Simplification of Carbon Bond Mechanism IV (CBM-IV) under Different Initial Conditions by Using Concentration Sensitivity Analysis
Abstract
1. Introduction
2. Mathematical Models and Methods
2.1. Reaction System
2.2. Concentration Sensitivity Analysis
2.3. Configurations of the Model
3. Results and Discussion
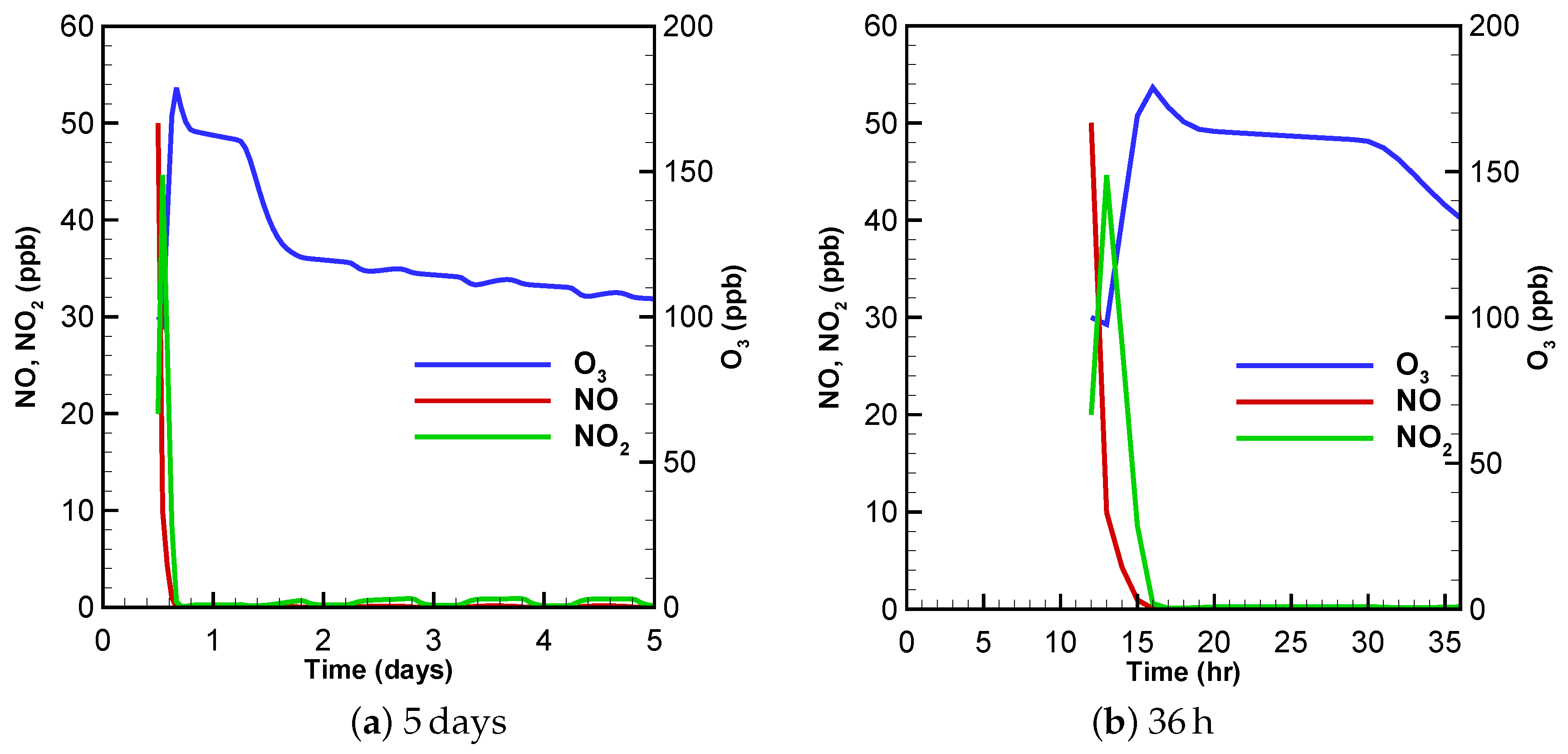
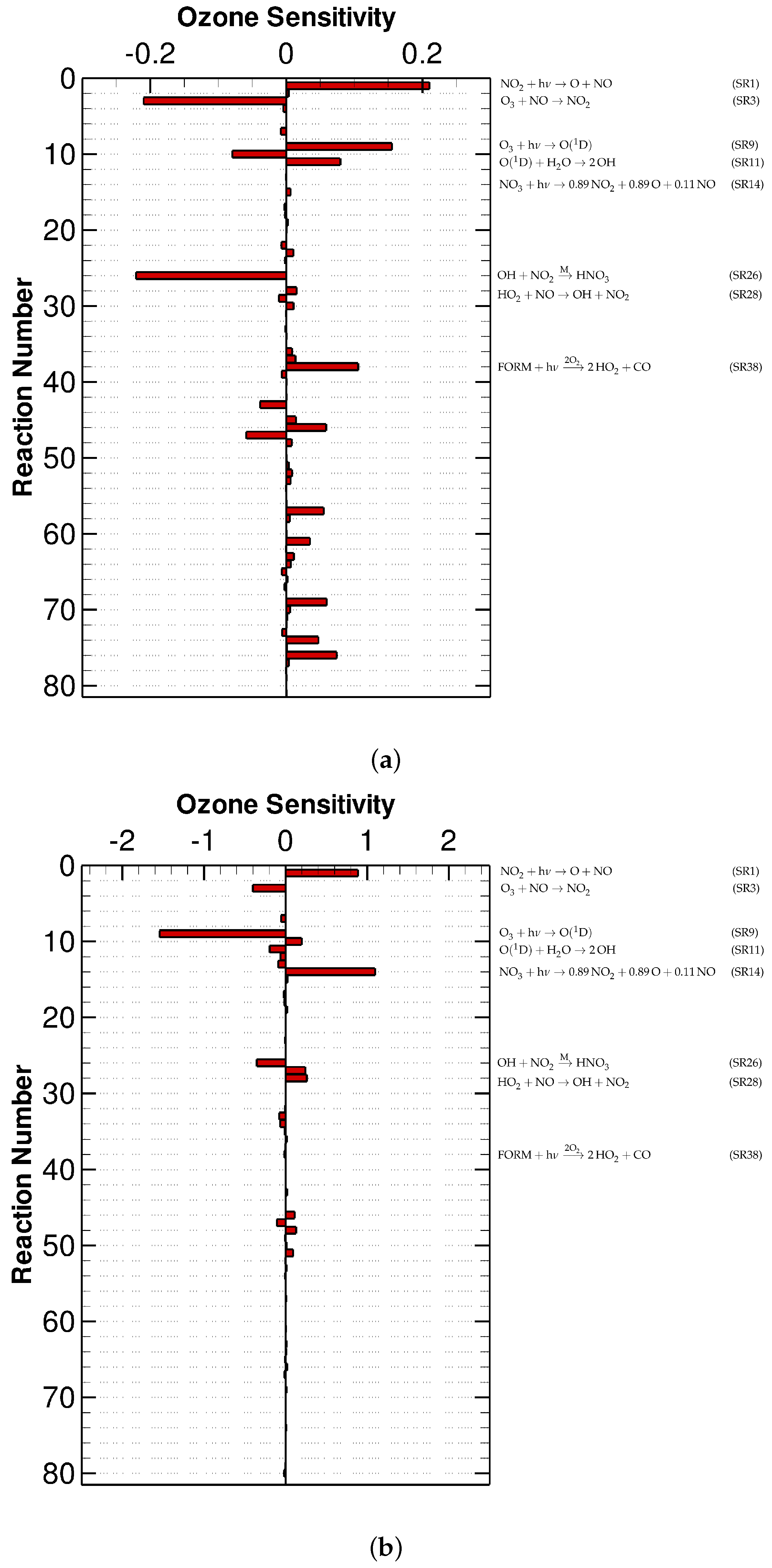
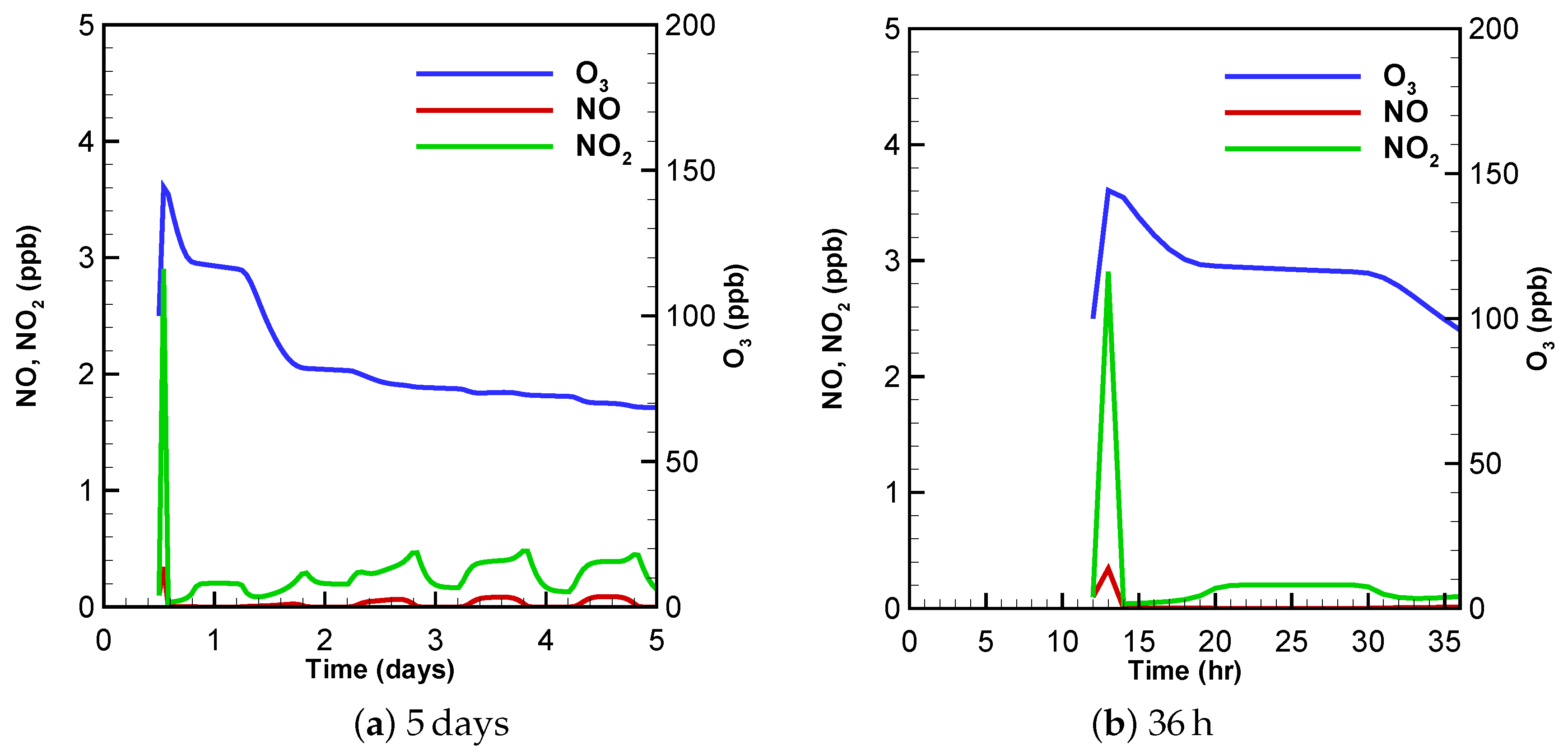
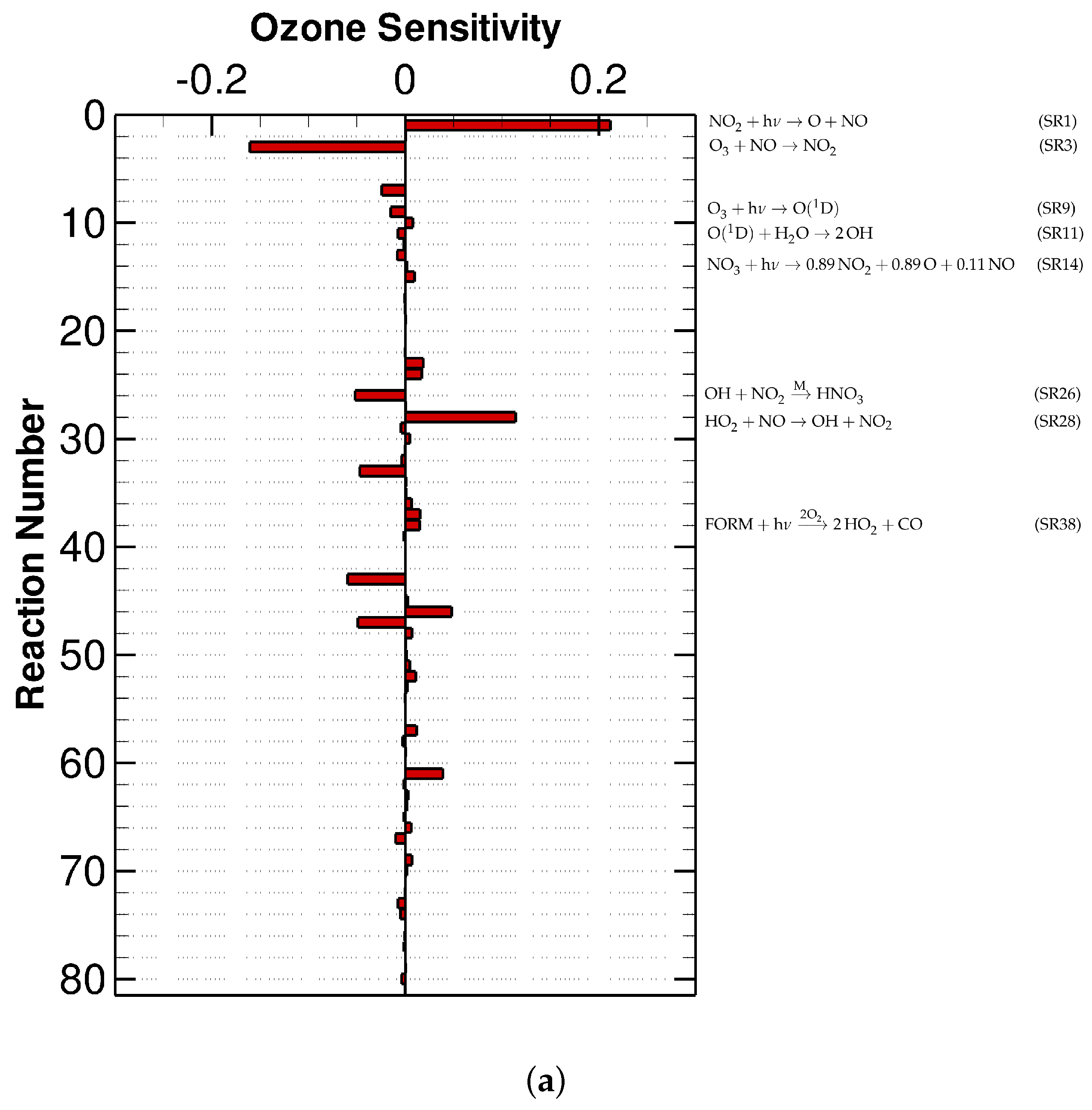
3.1. Urban Scenario
3.2. Low-NOx Scenario
4. Conclusions and Future Work
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Reaction Number | Reaction | Rate Constant (k) () |
|---|---|---|
| (SR1) | radiation dependent | |
| (SR2) | ||
| (SR3) | ||
| (SR4) | ||
| (SR5) | ||
| (SR6) | ||
| (SR7) | ||
| (SR8) | radiation dependent | |
| (SR9) | radiation dependent | |
| (SR10) | ||
| (SR11) | ||
| (SR12) | ||
| (SR13) | ||
| (SR14) | radiation dependent | |
| (SR15) | ||
| (SR16) | ||
| (SR17) | ||
| (SR18) | ||
| (SR19) | ||
| (SR20) | ||
