Magnetic Polyurea Nano-Capsules Synthesized via Interfacial Polymerization in Inverse Nano-Emulsion
Abstract
1. Introduction
2. Results and Discussions
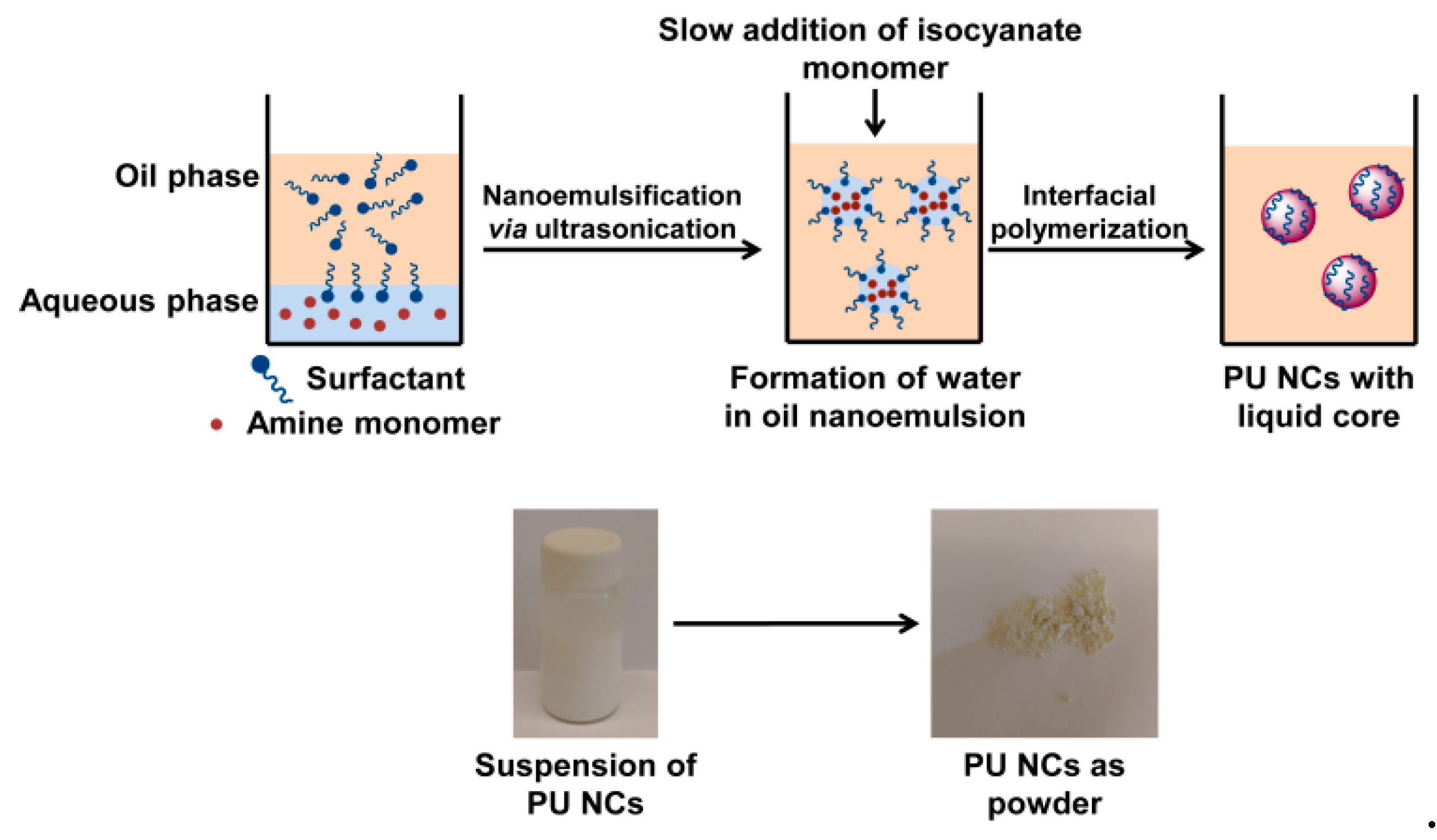
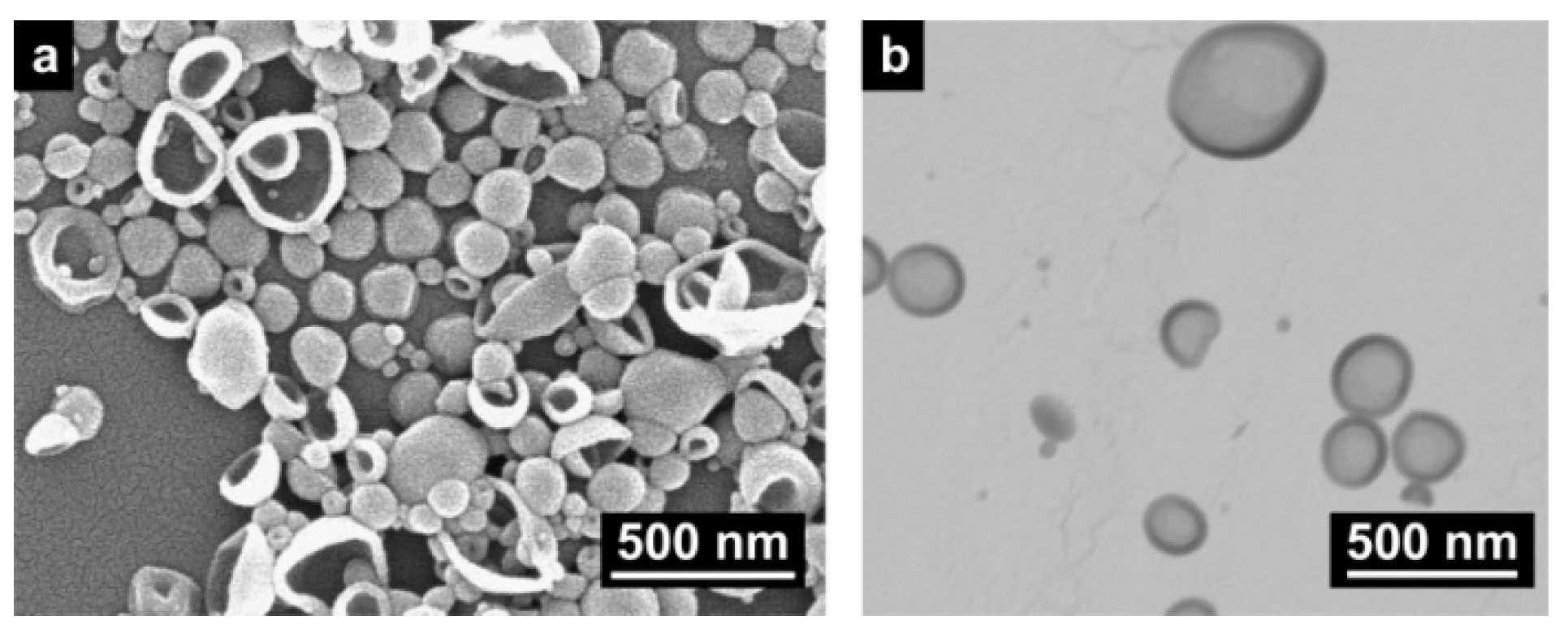
2.1. The Formation of Polyurea Nano-Capsules from Water-in-Oil (W/O) Nano-Emulsion
2.1.1. Variation of the Type and Amount of Surfactant
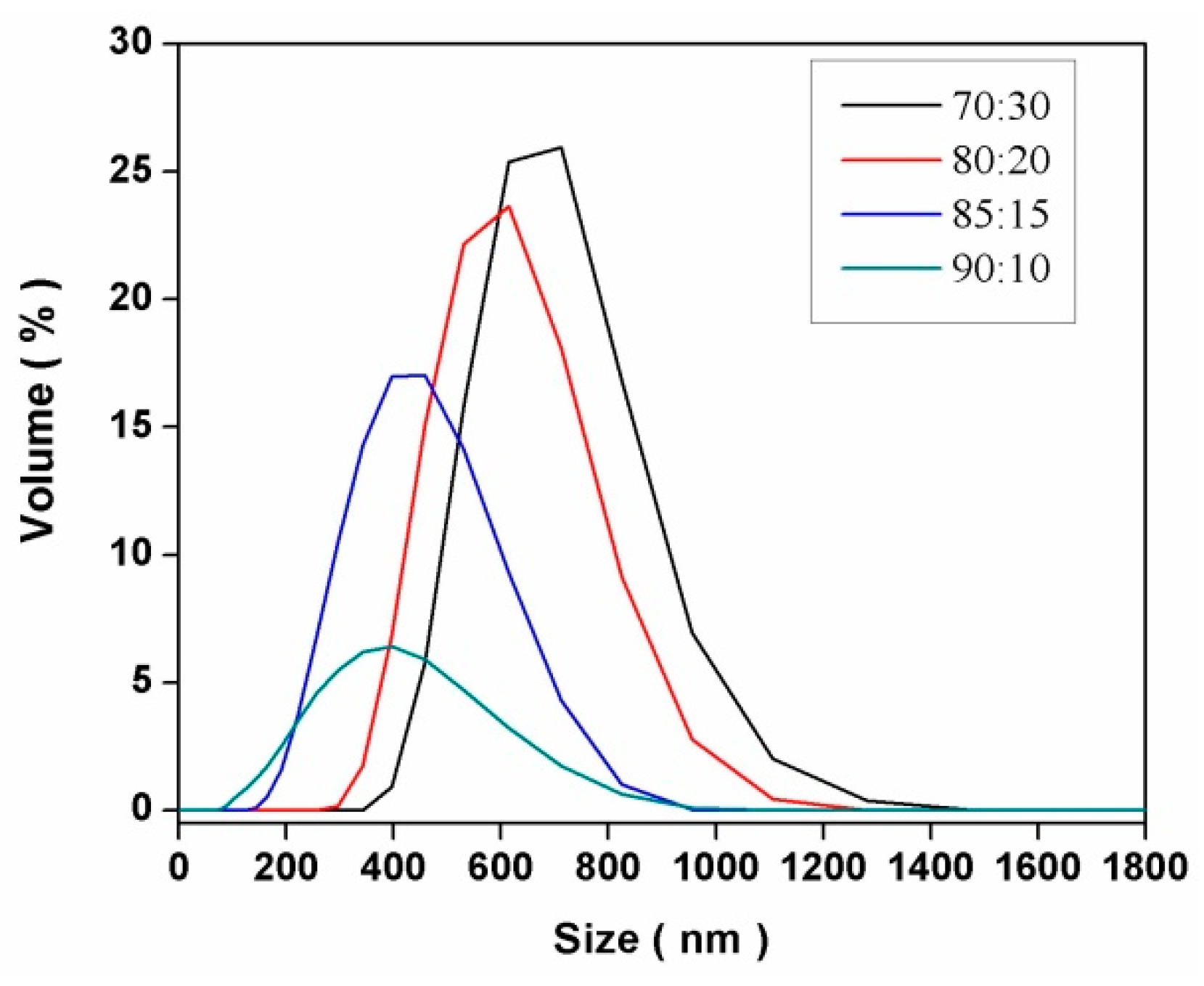
2.1.2. Variation of the Type and Percentage of the Continuous Organic Phase
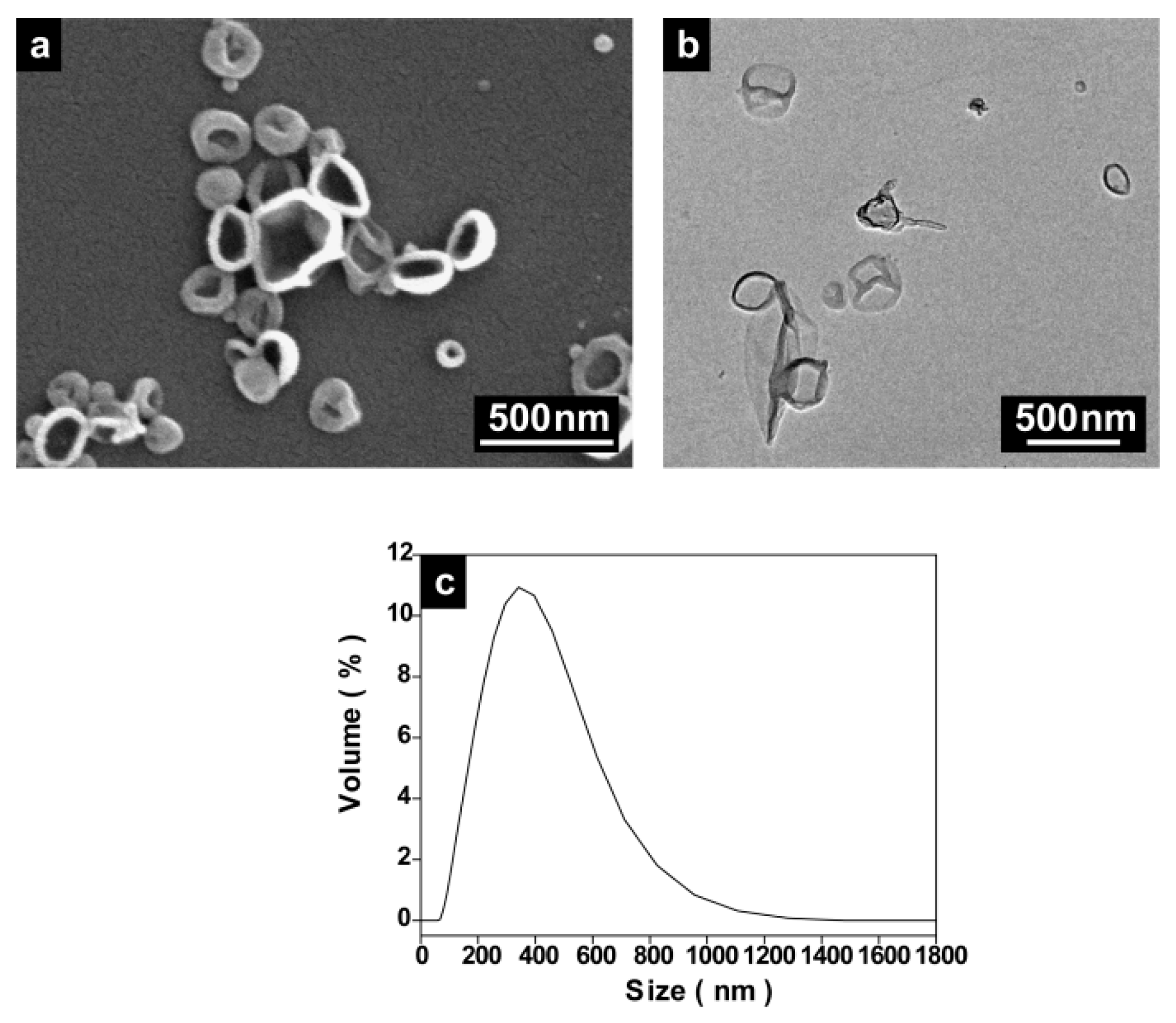
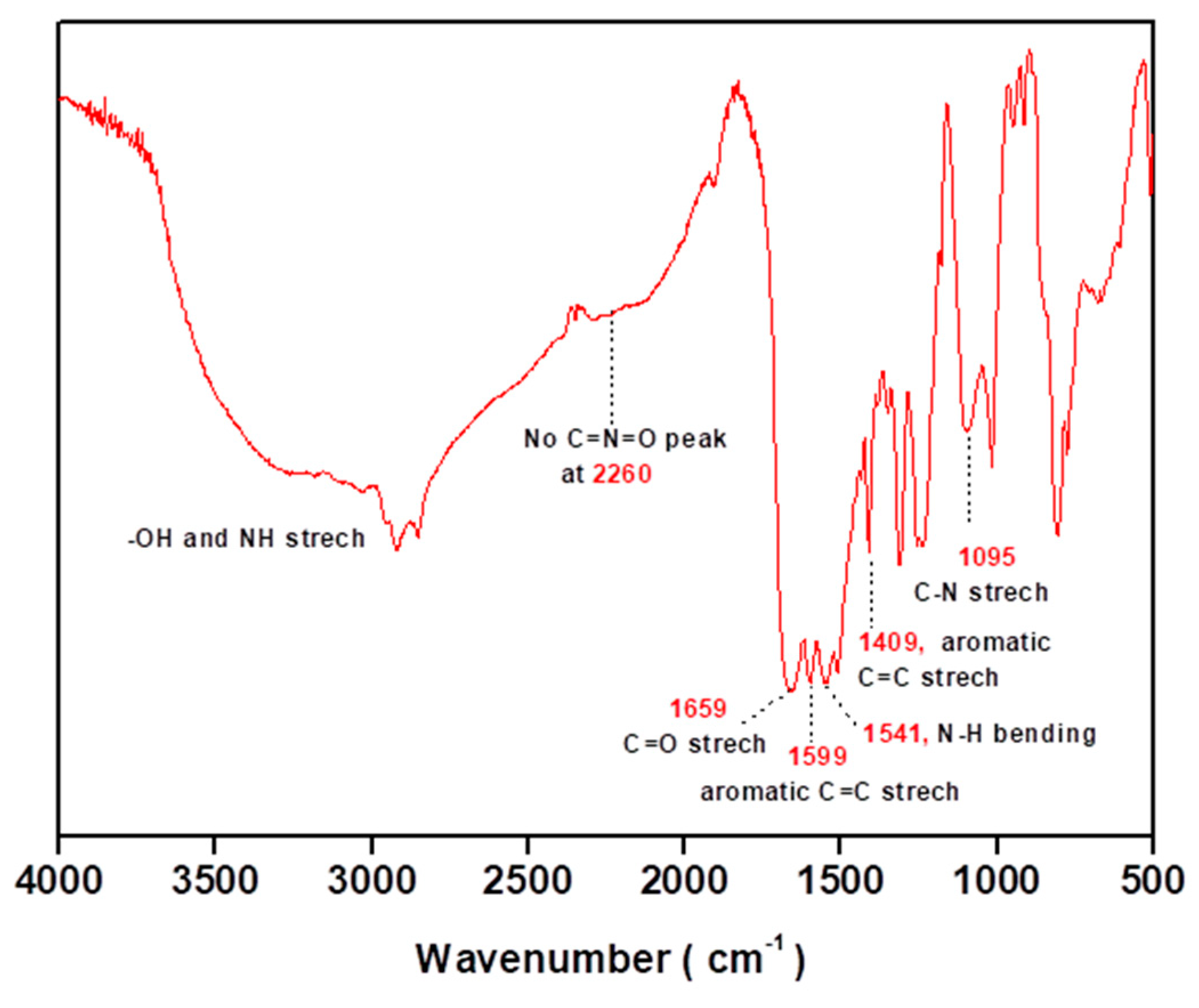
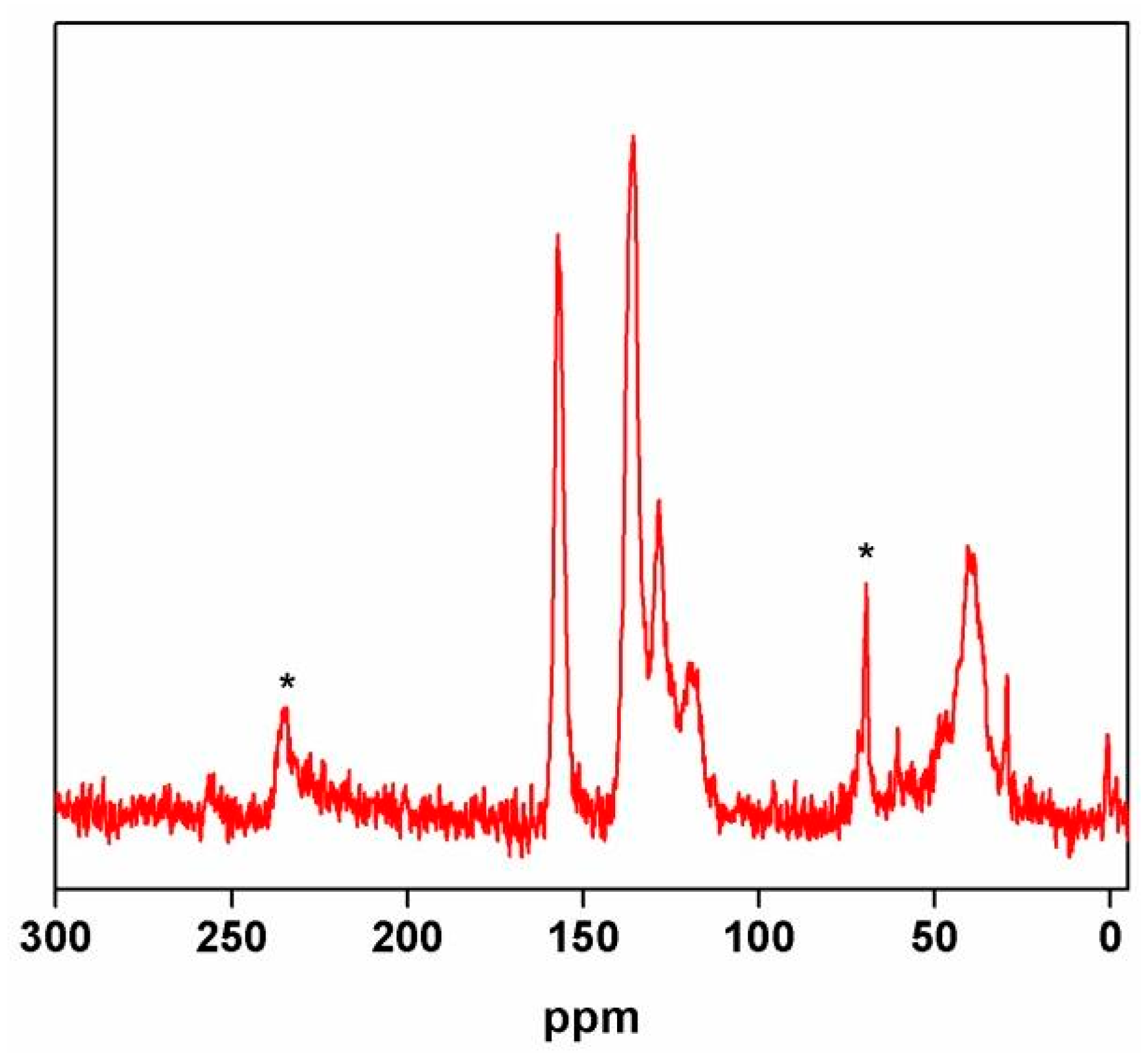
2.1.3. Variation of the Type of the Polyurea (PU) Monomers
2.1.4. The Influence of the Electrolyte
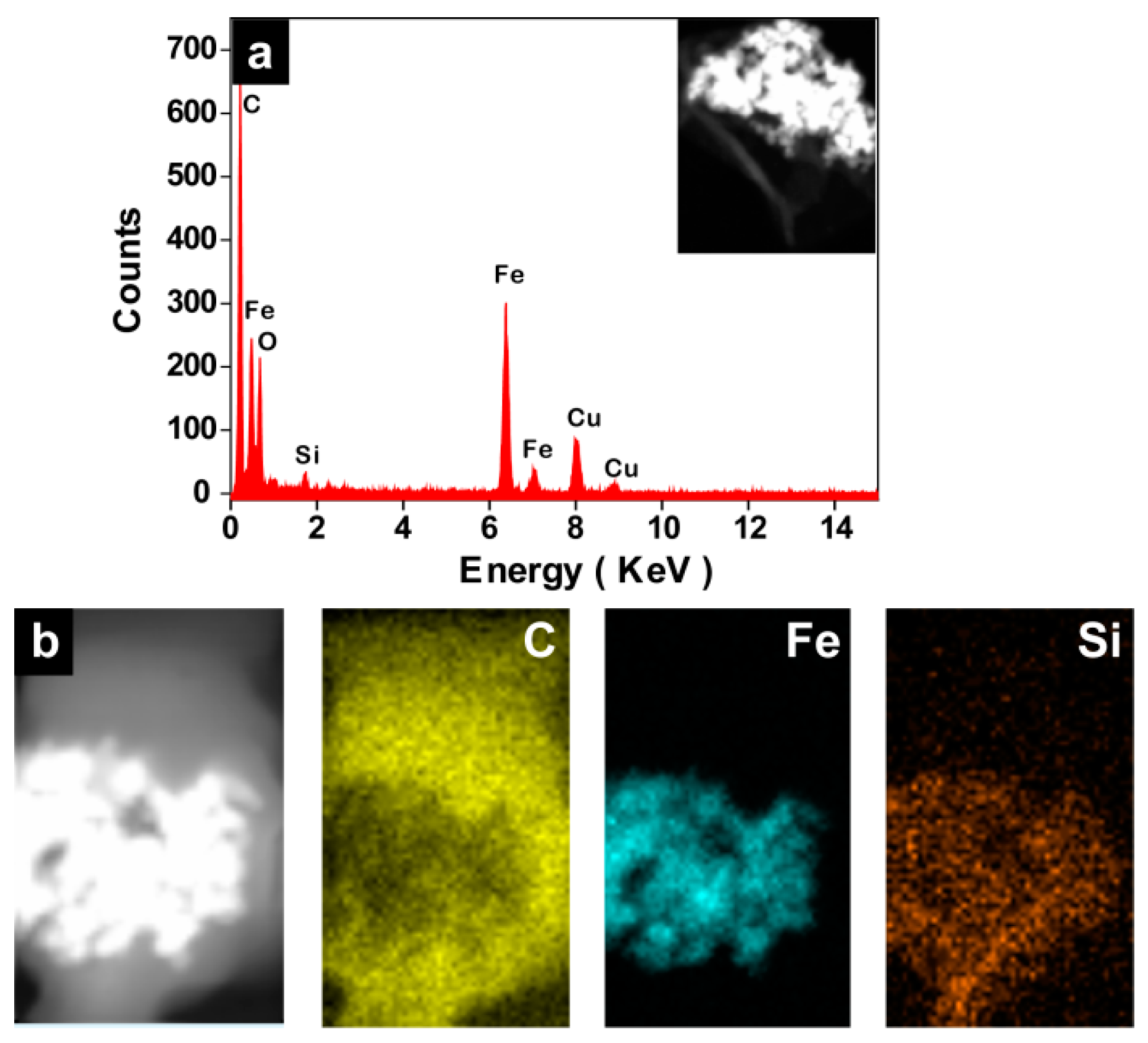
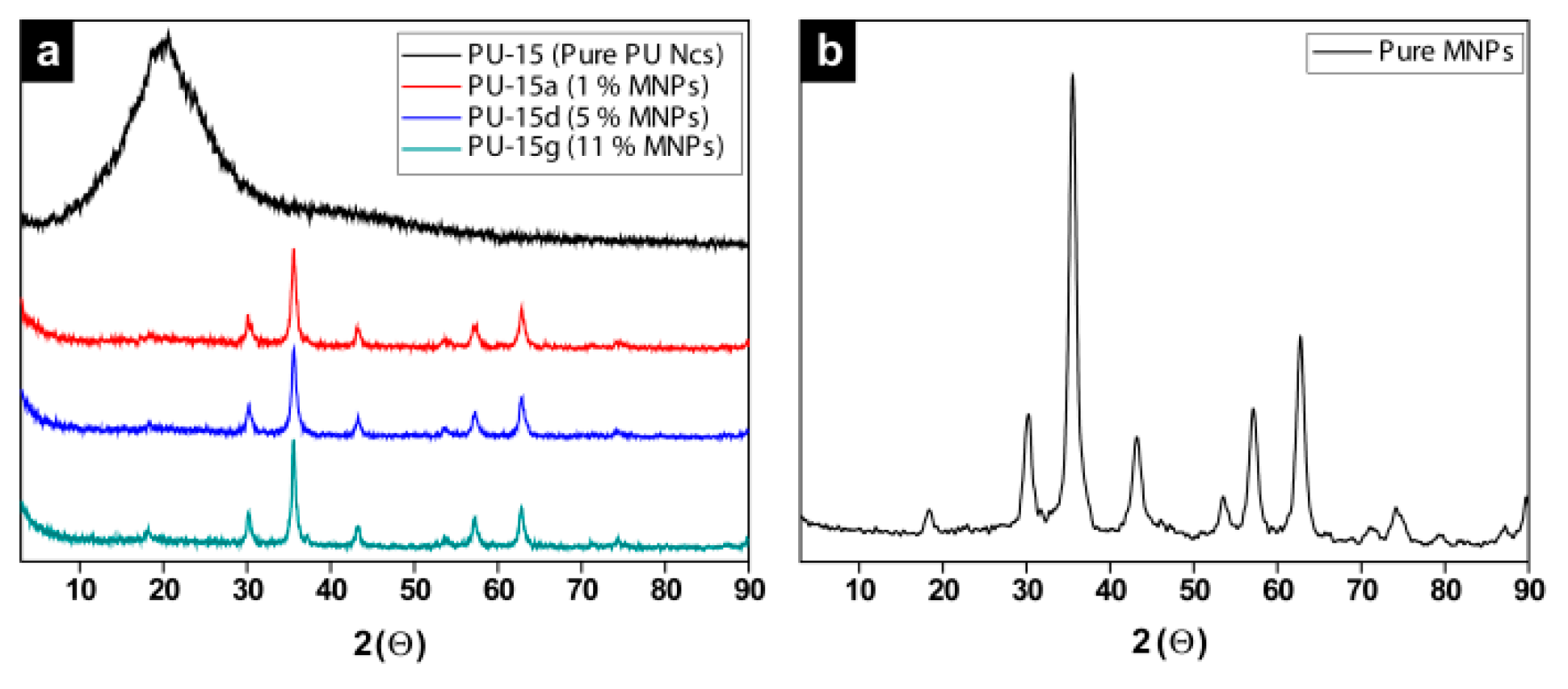
2.2. Encapsulation of Ionic Liquid-Modified Magnetite Nanoparticles (MNPs-IL-C4@PU NCs)
3. Materials and Methods
3.1. Synthesis of 1-Butyl-3-(3-(Trimethoxysilyl)Propyl)-1H-Imidazol-3-Cholride (IL-C4)
3.2. Preparation of Magnetite Nanoparticle-Supported IL-C4 (MNPs-IL-C4)
3.3. Preparation of Polyurea Nano-Capsules (PU NCs)
3.4. Preparation of Magnetic PU NCs (MNPs-IL-C4@PU NCs)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fu, G.-D.; Li, G.L.; Neoh, K.G.; Kang, E.T. Hollow polymeric nanostructures—Synthesis, morphology and function. Prog. Polym. Sci. 2011, 36, 127–167. [Google Scholar] [CrossRef]
