The Chemistry and Biology of Cyclophostin, the Cyclipostins and Related Compounds
Abstract
1. Introduction
2. Discussion
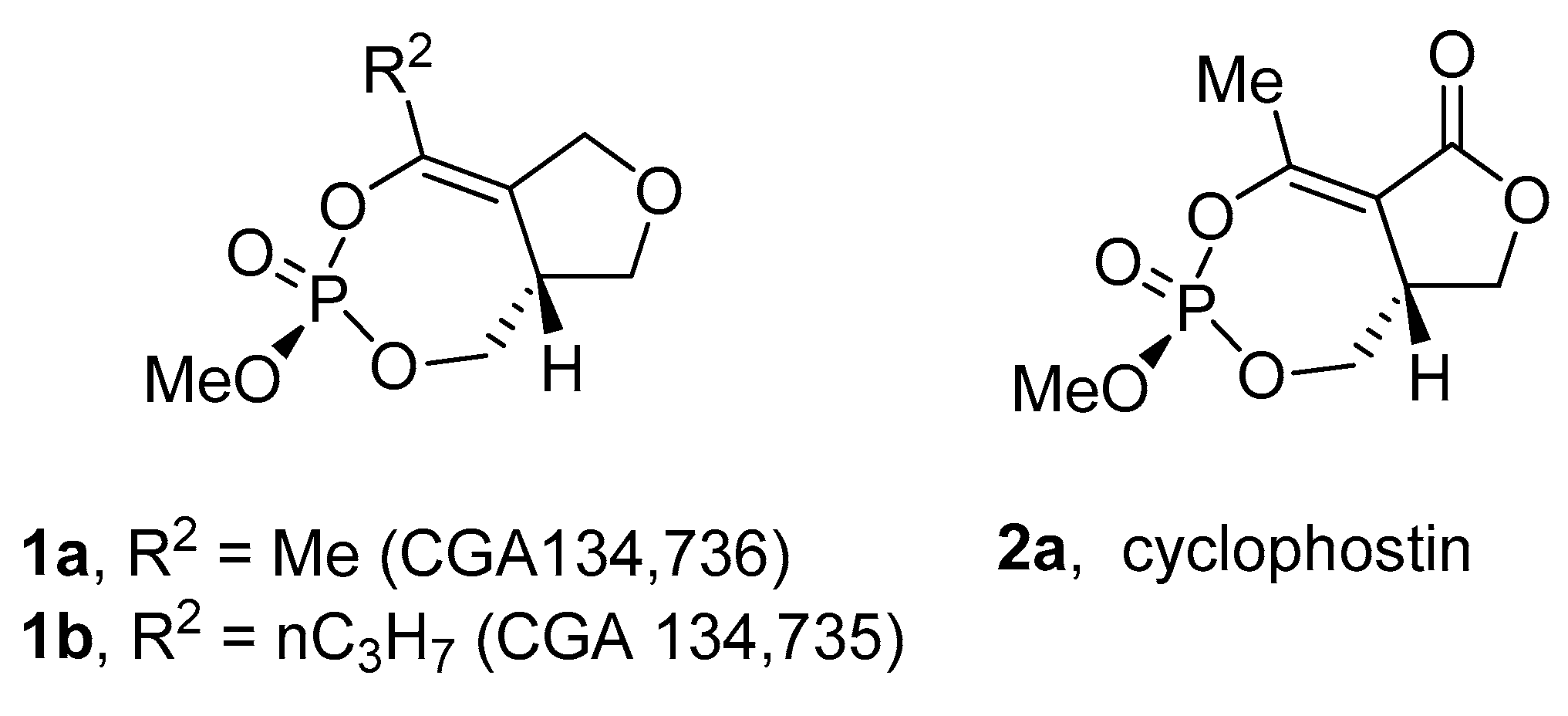
2.1. Isolation and Structure of Cyclophostin
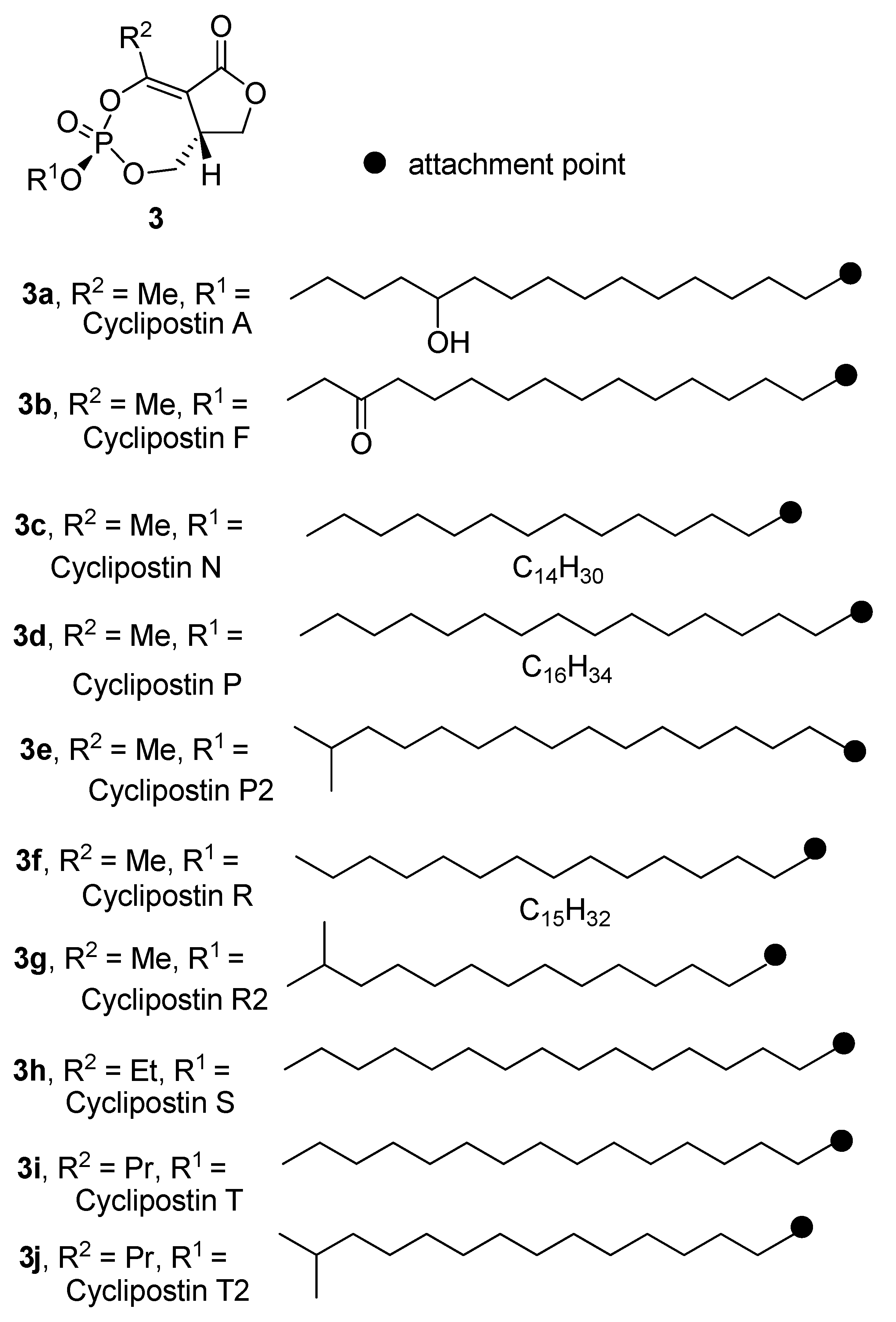
2.2. Isolation of the Cyclipostins
2.3. Isolation of the Salinipostins
2.4. Stereochemical Nomenclature
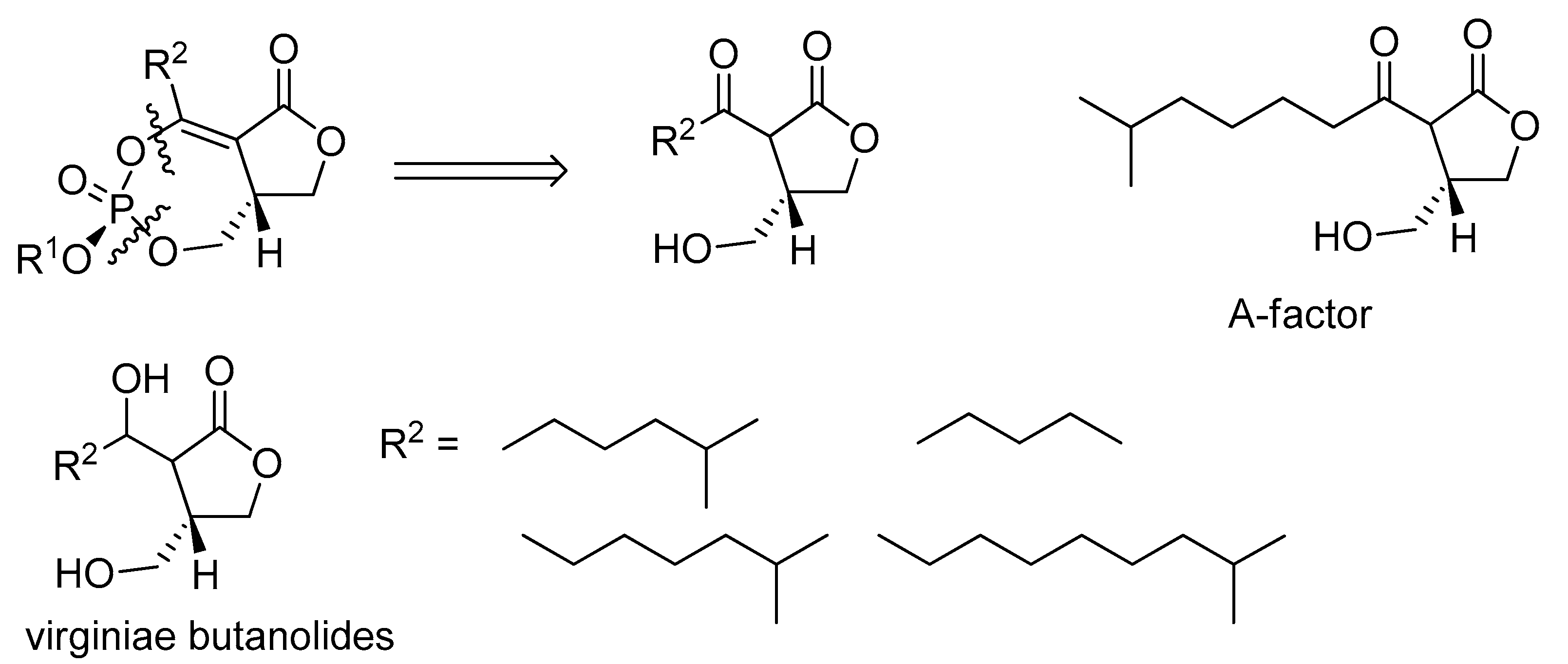
2.5. Biosynthesis
2.6. Synthesis of (±) Cyclophostin and (±) Cyclipostin P
2.7. Synthesis of the Salinipostins
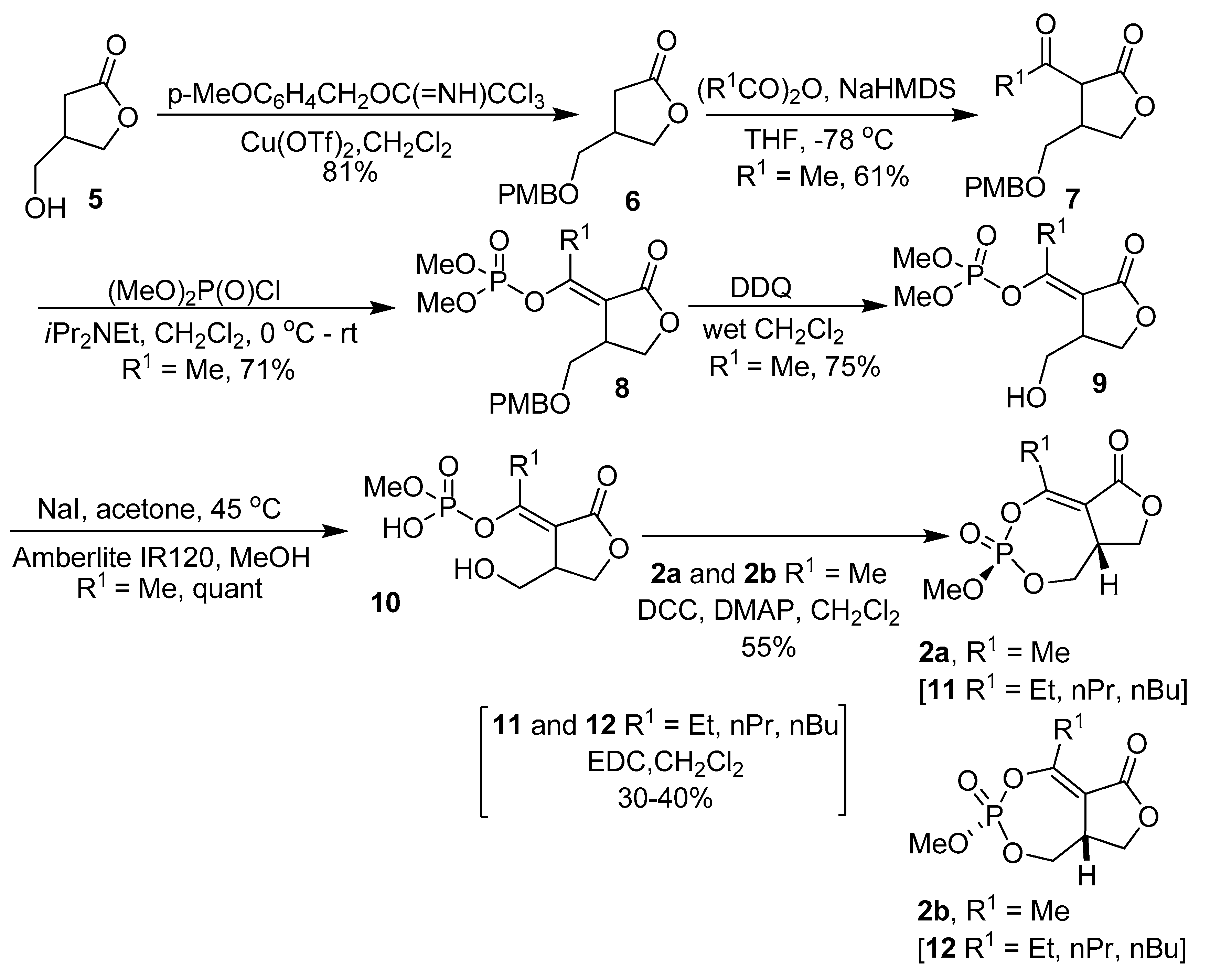
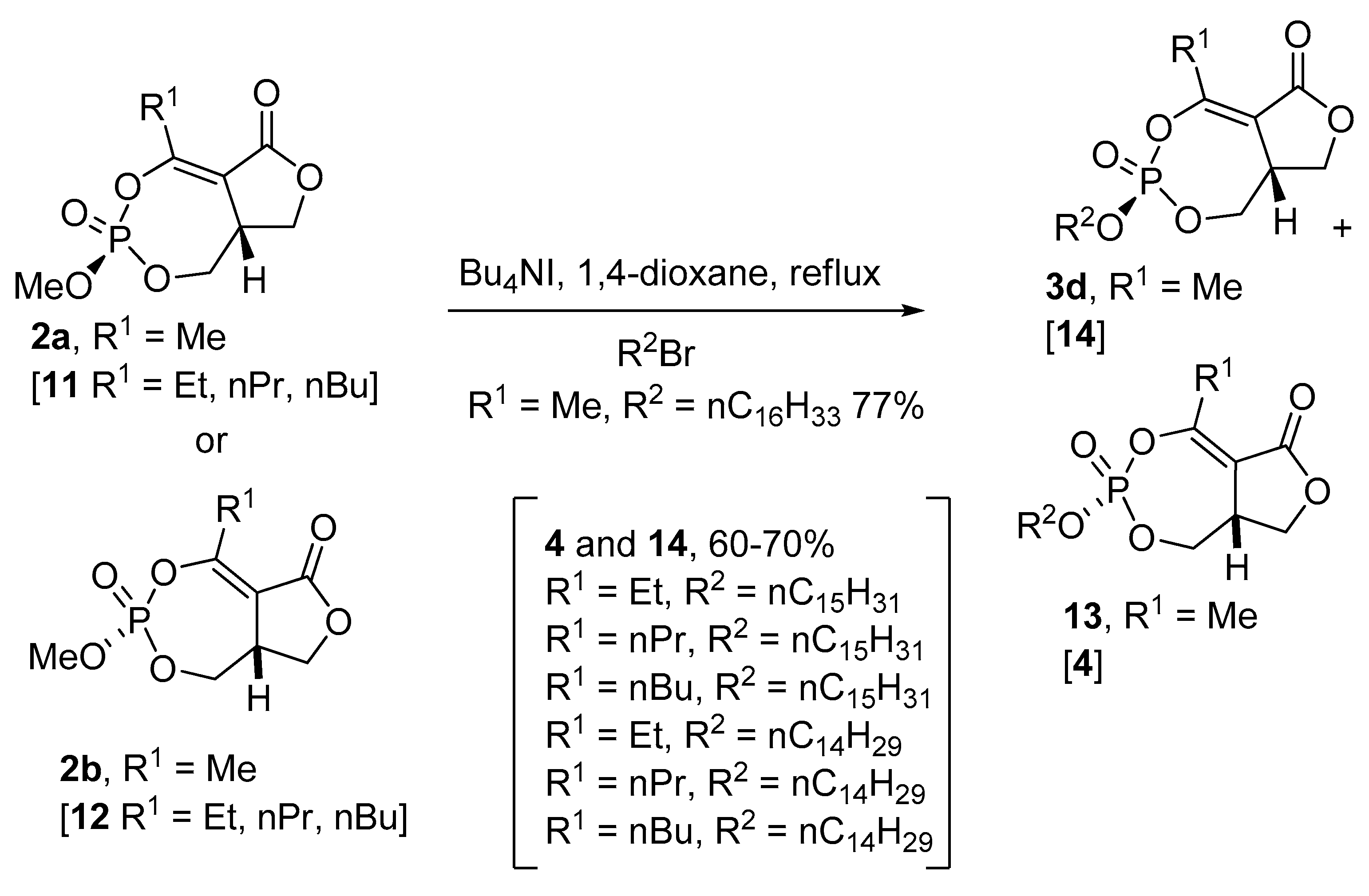
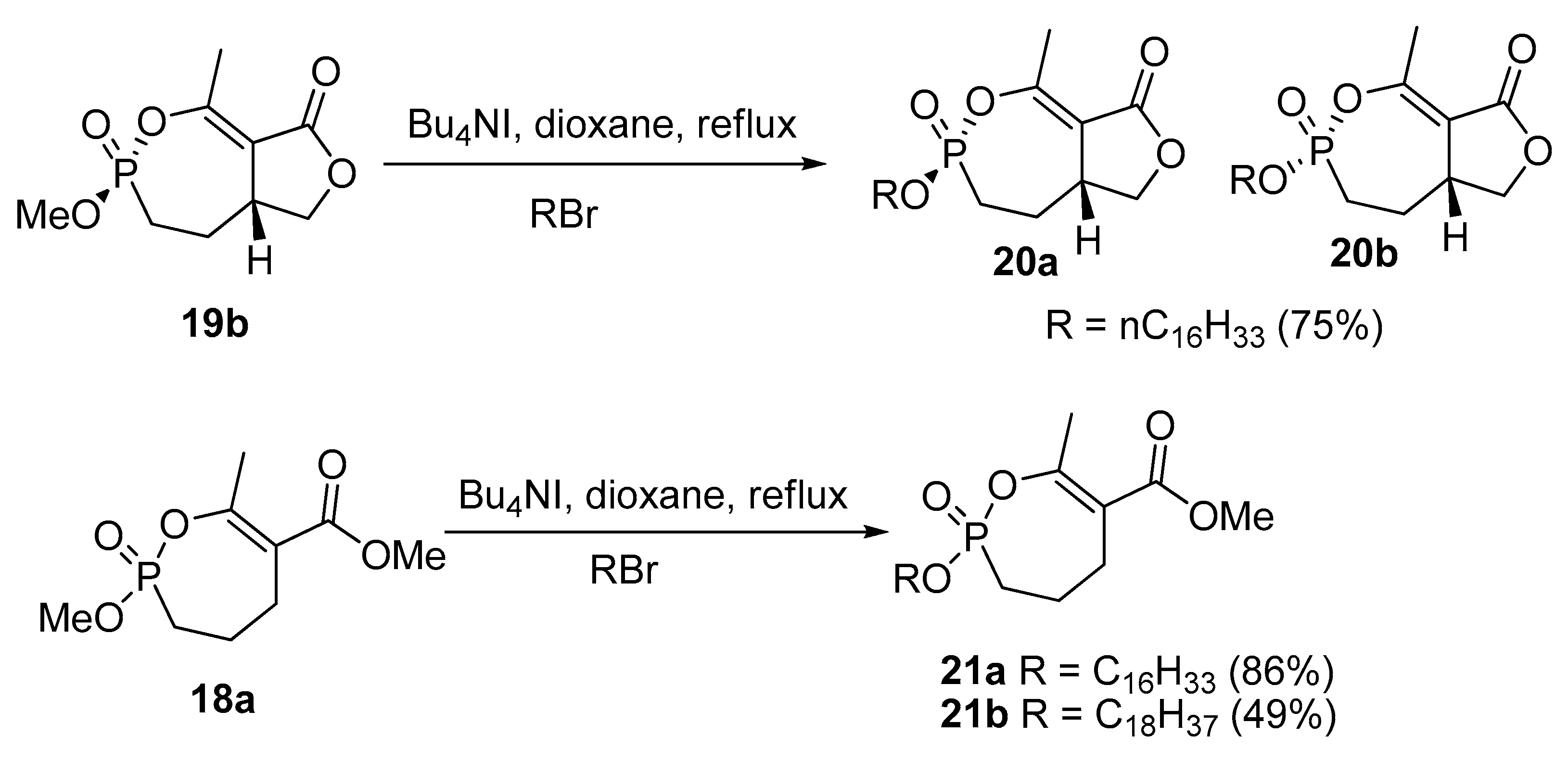
2.8. Synthesis of Mono and Bicyclic Phosphonate Analogs
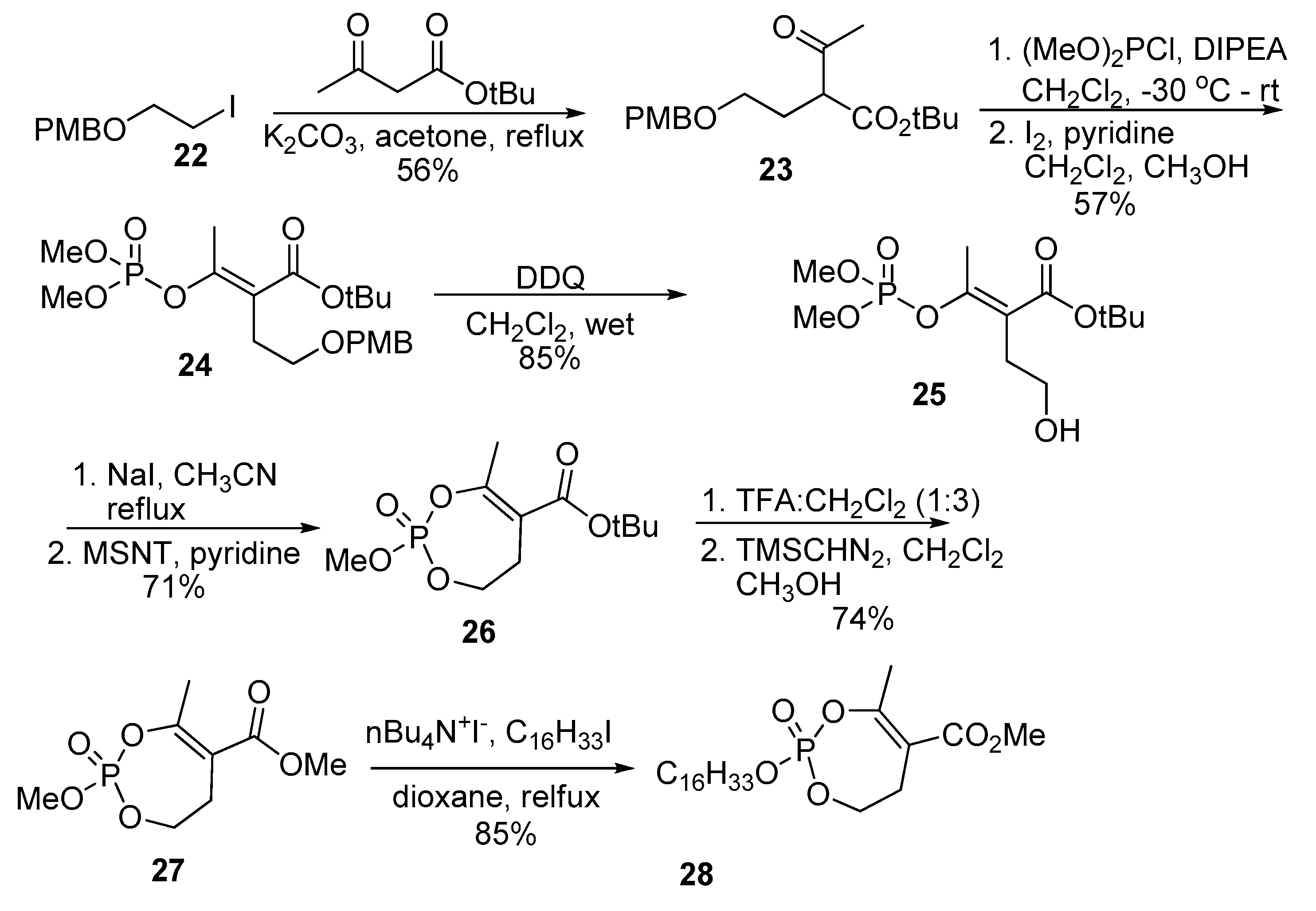
2.9. Synthesis of Monocyclic Phosphate Analogs
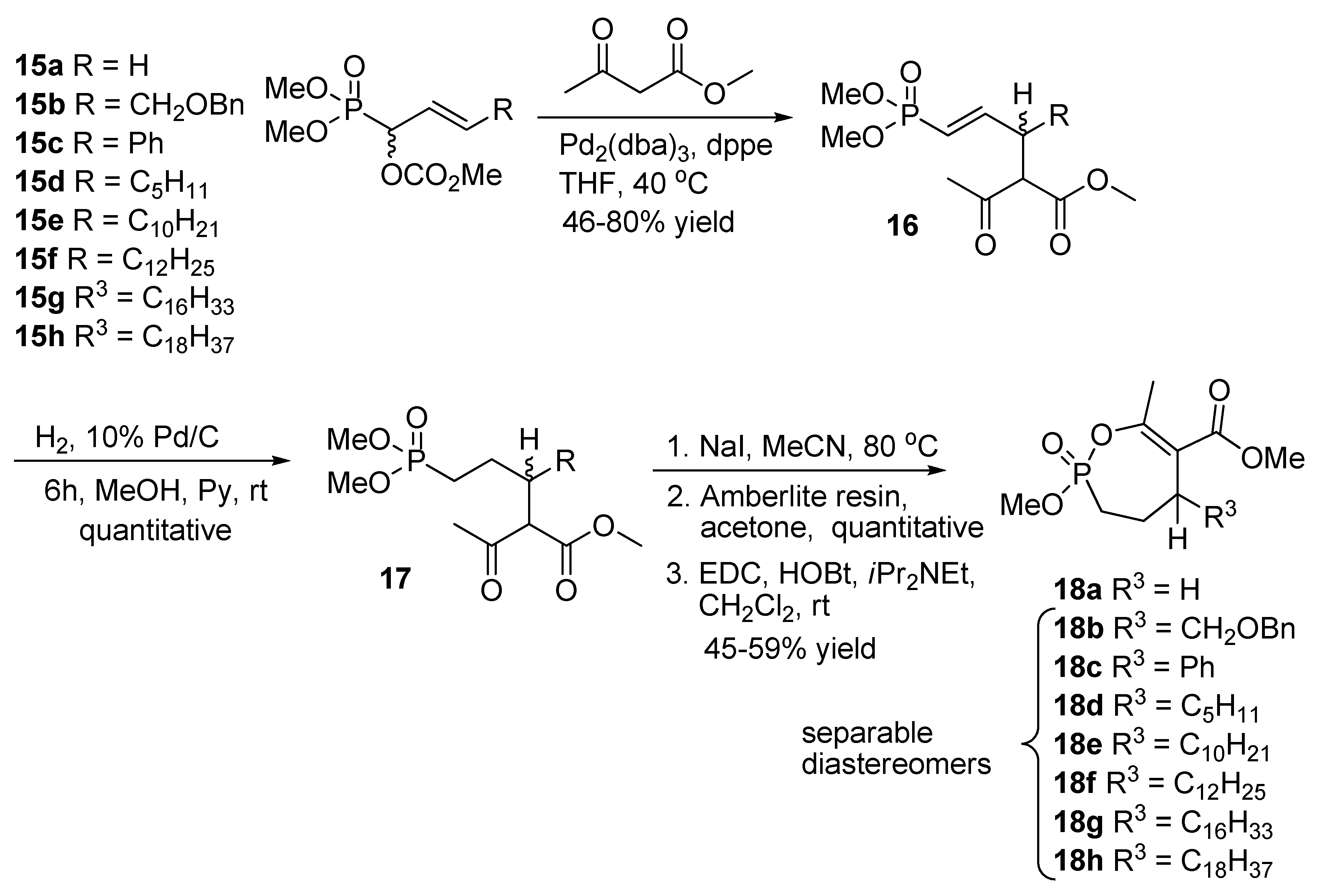
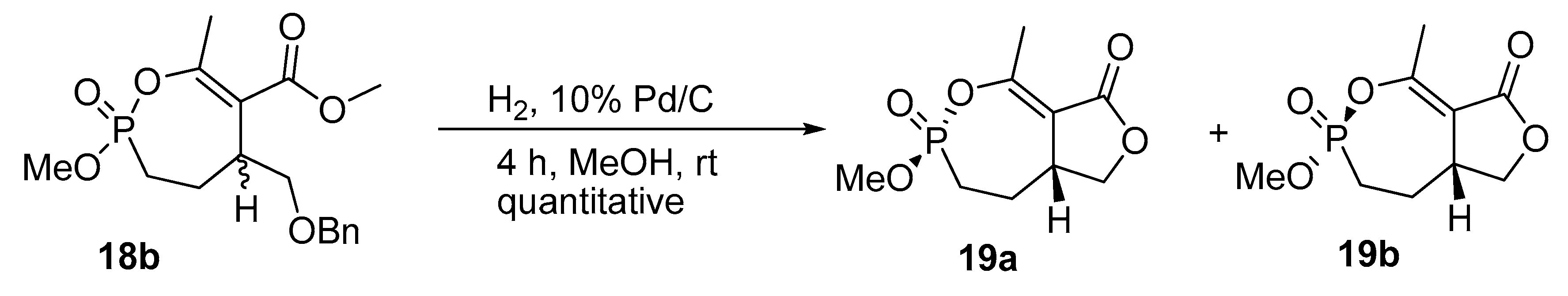
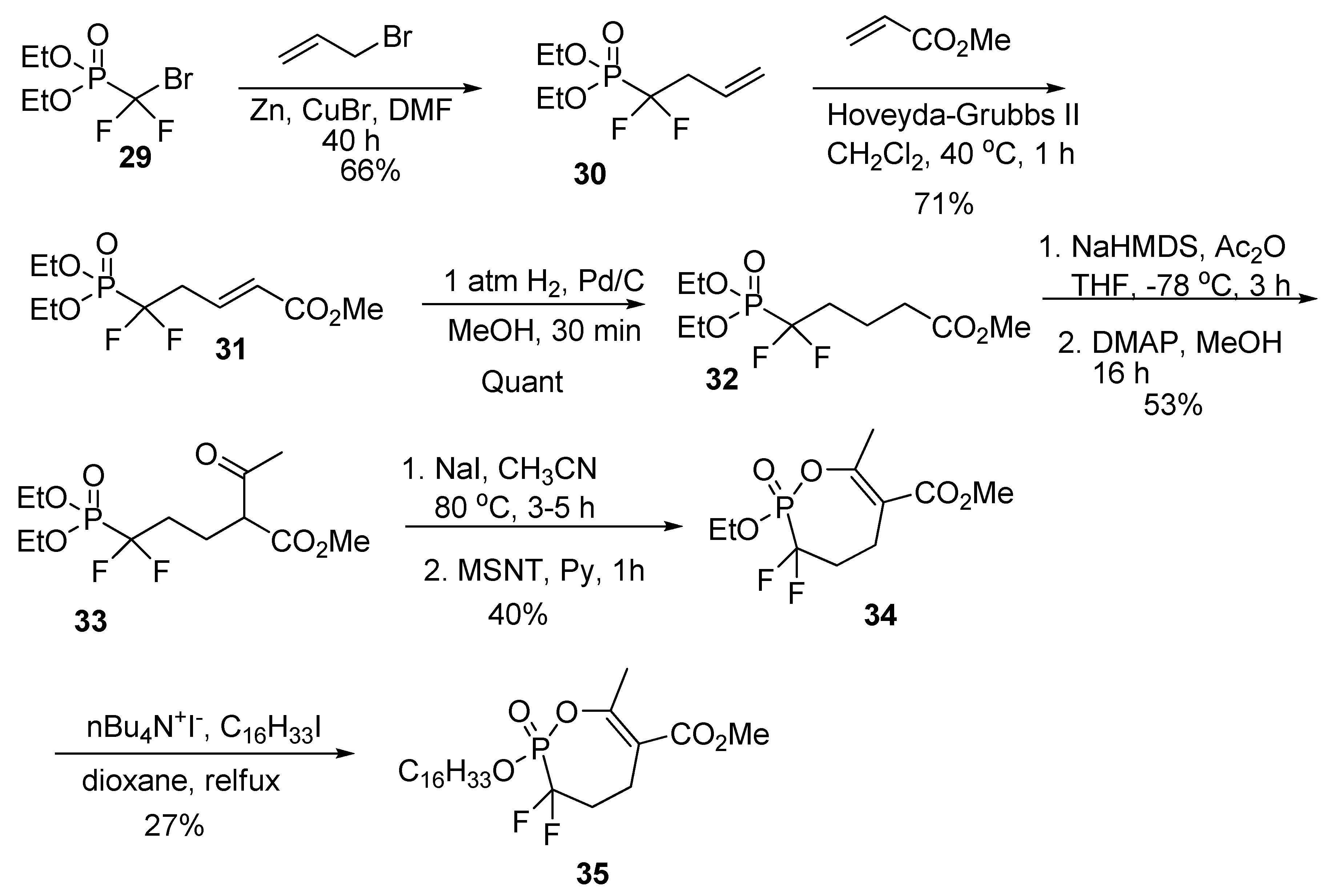
2.10. Synthesis of α,α-difluoro Phosphonate Analogs
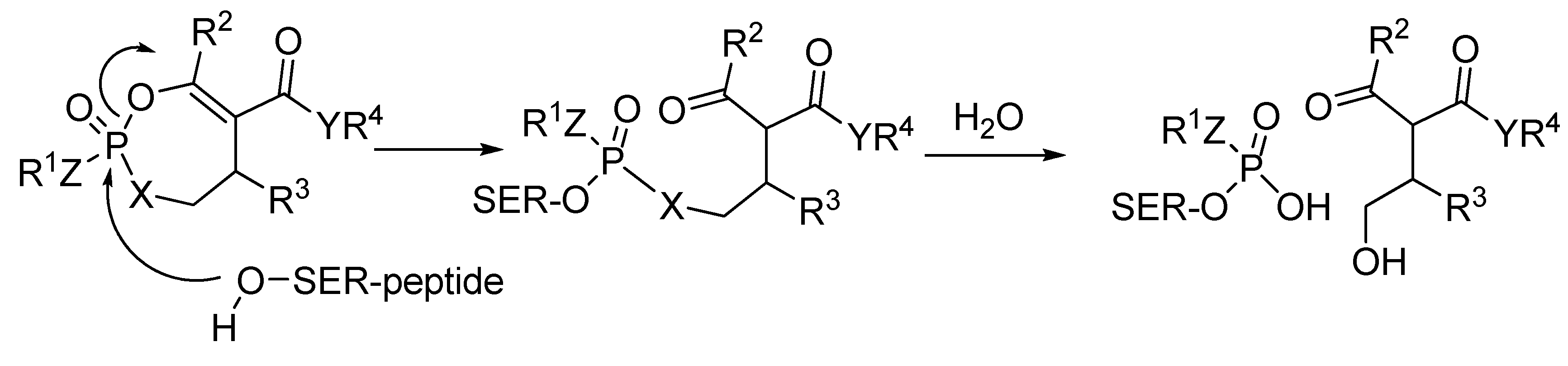
2.11. Biological Activities
