Essential Oil of Mentha aquatica var. Kenting Water Mint Suppresses Two-Stage Skin Carcinogenesis Accelerated by BRAF Inhibitor Vemurafenib
Abstract
1. Introduction
2. Results
2.1. Chemical Compositions of Mentha aquatica var. Kenting Water Mint Essential Oil
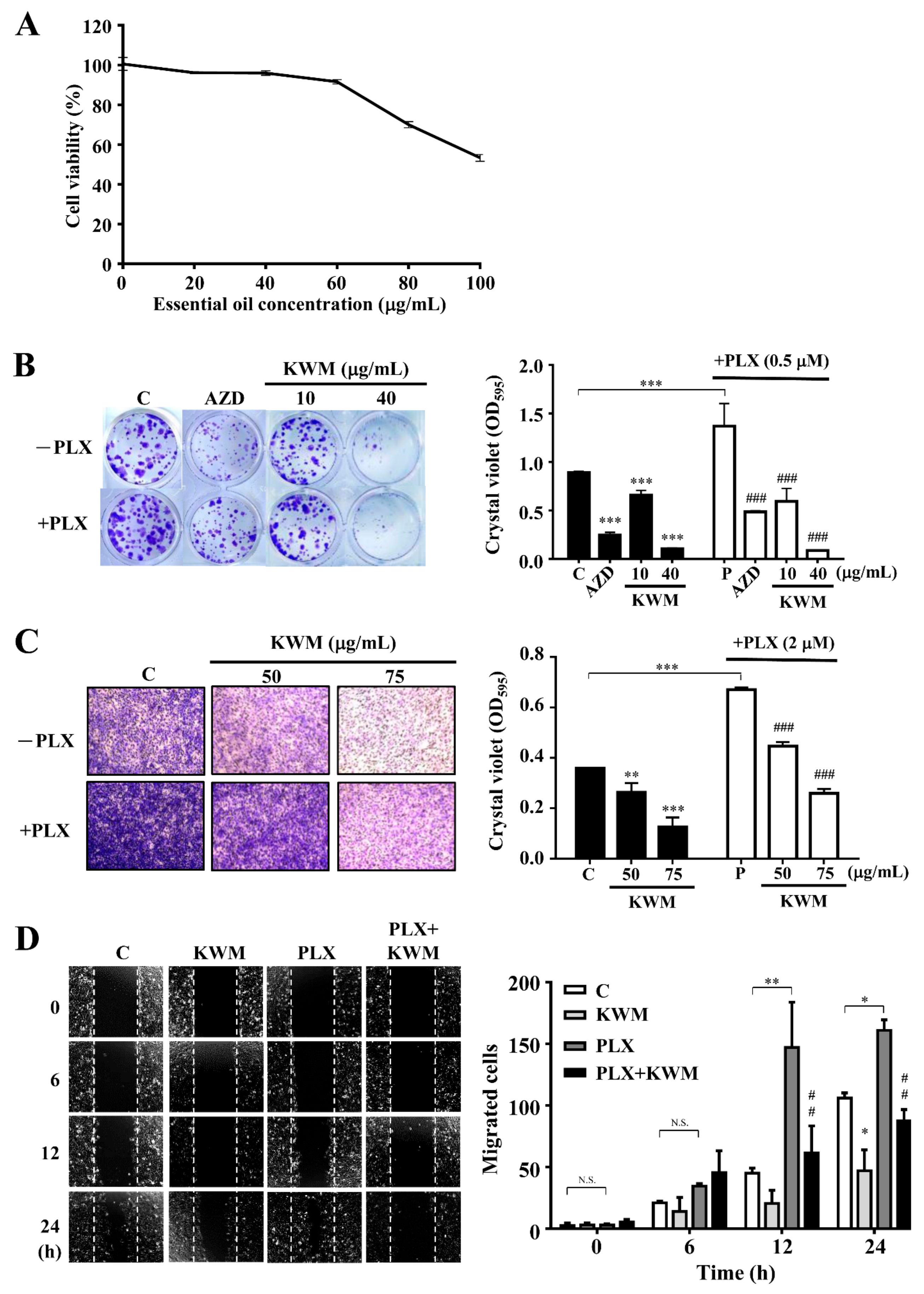
2.2. KWM-EO Effect on PDV Cell Proliferation, Invasion, and Migration
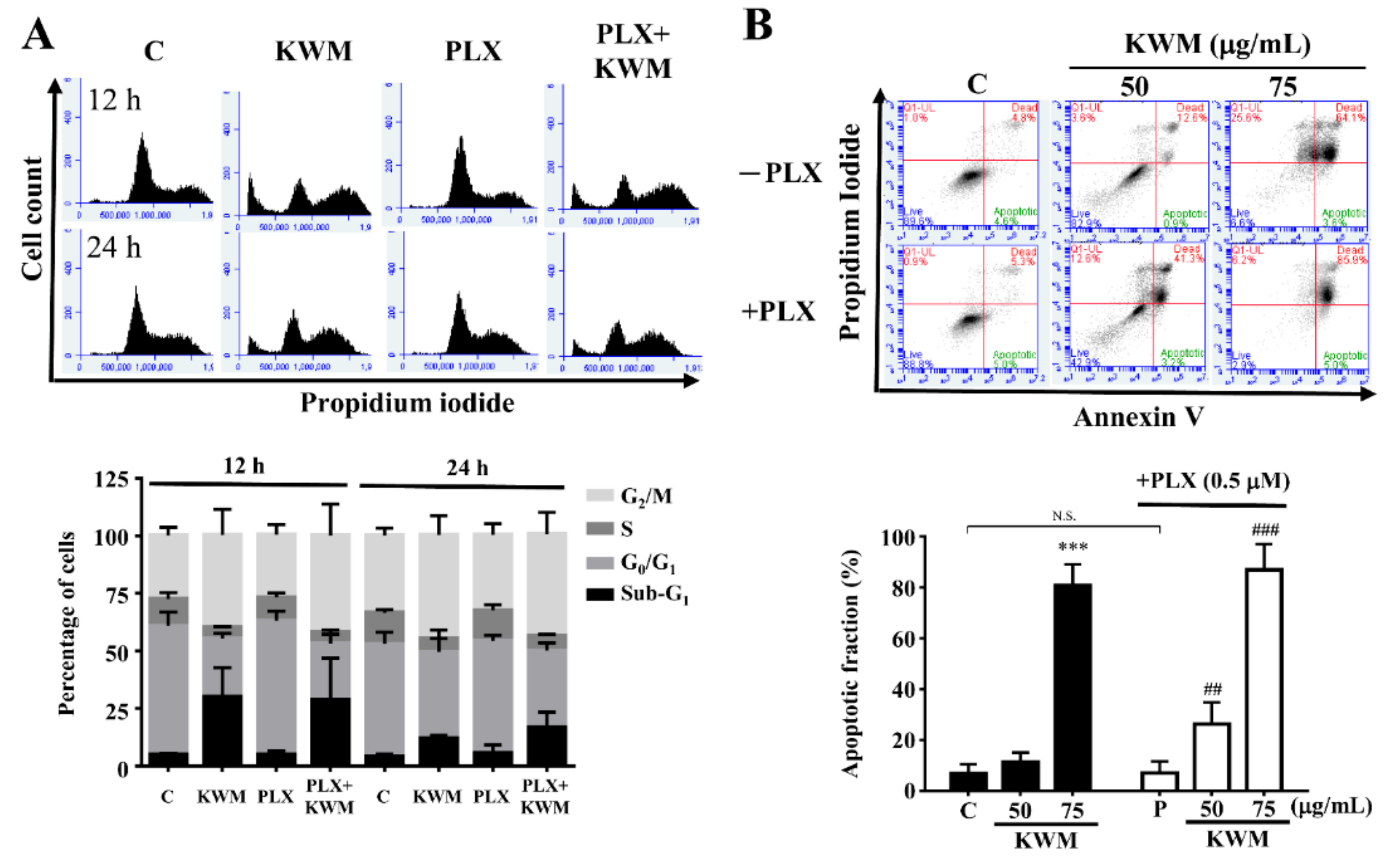
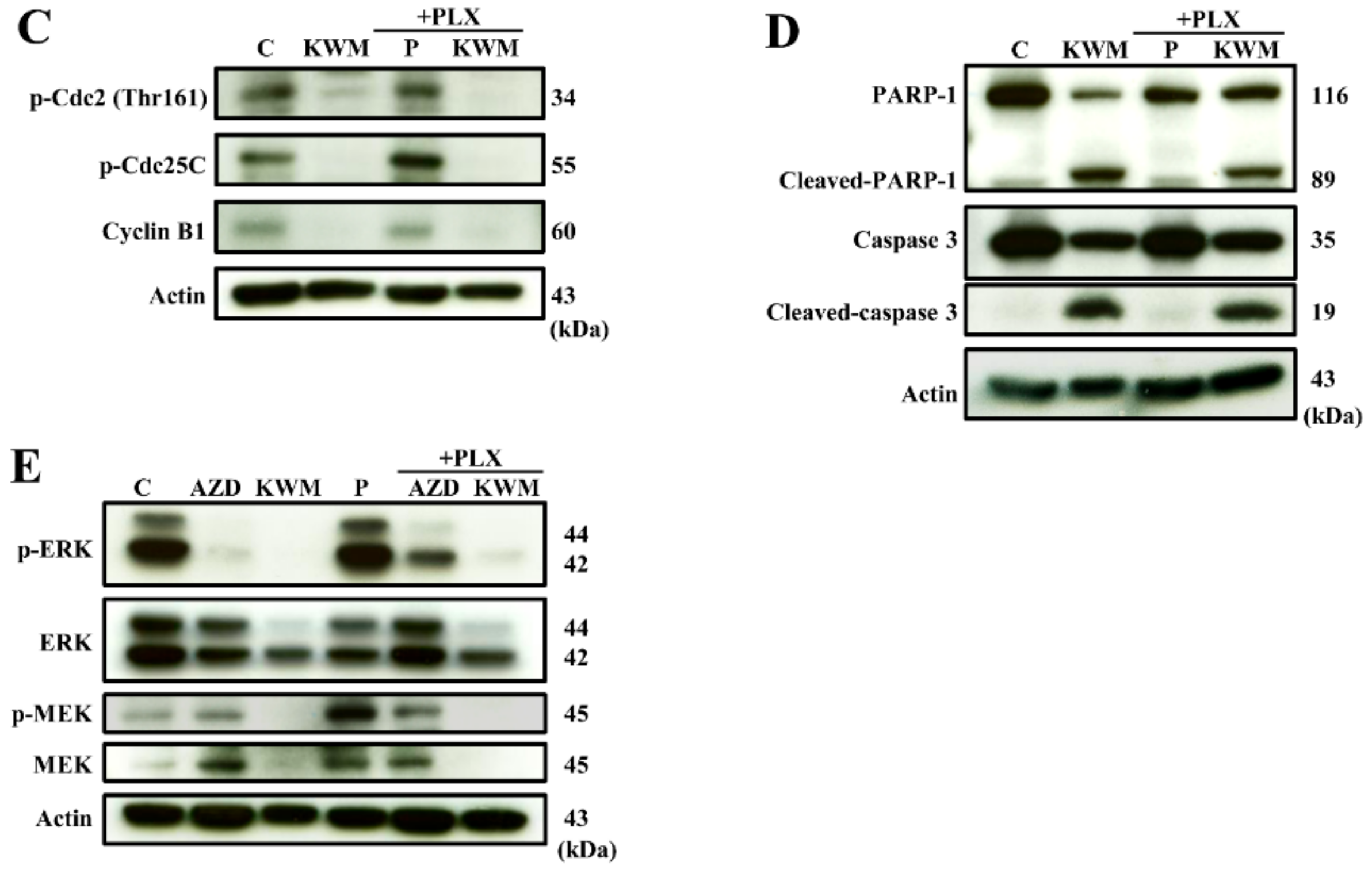
2.3. KWM-EO Induces Cell-Cycle Arrest and Apoptosis in PDV Cells
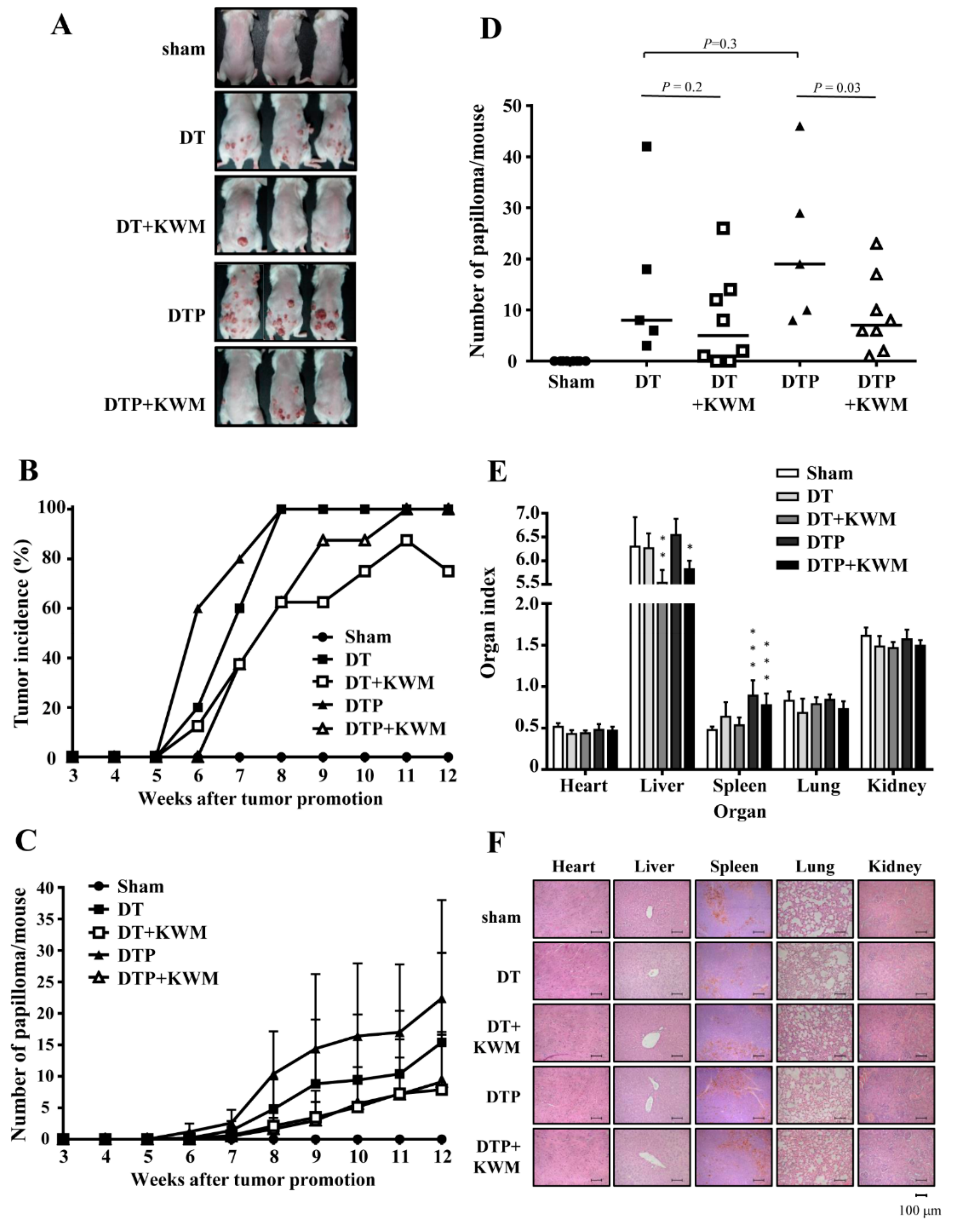
2.4. KWM-EO Inhibits Two-Stage Skin Carcinogenesis in FVB Mice
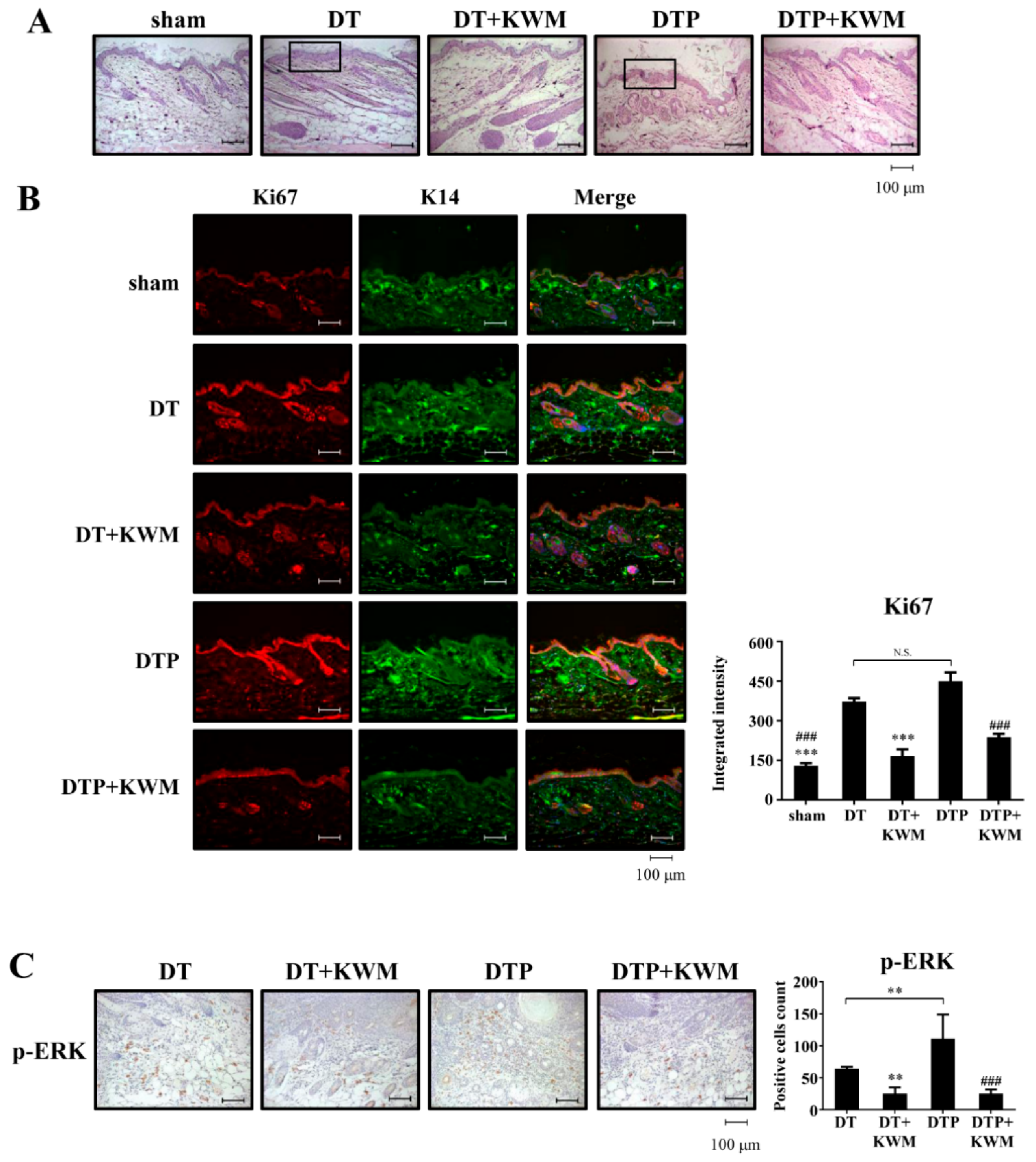
2.5. Skin Histology and Epidermal Cell Proliferation in KWM-EO-Treated Mice
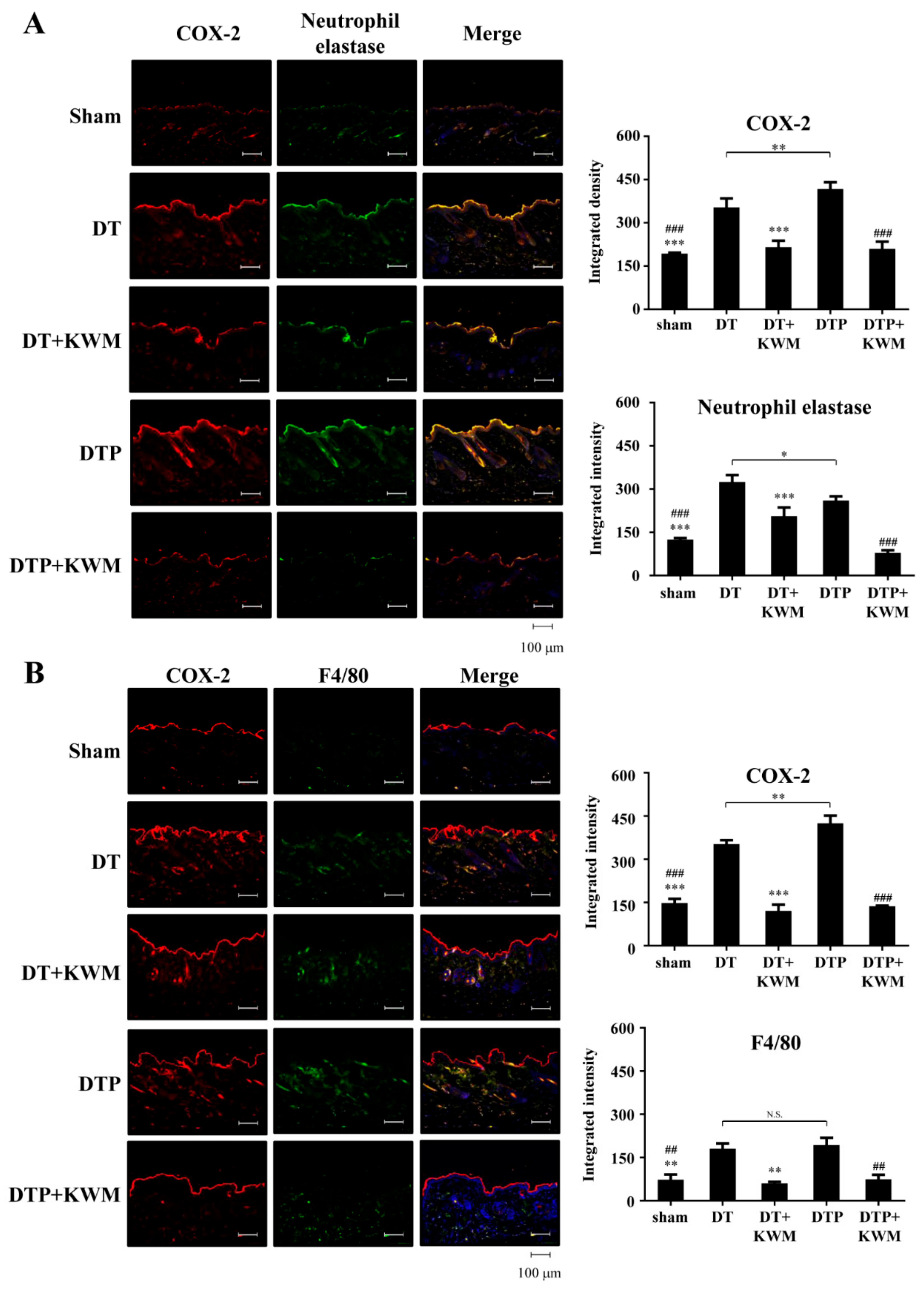
2.6. Anti-Inflammatory Effect of KWM-EO
3. Discussion
4. Materials and Methods
4.1. Mint Cultivation and Distillation of Essential Oils
4.2. Chemical Profiling of KWM-EO Composition by GC×GC−TOF MS
4.3. Cell Lines and Cell Culture
4.4. Measurement of Cell Viability
4.5. Colony Formation Assay
4.6. Cell Invasion Assay
4.7. Wound Healing Assay
4.8. Cell-Cycle Analysis
4.9. Apoptosis Assay
4.10. Western Blot Analysis
4.11. Two-Stage Skin Carcinogenesis Study
4.12. Histopathological and Immunohistochemical Analysis
