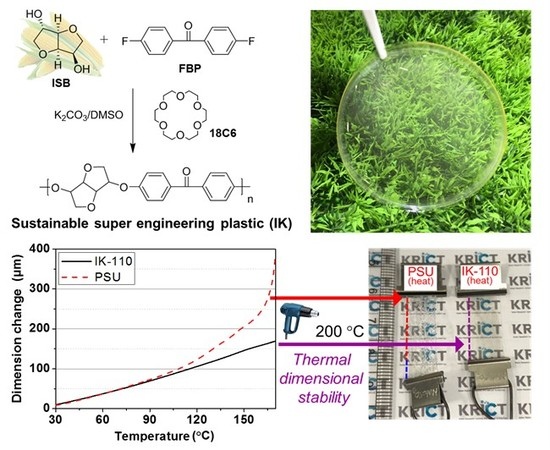
Study on the Synthetic Characteristics of Biomass-Derived Isosorbide-Based Poly(arylene ether ketone)s for Sustainable Super Engineering Plastic
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
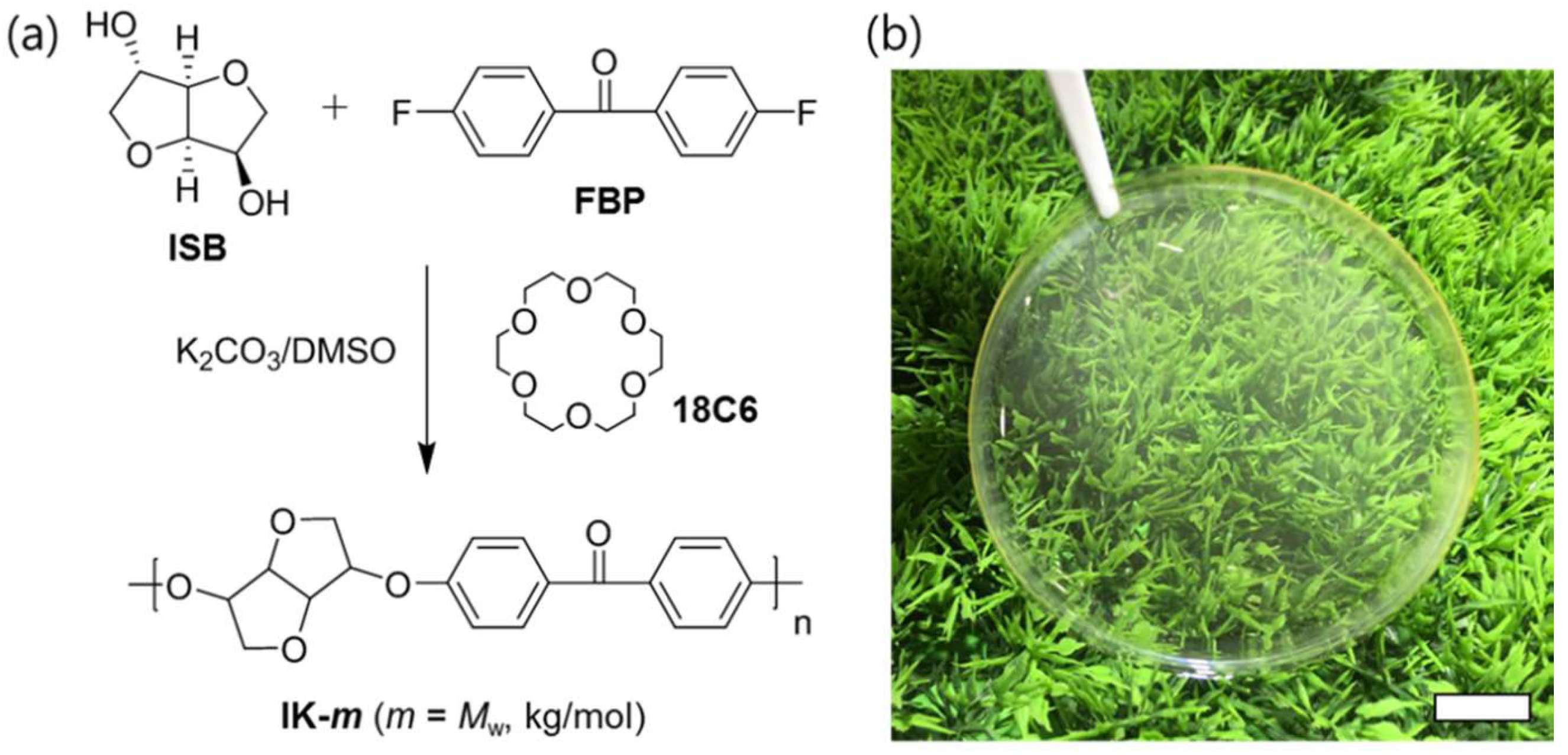
2.2. Polymerization of ISB-Based Poly(arylene ether ketone)s
2.3. Characterization
3. Results and Discussion
3.1. Polymerization Conditions for High MW
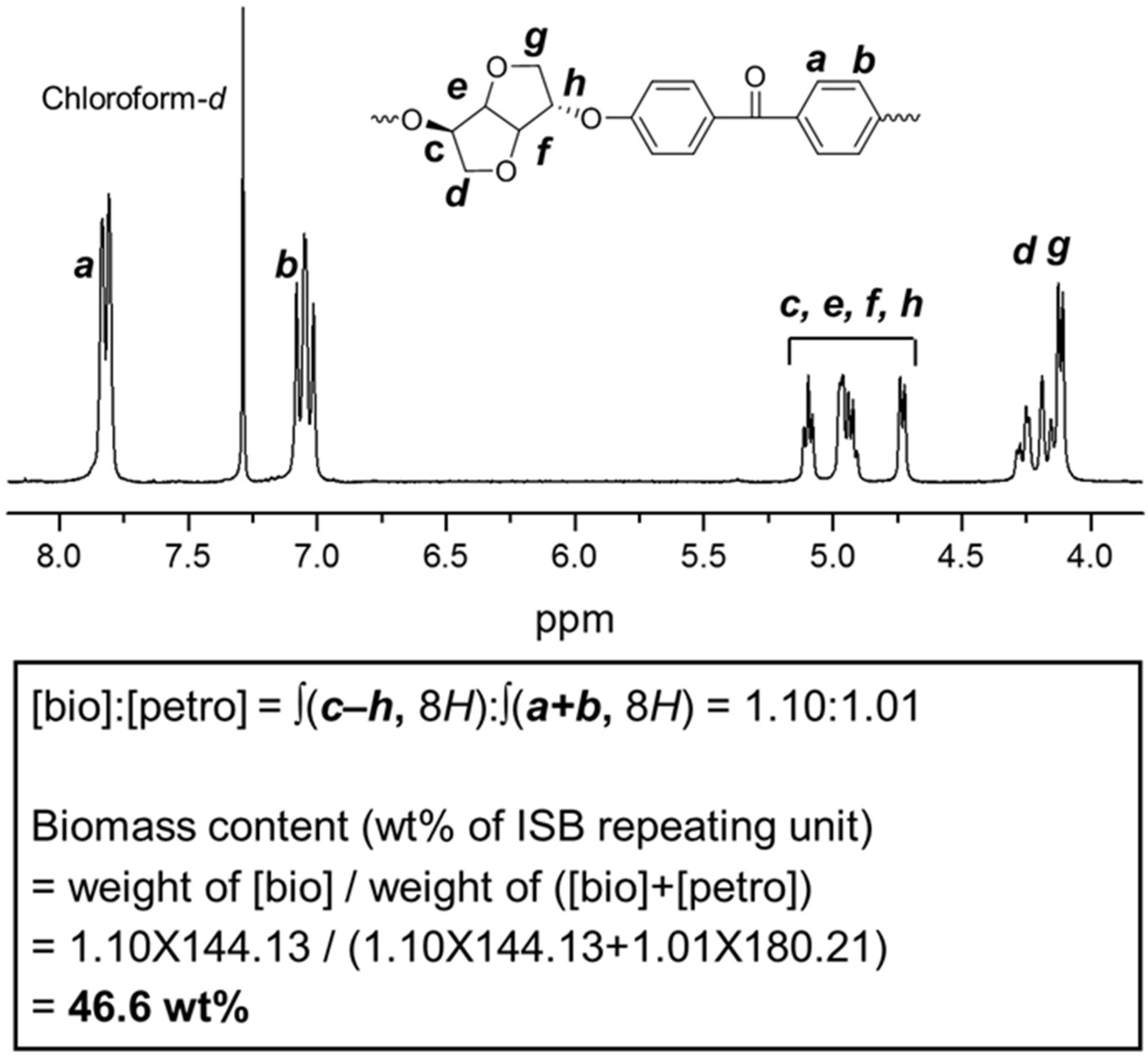
3.2. Determination of Biomass Contents of Polymers
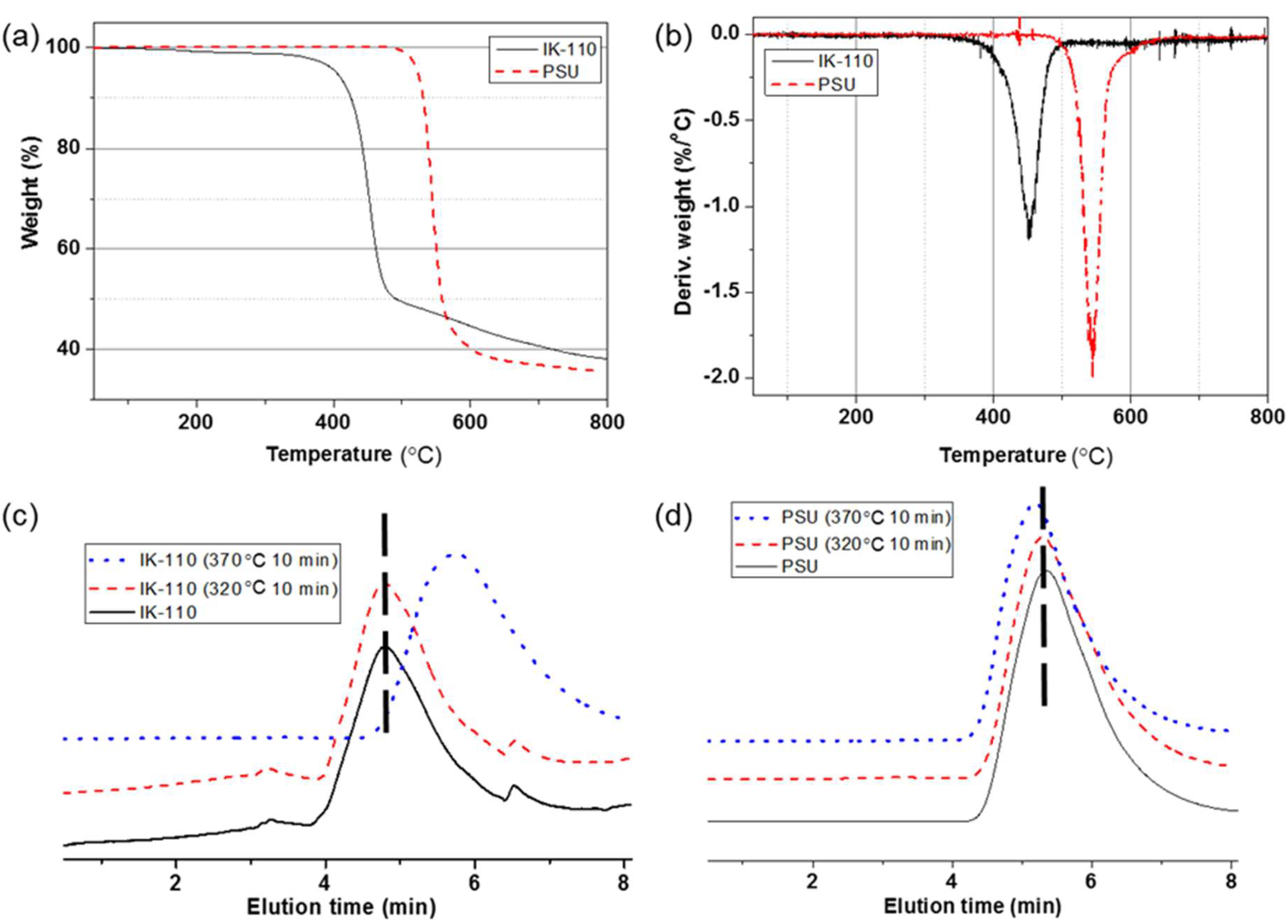
3.3. Thermal Degradation Stability Comparisons of Polymers from ISB with BPA
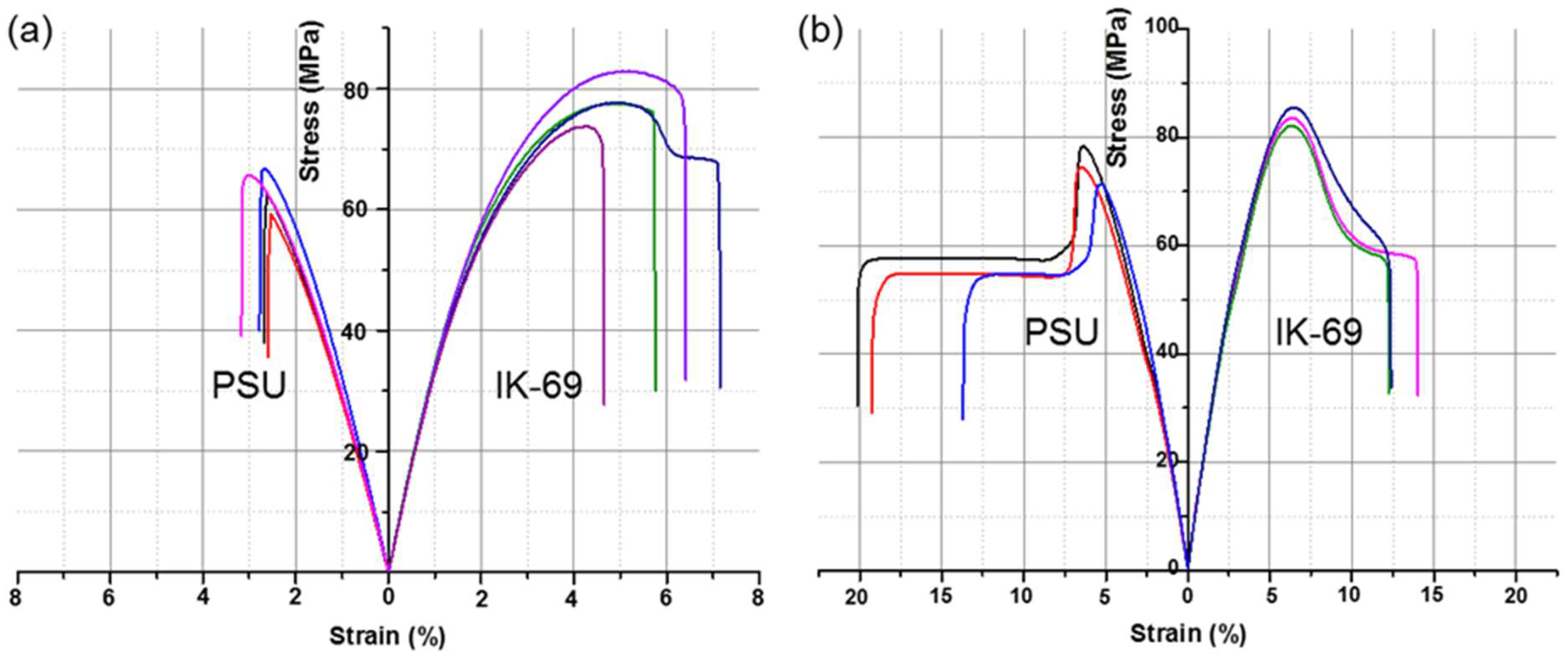
3.4. Preparation of Solution Casted Films and Melt Injection Molded Specimens for Mechanical Performances
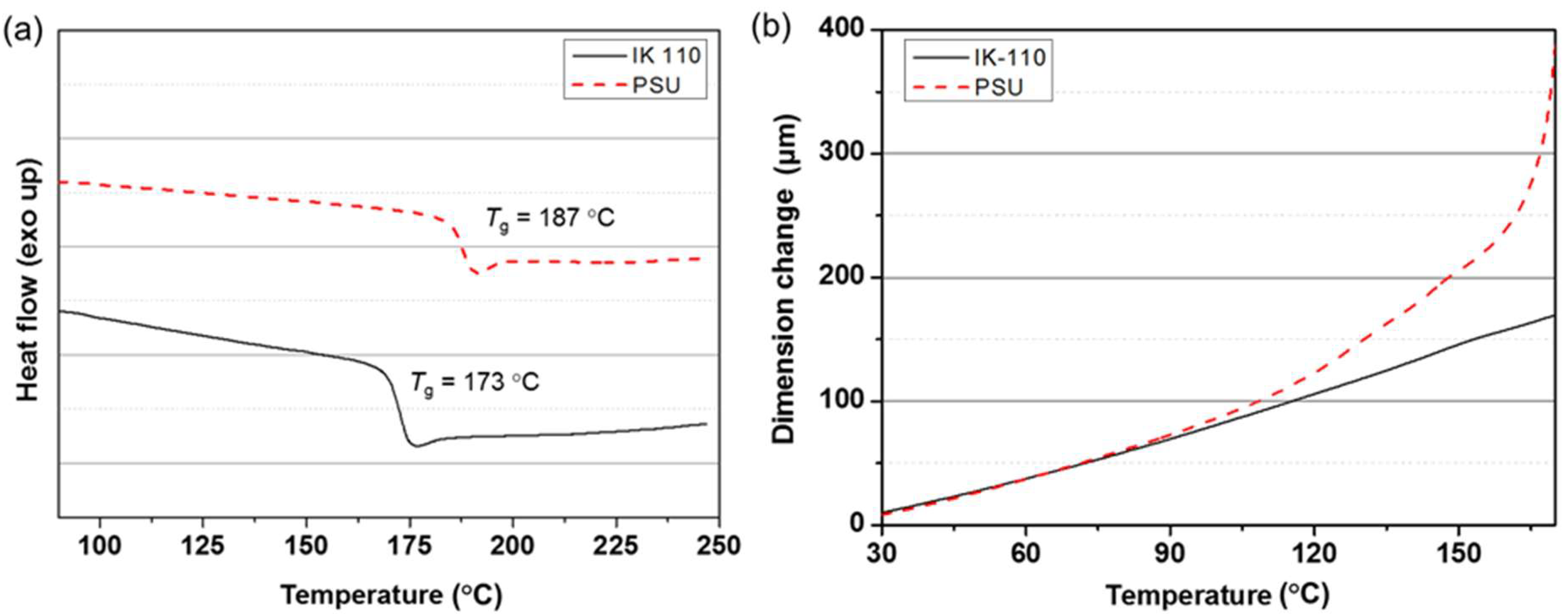
3.5. Thermal Dimensional Stability of Bio-SEP Meriting over Petrochemical PSU
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rose, J.B. Preparation and properties of poly(arylene ether sulphones). Polymer 1974, 15, 456–465. [Google Scholar] [CrossRef]
- Park, J.; Moon, M.; Seo, M.; Choi, H.; Kim, S.Y. Well-defined star-shaped rod-coil diblock copolymers as a new class of unimolecular micelles: Encapsulation of guests and thermoresponsive phase transition. Macromolecules 2010, 43, 8304–8313. [Google Scholar] [CrossRef]
- Labadie, J.W.; Hedrick, J.L.; Ueda, M. Poly(aryl ether) Synthesis. ACS Symp. Ser. 1996, 624, 210–225. [Google Scholar]
- Park, J.; Kim, J.; Seo, M.; Lee, J.; Kim, S.Y. Dual-mode fluorescence switching induced by self-assembly of well-defined poly (arylene ether sulfone)s containing pyrene and amide moieties. Chem. Commun. 2012, 48, 10556–10558. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Maier, G. Novel high Tg high-strength poly(aryl ether)s. Chem. Mater. 1999, 11, 2179–2184. [Google Scholar] [CrossRef]