- Guterres, S.S.; Alves, M.P.; Pohlmann, A.R. Polymeric nanoparticles, nanospheres and nanocapsules, for cutaneous applications. Drug Target Insights 2007, 2, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Pitaksuteepong, T.; Davies, N.M.; Tucker, I.G.; Rades, T. Factors influencing the entrapment of hydrophilic compounds in nanocapsules prepared by interfacial polymerisation of water-in-oil microemulsions. Eur. J. Pharm. Biopharm. 2002, 53, 335–342. [Google Scholar] [CrossRef]
- Klapper, M.; Nenov, S.; Haschick, R.; Müller, K.; Müllen, K. Oil-in-oil emulsions: A unique tool for the formation of polymer nanoparticles. Acc. Chem. Res. 2008, 41, 1190–1201. [Google Scholar] [CrossRef] [PubMed]
- Tiarks, F.; Landfester, K.; Antonietti, M. Preparation of polymeric nanocapsules by miniemulsion polymerization. Langmuir 2001, 17, 908–918. [Google Scholar] [CrossRef]
- Johnsen, H.; Schmid, R.B. Preparation of polyurethane nanocapsules by miniemulsion polyaddition. J. Microencapsul. 2007, 24, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Mora-Huertas, C.E.; Garrigues, O.; Fessi, H.; Elaissari, A. Nanocapsules prepared via nanoprecipitation and emulsification–diffusion methods: Comparative study. Eur. J. Pharm. Biopharm. 