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sosman, J.A.; Kim, K.B.; Schuchter, L.; Gonzalez, R.; Pavlick, A.C.; Weber, J.S.; McArthur, G.A.; Hutson, T.E.; Moschos, S.J.; Flaherty, K.T.; et al. Survial in BRAF V600-mutant advanced melanoma treated vemurafenib. N. Engl. J. Med. 2012, 366, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Gibney, G.T.; Messina, J.L.; Fedorenko, I.V.; Sondak, V.K.; Smalley, K.S.M. Paradoxical oncogenesis-the long-term effects of BRAF inhibition in melanoma. Nat. Rev. Clin. Oncol. 2013, 10, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Spevak, W.; Zhang, Y.; Burton, E.A.; Ma, Y.; Habets, G.; Zhang, J.; Lin, J.; Ewing, T.; Matusow, B.; et al. RAF inhibitors that evade paradoxical MAPK pathway activation. Nature 2015, 526, 583–586. [Google Scholar] [CrossRef] [PubMed]
- Doma, E.; Rupp, C.; Varga, A.; Kern, F.; Riegler, B.; Baccarini, M. Skin tumorigenesis stimulated by Raf inhibitors relies upon Raf functions that are dependent and independent of ERK. Cancer Res. 2013, 73, 6926–6937. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Viros, A.; Milagre, C.; Trunzer, K.; Bollag, G.; Splesis, O.; Reis-Filho, J.S.; Kong, X.; Koya, R.C.; Flasherty, K.T.; et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N. Engl. J. Med. 2012, 366, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Caulín, C.; Bauluz, C.; Gandarillas, A.; Cano, A.; Quintanilla, M. Changes in keratin expression during malignant progression of transformed mouse epidermal keratinocytes. Exp. Cell Res. 1993, 204, 11–21. [Google Scholar] [CrossRef]