- Lee, J.; Lee, B.; Park, J.; Oh, J.; Kim, T.; Seo, M.; Kim, S.Y. Synthesis and phase transition behavior of well-defined Poly (arylene ether sulfone)s by chain growth condensation polymerization in organic media. Polymer 2018, 153, 430–437. [Google Scholar] [CrossRef]
- Loan, S. Functionalized Polysulfones: Synthesis, Characterization, and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Ka, D.; Park, J.; Kim, S.D.; Lee, S.; Kim, S.Y. Grafting of trifluoromethylated poly(phenylene oxide)s on a single-walled carbon nanotube via surface-initiated chain-growth condensation polymerization. J. Polym. Sci. A 2017, 55, 3180–3184. [Google Scholar] [CrossRef]
- Dhara, M.G.; Banerjee, S. Fluorinated high-performance polymers: Poly(arylene ether)s and aromatic polyimides containing trifluoromethyl groups. Prog. Polum. Sci. 2010, 35, 1022–1077. [Google Scholar] [CrossRef]
- Park, J.; Seo, M.; Choi, H.; Kim, S.Y. Synthesis and physical gelation induced by self-assembly of well-defined poly(arylene ether sulfone)s with various numbers of arms. Polym. Chem. 2011, 2, 1174–1179. [Google Scholar] [CrossRef]
- Zimmerman, J.B.; Anastas, P.T. Toward substitution with no regrets. Science 2015, 347, 1198–1199. [Google Scholar] [CrossRef]
- Rochester, J.R. Bisphenol A and human health: A review of the literature. Reproductive Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, C.; Shin, H.; Kho, Y.; Choi, K. Comparison of thyroid hormone disruption potentials by bisphenols A, S, F, and Z in embryo-larval zebrafish. Chemosphere 2019, 221, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Fenouillot, F.; Rousseaua, A.; Colomines, G.; Saint-Loup, R.; Pascault, J.-P. Polymers from renewable 1,4:3,6-dianhydrohexitols (isosorbide, isomannide and isoidide): A review. Prog. Polym. Sci. 2010, 35, 578–622. [Google Scholar] [CrossRef]