2012, 80, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Landfester, K. Miniemulsions for nanoparticle synthesis. In Colloid Chemistry II; Antonietti, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2003; pp. 75–123. [Google Scholar]
- MacRitchie, F. Mechanism of interfacial polymerization. Trans. Faraday Soc. 1969, 65, 2503–2507. [Google Scholar] [CrossRef]
- Gaudin, F.; Sintes-Zydowicz, N. Core–shell biocompatible polyurethane nanocapsules obtained by interfacial step polymerisation in miniemulsion. Colloids Surf. A 2008, 331, 133–142. [Google Scholar] [CrossRef]
- Capek, I. On inverse miniemulsion polymerization of conventional water-soluble monomers. Adv. Colloid. Interface Sci. 2010, 156, 35–61. [Google Scholar] [CrossRef]
- Cao, Z.; Ziener, U.; Landfester, K. Synthesis of narrowly size-distributed thermosensitive poly(N-isopropylacrylamide) nanocapsules in inverse miniemulsion. Macromolecules 2010, 43, 6353–6360. [Google Scholar] [CrossRef]
- Cao, Z.; Ziener, U. Synthesis of nanostructured materials in inverse miniemulsions and their applications. Nanoscale 2013, 5, 10093–10107. [Google Scholar] [CrossRef] [PubMed]