- Abel, E.L.; Angel, J.M; Kiguchi, K.; DiGiovanni, J. Multi-stage chemical carcinogenesis in mouse skin: Fundamentals and applications. Nat. Protoc. 2009, 4, 1350–1362. [Google Scholar] [CrossRef]
- Thomas, G.; Tuk, B.; Song, J.Y.; Truong, H.; Gerritsen, H.C.; Gruijl, F.R.D.; Sterenborg, H.J. Studying skin tumourigenesis and progression in immunocompetent hairless SKH1-h mice using chronic 7,12-dimethylbenz(a)anthracene topical applications to develop a useful experimental skin cancer model. Lab. Anim. 2017, 51, 24–35. [Google Scholar] [CrossRef]
- Gonda, T.A.; Tu, S.; Wang, T.C. Chronic inflammation, the tumor microenvironment and carcinogenesis. Cell Cycle 2014, 8, 2005–2013. [Google Scholar] [CrossRef]
- Korniluk, A.; Koper, O.; Kemona, H.; Dymicka-Piekarska, D. From inflammation to cancer. Ir. J. Med. Sci. 2017, 186, 57–62. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Pollard, J.W. Trophic macrophages in development and disease. Nat. Rev. Immunol. 2009, 9, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Telerman, S.B.; Reimer, A.S.; Sequeira, I.; Liakath-Ali, K.; Arwert, E.N.; Watt, F.M. Macrophage infiltration and alternative activation during wound healing promote MEK1-induced skin carcinogenesis. Cancer Res. 2016, 76, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Alexa, E.; Dauciu, C.; Radulov, I.; Obistioiu, D.; Sumalan, R.M.; Morar, A.; Dehelean, C.A. Phytochemical screening and biological activity of Mentha x piperita L. and Lavandula angustifolia Mill. Extracts. Anal. Cell Pathol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ogaly, H.A.; Eltablawy, N.A.; Abd-Elsalam, R.M. Antifibrogenic influence of Mentha piperita L. essential oil against CCl4-induced liver fibrosis in rats. Oxid. Med. Cell Longev. 