| (SR21) | ||
| (SR22) | ||
| (SR23) | radiation dependent | |
| (SR24) | ||
| (SR25) | ||
| (SR26) | ||
| (SR27) | ||
| (SR28) | ||
| (SR29) | ||
| (SR30) | ||
| (SR31) | ||
| (SR32) | ||
| (SR33) | ||
| (SR34) | radiation dependent | |
| (SR35) | ||
| (SR36) | ||
| (SR37) | ||
| (SR38) | radiation dependent | |
| (SR39) | radiation dependent | |
| (SR40) | ||
| (SR41) | ||
| (SR42) | ||
| (SR43) | ||
| (SR44) | ||
| (SR45) | radiation dependent | |
| (SR46) | ||
| (SR47) | ||
| (SR48) | ||
| (SR49) | ||
| (SR50) | ||
| (SR51) | ||
| (SR52) | ||
| (SR53) | ||
| (SR54) | ||
| (SR55) | ||
| (SR56) | ||
| (SR57) | ||
| (SR58) | ||
| (SR59) | ||
| (SR60) | ||
| (SR61) | ||
| (SR62) | ||
| (SR63) | ||
| (SR64) | ||
| (SR65) | 4.2 | |
| (SR66) | ||
| (SR67) | ||
| (SR68) | ||
| (SR69) | ||
| (SR70) | ||
| (SR71) | radiation dependent | |
| (SR72) | ||
| (SR73) | ||
| (SR74) | radiation dependent | |
| (SR75) | ||
| (SR76) | ||
| (SR77) | ||
| (SR78) | ||
| (SR79) | ||
| (SR80) | ||
| (SR81) |
References
