Extraction of Carotenoids and Fat-Soluble Vitamins from Tetradesmus Obliquus Microalgae: An Optimized Approach by Using Supercritical CO2
Abstract
1. Introduction
2. Results and Discussion
2.1. Extraction via Matrix Solid-Phase Dispersion
2.2. Supercritical CO2 (SCCO2) Extraction
2.2.1. Evaluation of the Optimal Extraction Time
2.2.2. Addition of Diatomaceous Earth as a Dispersing Phase
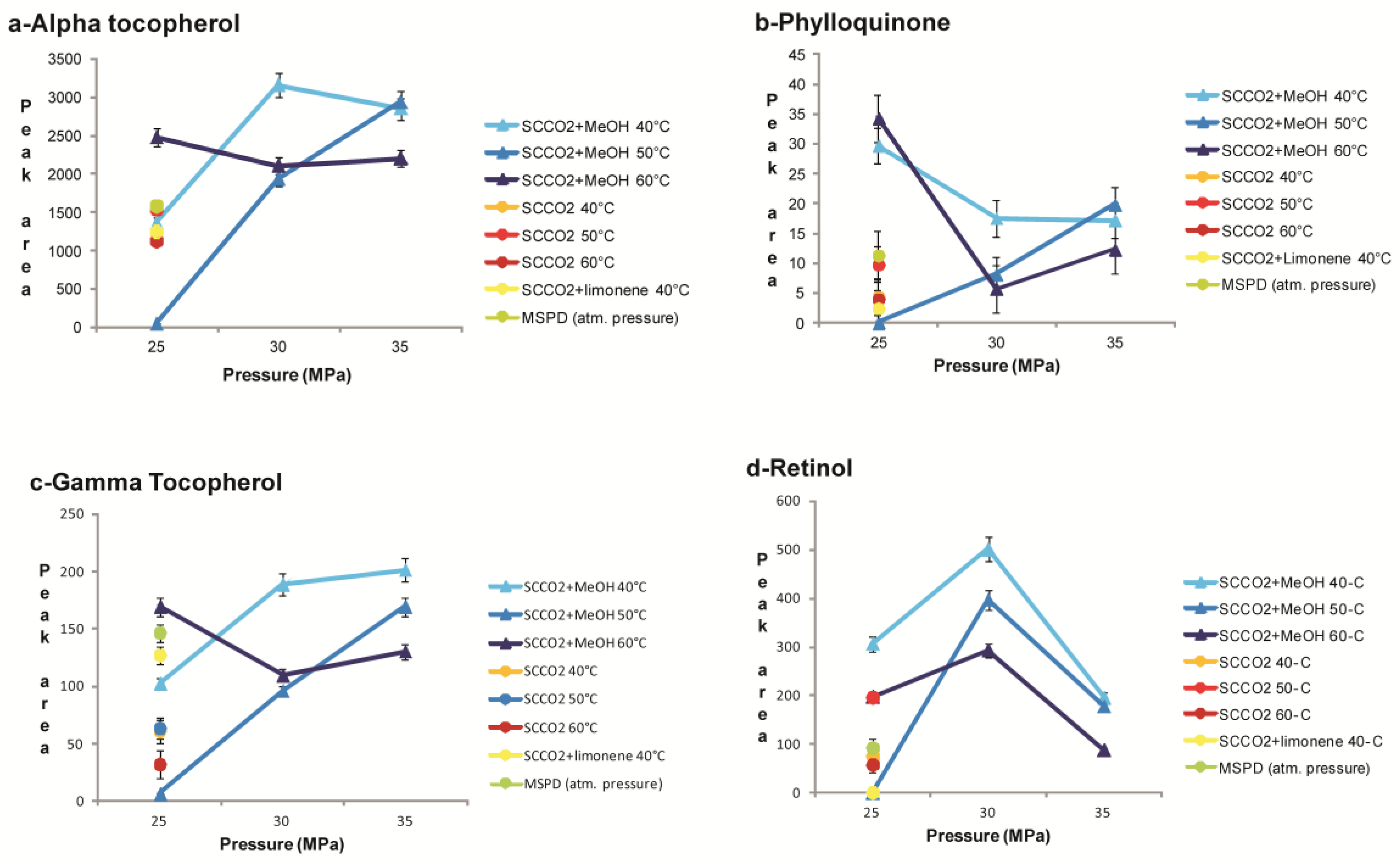
2.3. Evaluation of the Influence of Pressure and Temperature on the Recovery of Carotenoids from T. Obliquus
2.3.1. Extraction Conditions and Extraction Variables
2.3.2. SCCO2 Extraction of Fat-Soluble Vitamins and Carotenoids from T. Obliquus in Comparison with MSPD
3. Materials and Methods
3.1. Biomass Production
3.2. Chemicals and Solvents
3.3. Biomass Pretreatment
3.4. MSPD Extraction
3.5. Supercritical Fluid Extraction
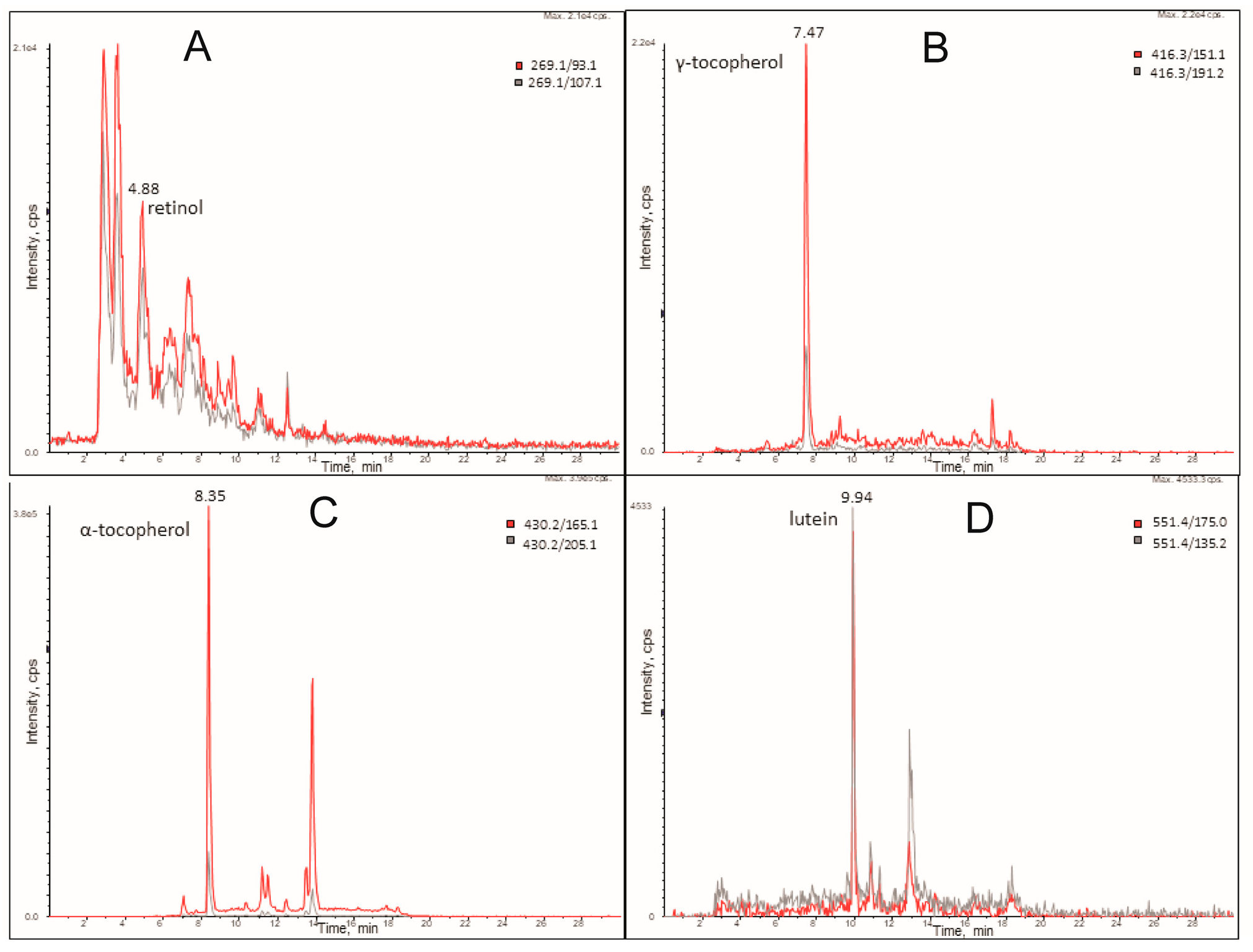
3.6. Mass Spectrometry Experiments
3.7. Liquid Chromatography
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sabu, S.; Bright-Singh, I.; Joseph, V. Molecular identification and comparative evaluation of tropical marine microalgae for biodiesel production. Mar. Biotechnol. 2017, 19, 328–344. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Nojima, D.; Nonoyama, T.; Ikeda, K.; Maeda, Y.; Yoshino, T.; Tanaka, T. Outdoor cultivation of marine diatoms for year-round production of biofuels. Mar. Drugs 2017, 15, 94. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.B.; Adel, M.; Karimi, P.; Peidayesh, M. Pharmaceutical, cosmeceutical, and traditional applications of marine carbohydrates. Adv. Food Nutr. Res. 2014, 73, 197–220. [Google Scholar] [PubMed]
- Nelsestuen, G.L.; Shah, A.M.; Harvey, S.B. Vitamin K-dependent proteins. Vitam. Horm. 2000, 58, 355–389. [Google Scholar] [PubMed]
- Gentili, A.; Miccheli, A.; Tomai, P.; Baldassarre, M.E.; Curini, R.; Pérez-Fernández, V. Liquid chromatography–tandem mass spectrometry method for the determination of vitamin K homologues in human milk after overnight cold saponification. J. Food Compos. Anal. 2016, 47, 21–30. [Google Scholar] [CrossRef]
- Diener, H.C.; Hart, R.G.; Koudstaal, P.J.; Lane, D.A.; Lip, G.Y.H. Atrial fibrillation and cognitive function: Jacc review topic of the week. J. Am. Coll. Cardiol. 2019, 73, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Brangier, A.; Ferland, G.; Rolland, Y.; Gautier, J.; Féart, C.; Annweiler, C. Vitamin K antagonists and cognitive decline in older adults: A 24-month follow-up. Nutrients 2018, 10, 666. [Google Scholar] [CrossRef]