2018. [Google Scholar] [CrossRef]
- Tsai, M.L.; Wu, C.T.; Lin, T.F.; Lin, W.C.; Huang, Y.C.; Yang, C.H. Chemical composition and biological properties of essential oils of two mint species. Trop. J. Pharm. Res. 2013, 12, 577–582. [Google Scholar] [CrossRef]
- Bouyahya, A.; Et-Touys, A.; Bakri, Y.; Talbaui, A.; Fellah, H.; Abrini, J.; Dakka, N. Chemical composition of Mentha pulegium and Rosmarinus officinalis essential oils and their antileishmanial, antibacterial and antioxidant activities. Microb. Pathog. 2017, 111, 41–49. [Google Scholar] [CrossRef]
- Zu, Y.; Yu, H.; Liang, L.; Fu, Y.; Efferth, T.; Liu, X.; Wu, N. Activities of ten essential oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 cancer cells. Molecules 2010, 15, 3200–3210. [Google Scholar] [CrossRef]
- Hara, M.; Abe, Y.; Tanaka, T.; Yamamto, T.; Okumura, E.; Kishimoto, T. Greatwall kinase and cyclin B-Cdk1 are both critical constituents of M-phase-promoting factor. Nat. Commun. 2012, 3, 1059–1067. [Google Scholar] [CrossRef]
- Gray, D.C.; Mahrus, S.; Wells, J.A. Activation of specific apoptotic caspases with an engineered small-molecule-activated protease. Cell 2010, 142, 637–646. [Google Scholar] [CrossRef]
