An Isoxazole Derivative SHU00238 Suppresses Colorectal Cancer Growth through miRNAs Regulation
Abstract
1. Introduction
2. Results
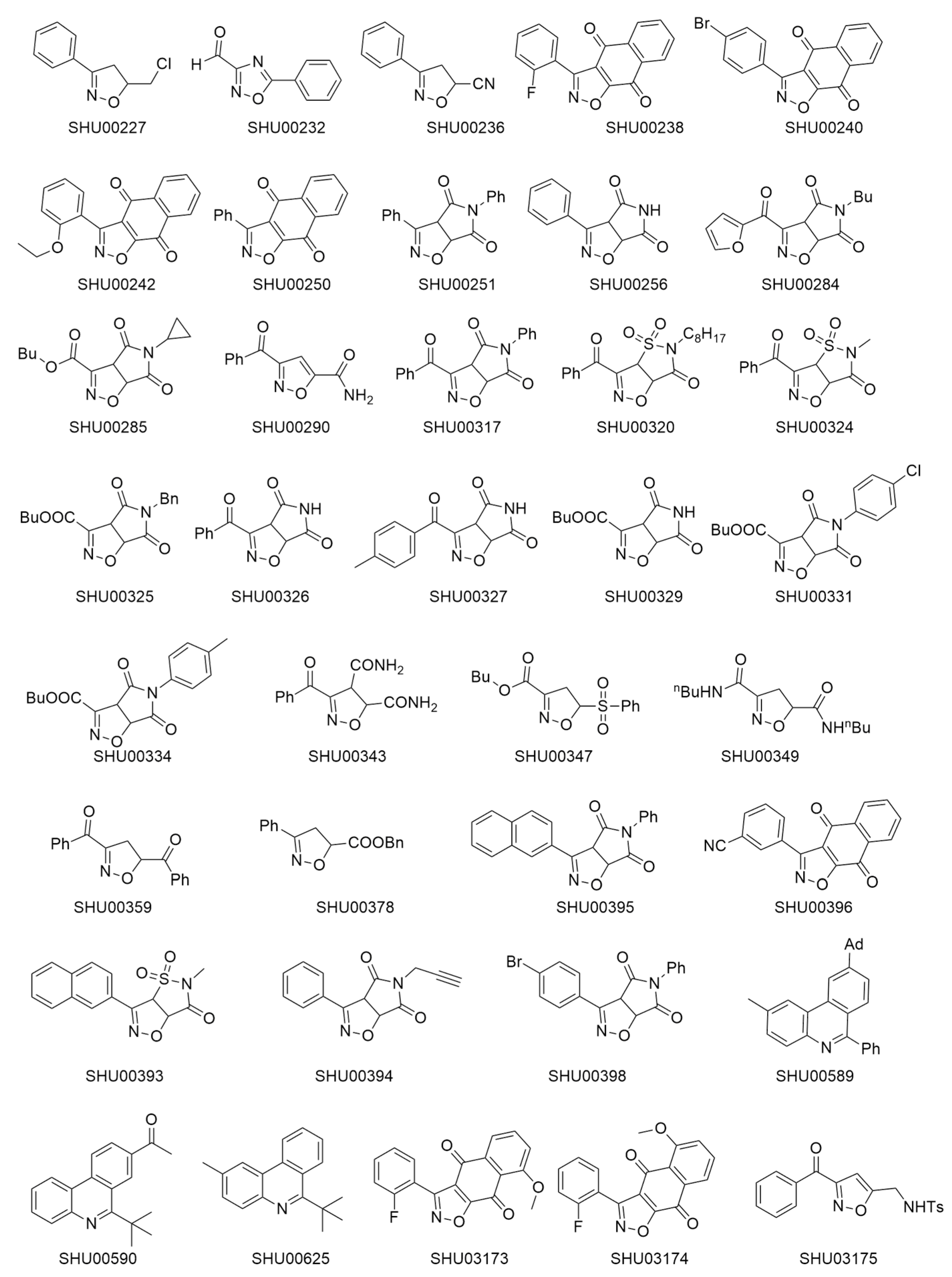
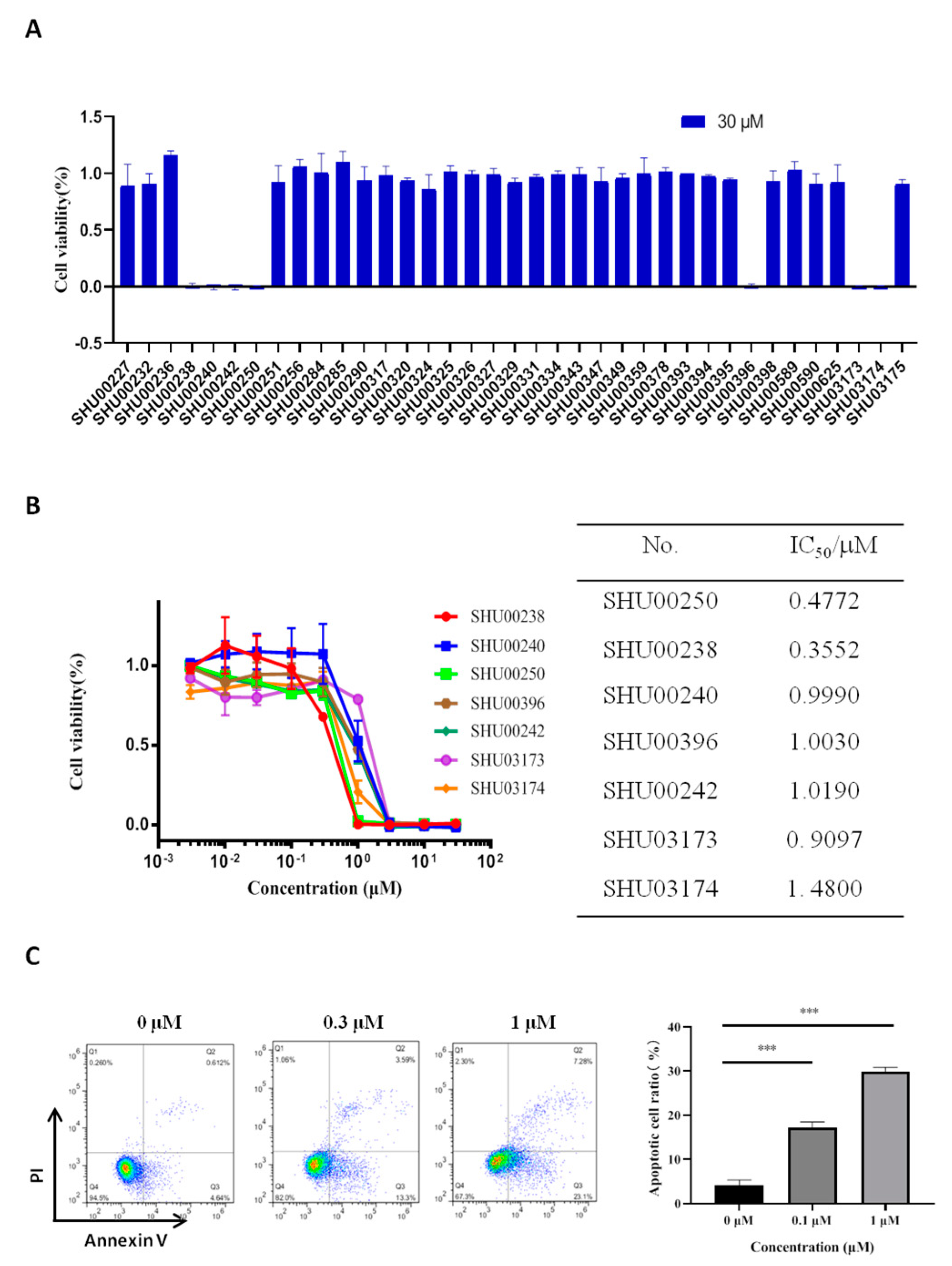
2.1. An Isoxazole Derivative SHU00238 has a Lower IC50 in HCT116 Cells
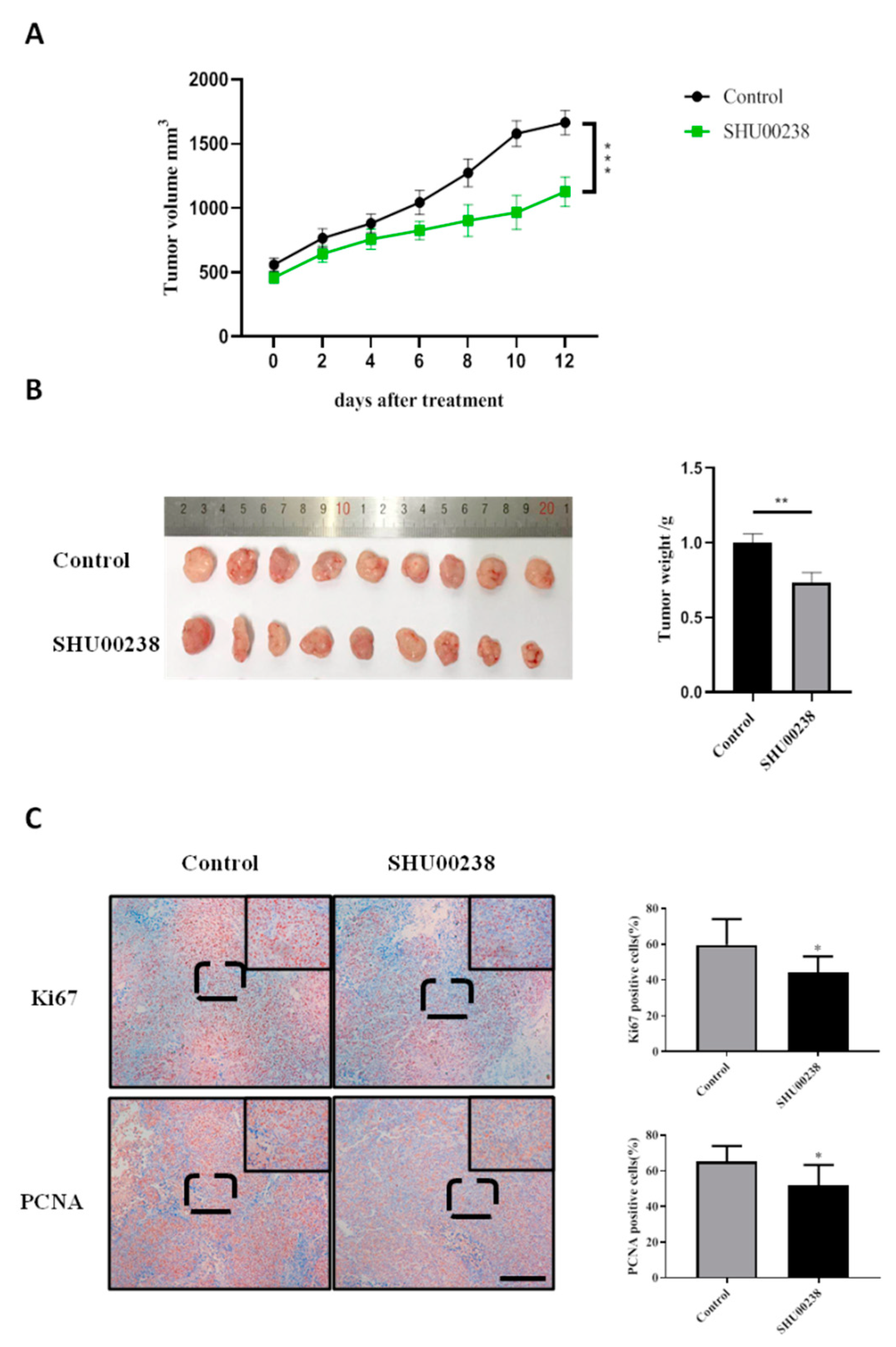
2.2. SHU00238 Suppresses Colonic Tumor Growth and Cell Proliferation in Xenograft Mice Model.
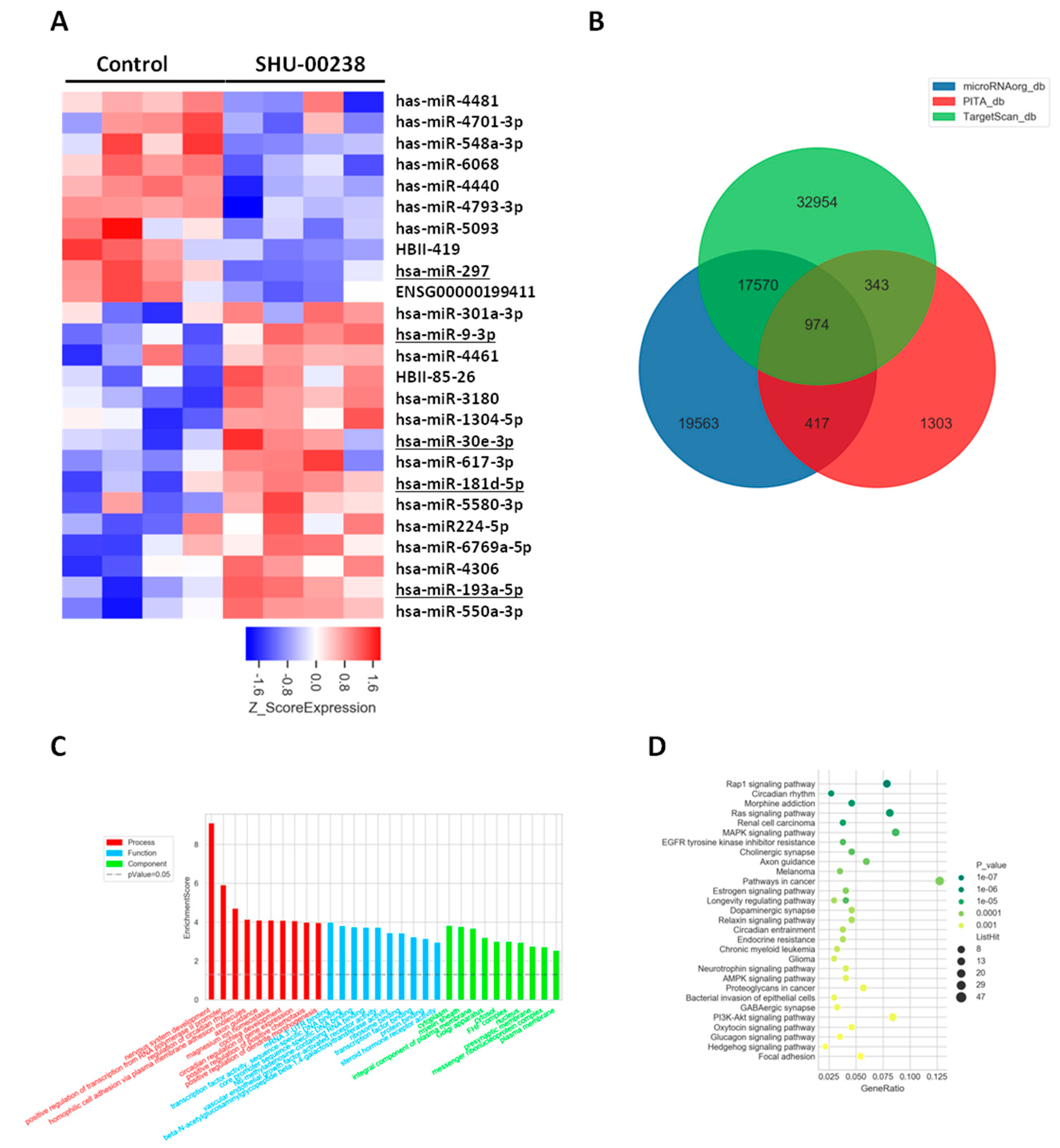
2.3. SHU00238 Treatment Affects Cell Fate by Regulating a Set of miRNAs
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cell Viability Assay
4.3. Cell Apoptosis Assay
4.4. Tissue Immunohistochemistry
4.5. Microarray Hybridization and Data Analysis
4.6. Mice and Treatment
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Cappellani, A.; Zanghi, A.; Di Vita, M.; Cavallaro, A.; Piccolo, G.; Veroux, P.; Lo Menzo, E.; Cavallaro, V.; de Paoli, P.; Veroux, M.; et al. Strong correlation between diet and development of colorectal cancer. Front. Biosci-Landmrk 2013, 18, 190–198. [Google Scholar]
- Abu-Remaileh, M.; Bender, S.; Raddatz, G.; Ansari, I.; Cohen, D.; Gutekunst, J.; Musch, T.; Linhart, H.; Breiling, A.; Pikarsky, E.; et al. Chronic inflammation induces a novel epigenetic program that is conserved in intestinal adenomas and in colorectal cancer. Cancer Res. 2015, 75, 2120–2130. [Google Scholar] [CrossRef]
