cell death signaling pathways. Cancer Biol. Ther. 2005, 4, 139–163. [Google Scholar] [CrossRef]
- Ciniglia, C.; Mastrobuoni, F.; Scortichini, M.; Petriccione, M. Oxidative damage and cell-programmed death induced in Zea mays L. by allelochemical stress. Ecotoxicology 2015, 24, 926–937. [Google Scholar] [CrossRef]
- Florea, A.M.; Busselberg, D. Cisplatin as an anti-tumor drug: Cellular mechanisms of activity, drug resistance and induced side effects. Cancers 2011, 3, 1351. [Google Scholar] [CrossRef]
- Siddik, Z.H. Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 22. [Google Scholar] [CrossRef]
- Ozkol, H.; Musa, D.; Tuluce, Y.; Koyuncu, I.; Asadi-Samani, M.; Rafieian-Kopaei, M.; Lorigooini, Z.; Shirzad, H. Ameliorative influence of Urtica dioica L against cisplatin-induced toxicity in mice bearing Ehrlich ascites carcinoma. Drug Chem. Toxicol. 2012, 35, 251–257. [Google Scholar] [CrossRef]
- Nollet, L.M.L.; Gutierrez-Uribe, J.A. Phenolic Compounds in Food: Characterization and Analysis; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Faramarzi, S.; Pacifico, S.; Yadollahi, A.; Lettieri, A.; Nocera, P.; Piccolella, S. Red-fleshed Apples: Old Autochthonous Fruits as a Novel Source of Anthocyanin Antioxidants. Plant Foods Hum. Nutr. 2015, 70, 324–330. [Google Scholar] [CrossRef]
- Keskin-Šašic, I.; Tahirovic, A.; Topˇcagic, A.; Klepo, L.; Salihovic, M.; Ibragic, S.; Toromanovi´, J.; Ajanovic, A.; Velispahic, E. Total phenolic content and antioxidant capacity of fruit juices. Bull. Chem. Technol. Bosnia Herzeg. 2012, 39, 25–28. [Google Scholar]
- Wang, C.; Yu, J.; Wang, H.; Zhang, J.; Wu, N. Lipid peroxidation and altered anti-oxidant status in breast adenocarcinoma patients. Drug Res. 2014, 64, 690–692. [Google Scholar] [CrossRef]
- Ray, G.; Batra, S.; Shukla, N.K.; Deo, S.; Raina, V.; Ashok, S.; Husain, S.A. Lipid peroxidation, free radical production and antioxidant status in breast cancer. Breast Cancer Res. Treat. 2000, 59, 163–170. [Google Scholar] [CrossRef]
- Khanzode, S.S.; Muddeshwar, M.G.; Khanzode, S.D.; Dakhale, G.N. Antioxidant Enzymes and Lipid Peroxidation in Different Stages of Breast Cancer. Free Radic. Res. 2004, 38, 81–85. [Google Scholar] [CrossRef]
- Benz, E.J.; Nathan, D.G.; Amaravadi, R.K.; Danial, N.N. Targeting the cell death-survival equation. Clin. Cancer Res. 2007, 13, 7250–7253. [Google Scholar] [CrossRef]
- Taraphdar, A.K.; Roy, M.; Bhattacharya, R.K. Natural products as inducers of apoptosis: Implication for cancer therapy and prevention. Curr. Sci. 2001, 80, 1387–1396. [Google Scholar]
- Wagner, H.; Willer, F.; Samtleben, R.; Boos, G. Search for the antiprostatic principle of stinging nettle (Urtica dioica) roots. Phytomedicine 1994, 1, 213–224. [Google Scholar] [CrossRef]
- Ghasemi, S.; Moradzadeh, M.; Mousavi, S.H.; Sadeghnia, H.R. Cytotoxic effects of Urtica dioica radix on human colon (HT29) and gastric (MKN45) cancer cells mediated through oxidative and apoptotic mechanisms. Cell. Mol. Biol. 2016, 62, 90–96. [Google Scholar]