- Zhu, Y.; Romain, C.; Williams, C.K. Sustainable polymers from renewable resources. Nature 2016, 540, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Editorial. The future of plastic. Nat. Commun. 2018, 9, 2157. Available online: https://www.nature.com/articles/s41467-018-04565-2.pdf (accessed on 8 July 2019).
- Moon, S.I.; Lee, C.W.; Miyamoto, M.; Kimura, Y. Melt polycondensation of L-lactic acid with Sn(II) catalysts activated by various proton acids: A direct manufacturing route to high molecular weight Poly(L-lactic acid). J. Polym. Sci. A 2000, 38, 1673–1679. [Google Scholar] [CrossRef]
- Ryu, M.H.; Park, J.; Oh, D.X.; Hwang, S.Y.; Jeon, H.; Im, S.; Jegal, J. Precisely controlled two-step synthesis of cellulose-graft-poly (L-lactide) copolymers: Effects of graft chain length on thermal behavior. Polym. Deg. Stab. 2017, 142, 226–233. [Google Scholar] [CrossRef]
- Tran, T.H.; Nguyen, H.-L.; Hao, L.T.; Kong, H.; Park, J.M.; Jung, S.-H.; Cha, H.G.; Lee, J.Y.; Kim, H.; Hwang, S.Y.; et al. A ball milling-based one-step transformation of chitin biomass to organo-dispersible strong nanofibers passing highly time and energy consuming processes. Int. J. Biol. Macromol. 2019, 125, 660–667. [Google Scholar] [CrossRef]
- Gigli, M.; Fabbri, M.; Lotti, N.; Gamberini, R.; Rimini, B.; Munari, A. Poly(butylene succinate)-based polyesters for biomedical applications: A review. Europ. Polym. J. 2016, 75, 431–460. [Google Scholar] [CrossRef]
- Kim, T.; Jeon, H.; Jegal, J.; Kim, J.H.; Yang, H.; Park, J.; Oh, D.X.; Hwang, S.Y. Trans crystallization behavior and strong reinforcement effect of cellulose nanocrystals on reinforced poly(butylene succinate) nanocomposites. RSC Adv. 2018, 8, 15389–15398. [Google Scholar] [CrossRef]