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Harvey, A.L.; Edrada-Ebel, R.; Quinn, R.J. The Re-emergence of natural products for drug discovery in the genomic era. Nat. Rev. Drug Discov. 2015, 14, 111–129. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L. Natural products in drug discovery. Drug Discov. Today 2008, 13, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Neumann, R.; Peter, H.H. Insecticidal organophosphates: Nature made them first. Experimentia 1987, 43, 1235–1237. [Google Scholar] [CrossRef]
- Kurokawa, T.; Suzuki, K.; Hayaoka, T.; Nakagawa, T.; Izawa, T.; Kobayashi, M.; Harada, N. Cyclophostin, Acetylcholinesterase inhibitor from Streptomyces lavendulae. J. Antibiot. 1993, 46, 1315–1318. [Google Scholar] [CrossRef] [PubMed]
- Wink, J.; Schmidt, F.-R.; Seibert, G.; Aretz, W. Cyclipostins: Novel hormone sensitive lipase inhibitors from Streptomyces sp. DSM 13381 I. Taxonomic studies of the producer microorganism and fermentation results. J. Antibiot. 2002, 55, 472–479. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vertesy, L.; Beck, B.; Brönstrup, M.; Ehrlich, K.; Kurz, M.; Müller, G.; Schummer, D.; Seibert, G. Cyclipostins: Novel hormone sensitive lipase inhibitors from Streptomyces sp. DSM 13381 II. Isolation, structure, elucidation and biological properties. J. Antibiot. 2002, 55, 480–494. [Google Scholar] [CrossRef][Green Version]