- Mohammadi, A.; Mansoori, B.; Aghapour, M.; Baradaran, P.C.; Shajari, N.; Davudian, S.; Salehi, S.; Baradaran, B. The Herbal Medicine Utrica Dioica Inhibits Proliferation of Colorectal Cancer Cell Line by Inducing Apoptosis and Arrest at the G2/M Phase. J. Gastrointest. Cancer 2016, 47, 187–195. [Google Scholar] [CrossRef]
- Lichius, J.J.; Muth, C. The inhibiting effects of Urtica dioica root extracts on experimentally induced prostatic hyperplasia in the mouse. Planta Med. 1997, 63, 307–310. [Google Scholar] [CrossRef]
- Konrad, L.; Müller, H.H.; Lenz, C.; Laubinger, H.; Aumüller, G.; Lichius, J.J. Antiproliferative effect on human prostate cancer cells by a stinging nettle root (Urtica dioica) extract. Planta Med. 2000, 66, 44–47. [Google Scholar] [CrossRef]
- Durak, I.; Biri, H.; Devrim, E.; Sözen, S.; Avci, A. Aqueous extract of Urtica dioica makes significant inhibition on adenosine deaminase activity in prostate tissue from patients with prostate cancer. Cancer Biol. Ther. 2004, 3, 855–857. [Google Scholar] [CrossRef]
- Nahata, A.; Dixit, V.K. Ameliorative effects of stinging nettle (Urtica dioica) on testosterone-induced prostatic hyperplasia in rats. Andrologia 2012, 44 (Suppl. 1), 396–409. [Google Scholar] [CrossRef]
- Mohammadi, A.; Mansoori, B.; Aghapour, M.; Baradaran, B. Urtica dioica dichloromethane extract induce apoptosis from intrinsic pathway on human prostate cancer cells (PC3). Cell. Mol. Biol. 2016, 62, 78–83. [Google Scholar]
- Wang, G.; Reed, E.; Li, Q.Q. Molecular basis of cellular response to cisplatin chemotherapy in non-small cell lung cancer (Review). Oncol. Rep. 2004, 12. [Google Scholar] [CrossRef]
- Abdelrahim, M.; Newman, K.; Vanderlaag, K.; Samudio, I.; Safe, S. 3, 3′-Diindolylmethane (DIM) and its derivatives induce apoptosis in pancreatic cancer cells through endoplasmic reticulum stress-dependent upregulation of DR5. Carcinogenesis 2006, 27. [Google Scholar] [CrossRef]
- Oyadomari, S.; Mori, M. Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ. 2004, 11. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Wang, H.G. CHOP is involved in endoplasmic reticulum stress-induced apoptosis by enhancing DR5 expression in human carcinoma cells. J. Biol. Chem. 2004, 279. [Google Scholar] [CrossRef]
- Kelley, S.K.; Ashkenazi, A. Targeting death receptors in cancer with Apo2L/TRAIL. Curr. Opin. Pharmacol. 2004, 4. [Google Scholar] [CrossRef]
- Randa, A.; Kamel, S. Bioactive oxylipins from the endophyte Khuskia oryzae isolated from the medicinal plant Bidens alba. Eur. J. Biomed. Pharm. Sci. 2015, 2, 630–639. [Google Scholar]
- Romano, G. Design and synthesis of pro-apoptotic compounds inspired by diatom oxylipins. Mar. Drugs 2013, 11, 4527. [Google Scholar] [CrossRef]
- Gerwick, W.H. Biologically active oxylipins from seaweeds. Hydrobiologia 1993, 260–261. [Google Scholar] [CrossRef]