- Garcia-Anguita, A.; Kakourou, A.; Tsilidis, K.K. Biomarkers of inflammation and immune function and risk of colorectal cancer. Curr. Colorect. Canc. Rep. 2015, 11, 250–258. [Google Scholar] [CrossRef]
- Xu, J.C.; Gao, C.; Lee, J.K.W.; Zhao, J.X. Pm2.5: A barrier to fitness and health promotion in china. J. Sport Health Sci. 2017, 6, 292–294. [Google Scholar] [CrossRef]
- Wu, S.; Luo, Y.F.; Qiu, X.; Bao, M.X. Building a healthy china by enhancing physical activity: Priorities, challenges, and strategies. J. Sport Health Sci. 2017, 6, 125–126. [Google Scholar] [CrossRef][Green Version]
- Dean, M. Cancer as a complex developmental disorder - nineteenth cornelius p. Rhoads memorial award lecture. Cancer Res. 1998, 58, 5633–5636. [Google Scholar]
- Kuipers, E.J.; Grady, W.M.; Lieberman, D.; Seufferlein, T.; Sung, J.J.; Boelens, P.G.; de Velde, C.J.H.V.; Watanabe, T. Colorectal cancer. Nat. Rev. Dis. Primers 2015, 1, 15065. [Google Scholar] [CrossRef]
- Moriarity, A.; O’Sullivan, J.; Kennedy, J.; Mehigan, B.; McCormick, P. Current targeted therapies in the treatment of advanced colorectal cancer: A review. Ther. Adv. Med. Oncol. 2016, 8, 276–293. [Google Scholar] [CrossRef]