- Spernath, L.; Magdassi, S. Polyurea nanocapsules obtained from nano-emulsions prepared by the phase inversion temperature method. Polym. Adv. Technol. 2011, 22, 2469–2473. [Google Scholar] [CrossRef]
- Landfester, K.; Willert, M.; Antonietti, M. Preparation of polymer particles in nonaqueous direct and inverse miniemulsions. Macromolecules 2000, 33, 2370–2376. [Google Scholar] [CrossRef]
- Utama, R.H.; Stenzel, M.H.; Zetterlund, P.B. Inverse miniemulsion periphery RAFT polymerization: A convenient route to hollow polymeric nanoparticles with an aqueous core. Macromolecules 2013, 46, 2118–2127. [Google Scholar] [CrossRef]
- Romio, A.P.; Rodrigues, H.H.; Peres, A.; Viegas, A.D.C.; Kobitskaya, E.; Ziener, U.; Landfester, K.; Sayer, C.; Araújo, P.H.H. Encapsulation of magnetic nickel nanoparticles via inverse miniemulsion polymerization. J. Appl. Polym. Sci. 2013, 129, 1426–1433. [Google Scholar] [CrossRef]
- Dsouza, R.; Sriramulu, D.; Valiyaveettil, S. Topology and porosity modulation of polyurea films using interfacial polymerization. RSC Adv. 2016, 6, 24508–24517. [Google Scholar] [CrossRef]
- Rosenbauer, E.-M.; Landfester, K.; Musyanovych, A. Surface-active monomer as a stabilizer for polyurea nanocapsules synthesized via interfacial polyaddition in inverse miniemulsion. Langmuir 2009, 25, 12084–12091. [Google Scholar] [CrossRef] [PubMed]