- Hall-Jackson, C.A; Eyers, P.A; Cohen, P.; Goedert, M.; Boyle, F.T.; Hewitt, N.; Plant, H.; Hedge, P. Paradoxical activation of Raf by a novel Raf inhibitor. Chem. Biol. 1999, 6, 559–568. [Google Scholar] [CrossRef]
- Li, Y.X.; Liu, Y.B.; Ma, A.Q.; Bao, Y.; Wang, M.; Sun, Z.L. In vitro antiviral, anti-inflammatory, and antioxidant activities of the ethanol extract of Mentha piperita L. Food Sci. Biotechnol. 2017, 26, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Neagu, M.; Caruntu, C.; Constantin, C.; Boda, D.; Zurac, S.; Spandidos, D.A.; Tsatsakis, A.M. Chemically induced skin carcinogenesis: Updates in experimental models (Review). Oncol. Rep. 2016, 35, 2516–2528. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Singh, P.; Tripathi, N.N. Chemistry and bioactivities of essential oils of some Ocimum species: An overview. Asian Pac. J. Trop. Biomed. 2014, 4, 682–694. [Google Scholar] [CrossRef]
- Matsuo, A.L.; Figueiredo, C.R.; Arruda, D.C.; Pereira, F.V.; Scutti, J.A.B.; Massaoka, M.H.; Travassos, L.R.; Sartorelli, P.; Lago, J.H.G. α-Pinene isolated from Schinus terebinthifolius Raddi (Anacardiaceae) induces apoptosis and confers antimetastatic protection in a melanoma model. Biochem. Biophys. Res. Commun. 2011, 411, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Pavithra, P.S.; Mehta, A.; Verma, R.S. Induction of apoptosis by essential oil from P. missionis in skin epidermoid cancer cells. Phytomedicine 2018, 50, 184–195. [Google Scholar] [PubMed]