- Hwang, J.H.; Voortman, J.; Giovannetti, E.; Steinberg, S.M.; Leon, L.G.; Kim, Y.T.; Funel, N.; Park, J.K.; Kim, M.A.; Kang, G.H.; et al. Identification of microRNA-21 as a biomarker for chemoresistance and clinical outcome following adjuvant therapy in resectable pancreatic cancer. PLoS ONE 2010, 5, e10630. [Google Scholar] [CrossRef]
- Verpoorte, R.; Choi, Y.H.; Kim, H.K. NMR-based metabolomics at work in phytochemistry. Phytochem. Rev. 2007, 6. [Google Scholar] [CrossRef]
- Gabbs, M.; Leng, S.; Devassy, J.G.; Monirujjaman, M.; Aukema, H.M. Advances in Our Understanding of Oxylipins Derived from Dietary PUFAs. Adv. Nutr. 2015, 6, 513–540. [Google Scholar] [CrossRef]
- Baldin, V.; Lukas, J.; Marcote, M.J.; Pagano, M.; Draetta, G. Cyclin D1 is a nuclear protein required for cell cycle progression in G1. Genes Dev 1993, 7, 812–821. [Google Scholar] [CrossRef]
- Altiok, N.; Mezzadra, H.; Patel, P.; Koyuturk, M.; Altiok, S. A plant oxylipin, 12-oxo-phytodienoic acid, inhibits proliferation of human breast cancer cells by targeting cyclin D1. Breast Cancer Res. Treat. 2008, 109. [Google Scholar] [CrossRef]
- Does, M.P.; Ng, D.K.; Dekker, H.L.; Peumans, W.J.; Houterman, P.M.; Van Damme, E.J.; Cornelissen, B.J. Characterization of Urtica dioica agglutinin isolectins and the encoding gene family. Plant Mol. Biol. 1999, 39, 335–347. [Google Scholar] [CrossRef]
- Wagner, H.; Geiger, W.N.; Boos, G.; Samtleben, R. Studies on the binding of Urtica dioica agglutinin (UDA) and other lectins in an in vitro epidermal growth factor receptor test. Phytomedicine 1995, 1, 287–290. [Google Scholar] [CrossRef]
- Steers, W.D. 5alpha-reductase activity in the prostate. Urology 2001, 58, 17–24. [Google Scholar] [CrossRef]
- Valitova, J.N.; Sulkarnayeva, A.G.; Minibayeva, F.V. Plant Sterols: Diversity, Biosynthesis, and Physiological Functions. Biochemistry 2016, 81, 819–834. [Google Scholar] [CrossRef]
- Berges, R.R.; Windeler, J.; Trampisch, H.J.; Senge, T. Randomised, placebo-controlled, double-blind clinical trial of beta-sitosterol in patients with benign prostatic hyperplasia. Beta-sitosterol Study Group. Lancet 1995, 345, 1529–1532. [Google Scholar] [CrossRef]
- Wilt, T.J.; MacDonald, R.; Ishani, A. beta-sitosterol for the treatment of benign prostatic hyperplasia: A systematic review. BJU Int. 1999, 83, 976–983. [Google Scholar] [CrossRef]
- Bourgaud, F.; Hehn, A.; Larbat, R.; Doerper, S.; Gontier, E.; Kellner, S.; Matern, U. Biosynthesis of coumarins in plants: A major pathway still to be unravelled for cytochrome P450 enzymes. Phytochem. Rev. 2006, 5, 293–308. [Google Scholar] [CrossRef]
- Ding, Z.; Dai, Y.; Hao, H.; Pan, R.; Yao, X.; Wang, Z. Anti-inflammatory effects of scopoletin and underlying mechanisms. Pharm. Biol. 2008, 46, 854–860. [Google Scholar] [CrossRef]
- Liu, X.L.; Zhang, L.; Fu, X.L.; Chen, K.; Qian, B.C. Effect of scopoletin on PC3 cell proliferation and apoptosis. Acta Pharmacol. Sin. 2001, 22, 929–933. [Google Scholar]