- Feng, X.; East, A.J.; Hammond, W.B.; Zhang, Y.; Jaffe, M. Overview of advances in sugar-based polymers. Polym. Adv. Technol. 2011, 22, 139–150. [Google Scholar] [CrossRef]
- Nelson, A.M.; Long, T.E. A perspective on emerging polymer technologies for bisphenol-A replacement. Polym. Int. 2012, 61, 1485–1491. [Google Scholar] [CrossRef]
- Rose, M.; Palkovits, R. Isosorbide as a renewable platform chemical for versatile applications-quo vadis? ChemSusChem 2012, 5, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Kucherov, F.A.; Gordeev, E.G.; Kashin, A.S.; Ananikov, V.P. Three-dimensional printing with biomass-derived PEF for carbon-neutral manufacturing. Angew. Chem. Int. Ed. 2017, 56, 15931–15935. [Google Scholar] [CrossRef] [PubMed]
- Rosenboom, J.-G.; Hohl, D.K.; Fleckenstein, P.; Storti, G.; Morbidelli, M. Bottle-grade polyethylene furanoate from ring-opening polymerisation of cyclic oligomers. Nat. Commun. 2018, 9, 2701. [Google Scholar] [CrossRef] [PubMed]
- Haurenstein, O.; Agarwal, S.; Greiner, A. Bio-based polycarbonate as synthetic toolbox. Nat. Commun. 2016, 7, 11862. [Google Scholar]
- Bolton, J.M.; Hillmyer, M.A.; Hoye, T.R. Sustainable thermoplastic elastomers from terpene-derived monomers. ACS Macro Lett. 2014, 3, 717–720. [Google Scholar] [CrossRef]
- Mialon, L.; Pemba, A.G.; Miller, S.A. Biorenewable polyethylene terephthalate mimics derived from lignin and acetic acid. Green Chem. 2010, 12, 1704–1706. [Google Scholar] [CrossRef]
- Kaneko, T.; Thi, T.H.; Shi, D.J.; Akashi, M. Environmentally degradable, high-performance thermoplastics from phenolic phytomonomers. Nat. Mater. 2006, 5, 966–970. [Google Scholar] [CrossRef]
- Nguyen, H.T.H.; Qi, P.; Rostagno, M.; Fetcha, A.; Miller, S.A. The quest for high glass transition temperature bioplastics. J. Mater. Chem. A. 2018, 6, 9298–9331. [Google Scholar] [CrossRef]