- Masuda, T.; Hayashi, N.; Kuroda, Y.; Ito, S.; Eguchi, H.; Mimori, K. Micrornas as biomarkers in colorectal cancer. Cancers 2017, 9, 124. [Google Scholar] [CrossRef]
- Zhai, Z.; Yu, X.; Yang, B.; Zhang, Y.; Zhang, L.; Li, X.; Sun, H. Colorectal cancer heterogeneity and targeted therapy: Clinical implications, challenges and solutions for treatment resistance. Semin. Cell Dev. Biol. 2017, 64, 107–115. [Google Scholar] [CrossRef]
- Barmade, M.A.; Murumkar, P.R.; Sharma, M.K.; Yadav, M.R. Medicinal chemistry perspective of fused isoxazole derivatives. Curr. Top. Med. Chem. 2016, 16, 2863–2883. [Google Scholar] [CrossRef]
- Sysak, A.; Obminska-Mrukowicz, B. Isoxazole ring as a useful scaffold in a search for new therapeutic agents. Eur. J. Med. Chem. 2017, 137, 292–309. [Google Scholar] [CrossRef]
- Jensen, M.R.; Schoepfer, J.; Radimerski, T.; Massey, A.; Guy, C.T.; Brueggen, J.; Quadt, C.; Buckler, A.; Cozens, R.; Drysdale, M.J.; et al. Nvp-auy922: A small molecule hsp90 inhibitor with potent antitumor activity in preclinical breast cancer models. Breast Cancer Res. 2008, 10. [Google Scholar] [CrossRef]
- Montenegro, R.C.; Clark, P.G.K.; Howarth, A.; Wan, X.; Ceroni, A.; Siejka, P.; Nunez-Alonso, G.A.; Monteiro, O.; Rogers, C.; Gamble, V.; et al. Bet inhibition as a new strategy for the treatment of gastric cancer. Oncotarget 2016, 7, 43997–44012. [Google Scholar] [CrossRef]