- Vos, M.; Esposito, G.; Edirisinghe, J.N.; Vilain, S.; Haddad, D.M.; Slabbaert, J.R.; Van Meensel, S.; Schaap, O.; De Strooper, B.; Meganathan, R.; et al. Vitamin K2 is a mitochondrial electron carrier that rescues pink1 deficiency. Science 2012, 336, 1306–1310. [Google Scholar] [CrossRef] [PubMed]
- Gentili, A.; Cafolla, A.; Gasperi, T.; Bellante, S.; Caretti, F.; Curini, R.; Pérez-Fernández, V. Rapid, high performance method for the determination of vitamin K1, menaquinone-4 and vitamin K1 2, 3-epoxide in human serum and plasma using liquid chromatography-hybrid quadrupole linear ion trap mass spectrometry. J. Chromatogr. A 2014, 1338, 102–110. [Google Scholar] [CrossRef]
- Roeck-Holtzhauer, Y.D.; Quere, I.; Claire, C. Vitamin analysis of five planktonic microalgae and one macroalga. J. Appl. Phycol. 1991, 3, 259–264. [Google Scholar] [CrossRef]
- Tarento, T.D.C.; McClure, D.D.; Vasiljevski, E.; Schindeler, A.; Dehghani, F.; Kavanagh, J.M. Microalgae as a source of vitamin K1. Algal Res. 2018, 36, 77–87. [Google Scholar] [CrossRef]
- Flores-Hidalgo, M.; Torres-Rivas, F.; Monzon-Bensojo, J.; Escobedo-Bretado, M.; Glossman-Mitnik, D.; Barraza-Jimenez, D. Electronic Structure of Carotenoids in Natural and Artificial Photosynthesis. In Carotenoids; Cvetkovic, D.J., Nikolic, G.S., Eds.; IntechOpen: London, UK, 2017; pp. 17–33. [Google Scholar] [CrossRef]
- Mein, J.R.; Lian, F.; Wang, X.D. Biological activity of lycopene metabolites: Implications for cancer prevention. Nutr. Rev. 2008, 66, 667–683. [Google Scholar] [CrossRef] [PubMed]
- Sathasivam, R.; Ki, J.S. A Review of the Biological Activities of Microalgal Carotenoids and Their Potential Use in Healthcare and Cosmetic Industries. Mar. Drugs 2018, 16, 26. [Google Scholar] [CrossRef] [PubMed]
- Higuera-Ciapara, I.; Félix-Valenzuela, L.; Goycoolea, F.M. Astaxanthin: A review of its chemistry and applications. Crit. Rev. Food Sci. Nutr. 2006, 46, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Gordon, H.T.; Bauernfeind, J.C. Carotenoids as food colorants. Crit. Rev. Food Sci. Nutr. 1982, 18, 59–97. [Google Scholar] [CrossRef] [PubMed]