- Vertesy, L.; Ehrlich, K.; Kurz, M.; Wink, J. Cyclopostins, Process for their Preparation and Use Thereof. U. S. Patent US 6,756,402 B2, 29 June 2004. [Google Scholar]
- Seibert, G.; Toti, L.; Wink, J. Treating Mycobacterial Infections with Cyclipostins. WO/2008/025449, 6 March 2008. [Google Scholar]
- Schulze, C.J.; Navarro, G.; Ebert, D.; DeRisi, J.; Linnington, R.G. Salinipostins A-K, long-chain bicyclic phosphotriesters as a potent and selective antimalarial chemotype. J. Org. Chem. 2015, 80, 1312–1320. [Google Scholar] [CrossRef]
- Malla, R.K.; Bandyopadhyay, S.; Spilling, C.D.; Dutta, S.; Dupureur, C.M. The first total synthesis of (±) cyclophostin and (±) cyclipostin P: Inhibitors of the serine hydrolases acetyl cholinesterase and hormone sensitive lipase. Org. Lett. 2011, 13, 3094–3097. [Google Scholar] [CrossRef][Green Version]
- Cahn, R.S.; Ingold, C.K.; Prelog, V. Specification of molecular chirality. Angew. Chem. Int. Ed. 1966, 5, 385–415. [Google Scholar] [CrossRef]
- Morin, J.B.; Adams, K.L.; Sello, J.K. Replication of biosynthetic reactions enables efficient synthesis of A-Factor, a γ-butyrolactone autoinducer from Streptomyces griseus. Org. Biomol. Chem. 2012, 10, 1517–1520. [Google Scholar] [CrossRef] [PubMed]