- Crespy, D.; Stark, M.; Hoffmann-Richter, C.; Ziener, U.; Landfester, K. Polymeric nanoreactors for hydrophilic reagents synthesized by interfacial polycondensation on miniemulsion droplets. Macromolecules 2007, 40, 3122–3135. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef]
- Zhu, Y.; Stubbs, L.P.; Ho, F.; Liu, R.; Ship, C.P.; Maguire, J.A.; Hosmane, N.S. Magnetic nanocomposites: A new perspective in catalysis. ChemCatChem 2010, 2, 365–374. [Google Scholar] [CrossRef]
- Faraji, M.; Yamini, Y.; Rezaee, M. Magnetic nanoparticles: Synthesis, stabilization, functionalization, characterization, and applications. J. Iran. Chem. Soc. 2010, 7, 1–37. [Google Scholar] [CrossRef]
- Baig, R.B.N.; Varma, R.S. Magnetically retrievable catalysts for organic synthesis. Chem. Commun. 2013, 49, 752–770. [Google Scholar] [CrossRef] [PubMed]
- Natour, S.; Abu-Reziq, R. Immobilization of palladium catalyst on magnetically separable polyurea nanosupport. RSC Adv. 2014, 4, 48299–48309. [Google Scholar] [CrossRef]
- Philippova, O.; Barabanova, A.; Molchanov, V.; Khokhlov, A. Magnetic polymer beads: Recent trends and developments in synthetic design and applications. Eur. Polym. J. 2011, 47, 542–559. [Google Scholar] [CrossRef]