- Macías-Sánchez, M.D.; Mantell Serrano, C.; Rodríguez Rodríguez, M.; Martínez de la Ossa, E.; Lubián, L.M.; Montero, O. Extraction of carotenoids and chlorophyll from microalgae with supercritical carbon dioxide and ethanol as cosolvent. J. Sep. Sci. 2008, 31, 1352–1362. [Google Scholar] [CrossRef] [PubMed]
- Di Caprio, F.; Altimari, P.; Iaquaniello, G.; Toro, L.; Pagnanelli, F. Heterotrophic cultivation of T. obliquus under non-axenic conditions by uncoupled supply of nitrogen and glucose. Biochem. Eng. J. 2019, 145, 127–136. [Google Scholar] [CrossRef]
- Ambati, R.R.; Gogisetty, D.; Aswathanarayana, R.G.; Ravi, S.; Bikkina, P.N.; Bo, L.; Yuepeng, S. Industrial potential of carotenoid pigments from microalgae: Current trends and future prospects. Crit. Rev. Food Sci. 2019, 59, 1880–1902. [Google Scholar] [CrossRef]
- Wu, Y.H.; Hu, H.Y.; Yu, Y.; Zhang, T.Y.; Zhu, S.F.; Zhuang, L.L.; Zhang, X.; Lu, Y. Microalgal species for sustainable biomass/lipid production using wastewater as resource: A review. Renew. Sust. Energ. Rev. 2014, 33, 675–688. [Google Scholar] [CrossRef]
- Breuer, G.; Lamers, P.P.; Martens, D.E.; Draaisma, R.B.; Wijffels, R.H. Effect of light intensity, pH, and temperature on triacylglycerol (TAG) accumulation induced by nitrogen starvation in Scenedesmus obliquus. Bioresour. Technol. 2013, 143, 1–9. [Google Scholar] [CrossRef]
- Amaro, H.M.; Fernandes, F.; Valentão, P.; Andrade, P.B.; Sousa-Pinto, I.; Malcata, F.X.; Guedes, A.C. Effect of Solvent System on Extractability of Lipidic Components of Scenedesmusobliquus (M2-1) and Gloeothece sp. on Antioxidant Scavenging Capacity Thereof. Mar. Drugs. 2015, 13, 6453–6471. [Google Scholar] [CrossRef] [PubMed]
- Postma, P.R.; ‘t Lam, G.P.; Barbosa, M.J.; Wijffels, R.H.; Eppink, M.H.M.; Olivieri, G. Microalgal Biorefinery for Bulk and High-Value Products: Product Extraction Within Cell Disintegration. In Handbook of Electroporation; Miklavcic, D., Ed.; Springer International Publishing: Berlin, Germany, 2016; pp. 1–20. [Google Scholar] [CrossRef]
- De Andrade Lima, M.; Kestekoglou, I.; Charalampopoulos, D.; Chatzifragkou, A. Supercritical Fluid Extraction of Carotenoids from Vegetable Waste Matrices. Molecules 2019, 24, 466. [Google Scholar] [CrossRef] [PubMed]