- Wu, J.; Eduard, P.; Jasinska-Walc, L.; Rozanski, A.; Noordover, B.A.J.; van Es, D.S.; Koning, C.E. Fully isohexide-based polyesters: Synthesis, characterization, and structure–properties relations. Macromolecules 2013, 46, 384–394. [Google Scholar] [CrossRef]
- Yoon, W.J.; Oh, K.S.; Koo, J.M.; Kim, J.R.; Lee, K.J.; Im, S.S. Advanced polymerization and properties of biobased high Tg polyester of isosorbide and 1,4-cyclohexanedicarboxylic acid through in situ acetylation. Macromolecules 2013, 46, 2930–2940. [Google Scholar] [CrossRef]
- Yoon, W.J.; Hwang, S.W.; Koo, J.M.; Lee, Y.J.; Lee, S.U.; Im, S.S. Synthesis and characteristics of a biobased high-Tg terpolyester of isosorbide, ethylene glycol, and 1,4-cyclohexane dimethanol: Effect of ethylene glycol as a chain linker on polymerization. Macromolecules 2013, 46, 7219–7231. [Google Scholar] [CrossRef]
- Koo, J.M.; Hwang, S.Y.; Yoon, W.J.; Lee, Y.G.; Kim, S.H.; Im, S.S. Structural and thermal properties of poly(1,4-cyclohexane dimethylene terephthalate) containing isosorbide. Polym. Chem. 2015, 6, 6973–6986. [Google Scholar] [CrossRef]
- Park, S.-A.; Choi, J.; Ju, S.; Jegal, J.; Lee, K.M.; Hwang, S.Y.; Oh, D.X.; Park, J. Copolycarbonates of bio-based rigid isosorbide and flexible 1,4-cyclohexanedimethanol: Merits over bisphenol-A based polycarbonates. Polymer 2017, 116, 153–159. [Google Scholar] [CrossRef]
- Kim, T.; Koo, J.M.; Ryu, M.H.; Jeon, H.; Kim, S.-K.; Park, S.-A.; Oh, D.X.; Park, J.; Hwang, S.Y. Sustainable terpolyester of high Tg based on bio heterocyclic monomer of dimethyl furan-2, 5-dicarboxylate and isosorbide. Polymer 2017, 132, 122–132. [Google Scholar] [CrossRef]
- Chatti, S.; Hani, M.A.; Bornhorst, K.; Kricheldorf, H.R. Poly(ether sulfone) of isosorbide, isomannide and isoidide. High. Perform. Polym. 2009, 21, 105–118. [Google Scholar] [CrossRef]