- Nihira, T.; Shimizu, Y.; Kim, H.S.; Yamada, Y. Structure-activity relationships of virginiae butanolide C, an inducer of virginiamycin production in Streptomyces virginiae. J. Antbiot. 1988, 1828–1837. [Google Scholar] [CrossRef]
- Sakuda, S.; Higashi, A.; Tanaka, S.; Nihira, T.; Yamada, Y. Biosynthesis of virginiae butanolide A, a butyrolactone autoregulator from Streptomyces. J. Am. Chem. Soc. 1992, 114, 663–668. [Google Scholar] [CrossRef]
- Sakuda, S.; Tanaka, S.; Mizuno, K.; Sukcharoen, O.; Nihira, T.; Yamada, Y. Biosynthetic studies on virginiae butanolide A, a butyrolactone autoregulator from Streptomyces. Part 2. Preparation of Possible Biosynthetic intermediates and conversion experiments in cell-free system. J. Chem. Soc. Perkin Trans. I 1993, 2309–2315. [Google Scholar] [CrossRef]
- Takano, E.; Nihira, T.; Hara, Y.; Jones, J.J.; Gershater, C.J.L.; Yamada, Y.; Bibb, M. Purification and structural determination of SCB1, a γ-butyrolactone that elicits antibiotic production in Streptomyces coelicolor A3(2). J. Biol. Chem. 2000, 275, 11010–11016. [Google Scholar] [CrossRef]