- El-Sherif, H.; El-Masry, M.; Emira, H.S. Magnetic polymer composite particles via in situ inverse miniemulsion polymerization process. J. Macromol. Sci. Part A 2010, 47, 1096–1103. [Google Scholar] [CrossRef]
- Park, S.; Lee, Y.; Kim, Y.S.; Lee, H.M.; Kim, J.H.; Cheong, I.W.; Koh, W.-G. Magnetic nanoparticle-embedded PCM nanocapsules based on paraffin core and polyurea shell. Colloids Surf. A 2014, 450, 46–51. [Google Scholar] [CrossRef]
- Kong, L.; Lu, X.; Jin, E.; Jiang, S.; Bian, X.; Zhang, W.; Wang, C. Constructing magnetic polyaniline/metal hybrid nanostructures using polyaniline/Fe3O4 composite hollow spheres as supports. J. Solid State Chem. 2009, 182, 2081–2087. [Google Scholar] [CrossRef]
- Awad, W.H.; Wilkie, C.A. Investigation of the thermal degradation of polyurea: The effect of ammonium polyphosphate and expandable graphite. Polymer 2010, 51, 2277–2285. [Google Scholar] [CrossRef]
- Schlegel, I.; Renz, P.; Simon, J.; Lieberwirth, I.; Pektor, S.; Bausbacher, N.; Miederer, M.; Mailänder, V.; Muñoz-Espí, R.; Crespy, D.; et al. Highly loaded semipermeable nanocapsules for magnetic resonance imaging. Macromol. Biosci. 2018, 18, 1700387. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R167. [Google Scholar] [CrossRef]