- Katsanakis, K.D.; Gorgoulis, V.; Papavassiliou, A.G.; Zoumpourlis, V.K. The progression in the mouse skin carcinogenesis model correlates. Mol. Med. 2002, 8, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, K.T.; Infante, J.R.; Daud, A.; Gonzalez, R.; Kefford, R.F.; Sosman, J.; Hamid, O.; Schuchter, L.; Cebon, J.; Ibrahim, N. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N. Engl. J. Med. 2012, 367, 1694–1703. [Google Scholar] [CrossRef]
- Escuin-Ordinas, H.; Atefi, M.; Fu, Y.; Cass, A.; Ng, C.; Huang, R.R.; Yashar, S.; Comin-Anduix, B.; Avramis, E.; Cochran, A.J.; et al. COX-2 inhibition prevents the appearance of cutaneous squamous cell carcinomas accelerated by BRAF inhibitors. Mol. Oncol. 2014, 8, 250–260. [Google Scholar] [CrossRef]
- Schomburg, G.; Dielmann, G. Identification by means of retention parameters. J. Chromatogr. Sci. 1973, 11, 151–159. [Google Scholar] [CrossRef]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of soluble tetrazolium/formazon assay for cell growth sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar] [PubMed]
- Franken, N.A.P.; Rodermond, H.M.; Stap, J.; Haveman, J.; Bree, C.V. Clonogenic assay of cells in vitro. Nat. Protoc. 2006, 1, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Apaya, M.K.; Lin, C.Y.; Chiou, C.Y.; Yang, C.C.; Ting, C.Y.; Shyur, L.F. Simvastatin and a plant galactolipid protect animals from septic shock by regulating oxylipin mediator dynamics through the MAPK-cPLA2 signaling pathway. Mol. Med. 2016, 21, 988–1001. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Chemical Compound | CAS no.a | RT1b | RT2c | KIexpd | KILite | Relative Percentage (%) | |