- Bennour, H.; Medimagh, R.; Fildier, A.; Raffin, G.; Hangouet, M.; Chatti, S.; Zina, M.S.; Kricheldorf, H.R. Hyperbranched cyclic and multicyclic poly(ether ketone)s by polycondensation of isosorbide and isomannide with 2,6,4’-trifluorobenzophenone and 1,3,5-tris(4-fluorobenzoyl) benzene. High. Perform. Polym. 2014, 26, 144–155. [Google Scholar] [CrossRef]
- Abderrazak, H.B.; Fildier, A.; Romdhane, A.B.; Chatti, S.; Kricheldorf, H.R. Synthesis of new poly(ether ketone)s derived from biobased diols. Macromol. Chem. Phys. 2013, 214, 1423–1433. [Google Scholar] [CrossRef]
- Belgacem, C.; Medimagh, R.; Fildier, A.; Bulete, A.; Kricheldorf, H.; Romdhane, H.B.; Chatti, S. Synthesis and characterization of isosorbide-based α,ω-dihydroxyethersulfone oligomers. Deg. Monomers Polym. 2015, 18, 64–72. [Google Scholar] [CrossRef][Green Version]
- Belgacem, C.; Medimagh, R.; Kricheldorf, H.; Romdhane, H.B.; Chatti, S. Copolyethersulfones of 1,4:3,6-dianhydrohexitols and bisphenol A. Deg. Monomers Polym. 2016, 19, 248–255. [Google Scholar] [CrossRef]
- Tsuchiya, K.; Ishida, Y.; Higashihara, T.; Kameyama, A.; Ueda, M. Synthesis of poly(arylene ether sulfone): 18-Crown-6 catalyzed phase-transfer polycondensation of bisphenol A with 4,4′-dichlorodiphenyl sulfone. Polym. J. 2015, 47, 353–354. [Google Scholar] [CrossRef]
- Vlasov, V.M. Fluoride ion as a nucleophile and a leaving group in aromatic nucleophilic substitution reactions. J. Fluorine Chem. 1993, 61, 193–216. [Google Scholar] [CrossRef]
- ASTM International, ASTM D6866-18 Method B: Standard Test Methods for Determining the Biobased Content of Solid, Liquid and Gaseous Samples Using Radiocarbon Analysis. Available online: https://www.astm.org/Standards/D6866.htm (accessed on 8 July 2019).