- Kato, J.; Funa, N.; Watanabe, H.; Ohnishi, Y.; Horinouchi, S. Biosynthesis of γ-butyrolactone autoregulators that switch on secondary metabolism and morphological development in Streptomyces. Proc. Natl. Acad. Sci. USA 2007, 104, 2378–2383. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wei, X.; Liu, X.; Dong, X.; Yu, R.; Wan, S.; Jiang, T. Total synthesis of marine cyclic enol-phosphotriester salinipostin compounds. J. Ocean. Univ. China 2018, 17, 683–689. [Google Scholar] [CrossRef]
- Bandyopadhyay, S.; Dutta, S.; Spilling, C.D.; Dupureur, C.M.; Rath, N.P. Synthesis and biological evaluation of a phosphonate analog of the natural acetyl cholinesterase inhibitor cyclophostin. J. Org. Chem. 2008, 73, 8386–8391. [Google Scholar] [CrossRef]
- Dutta, S.; Malla, R.K.; Bandyopadhyay, S.; Spilling, C.D.; Dupureur, C.M. Synthesis and kinetic analysis of some phosphonate analogs of cyclophostin as inhibitors of human acetylcholinesterase. Bioorg. Med. Chem. 2010, 18, 2265–2274. [Google Scholar] [CrossRef][Green Version]