|---|---|---|---|---|---|---|---|
| 1 | α-Pinene | 7785-70-8 | 6.27 | 0.0245 | 936 | 929 | 10.49 |
| 2 | β-Pinene | 127-91-3 | 7.00 | 0.0250 | 981 | 973 | 15.41 |
| 3 | β-Myrcene | 123-35-3 | 7.20 | 0.0243 | 992 | 993 | 4.86 |
| 4 | β-Cymene | 535-77-3 | 7.73 | 0.0253 | 1027 | 1031 | 3.07 |
| 5 | Eucalyptol | 470-82-6 | 7.93 | 0.0258 | 1040 | 1041 | 12.87 |
| 6 | β-Ocimene | 3338-55-4 | 8.07 | 0.0245 | 1049 | 1036 | 22.18 |
| 7 | Linalool | 78-70-6 | 8.87 | 0.0262 | 1097 | 1101 | 0.25 |
| 8 | Menthone | 89-80-5 | 9.80 | 0.0325 | 1160 | 1154 | 0.04 |
| 9 | Menthofuran | 494-90-6 | 9.93 | 0.0267 | 1169 | 1169 | 0.04 |
| 10 | Levomenthol | 2216-51-5 | 10.07 | 0.0277 | 1178 | 1172 | 0.05 |
| 11 | p-Menth-8-en-2-one | 5948-4-9 | 10.60 | 0.0330 | 1213 | 1218 | 0.07 |
| 12 | Carveol | 1197-07-5 | 10.73 | 0.0272 | 1222 | 1223 | 0.13 |
| 13 | Carvone | 6485-40-1 | 11.13 | 0.0327 | 1251 | 1249 | 1.59 |
| 14 | Linalyl acetate | 115-95-7 | 11.20 | 0.0270 | 1256 | 1257 | 0.08 |
| 15 | Dihydroedulan I | 63335-66-0 | 11.87 | 0.0258 | 1302 | 1292 | 0.28 |
| 16 | β-Bourbonene | 5208-59-3 | 13.13 | 0.0250 | 1396 | 1386 | 0.78 |
| 17 | Caryophyllene | 87-44-5 | 13.60 | 0.0258 | 1434 | 1431 | 2.80 |
| 18 | Humulene | 6753-98-6 | 14.00 | 0.0318 | 1466 | 1465 | 0.29 |
| 19 | Ethyl 4-ethoxybenzoate | 23676-09-7 | 14.73 | 0.0302 | 1524 | 1521 | 2.37 |