- Kunioka, M. Possible incorporation of petroleum-based carbons in biochemicals produced by bioprocess. Appl. Microbiol. Biotechnol. 2010, 87, 491–497. [Google Scholar] [CrossRef] [PubMed]
- van Krevelen, D.W.; te Nijenhuis, K. Properties of Polymers: Their Correlation with Chemical Structure; their Numerical Estimation and Prediction from Additive Group Contributions, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Park, S.-A.; Jeon, H.; Kim, H.; Kim, S.-H.; Choy, S.; Hwang, D.S.; Koo, J.M.; Jegal, J.; Hwang, S.W.; Park, J.; et al. Sustainable and recyclable super engineering thermoplastic from biorenewable monomer. Nat. Commun. 2019, 10, 2601. [Google Scholar] [CrossRef] [PubMed]
- Perepechko, I. Low-Temperature Properties of Polymers; Elsevier: Moscow, Russia, 2013. [Google Scholar]
- Courtney, T.H. Mechanical Behavior of Materials, 2nd ed.; Waveland Press: Long Grove, IL, USA, 2005. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors upon reasonable request. |
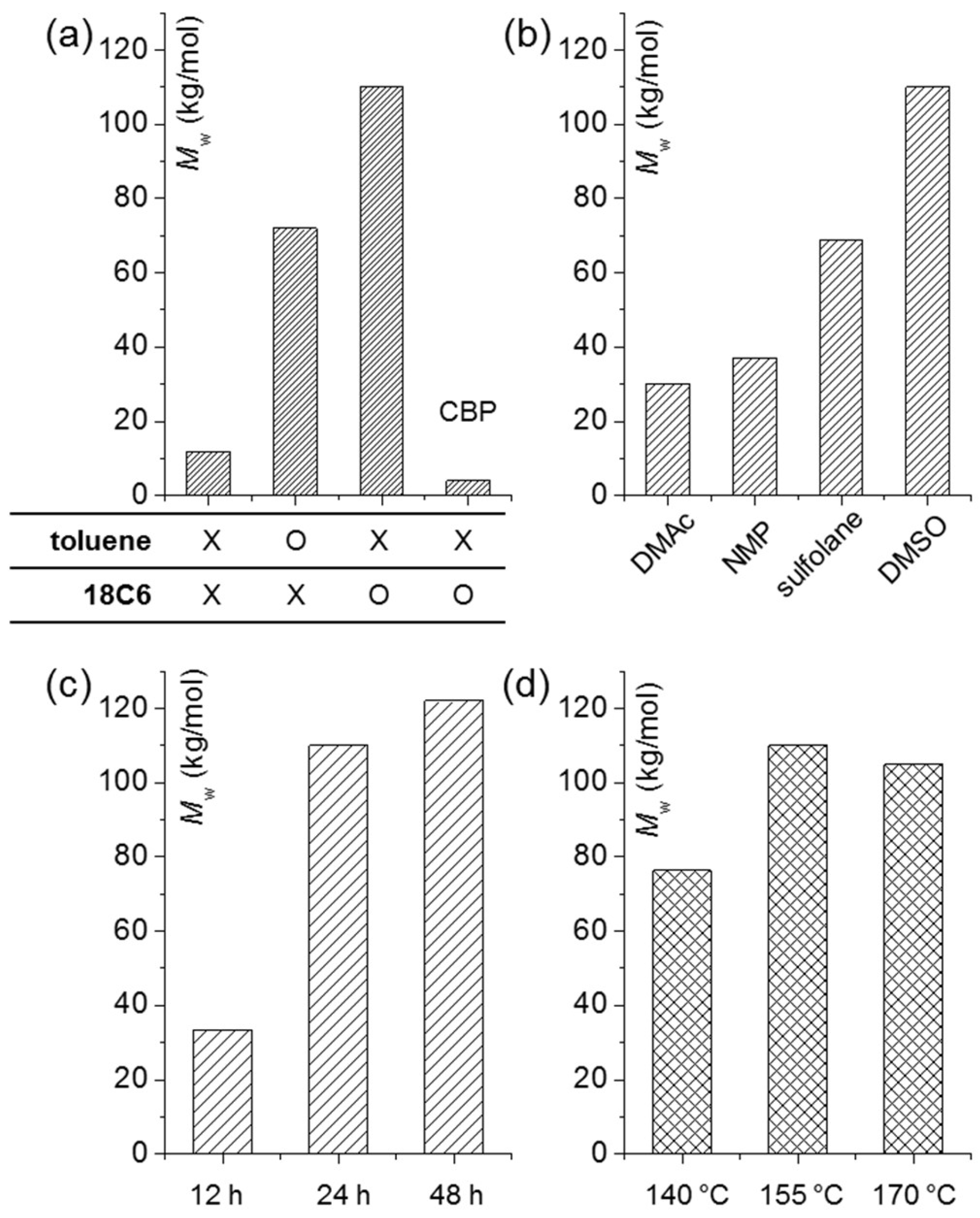

| Entry (IK-m) a | Halogen b | 18C6 c | Toluene d | Solvent e | Time/Temperature (h/°C) f | Mw (kg/mol) g | PDI g |
|---|---|---|---|---|---|---|---|
| IK-12 | FBP | X | X | DMSO | 24/155 | 11.6 | 1.86 |
| IK-72 | FBP | X | O | DMSO | 24/155 | 72.2 | 1.73 |
| IK-110 | FBP | O | X | DMSO | 24/155 | 110 | 1.68 |
| IK-4 | CBP | O | X | DMSO | 24/155 | 4.0 | 1.65 |
| IK-30 | FBP | O | X | DMAc | 24/155 | 30.0 | 2.21 |
| IK-37 | FBP | O | X | NMP | 24/155 | 37.0 | 2.19 |
| IK-69 | FBP | O | X | sulfolane | 24/155 | 68.8 | 1.62 |
| IK-34 | FBP | O | X | DMSO | 12/155 | 33.5 | 2.11 |
| IK-122 | FBP | O | X | DMSO | 48/155 | 122 | 2.08 |
| IK-76 | FBP | O | X | DMSO | 24/140 | 76.3 | 1.56 |
| IK-105 | FBP | O | X | DMSO | 24/170 | 105 | 2.06 |
| PSU | - | - | - | - | - | 63.7 | 1.64 |
| pMC a | Biocarbon Content (%) b | |
|---|---|---|
| IK-110 | 32.17 | 32 (±3) |
| PSU | 0.24 | 0 (±3) |
| IK-110 | PSU | |
|---|---|---|
| Td5 (°C) a | 411 | 522 |
| Tmax (°C) b | 454 | 545 |
| Weight loss at 320 °C for 1 h (wt%) c | 2.5 | 0.1 |
| Weight loss at 370 °C for 1 h (wt%) c | 50.3 | 0.1 |