- Mo, Z.Z.; Lin, Z.X.; Su, Z.R.; Zheng, L.; Li, H.L.; Xie, J.H.; Xian, Y.F.; Yi, T.G.; Huang, S.Q.; Chen, J.P. Angelica sinensis Supercritical Fluid CO2 Extract Attenuates D-Galactose-Induced Liver and Kidney Impairment in Mice by Suppressing Oxidative Stress and Inflammation. J. Med. Food 2018, 21, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Chronopoulou, L.; Agatone, A.; Palocci, C. Supercritical CO2 extraction of oleanolic acid from grape pomace. Int. J. Food Sci. Tech. 2013, 48, 1854–1860. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Di Sanzo, G.; Martino, M.; Casella, P.; Marino, T.; Karatza, D.; Musmarra, D. Extraction of Bioactive Compounds Using Supercritical Carbon Dioxide. Molecules 2019, 24, 782. [Google Scholar] [CrossRef] [PubMed]
- Tyśkiewicz, K.; Dębczak, A.; Gieysztor, R.; Szymczak, T.; Rój, E. Determination of fat- and water-soluble vitamins by supercritical fluid chromatography: A review. J. Sep. Sci. 2018, 41, 336–350. [Google Scholar] [CrossRef] [PubMed]
- Gallego, R.; Bueno, M.; Herrero, M. Sub- and supercritical fluid extraction of bioactive compounds from plants, food-by-products, seaweeds and microalgae: An update. TrAC, Trends Anal. Chem. 2019, 116, 198–213. [Google Scholar] [CrossRef]
- Gentili, A.; Dal Bosco, C.; Fanali, S.; Fanali, C. Large-scale profiling of carotenoids by using non aqueous reversed phase liquid chromatography—Photodiode array detection—Triple quadrupole linear ion trap mass spectrometry: Application to some varieties of sweet pepper (Capsicum annuum L.). J. Pharm. Biomed. Anal. 2019, 164, 759–767. [Google Scholar] [CrossRef]
- Gentili, A.; Caretti, F.; Ventura, S.; Pérez-Fernández, V.; Venditti, A.; Curini, R. Screening of carotenoids in tomato fruits by using liquid chromatography with diode array−linear ion trap mass spectrometry detection. J. Agric. Food Chem. 2015, 63, 7428–7439. [Google Scholar] [CrossRef]
- Mudimu, O.; Koopmann, I.K.; Rybalka, N.; Friedl, T.; Schulz, R.; Bilger, W. Screening of microalgae and cyanobacteria strains for α-tocopherol content at different growth phases and the influence of nitrate reduction on α-tocopherol production. J. Appl. Phycol. 2017, 29, 2867–2875. [Google Scholar] [CrossRef]