- Shiro, T.; Fukaya, T.; Tobe, M. The chemistry and biological activity of heterocycle-fused quinolinone derivatives: A review. Eur. J. Med. Chem. 2015, 97, 397–408. [Google Scholar] [CrossRef]
- Lin, Y.; Zhang, K.; Gao, M.; Jiang, Z.; Liu, J.; Ma, Y.; Wang, H.; Tan, Q.; Xiao, J.; Xu, B. Copper nitrate-mediated synthesis of 3-aryl isoxazolines and isoxazoles from olefinic azlactones. Org. Biomol. Chem. 2019, 17, 5509–5513. [Google Scholar] [CrossRef]
- Amirkhah, R.; Schmitz, U.; Linnebacher, M.; Wolkenhauer, O.; Farazmand, A. Microrna-mrna interactions in colorectal cancer and their role in tumor progression. Gene. Chromosome. Canc. 2015, 54, 129–141. [Google Scholar] [CrossRef]
- Krol, J.; Loedige, I.; Filipowicz, W. The widespread regulation of microrna biogenesis, function and decay. Nat. Rev. Genet. 2010, 11, 597–610. [Google Scholar] [CrossRef]
- Wang, L.J.; Lv, Y.C.; Li, G.P.; Xiao, J.J. Micrornas in heart and circulation during physical exercise. J. Sport Health Sci. 2018, 7, 433–441. [Google Scholar] [CrossRef]
- Hayes, J.; Peruzzi, P.P.; Lawler, S. Micrornas in cancer: Biomarkers, functions and therapy. Trends Mol. Med. 2014, 20, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Catalanotto, C.; Cogoni, C.; Zardo, G. Microrna in control of gene expression: An overview of nuclear functions. Int. J. Mol. Sci. 2016, 17, 1712. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.C.; Farh, K.K.H.; Burge, C.B.; Bartel, D.P. Most mammalian mrnas are conserved targets of micrornas. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Chen, J.S.; Wen, J.K.; Gao, H.T.; Zheng, B.; Qu, C.B.; Liu, K.L.; Zhang, M.L.; Gu, J.F.; Li, J.D.; et al. Silencing of mir-193a-5p increases the chemosensitivity of prostate cancer cells to docetaxel. J. Exp. Cli. Can. Res. 2017, 36, 178. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Liang, X.; Shen, K.; Cui, D.L.; Zheng, Y.H.; Xu, J.H.; Fan, Z.Z.; Qiu, Y.Y.; Li, Q.; Ni, L.; et al. Mir-297 modulates multidrug resistance in human colorectal carcinoma by down-regulating mrp-2. Biochem. J. 2012, 446, 291–300. [Google Scholar] [CrossRef]
- Ma, Z.; Qiu, X.; Wang, D.; Li, Y.; Zhang, B.; Yuan, T.; Wei, J.; Zhao, B.; Zhao, X.; Lou, J.; et al. Mir-181a-5p inhibits cell proliferation and migration by targeting kras in non-small cell lung cancer a549 cells. Acta Bioch. Bioph. Sin. 2015, 47, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kuscu, C.; Banach, A.; Zhang, Q.; Pulkoski-Gross, A.; Kim, D.; Liu, J.; Roth, E.; Li, E.; Shroyer, K.R.; et al. Mir-181a-5p inhibits cancer cell migration and angiogenesis via downregulation of matrix metalloproteinase-14. Cancer Res. 2015, 75, 2674–2685. [Google Scholar] [CrossRef]