| Mw after 10 min at 320 °C (kg/mol) d | 114 | 63.3 |
| Mw after 10 min at 370 °C (kg/mol) d | 29.8 | 63.5 |
| Transmittance at 550 nm (%) e | 97.2 | 99.4 |
| Pencil hardness | HB | HB |
| CTE (ppm, 30–80 °C) f | 53.2 | 65.4 |
| CTE (ppm, 80–170 °C) f | 70.4 | 227 |
| Type | PSU | IK-69 | ||||||
|---|---|---|---|---|---|---|---|---|
| Young’s Modulus (GPa) | UTS (MPa) | Elongation at Break (%) | Toughness (MJ/m3) | Young’s Modulus (GPa) | UTS (MPa) | Elongation at Break (%) | Toughness (MJ/m3) | |
| Solution casted film | 3.1 (± 0.09) | 64 (±1.7) | 2.8 (±0.13) | 1.1 (±0.09) | 3.7 (± 0.05) | 78 (±2.2) | 6.0 (±0.73) | 3.5 (±0.57) |
| Injection molded specimen | 2.0 (± 0.03) | 74 (±2.1) | 18 (±2.0) | 9.3 (±1.3) | 2.2 (± 0.03) | 84 (±1.0) | 13 (±0.6) | 7.8 (±0.34) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.-A.; Im, C.; Oh, D.X.; Hwang, S.Y.; Jegal, J.; Kim, J.H.; Chang, Y.-W.; Jeon, H.; Park, J. Study on the Synthetic Characteristics of Biomass-Derived Isosorbide-Based Poly(arylene ether ketone)s for Sustainable Super Engineering Plastic. Molecules 2019, 24, 2492. https://doi.org/10.3390/molecules24132492
Park S-A, Im C, Oh DX, Hwang SY, Jegal J, Kim JH, Chang Y-W, Jeon H, Park J. Study on the Synthetic Characteristics of Biomass-Derived Isosorbide-Based Poly(arylene ether ketone)s for Sustainable Super Engineering Plastic. Molecules. 2019; 24(13):2492. https://doi.org/10.3390/molecules24132492
Chicago/Turabian StylePark, Seul-A, Changgyu Im, Dongyeop X. Oh, Sung Yeon Hwang, Jonggeon Jegal, Ji Hyeon Kim, Young-Wook Chang, Hyeonyeol Jeon, and Jeyoung Park. 2019. "Study on the Synthetic Characteristics of Biomass-Derived Isosorbide-Based Poly(arylene ether ketone)s for Sustainable Super Engineering Plastic" Molecules 24, no. 13: 2492. https://doi.org/10.3390/molecules24132492
APA StylePark, S.-A., Im, C., Oh, D. X., Hwang, S. Y., Jegal, J., Kim, J. H., Chang, Y.-W., Jeon, H., & Park, J. (2019). Study on the Synthetic Characteristics of Biomass-Derived Isosorbide-Based Poly(arylene ether ketone)s for Sustainable Super Engineering Plastic. Molecules, 24(13), 2492. https://doi.org/10.3390/molecules24132492