- Schwender, J.; Seemann, M.; Lichtenthaler, H.K.; Rohmer, M. Biosynthesis of isoprenoids (carotenoids, sterols, prenyl side-chains of chlorophylls and plastoquinone) via a novel pyruvate/glyceraldehyde 3-phosphate non-mevalonate pathway in the green alga Scenedesmus obliquus. Biochem. J. 1996, 316, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Lian, J.; Wijffels, R.H.; Smidt, H.; Sipkema, D. The effect of the algal microbiome on industrial production of microalgae. Microb. Biotechnol. 2018, 11, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Croft, M.T.; Lawrence, A.D.; Raux-Deery, E.; Warren, M.J.; Smith, A.G. Algae acquire vitamin B12 through a symbiotic relationship with bacteria. Nature 2005, 438, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Pour, H.S.R.; Tavakoli, O.; Sarrafzadeh, M.H. Experimental optimization of SC-CO2 extraction of carotenoids from Dunaliella salina. J. Supercrit. Fluids 2017, 121, 89–95. [Google Scholar] [CrossRef]
- Di Caprio, F.; Scarponi, P.; Altimari, P.; Iaquaniello, G.; Pagnanelli, F. The influence of phenols extracted from olive mill wastewater on the heterotrophic and mixotrophic growth of Scenedesmus sp. J. Chem. Technol. Biotechnol. 2018, 93, 3619–3626. [Google Scholar] [CrossRef]
- Wynne, M.J.; Hallan, J.K. Reinstatement of Tetradesmus, G.M. Smith (Sphaeropleales, Chlorophyta). Feddes Repertorium 2016, 126, 83–86. [Google Scholar] [CrossRef]
- Di Caprio, F.; Altimari, P.; Pagnanelli, F. Integrated microalgae biomass production and olive mill wastewater biodegradation: Optimization of the wastewater supply strategy. Chem. Eng. J. 2018, 349, 539–546. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


| Compound | SCCO2 25 MPa | SCCO2 + limonene | SCCO2 + MeOH 25 MPa | SCCO2 + MeOH 30 MPa | SCCO2 + MeOH 35 MPa | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 40 °C | 50 °C | 60 °C | 40 °C | 50 °C | 60 °C | 40 °C | 50 °C | 60 °C | 40 °C | 50 °C | 60 °C | ||
| Alpha-tocopherol | (77.71 ± 4.95)% | (97.3 ± 3.59)% | (71.12 ± 3.91)% | (78.73 ± 3.28)% | (86.25 ± 5.16)% | (2.88 ± 2.76)% | (156.95 ± 5.31)% | (200.15 ± 4.64)% | (122.39 ± 4.61)% | (133.44 ± 5.45)% | (180.73 ± 5.33)% | (185.79 ± 3.78)% | (139.39 ± 3.74)% |
| Phylloquinone | (38.83 ± 4.61)% | (86.50 ± 4.31)% | (34.77 ± 4.31)% | (21.45 ± 4.15)% | (262.14 ± 5.16)% | 0 | (302.74 ± 5.89)% | (154.46 ± 5.72)% | (71.67 ± 6.11)% | (50.31 ± 6.73)% | (151.81 ± 5.63)% | (174.76 ± 6.28)% | (108.56 ± 6.45)% |
| Gamma-tocopherol | (41.42 ± 3.76)% | (43.26 ± 3.73)% | (21.77 ± 5.71)% | (86.73 ± 3.82)% | (69.74 ± 3.16)% | (4.09 ± 3.16)% | (115.46 ± 3.81)% | (128.90 ± 3.45)% | (65.30 ± 3.18)% | (74.72 ± 3.62)% | (137.43 ± 3.92)% | (115.52 ± 3.75)% | (88.98 ± 3.49)% |