- Kohler, B.A.; Sherman, R.L.; Howlader, N.; Jemal, A.; Ryerson, A.B.; Henry, K.A.; Boscoe, F.P.; Cronin, K.A.; Lake, A.; Noone, A.-M.; et al. Annual Report to the Nation on the Status of Cancer, 1975–2011, Featuring Incidence of Breast Cancer Subtypes by Race/Ethnicity, Poverty, and State. JNCI J. Natl. Cancer Inst. 2015, 107. [Google Scholar] [CrossRef]
- DeSantis, C.; Ma, J.; Bryan, L.; Jemal, A. Breast cancer statistics, 2013. CA Cancer J. Clin. 2014, 64, 52–62. [Google Scholar] [CrossRef]
- Sharma, G.N.; Dave, R.; Sanadya, J.; Sharma, P.; Sharma, K.K. Various types and management of breast cancer: An overview. J. Adv. Pharm. Technol. Res. 2010, 1, 109–126. [Google Scholar]
- Nounou, M.I.; ElAmrawy, F.; Ahmed, N.; Abdelraouf, K.; Goda, S.; Syed-Sha-Qhattal, H. Breast Cancer: Conventional Diagnosis and Treatment Modalities and Recent Patents and Technologies. Breast Cancer 2015, 9, 17–34. [Google Scholar] [CrossRef]
- Mitra, S.; Dash, R. Natural Products for the Management and Prevention of Breast Cancer. Evid. Based Complement. Altern. Med. 2018, 2018, 8324696. [Google Scholar] [CrossRef]
- Lin, B.; Kolluri, S.K.; Lin, F.; Liu, W.; Han, Y.H.; Cao, X.; Dawson, M.I.; Reed, J.C.; Zhang, X.K. Conversion of Bcl-2 from protector to killer by interaction with nuclear orphan receptor Nur77/TR3. Cell 2004, 116, 527–540. [Google Scholar] [CrossRef]
- Fattahi, S.; Ghadami, E.; Asouri, M.; Motevalizadeh Ardekanid, A.; Akhavan-Niaki, H. Urtica dioica inhibits cell growth and induces apoptosis by targeting Ornithine decarboxylase and Adenosine deaminase as key regulatory enzymes in adenosine and polyamines homeostasis in human breast cancer cell lines. Cell. Mol. Biol. 2018, 64, 97–102. [Google Scholar] [CrossRef]
- Tullo, A.; Mastropasqua, G.; Bourdon, J.C.; Centonze, P.; Gostissa, M.; Costanzo, A.; Levrero, M.; Del Sal, G.; Saccone, C.; Sbisà, E. Adenosine deaminase, a key enzyme in DNA precursors control, is a new p73 target. Oncogene 2003, 22, 8738–8748. [Google Scholar] [CrossRef][Green Version]
- Zhu, Q.; Jin, L.; Casero, R.A.; Davidson, N.E.; Huang, Y. Role of ornithine decarboxylase in regulation of estrogen receptor alpha expression and growth in human breast cancer cells. Breast Cancer Res. Treat. 2012, 136, 57–66. [Google Scholar] [CrossRef][Green Version]
- Apasov, S.G.; Blackburn, M.R.; Kellems, R.E.; Smith, P.T.; Sitkovsky, M.V. Adenosine deaminase deficiency increases thymic apoptosis and causes defective T cell receptor signaling. J. Clin. Investig. 2001, 108, 131–141. [Google Scholar] [CrossRef]
- Nowotarski, S.L.; Woster, P.M.; Casero, R.A. Polyamines and cancer: Implications for chemotherapy and chemoprevention. Expert Rev. Mol. Med. 2013, 15, e3. [Google Scholar] [CrossRef]
- Gerner, E.W.; Meyskens, F.L. Polyamines and cancer: Old molecules, new understanding. Nat. Rev. Cancer 2004, 4, 781–792. [Google Scholar] [CrossRef]