- Higashi, T.; Hayashi, H.; Ishimoto, T.; Takeyama, H.; Kaida, T.; Arima, K.; Taki, K.; Sakamoto, K.; Kuroki, H.; Okabe, H.; et al. Mir-9-3p plays a tumour-suppressor role by targeting taz (wwtr1) in hepatocellular carcinoma cells. Brit. J. Cancer 2015, 113, 252–258. [Google Scholar] [CrossRef]
- Wang, D.Y.; Zhu, C.; Zhang, Y.F.; Zheng, Y.N.; Ma, F.J.; Su, L.; Shao, G.J. Microrna-30e-3p inhibits cell invasion and migration in clear cell renal cell carcinoma by targeting snail1. Oncol. Lett. 2017, 13, 2053–2058. [Google Scholar] [CrossRef]
- Zhu, J.; Mo, J.; Lin, H.Z.; Chen, Y.; Sun, H.P. The recent progress of isoxazole in medicinal chemistry. Bioorgan. Med. Chem. 2018, 26, 3065–3075. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Schetter, A.J.; Leung, S.Y.; Sohn, J.J.; Zanetti, K.A.; Bowman, E.D.; Yanaihara, N.; Yuen, S.T.; Chan, T.L.; Kwong, D.L.W.; Au, G.K.H.; et al. Microrna expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. Jama-J. Am. Med. Assoc. 2008, 299, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Anvarnia, A.; Mohaddes-Gharamaleki, F.; Asadi, M.; Akbari, M.; Yousefi, B.; Shanehbandi, D. Dysregulated micrornas in colorectal carcinogenesis: New insight to cell survival and apoptosis