- Kalia, S.; Kango, S.; Kumar, A.; Haldorai, Y.; Kumari, B.; Kumar, R. Magnetic polymer nanocomposites for environmental and biomedical applications. Colloid. Polym. Sci. 2014, 292, 2025–2052. [Google Scholar] [CrossRef]
- Alfadhel, A.; Li, B.; Kosel, J. Magnetic polymer nanocomposites for sensing applications. In Proceedings of the Sensors, Valencia, Spain, 2–5 November 2014; pp. 2066–2069. [Google Scholar]
- Srinivasan, S.Y.; Paknikar, K.M.; Gajbhiye, V.; Bodas, D. Magneto-conducting core/shell nanoparticles for biomedical applications. ChemNanoMat 2018, 4, 151–164. [Google Scholar] [CrossRef]
- Perera, A.S.; Zhang, S.; Homer-Vanniasinkam, S.; Coppens, M.-O.; Edirisinghe, M. Polymer–magnetic composite fibers for remote-controlled drug release. ACS Appl. Mat. Interf. 2018, 10, 15524–15531. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Li, S.; Zhu, X.; Jiang, X.; Kong, X.Z. One step preparation of porous polyurea by reaction of toluene diisocyanate with water and its characterization. RSC Adv. 2014, 4, 33520–33529. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compound 1-butyl-3-(3-(trimethoxysilyl)propyl)-1H-imidazol-3-cholride and MNPs-IL-C4@PU NCs are available from the authors. |







| Entry | Amine Monomer | Isocyanate Monomer | MNPs-IL-C4 | Size (nm) b |
|---|---|---|---|---|
| PU-15 | DETA/EDA | 4,4′-MDI | 0% | 365.5 |