- Jalili, C.; Salahshoor, M.R.; Yousefi, D.; Khazaei, M.; Shabanizadeh Darehdori, A.; Mokhtari, T. Morphometric and Hormonal Study of the Effect of Utrica diocia Extract on Mammary Glands in Rats. Int. J. Morphol. 2015, 983–987. [Google Scholar] [CrossRef]
- Abu-Dahab, R.; Afifi, F. Antiproliferative activity of selected medicinal plants of Jordan against a breast adenocarcinoma cell line (MCF7). Sci. Pharm. 2007, 75. [Google Scholar] [CrossRef]
- Mohammadi, A.; Mansoori, B.; Goldar, S.; Shanehbandi, D.; Khaze, V.; Mohammadnejad, L.; Baghbani, E.; Baradaran, B. Effects of Urtica dioica dichloromethane extract on cell apoptosis and related gene expression in human breast cancer cell line (MDA-MB-468). Cell. Mol. Biol. 2016, 62, 62–67. [Google Scholar]
- Mohammadi, A.; Mansoori, B.; Aghapour, M.; Shirjang, S.; Nami, S.; Baradaran, B. The Urtica dioica extract enhances sensitivity of paclitaxel drug to MDA-MB-468 breast cancer cells. Biomed. Pharmacother. 2016, 83, 835–842. [Google Scholar] [CrossRef]
- Mansoori, B.; Mohammadi, A.; Hashemzadeh, S.; Shirjang, S.; Baradaran, A.; Asadi, M.; Doustvandi, M.A.; Baradaran, B. Urtica dioica extract suppresses miR-21 and metastasis-related genes in breast cancer. Biomed. Pharmacother. 2017, 93, 95–102. [Google Scholar] [CrossRef]
- Mohammadi, A.; Mansoori, B.; Baradaran, P.C.; Khaze, V.; Aghapour, M.; Farhadi, M.; Baradaran, B. Urtica dioica Extract Inhibits Proliferation and Induces Apoptosis and Related Gene Expression of Breast Cancer Cells In Vitro and In Vivo. Clin. Breast Cancer 2017, 17, 463–470. [Google Scholar] [CrossRef]
- Telo, S.; Halifeoglu, I.; Ozercan, I.H. Effects of Stinging Nettle (Urtica dioica L.,) on Antioxidant Enzyme Activities in Rat Model of Mammary Gland Cancer. Iran. J. Pharm. Res. 2017, 16, 164–170. [Google Scholar]
- Mekhail, T.M.; Markman, M. Paclitaxel in cancer therapy. Expert Opin. Pharmacother. 2002, 3, 755–766. [Google Scholar]
- Yarrow, J.C.; Perlman, Z.E.; Westwood, N.J.; Mitchison, T.J. A high-throughput cell migration assay using scratch wound healing, a comparison of image-based readout methods. BMC Biotechnol. 2004, 4, 21. [Google Scholar] [CrossRef]
- Savary, K.; Caglayan, D.; Caja, L.; Tzavlaki, K.; Bin Nayeem, S.; Bergström, T.; Jiang, Y.; Uhrbom, L.; Forsberg-Nilsson, K.; Westermark, B.; et al. Snail depletes the tumorigenic potential of glioblastoma. Oncogene 2013, 32, 5409. [Google Scholar] [CrossRef]
- Waldmann, J.; Feldmann, G.; Slater, E.P.; Langer, P.; Buchholz, M.; Ramaswamy, A.; Saeger, W.; Rothmund, M.; Fendrich, V. Expression of the zinc-finger transcription factor Snail in adrenocortical carcinoma is associated with decreased survival. Br. J. Cancer 2008, 99, 1900–1907. [Google Scholar] [CrossRef]
- Batlle, E.; Sancho, E.; Francí, C.; Domínguez, D.; Monfar, M.; Baulida, J.; García De Herreros, A. The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat. Cell Biol. 2000, 2, 84–89. [Google Scholar] [CrossRef]