regulation. J. Cell. Physiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Shekari, N.; Baradaran, B.; Shanehbandi, D.; Kazemi, T. Circulating micrornas: Valuable biomarkers for the diagnosis and prognosis of gastric cancer. Curr. Med. Chem. 2018, 25, 698–714. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compound SHU00238 are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Ma, Y.; Lin, Y.; Liu, J.; Chen, R.; Xu, B.; Liang, Y. An Isoxazole Derivative SHU00238 Suppresses Colorectal Cancer Growth through miRNAs Regulation. Molecules 2019, 24, 2335. https://doi.org/10.3390/molecules24122335
Wang H, Ma Y, Lin Y, Liu J, Chen R, Xu B, Liang Y. An Isoxazole Derivative SHU00238 Suppresses Colorectal Cancer Growth through miRNAs Regulation. Molecules. 2019; 24(12):2335. https://doi.org/10.3390/molecules24122335
Chicago/Turabian StyleWang, Haoyu, Yurui Ma, Yifan Lin, Jiajie Liu, Rui Chen, Bin Xu, and Yajun Liang. 2019. "An Isoxazole Derivative SHU00238 Suppresses Colorectal Cancer Growth through miRNAs Regulation" Molecules 24, no. 12: 2335. https://doi.org/10.3390/molecules24122335
APA StyleWang, H., Ma, Y., Lin, Y., Liu, J., Chen, R., Xu, B., & Liang, Y. (2019). An Isoxazole Derivative SHU00238 Suppresses Colorectal Cancer Growth through miRNAs Regulation. Molecules, 24(12), 2335. https://doi.org/10.3390/molecules24122335