| Retinol | (81.44 ± 3.18)% | (212.56 ± 3.47)% | (62.03 ± 3.94)% | 0 | (332.72 ± 2.58)% | 0 | (213.75 ± 5.76)% | (543.00 ± 5.73)% | (430.97 ± 8.56)% | (317.32 ± 4.61)% | (213.00 ± 3.94)% | (192.93 ± 6.31)% | (94.78 ± 4.72)% |
| Canthaxanthin | 0 | 0 | 0 | (8.09 ± 2.98)% | (11.02 ± 3.11)% | (0.56 ± 0.44)% | (9.16 ± 3.26)% | (12.99 ± 3.45)% | (4.99 ± 2.31)% | (12.99 ± 4.23)% | (9.25 ± 3.62)% | (16.14 ± 3.15)% | (9.25 ± 3.52)% |
| Phytofluene | (18.24 ± 5.64)% | (23.50 ± 4.25)% | (15.21 ± 2.58)% | (142.65 ± 7.51)% | (36.43 ± 4.39)% | (94.04 ± 3.98)% | (56.80 ± 4.58)% | (3.34 ± 2.13)% | (23.50 ± 8.57)% | (58.92 ± 8.63)% | (39.46 ± 8.91)% | (8.44 ± 7.32)% | (41.13 ± 8.69)% |
| Lutein | 0 | 0 | 0 | 0 | (0.74 ± 0.53)% | 0 | (0.70 ± 0.45)% | (1.03 ± 0.74)% | (0.29 ± 0.25)% | (0.22 ± 0.20)% | (0.67 ± 0.64)% | (1.25 ± 0.50)% | (0.40 ± 0.40)% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chronopoulou, L.; Dal Bosco, C.; Di Caprio, F.; Prosini, L.; Gentili, A.; Pagnanelli, F.; Palocci, C. Extraction of Carotenoids and Fat-Soluble Vitamins from Tetradesmus Obliquus Microalgae: An Optimized Approach by Using Supercritical CO2. Molecules 2019, 24, 2581. https://doi.org/10.3390/molecules24142581
Chronopoulou L, Dal Bosco C, Di Caprio F, Prosini L, Gentili A, Pagnanelli F, Palocci C. Extraction of Carotenoids and Fat-Soluble Vitamins from Tetradesmus Obliquus Microalgae: An Optimized Approach by Using Supercritical CO2. Molecules. 2019; 24(14):2581. https://doi.org/10.3390/molecules24142581
Chicago/Turabian StyleChronopoulou, Laura, Chiara Dal Bosco, Fabrizio Di Caprio, Letizia Prosini, Alessandra Gentili, Francesca Pagnanelli, and Cleofe Palocci. 2019. "Extraction of Carotenoids and Fat-Soluble Vitamins from Tetradesmus Obliquus Microalgae: An Optimized Approach by Using Supercritical CO2" Molecules 24, no. 14: 2581. https://doi.org/10.3390/molecules24142581
APA StyleChronopoulou, L., Dal Bosco, C., Di Caprio, F., Prosini, L., Gentili, A., Pagnanelli, F., & Palocci, C. (2019). Extraction of Carotenoids and Fat-Soluble Vitamins from Tetradesmus Obliquus Microalgae: An Optimized Approach by Using Supercritical CO2. Molecules, 24(14), 2581. https://doi.org/10.3390/molecules24142581