- Tran, H.D.; Luitel, K.; Kim, M.; Zhang, K.; Longmore, G.D.; Tran, D.D. Transient SNAIL1 expression is necessary for metastatic competence in breast cancer. Cancer Res. 2014, 74, 6330–6340. [Google Scholar] [CrossRef]
- Lin, H.-C.; Cheng, Y.-W.; Hsu, N.-Y. The association of miR-21, HER-2/neu, and PTEN expression and clinical outcome of breast cancer. Cancer Res. 2014, 74 (Suppl. 19), 1470. [Google Scholar]
- Liu, Z.L.; Wang, H.; Liu, J.; Wang, Z.X. MicroRNA-21 (miR-21) expression promotes growth, metastasis, and chemo- or radioresistance in non-small cell lung cancer cells by targeting PTEN. Mol. Cell. Biochem. 2013, 372, 35–45. [Google Scholar] [CrossRef]
- Cheng, H.; Shi, S.; Cai, X.; Long, J.; Xu, J.; Liu, C.; Yu, X. microRNA signature for human pancreatic cancer invasion and metastasis. Exp. Ther. Med. 2012, 4, 181–187. [Google Scholar] [CrossRef]
- Medina, P.P.; Nolde, M.; Slack, F.J. OncomiR addiction in an in vivo model of microRNA-21-induced pre-B-cell lymphoma. Nature 2010, 467, 86–90. [Google Scholar] [CrossRef]
- Huang, T.H.; Wu, F.; Loeb, G.B.; Hsu, R.; Heidersbach, A.; Brincat, A.; Horiuchi, D.; Lebbink, R.J.; Mo, Y.Y.; Goga, A.; et al. Up-regulation of miR-21 by HER2/neu signaling promotes cell invasion. J. Biol. Chem. 2009, 284, 18515–18524. [Google Scholar] [CrossRef]
- Nicoloso, M.S.; Spizzo, R.; Shimizu, M.; Rossi, S.; Calin, G.A. MicroRNAs—The micro steering wheel of tumour metastases. Nat. Rev. Cancer 2009, 9, 293–302. [Google Scholar] [CrossRef]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef]
- Merdad, A.; Karim, S.; Schulten, H.J.; Dallol, A.; Buhmeida, A.; Al-Thubaity, F.; Gari, M.A.; Chaudhary, A.G.; Abuzenadah, A.M.; Al-Qahtani, M.H. Expression of matrix metalloproteinases (MMPs) in primary human breast cancer: MMP-9 as a potential biomarker for cancer invasion and metastasis. Anticancer Res. 2014, 34, 1355–1366. [Google Scholar]
- Kokkinos, M.I.; Wafai, R.; Wong, M.K.; Newgreen, D.F.; Thompson, E.W.; Waltham, M. Vimentin and epithelial-mesenchymal transition in human breast cancer-observations in vitro and in vivo. Cells Tissues Organs 2007, 185, 191–203. [Google Scholar] [CrossRef]
- Catherine, C.M.; Shaffer, C.V.; Jacqueline, F. A novel CXCR4 pathway is required for migration of metastatic breast cancer cells. Cancer Res. 2015, 75 (Suppl. 15), 4043. [Google Scholar]
- Mukherjee, D.; Zhao, J. The Role of chemokine receptor CXCR4 in breast cancer metastasis. Am. J. Cancer Res. 2013, 3, 46–57. [Google Scholar]
- Canel, M.; Serrels, A.; Frame, M.C.; Brunton, V.G. E-cadherin-integrin crosstalk in cancer invasion and metastasis. J. Cell Sci. 2013, 126, 393–401. [Google Scholar] [CrossRef]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Gansser, D.; Spiteller, G. Aromatase inhibitors from Urtica dioica roots. Planta Med. 1995, 61, 138–140. [Google Scholar] [CrossRef]
| U. dioica Extracts * | Cancer Cell Lines | IC50 | Effects | References |