| 20 | Viridiflorol | 552-02-3 | 15.73 | 0.0272 | 1608 | 1603 | 3.47 |
| Monoterpene hydrocarbons identified | 56.01 | ||||||
| Oxygenated monoterpene identified | 15.86 | ||||||
| Sesquiterpene hydrocarbon identified | 3.87 | ||||||
| Oxygenated sesquiterpene identified | 3.47 | ||||||
| Other | 2.56 | ||||||
| Identified components | 81.86 | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-T.; Soo, W.-N.; Chen, Y.-H.; Shyur, L.-F. Essential Oil of Mentha aquatica var. Kenting Water Mint Suppresses Two-Stage Skin Carcinogenesis Accelerated by BRAF Inhibitor Vemurafenib. Molecules 2019, 24, 2344. https://doi.org/10.3390/molecules24122344
Chang C-T, Soo W-N, Chen Y-H, Shyur L-F. Essential Oil of Mentha aquatica var. Kenting Water Mint Suppresses Two-Stage Skin Carcinogenesis Accelerated by BRAF Inhibitor Vemurafenib. Molecules. 2019; 24(12):2344. https://doi.org/10.3390/molecules24122344
Chicago/Turabian StyleChang, Chih-Ting, Wen-Ni Soo, Yu-Hsin Chen, and Lie-Fen Shyur. 2019. "Essential Oil of Mentha aquatica var. Kenting Water Mint Suppresses Two-Stage Skin Carcinogenesis Accelerated by BRAF Inhibitor Vemurafenib" Molecules 24, no. 12: 2344. https://doi.org/10.3390/molecules24122344
APA StyleChang, C.-T., Soo, W.-N., Chen, Y.-H., & Shyur, L.-F. (2019). Essential Oil of Mentha aquatica var. Kenting Water Mint Suppresses Two-Stage Skin Carcinogenesis Accelerated by BRAF Inhibitor Vemurafenib. Molecules, 24(12), 2344. https://doi.org/10.3390/molecules24122344