- Akimoto, H. Global air quality and pollution. Science 2003, 302, 1716–1719. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.K.; Yao, X. Air pollution in mega cities in China. Atmos. Environ. 2008, 42, 1–42. [Google Scholar] [CrossRef]
- WHO. Ambient Air Pollution: A Global Assessment of Exposure and Burden of Disease; Technical Report; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Dodge, M.C. Chemical oxidant mechanisms for air quality modeling: Critical review. Atmos. Environ. 2000, 34, 2103–2130. [Google Scholar] [CrossRef]
- Jimenez, P.; Baldasano, J.M.; Dabdub, D. Comparison of photochemical mechanisms for air quality modeling. Atmos. Environ. 2003, 37, 4179–4194. [Google Scholar] [CrossRef]
- Gery, M.W.; Whitten, G.; Killus, J.P.; Dodge, M.C. A photochemical kinetics mechanism for urban and regional scale computer modeling. J. Geophys. Res. 1989, 94, 12925–12956. [Google Scholar] [CrossRef]
- Zaveri, R.A.; Peters, L.K. A new lumped structure photochemical mechanism for large-scale applications. J. Geophys. Res. Atmos. 1999, 104, 30387–30415. [Google Scholar] [CrossRef]
- Yarwood, G.; Rao, S.; Yocke, M.; Whitten, G. Updates to the Carbon Bond Chemical Mechanism: CB05; Technical Report; U.S. Environmental Protection Agency: Washington, DC, USA, 2005.
- Yarwood, G.; Jung, J.; Whitten, G.; Heo, G.; J, M.; M, E. Updates to the Carbon Bond Mechanism for Version 6 (CB6). In Proceedings of the 9th Annual CMAS Conference, Chapel Hill, NC, USA, 11–13 October 2010; pp. 1–4. [Google Scholar]
- Carter, W.P. Documentation of the SAPRC-99 Chemical Mechanism for VOC Reactivity Assessment; Technical Report 329; California Air Resources Board: Sacramento, CA, USA, 2000.
- Carter, W.P. Implementation of the SAPRC-99 Chemical Mechanism into the Models-3 Framework; Technical Report; United States Environmental Protection Agency: Washington, DC, USA, 2000.
- Carter, W.P. Development of the SAPRC-07 chemical mechanism. Atmos. Environ. 2010, 44, 5324–5335. [Google Scholar] [CrossRef]
- Pitts, J.N.; Darnall, K.; Carter, W.; Winer, A.; Atkinson, R. Mechanisms of Photochemical Reactions in Urban Air; Technical Report; Environmental Sciences Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency: Washington, DC, USA, 1977.
- Jeffries, H.E.; Kamens, R.M.; Sexton, K.G.; Gerhardt, A.A. Outdoor Smog Chamber Experiments to Test Photochemical Models; Technical Report; U.S. Environmental Protection Agency, Environmental Sciences Research Laboratory: Research Triangle Park, NC, USA, 1982.
- Jeffries, H.E.; Sexton, K.G.; Kamens, R.M.; Holleman, M.S. Outdoor Smog Chamber Experiments to Test Photochemical Models: Phase II; Technical Report; U.S. Environmental Protection Agency, Atmospheric Sciences Research Laboratory: Research Triangle Park, NC, USA, 1985.
- Carter, W.P.L.; Dodd, M.C.; Long, W.D.; Atkinson, R.; . Dodge, M.C. Outdoor Chamber Study to Test Multi-Day Effects; Technical Report; U.S. Environmental Protection Agency, Atmospheric Sciences Research Laboratory: Research Triangle Park, NC, USA, 1985; Volumes I–III.