|---|---|---|---|---|
| U. dioica L. (Germany) roots aqueous extract U. dioica agglutinine (UDA) | HeLa human cervical cancer A431 human epidermoid carcinoma | 5 μg/mL (24 h treatment) 21 μg/mL (24 h treatment) | ↓ Proliferation ↓ EGF binding | Wagner 1994 [85] |
| U. dioica L. (Iran) roots ethanolic extract | HT29 human colon cancer MKN45 human gastric cancer | 24.7 μg/mL (72 h treatment) 249.9 μg/mL (72 h treatment) | ↓ Proliferation ↑ Apoptosis | Ghasemi 2016 [86] |
| U. dioica L. (Iran) aerial parts dichloromethane extract | HCT-116 human colon cancer | 23.61 μg/mL (48 h treatment) | ↓ Proliferation ↑ Apoptosis G2/M arrest | Mohammadi 2016 [87] |
| U. dioica L. (Italy) leaves methanolic extract, oxylipins | NSCLC H1299 human non-small cell lung cancer NSCLC A549 human non-small cell lung cancer | 52.3 μg/mL (72 h treatment) 47.5 μg/mL (72 h treatment) | ↓ Proliferation ↑ Apoptosis extrinsic pathway ↑ caspase 3 ↑ caspase 8 ↑ cPARP ↑ tBid ↑ GADD153 ↑ DR5 G2/M arrest | D’Abrosca 2019 [12] |
| U. dioica Extracts * | Cancer Cell Lines/Tissues/Animal Models | IC50 | Effects | References |
|---|---|---|---|---|
| U. dioica L.(Germany) roots methanolic extract | Balb/c mouse model of benign prostatic hyperplasia (28 days, 5 mg oral treatment) | ↓ hyperplasia 51.4 % growth inhibition | Lichius 1997 [88] | |
| U. dioica L.(Germany) roots methanolic extract | LNCaP human prostate cancer | ↓ Proliferation 30% (5 day treatment with 1 μg/mL) | Konrad 2000 [89] | |
| U. dioica L. leaves aqueous extract | prostate tissue from prostate cancer patients | 50 μg/mL (30 min treatment) | ↓ ADA | Durak 2004 [90] |
| U. dioica L. (India) roots petroleum ether extract, β-sitosterol | rat model of benign prostatic hyperplasia (28 days, 50 mg/Kg oral treatment) | 0.19 mg/mL (28 day treatment) | ↓ hyperplasia ↓ 5α-reductase | Nahata 2012 [91] |
| roots ethanolic extract, β-sitosterol and scopoletin | 0.12 mg/mL (28 day treatment) | |||
| U. dioica L. (Iran) leaves dichloromethane extract | PC3 human prostate cancer | 15.54 μg/mL (48 h treatment) | ↓ Proliferation ↑ Apoptosis intrinsic pathway ↑ caspase 3 ↑ caspase 9 ↓ Bcl-2 G2/M arrest | Mohammadi 2016 [92] |
| Molecules | Biological Activities | Cells | References |
|---|---|---|---|
| 13-S-hydroxy-9Z, 11E, 15Z-octadecantrienoic acid (oxylipin) | Anti-proliferation ro-apoptotis Stop cell cycle Anti-inflammation | lung cancer chondrocytes | [12,41] |
| U. dioica agglutinine (UDA) | Anti-proliferation Anti-EGF binding | cervical cancer epidermoid carcinoma | [85,107] |
| β-sitosterol | Anti-proliferation Inhibition 5α-reductase | prostate | [91,110,111] |
| scopoletin | Anti-proliferation Inhibition 5α-reductase Anti-inflammation Pro-apoptotis | prostate prostate cancer | [91,113,114] |
| U. dioica Extracts * | Cancer Cell Lines/ Animal Models | IC50 | Effects | References |
|---|---|---|---|---|