- Point, V.; Malla, R.K.; Diomande, S.; Martin, B.P.; Delorme, V.; Carrière, F.; Canaan, S.; Rath, N.P.; Spilling, C.D.; Cavalier, J.-F. Synthesis and kinetic evaluation of cyclophostin and cyclipostins phosphonate analogs as selective and potent inhibitors of microbial lipases. J. Med. Chem. 2012, 55, 10204–10219. [Google Scholar] [CrossRef]
- Point, V.; Malla, R.K.; Carrière, F.; Canaan, S.; Spilling, C.D.; Cavalier, J.-F. Enantioselective inhibition of microbial lipolytic enzymes by nonracemic monocyclic enolphosphonate analogs of cyclophostin. J. Med. Chem. 2013, 56, 4393–4401. [Google Scholar] [CrossRef] [PubMed]
- Vasilieva, E.; Dutta, S.; Malla, R.K.; Martin, B.P.; Spilling, C.D.; Dupureur, C.M. Rat hormone sensitive lipase inhibition by cyclipostins and their analogs. Bioorg. Med. Chem. 2015, 23, 944–952. [Google Scholar] [CrossRef][Green Version]
- Martin, B.P.; Vasilieva, E.; Dupureur, C.M.; Spilling, C.D. Synthesis and comparison of the biological activity of monocyclic phosphonate, difluorophosphonate and phosphate analogs of the natural AChE inhibitor cyclophostin. Bioorg. Med. Chem. 2015, 23, 7529–7534. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.C.; Delorme, V.; Bénarouche, A.; Martin, B.P.; Paudel, R.; Gnawali, G.R.; Madani, A.; Puppo, R.; Landry, V.; Kremer, L.; et al. Cyclipostins and cyclophostin analogs as promising compounds in the fight against tuberculosis. Sci. Rep. 2017, 7, 11751. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.C.; Madani, A.; Santucci, P.; Martin, B.P.; Paudel, R.; Delattre, S.; Herrmann, J.-L.; Spilling, C.D.; Kremer, L.; Canaan, S. Cyclophostin and cyclipostins analogs, new promising molecules to treat mycobacterial-related diseases. Int. J. Antimicrob Agents 2018, 51, 651–654. [Google Scholar] [CrossRef] [PubMed]
- Viljoen, A.; Richard, M.; Nguyen, P.C.; Fourquet, P.; Camoin, L.; Paudal, R.; Gnawali, G.R.; Spilling, C.D.; Cavalier, J.-F.; Canaan, S.; et al. Cyclipostins and cyclophostin analogs inhibit the antigen 85C from Mycobacterium tuberculosis both in vitro and in vivo. J. Biol. Chem. 2018, 293, 2755–2769. [Google Scholar] [CrossRef]
- Nguyen, P.C.; Nguyen, V.S.; Martin, B.P.; Fourquet, P.; Camoin, L.; Spilling, C.D.; Cavalier, J.-F.; Cambillau, C.; Canaan, S. Biochemical and structural characterization of TesA, a major thioesterase required for outer-envelope lipid biosynthesis in M. tuberculosis. J. Mol. Biol. 2018, 430, 5120–5136. [Google Scholar] [CrossRef]