- Grell, G.A.; Peckham, S.E.; Schmitz, R.; McKeen, S.A.; Frost, G.; Skamarock, W.C.; Eder, B. Fully coupled “online” chemistry within the WRF model. Atmos. Environ. 2005, 39, 6957–6975. [Google Scholar] [CrossRef]
- Damian, V.; Sandu, A.; Damian, M.; Potra, F.A.; Carmichael, G.R. The kinetic preprocessor KPP—A software environment for solving chemical kinetics. Comput. Chem. Eng. 2002, 26, 1567–1579. [Google Scholar] [CrossRef]
- Sandu, A.; Sander, R. Technical note: Simulating chemical systems in Fortran90 and Matlab with the Kinetic PreProcessor KPP-2.1. Atmos. Chem. Phys. 2006, 6, 187–195. [Google Scholar] [CrossRef]
- Kang, D.; Aneja, V.P.; Mathur, R.; Ray, J.D. Observed and modeled VOC chemistry under high VOC/NO conditions in the Southeast United States national parks. Atmos. Environ. 2004, 38, 4969–4974. [Google Scholar] [CrossRef]
- Matthes, S.; Grewe, V.; Sausen, R.; Roelofs, G.J. Global impact of road traffic emissions on tropospheric ozone. Atmos. Chem. Phys. 2007, 7, 1707–1718. [Google Scholar] [CrossRef]
- Wang, Q.; Han, Z.; Wang, T.; Zhang, R. Impacts of biogenic emissions of VOC and NOx on tropospheric ozone during summertime in eastern China. Sci. Total Environ. 2008, 395, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Oshima, N.; Koike, M.; Zhang, Y.; Kondo, Y.; Moteki, N.; Takegawa, N.; Miyazaki, Y. Aging of black carbon in outflow from anthropogenic sources using a mixing state resolved model: Model development and evaluation. J. Geophys. Res. Atmos. 2009, 114. [Google Scholar] [CrossRef]
- Liu, C.; Chung, C.E.; Yin, Y.; Schnaiter, M. The absorption Ångström exponent of black carbon: from numerical aspects. Atmos. Chem. Phys. 2018, 18, 6259–6273. [Google Scholar] [CrossRef]
- Garmory, A.; Kim, I.; Britter, R.; Mastorakos, E. Simulations of the dispersion of reactive pollutants in a street canyon, considering different chemical mechanisms and micromixing. Atmos. Environ. 2009, 43, 4670–4680. [Google Scholar] [CrossRef]
- Kwak, K.H.; Baik, J.J. A CFD modeling study of the impacts of NOx and VOC emissions on reactive pollutant dispersion in and above a street canyon. Atmos. Environ. 2012, 46, 71–80. [Google Scholar] [CrossRef]
- Kwak, K.H.; Baik, J.J.; Lee, K.Y. Dispersion and photochemical evolution of reactive pollutants in street canyons. Atmos. Environ. 2013, 70, 98–107. [Google Scholar] [CrossRef]
- Kwak, K.H.; Baik, J.J. Diurnal variation of NOx and ozone exchange between a street canyon and the overlying air. Atmos. Environ. 2014, 86, 120–128. [Google Scholar] [CrossRef]
- Turányi, T. KINAL—A program package for kinetic analysis of reaction mechanisms. Comput. Chem. 1990, 14, 253–254. [Google Scholar] [CrossRef]
- Sandu, A.; Daescu, D.N.; Carmichael, G.R. Direct and adjoint sensitivity analysis of chemical kinetic systems with KPP: Part I—Theory and software tools. Atmos. Environ. 2003, 37, 5083–5096. [Google Scholar] [CrossRef]
- Daescu, D.N.; Sandu, A.; Carmichael, G.R. Direct and adjoint sensitivity analysis of chemical kinetic systems with KPP: II—Numerical validation and applications. Atmos. Environ. 2003, 37, 5097–5114. [Google Scholar] [CrossRef]
- Cao, L.; Gao, M.; Li, S.; Yi, Z.; Meng, X. Sensitivity analysis of the dependence of the carbon bond mechanism IV (CBM-IV) on the initial air composition under an urban condition. Atmos. Environ. 2019. under review. [Google Scholar]