| U. dioica L. (Jordan) leaves and stems ethanol extract | MCF-7 human breast cancer | ↓ Proliferation 7% (72 h treatment with 50 μg/mL) | Abu-Dahab 2007 [128] | |
| U. dioica, L. (Iran) leaves aqueous extract | MCF-7 human breast cancer | 2 mg/mL (72 h treatment) | ↓ Proliferation ↑ Apoptosis intrinsic pathway ↑ caspase 3 ↑ caspase 9 ↑ Bax ↑ Bcl-2 ↑ calpain 1 ↑ calpastatin | Fattahi 2013 [45] |
| U. dioica, L. (Iran) leaves dichloromethane extract | MDA-MB-468 human breast cancer | 15.54 μg/mL (48 h treatment) | ↓ Proliferation ↑ Apoptosis intrinsic pathway ↑ caspase 3 ↑ caspase 9 ↓ Bcl-2 | Mohammadi 2016 [129] |
| U. dioica, L. (Iran) leaves dichloromethane extract | MDA-MB-468 human breast cancer | 0.59 μM (24 h co-treatment paclitaxel + extract) | ↓ Proliferation ↑ Apoptosis ↓ Migration ↓ Snail-1 ↓ ZEB1, ZEB2, twist G2/M arrest ↓ Cdc2 | Mohammadi 2016 [130] |
| U. dioica, L. (Iran) leaves dichloromethane extract | MCF-7 human breast cancer MDA-MB-231human breast cancer 4T1 mouse breast cancer Balb/c mouse model of breast cancer (28 day, 20 mg/Kg injection treatment) | 31.37 mg/mL (48 h treatment) 38.14 mg/mL (48 h treatment) 35.21 mg/mL (48 h treatment) | ↓ Proliferation ↓ Migration ↓ miR-21 ↓ MMP1, MMP9, MMP13, vimentin, CXCR4 ↑ E-cadherin | Mansoori 2017 [131] |
| U. dioica, L. (Iran) leaves dichloromethane extract | Balb/c mouse model of breast cancer (28 day, 20 mg/Kg injection treatment) | ↓ Metastasis ↑ Apoptosis intrinsic pathway ↑ caspase 3 ↑ caspase 9 ↓ Bcl-2 ↓ Ki-67 | Mohammadi 2017 [132] | |
| U. dioica, aqueous extract | rat model of breast cancer (5.5 months, 50 g/kg food treatment) | ↓ Metastasis ↓ lipid peroxidation ↑ catalase | Telo 2017 [133] | |
| U. dioica, L. (Iran) leaves aqueous extract | MCF-7 human breast cancer | 2 mg/mL (72 h treatment) | ↓ Proliferation ↑ Apoptosis ↑ ADA ↑ ODC1 | Fattahi 2018 [121] |
| MDA-MB-231 human breast cancer | 2 mg/mL (72 h treatment) | ↓ Proliferation ↑ Apoptosis = ADA ↑ ODC1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, S.; Bianco, A.; Russo, R.; Di Maro, A.; Isernia, C.; Pedone, P.V. Therapeutic Perspectives of Molecules from Urtica dioica Extracts for Cancer Treatment. Molecules 2019, 24, 2753. https://doi.org/10.3390/molecules24152753
Esposito S, Bianco A, Russo R, Di Maro A, Isernia C, Pedone PV. Therapeutic Perspectives of Molecules from Urtica dioica Extracts for Cancer Treatment. Molecules. 2019; 24(15):2753. https://doi.org/10.3390/molecules24152753
Chicago/Turabian StyleEsposito, Sabrina, Alessandro Bianco, Rosita Russo, Antimo Di Maro, Carla Isernia, and Paolo Vincenzo Pedone. 2019. "Therapeutic Perspectives of Molecules from Urtica dioica Extracts for Cancer Treatment" Molecules 24, no. 15: 2753. https://doi.org/10.3390/molecules24152753
APA StyleEsposito, S., Bianco, A., Russo, R., Di Maro, A., Isernia, C., & Pedone, P. V. (2019). Therapeutic Perspectives of Molecules from Urtica dioica Extracts for Cancer Treatment. Molecules, 24(15), 2753. https://doi.org/10.3390/molecules24152753