| PU-15a | DETA/EDA | 4,4′-MDI | 1% | 233.7 |
| PU-15b | DETA/EDA | 4,4′-MDI | 2% | 216.4 |
| PU-15c | DETA/EDA | 4,4′-MDI | 3% | 221.8 |
| PU-15d | DETA/EDA | 4,4′-MDI | 5% | 195.1 |
| PU-15e | DETA/EDA | 4,4′-MDI | 7% | 198.5 |
| PU-15f | DETA/EDA | 4,4′-MDI | 9% | 246.8 |
| PU-15g | DETA/EDA | 4,4′-MDI | 11% | 238.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Natour, S.; Levi-Zada, A.; Abu-Reziq, R. Magnetic Polyurea Nano-Capsules Synthesized via Interfacial Polymerization in Inverse Nano-Emulsion. Molecules 2019, 24, 2663. https://doi.org/10.3390/molecules24142663
Natour S, Levi-Zada A, Abu-Reziq R. Magnetic Polyurea Nano-Capsules Synthesized via Interfacial Polymerization in Inverse Nano-Emulsion. Molecules. 2019; 24(14):2663. https://doi.org/10.3390/molecules24142663
Chicago/Turabian StyleNatour, Suzana, Anat Levi-Zada, and Raed Abu-Reziq. 2019. "Magnetic Polyurea Nano-Capsules Synthesized via Interfacial Polymerization in Inverse Nano-Emulsion" Molecules 24, no. 14: 2663. https://doi.org/10.3390/molecules24142663
APA StyleNatour, S., Levi-Zada, A., & Abu-Reziq, R. (2019). Magnetic Polyurea Nano-Capsules Synthesized via Interfacial Polymerization in Inverse Nano-Emulsion. Molecules, 24(14), 2663. https://doi.org/10.3390/molecules24142663