- Santucci, P.; Point, V.; Poncin, I.; Guy, A.; Crauste, C.; Serveau-Avesque, C.; Galano, J.M.; Spilling, C.D.; Cavalier, J.F.; Canaan, S. LipG a bifunctional phospholipase/thioesterase involved in mycobacterial envelope remodeling. Biosci. Rep. 2018, 38, BSR20181953. [Google Scholar] [CrossRef]
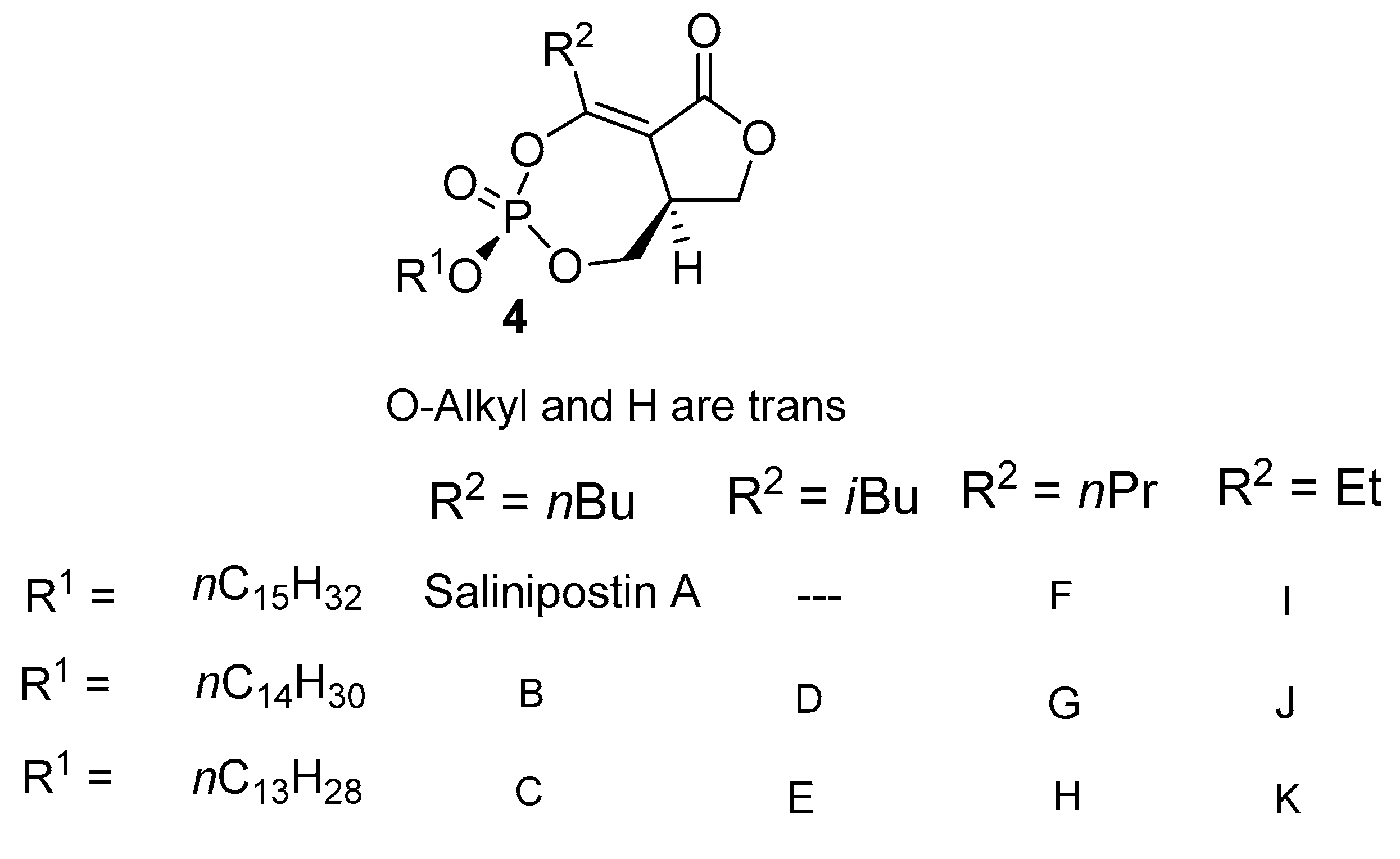
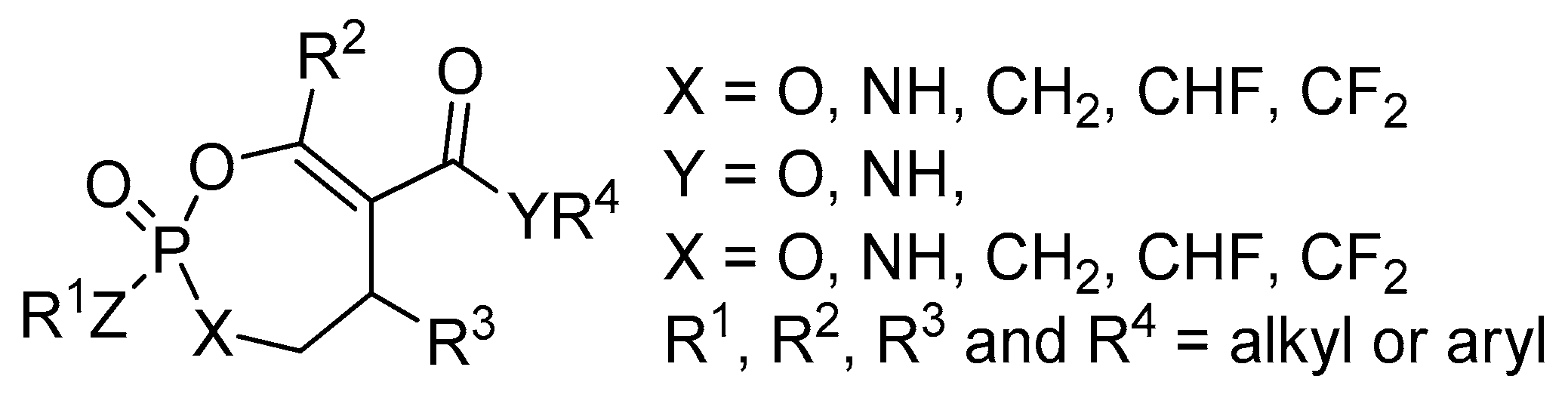
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spilling, C.D. The Chemistry and Biology of Cyclophostin, the Cyclipostins and Related Compounds. Molecules 2019, 24, 2579. https://doi.org/10.3390/molecules24142579
Spilling CD. The Chemistry and Biology of Cyclophostin, the Cyclipostins and Related Compounds. Molecules. 2019; 24(14):2579. https://doi.org/10.3390/molecules24142579
Chicago/Turabian StyleSpilling, Christopher D. 2019. "The Chemistry and Biology of Cyclophostin, the Cyclipostins and Related Compounds" Molecules 24, no. 14: 2579. https://doi.org/10.3390/molecules24142579
APA StyleSpilling, C. D. (2019). The Chemistry and Biology of Cyclophostin, the Cyclipostins and Related Compounds. Molecules, 24(14), 2579. https://doi.org/10.3390/molecules24142579