- Cao, L.; Wang, C.; Mao, M.; Grosshans, H.; Cao, N. Derivation of the reduced reaction mechanisms of ozone depletion events in the Arctic spring by using concentration sensitivity analysis and principal component analysis. Atmos. Chem. Phys. 2016, 16, 14853–14873. [Google Scholar] [CrossRef]
- Sandu, A.; Verwer, J.; Loon, M.V.; Carmichael, G.; Potra, F.; Dabdub, D.; Seinfeld, J. Benchmarking stiff ode solvers for atmospheric chemistry problems-I. implicit vs explicit. Atmos. Environ. 1997, 31, 3151–3166. [Google Scholar] [CrossRef]
- Atkins, P.; de Paula, J. Atkins’ Physical Chemistry; OUP Oxford: Oxford, UK, 2010. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |

| Species | Urban | Low-NOx |
|---|---|---|
| 50 | 0.1 | |
| 20 | 0.1 | |
| 1 | 30 | |
| 100 | 100 | |
| 300 | 300 | |
| 10 | 10 | |
| 10 | 10 | |
| 1 | 1 | |
| 10 | 10 | |
| 10 | 10 | |
| 10 | 10 | |
| 10 | 10 | |
| Relative humidity | 30% | |
| Temperature | 288.15 K | |
| Altitude | 0 km | |
| Pressure | hPa | |
| Air density | molec. cm |
| Species | Maximum Deviation (%) | Species | Maximum Deviation (%) | Species | Maximum Deviation (%) |
|---|---|---|---|---|---|
| NO | 3.4 | TOL | 0.5 | PNA | 3.1 |
| NO2 | 3.2 | XYL | 0.8 | 2.9 | |
| HONO | 1.9 | ISOP | 4.1 | MGLY | 0.5 |
| O3 | 0.1 | H2O | 0.0 | O(1D) | 0.1 |
| CO | 0.0 | HO2 | 0.4 | O(3P) | 0.4 |
| FORM | 0.2 | H2O2 | 1.2 | CRES | 1.0 |
| 0.3 | OH | 0.4 | CRO | 2.3 | |
| PAN | 0.4 | XO2 | 1.2 | NO3 | 1.7 |
| PAR | 0.2 | ROR | 0.2 | 0.4 | |
| OLE | 0.6 | XO2N | 1.5 | OPEN | 0.5 |
| ETH | 0.4 | HNO3 | 0.5 | N2O5 | 4.6 |
| Species | Maximum Deviation (%) | Species | Maximum Deviation (%) | Species | Maximum Deviation (%) |
|---|---|---|---|---|---|
| NO | 1.8 | TOL | 0.2 | PNA | 0.4 |
| NO2 | 0.4 | XYL | 0.3 | 0.4 | |
| HONO | 0.5 | ISOP | 0.6 | MGLY | 0.2 |
| O3 | 0.0 | H2O | 0.0 | O(1D) | 0.0 |
| CO | 0.0 | HO2 | 0.1 | O(3P) | 0.0 |
| FORM | 0.0 | H2O2 | 0.0 | CRES | 2.2 |
| 1.2 | OH | 0.1 | CRO | 0.3 | |
| PAN | 0.0 | XO2 | 0.2 | NO3 | 2.1 |
| PAR | 0.0 | ROR | 0.1 | 0.2 | |
| OLE | 0.3 | XO2N | 0.4 | OPEN | 0.2 |
| ETH | 0.5 | HNO3 | 0.1 | N2O5 | 2.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, L.; Li, S.; Yi, Z.; Gao, M. Simplification of Carbon Bond Mechanism IV (CBM-IV) under Different Initial Conditions by Using Concentration Sensitivity Analysis. Molecules 2019, 24, 2463. https://doi.org/10.3390/molecules24132463
Cao L, Li S, Yi Z, Gao M. Simplification of Carbon Bond Mechanism IV (CBM-IV) under Different Initial Conditions by Using Concentration Sensitivity Analysis. Molecules. 2019; 24(13):2463. https://doi.org/10.3390/molecules24132463
Chicago/Turabian StyleCao, Le, Simeng Li, Ziwei Yi, and Mengmeng Gao. 2019. "Simplification of Carbon Bond Mechanism IV (CBM-IV) under Different Initial Conditions by Using Concentration Sensitivity Analysis" Molecules 24, no. 13: 2463. https://doi.org/10.3390/molecules24132463
APA StyleCao, L., Li, S., Yi, Z., & Gao, M. (2019). Simplification of Carbon Bond Mechanism IV (CBM-IV) under Different Initial Conditions by Using Concentration Sensitivity Analysis. Molecules, 24(13), 2463. https://doi.org/10.3390/molecules24132463





